Diversity and Current Classification of dsRNA Bacteriophages
Abstract
:1. Introduction
2. Discovery of dsRNA Phages
| Method 1 | Phi6 | PhiNN | Phi2954 | Phi8 | Phi12 | Phi13 | PhiYY | PhiNY | PhiZ98 | CAP3 2 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Genome type | Gel electrophoresis | [34] | [16] | [15] | [11] | [15] | [14] | [17] | [1] | [8] | [3] |
| RNase sensitivity | [34] | [17] | [1] | [8] | [3] | ||||||
| Virion morphology | TEM imaging | [9,35] | [16] | [17] | [1] | [8] | [3] | ||||
| Cryo-EM/Cryo-ET imaging | [27,28] | [36] | [28] | [36,37] | |||||||
| Virion composition | PAGE of structural proteins | [38] | [16] | [15] | [11] | [37] | [14] | [17] | [8] | [3] | |
| Chloroform sensitivity | [9] | [16] | [15] | [10] | [10] | [10] | [17] | [1] | [8] | [3] | |
| Detergent sensitivity | [35] | [16] | [15] | [11] | [12,13] | [3] |
3. Diversity of dsRNA Phages
3.1. Host Range of dsRNA Phages
3.2. Host Cell Receptors for dsRNA Phages
3.3. Infection Strategies of dsRNA Phages
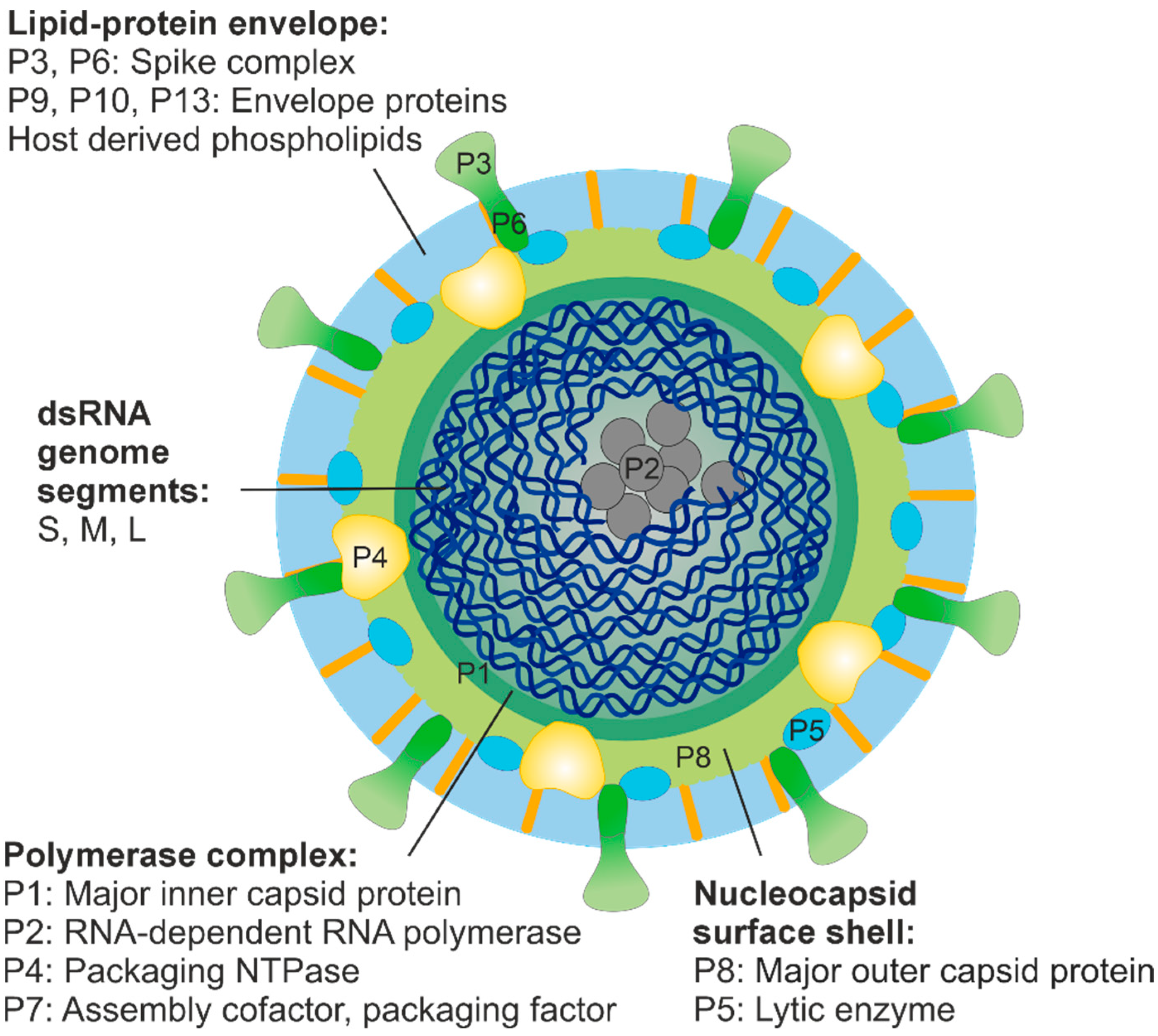
3.4. Organisation of the dsRNA Phage Virions and Structural Relationships with Eukaryotic RNA Viruses
3.5. Genomes of dsRNA Phages
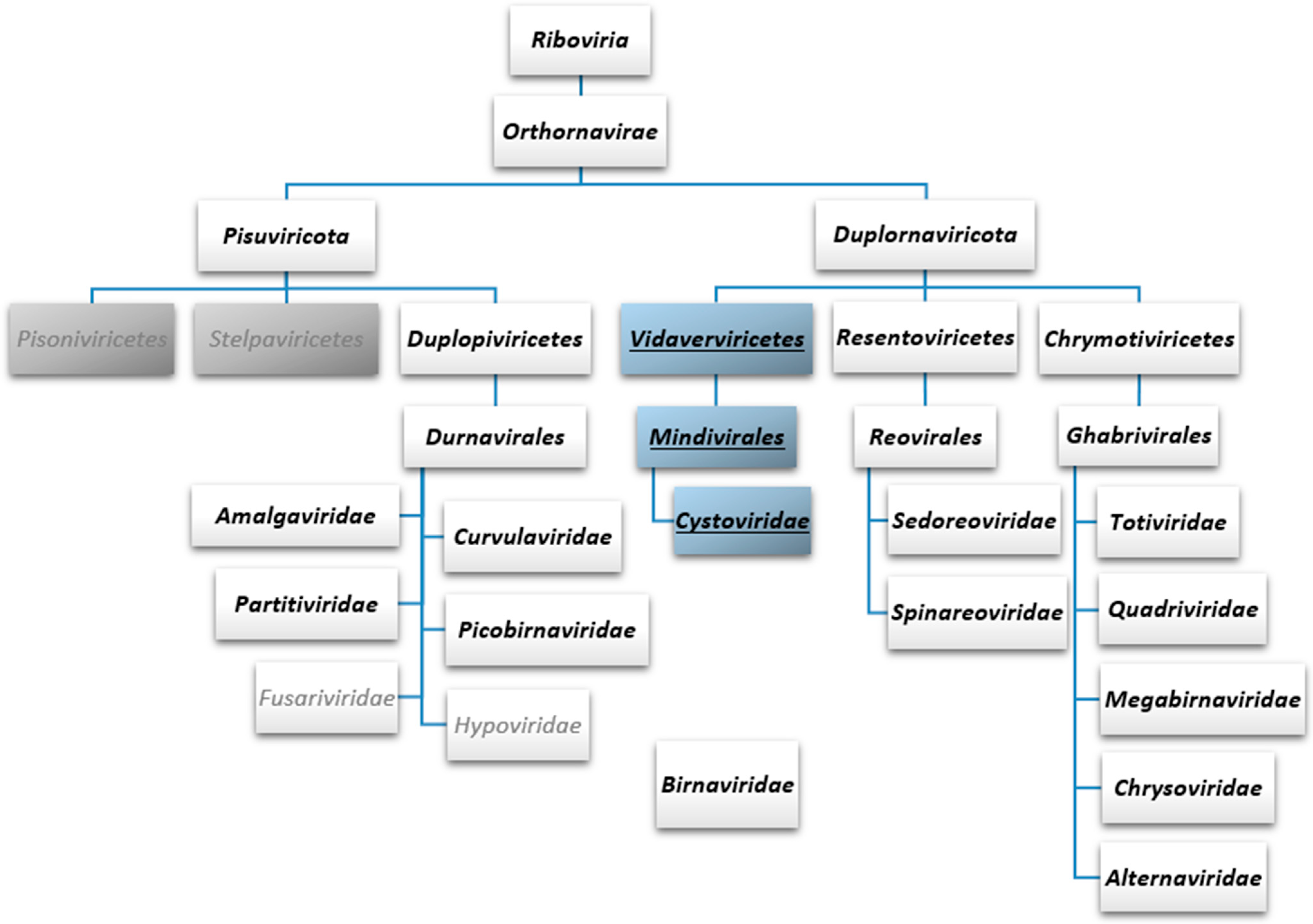
4. Current Taxonomic Classification of dsRNA Phages
5. Concluding Remarks and Future Research Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cai, X.; Tian, F.; Teng, L.; Liu, H.; Tong, Y.; Le, S.; Zhang, T. Cultivation of a lytic double-stranded RNA bacteriophage infecting Microvirgula aerodenitrificans reveals a mutualistic parasitic lifestyle. J. Virol. 2021, 95, 0039921. [Google Scholar] [CrossRef]
- Chen, Y.M.; Sadiq, S.; Tian, J.H.; Chen, X.; Lin, X.D.; Shen, J.J.; Chen, H.; Hao, Z.Y.; Wille, M.; Zhou, Z.C.; et al. RNA viromes from terrestrial sites across China expand environmental viral diversity. Nat. Microbiol. 2022, 7, 1312–1323. [Google Scholar] [CrossRef]
- Crippen, C.S.; Zhou, B.; Andresen, S.; Patry, R.T.; Muszynski, A.; Parker, C.T.; Cooper, K.K.; Szymanski, C.M. RNA and sugars, unique properties of bacteriophages infecting multidrug resistant Acinetobacter radioresistens strain LH6. Viruses 2021, 13, 1652. [Google Scholar] [CrossRef]
- Krishnamurthy, S.R.; Janowski, A.B.; Zhao, G.; Barouch, D.; Wang, D. Hyperexpansion of RNA bacteriophage diversity. PLoS Biol. 2016, 14, e1002409. [Google Scholar] [CrossRef] [PubMed]
- Neri, U.; Wolf, Y.I.; Roux, S.; Camargo, A.P.; Lee, B.; Kazlauskas, D.; Chen, I.M.; Ivanova, N.; Zeigler Allen, L.; Paez-Espino, D.; et al. Expansion of the global RNA virome reveals diverse clades of bacteriophages. Cell 2022, 185, 4023–4037.e18. [Google Scholar] [CrossRef] [PubMed]
