An Appraisal of the Current Scenario in Vaccine Research for COVID-19
Abstract
:1. Introduction
2. Vaccination as a Promising Strategy Against COVID-19
3. COVID-19 Vaccine Development
3.1. BNT162b2
3.2. Sputnik V
3.3. EpiVacCorona
3.4. CoronaVac (PiCoVacc)
3.5. mRNA-1273
3.6. AZD1222
3.7. Covaxin (BBV152)
3.8. Ad26.COV2
3.9. BBIBP-CorV
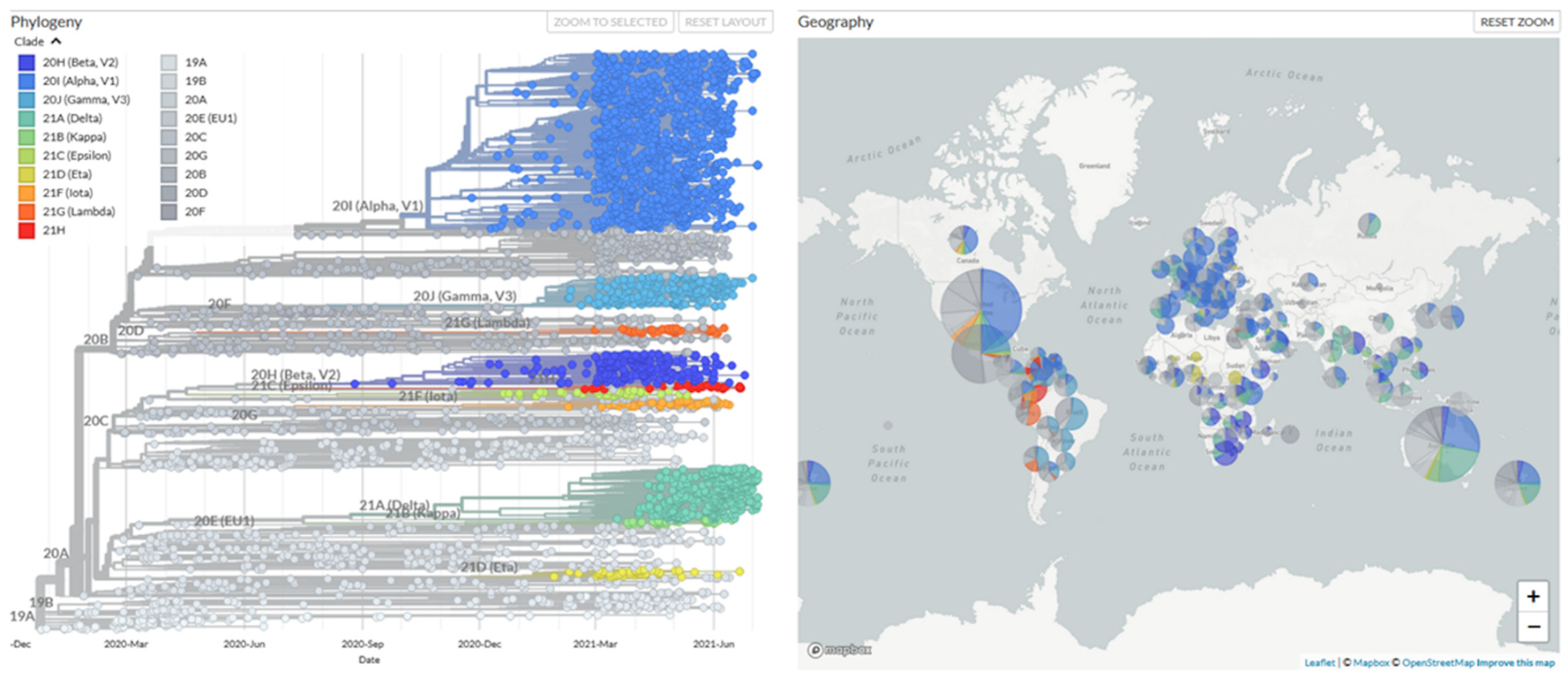
4. Efficacy of Vaccine Against Emerging SARS-CoV-2 Variant Strains
5. Conclusions
6. Opinion
7. Key Findings
- SARS-CoV-2 has been the causative viral pathogen responsible for the COVID-19 outbreak;
- Vaccination against SARS-CoV-2 has now become the main therapeutic strategy for eradicating COVID-19 from the community;
- Numerous vaccine candidates have been introduced, of which mRNA 1273, BNT162, AZD1222, CoronaVac, Sputnik V, EpiVaCorona, and Covaxin have been the leading vaccine candidates against SARS-CoV-2.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dong, E.; Du, H.; Gardner, L. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). Available online: https://www.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6 (accessed on 27 March 2021).
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet 2020, 20, 533–534. [Google Scholar] [CrossRef]
- NIH. Coronaviruses. Available online: https://www.niaid.nih.gov/diseases-conditions/coronaviruses (accessed on 27 March 2021).
- Shastri, M.D.; Shukla, S.D.; Chong, W.C.; Kc, R.; Dua, K.; Patel, R.P.; Peterson, G.M.; O’Toole, R.F. Smoking and COVID-19: What we know so far. Respir. Med. 2020, 106237. [Google Scholar] [CrossRef]
- Zimmer, C.; Corum, J.; Wee, S.-L. Coronavirus Vaccine Tracker. Available online: https://www.nytimes.com/interactive/2020/science/coronavirus-vaccine-tracker.html (accessed on 25 March 2021).
