Understanding Post Entry Sorting of Adenovirus Capsids; A Chance to Change Vaccine Vector Properties
Abstract
1. Introduction
1.1. Adenoviruses Constitute a Diverse Family of Infectious Pathogens
1.2. Adenovirus Capsids Are Metastable Structures
1.3. Adenoviruses Follow a Lytic Life Cycle
2. Immune Detection of Adenovirus
2.1. Adenoviruses Trigger Cell Intrinsic Immunity
2.2. Adenoviruses Provoke an Aadaptive Immune Response and Subvert Antiviral Autophagy upon Cell Entry
3. Adenoviruses as (Vaccine) Vectors
3.1. Adenovirus Vector Development, a Generational Approach
3.2. Adenovirus as a Vaccine Vector
4. Modulating Adenovirus (Vaccine) Vector Efficacy, the Capsid Leads the Way
4.1. Stability Lies in the Species
4.2. Fine-Tuning the Capsid Structure, the Example of Protein VI in Species C Ad2/5
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ghosh, S.S.; Gopinath, P.; Ramesh, A. Adenoviral vectors: A promising tool for gene therapy. Appl. Biochem. Biotechnol. 2006, 133, 9–29. [Google Scholar] [CrossRef]
- Adams, W.; Loré, K. Recombinant adenovirus vector infection of human dendritic cells. Viral Gene Ther. 2011. [Google Scholar] [CrossRef]
- Mercier, S.; Gahéry-Segard, H.; Monteil, M.; Lengagne, R.; Guillet, J.-G.; Eloit, M.; Denesvre, C. Distinct roles of adenovirus vector-transduced dendritic cells, myoblasts, and endothelial cells in mediating an immune response against a transgene product. J. Virol. 2002, 76, 2899–2911. [Google Scholar] [CrossRef]
- Rowe, W.P.; Huebner, R.J.; Gilmore, L.K.; Parrott, R.H.; Ward, T.G. Isolation of a cytopathogenic agent from human adenoids undergoing spontaneous degeneration in tissue culture. Proc. Soc. Exp. Biol. Med. 1953, 84, 570–573. [Google Scholar] [CrossRef]
- Enders, J.F.; Bell, J.A.; Dingle, J.H.; Francis, T.; Hilleman, M.R.; Huebner, R.J.; Payne, A.M.M. “Adenoviruses”: Group name proposed for new respiratory-tract viruses. Science 1956, 124, 119–120. [Google Scholar] [CrossRef]
- Lefkowitz, E.J.; Dempsey, D.M.; Hendrickson, R.C.; Orton, R.J.; Siddell, S.G.; Smith, D.B. Virus taxonomy: The database of the International Committee on Taxonomy of Viruses (ICTV). Nucleic Acids Res. 2018, 46, D708–D717. [Google Scholar] [CrossRef]
- Harrach, B.; Tarján, Z.L.; Benkő, M. Adenoviruses across the animal kingdom: A walk in the zoo. FEBS Lett. 2019, 593, 3660–3673. [Google Scholar] [CrossRef] [PubMed]
- Hage, E.; Liebert, U.G.; Bergs, S.; Ganzenmueller, T.; Heim, A. Human mastadenovirus type 70: A novel, multiple recombinant species D mastadenovirus isolated from diarrhoeal faeces of a haematopoietic stem cell transplantation recipient. J. Gen. Virol. 2015, 96, 2734–2742. [Google Scholar] [CrossRef]
- Sarantis, H.; Johnson, G.; Brown, M.; Petric, M.; Tellier, R. Comprehensive detection and serotyping of human adenoviruses by PCR and sequencing. J. Clin. Microbiol. 2004, 42, 3963–3969. [Google Scholar] [CrossRef] [PubMed]
- Crenshaw, B.J.; Jones, L.B.; Bell, C.R.; Kumar, S.; Matthews, Q.L. Perspective on adenoviruses: Epidemiology, pathogenicity, and gene therapy. Biomedicines 2019, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Ghebremedhin, B. Human adenovirus: Viral pathogen with increasing importance. Eur. J. Microbiol. Immunol. 2014, 4, 26–33. [Google Scholar] [CrossRef]
- Lion, T. Adenovirus infections in immunocompetent and immunocompromised patients. Clin. Microbiol. Rev. 2014, 27, 441–462. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, S.; Seto, J.; Liu, E.B.; Walsh, M.P.; Dyer, D.W.; Chodosh, J.; Seto, D. Computational analysis of four human adenovirus type 4 genomes reveals molecular evolution through two interspecies recombination events. Virology 2013, 443, 197–207. [Google Scholar] [CrossRef]
- Dehghan, S.; Seto, J.; Liu, E.B.; Ismail, A.M.; Madupu, R.; Heim, A.; Jones, M.S.; Dyer, D.W.; Chodosh, J.; Seto, D. A zoonotic adenoviral human pathogen emerged through genomic recombination among human and nonhuman simian hosts. J. Virol. 2019, 93. [Google Scholar] [CrossRef] [PubMed]