- Starr, E.P.; Nuccio, E.E.; Pett-Ridge, J.; Banfield, J.F.; Firestone, M.K. Metatranscriptomic reconstruction reveals RNA viruses with the potential to shape carbon cycling in soil. Proc. Natl. Acad. Sci. USA 2019, 116, 25900–25908. [Google Scholar] [CrossRef]
- Zayed, A.A.; Wainaina, J.M.; Dominguez-Huerta, G.; Pelletier, E.; Guo, J.; Mohssen, M.; Tian, F.; Pratama, A.A.; Bolduc, B.; Zablocki, O.; et al. Cryptic and abundant marine viruses at the evolutionary origins of Earth’s RNA virome. Science 2022, 376, 156–162. [Google Scholar] [CrossRef]
- Li, D.; Li, Y.; Li, P.; Han, Q.; Zhang, T.; Yang, B.; Wu, W.; Yang, H. Phage phiZ98: A novel tri-segmented dsRNA cystovirus for controlling Pseudomonas strains with defective lipopolysaccharides in foods. Food Res. Int. 2022, 162, 112197. [Google Scholar] [CrossRef] [PubMed]
- Vidaver, A.K.; Koski, R.K.; Van Etten, J.L. Bacteriophage phi6: A Lipid-containing virus of Pseudomonas phaseolicola. J. Virol. 1973, 11, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Mindich, L.; Qiao, X.; Qiao, J.; Onodera, S.; Romantschuk, M.; Hoogstraten, D. Isolation of additional bacteriophages with genomes of segmented double-stranded RNA. J. Bacteriol. 1999, 181, 4505–4508. [Google Scholar] [CrossRef]
- Hoogstraten, D.; Qiao, X.; Sun, Y.; Hu, A.; Onodera, S.; Mindich, L. Characterization of phi8, a bacteriophage containing three double-stranded RNA genomic segments and distantly related to phi6. Virology 2000, 272, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, P.; Potgieter, C.; Wei, H.; Toporovsky, I. Characterization of phi12, a bacteriophage related to phi6: Nucleotide sequence of the large double-stranded RNA. Virology 2002, 295, 266–271. [Google Scholar] [CrossRef]
- Gottlieb, P.; Wei, H.; Potgieter, C.; Toporovsky, I. Characterization of phi12, a bacteriophage related to phi6: Nucleotide sequence of the small and middle double-stranded RNA. Virology 2002, 293, 118–124. [Google Scholar] [CrossRef]
- Qiao, X.; Qiao, J.; Onodera, S.; Mindich, L. Characterization of phi13, a bacteriophage related to phi6 and containing three dsRNA genomic segments. Virology 2000, 275, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Qiao, X.; Sun, Y.; Qiao, J.; Di Sanzo, F.; Mindich, L. Characterization of phi2954, a newly isolated bacteriophage containing three dsRNA genomic segments. BMC Microbiol. 2010, 10, 55. [Google Scholar] [CrossRef]
- Mäntynen, S.; Laanto, E.; Kohvakka, A.; Poranen, M.M.; Bamford, J.K.H.; Ravantti, J.J. New enveloped dsRNA phage from freshwater habitat. J. Gen. Virol. 2015, 96, 1180–1189. [Google Scholar] [CrossRef]
- Yang, Y.; Lu, S.; Shen, W.; Zhao, X.; Shen, M.; Tan, Y.; Li, G.; Li, M.; Wang, J.; Hu, F.; et al. Characterization of the first double-stranded RNA bacteriophage infecting Pseudomonas aeruginosa. Sci. Rep. 2016, 6, 38795. [Google Scholar] [CrossRef]
