Nanoluciferase Reporter Zika Viruses as Tools for Assessing Infection Kinetics and Antibody Potency
Abstract
:1. Introduction
2. Methods
2.1. Cell Lines
2.2. Antibodies
2.3. Viruses and Infectious Clones
2.4. Assessment of ZIKV Infectivity and Antibody Mediated Neutralization
2.5. Assessment of Transcytosis of Immune Complexes (IC) across Epithelial Cell Layers
2.6. In Vivo Bioluminescence Imaging (BLI)
Animals
2.7. Statistical Analysis
3. Results
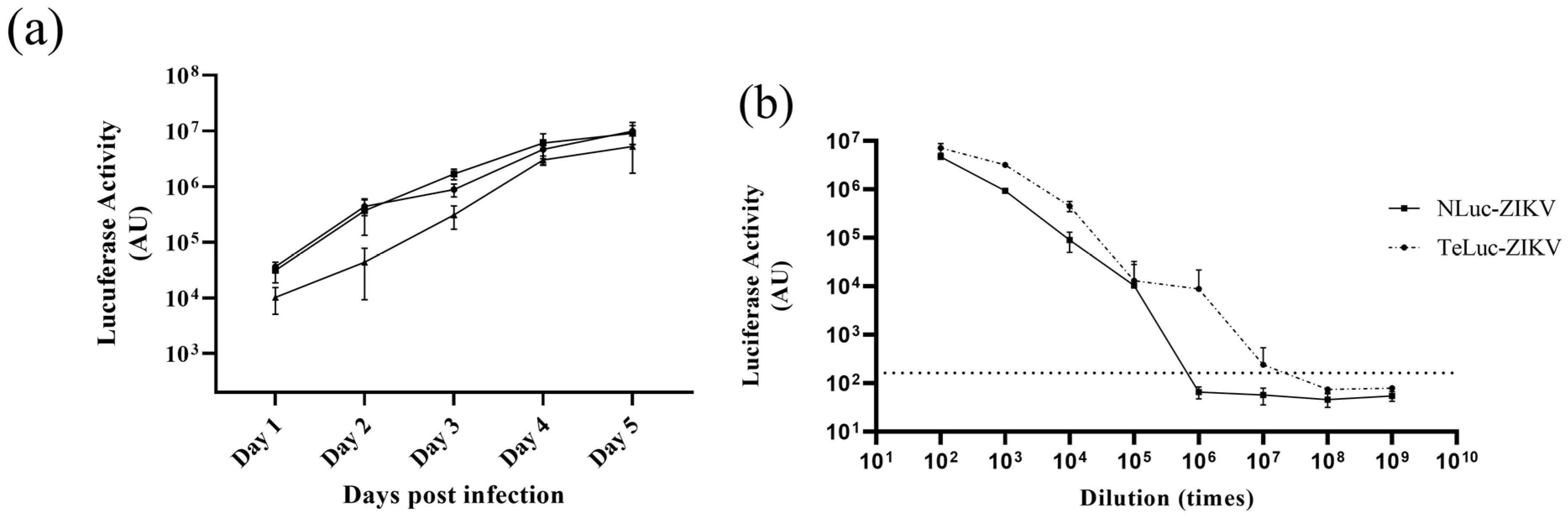
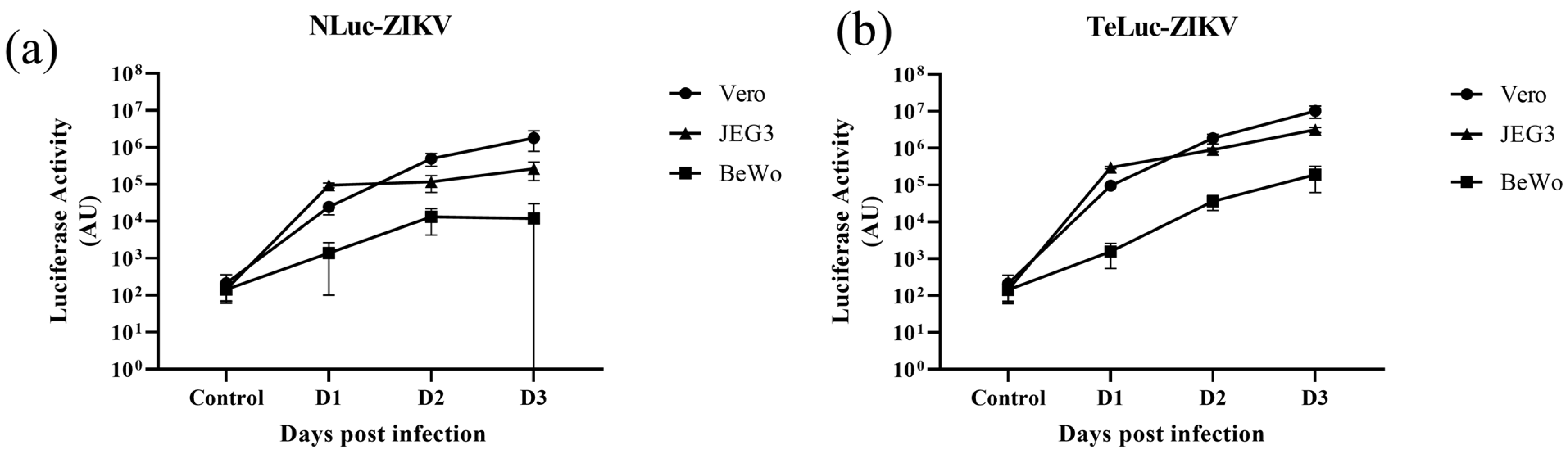
3.1. Assessment of Infectivity of ZIKV Infectious Clones Containing Luciferase Reporters
