Adjustment of Immunosuppressants to Facilitate Anti-COVID-19 Antibody Production after mRNA Vaccination in Liver Transplant Recipients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Cell Culture and Virus
2.3. Wild-Type COVID-19 Neutralizing Antibody Assay
2.4. Adjustment of Immunosuppressive Regimen
2.5. Clinical Following Up
2.6. Statistical Analysis
3. Results
3.1. Patients
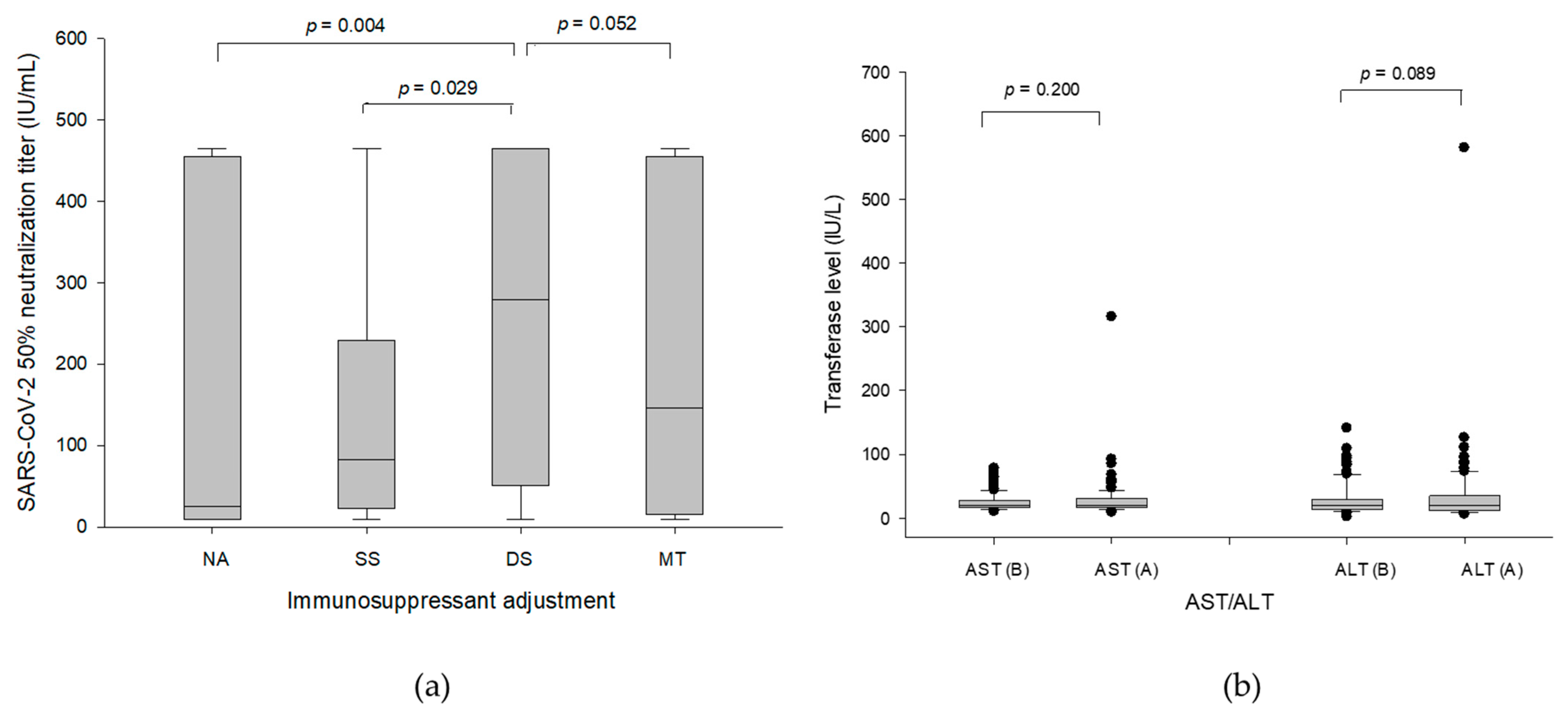
3.2. Immunosuppressive Regimens
3.3. Humoral Responses to Moderna mRNA Vaccine
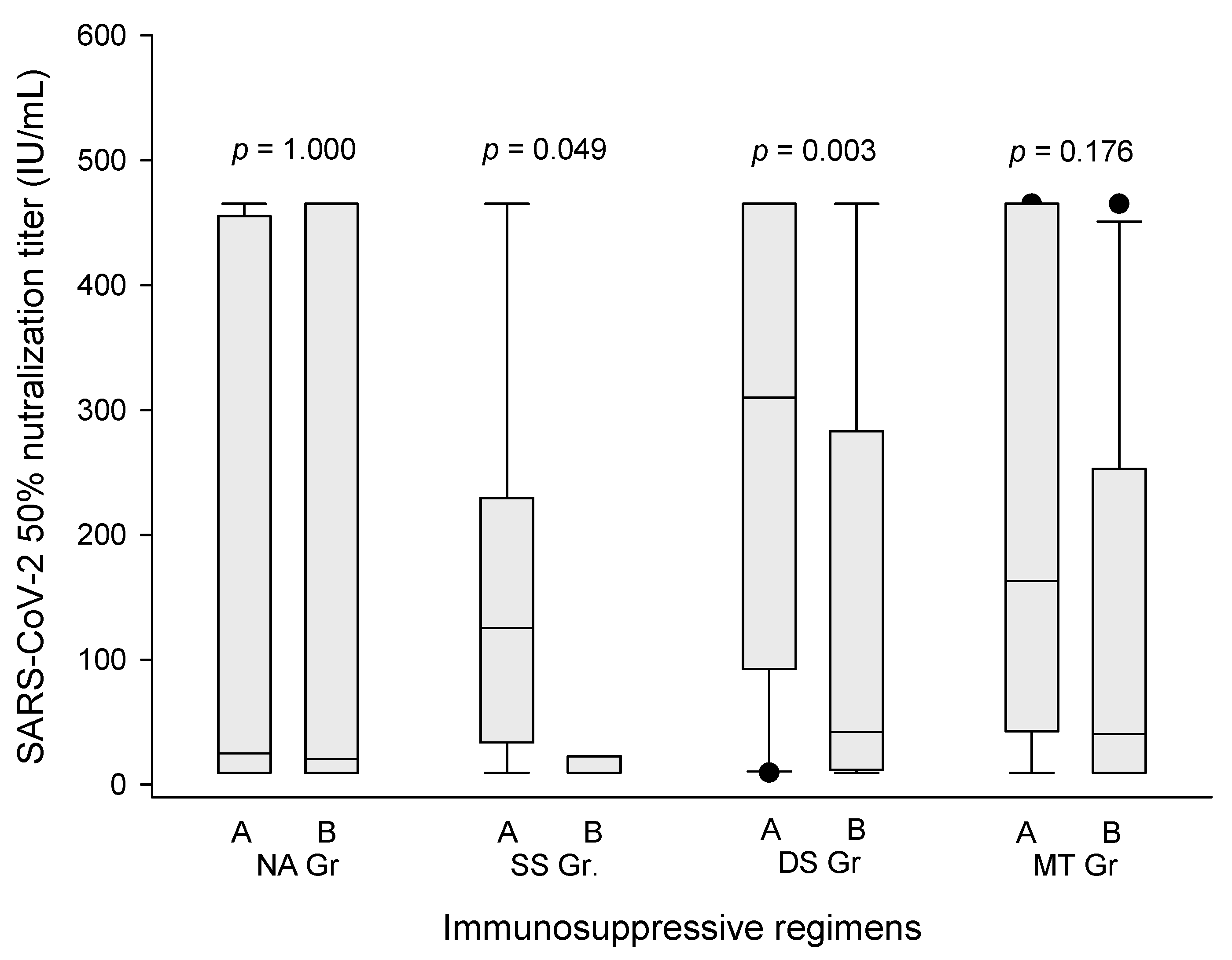
3.4. Antibody Production and Liver Function in Temporary Suspension of MMF/EVR
3.5. Significance of Tacrolimus and Steroid
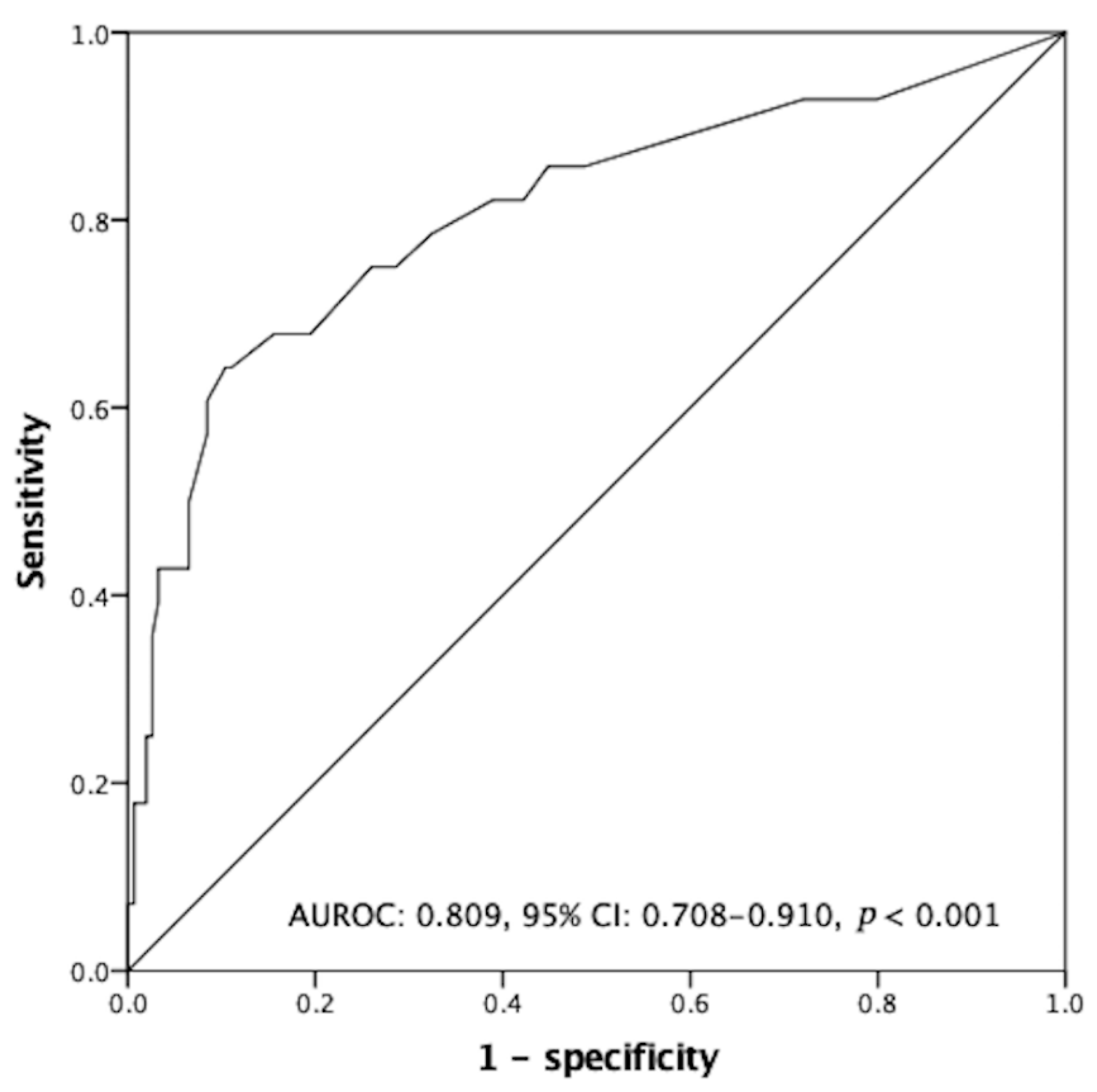
3.6. Independent Predictors Associated with Negative Humoral Response
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tsang, H.F.; Chan, L.W.C.; Cho, W.C.S.; Yu, A.C.S.; Yim, A.K.Y.; Chan, A.K.C.; Ng, L.P.W.; Wong, Y.K.E.; Pei, X.M.; Li, M.J.W.; et al. An update on COVID-19 pandemic: The epidemiology, pathogenesis, prevention and treatment strategies. Expert Rev. Anti-Infect. Ther. 2021, 19, 877–888. [Google Scholar] [CrossRef]
- Zheng, C.; Shao, W.; Chen, X.; Zhang, B.; Wang, G.; Zhang, W. Real-world effectiveness of COVID-19 vaccines: A literature review and meta-analysis. Int. J. Infect. Dis. 2022, 114, 252–260. [Google Scholar] [CrossRef]
- Karim, S.S.A.; Karim, Q.A. Omicron SARS-CoV-2 variant: A new chapter in the COVID-19 pandemic. Lancet 2021, 398, 2126–2128. [Google Scholar] [CrossRef] [PubMed]
- Stainer, A.; Amati, F.; Suigo, G.; Simonetta, E.; Gramegna, A.; Voza, A.; Aliberti, S. COVID-19 in Immunocompromised Patients: A Systematic Review. Semin. Respir. Crit. Care Med. 2021, 42, 839–858. [Google Scholar] [CrossRef] [PubMed]