- Poranen, M.M.; Bamford, D.H. Assembly of large icosahedral double-stranded RNA viruses. Adv. Exp. Med. Biol. 2012, 726, 379–402. [Google Scholar]
- Niehl, A.; Soininen, M.; Poranen, M.M.; Heinlein, M. Synthetic biology approach for plant protection using dsRNA. Plant Biotechnol. J. 2018, 16, 1679–1687. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Osterlund, P.; Westenius, V.; Guo, D.; Poranen, M.M.; Bamford, D.H.; Julkunen, I. Efficient inhibition of avian and seasonal influenza A viruses by a virus-specific dicer-substrate small interfering RNA swarm in human monocyte-derived macrophages and dendritic cells. J. Virol. 2019, 93, e01916-18. [Google Scholar] [CrossRef] [PubMed]
- Kalke, K.; Lund, L.M.; Nyman, M.C.; Levanova, A.A.; Urtti, A.; Poranen, M.M.; Hukkanen, V.; Paavilainen, H. Swarms of chemically modified antiviral siRNA targeting herpes simplex virus infection in human corneal epithelial cells. PLoS Pathog. 2022, 18, e1010688. [Google Scholar] [CrossRef] [PubMed]
- Levanova, A.A.; Kalke, K.M.; Lund, L.M.; Sipari, N.; Sadeghi, M.; Nyman, M.C.; Paavilainen, H.; Hukkanen, V.; Poranen, M.M. Enzymatically synthesized 2’-fluoro-modified Dicer-substrate siRNA swarms against herpes simplex virus demonstrate enhanced antiviral efficacy and low cytotoxicity. Antivir. Res. 2020, 182, 104916. [Google Scholar] [CrossRef] [PubMed]
- Adcock, N.J.; Rice, E.W.; Sivaganesan, M.; Brown, J.D.; Stallknecht, D.E.; Swayne, D.E. The use of bacteriophages of the family Cystoviridae as surrogates for H5N1 highly pathogenic avian influenza viruses in persistence and inactivation studies. J. Environ. Sci. Health A Tox Hazard. Subst. Environ. Eng. 2009, 44, 1362–1366. [Google Scholar] [CrossRef] [PubMed]
- Fedorenko, A.; Grinberg, M.; Orevi, T.; Kashtan, N. Survival of the enveloped bacteriophage phi6 (a surrogate for SARS-CoV-2) in evaporated saliva microdroplets deposited on glass surfaces. Sci. Rep. 2020, 10, 22419. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.P.; Richter, W.; Sunderman, M.; Calfee, M.W.; Serre, S.; Mickelsen, L. Evaluating the environmental persistence and inactivation of MS2 bacteriophage and the presumed Ebola virus surrogate phi6 using low concentration hydrogen peroxide vapor. Environ. Sci. Technol. 2020, 54, 3581–3590. [Google Scholar] [CrossRef]
- Oksanen, L.; Auvinen, M.; Kuula, J.; Malmgren, R.; Romantschuk, M.; Hyvärinen, A.; Laitinen, S.; Maunula, L.; Sanmark, E.; Geneid, A.; et al. Combining phi6 as a surrogate virus and computational large-eddy simulations to study airborne transmission of SARS-CoV-2 in a restaurant. Indoor Air 2022, 32, e13165. [Google Scholar] [CrossRef]
- Kenney, J.M.; Hantula, J.; Fuller, S.D.; Mindich, L.; Ojala, P.M.; Bamford, D.H. Bacteriophage phi6 envelope elucidated by chemical cross-linking, immunodetection, and cryoelectron microscopy. Virology 1992, 190, 635–644. [Google Scholar] [CrossRef]