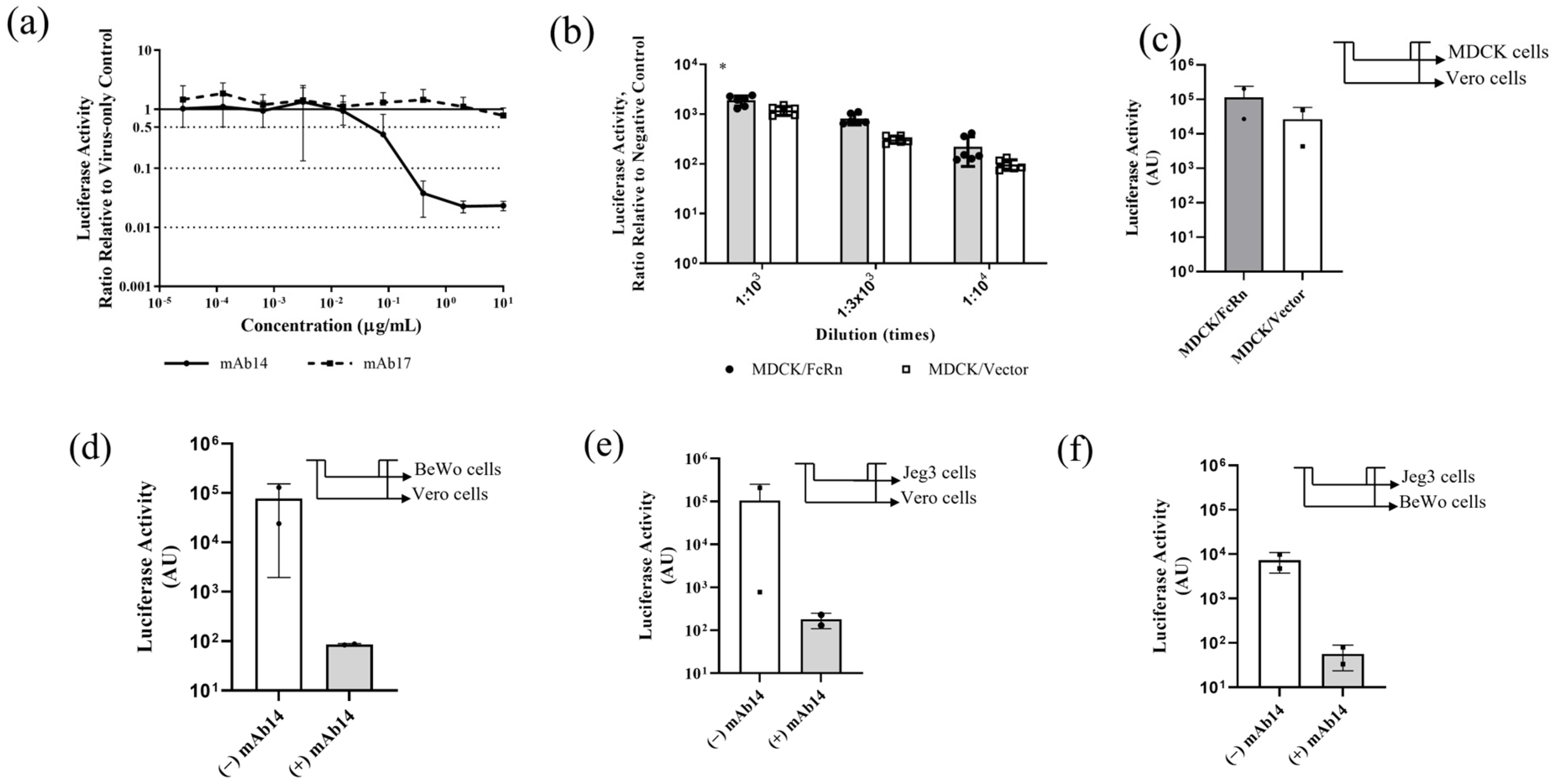
3.2. Antibody Neutralization, Infection of Cells Overexpressing FcRn and Transcytosis of nLuc-ZIKV across Epithelial Cell Layers
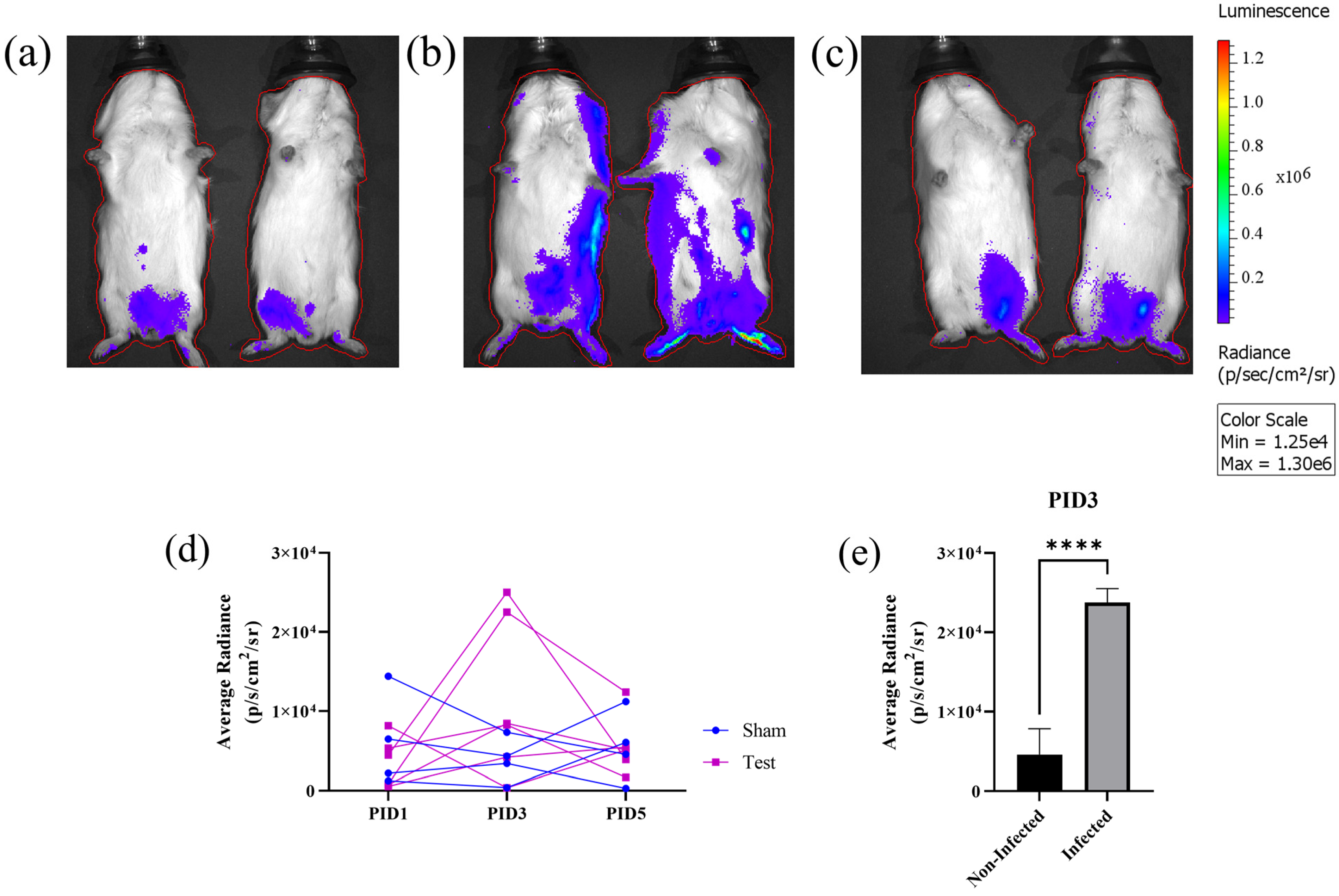
3.3. In Vivo Bioluminescent Imaging (BLI) with nLuc-ZIKV
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Freitas, D.A.; Souza-Santos, R.; Carvalho, L.M.A.; Barros, W.B.; Neves, L.M.; Brasil, P.; Wakimoto, M.D. Congenital Zika syndrome: A systematic review. PLoS ONE 2020, 15, e0242367. [Google Scholar] [CrossRef] [PubMed]