- Schinas, G.; Polyzou, E.; Mitropetrou, F.; Pazionis, A.; Gogos, C.; Triantos, C.; Akinosoglou, K. COVID-19 Vaccination in Patients with Chronic Liver Disease. Viruses 2022, 14, 2778. [Google Scholar] [CrossRef]
- Yi, S.G.; Knight, R.J.; Graviss, E.A.; Moore, L.W.; Nguyen, D.T.; Ghobrial, R.M.; Gaber, A.O.; Huang, H.J. Kidney Transplant Recipients Rarely Show an Early Antibody Response Following the First COVID-19 Vaccine Administration. Transplantation 2021, 105, e72–e73. [Google Scholar] [CrossRef] [PubMed]
- Shostak, Y.; Shafran, N.; Heching, M.; Rosengarten, D.; Shtraichman, O.; Shitenberg, D.; Amor, S.M.; Yahav, D.; Ben Zvi, H.; Pertzov, B.; et al. Early humoral response among lung transplant recipients vaccinated with BNT162b2 vaccine. Lancet Respir. Med. 2021, 9, e52–e53. [Google Scholar] [CrossRef]
- Rabinowich, L.; Grupper, A.; Baruch, R.; Ben-Yehoyada, M.; Halperin, T.; Turner, D.; Katchman, E.; Levi, S.; Houri, I.; Lubezky, N.; et al. Low immunogenicity to SARS-CoV-2 vaccination among liver transplant recipients. J. Hepatol. 2021, 75, 435–438. [Google Scholar] [CrossRef]
- Huang, C.-G.; Lee, K.-M.; Hsiao, M.-J.; Yang, S.-L.; Huang, P.-N.; Gong, Y.-N.; Hsieh, T.-H.; Huang, P.-W.; Lin, Y.-J.; Liu, Y.-C.; et al. Culture-Based Virus Isolation To Evaluate Potential Infectivity of Clinical Specimens Tested for COVID-19. J. Clin. Microbiol. 2020, 58, e01068-20. [Google Scholar] [CrossRef] [PubMed]
