Nanotechnology as a Shield against COVID-19: Current Advancement and Limitations
Abstract
:1. Introduction
2. Risk of Comorbidities in SARS-CoV-2 Infection
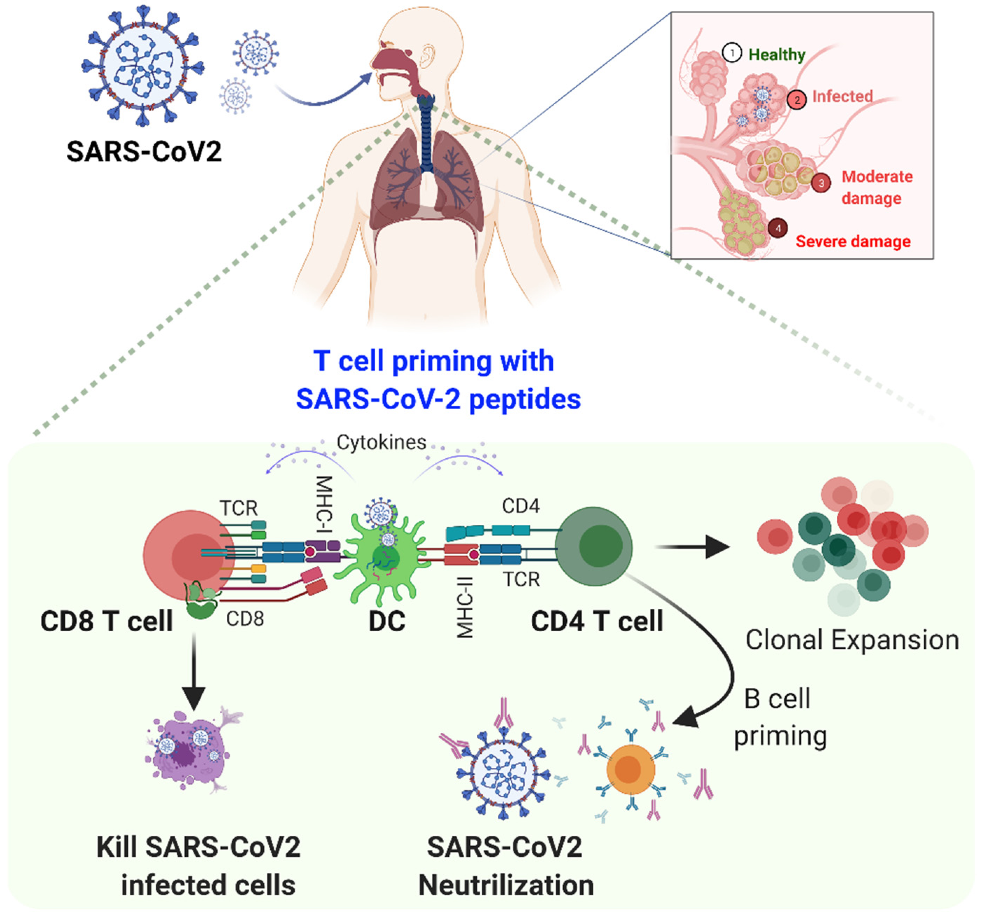
3. Mechanism of Immune Response after Infection of SARS-CoV-2
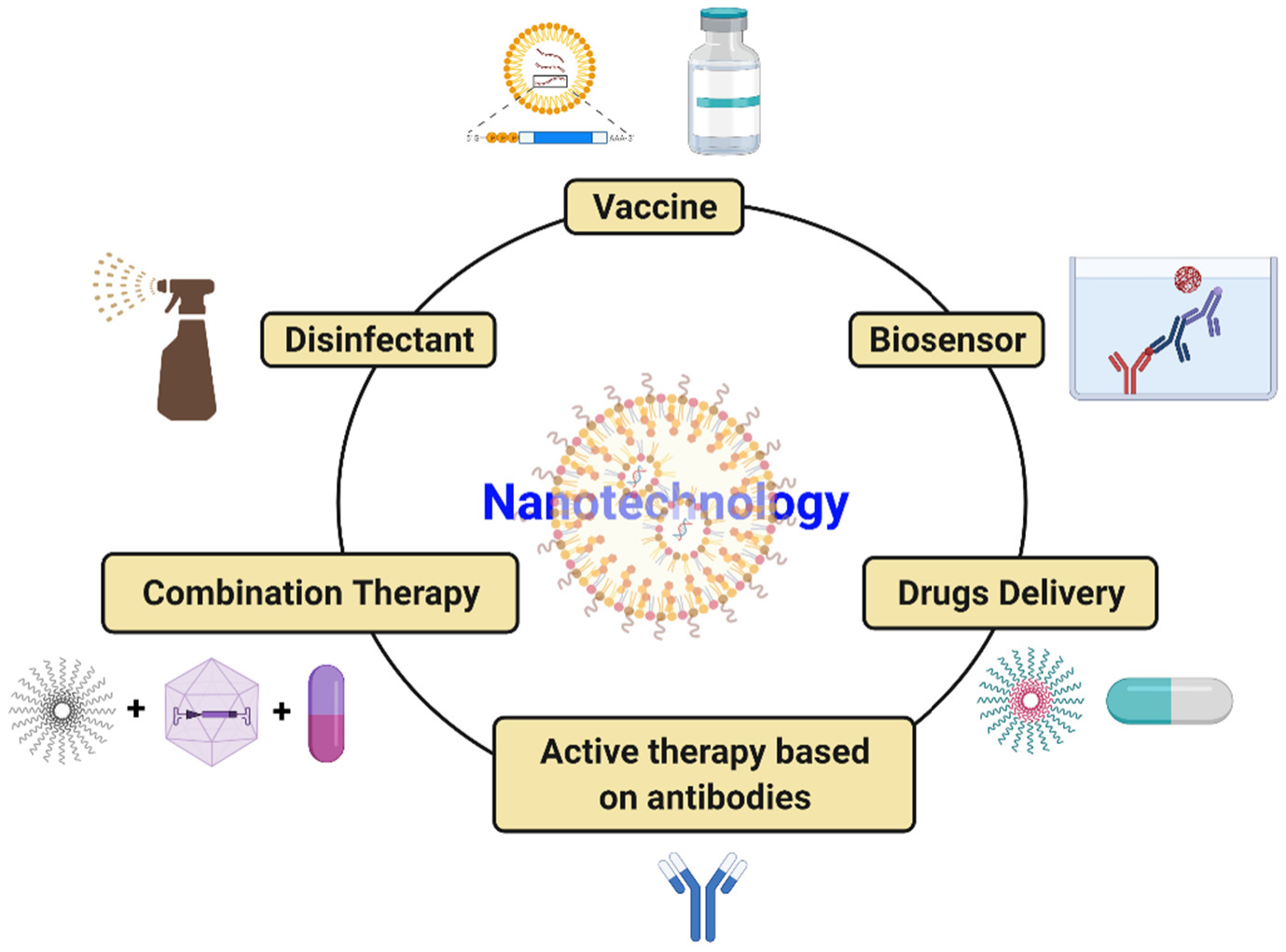
4. State-of-the-Art of Nanomaterials as Anti-SARS-CoV-2
4.1. Nanobiosensors
4.1.1. Affinity-Based Nanobiosensor
4.1.2. Optical Nanobiosensor
4.1.3. Electrochemical Nanobiosensors
4.1.4. Chiral Nanobiosensors
4.1.5. Nanoimaging System
4.2. PPE (Personal Protective Equipment) Kits
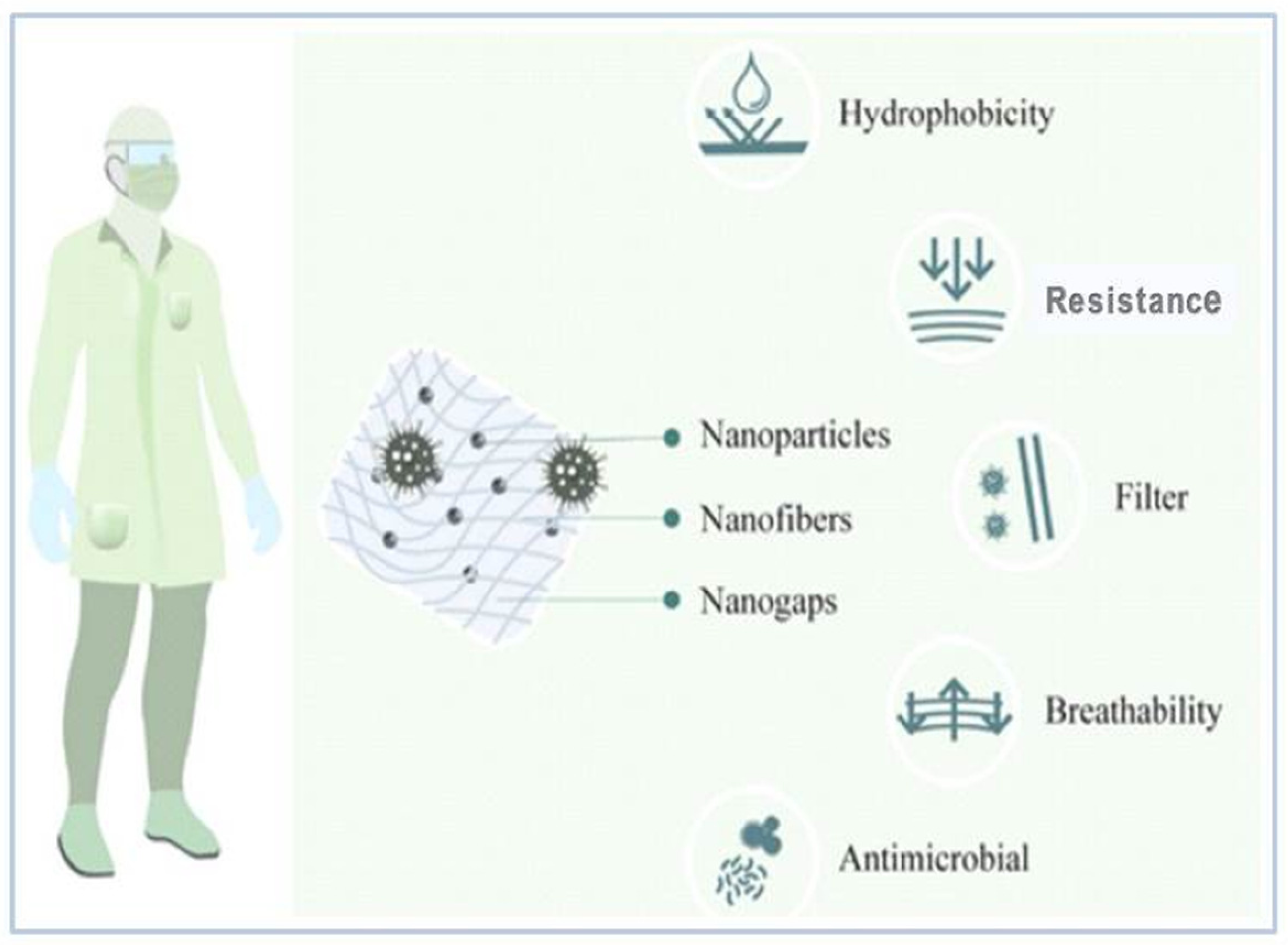
4.3. Nanomasks
4.4. Sanitizers and Disinfectants
4.5. Antiviral Coatings
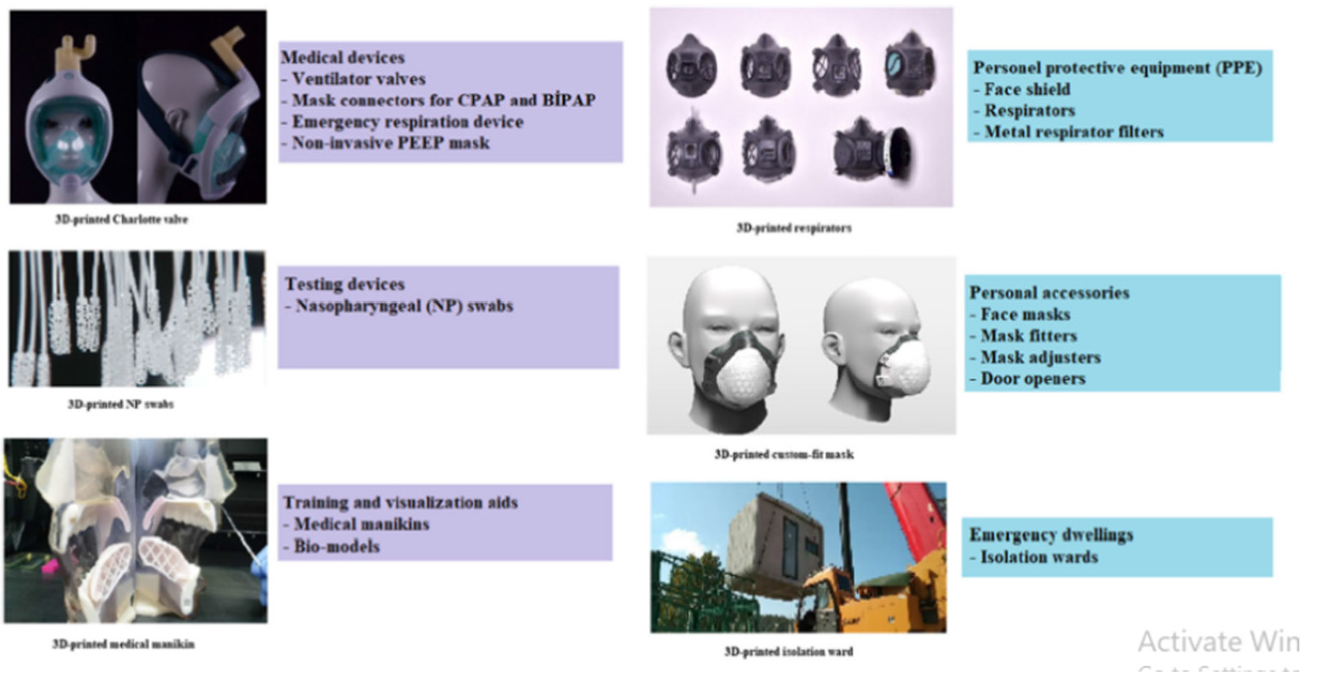
4.6. 3D-Printing
5. Current Advancements on Nanomedicine: Therapeutics and Vaccine Development
Nanotechnological Ways for Vaccine Development
6. Nanotechnology-Based Approaches in Preclinical and Clinical Studies: In Vitro and In Vivo
6.1. Nano-Based Approaches in Pre-Clinical Studies
6.2. Nano-Based Approaches in Clinical Studies
7. Future Perspectives to Tackle COVID-19 Using Nanotechnology
8. Challenges and Limitations of Nanotechnology in COVID-19
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef] [Green Version]
- Cevik, M.; Kuppalli, K.; Kindrachuk, J.; Peiris, M. Virology, transmission, and pathogenesis of SARS-CoV-2. BMJ 2020, 371, m3862. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Zhang, X.; Shu, Y.; Guo, M.; Zhang, H.; Tao, W. Insights from nanotechnology in COVID-19 treatment. Nano Today 2020, 36, 101019. [Google Scholar] [CrossRef] [PubMed]
- Rangayasami, A.; Kannan, K.; Murugesan, S.; Radhika, D.; Sadasivuni, K.K.; Reddy, K.R.; Raghu, A.V. Influence of nanotechnology to combat against COVID-19 for global health emergency: A review. Sens. Int. 2021, 2, 100079. [Google Scholar] [CrossRef]
- Honardoost, M.; Jananic, L.; Aghilia, R.; Emamia, Z.; Khamseh, M.E. The Association between Presence of Comorbidities and COVID-19 Severity: A Systematic Review and Meta-Analysis. Cerebrovasc. Dis. 2021, 50, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Biswas, M.; Rahaman, S.; Biswas, T.K.; Haque, Z.; Ibrahim, B. Association of sex, age, and comorbidities with mortality in COVID-19 patients: A systematic review and meta-analysis. Intervirology 2021, 64, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Nishiga, M.; Wang, D.W.; Han, Y.; Lewis, D.B.; Wu, J.C. COVID-19 and cardiovascular disease: From basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020, 17, 543–558. [Google Scholar] [CrossRef] [PubMed]