- Rice, M.E.; Galang, R.R.; Roth, N.M.; Ellington, S.R.; Moore, C.A.; Valencia-Prado, M.; Ellis, E.M.; Tufa, A.J.; Taulung, L.A.; Alfred, J.M.; et al. Vital Signs: Zika-Associated Birth Defects and Neurodevelopmental Abnormalities Possibly Associated with Congenital Zika Virus Infection—U.S. Territories and Freely Associated States, 2018. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 858–867. [Google Scholar] [CrossRef] [PubMed]
- Ades, A.E.; Soriano-Arandes, A.; Alarcon, A.; Bonfante, F.; Thorne, C.; Peckham, C.S.; Giaquinto, C. Vertical transmission of Zika virus and its outcomes: A Bayesian synthesis of prospective studies. Lancet Infect. Dis. 2021, 21, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Soares de Souza, A.; Moraes Dias, C.; Braga, F.D.; Terzian, A.C.B.; Estofolete, C.F.; Oliani, A.H.; Oliveira, G.H.; de Mattos, C.C.B.; de Mattos, L.C.; Nogueira, M.L.; et al. Fetal Infection by Zika Virus in the Third Trimester: Report of 2 Cases. Clin. Infect. Dis. 2016, 63, 1622–1625. [Google Scholar] [CrossRef]
- Pan American Health Organization. Epidemiological Update for Dengue, Chikungunya and Zika in 2022. Updated 29 March 2023. Available online: https://ais.paho.org/ha_viz/Arbo/Arbo_Bulletin_2022.asp?env=pri (accessed on 29 March 2023).