- Kuo, T.Y.; Lin, M.Y.; Coffman, R.L.; Campbell, J.D.; Traquina, P.; Lin, Y.J.; Liu, L.T.; Cheng, J.; Wu, Y.C.; Wu, C.C.; et al. Development of CpG-adjuvanted stable prefusion SARS-CoV-2 spike antigen as a subunit vaccine against COVID-19. Sci. Rep. 2020, 10, 20085. [Google Scholar] [CrossRef]
- Nacif, L.S.; Zanini, L.Y.; Waisberg, D.R.; Pinheiro, R.S.; Galvão, F.; Andraus, W.; D’Albuquerque, L.C. COVID-19 in solid organ transplantation patients: A systematic review. Clinics 2020, 75, e1983. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, A.L.; Pillai, A. The effect of COVID-19 on liver transplantation: Impact, practice patterns, therapeutics, and next steps. Curr. Opin. Organ Transplant. 2021, 26, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Korth, J.; Jahn, M.; Dorsch, O.; Anastasiou, O.E.; Sorge-Hädicke, B.; Eisenberger, U.; Gäckler, A.; Dittmer, U.; Witzke, O.; Wilde, B.; et al. Impaired Humoral Response in Renal Transplant Recipients to SARS-CoV-2 Vaccination with BNT162b2 (Pfizer-BioNTech). Viruses 2021, 13, 756. [Google Scholar] [CrossRef] [PubMed]
- Boyarsky, B.J.; Werbel, W.A.; Avery, R.K.; Tobian, A.A.; Massie, A.B.; Segev, D.L.; Garonzik-Wang, J.M. Antibody Response to 2-Dose SARS-CoV-2mRNA Vaccine Series in Solid Organ Transplant Recipients. JAMA 2021, 325, 2204–2206. [Google Scholar] [CrossRef] [PubMed]
- Thuluvath, P.J.; Robarts, P.; Chauhan, M. Analysis of antibody responses after COVID-19 vaccination in liver transplant recipients and those with chronic liver diseases. J. Hepatol. 2021, 75, 1434–1439. [Google Scholar] [CrossRef]
- Guarino, M.; Esposito, I.; Portella, G.; Cossiga, V.; Loperto, I.; Tortora, R.; Cennamo, M.; Capasso, M.; Terracciano, D.; Lanza, A.G.; et al. Humoral Response to 2-dose BNT162b2 mRNA COVID-19 Vaccination in Liver Transplant Recipients. Clin. Gastroenterol. Hepatol. 2022, 20, 1534–1541.e4. [Google Scholar] [CrossRef]
