Hop Latent Viroid: A Hidden Threat to the Cannabis Industry
Abstract
1. Introduction
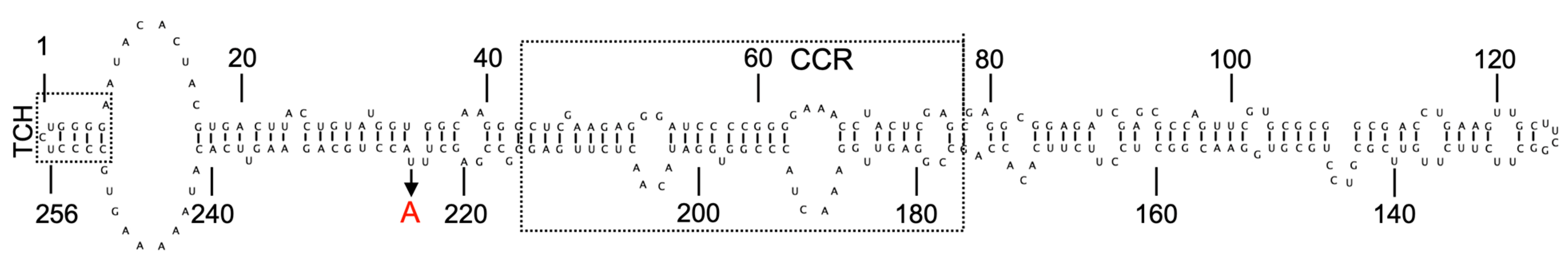
2. Viroids
3. Hop Latent Viroid
4. Host Range and Transmission of HLVd
5. HLVd-Associated Symptoms in Hops
6. Effect of HLVd on the Terpene and Essential Oil Contents
7. HLVd Disease in Cannabis
8. Comparison between HLVd Isolated from Hops and Cannabis
9. Control of HLVd Disease
10. Detection and Management of HLVd Infection
11. Viroid Species Known to Infect Hops
12. Conclusions and Prospective
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Diener, T.O. Potato spindle tuber “virus”. Virology 1971, 45, 411–428. [Google Scholar] [CrossRef]
- Diener, T.O. Discovering viroids–a personal perspective. Nat. Rev. Microbiol. 2003, 1, 75–80. [Google Scholar] [CrossRef]
- Semancik, J.S.; Weathers, L.G. Exocortis virus: An infectious free-nucleic acid plant virus with unusual properties. Virology 1972, 47, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Adkar-Purushothama, C.R.; Perreault, J. Current overview on viroid–host interactions. WIREs RNA 2020, 11, e1570. [Google Scholar] [CrossRef]
- Chiumenti, M.; Navarro, B.; Candresse, T.; Flores, R.; Di Serio, F. Reassessing species demarcation criteria in viroid taxonomy by pairwise identity matrices. Virus Evol. 2021, 7, veab001. [Google Scholar] [CrossRef] [PubMed]
- Pallas, V.; Navarro, A.; Flores, R. Isolation of a viroid-like RNA from hop different from hop stunt viroid. J. Gen. Virol. 1987, 68, 3201–3205. [Google Scholar] [CrossRef]
- Puchta, H.; Ramm, K.; Sänger, H.L. The molecular structure of hop latent viroid (HLV), a new viroid occurring worldwide in hops. Nucleic Acids Res. 1988, 16, 4197–4216. [Google Scholar] [CrossRef]
- Barbara, D.J.; Morton, A.; Adams, A.N.; Green, C.P. Some effects of hop latent viroid on two cultivars of hop (Humulus lupulus) in the UK. Ann. Appl. Biol. 1990, 117, 359–366. [Google Scholar] [CrossRef]
- Faggioli, F.; Duran-Vila, N.; Tsagris, M.; Pallás, V. Geographical distribution of viroids in Europe. In Viroids and Satellites; Elsevier: Amsterdam, The Netherlands, 2017; pp. 473–484. [Google Scholar]
- Pethybridge, S.J.; Hay, F.S.; Barbara, D.J.; Eastwell, K.C.; Wilson, C.R. Viruses and viroids Infecting hop: Significance, epidemiology, and management. Plant Dis. 2008, 92, 324–338. [Google Scholar] [CrossRef]
- Matoušek, J. Hop latent viroid (HLVd) microevolution: An experimental transmission of HLVd “thermomutants” to Solanaceous Species. Biol. Plant. 2003, 46, 607–610. [Google Scholar] [CrossRef]
- Bektaş, A.; Hardwick, K.M.; Waterman, K.; Kristof, J. Occurrence of hop latent viroid in Cannabis sativa with symptoms of cannabis stunting disease in California. Plant Dis. 2019, 103, 2699. [Google Scholar] [CrossRef]
- Warren, J.G.; Mercado, J.; Grace, D. Occurrence of hop latent viroid causing disease in Cannabis sativa in California. Plant Dis. 2019, 103, 2699. [Google Scholar] [CrossRef]