- Tebas, P.; Roberts, C.C.; Muthumani, K.; Reuschel, E.L.; Kudchodkar, S.B.; Zaidi, F.I.; White, S.; Khan, A.S.; Racine, T.; Choi, H.; et al. Safety and Immunogenicity of an Anti-Zika Virus DNA Vaccine. N. Engl. J. Med. 2021, 385, e35. [Google Scholar] [CrossRef]
- White, J.; Tunga, P.; Anderson, D.M.; Iledan, K.; Loreth, T.; Parrera, G.S.; Astacio, H.; Drobic, B.; Richardson, J.S. Results of a Double-Blind, Randomized, Placebo-Controlled Phase 1 Study to Evaluate the Safety and Pharmacokinetics of Anti-Zika Virus Immunoglobulin. Am. J. Trop. Med. Hyg. 2021, 105, 1552–1562. [Google Scholar] [CrossRef]
- Struble, E.B.; Rawson, J.M.O.; Stantchev, T.; Scott, D.; Shapiro, M.A. Uses and Challenges of Antiviral Polyclonal and Monoclonal Antibody Therapies. Pharmaceutics 2023, 15, 1538. [Google Scholar] [CrossRef] [PubMed]
- Volkova, E.; Tsetsarkin, K.A.; Sippert, E.; Assis, F.; Liu, G.; Rios, M.; Pletnev, A.G. Novel Approach for Insertion of Heterologous Sequences into Full-Length ZIKV Genome Results in Superior Level of Gene Expression and Insert Stability. Viruses 2020, 12, 61. [Google Scholar] [CrossRef]
- England, C.G.; Ehlerding, E.B.; Cai, W. NanoLuc: A Small Luciferase Is Brightening up the Field of Bioluminescence. Bioconjug. Chem. 2016, 27, 1175–1187. [Google Scholar] [CrossRef]
- Yeh, H.W.; Karmach, O.; Ji, A.; Carter, D.; Martins-Green, M.M.; Ai, H.W. Red-shifted luciferase-luciferin pairs for enhanced bioluminescence imaging. Nat. Methods 2017, 14, 971–974. [Google Scholar] [CrossRef]
- Xu, Y.; He, Y.; Momben-Abolfath, S.; Vertrees, D.; Li, X.; Norton, M.G.; Struble, E.B. Zika Virus Infection and Antibody Neutralization in FcRn Expressing Placenta and Engineered Cell Lines. Vaccines 2022, 10, 2059. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; He, Y.; Momben-Abolfath, S.; Eller, N.; Norton, M.; Zhang, P.; Scott, D.; Struble, E.B. Entry and Disposition of Zika Virus Immune Complexes in a Tissue Culture Model of the Maternal-Fetal Interface. Vaccines 2021, 9, 145. [Google Scholar] [CrossRef] [PubMed]
- Hierholzer, J.C.; Killington, R.A. Virus isolation and quantitation. In Virology Methods Manual; Elsevier: Amsterdam, The Netherlands, 1996; pp. 25–46. [Google Scholar] [CrossRef]
- Ramos da Silva, S.; Cheng, F.; Huang, I.C.; Jung, J.U.; Gao, S.J. Efficiencies and kinetics of infection in different cell types/lines by African and Asian strains of Zika virus. J. Med. Virol. 2019, 91, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Krause, K.K.; Azouz, F.; Nakano, E.; Nerurkar, V.R. A guinea pig model of Zika virus infection. Virol. J. 2017, 14, 75. [Google Scholar] [CrossRef]