- Davidov, Y.; Tsaraf, K.; Cohen-Ezra, O.; Likhter, M.; Ben Yakov, G.; Levy, I.; Levin, E.G.; Lustig, Y.; Mor, O.; Rahav, G.; et al. Immunogenicity and Adverse Effects of the 2-Dose BNT162b2 Messenger RNA Vaccine Among Liver Transplantation Recipients. Liver Transplant. 2021, 28, 215–223. [Google Scholar] [CrossRef]
- Fix, O.K.; Blumberg, E.A.; Chang, K.-M.; Chu, J.; Chung, R.T.; Goacher, E.K.; Hameed, B.; Kaul, D.R.; Kulik, L.M.; Kwok, R.M.; et al. American Association for the Study of Liver Diseases Expert Panel Consensus Statement: Vaccines to Prevent Coronavirus Disease 2019 Infection in Patients With Liver Disease. Hepatology 2021, 74, 1049–1064. [Google Scholar] [CrossRef]
- Negahdaripour, M.; Shafiekhani, M.; Moezzi, S.M.I.; Amiri, S.; Rasekh, S.; Bagheri, A.; Mosaddeghi, P.; Vazin, A. Administration of COVID-19 Vaccines in ImmunocompromisedPatients. Int. Immunopharmacol. 2021, 99, 108021. [Google Scholar] [CrossRef]
- Klupp, J.; Pfitzmann, R.; Langrehr, J.M.; Neuhaus, P. Indications of Mycophenolate Mofetil in Liver Transplantation. Transplantation 2005, 80, S142–S146. [Google Scholar] [CrossRef]
- Kawahara, T.; Asthana, S.; Kneteman, N.M. m-TOR inhibitors: What role in liver transplantation? J. Hepatol. 2011, 55, 1441–1451. [Google Scholar] [CrossRef] [PubMed]
- Grigoryan, L.; Pulendran, B. The immunology of SARS-CoV-2 infections and vaccines. Semin. Immunol. 2020, 50, 101422. [Google Scholar] [CrossRef] [PubMed]