- Fonseca, M.E.N. Hop latent viroid in hop germ plasm introduced into Brazil from the United States. Plant Dis. 1993, 77, 952E. [Google Scholar] [CrossRef]
- Lavagi, I.; Matoušek, J.; Vidalakis, G. Other Cocadviroids. In Viroids and Satellites; Elsevier: Amsterdam, The Netherlands, 2017; pp. 275–287. [Google Scholar]
- Matoušek, J.; Orctová, L.; Škopek, J.; Pešina, K.; Steger, G. Elimination of hop latent viroid upon developmental activation of pollen nucleases. Biol. Chem. 2008, 389, 905–918. [Google Scholar] [CrossRef]
- Adams, A.N.; Morton, A.; Barbarra, D.J.; Ridout, M.S. The distribution and spread of hop latent viroid within two commercial plantings of hop (Humulus lupulus). Ann. Appl. Biol. 1992, 121, 585–592. [Google Scholar] [CrossRef]
- Matoušek, J.; Patzak, J.; Orctová, L.; Schubert, J.; Vrba, L.; Steger, G.; Riesner, D. The variability of hop latent viroid as induced upon heat treatment. Virology 2001, 287, 349–358. [Google Scholar] [CrossRef]
- Patzak, J.; Henychová, A.; Krofta, K.; Svoboda, P.; Malířová, I. The influence of hop latent viroid (HLVd) infection on gene expression and secondary metabolite contents in hop (Humulus lupulus L.) glandular trichomes. Plants 2021, 10, 2297. [Google Scholar] [CrossRef] [PubMed]
- Patzak, J.; Matoušek, J.; Krofta, K.; Svoboda, P. Hop latent viroid (HLVd)-caused pathogenesis: Effects of HLVd infection on lupulin composition of meristem culture-derived Humulus lupulus. Biol. Plant. 2001, 44, 579–585. [Google Scholar] [CrossRef]
- Kovačevič, M.; Kač, M. Determination and verification of hop varieties by analysis of essential oils. Food Chem. 2002, 77, 489–494. [Google Scholar] [CrossRef]
- Flores, R.; Grubb, D.; Elleuch, A.; Nohales, M.-Á.; Delgado, S.; Gago, S. Rolling-circle replication of viroids, viroid-like satellite RNAs and hepatitis delta virus: Variations on a theme. RNA Biol. 2011, 8, 200–206. [Google Scholar] [CrossRef]
- Flores, R.; Hernández, C.; de Alba, A.E.M.; Daròs, J.-A.; Serio, F. Di Viroids and viroid-host interactions. Annu. Rev. Phytopathol. 2005, 43, 117–139. [Google Scholar] [CrossRef] [PubMed]
- Eigen, M. Selforganization of matter and the evolution of biological macromolecules. Naturwissenschaften 1971, 58, 465–523. [Google Scholar] [CrossRef] [PubMed]
- Brass, J.R.J.; Owens, R.A.; Matoušek, J.; Steger, G. Viroid quasispecies revealed by deep sequencing. RNA Biol. 2017, 14, 317–325. [Google Scholar] [CrossRef]
- Adkar-Purushothama, C.R.; Bolduc, F.; Bru, P.; Perreault, J.-P. Insights into potato spindle tuber viroid quasi-species from infection to disease. Front. Microbiol. 2020, 11, 1235. [Google Scholar] [CrossRef] [PubMed]
- Eastwell, K.C.; Nelson, M.E. Occurrence of viroids in commercial hop (Humulus lupulus L.) production areas of Washington State. Plant Health Prog. 2007, 8. [Google Scholar] [CrossRef]