- Paavonen, J.; Naud, P.; Salmerón, J.; Wheeler, C.M.; Chow, S.-N.; Apter, D.; Kitchener, H.; Castellsagué, X.; Teixeira, J.C.; Skinner, S.R. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): Final analysis of a double-blind, randomised study in young women. Lancet 2009, 374, 301–314. [Google Scholar] [CrossRef]
- Giuliano, A.R.; Palefsky, J.M.; Goldstone, S.; Moreira, E.D., Jr.; Penny, M.E.; Aranda, C.; Vardas, E.; Moi, H.; Jessen, H.; Hillman, R. Efficacy of quadrivalent HPV vaccine against HPV Infection and disease in males. N. Engl. J. Med. 2011, 364, 401–411. [Google Scholar] [CrossRef] [Green Version]
- Riedel, S. Edward Jenner and the history of smallpox and vaccination. In Baylor University Medical Center Proceedings; Taylor & Francis: Abingdon, UK, 2005; Volume 18, pp. 21–25. [Google Scholar]
- Patel, M.; Zipursky, S.; Orenstein, W.; Garon, J.; Zaffran, M. Polio endgame: The global introduction of inactivated polio vaccine. Expert Rev. Vaccines 2015, 14, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Masihi, K.N. Fighting infection using immunomodulatory agents. Expert Opin. Biol. Ther. 2001, 1, 641–653. [Google Scholar] [CrossRef] [PubMed]
- NCBI. NCBI SARS-CoV-2 Resources; National Library of Medicine: Bethesda, MD, USA, 2020. [Google Scholar]
- Forster, P.; Forster, L.; Renfrew, C.; Forster, M. Phylogenetic network analysis of SARS-CoV-2 genomes. Proc. Natl. Acad. Sci. USA 2020, 117, 9241–9243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sah, R.; Rodriguez-Morales, A.J.; Jha, R.; Chu, D.K.; Gu, H.; Peiris, M.; Bastola, A.; Lal, B.K.; Ojha, H.C.; Rabaan, A.A. Complete genome sequence of a 2019 novel coronavirus (SARS-CoV-2) strain isolated in Nepal. Microbiol. Resour. Announc. 2020, 9, 1–3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, M.I.; Khan, Z.A.; Baig, M.H.; Ahmad, I.; Farouk, A.-E.; Song, Y.G.; Dong, J.-J. Comparative genome analysis of novel coronavirus (SARS-CoV-2) from different geographical locations and the effect of mutations on major target proteins: An in silico insight. PLoS ONE 2020, 15, e0238344. [Google Scholar] [CrossRef]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef] [Green Version]
- van Riel, D.; de Wit, E. Next-generation vaccine platforms for COVID-19. Nat. Mater. 2020, 19, 810–812. [Google Scholar] [CrossRef]
- Belshe, R.B.; Edwards, K.M.; Vesikari, T.; Black, S.V.; Walker, R.E.; Hultquist, M.; Kemble, G.; Connor, E.M. Live attenuated versus inactivated influenza vaccine in infants and young children. N. Engl. J. Med. 2007, 356, 685–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monto, A.S.; Ohmit, S.E.; Petrie, J.G.; Johnson, E.; Truscon, R.; Teich, E.; Rotthoff, J.; Boulton, M.; Victor, J.C. Comparative efficacy of inactivated and live attenuated influenza vaccines. N. Engl. J. Med. 2009, 361, 1260–1267. [Google Scholar] [CrossRef] [PubMed]
- Siedler, A.; Koch, J.; Ultsch, B.; Garbe, E.; von Kries, R.; Ledig, T.; Mertens, T.; Überla, K.; Zepp, F.; Hengel, H. Background paper to the decision not to recommend a standard vaccination with the live attenuated herpes zoster vaccine for the elderly in Germany. Bundesgesundheitsbl 2017, 60, 1162–1179. [Google Scholar]
- Kamboj, M.; Sepkowitz, K.A. Risk of transmission associated with live attenuated vaccines given to healthy persons caring for or residing with an immunocompromised patient. Infect. Control Hosp. Epidemiol. 2007, 28, 702–707. [Google Scholar] [CrossRef] [PubMed]
- Shinjoh, M.; Miyairi, I.; Hoshino, K.; Takahashi, T.; Nakayama, T. Effective and safe immunizations with live-attenuated vaccines for children after living donor liver transplantation. Vaccine 2008, 26, 6859–6863. [Google Scholar] [CrossRef] [PubMed]
- Chong, P.P.; Avery, R.K. A comprehensive review of immunization practices in solid organ transplant and hematopoietic stem cell transplant recipients. Clin. Ther. 2017, 39, 1581–1598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moyle, P.M.; Toth, I. Modern subunit vaccines: Development, components, and research opportunities. ChemMedChem 2013, 8, 360–376. [Google Scholar] [CrossRef]
- Tan, M.; Jiang, X. Recent advancements in combination subunit vaccine development. Hum. Vaccines Immunother. 2017, 13, 180–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, J.; Wang, W.; Chen, Z.; Lu, S.; Yang, F.; Bi, Z.; Bao, L.; Mo, F.; Li, X.; Huang, Y. A vaccine targeting the RBD of the S protein of SARS-CoV-2 induces protective immunity. Nature 2020, 586, 572–577. [Google Scholar] [CrossRef]
- Chi, X.; Yan, R.; Zhang, J.; Zhang, G.; Zhang, Y.; Hao, M.; Zhang, Z.; Fan, P.; Dong, Y.; Yang, Y. A neutralizing human antibody binds to the N-terminal domain of the Spike protein of SARS-CoV-2. Science 2020, 369, 650–655. [Google Scholar] [CrossRef]
- Cao, Y.; Zhu, X.; Hossen, M.N.; Kakar, P.; Zhao, Y.; Chen, X. Augmentation of vaccine-induced humoral and cellular immunity by a physical radiofrequency adjuvant. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Rauch, S.; Jasny, E.; Schmidt, K.E.; Petsch, B. New vaccine technologies to combat outbreak situations. Front. Immunol. 2018, 9, 1963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henao-Restrepo, A.M.; Longini, I.M.; Egger, M.; Dean, N.E.; Edmunds, W.J.; Camacho, A.; Carroll, M.W.; Doumbia, M.; Draguez, B.; Duraffour, S. Efficacy and effectiveness of an rVSV-vectored vaccine expressing Ebola surface glycoprotein: Interim results from the Guinea ring vaccination cluster-randomised trial. Lancet 2015, 386, 857–866. [Google Scholar] [CrossRef]
- Ura, T.; Okuda, K.; Shimada, M. Developments in viral vector-based vaccines. Vaccines 2014, 2, 624–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Vries, R.D.; Rimmelzwaan, G.F. Viral vector-based influenza vaccines. Hum. Vaccines Immunother. 2016, 12, 2881–2901. [Google Scholar] [CrossRef]
- Condit, R.C.; Kim, D.; Robertson, J.S.; Excler, J.-L.; Gurwith, M.; Monath, T.P.; Pavlakis, G.; Fast, P.E.; Smith, J.; Smith, E.R. The Brighton Collaboration standardized template for collection of key information for benefit-risk assessment of viral vector vaccines. Vaccine 2020, 38, 7708–7715. [Google Scholar] [CrossRef] [PubMed]
- Ulmer, J.B.; Wahren, B.; Liu, M.A. Gene-based vaccines: Recent technical and clinical advances. Trends Mol. Med. 2006, 12, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Pascolo, S. Messenger RNA-based vaccines. Expert Opin. Biol. Ther. 2004, 4, 1285–1294. [Google Scholar] [CrossRef] [PubMed]
- Ulmer, J.B.; Mason, P.W.; Geall, A.; Mandl, C.W. RNA-based vaccines. Vaccine 2012, 30, 4414–4418. [Google Scholar] [CrossRef]
- Jahanafrooz, Z.; Baradaran, B.; Mosafer, J.; Hashemzaei, M.; Rezaei, T.; Mokhtarzadeh, A.; Hamblin, M.R. Comparison of DNA and mRNA vaccines against cancer. Drug Discov. Today 2020, 25, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, A.; Aljabbari, A.; Lokras, A.; Foged, C.; Thakur, A. Opportunities and Challenges in the Delivery of mRNA-based Vaccines. Pharmaceutics 2020, 12, 102. [Google Scholar] [CrossRef] [Green Version]
- WHO. WHO R&D Blueprint Novel Coronavirus (nCov) Vaccine Prioritization for Clinical Trials. Available online: https://www.who.int/publications/i/item/who-r-d-blueprint-novel-coronavirus-ncov-vaccine-prioritization-for-clinical-trials (accessed on 17 December 2020).