- Abu-Farha, M.; Al-Mulla, F.; Thanaraj, T.A.; Kavalakatt, S.; Ali, H.; Ghani, M.A.; Abubaker, J. Impact of diabetes in patients diagnosed with COVID-19. Front. Immunol. 2020, 11, 576818. [Google Scholar] [CrossRef] [PubMed]
- Grivas, P.; Khaki, A.R.; Wise-Draper, T.M.; French, B.; Hennessy, C.; Hsu, C.-Y.; Shyr, Y.; Li, X.; Choueiri, T.K.; Painter, C.A.; et al. Association of clinical factors and recent anticancer therapy with COVID-19 severity among patients with cancer: A report from the COVID-19 and Cancer Consortium. Ann. Oncol 2021, 31, 787–800. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, M.; Garcia, G., Jr.; Tian, E.; Cui, Q.; Chen, X.; Sun, G.; Wang, J.; Arumugaswami, V.; Shi, Y. ApoE-isoform-dependent SARS-CoV-2 neurotropism and cellular response. Cell. Stem. Cell. 2021, 28, 331–342. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Davis, P.B.; Gurney, M.E.; Xu, R. COVID-19 and dementia: Analyses of risk, disparity, and outcomes from electronic health records in the US. Alzheimer’s Dement 2021, alz.12296. [Google Scholar] [CrossRef]
- Yu, I.T.S.; Li, Y.; Wong, T.W.; Tam, W.; Chan, A.T.; Lee, J.H.W.; Leung, D.Y.C.; Tommy Ho, B.S. Evidence of Airborne Transmission of the Severe Acute Respiratory Syndrome Virus. N. Engl. J. Med. 2004, 350, 1731–1739. [Google Scholar] [CrossRef] [Green Version]
- Otter, J.A.; Donskey, C.; Yezli, S.; Douthwaite, S.; Goldenberg, S.D.; Weber, D.J. Transmission of SARS and MERS coronaviruses and influenza virus in healthcare settings: The possible role of dry surface contamination. J. Hosp. Infect 2016, 92, 235–250. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Hu, C.; Haerter, R.; Englert, U. Isosteric molecules in non-isomorphous structures: A new route to new structures. The example of 9,10-dihaloanthracene. CrystEngComm 2004, 6, 83–86. [Google Scholar] [CrossRef]
- Kuba, K.; Imai, Y.; Rao, S.; Gao, H.; Guo, F.; Guan, B.; Huan, Y.; Yang, P.; Zhang, Y.; Deng, W.; et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus–induced lung injury. Nat. Med. 2005, 11, 875–879. [Google Scholar] [CrossRef]