- Jäälinoja, H.T.; Huiskonen, J.T.; Butcher, S.J. Electron cryomicroscopy comparison of the architectures of the enveloped bacteriophages phi6 and phi8. Structure 2007, 15, 157–167. [Google Scholar] [CrossRef]
- Ilca, S.L.; Sun, X.; El Omari, K.; Kotecha, A.; de Haas, F.; DiMaio, F.; Grimes, J.M.; Stuart, D.I.; Poranen, M.M.; Huiskonen, J.T. Multiple liquid crystalline geometries of highly compacted nucleic acid in a dsRNA virus. Nature 2019, 570, 252–256. [Google Scholar] [CrossRef]
- Sun, X.; Ilca, S.L.; Huiskonen, J.T.; Poranen, M.M. Dual role of a viral polymerase in viral genome replication and particle self-assembly. mBio 2018, 9, e01242-18. [Google Scholar] [CrossRef]
- O’Keefe, K.J.; Silander, O.K.; McCreery, H.; Weinreich, D.M.; Wright, K.M.; Chao, L.; Edwards, S.V.; Remold, S.K.; Turner, P.E. Geographic differences in sexual reassortment in RNA phage. Evolution 2010, 64, 3010–3023. [Google Scholar] [CrossRef]
- Silander, O.K.; Weinreich, D.M.; Wright, K.M.; O’Keefe, K.J.; Rang, C.U.; Turner, P.E.; Chao, L. Widespread genetic exchange among terrestrial bacteriophages. Proc. Natl. Acad. Sci. USA 2005, 102, 19009–19014. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, J.M.; Hartmann, D.; Bartosova-Sojkova, P.; Debat, H.; Moos, M.; Simek, P.; Fara, J.; Palus, M.; Kucera, M.; Hajdusek, O.; et al. Blood-feeding adaptations and virome assessment of the poultry red mite Dermanyssus gallinae guided by RNA-seq. Commun. Biol. 2023, 6, 517. [Google Scholar] [CrossRef]
- Semancik, J.S.; Vidaver, A.K.; Van Etten, J.L. Characterization of segmented double-helical RNA from bacteriophage phi6. J. Mol. Biol. 1973, 78, 617–625. [Google Scholar] [CrossRef]
- Bamford, D.H.; Palva, E.T.; Lounatmaa, K. Ultrastructure and life cycle of the lipid-containing bacteriophage phi6. J. Gen. Virol. 1976, 32, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Leo-Macias, A.; Katz, G.; Wei, H.; Alimova, A.; Katz, A.; Rice, W.J.; Diaz-Avalos, R.; Hu, G.B.; Stokes, D.L.; Gottlieb, P. Toroidal surface complexes of bacteriophage phi12 are responsible for host-cell attachment. Virology 2011, 414, 103–109. [Google Scholar] [CrossRef]
- Hu, G.B.; Wei, H.; Rice, W.J.; Stokes, D.L.; Gottlieb, P. Electron cryo-tomographic structure of cystovirus phi12. Virology 2008, 372, 1–9. [Google Scholar] [CrossRef]
- Sinclair, J.F.; Tzagoloff, A.; Levine, D.; Mindich, L. Proteins of bacteriophage phi6. J. Virol. 1975, 16, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Roine, E.; Raineri, D.M.; Romantschuk, M.; Wilson, M.; Nunn, D.N. Characterization of type IV pilus genes in Pseudomonas syringae pv. tomato DC3000. Mol. Plant Microbe Interact. 1998, 11, 1048–1056. [Google Scholar] [CrossRef]