- Pfizer. A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates against COVID-19 in Healthy Individuals; Pfizer: New York, NY, USA, 2020; pp. 1–137. [Google Scholar]
- Vogel, A.; Kanevsky, I.; Che, Y.; Swanson, K.; Muik, A.; Vormehr, M.; Kranz, L.; Walzer, K.; Hein, S.; Gueler, A. A prefusion SARS-CoV-2 spike RNA vaccine is highly immunogenic and prevents lung infection in non-human primates. bioRxiv 2020, 1–38. [Google Scholar] [CrossRef]
- Walsh, E.E.; Frenck, R.W., Jr.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R. Safety and immunogenicity of two RNA-based Covid-19 vaccine candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef] [PubMed]
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D. COVID-19 vaccine BNT162b1 elicits human antibody and TH 1 T cell responses. Nature 2020, 586, 594–599. [Google Scholar] [CrossRef]
- MHRA (Ed.) Regulatory Approval of Pfizer/BioNTech Vaccine for COVID-19; Government of the United Kingdom: London, UK, 2020. [Google Scholar]
- Jansen, K.; Gruber, W. BNT162b2 Vaccine Candidate against COVID-19. FDA: Silver Spring, MD, USA, 2020; p. 64. Available online: https://stacks.cdc.gov/view/cdc/105431 (accessed on 17 December 2020).
- Hunter, P.R.; Brainard, J.S. Estimating the effectiveness of the Pfizer COVID-19 BNT162b2 vaccine after a single dose. A reanalysis of a study of’real-world’vaccination outcomes from Israel. Medrxiv 2021. [Google Scholar] [CrossRef]
- WHO. WHO Issues its First Emergency Use Validation for a COVID-19 Vaccine and Emphasizes Need for Equitable Global Access. Available online: https://www.who.int/news/item/31-12-2020-who-issues-its-first-emergency-use-validation-for-a-covid-19-vaccine-and-emphasizes-need-for-equitable-global-access (accessed on 31 December 2020).
- RioTimes. Argentina Grants Emergency Approval to Pfizer/BioNTech Vaccine. The RioTimes, 23 December 2020. [Google Scholar]
- HealthCanada. Pfizer-BioNTech COVID-19 Vaccine: Authorization Information. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/covid19-industry/drugs-vaccines-treatments/vaccines/pfizer-biontech/authorization.html (accessed on 8 January 2021).
- Montes, R.; Espana, S. Chile y Ecuador se adelantan en Sudamérica y autorizan la vacuna de Pfizer. El Pais, 17 December 2020. [Google Scholar]
- Zuniga, A. Costa Rica authorizes Pfizer-BioNTech coronavirus vaccine. The TicoTimes, 16 December 2020. [Google Scholar]
- RoyaNews. Obeidat confirms arrival of first batch of Pfizer vaccine Monday. RoyaNews, 10 January 2021. [Google Scholar]
- ABCNews. Bahrain approves Chinese COVID-19 vaccine for use. ABC News, 13 December 2020. [Google Scholar]
- NewYorkTimes. Covid-19: F.D.A. Clears Pfizer Vaccine and Millions of Doses Will Be Shipped Right Away. The New York Times, 15 December 2020. [Google Scholar]
- Castrellon, T. Panamá aprueba uso por emergencia de la vacuna Pfizer-BioNTech contra COVID-19. Telemetro, 15 December 2020. [Google Scholar]
- HSA. HSA Grants Interim Authorisation for First COVID-19 Vaccine in Singapore; Government of Singapore: Singapore, 2020. [Google Scholar]
- Radwan, R. Saudi Arabia ready to launch vaccine campaign. Arab News, 12 December 2020. [Google Scholar]
- FOPH (Ed.) Coronavirus: Vaccination; Swiss Government: Bern, Switzerland, 2021. [Google Scholar]
- Levine-Tiefenbrun, M.; Yelin, I.; Katz, R.; Herzel, E.; Golan, Z.; Schreiber, L.; Wolf, T.; Nadler, V.; Ben-Tov, A.; Kuint, J. Initial report of decreased SARS-CoV-2 viral load after inoculation with the BNT162b2 vaccine. Nat. Med. 2021, 27, 790–792. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrial. National Cohort Study of Effectiveness and Safety of SARS-CoV-2/COVID-19 Vaccines (ENFORCE) (ENFORCE). Available online: https://www.clinicaltrials.gov/ct2/show/NCT04760132?term=vaccine%2C+phase+4&cond=Covid19&draw=2 (accessed on 6 July 2021).
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatullin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: Two open, non-randomised phase 1/2 studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- ClinicalTrial. The Study of “Gam-COVID-Vac” Vaccine against COVID-19 with the Participation of Volunteers of 60 y.o and Older. Available online: https://clinicaltrials.gov/ct2/show/NCT04587219?term=NCT04587219&draw=2&rank=1 (accessed on 17 December 2020).
- ClinicalTrial. Clinical Trial of Efficacy, Safety, and Immunogenicity of Gam-COVID-Vac Vaccine against COVID-19 (RESIST). Available online: https://clinicaltrials.gov/ct2/show/NCT04530396?term=NCT04530396&draw=2&rank=1 (accessed on 17 December 2020).