- Lucke, A.C.; Vom Hemdt, A.; Wieseler, J.; Fischer, C.; Feldmann, M.; Rothenfusser, S.; Drexler, J.F.; Kümmerer, B.M. High-Throughput Platform for Detection of Neutralizing Antibodies Using Flavivirus Reporter Replicon Particles. Viruses 2022, 14, 346. [Google Scholar] [CrossRef]
- Matsuda, M.; Yamanaka, A.; Yato, K.; Yoshii, K.; Watashi, K.; Aizaki, H.; Konishi, E.; Takasaki, T.; Kato, T.; Muramatsu, M.; et al. High-throughput neutralization assay for multiple flaviviruses based on single-round infectious particles using dengue virus type 1 reporter replicon. Sci. Rep. 2018, 8, 16624. [Google Scholar] [CrossRef]
- Whitbeck, J.C.; Thomas, A.; Kadash-Edmondson, K.; Grinyo-Escuer, A.; Stafford, L.J.; Cheng, C.; Liao, G.C.; Holtsberg, F.W.; Aman, M.J.; Simmons, G.; et al. Antigenicity, stability, and reproducibility of Zika reporter virus particles for long-term applications. PLoS Negl. Trop. Dis. 2020, 14, e0008730. [Google Scholar] [CrossRef]
- Pyzik, M.; Sand, K.M.K.; Hubbard, J.J.; Andersen, J.T.; Sandlie, I.; Blumberg, R.S. The Neonatal Fc Receptor (FcRn): A Misnomer? Front. Immunol. 2019, 10, 1540. [Google Scholar] [CrossRef]
- Liu, X.; Palaniyandi, S.; Zhu, I.; Tang, J.; Li, W.; Wu, X.; Ochsner, S.P.; Pauza, C.D.; Cohen, J.I.; Zhu, X. Human cytomegalovirus evades antibody-mediated immunity through endoplasmic reticulum-associated degradation of the FcRn receptor. Nat. Commun. 2019, 10, 3020. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, G.; Liu, S.; Chen, X.; Peng, R.; Dai, L.; Qu, X.; Li, S.; Song, H.; Gao, Z.; et al. Human Neonatal Fc Receptor Is the Cellular Uncoating Receptor for Enterovirus B. Cell 2019, 177, 1553–1565.e16. [Google Scholar] [CrossRef]
- Anwar, A.; Helou, M.; Hervol, J.; Levine, A.D. Impact of Caveolin-Mediated Endocytosis on the Trafficking of HIV within the Colonic Barrier. J. Virol. 2022, 96, e0020222. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.K.; Hassert, M.; Han, X.; Barker, D.; Carnelley, T.; Branche, E.; Steffen, T.L.; Stone, E.T.; Geerling, E.; Viramontes, K.M.; et al. The Ability of Zika virus Intravenous Immunoglobulin to Protect from or Enhance Zika Virus Disease. Front. Immunol. 2021, 12, 717425. [Google Scholar] [CrossRef] [PubMed]