- Romantschuk, M.; Bamford, D.H. Function of pili in bacteriophage phi6 penetration. J. Gen. Virol. 1985, 66, 2461–2469. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Shen, W.; Zhong, Q.; Chen, Q.; He, X.; Baker, J.L.; Xiong, K.; Jin, X.; Wang, J.; Hu, F.; et al. Development of a bacteriophage cocktail to constrain the emergence of phage-resistant Pseudomonas aeruginosa. Front. Microbiol. 2020, 11, 327. [Google Scholar] [CrossRef]
- Mäntynen, S.; Laanto, E.; Oksanen, H.M.; Poranen, M.M.; Diaz-Munoz, S.L. Black box of phage-bacterium interactions: Exploring alternative phage infection strategies. Open Biol. 2021, 11, 210188. [Google Scholar] [CrossRef]
- Onodera, S.; Olkkonen, V.M.; Gottlieb, P.; Strassman, J.; Qiao, X.Y.; Bamford, D.H.; Mindich, L. Construction of a transducing virus from double-stranded RNA bacteriophage phi6: Establishment of carrier states in host cells. J. Virol. 1992, 66, 190–196. [Google Scholar] [CrossRef]
- Romantschuk, M.; Bamford, D.H. Phi6-resistant phage-producing mutants of Pseudomonas phaseolicola. J. Gen. Virol. 1981, 56, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Ktistakis, N.T.; Lang, D. The dodecahedral framework of the bacteriophage phi6 nucleocapsid is composed of protein P1. J. Virol. 1987, 61, 2621–2623. [Google Scholar] [CrossRef] [PubMed]
- Olkkonen, V.M.; Bamford, D.H. The nucleocapsid of the lipid-containing double-stranded RNA bacteriophage phi6 contains a protein skeleton consisting of a single polypeptide species. J. Virol. 1987, 61, 2362–2367. [Google Scholar] [CrossRef] [PubMed]
- Butcher, S.J.; Dokland, T.; Ojala, P.M.; Bamford, D.H.; Fuller, S.D. Intermediates in the assembly pathway of the double-stranded RNA virus phi6. EMBO J. 1997, 16, 4477–4487. [Google Scholar] [CrossRef]
- Huiskonen, J.T.; de Haas, F.; Bubeck, D.; Bamford, D.H.; Fuller, S.D.; Butcher, S.J. Structure of the bacteriophage phi6 nucleocapsid suggests a mechanism for sequential RNA packaging. Structure 2006, 14, 1039–1048. [Google Scholar] [CrossRef]
- Wei, H.; Cheng, R.H.; Berriman, J.; Rice, W.J.; Stokes, D.L.; Katz, A.; Morgan, D.G.; Gottlieb, P. Three-dimensional structure of the enveloped bacteriophage phi12: An incomplete T = 13 lattice is superposed on an enclosed T = 1 shell. PLoS ONE 2009, 4, e6850. [Google Scholar] [CrossRef]
- Sun, Z.; El Omari, K.; Sun, X.; Ilca, S.L.; Kotecha, A.; Stuart, D.I.; Poranen, M.M.; Huiskonen, J.T. Double-stranded RNA virus outer shell assembly by bona fide domain-swapping. Nat. Commun. 2017, 8, 14814. [Google Scholar] [CrossRef]
- Cvirkaite-Krupovic, V.; Poranen, M.M.; Bamford, D.H. Phospholipids act as secondary receptor during the entry of the enveloped, double-stranded RNA bacteriophage phi6. J. Gen. Virol. 2010, 91, 2116–2120. [Google Scholar] [CrossRef] [PubMed]