- Jia, H.P.; Look, D.C.; Shi, L.; Hickey, M.; Pewe, L.; Netland, J.; Farzan, M.; Wohlford-Lenane, C.; Perlman, S.; McCray, P.B., Jr. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J. Virol. 2005, 79, 14614–14621. [Google Scholar] [CrossRef] [Green Version]
- Hamming, I.; Timens, W.; Bulthuis, M.L.C.; Lely, A.T.; Navis, G.J.; Van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef]
- Ziegler, C.G.K.; Allon, S.J.; Nyquist, S.K.; Mbano, I.M.; Miao, V.N.; Tzouanas, C.N.; Tzouanas, C.N.; Cao, Y.; Yousif, A.S.; Bals, J.; et al. SARS-CoV-2 receptor ACE2 is an interferon-stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell 2020, 181, 1016–1035. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus–Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Chan, J.F.W.; Yuan, S.; Kok, K.H.; To, K.K.W.; Chu, H.; Yang, J.; Xing, F.; Nurs, J.L.B.; Yip, C.C.-Y.; Poon, R.W.-S.; et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. Lancet 2020, 395, 514–523. [Google Scholar] [CrossRef] [Green Version]
- Ksiazek, T.G.; Erdman, D.; Goldsmith, C.S.; Zaki, S.R.; Peret, T.; Emery, S.; Tong, S.; Urbani, C.; Comer, J.A.; Lim, M.P.H.W.; et al. A novel coronavirus associated with severe acute respiratory syndrome. N. Engl. J. Med. 2003, 348, 1953–1966. [Google Scholar] [CrossRef]
- Guery, B.; Poissy, J.; elMansouf, L.; Séjourné, C.; Ettahar, N.; Lemaire, X.; Vuotto, F.; Goffard, A.; PharmD, S.B.; Enouf, V.; et al. Clinical features and viral diagnosis of two cases of infection with Middle East Respiratory Syndrome coronavirus: A report of nosocomial transmission. Lancet 2013, 381, 2265–2272. [Google Scholar] [CrossRef] [Green Version]
- Totura, A.L.; Baric, R.S. SARS coronavirus pathogenesis: Host innate immune responses and viral antagonism of interferon. Curr. Opin. Virol. 2012, 2, 264–275. [Google Scholar] [CrossRef]
- Hadjadj, J.; Yatim, N.; Barnabei, L.; Corneau, A.; Boussier, J.; Smith, N.; Péré, H.; Charbit, B.; Bondet, V.; Chenevier-Gobeaux, C.; et al. Impaired type I interferon activity and exacerbated inflammatory responses in severe Covid-19 patients. MedRxiv 2020, 369, 718–724. [Google Scholar]
- Blanco-Melo, D.; Nilsson-Payant, B.E.; Liu, W.C.; Uhl, S.; Hoagland, D.; Møller, R.; Jordan, T.X.; Oishi, K.; Panis, M.; Sachs, D.; et al. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell 2020, 181, 1036–1045.e9. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, P.L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Jansen, J.M.; Gerlach, T.; Elbahesh, H.; Rimmelzwaan, G.F.; Saletti, G. Influenza virus-specific CD4+ and CD8+ T cell-mediated immunity induced by infection and vaccination. J. Clin. Virol. 2019, 119, 44–52. [Google Scholar] [CrossRef]