- Cholankeril, G.; Al-Hillan, A.; Tarlow, B.; Abrams, D.; Jacobs, J.S.; Flores, N.P.; Rana, A.; Kanwal, F.; Goss, J.A. Clinical Factors Associated With Lack of Serological Response to SARS-CoV-2 Messenger RNA Vaccine in Liver Transplantation Recipients. Liver Transpl. 2022, 28, 123–126. [Google Scholar] [CrossRef]
- Toor, S.M.; Saleh, R.; Sasidharan Nair, V.; Taha, R.Z.; Elkord, E. T-cell responses and therapies against SARS-CoV-2 infection. Immunology 2021, 162, 30–43. [Google Scholar] [CrossRef] [PubMed]
| Parameters | All Recipients (n = 183) | Negative (n = 28) | Positive (n = 155) | p-Value |
|---|---|---|---|---|
| Age | 65.7 ± 7.7 | 63.9 ± 7.9 | 66.2 ± 7.4 | 0.144 |
| Age, ≥65-year-old | 117 (63.9) | 15 (53.6) | 102 (65.8) | 0.215 |
| Gender, male | 140 (76.5) | 19 (67.9) | 121 (78.1) | 0.121 |
| Type of transplantation, LDLT | 141 (77.0) | 17 (60.7) | 124 (80.0) | 0.026 |
| Maintenance of mTOR inhibitor | 21 (11.5) | 2 (7.1) | 19 (12.3) | 0.434 |
| Maintenance of MMF | 121 (66.1) | 18 (64.3) | 103 (66.5) | 0.824 |
| Maintenance of corticosteroid | 5 (2.7) | 3 (10.7) | 2 (1.3) | 0.005 |
| Time from LT to vaccination, months | 108.8 ± 59.9 | 87.4 ± 54.6 | 115.5 ± 60.3 | 0.023 |
| Time from LT to vaccination, ≥120 months | 77 (42.1) | 6 (21.4) | 71 (45.8) | 0.016 |
| Interval between two vaccinations, days | 83.9 ± 21.2 | 76.2 ± 25.3 | 84.8 ± 20.3 | 0.097 |
| Interval between two vaccinations, ≥28 days | 180 (98.4) | 28 (100.0) | 152 (98.1) | 0.458 |
| Immunosuppressant settings | 0.003 | |||
| No adjustment | 23 (12.6) | 9 (32.1) | 14 (9.0) | |
| Single suspension of MMF/EVR | 19 (10.4) | 2 (7.1) | 17 (11.0) | |
| Double suspension of MMF/EVR | 100 (54.6) | 9 (32.1) | 91 (58.7) | |