- Poranen, M.M.; Daugelavicius, R.; Ojala, P.M.; Hess, M.W.; Bamford, D.H. A novel virus-host cell membrane interaction: Membrane voltage-dependent endocytic-like entry of bacteriophage phi6 nucleocapsid. J. Cell Biol. 1999, 147, 671–682. [Google Scholar] [CrossRef]
- Romantschuk, M.; Olkkonen, V.M.; Bamford, D.H. The nucleocapsid of bacteriophage phi6 penetrates the host cytoplasmic membrane. EMBO J. 1988, 7, 1821–1829. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Qiao, X.; Qiao, J.; Onodera, S.; Mindich, L. Unique properties of the inner core of bacteriophage phi8, a virus with a segmented dsRNA genome. Virology 2003, 308, 354–361. [Google Scholar] [CrossRef]
- Nemecek, D.; Boura, E.; Wu, W.; Cheng, N.; Plevka, P.; Qiao, J.; Mindich, L.; Heymann, J.B.; Hurley, J.H.; Steven, A.C. Subunit folds and maturation pathway of a dsRNA virus capsid. Structure 2013, 21, 1374–1383. [Google Scholar] [CrossRef]
- Ilca, S.L.; Kotecha, A.; Sun, X.; Poranen, M.M.; Stuart, D.I.; Huiskonen, J.T. Localized reconstruction of subunits from electron cryomicroscopy images of macromolecular complexes. Nat. Commun. 2015, 6, 8843. [Google Scholar] [CrossRef] [PubMed]
- El Omari, K.; Sutton, G.; Ravantti, J.J.; Zhang, H.; Walter, T.S.; Grimes, J.M.; Bamford, D.H.; Stuart, D.I.; Mancini, E.J. Plate tectonics of virus shell assembly and reorganization in phage phi8, a distant relative of mammalian reoviruses. Structure 2013, 21, 1384–1395. [Google Scholar] [CrossRef]
- Luque, D.; Mata, C.P.; Suzuki, N.; Ghabrial, S.A.; Caston, J.R. Capsid structure of dsRNA fungal viruses. Viruses 2018, 10, 481. [Google Scholar] [CrossRef]
- Miyazaki, N.; Salaipeth, L.; Kanematsu, S.; Iwasaki, K.; Suzuki, N. Megabirnavirus structure reveals a putative 120-subunit capsid formed by asymmetrical dimers with distinctive large protrusions. J. Gen. Virol. 2015, 96, 2435–2441. [Google Scholar] [CrossRef] [PubMed]
- Bamford, D.H.; Burnett, R.M.; Stuart, D.I. Evolution of viral structure. Theor. Popul. Biol. 2002, 61, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Butcher, S.J.; Grimes, J.M.; Makeyev, E.V.; Bamford, D.H.; Stuart, D.I. A mechanism for initiating RNA-dependent RNA polymerization. Nature 2001, 410, 235–240. [Google Scholar] [CrossRef]
- Ren, Z.; Franklin, C.M.; Ghose, R. Structure of the RNA-directed RNA polymerase from the cystovirus phi12. Proteins 2013, 81, 1479–1484. [Google Scholar] [CrossRef] [PubMed]
- Mönttinen, H.A.M.; Ravantti, J.J.; Poranen, M.M. Structure unveils relationships between RNA virus polymerases. Viruses 2021, 13, 313. [Google Scholar] [CrossRef]
- Gottlieb, P.; Alimova, A. RNA packaging in the cystovirus bacteriophages: Dynamic interactions during capsid maturation. Int. J. Mol. Sci. 2022, 23, 2677. [Google Scholar] [CrossRef] [PubMed]