- Chiginsky, J.; Langemeier, K.; MacWilliams, J.; Albrecht, T.; Cranshaw, W.; Fulladolsa, A.C.; Kapuscinski, M.; Stenglein, M.; Nachappa, P. First insights into the virus and viroid communities in hemp (Cannabis sativa). Front. Agron. 2021, 3, 778433. [Google Scholar] [CrossRef]
- Tsushima, T.; Murakami, S.; Ito, H.; He, Y.H.; Adkar-Purushothama, C.R.; Sano, T. Molecular characterization of potato spindle tuber viroid in dahlia. J. Gen. Plant Pathol. 2011, 77, 253–256. [Google Scholar] [CrossRef]
- Kitabayashi, S.; Tsushima, D.; Adkar-Purushothama, C.R.; Sano, T. Identification and molecular mechanisms of key nucleotides causing attenuation in pathogenicity of dahlia isolate of potato spindle tuber viroid. Int. J. Mol. Sci. 2020, 21, 7352. [Google Scholar] [CrossRef]
- Zhang, Z.; Xia, C.; Matsuda, T.; Taneda, A.; Murosaki, F.; Hou, W.; Owens, R.A.; Li, S.; Sano, T. Effects of host-adaptive mutations on hop stunt viroid pathogenicity and small RNA biogenesis. Int. J. Mol. Sci. 2020, 21, 7383. [Google Scholar] [CrossRef]
- Grudzińska, M.; Solarska, E. The Elimination of viruses and hop latent viroid from hop (Humulus lupulus L.) in Poland. Acta Hortic. 2005, 668, 149–152. [Google Scholar] [CrossRef]
- Matoušek, J.; Trněná, L.; Svoboda, P.; Oriniaková, P.; Lichtenstein, C.P. The gradual reduction of viroid levels in hop mericlones following heat therapy: A possible role for a nuclease degrading dsRNA. Biol. Chem. Hoppe. Seyler. 1995, 376, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Morton, A.; Barbara, D.J.; Adams, A.N. The distribution of hop latent viroid within plants of Humulus lupulus and attempts to obtain viroid-free plants. Ann. Appl. Biol. 1993, 123, 47–53. [Google Scholar] [CrossRef]
- Hataya, T.; Hikage, K.; Suda, N.; Nahata, T.; Li, S.; Itoga, Y.; Tanikoshi, T.; Shikata, E. Detection of hop latent viroid (HLVd) using reverse transcription and polymerase chain reaction (RT-PCR). Jpn. J. Phytopathol. 1992, 58, 677–684. [Google Scholar] [CrossRef]
- Adams, A.N.; Barrbara, D.J.; Morton, A.; Darby, P. The experimental transmission of hop latent viroid and its elimination by low temperature treatment and meristem culture. Ann. Appl. Biol. 1996, 128, 37–44. [Google Scholar] [CrossRef]
- Takahashi, T. Diagnosis and control of hop stunt disease. Agric. Hortic. 1979, 54, 1031–1034. [Google Scholar]
- Li, R.; Baysal-Gurel, F.; Abdo, Z.; Miller, S.A.; Ling, K.-S. Evaluation of disinfectants to prevent mechanical transmission of viruses and a viroid in greenhouse tomato production. Virol. J. 2015, 12, 5. [Google Scholar] [CrossRef]
- Shahid, M.A.; Abubakar, M.; Hameed, S.; Hassan, S. Avian influenza virus (H5N1); effects of physico-chemical factors on its survival. Virol. J. 2009, 6, 38. [Google Scholar] [CrossRef]
- Zou, S.; Guo, J.; Gao, R.; Dong, L.; Zhou, J.; Zhang, Y.; Dong, J.; Bo, H.; Qin, K.; Shu, Y. Inactivation of the novel avian influenza A (H7N9) virus under physical conditions or chemical agents treatment. Virol. J. 2013, 10, 289. [Google Scholar] [CrossRef]