- Behrens, E.M.; Koretzky, G.A. Cytokine storm syndrome: Looking toward the precision medicine era. Arthritis Rheumatol. 2017, 69, 1135–1143. [Google Scholar] [CrossRef] [Green Version]
- Alhalaili, B.; Popescu, I.N.; Kamoun, O.; Alzubi, F.; Alawadhia, S.; Vidu, R. Nanobiosensors for the detection of novel coronavirus 2019-nCoV and other pandemic/Epidemic Respiratory viruses: A review. Sensors 2020, 20, 6591. [Google Scholar] [CrossRef]
- Antiochia, R. Nanobiosensors as new diagnostic tools for SARS, MERS and COVID-19: From past to perspectives. Microchim. Acta 2020, 187, 639. [Google Scholar] [CrossRef]
- Jindal, S.; Gopinath, P. Nanotechnology based approaches for combating COVID-19 viral infection. Nano Express 2020, 1, 022003. [Google Scholar] [CrossRef]
- Martinez-Paredes, G.; Gonzalez-Garcia, M.B.; Costa-Garcia, A. Genosensor for SARS virus detection based on gold nanostructured screen-printed carbon electrodes. Electroanal 2009, 21, 379–385. [Google Scholar] [CrossRef]
- Iravani, S. Nano-and biosensors for the detection of SARS-CoV-2: Challenges and opportunities. Mater. Adv. 2020, 1, 3092. [Google Scholar] [CrossRef]
- Ahmed, S.R.; Nagy, E.; Neethirajan, S. Self-assembled star-shaped chiroplasmonic gold nanoparticles for an ultrasensitive chiroimmunosensor for viruses. RSC Adv. 2017, 7, 40849–40857. [Google Scholar] [CrossRef] [Green Version]
- Shiaelis, N.; Tometzki, A.; Peto, L.; McMahon, A.; Hepp, C.; Bickerton, E.; Favard, C.; Muriaux, D.; Andersson, M.; Oakley, S.; et al. Virus detection and identification in minutes using single-particle imaging and deep learning. MedRxiv 2021. [Google Scholar] [CrossRef]
- Campos, E.V.R.; Pereira, A.E.S.; de Oliveira, J.L.; Carvalho, L.B.; Guilger-Casagrande, M.; de Lima, R.; Fraceto, L.F. How can nanotechnology help to combat COVID-19? Opportunities and urgent need. J. Nanobio. 2020, 18, 125. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Joshi, R.K.; Chughtai, A.A.; Macintyre, C.R. Graphene modified multifunctional personal protective clothing. Adv. Mater. Interfaces 2019, 6, 1900622. [Google Scholar] [CrossRef] [Green Version]