- El Omari, K.; Meier, C.; Kainov, D.; Sutton, G.; Grimes, J.M.; Poranen, M.M.; Bamford, D.H.; Tuma, R.; Stuart, D.I.; Mancini, E.J. Tracking in atomic detail the functional specializations in viral RecA helicases that occur during evolution. Nucleic Acids Res. 2013, 41, 9396–9410. [Google Scholar] [CrossRef] [PubMed]
- Mancini, E.J.; Kainov, D.E.; Grimes, J.M.; Tuma, R.; Bamford, D.H.; Stuart, D.I. Atomic snapshots of an RNA packaging motor reveal conformational changes linking ATP hydrolysis to RNA translocation. Cell 2004, 118, 743–755. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Y.; Samad, A.; Zheng, P.; Ji, Z.; Chen, F.; Zhang, H.; Jin, T. Structure and mutation analysis of the hexameric P4 from Pseudomonas aeruginosa phage phiYY. Int. J. Biol. Macromol. 2022, 194, 42–49. [Google Scholar] [CrossRef]
- Mäntynen, S.; Sundberg, L.R.; Poranen, M.M. Recognition of six additional cystoviruses: Pseudomonas virus phi6 is no longer the sole species of the family Cystoviridae. Arch. Virol. 2018, 163, 1117–1124. [Google Scholar] [CrossRef]
- Gottlieb, P.; Qiao, X.; Strassman, J.; Frilander, M.; Mindich, L. Identification of the packaging regions within the genomic RNA segments of bacteriophage phi6. Virology 1994, 200, 42–47. [Google Scholar] [CrossRef]
- Frilander, M.; Gottlieb, P.; Strassman, J.; Bamford, D.H.; Mindich, L. Dependence of minus-strand synthesis on complete genomic packaging in the double-stranded RNA bacteriophage phi6. J. Virol. 1992, 66, 5013–5017. [Google Scholar] [CrossRef]
- Poranen, M.M.; Tuma, R.; Bamford, D.H. Assembly of double-stranded RNA bacteriophages. Adv. Virus Res. 2005, 64, 15–43. [Google Scholar]
- Qiao, X.; Qiao, J.; Mindich, L. Interference with bacteriophage phi6 genomic RNA packaging by hairpin structures. J. Virol. 1995, 69, 5502–5505. [Google Scholar] [CrossRef]
- Gottlieb, P.; Metzger, S.; Romantschuk, M.; Carton, J.; Strassman, J.; Bamford, D.H.; Kalkkinen, N.; Mindich, L. Nucleotide sequence of the middle dsRNA segment of bacteriophage phi6: Placement of the genes of membrane-associated proteins. Virology 1988, 163, 183–190. [Google Scholar] [CrossRef]
- Fenner, F. Classification and nomenclature of viruses. Second report of the International Committee on Taxonomy of Viruses. Intervirology 1976, 7, 1–115. [Google Scholar] [CrossRef]
- ICTV. Plenary Session vote. In Proceedings of the Minutes of the 4th meeting of the ICTV, The Hague, The Netherlands, 30 August 1978. [Google Scholar]
- Poranen, M.M.; Mäntynen, S. Cystoviridae. In The Online Report of the International Committee on Taxonomy of Viruses; International Committee on Taxonomy of Viruses (ICTV): Online, 2019; Available online: http://taxonomy.cvr.gla.ac.uk/PDF/Cystoviridae.pdf (accessed on 1 September 2023).