- Sano, T.; Yoshida, H.; Goshono, M.; Monma, T.; Kawasaki, H.; Ishizaki, K. Characterization of a new viroid strain from hops: Evidence for viroid speciation by isolation in different host species. J. Gen. Plant Pathol. 2004, 70, 181–187. [Google Scholar] [CrossRef]
- Marquez-Molins, J.; Gomez, G.; Pallas, V. Hop stunt viroid: A polyphagous pathogenic RNA that has shed light on viroid–host interactions. Mol. Plant Pathol. 2021, 22, 153–162. [Google Scholar] [CrossRef]
- Sano, T.; Kashiwagi, A. Host selection-producing variations in the genome of hop stunt viroid. Virus Res. 2022, 311, 198706. [Google Scholar] [CrossRef] [PubMed]
- Kappagantu, M.; Nelson, M.E.; Bullock, J.M.; Kenny, S.T.; Eastwell, K.C. Hop stunt viroid: Effects on vegetative growth and yield of hop cultivars, and its distribution in central Washington state. Plant Dis. 2017, 101, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Michelutti, R.; Al Rwahnih, M.; Torres, H.; Gomez, G.; Luffman, M.; Myrta, A.; Pallás, V. First record of hop stunt viroid in Canada. Plant Dis. 2004, 88, 1162. [Google Scholar] [CrossRef] [PubMed]
- Jakse, J.; Radisek, S.; Pokorn, T.; Matousek, J.; Javornik, B. Deep-sequencing revealed citrus bark cracking viroid (CBCVd) as a highly aggressive pathogen on hop. Plant Pathol. 2015, 64, 831–842. [Google Scholar] [CrossRef]
- Štajner, N.; Radišek, S.; Mishra, A.K.; Nath, V.S.; Matoušek, J.; Jakše, J. Evaluation of disease severity and global transcriptome response induced by citrus bark cracking viroid, hop latent viroid, and their co-infection in hop (Humulus lupulus L.). Int. J. Mol. Sci. 2019, 20, 3154. [Google Scholar] [CrossRef]



| Features | Characteristics |
|---|---|
| Structure of mature viroid RNA | Rod-like |
| Central conserved region (CCR) | Present |
| Terminal conserved hairpin (TCH) | Present |
| Self-cleaving activity (i.e., ribozyme) | absent |
| Replication site in the host | Nucleolus |
| Mode of Replication | Asymmetric rolling circle |
| Enzymes involved in replication | DNA-dependent RNA polymerase II |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adkar-Purushothama, C.R.; Sano, T.; Perreault, J.-P. Hop Latent Viroid: A Hidden Threat to the Cannabis Industry. Viruses 2023, 15, 681. https://doi.org/10.3390/v15030681
Adkar-Purushothama CR, Sano T, Perreault J-P. Hop Latent Viroid: A Hidden Threat to the Cannabis Industry. Viruses. 2023; 15(3):681. https://doi.org/10.3390/v15030681
Chicago/Turabian StyleAdkar-Purushothama, Charith Raj, Teruo Sano, and Jean-Pierre Perreault. 2023. "Hop Latent Viroid: A Hidden Threat to the Cannabis Industry" Viruses 15, no. 3: 681. https://doi.org/10.3390/v15030681
APA StyleAdkar-Purushothama, C. R., Sano, T., & Perreault, J.-P. (2023). Hop Latent Viroid: A Hidden Threat to the Cannabis Industry. Viruses, 15(3), 681. https://doi.org/10.3390/v15030681







