Antiviral Effect of Budesonide against SARS-CoV-2
Abstract
:1. Introduction
2. Materials and Methods
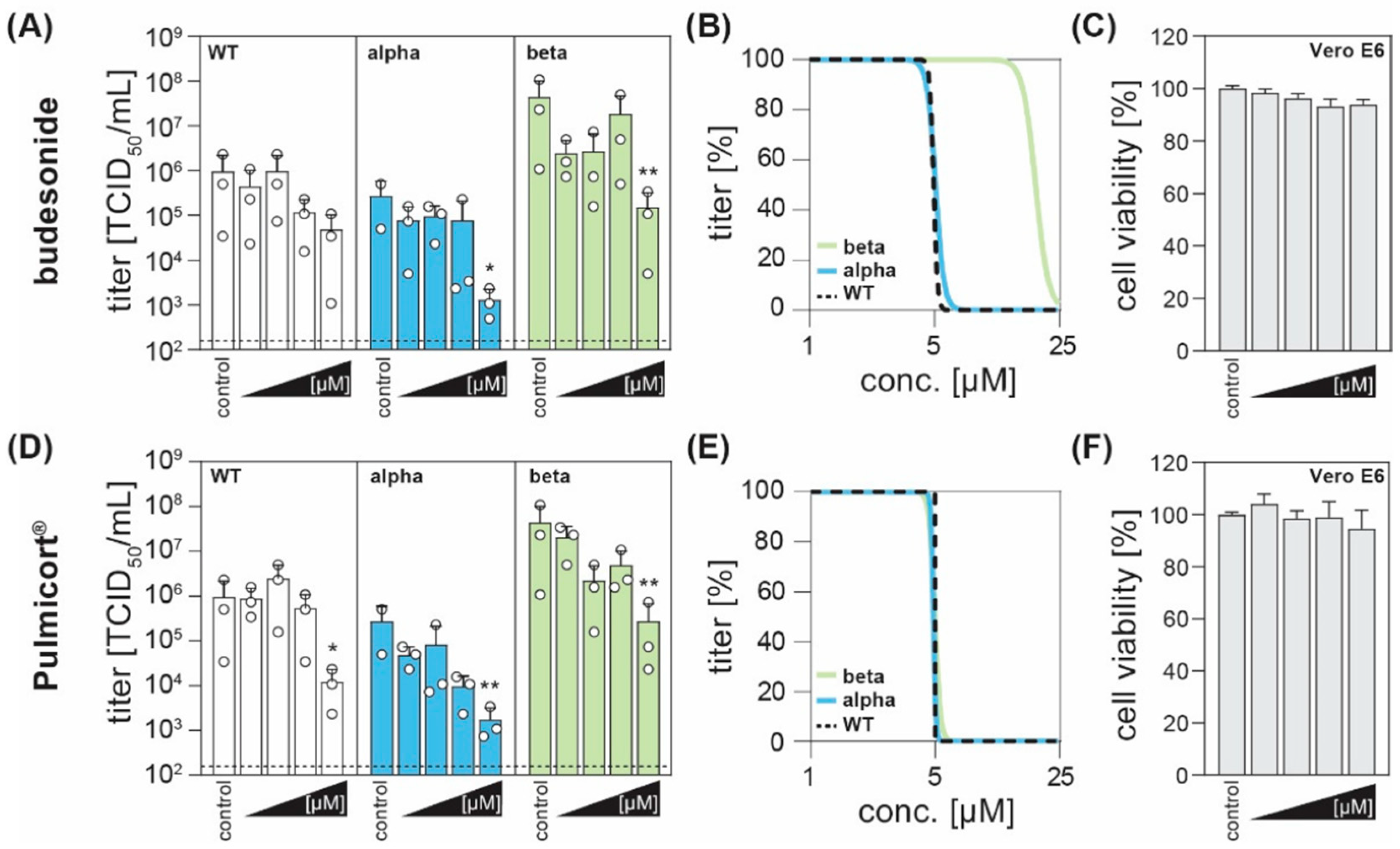
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Food and Drug Administration. Fact Sheet for Health Care Providers Emergency Use Authorization (EUA) of Bamlanivimab and Etesevimab. Available online: https://www.fda.gov/media/145802/download (accessed on 18 May 2021).
- Wang, Y.; Zhang, D.; Du, G.; Du, R.; Zhao, J.; Jin, Y.; Fu, S.; Gao, L.; Cheng, Z.; Lu, Q.; et al. Remdesivir in adults with severe COVID-19: A randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2020, 395, 1569–1578. [Google Scholar] [CrossRef]
- National Institutes of Health. Therapeutic Management of Adults With COVID-19. Available online: https://www.covid19treatmentguidelines.nih.gov/therapeutic-management/ (accessed on 18 May 2021).