| Monotherapy (tacrolimus) | 41 (22.4) | 8 (28.6) | 33 (21.9) | |
| Hx of hepatocellular carcinoma | 80 (43.7) | 10 (35.7) | 70 (45.2) | 0.354 |
| Viral hepatitis | 0.371 | |||
| NBNC | 32 (17.5) | 5 (17.9) | 27 (17.4) | |
| HBV | 111 (60.7) | 15 (53.6) | 96 (61.9) | |
| HCV | 34 (18.6) | 8 (28.6) | 26 (16.8) | |
| Co-infection of HBV and HCV | 6 (3.3) | 0 (0.0) | 6 (3.9) | |
| Hx of alcoholism | 22 (12) | 6 (21.4) | 16 (10.3) | 0.096 |
| Hx of autoimmune liver disease | 7 (3.8) | 1 (3.6) | 6 (3.9) | 0.939 |
| Hx of any autoimmune disease | 10 (5.5) | 1 (3.6) | 9 (5.8) | 0.632 |
| Hx of solid tumor malignancy | 87 (47.5) | 12 (42.9) | 75 (48.4) | 0.590 |
| Creatinine, mg/dL | 1.8 ± 1.9 | 2.1 ± 2.4 | 1.8 ± 1.8 | 0.410 |
| Creatinine, ≥1.2 mg/dL | 84 (45.9) | 14 (50.0) | 70 (45.2) | 0.636 |
| eGFR, mL/min/1.73 m2 | 57.4 ± 30.2 | 48.9 ± 24.3 | 59.3 ± 31.1 | 0.097 |
| eGFP, ≥60 mL/min/1.73 m2 | 84 (45.9) | 9 (32.1) | 75(48.4) | 0.112 |
| Hemodialysis | 17 (9.3) | 3 (10.7) | 14 (9.0) | 0.778 |
| Total bilirubin, mg/dL | 0.86 ± 0.56 | 0.85 ± 0.42 | 0.86 ± 0.61 | 0.857 |
| Total bilirubin, ≥1.2 mg/dL | 24 (13.1) | 4 (14.3) | 20 (12.9) | 0.842 |
| AST, U/L | 25.2 ± 16.7 | 28.2 ± 16.7 | 23.8 ± 15.3 | 0.170 |
| AST, ≥34 U/L | 24 (13.1) | 7 (25.0) | 17 (11.0) | 0.043 |
| ALT, U/L | 28.8 ± 25.8 | 35.9 ± 32.0 | 27.1 ± 24.2 | 0.093 |
| ALT, ≥36 U/L | 36 (19.8) | 9 (32.7) | 27 (17.5) | 0.074 |
| Albumin, g/dL | 4.3 ± 0.3 | 4.4 ± 0.3 | 4.3 ± 0.3 | 0.498 |
| Albumin, <3.5 g/dL | 3 (1.6) | 1 (3.6) | 2 (1.3) | 0.382 |
| WBC count, /uL | 5800.1 ± 1682.3 | 5435.7 ± 2298.8 | 5869.0 ± 1520.1 | 0.344 |
| WBC count, <4000/uL | 29 (15.8) | 8 (28.6) | 21 (13.5) | 0.045 |