- BBC. Coronavirus: Putin says vaccine has been approved for use. BBC News, 11 August 2020. [Google Scholar]
- Callaway, E. Russia’s fast-track coronavirus vaccine draws outrage over safety. Nature 2020, 584, 334–335. [Google Scholar] [CrossRef] [PubMed]
- Mahase, E. Covid-19: Russia approves vaccine without large scale testing or published results. BMJ 2020, 370, m3205. [Google Scholar] [CrossRef] [PubMed]
- Grzegorczyk, M. Belarus begins rollout of Russian Covid-19 vaccine. Emerging Europe, 29 December 2020. [Google Scholar]
- LiveMint. First 300,000 doses of Sputnik V vaccine arrive in Argentina to fight Covid-19. LiveMint, 24 December 2020. [Google Scholar]
- SouthernTimes. Algeria first in Africa to approve Sputnik V vaccine. The Southern Times, 11 January 2021. [Google Scholar]
- Wadhwa, T. Bolivia approves emergency use of Russia’s Sputnik V vaccine. People Dispatch, 7 January 2021. [Google Scholar]
- RFERL. Serbia Starts Inoculations With Russia’s Sputnik-V Vaccine. Radio Free Europe Radio Liberty, 6 January 2021. [Google Scholar]
- Reuters. Palestinians approve Russian COVID-19 vaccine for use in self-rule areas. Reuters, 11 January 2021. [Google Scholar]
- Reuters. Russia Completes Clinical Trials of Second Potential COVID-19 Vaccine: RIA. Available online: https://www.reuters.com/article/uk-health-coronavirus-russia-vaccine-vec/russia-completes-clinical-trials-of-second-potential-covid-19-vaccine-ria-idUSKBN26L13B (accessed on 17 December 2020).
- Ryzhikov, A.; Ryzhikov, Е.; Bogryantseva, M.; Usova, S.; Danilenko, E.; Nechaeva, E.; Pyankov, O.; Pyankova, O.; Gudymo, A.; Bodnev, S. A single blind, placebo-controlled randomized study of the safety, reactogenicity and immunogenicity of the “EpiVacCorona” Vaccine for the prevention of COVID-19, in volunteers aged 18–60 years (phase I–II). Russ. J. Infect. Immun. 2021, 11, 283–296. [Google Scholar] [CrossRef]
- ClinicalTrial. Study of the Tolerability, Safety, Immunogenicity and Preventive Efficacy of the EpiVacCorona Vaccine for the Prevention of COVID-19. Available online: https://www.clinicaltrials.gov/ct2/show/NCT04780035?term=vaccine&cond=Covid19&draw=2 (accessed on 6 July 2021).
- Bureau, H. Russia Approves 2nd Covid-19 Vaccine ‘EpiVacCorona’ after Sputnik V. Available online: https://www.healthwire.co/russia-approves-2nd-covid-19-vaccine-epivaccorona-after-sputnik-v/ (accessed on 17 December 2020).
- ClinicalTrial. Study of the Safety, Reactogenicity and Immunogenicity of “EpiVacCorona” Vaccine for the Prevention of COVID-19 (EpiVacCorona). Available online: https://clinicaltrials.gov/ct2/show/NCT04527575?term=NCT04527575&draw=2&rank=1 (accessed on 17 December 2020).
- Gao, Q.; Bao, L.; Mao, H.; Wang, L.; Xu, K.; Yang, M.; Li, Y.; Zhu, L.; Wang, N.; Lv, Z. Development of an inactivated vaccine candidate for SARS-CoV-2. Science 2020, 369, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Liu, W.; Zheng, Y.; Jiang, X.; Kou, G.; Ding, J.; Wang, Q.; Huang, Q.; Ding, Y.; Ni, W. A preliminary study on serological assay for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in 238 admitted hospital patients. Microbes Infect. 2020, 22, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zeng, G.; Pan, H.; Li, C.; Hu, Y.; Chu, K.; Han, W.; Chen, Z.; Tang, R.; Yin, W. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet 2020. [Google Scholar] [CrossRef]
- Wu, Z.; Hu, Y.; Xu, M.; Chen, Z.; Yang, W.; Jiang, Z.; Li, M.; Jin, H.; Cui, G.; Chen, P. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy adults aged 60 years and older: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect. Dis. 2021, 21, 803–812. [Google Scholar] [CrossRef]
- Reuters. Sinovac’s Coronavirus Vaccine Candidate Approved for Emergency Use in China—Source. Available online: https://www.reuters.com/article/us-health-coronavirus-china-vaccines/sinovacs-coronavirus-vaccine-candidate-approved-for-emergency-use-in-china-source-idUSKBN25O0Z3 (accessed on 17 December 2020).
- ClinicalTrial. Clinical Trial of Efficacy and Safety of Sinovac’s Adsorbed COVID-19 (Inactivated) Vaccine in Healthcare Professionals (PROFISCOV). Available online: https://clinicaltrials.gov/ct2/show/NCT04456595?term=NCT04456595&draw=2&rank=1 (accessed on 17 December 2020).
- ClinicalTrial. Clinical Trial for SARS-CoV-2 Vaccine (COVID-19). Available online: https://clinicaltrials.gov/ct2/show/NCT04582344?term=NCT04582344&draw=2&rank=1 (accessed on 17 December 2020).
- ClinicalTrial. Efficacy, Safety and Immunogenicity Study of SARS-CoV-2 Inactivated Vaccine (COVID-19). Available online: https://clinicaltrials.gov/ct2/show/NCT04508075?term=NCT04508075&draw=2&rank=1 (accessed on 17 December 2020).
- ModernaTX, Inc. Phase 3, Randomized, Stratified, Observer-Blind, Placebo-Controlled Study to Evaluate the Efficacy, Safety, and Immunogenicity of mRNA-1273 SARS-CoV-2 Vaccine in Adults Aged 18 Years and Older. ClinicalTrials.gov, 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04470427 (accessed on 17 December 2020).
- Hoffmann, M.; Kleine-Weber, H.; Pöhlmann, S. A multibasic cleavage site in the spike protein of SARS-CoV-2 is essential for infection of human lung cells. Mol. Cell 2020, 78, 779–784. [Google Scholar] [CrossRef] [PubMed]
- Toelzer, C.; Gupta, K.; Yadav, S.K.; Borucu, U.; Davidson, A.D.; Williamson, M.K.; Shoemark, D.K.; Garzoni, F.; Staufer, O.; Milligan, R. Free fatty acid binding pocket in the locked structure of SARS-CoV-2 spike protein. Science 2020, 370, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Salazar, E.; Kuchipudi, S.V.; Christensen, P.A.; Eagar, T.; Yi, X.; Zhao, P.; Jin, Z.; Long, S.W.; Olsen, R.J.; Chen, J. Convalescent plasma anti–SARS-CoV-2 spike protein ectodomain and receptor-binding domain IgG correlate with virus neutralization. J. Clin. Investig. 2020, 130, 6728–6738. [Google Scholar] [CrossRef]
- Chen, X.; Li, R.; Pan, Z.; Qian, C.; Yang, Y.; You, R.; Zhao, J.; Liu, P.; Gao, L.; Li, Z. Human monoclonal antibodies block the binding of SARS-CoV-2 spike protein to angiotensin converting enzyme 2 receptor. Cell. Mol. Immunol. 2020, 17, 647–649. [Google Scholar] [CrossRef] [PubMed]
- Corbett, K.S.; Edwards, D.K.; Leist, S.R.; Abiona, O.M.; Boyoglu-Barnum, S.; Gillespie, R.A.; Himansu, S.; Schäfer, A.; Ziwawo, C.T.; DiPiazza, A.T.; et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 2020, 586, 567–571. [Google Scholar] [CrossRef]
- Corbett, K.S.; Flynn, B.; Foulds, K.E.; Francica, J.R.; Boyoglu-Barnum, S.; Werner, A.P.; Flach, B.; O’Connell, S.; Bock, K.W.; Minai, M. Evaluation of the mRNA-1273 vaccine against SARS-CoV-2 in nonhuman primates. N. Engl. J. Med. 2020, 383, 1544–1555. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J. An mRNA vaccine against SARS-CoV-2—Preliminary report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J. Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef] [PubMed]
- Latestly. COVID-19 Vaccine EpiVacCorona Creates Immunity 1 Month After Inoculation, Says Russia’s Vector. Yahoo News, 27 November 2020. [Google Scholar]
- ClinicalTrial. Dose-Confirmation Study to Evaluate the Safety, Reactogenicity, and Immunogenicity of mRNA-1273 COVID-19 Vaccine in Adults Aged 18 Years and Older. Available online: https://clinicaltrials.gov/ct2/show/NCT04405076?term=NCT04405076&draw=2&rank=1 (accessed on 17 December 2020).