- Chaudhary, V.; Royal, A.; Chavali, M.; Yadav, S.K. Advancements in Research and development to combat COVID-19 using Nanotechnology. Nanotech. Environ. Eng. 2021, 6, 8. [Google Scholar] [CrossRef]
- Talebian, S.; Wallace, G.G.; Schroeder, A.; Stellacci, F.; Conde, J. Nanotechnology-based disinfectants and sensors for SARS-CoV-2. Nat. Nanotechnol. 2020, 15, 615–628. [Google Scholar] [CrossRef]
- Shirvanimoghaddam, K.; Akbari, M.K.; Yadav, R.; Al-Tamimi, A.K.; Naebe, M. Fight against COVID-19: The case of antiviral surfaces. APL Mater. 2021, 9, 031112. [Google Scholar] [CrossRef]
- Basak, S.; Packirisamy, G. Nano-based antiviral coatings to combat viral infections. Nanostruct. Nanoobjects 2020, 24, 100620. [Google Scholar]
- Erkoc, P.; Ulucan-Karnak, F. Nanotechnology-based antimicrobial and antiviral surface coating strategies. Prosthesis 2021, 3, 25–52. [Google Scholar] [CrossRef]
- Aydin, A.; Demirtas, Z.; Ok, M.; Erkus, H.; Cebi, G.; Uysal, E.; Gunduz, O.; Ustundag, C.B. 3D printing in the battle against COVID-19. Emerg. Mater. 2021, 4, 363–386. [Google Scholar] [CrossRef]
- Oladapo, B.I.; Ismail, S.O.; Afolalu, T.D.; Olawade, D.B.; Zahedi, M. Review on 3D printing: Fight against COVID-19. Mat. Chem. Phys. 2021, 258, 123943. [Google Scholar] [CrossRef]
- Jain, K.; Shukla, R.; Yadav, A.; Ujjwal, R.R.; Flora, S.J.S. 3D printing in development of Nanomedicine. Nanomater 2021, 11, 420. [Google Scholar] [CrossRef]
- McNamara, K.; Tofail, S.A. Nanosystems: The use of nanoalloys, metallic, bimetallic, and magnetic nanoparticles in biomedical applications. Phys. Chem. Chem. Phys. 2015, 17, 27981–27995. [Google Scholar] [CrossRef]
- Saadeh, Y.; Vyas, D. Nanorobotic applications in medicine: Current proposals and designs. Am. J. Robot. Surg. 2014, 1, 4–11. [Google Scholar] [CrossRef]
- Oliveira, O.N., Jr.; Iost, R.M.; Siqueira, J.R., Jr.; Crespilho, F.N.; Caseli, L. Nanomaterials for diagnosis: Challenges and applications in smart devices based on molecular recognition. ACS Appl. Mater. Interfaces 2014, 6, 14745–14766. [Google Scholar] [CrossRef]
- De Jong, W.H.; Borm, P.J. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133. [Google Scholar] [CrossRef] [Green Version]
- Holzinger, M.; Le Goff, A.; Cosnier, S. Nanomaterials for biosensing applications: A review. Front. Chem. 2014, 2, 63. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Yang, X.L.; Ho, W.S. Preparation of uniform-sized multiple emulsions and micro/nano particulates for drug delivery by membrane emulsification. J. Pharm. Sci. 2011, 100, 75–93. [Google Scholar] [CrossRef]