- Rathore, A.P.S.; Saron, W.A.A.; Lim, T.; Jahan, N.; St. John, A.L. Maternal immunity and antibodies to dengue virus promote infection and Zika virus-induced microcephaly in fetuses. Sci. Adv. 2019, 5, eaav3208. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, M.G.; Quicke, K.M.; O’Neal, J.T.; Arora, N.; Machiah, D.; Priyamvada, L.; Kauffman, R.C.; Register, E.; Adekunle, O.; Swieboda, D.; et al. Cross-Reactive Dengue Virus Antibodies Augment Zika Virus Infection of Human Placental Macrophages. Cell Host Microbe 2018, 24, 731–742.e6. [Google Scholar] [CrossRef] [PubMed]
- Westrich, J.A.; McNulty, E.E.; Edmonds, M.J.; Nalls, A.V.; Miller, M.R.; Foy, B.D.; Rovnak, J.; Perera, R.; Mathiason, C.K. Characterization of subclinical ZIKV infection in immune-competent guinea pigs and mice. J. Gen. Virol. 2021, 102, 001641. [Google Scholar] [CrossRef]
- Saver, A.E.; Crawford, S.A.; Joyce, J.D.; Bertke, A.S. Route of Infection Influences Zika Virus Shedding in a Guinea Pig Model. Cells 2019, 8, 1437. [Google Scholar] [CrossRef]
- Miller, M.R.; Fagre, A.C.; Clarkson, T.C.; Markle, E.D.; Foy, B.D. Three Immunocompetent Small Animal Models That Do Not Support Zika Virus Infection. Pathogens 2021, 10, 971. [Google Scholar] [CrossRef]
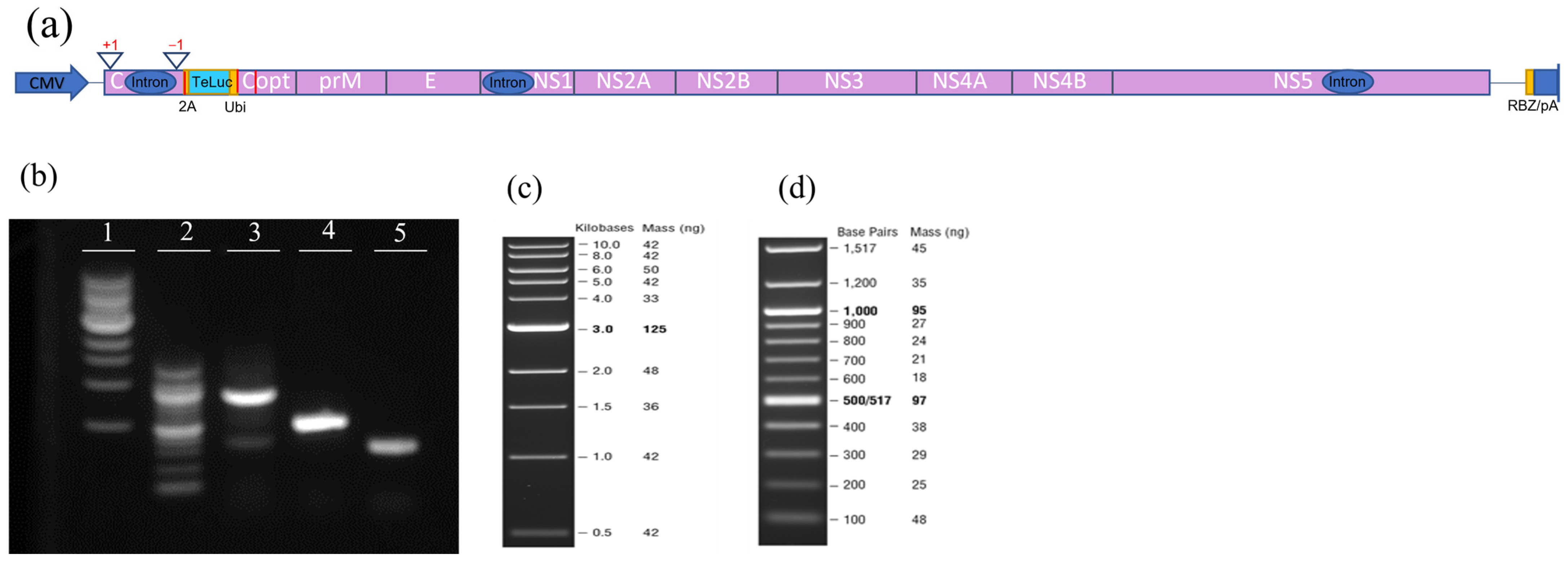
| Name | Sequence | Amplicon Size, Base Pairs |
|---|---|---|
| ZIKV-2A-teLuc-Dir | ATGCAATCCCGGGCCCatggtcttcaca | 541 |
| teLuc-Ubi-Rev | GAAGATCTGCATcgccagaatgcg | |
| teLuc-Ubi-Dir | cgcattctggcgATGCAGATCTTC | 331 |
| ZIKV-Mlu-Rev | TGAGACCACCgAATGGTGACA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.; Vertrees, D.; He, Y.; Momben-Abolfath, S.; Li, X.; Brewah, Y.A.; Scott, D.E.; Konduru, K.; Rios, M.; Struble, E.B. Nanoluciferase Reporter Zika Viruses as Tools for Assessing Infection Kinetics and Antibody Potency. Viruses 2023, 15, 2190. https://doi.org/10.3390/v15112190
Xu Y, Vertrees D, He Y, Momben-Abolfath S, Li X, Brewah YA, Scott DE, Konduru K, Rios M, Struble EB. Nanoluciferase Reporter Zika Viruses as Tools for Assessing Infection Kinetics and Antibody Potency. Viruses. 2023; 15(11):2190. https://doi.org/10.3390/v15112190
Chicago/Turabian StyleXu, Yanqun, Devin Vertrees, Yong He, Sanaz Momben-Abolfath, Xiaohong Li, Yambasu A. Brewah, Dorothy E. Scott, Krishnamurthy Konduru, Maria Rios, and Evi B. Struble. 2023. "Nanoluciferase Reporter Zika Viruses as Tools for Assessing Infection Kinetics and Antibody Potency" Viruses 15, no. 11: 2190. https://doi.org/10.3390/v15112190
APA StyleXu, Y., Vertrees, D., He, Y., Momben-Abolfath, S., Li, X., Brewah, Y. A., Scott, D. E., Konduru, K., Rios, M., & Struble, E. B. (2023). Nanoluciferase Reporter Zika Viruses as Tools for Assessing Infection Kinetics and Antibody Potency. Viruses, 15(11), 2190. https://doi.org/10.3390/v15112190