- ClinicalTrial. A Study to Evaluate Efficacy, Safety, and Immunogenicity of mRNA-1273 Vaccine in Adults Aged 18 Years and Older to Prevent COVID-19. Available online: https://clinicaltrials.gov/ct2/show/NCT04470427?term=NCT04470427&draw=2&rank=1 (accessed on 17 December 2020).
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- FDA. Coronavirus (COVID-19) Update: FDA Announces Advisory Committee Meeting to Discuss Second COVID-19 Vaccine Candidate. FDA News Release, 30 November 2020. [Google Scholar]
- EMA. EMA receives application for conditional marketing authorisation of Moderna COVID-19 vaccine. News, 1 December 2020. [Google Scholar]
- Reuters. UK starts real-time review of Moderna’s COVID-19 vaccine candidate. Reuters, 28 October 2020. [Google Scholar]
- Reuters. Switzerland starts ‘rolling review’ of Moderna COVID-19 vaccine. Reuters, 13 November 2020. [Google Scholar]
- ABC. US Food and Drug Administration authorises Moderna’s COVID-19 vaccine for emergency use. ABC News, 19 December 2020. [Google Scholar]
- HealthCanada (Ed.) Health Canada Authorizes Moderna COVID-19 Vaccine; Government of Canada: Ottawa, ON, Canada, 2020.
- EMA. EMA Recommends COVID-19 Vaccine Moderna for Authorisation in the EU; European Medicines Agency: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Hussey, C. Israeli Ministry of Health Authorizes COVID-19 Vaccine Moderna for Use in Israel; Moderna: Cambridge, MA, USA, 2021. [Google Scholar]
- Hussey, C. Swissmedic Authorizes COVID-19 Vaccine Moderna for Use in Switzerland. Businesswire, 12 January 2021. [Google Scholar]
- Hussey, C. United Kingdom Medicines and Healthcare Products Regulatory Agency Authorizes Use of COVID-19 Vaccine Moderna; Moderna: Cambridge, MA, USA, 2021. [Google Scholar]
- Van Doremalen, N.; Lambe, T.; Spencer, A.; Belij-Rammerstorfer, S.; Purushotham, J.N.; Port, J.R.; Avanzato, V.A.; Bushmaker, T.; Flaxman, A.; Ulaszewska, M.; et al. ChAdOx1 nCoV-19 vaccine prevents SARS-CoV-2 pneumonia in rhesus macaques. Nature 2020, 586, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet 2020, 396, 467–478. [Google Scholar] [CrossRef]
- ABCNews. Oxford says patient illness which forced pause to AstraZeneca coronavirus clinical trial ‘unlikely’ related to vaccine. ABCNews, 17 September 2020. [Google Scholar]
- Schwartz, L. Brazil Confirms Death of Volunteer in COVID-19 Vaccine Trial. MedScape, 21 October 2020. [Google Scholar]
- AstraZeneca. AZD1222 Clinical Trials Now Resumed Globally; AstraZeneca: Cambridge, MA, USA, 2020. [Google Scholar]
- Ramasamy, M.N.; Minassian, A.M.; Ewer, K.J.; Flaxman, A.L.; Folegatti, P.M.; Owens, D.R.; Voysey, M.; Aley, P.K.; Angus, B.; Babbage, G. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): A single-blind, randomised, controlled, phase 2/3 trial. Lancet 2020, 396, 1979–1993. [Google Scholar] [CrossRef]
- Duffy, K. AstraZeneca Admits the Best Results in Its COVID-19 Vaccine Study Came from a Dosing Error, and Experts Are Raising New Questions about the Shot; Business Insider: Sydney, Australia, 2020. [Google Scholar]
- MHRA (Ed.) Conditions of Authorisation for COVID-19 Vaccine AstraZeneca Agency; Government of United Kingdom: London, UK, 2021.
- PharmaLetter. Argentina Second to Back AstraZeneca’s COVID-19 Vaccine; The PharmaLetter: London, UK, 2020. [Google Scholar]
- VOA. Mexico Grants Emergency Approval to AstraZeneca-Oxford Coronavirus Vaccine. VOA News, 5 January 2021. [Google Scholar]
- AstraZeneca. AstraZeneca’s COVID-19 Vaccine Authorised in Five Other Countries; AstraZeneca: Cambridge, MA, USA, 2021. [Google Scholar]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Tiede, A.; Sachs, U.J.; Czwalinna, A.; Werwitzke, S.; Bikker, R.; Krauss, J.K.; Donnerstag, F.; Weißenborn, K.; Höglinger, G.; Maasoumy, B. Prothrombotic immune thrombocytopenia after COVID-19 vaccine. Blood 2021. [Google Scholar] [CrossRef]
- Sah, R.; Shrestha, S.; Mehta, R.; Sah, S.K.; Raaban, A.R.; Dharma, K.; Rodriguez-Morales, A.J. AZD1222 (Covishield) vaccination for COVID-19: Experiences, challenges and solutions in Nepal. Travel Med. Infect. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Román, G.C.; Gracia, F.; Torres, A.; Palacios, A.; Gracia, K.; Harris, D. Acute transverse myelitis (ATM): Clinical review of 43 patients with COVID-19-associated ATM and 3 post-vaccination ATM serious adverse events with the ChAdOx1 nCoV-19 vaccine (AZD1222). Front. Immunol. 2021, 12, 879. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Politou, M.; Ntanasis-Stathopoulos, I.; Karalis, V.; Merkouri, E.; Fotiou, D.; Gavriatopoulou, M.; Malandrakis, P.; Kastritis, E.; Trougakos, I.P. High Prevalence of Anti-PF4 Antibodies Following ChAdOx1 nCov-19 (AZD1222) Vaccination Even in the Absence of Thrombotic Events. Vaccines 2021, 9, 712. [Google Scholar] [CrossRef]
- BharatBiotech. COVAXIN—India’s First indigenous COVID-19 Vaccine. Available online: https://www.bharatbiotech.com/covaxin.html (accessed on 15 January 2021).