- Onoue, S.; Yamada, S.; Chan, H.K. Nanodrugs: Pharmacokinetics and safety. Int. J. Nanomed. 2014, 20, 1025–1037. [Google Scholar] [CrossRef] [Green Version]
- Dawidczyk, C.M.; Kim, C.; Park, J.H.; Russell, L.M.; Lee, K.H.; Pomper, M.G.; Searson, P.C. State-of-the-art in design rules for drug delivery platforms: Lessons learned from FDA-approved nanomedicines. J. Control. Release 2014, 10, 133–144. [Google Scholar] [CrossRef] [Green Version]
- Charlene, M.D.; Luisa, M.R.; Peter, C.S. Nanomedicines for cancer therapy: State-of-the-art and limitations to pre-clinical studies that hinder future developments. Front. Chem. 2014, 2, 69. [Google Scholar]
- Kumari, A.; Yadav, S.K. Cellular interactions of therapeutically delivered nanoparticles. Exp. Opinion Drug. Deliv. 2011, 8, 141–151. [Google Scholar] [CrossRef]
- Shi, X.; Sun, K.; Baker, J.R., Jr. Spontaneous formation of functionalized dendrimer-stabilized gold nanoparticles. J. Phys. Chem. C. 2008, 112, 8251–8258. [Google Scholar] [CrossRef] [Green Version]
- Park, S.H.; Oh, S.G.; Mun, J.Y.; Han, S.S. Loading of gold nanoparticles inside the DPPC bilayers of liposome and their effects on membrane fluidities. Coll. Surf. B 2006, 48, 112–118. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [Green Version]
- Behera, S.; Rana, G.; Satapathy, S.; Mohanty, M.; Pradhan, S.; Panda, M.K.; Ningthoujam, R.; Hazarika, B.N.; Singh, Y.D. Biosensors in diagnosing COVID-19 and recent development. Sens. Internat. 2020, 1, 100054. [Google Scholar] [CrossRef]
- Bonifácio, B.V.; da Silva, P.B.; dos Santos Ramos, M.A.; Negri, K.M.S.; Bauab, T.M.; Chorilli, M. Nanotechnology-based drug delivery systems and herbal medicines: A review. Int. J. Nanomed. 2014, 9, 1–15. [Google Scholar]
- Chakravarty, M.; Vora, A. Drug Delivery and Translational Research. Drug. Deliv. Transl. Res. 2014, 1–10. [Google Scholar]
- Reddy, S.T.; Van der Vlies, A.J.; Simeoni, E.; Angeli, V.; Randolph, G.J.; O’Neil, C.P.; Lee, L.K.; Swartz, M.A.; Hubbell, J.A. Exploiting lymphatic transport and complement activation in nanoparticle vaccines. Nat. Biotechnol. 2007, 25, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.T.; Berk, D.A.; Jain, R.K.; Swartz, M.A. A sensitive in vivo model for quantifying interstitial convective transport of injected macromolecules and nanoparticles. J. Appl. Physiol. 2006, 101, 1162–1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bachmann, M.F.; Jennings, G.T. Vaccine delivery: A matter of size, geometry, kinetics and molecular patterns. Nat. Rev. Immunol. 2010, 10, 787–796. [Google Scholar] [CrossRef]