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Hohmann, E.; Chu, H.Y.; Luetkemeyer, A.; Kline, S.; et al. Remdesivir for the Treatment of Covid-19—Final Report. N. Engl. J. Med. 2020, 383, 1813–1826. [Google Scholar] [CrossRef]
- Zhou, D.; Dejnirattisai, W.; Supasa, P.; Liu, C.; Mentzer, A.J.; Ginn, H.M.; Zhao, Y.; Duyvesteyn, H.M.E.; Tuekprakhon, A.; Nutalai, R.; et al. Evidence of escape of SARS-CoV-2 variant B.1.351 from natural and vaccine-induced sera. Cell 2021, 184, 2348–2361. [Google Scholar] [CrossRef]
- COVID-19 Genomics UK Consortium. COG-UK report on SARS-CoV-2 Spike mutations of interestin the UK. Available online: https://www.cogconsortium.uk/wp-content/uploads/2021/01/Report-2_COG-UK_SARS-CoV-2-Mutations.pdf (accessed on 18 May 2021).
- Ramakrishnan, S.; Nicolau, D.V.; Langford, B.; Mahdi, M.; Jeffers, H.; Mwasuku, C.; Krassowska, K.; Fox, R.; Binnian, I.; Glover, V.; et al. Inhaled budesonide in the treatment of early COVID-19 (STOIC): A phase 2, open-label, randomised controlled trial. Lancet Respir. Med. 2021. [Google Scholar] [CrossRef]
- Halpin, D.M.G.; Faner, R.; Sibila, O.; Badia, J.R.; Agusti, A. Do chronic respiratory diseases or their treatment affect the risk of SARS-CoV-2 infection? Lancet Respir. Med. 2020, 8, 436–438. [Google Scholar] [CrossRef]
- Zetterström, O.; Buhl, R.; Mellem, H.; Perpiñá, M.; Hedman, J.; O’Neill, S.; Ekström, T. Improved asthma control with budesonide/formoterol in a single inhaler, compared with budesonide alone. Eur. Respir. J. 2001, 18, 262–268. [Google Scholar] [CrossRef] [Green Version]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. Available online: https://ginasthma.org/wp-content/uploads/2021/04/GINA-2021-Main-Report_FINAL_21_04_28-WMS.pdf (accessed on 18 May 2021).
- Szafranski, W.; Cukier, A.; Ramirez, A.; Menga, G.; Sansores, R.; Nahabedian, S.; Peterson, S.; Olsson, H. Efficacy and safety of budesonide/formoterol in the management of chronic obstructive pulmonary disease. Eur. Respir. J. 2003, 21, 74–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.-R.; Song, J.-H.; Ahn, J.-H.; Lee, G.-S.; Ahn, H.; Yoon, S.-I.; Kang, S.G.; Kim, P.-H.; Jeon, S.-M.; Choi, E.-J.; et al. Antiviral and anti-inflammatory activity of budesonide against human rhinovirus infection mediated via autophagy activation. Antiviral Res. 2018, 151, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, S.; Kawase, M.; Nao, N.; Shirato, K.; Ujike, M.; Kamitani, W.; Shimojima, M.; Fukushi, S. The Inhaled Steroid Ciclesonide Blocks SARS-CoV-2 RNA Replication by Targeting the Viral Replication-Transcription Complex in Cultured Cells. J. Virol. 2020, 95. [Google Scholar] [CrossRef]
- Yamaya, M.; Nishimura, H.; Deng, X.; Sugawara, M.; Watanabe, O.; Nomura, K.; Shimotai, Y.; Momma, H.; Ichinose, M.; Kawase, T. Inhibitory effects of glycopyrronium, formoterol, and budesonide on coronavirus HCoV-229E replication and cytokine production by primary cultures of human nasal and tracheal epithelial cells. Respir. Investig. 2020, 58, 155–168. [Google Scholar] [CrossRef]
- World Health Organization. Therapeutics and COVID-19. Available online: https://apps.who.int/iris/bitstream/handle/10665/336729/WHO-2019-nCov-remdesivir-2020.1-eng.pdf (accessed on 18 May 2021).