- Mohandas, S.; Yadav, P.D.; Shete, A.; Abraham, P.; Mohan, K.; Sapkal, G.; Mote, C.; Nyayanit, D.; Gupta, N.; Srinivas, V. Immunogenicity and protective efficacy of BBV152: A whole virion inactivated SARS CoV-2 vaccine in the Syrian hamster model. Res. Sq. 2020, 1, 1–25. [Google Scholar] [CrossRef]
- Yadav, P.; Ella, R.; Kumar, S.; Patil, D.; Mohandas, S.; Shete, A.; Bhati, G.; Sapkal, G.; Kaushal, H.; Patil, S. Remarkable immunogenicity and protective efficacy of BBV152, an inactivated SARS-CoV-2 vaccine in rhesus macaques. Res. Sq. 2020, 1, 1–17. [Google Scholar] [CrossRef]
- Ella, R.; Vadrevu, K.M.; Jogdand, H.; Prasad, S.; Reddy, S.; Sarangi, V.; Ganneru, B.; Sapkal, G.; Yadav, P.; Abraham, P.; et al. A Phase 1: Safety and Immunogenicity Trial of an Inactivated SARS-CoV-2 Vaccine-BBV152. medRxiv 2020, 1–34. [Google Scholar] [CrossRef]
- Ella, R.; Reddy, S.; Jogdand, H.; Sarangi, V.; Ganneru, B.; Prasad, S.; Das, D.; Raju, D.; Praturi, U.; Sapkal, G.; et al. Safety and immunogenicity clinical trial of an inactivated SARS-CoV-2 vaccine, BBV152 (a phase 2, double-blind, randomised controlled trial) and the persistence of immune responses from a phase 1 follow-up report. medRxiv 2020, 1–34. [Google Scholar] [CrossRef]
- BBCNews. Coronavirus: India approves vaccines from Bharat Biotech and Oxford/AstraZeneca. BBC News, 3 January 2021. [Google Scholar]
- 24GlobeNews. Bharat Biotech: Indian well being specialists voice their doubts, dismay on the hurried vaccine approval course of. 24 Globe News, 12 January 2021. [Google Scholar]
- FirstPost. Health experts voice doubts, concerns about hurried approval process for Bharat Biotech’s Covaxin. FirstPost, 12 January 2021. [Google Scholar]
- Frayer, L. India Prepares for Massive Vaccine Drive, But Some Fear It’s Moving Too Quickly. National Public Radio, 13 January 2021. [Google Scholar]
- Janssen. Clinical Trial COVID-19 Vaccine Candidate Underway; Janssen: Beerse, Belgium, 2021. [Google Scholar]
- WHO. Background Document on the Janssen Ad26.COV2.S (COVID-19) Vaccine; World Health Organization: Italy, Rome, 2021; Volume 1, p. 54. [Google Scholar]
- He, X.; Chandrashekar, A.; Zahn, R.; Wegmann, F.; Yu, J.; Mercado, N.B.; McMahan, K.; Martinot, A.J.; Piedra-Mora, C.; Beecy, S. Low-dose Ad26. COV2. S protection against SARS-CoV-2 challenge in rhesus macaques. Cell 2021. [Google Scholar] [CrossRef]
- Solforosi, L.; Kuipers, H.; Huber, S.K.R.; van der Lubbe, J.E.; Dekking, L.; Czapska-Casey, D.N.; Gil, A.I.; Baert, M.; Drijver, J.; Vaneman, J. Immunogenicity of one-and two-dose regimens of the Ad26. COV2. S COVID-19 vaccine candidate in adult and aged rhesus macaques. BioRxiv 2020. [Google Scholar] [CrossRef]
- Roozendaal, R.; Solforosi, L.; Stieh, D.; Serroyen, J.; Straetemans, R.; Wegmann, F.; Huber, S.K.R.; van der Lubbe, J.E.; Hendriks, J.; le Gars, M. SARS-CoV-2 binding and neutralizing antibody levels after vaccination with Ad26. COV2. S predict durable protection in rhesus macaques. bioRxiv 2021. [Google Scholar] [CrossRef]
- Van der Lubbe, J.E.; Huber, S.K.R.; Vijayan, A.; Dekking, L.; van Huizen, E.; Vreugdenhil, J.; Choi, Y.; Baert, M.R.; Feddes-de Boer, K.; Gil, A.I. Ad26. COV2. S-elicited immunity protects against G614 spike variant SARS-CoV-2 infection in Syrian hamsters and does not enhance respiratory disease in challenged animals with breakthrough infection after sub-optimal vaccine dosing. bioRxiv 2021. [Google Scholar] [CrossRef]
- Sadoff, J.; Le Gars, M.; Shukarev, G.; Heerwegh, D.; Truyers, C.; de Groot, A.M.; Stoop, J.; Tete, S.; Van Damme, W.; Leroux-Roels, I. Interim results of a phase 1–2a trial of Ad26. COV2. S Covid-19 vaccine. N. Engl. J. Med. 2021, 384, 1824–1835. [Google Scholar] [CrossRef] [PubMed]
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cárdenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.; Truyers, C.; Fennema, H.; Spiessens, B. Safety and efficacy of single-dose Ad26. COV2. S vaccine against Covid-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef] [PubMed]
- FDA. FDA Issues Emergency Use Authorization for Third COVID-19 Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/fda-issues-emergency-use-authorization-third-covid-19-vaccine (accessed on 17 December 2020).