- Ma, C.Q.; Wang, L.L.; Tao, X.R.; Zhang, N.; Yang, Y.; Tseng, C.-T.K.; Li, F.; Zhou, Y.; Jiang, S.; Dua, L. Searching for an ideal vaccine candidate among different MERS coronavirus receptor-binding fragments-The importance of immune focusing in subunit vaccine design. Vaccine 2014, 32, 6170–6176. [Google Scholar] [CrossRef]
- Wang, L.S.; Shi, W.; Joyce, M.G.; Modjarrad, K.; Zhang, Y.; Leung, K.; Lees, C.R.; Zhou, T.; Yassine, H.M.; Kanekiyo, M.; et al. Evaluation of candidate vaccine approaches for MERS-CoV. Nat. Commun. 2015, 6, 7712. [Google Scholar] [CrossRef]
- Milane, L.; Amiji, M. Clinical approval of nanotechnology-based SARS-CoV-2 mRNA vaccines: Impact on translational nanomedicine. Drug. Deliv. Transl. Res. 2021, 29, 1–7. [Google Scholar]
- Dhama, K.; Sharun, K.; Tiwari, R.; Dadar, M.; Malik, Y.S.; Singh, K.P.; Chaicumpa, W. COVID-19, an emerging coronavirus infection: Advances and prospects in designing and developing vaccines, immunotherapeutics, and therapeutics. Hum. Vaccines Immunother. 2020, 16, 1232–1238. [Google Scholar] [CrossRef] [Green Version]
- Marston, H.D.; Paules, C.I.; Fauci, A.S. Monoclonal Antibodies for Emerging Infectious Diseases-Borrowing from History. N. Engl. J. Med. 2018, 378, 1469–1472. [Google Scholar] [CrossRef]
- Rab, S.; Afjal, A.; Javaid, M.; Haleem, A.; Vaishya, R. An update on the global vaccine development for coronavirus. Diabetes Metab. Syndr. 2020, 14, 2053–2055. [Google Scholar] [CrossRef]
- Chen, Y.N.; Hsueh, Y.H.; Hsieh, C.T.; Tzou, D.Y.; Chang, P.L. Antiviral Activity of Graphene–Silver Nanocomposites against Non-Enveloped and Enveloped Viruses. Int. J. Environ. Res. Public Health 2016, 13, 430. [Google Scholar] [CrossRef] [Green Version]
- Florindo, H.F.; Kleiner, R.; Vaskovich-Koubi, D.; Acúrcio, R.C.; Carreira, B.; Yeini, E.; Tiram, G.; Liubomirski, Y.; Satchi-Fainaro, R. Immune-mediated approaches against COVID-19. Nat. Nanotechnol. 2020, 15, 630–645. [Google Scholar] [CrossRef]
- Shah, V.K.; Firmal, P.; Alam, A.; Ganguly, D.; Chattopadhyay, S. Overview of Immune Response During SARS-CoV-2 Infection: Lessons From the Past. Front. Immunol. 2020, 11, 1949. [Google Scholar] [CrossRef]