| Neutrophil, % | 62.5 ± 9.3 | 66.0 ± 9.5 | 62.1 ± 9.2 | 0.041 |
| Neutrophil, ≥74% | 20 (10.9) | 7 (25.0) | 13 (8.4) | 0.010 |
| Lymphocyte, % | 27.2 ± 8.8 | 24.0 ± 9.0 | 27.9 ± 8.5 | 0.029 |
| Lymphocyte, <20% | 38 (20.8) | 11 (39.3) | 27 (17.4) | 0.009 |
| NLR | 2.7 ± 1.7 | 3.2 ± 1.4 | 2.6 ± 1.3 | 0.027 |
| NLR ≥2.25 | 102 (55.7) | 22 (78.6) | 80 (51.6) | 0.008 |
| Platelet count <100 × 109/L | 173.0 ± 55.9 | 166.8 ± 77.0 | 174.9 ± 50.3 | 0.475 |
| Platelet count, <100 × 109/L | 13 (7.1) | 2 (7.1) | 11 (7.1) | 0.993 |
| FK506 trough level, ng/mL | 5.51 ± 4.41 | 5.5 ± 3.4 | 5.1 ± 3.6 | 0.534 |
| FK506 trough level, ≥6.8 ng/mL | 31 (16.9) | 9 (32.1) | 21 (14.2) | 0.020 |
| Parameters (Reference) | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | |
| Type of LT, DDLT (LDLT) | 2.59 | 1.10–6.08 | 0.029 | 2.87 | 1.06–7.81 | 0.038 |
| Maintenance of steroid—yes (no) | 9.18 | 1.46–57.71 | 0.018 | |||
| Triple immunosuppressants—yes (no) | 5.89 | 0.79 –43.64 | 0.083 | |||
| Time from LT to vaccination <120 (≥120) months | 3.10 | 1.19–8.07 | 0.020 | 2.97 | 0.99–8.93 | 0.053 |
| AST ≥ 34 (<34) U/L | 2.71 | 1.01–7.30 | 0.049 | |||
| ALT ≥ 36 (<36) U/L | 2.23 | 0.91–5.45 | 0.079 | |||
| WBC count < 4000 (≥4000)/uL | 2.55 | 0.99–6.54 | 0.051 | 3.96 | 1.28–12.20 | 0.017 |
| Lymphocyte < 20 (≥20) % | 3.07 | 1.29–7.28 | 0.011 | 3.38 | 1.24–9.24 | 0.018 |
| Neutrophil ≥ 74 (<74) % | 3.64 | 1.30–10.17 | 0.014 | |||
| NLR ≥ 2.25 (<2.25) | 3.44 | 1.32–8.94 | 0.011 | |||
| Temporary suspension of immunosuppressant | ||||||
| Single suspension (NA) | 0.18 | 0.34–0.99 | 0.049 | 0.10 | 0.02–0.71 | 0.021 |