- EMA. EMA Recommends COVID-19 Vaccine Janssen for Authorisation in the EU; European Medicines Agency: Amsterdam, The Netherlands, 2021. [Google Scholar]
- MHRA. Regulatory Approval of COVID-19 Vaccine Janssen; Medicines and Healthcare products Regulatory Agency: London, UK, 2021. [Google Scholar]
- Muir, K.-L.; Kallam, A.; Koepsell, S.A.; Gundabolu, K. Thrombotic thrombocytopenia after Ad26. COV2. S vaccination. N. Engl. J. Med. 2021, 384, 1964–1965. [Google Scholar] [CrossRef] [PubMed]
- Sadoff, J.; Davis, K.; Douoguih, M. Thrombotic thrombocytopenia after Ad26. COV2. S vaccination—response from the manufacturer. N. Engl. J. Med. 2021, 384, 1965–1966. [Google Scholar] [CrossRef] [PubMed]
- See, I.; Su, J.R.; Lale, A.; Woo, E.J.; Guh, A.Y.; Shimabukuro, T.T.; Streiff, M.B.; Rao, A.K.; Wheeler, A.P.; Beavers, S.F. US case reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26. COV2. S vaccination, March 2 to April 21, 2021. JAMA 2021. [Google Scholar] [CrossRef] [PubMed]
- WHO (Ed.) Background Document on the Inactivated COVID-19 Vaccine BIBP Developed by China National Biotec Group (CNBG); World Health Organization: Rome, Italy, 2021; p. 23. [Google Scholar]
- Wang, H.; Zhang, Y.; Huang, B.; Deng, W.; Quan, Y.; Wang, W.; Xu, W.; Zhao, Y.; Li, N.; Zhang, J. Development of an inactivated vaccine candidate, BBIBP-CorV, with potent protection against SARS-CoV-2. Cell 2020, 182, 713–721. [Google Scholar] [CrossRef]
- Xia, S.; Zhang, Y.; Wang, Y.; Wang, H.; Yang, Y.; Gao, G.F.; Tan, W.; Wu, G.; Xu, M.; Lou, Z. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBIBP-CorV: A randomised, double-blind, placebo-controlled, phase 1/2 trial. Lancet Infect. Dis. 2021, 21, 39–51. [Google Scholar] [CrossRef]
- Taylor, A. WHO grants emergency use authorization for Chinese-made Sinopharm coronavirus vaccine. The Washington Post, 2021; 1. [Google Scholar]
- WHO. WHO Lists Additional COVID-19 Vaccine for Emergency use and Issues Interim Policy Recommendations; World Health Organization: Rome, Italy, 2021. [Google Scholar]
- WHO (Ed.) Tracking SARS-CoV-2 Variants; World Health Organization: Rome, Italy, 2021; Volume 1, p. 33. [Google Scholar]
- Zhou, B.; Thao, T.T.N.; Hoffmann, D.; Taddeo, A.; Ebert, N.; Labroussaa, F.; Pohlmann, A.; King, J.; Steiner, S.; Kelly, J.N. SARS-CoV-2 spike D614G change enhances replication and transmission. Nature 2021, 592, 122–127. [Google Scholar] [CrossRef]
- Volz, E.; Hill, V.; McCrone, J.T.; Price, A.; Jorgensen, D.; O’Toole, Á.; Southgate, J.; Johnson, R.; Jackson, B.; Nascimento, F.F. Evaluating the effects of SARS-CoV-2 spike mutation D614G on transmissibility and pathogenicity. Cell 2021, 184, 64–75. [Google Scholar] [CrossRef]
- Korber, B.; Fischer, W.M.; Gnanakaran, S.; Yoon, H.; Theiler, J.; Abfalterer, W.; Hengartner, N.; Giorgi, E.E.; Bhattacharya, T.; Foley, B. Tracking changes in SARS-CoV-2 spike: Evidence that D614G increases infectivity of the COVID-19 virus. Cell 2020, 182, 812–827. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Garcia-Knight, M.A.; Khalid, M.M.; Servellita, V.; Wang, C.; Morris, M.K.; Sotomayor-González, A.; Glasner, D.R.; Reyes, K.R.; Gliwa, A.S. Transmission, infectivity, and antibody neutralization of an emerging SARS-CoV-2 variant in California carrying a L452R spike protein mutation. MedRxiv 2021. [Google Scholar] [CrossRef]
- Wang, P.; Nair, M.S.; Liu, L.; Iketani, S.; Luo, Y.; Guo, Y.; Wang, M.; Yu, J.; Zhang, B.; Kwong, P.D. Antibody resistance of SARS-CoV-2 variants B. 1.351 and B. 1.1. 7. Nature 2021, 593, 130–135. [Google Scholar] [CrossRef]
- Edara, V.V.; Floyd, K.; Lai, L.; Gardner, M.; Hudson, W.; Piantadosi, A.; Waggoner, J.; Babiker, A.; Ahmed, R.; Xie, X. Infection and mRNA-1273 vaccine antibodies neutralize SARS-CoV-2 UK variant. medRxiv 2021. [Google Scholar] [CrossRef]
- Wang, P.; Casner, R.G.; Nair, M.S.; Wang, M.; Yu, J.; Cerutti, G.; Liu, L.; Kwong, P.D.; Huang, Y.; Shapiro, L. Increased resistance of SARS-CoV-2 variant P. 1 to antibody neutralization. Cell Host Microbe 2021, 29, 747–751.e744. [Google Scholar] [CrossRef]
- Garcia-Beltran, W.F.; Lam, E.C.; Denis, K.S.; Nitido, A.D.; Garcia, Z.H.; Hauser, B.M.; Feldman, J.; Pavlovic, M.N.; Gregory, D.J.; Poznansky, M.C. Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell 2021, 184, 2372–2383. [Google Scholar] [CrossRef]
- Edara, V.-V.; Lai, L.; Sahoo, M.; Floyd, K.; Sibai, M.; Solis, D.; Flowers, M.W.; Hussaini, L.; Ciric, C.R.; Bechnack, S. Infection and vaccine-induced neutralizing antibody responses to the SARS-CoV-2 B. 1.617. 1 variant. bioRxiv 2021. [Google Scholar] [CrossRef]
- PHE. Investigation of Novel SARS-CoV-2 Variant: Variant of Concern 202012/01, Technical Briefing 3; Public Health England: London, UK, 2020. [Google Scholar]
- Mwenda, M.; Saasa, N.; Sinyange, N.; Busby, G.; Chipimo, P.J.; Hendry, J.; Kapona, O.; Yingst, S.; Hines, J.Z.; Minchella, P. Detection of B. 1.351 SARS-CoV-2 Variant Strain—Zambia.MMWR Morb. Mortal. Wkly Rep. 2021, 70, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Hirotsu, Y.; Omata, M. Discovery of a SARS-CoV-2 variant from the P. 1 lineage harboring K417T/E484K/N501Y mutations in Kofu, Japan. J. Infect. 2021, 82, 276–316. [Google Scholar] [CrossRef]
- Annavajhala, M.K.; Mohri, H.; Zucker, J.E.; Sheng, Z.; Wang, P.; Gomez-Simmonds, A.; Ho, D.D.; Uhlemann, A.-C. A novel SARS-CoV-2 variant of concern, B. 1.526, identified in New York. medRxiv 2021. [Google Scholar] [CrossRef]