- Fleischmann, R.; Genovese, M.C.; Lin, Y.; St John, G.; Van der Heijde, D.; Wang, S.; Gomez-Reino, J.J.; Maldonado-Cocco, J.A.; Stanislav, M.; Kivitz, A.J.; et al. Long-term safety of sarilumab in rheumatoid arthritis: An integrated analysis with up to 7 years’ follow-up. Rheumatology 2020, 59, 292–302. [Google Scholar] [CrossRef] [Green Version]
- Chan, W.C.W. Nano research for COVID-19. ACS Nano. 2020, 14, 3719–3720. [Google Scholar] [CrossRef] [Green Version]
- Polyak, B.; Cordovez, B. How can we predict behavior of nanoparticles in vivo? Nanomedicine 2016, 11, 189–192. [Google Scholar] [CrossRef] [Green Version]
- Beyth, N.; Houri-Haddad, Y.; Domb, A.; Khan, W.; Hazan, R. Alternative antimicrobial approach: Nano-antimicrobial materials. Evid. Based Complement. Altern. Med. 2015, 1, 246012. [Google Scholar] [CrossRef] [Green Version]
- Ventola, C.L. Progress in nanomedicine: Approved and investigational nanodrugs. Pharm. Ther. 2017, 42, 742–755. [Google Scholar]
- Caster, J.M.; Patel, A.N.; Zhang, T.; Wang, A. Investigational nanomedicines in 2016: A review of nanotherapeutics currently undergoing clinical trials. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1416. [Google Scholar] [CrossRef]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on Their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 44. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rai, M.; Bonde, S.; Yadav, A.; Bhowmik, A.; Rathod, S.; Ingle, P.; Gade, A. Nanotechnology as a Shield against COVID-19: Current Advancement and Limitations. Viruses 2021, 13, 1224. https://doi.org/10.3390/v13071224
Rai M, Bonde S, Yadav A, Bhowmik A, Rathod S, Ingle P, Gade A. Nanotechnology as a Shield against COVID-19: Current Advancement and Limitations. Viruses. 2021; 13(7):1224. https://doi.org/10.3390/v13071224
Chicago/Turabian StyleRai, Mahendra, Shital Bonde, Alka Yadav, Arpita Bhowmik, Sanjay Rathod, Pramod Ingle, and Aniket Gade. 2021. "Nanotechnology as a Shield against COVID-19: Current Advancement and Limitations" Viruses 13, no. 7: 1224. https://doi.org/10.3390/v13071224
APA StyleRai, M., Bonde, S., Yadav, A., Bhowmik, A., Rathod, S., Ingle, P., & Gade, A. (2021). Nanotechnology as a Shield against COVID-19: Current Advancement and Limitations. Viruses, 13(7), 1224. https://doi.org/10.3390/v13071224