| Double suspension (NA) | 0.15 | 0.05–0.45 | 0.001 | 0.13 | 0.04–0.45 | 0.001 |
| Monotherapy (NA) | 0.38 | 0.12–1.18 | 0.093 | 0.22 | 0.06–0.84 | 0.026 |
| FK506 trough level ≥ 6.8 (<6.8) ng/mL | 2.84 | 1.14–7.08 | 0.025 | 3.00 | 1.03–8.72 | 0.044 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, W.-C.; Hung, H.-C.; Lee, J.-C.; Huang, C.-G.; Huang, P.-W.; Gu, P.-W.; Wang, Y.-C.; Cheng, C.-H.; Wu, T.-H.; Lee, C.-F.; et al. Adjustment of Immunosuppressants to Facilitate Anti-COVID-19 Antibody Production after mRNA Vaccination in Liver Transplant Recipients. Viruses 2023, 15, 678. https://doi.org/10.3390/v15030678
Lee W-C, Hung H-C, Lee J-C, Huang C-G, Huang P-W, Gu P-W, Wang Y-C, Cheng C-H, Wu T-H, Lee C-F, et al. Adjustment of Immunosuppressants to Facilitate Anti-COVID-19 Antibody Production after mRNA Vaccination in Liver Transplant Recipients. Viruses. 2023; 15(3):678. https://doi.org/10.3390/v15030678
Chicago/Turabian StyleLee, Wei-Chen, Hao-Chien Hung, Jin-Chiao Lee, Chung-Guei Huang, Po-Wei Huang, Po-Wen Gu, Yu-Chao Wang, Chih-Hsien Cheng, Tsung-Han Wu, Chen-Fang Lee, and et al. 2023. "Adjustment of Immunosuppressants to Facilitate Anti-COVID-19 Antibody Production after mRNA Vaccination in Liver Transplant Recipients" Viruses 15, no. 3: 678. https://doi.org/10.3390/v15030678
APA StyleLee, W.-C., Hung, H.-C., Lee, J.-C., Huang, C.-G., Huang, P.-W., Gu, P.-W., Wang, Y.-C., Cheng, C.-H., Wu, T.-H., Lee, C.-F., Wu, T.-J., Chou, H.-S., & Chan, K.-M. (2023). Adjustment of Immunosuppressants to Facilitate Anti-COVID-19 Antibody Production after mRNA Vaccination in Liver Transplant Recipients. Viruses, 15(3), 678. https://doi.org/10.3390/v15030678