- Hadfield, J.; Megill, C.; Bell, S.M.; Huddleston, J.; Potter, B.; Callender, C.; Sagulenko, P.; Bedford, T.; Neher, R.A. Nextstrain: Real-time tracking of pathogen evolution. Bioinformatics 2018, 34, 4121–4123. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.L.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Tessier, E.; Groves, N.; Dabrera, G.; Myers, R. Effectiveness of COVID-19 vaccines against the B. 1.617. 2 variant. medRxiv 2021. [Google Scholar] [CrossRef]
- Abu-Raddad, L.J.; Chemaitelly, H.; Butt, A.A. Effectiveness of the BNT162b2 Covid-19 Vaccine against the B. 1.1. 7 and B. 1.351 Variants. N. Engl. J. Med. 2021, 385, 187–189. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Dai, L.; Wang, H.; Hu, Z.; Tan, W.; Gao, G.F.; Yang, X. Neutralization of SARS-CoV-2 VOC 501Y. V2 by human antisera elicited by both inactivated BBIBP-CorV and recombinant dimeric RBD ZF2001 vaccines. BioRxiv 2021. [Google Scholar] [CrossRef]
- Wu, K.; Werner, A.P.; Koch, M.; Choi, A.; Narayanan, E.; Stewart-Jones, G.B.; Colpitts, T.; Bennett, H.; Boyoglu-Barnum, S.; Shi, W. Serum neutralizing activity elicited by mRNA-1273 vaccine. N. Engl. J. Med. 2021, 384, 1468–1470. [Google Scholar] [CrossRef] [PubMed]
| Vaccine Type | Candidate Vaccine | Developers | Trial Phase | Number of Virologically Confirmed Symptomatic Cases of COVID-19 | Efficacy Compared to Placebo for the Prevention of SARS-CoV-2 | Efficacy of Vaccine Against Severe and non-Severe COVID-19 | Efficacy: Seroconversion Rates | Assess Humoral Immunogenicity | Safety and Immunogenicity of a Booster Dose |
|---|---|---|---|---|---|---|---|---|---|
| Inactivated virus | CoronaVac | Sinovac Research and Development Co., Ltd | 4 | X | X | X | X | X | X |
| Inactivated virus | Vero cell | Sinopharm + China National Biotec Group Co + Wuhan Institute of Biological Products | 3 | X | X | X | X | X | |
| Inactivated virus | BBIBP-CorV | Sinopharm + China National Biotec Group Co + Beijing Institute of Biological Products | 4 | X | X | X | X | X | X |
| Viral vector (Non-replicating) | AZD1222 | AstraZeneca + University of Oxford | 4 | X | X | X | X | X | |
| Viral vector (Non-replicating) | Recombinant novel coronavirus vaccine (Adenovirus type 5 vector) | CanSino Biological Inc./Beijing Institute of Biotechnology | 4 | X | X | X | X | X | |
| Viral vector (Non-replicating) | Gam-COVID-Vac | Gamaleya Research Institute; Health Ministry of the Russian Federation | 3 | X | X | X | X | ||
| Viral vector (Non-replicating) | Ad26.COV2.S | Janssen Pharmaceutical | 4 | X | X | X | X | X | X |
| Protein subunit | NVX-CoV2373 | Novavax | 3 | X | X | X | X | X | X |
| RNA based vaccine | mRNA-1273 | Moderna + National Institute of Allergy and Infectious Diseases (NIAID) | 4 | X | X | X | X | X | X |
| RNA based vaccine | BNT162b2 (Comirnaty) | Pfizer/BioNTech + Fosun Pharma | 4 | X | X | X | X | X | |
| Protein subunit | CHO Cell | Anhui Zhifei Longcom Biopharmaceutical + Institute of Microbiology, Chinese Academy of Sciences | 3 | ||||||
| RNA based vaccine | CVnCoV Vaccine | CureVac AG | 3 | X | X | X | X | X | X |
| Inactivated virus | SARS-CoV-2 vaccine (vero cells) | Institute of Medical Biology + Chinese Academy of Medical Sciences | 3 | X | X | X | X | X | |
| Inactivated virus | QazCovid-in® | Research Institute for Biological Safety Problems, Rep of Kazakhstan | 3 | X | X | X | X | X | |
| DNA based vaccine | INO-4800 | Inovio Pharmaceuticals + International Vaccine Institute + Advaccine (Suzhou) Biopharmaceutical Co., Ltd | 2/3 | X | X | X | X | X | X |
| DNA based vaccine | AG0301-COVID19 | AnGes + Takara Bio + Osaka University | 2/3 | X | X | X | X | X | |
| DNA based vaccine | nCov vaccine | Zydus Cadila | 3 | X | X | X | X | X | |
| Inactivated virus | Covaxin | Bharat Biotech International Limited | 3 | X | X | X | X | X | X |
| Inactivated virus | Inactivated SARS-CoV-2 vaccine (Vero cell) | Shenzhen Kangtai Biological Products Co., Ltd. | 3 | X | X | X | X | X | |
| Protein subunit | EpiVacCorona | Federal Budgetary Research Institution State Research Center of Virology and Biotechnology “Vector” | 3 | X | X | X | X | X | X |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chong, W.C.; Chellappan, D.K.; Shukla, S.D.; Peterson, G.M.; Patel, R.P.; Jha, N.K.; Eri, R.D.; Dua, K.; Tambuwala, M.M.; Shastri, M.D. An Appraisal of the Current Scenario in Vaccine Research for COVID-19. Viruses 2021, 13, 1397. https://doi.org/10.3390/v13071397
Chong WC, Chellappan DK, Shukla SD, Peterson GM, Patel RP, Jha NK, Eri RD, Dua K, Tambuwala MM, Shastri MD. An Appraisal of the Current Scenario in Vaccine Research for COVID-19. Viruses. 2021; 13(7):1397. https://doi.org/10.3390/v13071397
Chicago/Turabian StyleChong, Wai Chin, Dinesh K. Chellappan, Shakti D. Shukla, Gregory M. Peterson, Rahul P. Patel, Niraj Kumar Jha, Rajaraman D. Eri, Kamal Dua, Murtaza M. Tambuwala, and Madhur D. Shastri. 2021. "An Appraisal of the Current Scenario in Vaccine Research for COVID-19" Viruses 13, no. 7: 1397. https://doi.org/10.3390/v13071397
APA StyleChong, W. C., Chellappan, D. K., Shukla, S. D., Peterson, G. M., Patel, R. P., Jha, N. K., Eri, R. D., Dua, K., Tambuwala, M. M., & Shastri, M. D. (2021). An Appraisal of the Current Scenario in Vaccine Research for COVID-19. Viruses, 13(7), 1397. https://doi.org/10.3390/v13071397