- Ding, C.; Feng, X.; Chen, Y.; Yuan, J.; Yi, P.; Li, Y.; Ni, Q.; Zou, R.; Li, X.; Sheng, J.; et al. Effect of Corticosteroid Therapy on the Duration of SARS-CoV-2 Clearance in Patients with Mild COVID-19: A Retrospective Cohort Study. Infect. Dis. Ther. 2020, 9, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, L. Influence of Corticosteroid Dose on Viral Shedding Duration in Patients With COVID-19. Clin. Infect. Dis. 2021, 72, 1298–1300. [Google Scholar] [CrossRef]
- Arabi, Y.M.; Mandourah, Y.; Al-Hameed, F.; Sindi, A.A.; Almekhlafi, G.A.; Hussein, M.A.; Jose, J.; Pinto, R.; Al-Omari, A.; Kharaba, A.; et al. Corticosteroid Therapy for Critically Ill Patients with Middle East Respiratory Syndrome. Am. J. Respir. Crit. Care Med. 2018, 197, 757–767. [Google Scholar] [CrossRef]
- Li, S.; Hu, Z.; Song, X. High-dose but Not Low-dose Corticosteroids Potentially Delay Viral Shedding of Patients With COVID-19. Clin. Infect. Dis. 2021, 72, 1297–1298. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Murthy, S.; Diaz, J.V.; Slutsky, A.S.; Villar, J.; Angus, D.C.; Annane, D.; Azevedo, L.C.P.; Berwanger, O.; Cavalcanti, A.B.; et al. Association Between Administration of Systemic Corticosteroids and Mortality Among Critically Ill Patients With COVID-19: A Meta-analysis. JAMA 2020, 324, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in Hospitalized Patients with Covid-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health. Corticosteroids. Available online: https://www.covid19treatmentguidelines.nih.gov/immunomodulators/corticosteroids/ (accessed on 18 May 2021).
- Torres Acosta, M.A.; Singer, B.D. Pathogenesis of COVID-19-induced ARDS: Implications for an ageing population. Eur. Respir. J. 2020, 56. [Google Scholar] [CrossRef] [PubMed]
- Deliloglu, B.; Tuzun, F.; Cengiz, M.M.; Ozkan, H.; Duman, N. Endotracheal Surfactant Combined With Budesonide for Neonatal ARDS. Front. Pediatr. 2020, 8, 210. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.S.; Meguid, M.M.A. Effect of nebulized budesonide on respiratory mechanics and oxygenation in acute lung injury/acute respiratory distress syndrome: Randomized controlled study. Saudi J. Anaesth. 2017, 11, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.M.; Carroll, M.L.; Li, H.; Poh, A.M.; Kirkegard, D.; Towers, M.; Upham, J.W. Budesonide and formoterol reduce early innate anti-viral immune responses in vitro. PLoS ONE 2011, 6, e27898. [Google Scholar] [CrossRef] [Green Version]
- Gibson, P.G.; Saltos, N.; Fakes, K. Acute anti-inflammatory effects of inhaled budesonide in asthma: A randomized controlled trial. Am. J. Respir. Crit. Care Med. 2001, 163, 32–36. [Google Scholar] [CrossRef]
- Kelly, E.A.; Busse, W.W.; Jarjour, N.N. Inhaled budesonide decreases airway inflammatory response to allergen. Am. J. Respir. Crit. Care Med. 2000, 162, 883–890. [Google Scholar] [CrossRef]
- Mendes, E.S.; Rebolledo, P.; Campos, M.; Wanner, A. Immediate antiinflammatory effects of inhaled budesonide in patients with asthma. Ann. Am. Thorac. Soc. 2014, 11, 706–711. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heinen, N.; Meister, T.L.; Klöhn, M.; Steinmann, E.; Todt, D.; Pfaender, S. Antiviral Effect of Budesonide against SARS-CoV-2. Viruses 2021, 13, 1411. https://doi.org/10.3390/v13071411
Heinen N, Meister TL, Klöhn M, Steinmann E, Todt D, Pfaender S. Antiviral Effect of Budesonide against SARS-CoV-2. Viruses. 2021; 13(7):1411. https://doi.org/10.3390/v13071411
Chicago/Turabian StyleHeinen, Natalie, Toni Luise Meister, Mara Klöhn, Eike Steinmann, Daniel Todt, and Stephanie Pfaender. 2021. "Antiviral Effect of Budesonide against SARS-CoV-2" Viruses 13, no. 7: 1411. https://doi.org/10.3390/v13071411
APA StyleHeinen, N., Meister, T. L., Klöhn, M., Steinmann, E., Todt, D., & Pfaender, S. (2021). Antiviral Effect of Budesonide against SARS-CoV-2. Viruses, 13(7), 1411. https://doi.org/10.3390/v13071411