- International Committee on Taxonomy of Viruses Executive. The new scope of virus taxonomy: Partitioning the virosphere into 15 hierarchical ranks. Nat. Microbiol. 2020, 5, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, P.; Adriaenssens, E.M.; Zerbini, F.M.; Abrescia, N.G.A.; Aiewsakun, P.; Alfenas-Zerbini, P.; Bao, Y.; Barylski, J.; Drosten, C.; Duffy, S.; et al. Four principles to establish a universal virus taxonomy. PLoS Biol. 2023, 21, e3001922. [Google Scholar] [CrossRef] [PubMed]
- Wolf, Y.I.; Kazlauskas, D.; Iranzo, J.; Lucia-Sanz, A.; Kuhn, J.H.; Krupovic, M.; Dolja, V.V.; Koonin, E.V. Origins and evolution of the global RNA virome. mBio 2018, 9, e02329-18. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, L.A.M.; Pereira, C.; Frazao, C.; Balcao, V.M.; Almeida, A. Efficiency of phage phi6 for biocontrol of Pseudomonas syringae pv. syringae: An in vitro preliminary study. Microorganisms 2019, 7, 286. [Google Scholar]
- Li, L.; Zhong, Q.; Zhao, Y.; Bao, J.; Liu, B.; Zhong, Z.; Wang, J.; Yang, L.; Zhang, T.; Cheng, M.; et al. First-in-human application of double-stranded RNA bacteriophage in the treatment of pulmonary Pseudomonas aeruginosa infection. Microb. Biotechnol. 2023, 16, 862–867. [Google Scholar] [CrossRef] [PubMed]
| Virus Species 1, Phage Isolate | GenBank Accession Number (Segments L, M and S) | Isolation Host | Isolation Source, Country | Reference |
|---|---|---|---|---|
| Cystovirus phi6, Pseudomonas phage phi6 |
M17461, M17462, M12921 | Pseudomonas syringae pv. phaseolicola HB10Y | Phaseolus vulgaris (common bean), USA | [9] |
| Cystovirus phi8, Pseudomonas phage phi8 | AF226851, AF226852, AF226853 | P. syringae pv. phaseolicola LM2333 | Pisum sativum (pea), USA | [10,11] |
| Cystovirus phi12, Pseudomonas phage phi12 | AF408636, AY039807, AY034425 | P. syringae pv. phaseolicola LM2333 | Ocimum basilicum (bacil), USA | [10,12,13] |
| Cystovirus phi13, Pseudomonas phage phi13 | AF261668, AF261667, AF261666 | P. syringae pv. phaseolicola LM2333 | Raphanus sativum (radish), USA | [10,14] |
| Cystovirus phi2954, Pseudomonas phage phi2954 |
FJ608823, FJ608824, FJ608825 | P. syringae pv. phaseolicola LM2489 | Raphanus sativum , USA | [15] |
| Cystovirus phiNN, Pseudomonas phage phiNN |
KJ957164, KJ957165, KJ957166 | Pseudomonas sp. B314 | Lake water, Finland | [16] |
| Cystovirus phiYY, Pseudomonas phage phiYY | KX074201, KX074202, KX074203 | Pseudomonas aeruginosa PAO38 | Hospital sewage, China | [17] |
| ND, Microvirgula aerodenitrificans phage phiNY | MW471133, MW471134, MW471135 | Microvirgula aerodenitrificans LH11-4 | Fermented sour soup, China | [1] |
| ND, Pseudomonas phage phiZ98 | ON960064.1, ON960065.1, ON960066.1 | P. aeruginosa SK98 | Horse manure, China | [8] |
| ND, Pseudomonas phage CAP3 | MZ558504, MZ558505, MZ558506 | Acinetobacter radioresistens LH6 | Duck faeces, USA | [3] |
| ND, Pseudomonas phage CAP4 | MZ558507, MZ558508, MZ558509 | A. radioresistens LH6 | Turkey faeces, USA | [3] |
| ND, Pseudomonas phage CAP5 | MZ558510, MZ558511, MZ558512 | A. radioresistens LH6 | Turkey faeces, USA | [3] |
| ND, Pseudomonas phage CAP6 | MZ558513, MZ558514, MZ558515 | A. radioresistens LH6 | Turkey faeces, USA | [3] |
| ND, Pseudomonas phage CAP7 | MZ558516, MZ558517, MZ558518 | A. radioresistens LH6 | Turkey faeces, USA | [3] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mäntynen, S.; Salomaa, M.M.; Poranen, M.M. Diversity and Current Classification of dsRNA Bacteriophages. Viruses 2023, 15, 2154. https://doi.org/10.3390/v15112154
Mäntynen S, Salomaa MM, Poranen MM. Diversity and Current Classification of dsRNA Bacteriophages. Viruses. 2023; 15(11):2154. https://doi.org/10.3390/v15112154
Chicago/Turabian StyleMäntynen, Sari, Meri M. Salomaa, and Minna M. Poranen. 2023. "Diversity and Current Classification of dsRNA Bacteriophages" Viruses 15, no. 11: 2154. https://doi.org/10.3390/v15112154
APA StyleMäntynen, S., Salomaa, M. M., & Poranen, M. M. (2023). Diversity and Current Classification of dsRNA Bacteriophages. Viruses, 15(11), 2154. https://doi.org/10.3390/v15112154