- Medkour, H.; Amona, I.; Akiana, J.; Davoust, B.; Bitam, I.; Levasseur, A.; Tall, M.L.; Diatta, G.; Sokhna, C.; Hernandez-Aguilar, R.A.; et al. Adenovirus infections in African humans and wild non-human primates: Great diversity and cross-species transmission. Viruses 2020, 12, 657. [Google Scholar] [CrossRef]
- Hodgson, S.H.; Ewer, K.J.; Bliss, C.M.; Edwards, N.J.; Rampling, T.; Anagnostou, N.A.; de Barra, E.; Havelock, T.; Bowyer, G.; Poulton, I.D.; et al. Evaluation of the efficacy of ChAd63-MVA vectored vaccines expressing circumsporozoite protein and ME-TRAP against controlled human malaria infection in malaria-naive individuals. J. Infect. Dis. 2015, 211, 1076–1086. [Google Scholar] [CrossRef]
- Tapia, M.D.; Sow, S.O.; Lyke, K.E.; Haidara, F.C.; Diallo, F.; Doumbia, M.; Traore, A.; Coulibaly, F.; Kodio, M.; Onwuchekwa, U.; et al. Use of ChAd3-EBO-Z Ebola virus vaccine in Malian and US adults, and boosting of Malian adults with MVA-BN-Filo: A phase 1, single-blind, randomised trial, a phase 1b, open-label and double-blind, dose-escalation trial, and a nested, randomised, double-bli. Lancet Infect. Dis. 2016, 16, 31–42. [Google Scholar] [CrossRef]
- Guo, J.; Mondal, M.; Zhou, D. Development of novel vaccine vectors: Chimpanzee adenoviral vectors. Hum. Vaccines Immunother. 2018, 14, 1679–1685. [Google Scholar] [CrossRef]
- Hollingdale, M.R.; Sedegah, M.; Limbach, K. Development of replication-deficient adenovirus malaria vaccines. Expert Rev. Vaccines 2017, 16, 261–271. [Google Scholar] [CrossRef]
- Kümin, D.; Hofmann, C.; Rudolph, M.; Both, G.W.; Löser, P. Biology of ovine adenovirus infection of nonpermissive cells. J. Virol. 2002, 76, 10882–10893. [Google Scholar] [CrossRef][Green Version]
- Rasmussen, U.B.; Benchaibi, M.; Meyer, V.; Schlesinger, Y.; Schughart, K. Novel human gene transfer vectors: Evaluation of wild-type and recombinant animal adenoviruses in human-derived cells. Hum. Gene Ther. 1999, 10, 2587–2599. [Google Scholar] [CrossRef]
- De Vleeschauwer, A.R.; Zhou, X.; Lefebvre, D.J.; Garnier, A.; Watier, F.; Pignon, C.; Lacour, S.A.; Zientara, S.; Bakkali-Kassimi, L.; de Clercq, K.; et al. A canine adenovirus type 2 vaccine vector confers protection against foot-and-mouth disease in guinea pigs. Vaccine 2018, 36, 2193–2198. [Google Scholar] [CrossRef]
- Chen, E.C.; Yagi, S.; Kelly, K.R.; Mendoza, S.P.; Maninger, N.; Rosenthal, A.; Spinner, A.; Bales, K.L.; Schnurr, D.P.; Lerche, N.W.; et al. Cross-species transmission of a novel adenovirus associated with a fulminant pneumonia outbreak in a new world monkey colony. PLoS Pathog. 2011, 7, e1002155. [Google Scholar] [CrossRef]
- Pérez-Illana, M.; Martínez, M.; Condezo, G.N.; Hernando-Pérez, M.; Mangroo, C.; Brown, M.; Marabini, R.; Martín, C.S. Cryo-EM structure of enteric adenovirus HAdV-F41 highlights structural variations among human adenoviruses. Sci. Adv. 2021, 7, 1–15. [Google Scholar] [CrossRef]
- Berciaud, S.; Rayne, F.; Kassab, S.; Jubert, C.; Faure-Della Corte, M.; Salin, F.; Wodrich, H.; Lafon, M.E.; Barat, P.; Fayon, M.; et al. Adenovirus infections in Bordeaux University Hospital 2008–2010: Clinical and virological features. J. Clin. Virol. 2012, 54, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Khanal, S.; Ghimire, P.; Dhamoon, A.S. The repertoire of adenovirus in human disease: The innocuous to the deadly. Biomedicines 2018, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P.; Kajon, A.E. Adenovirus: Epidemiology, global spread of novel serotypes, and advances in treatment and prevention. Semin. Respir. Crit. Care Med. 2016, 37, 586–602. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.M.; Seto, D.; Jones, M.S.; Dyer, D.W.; Chodosh, J. Molecular evolution of human species D adenoviruses. Infect. Genet. Evol. 2011, 11, 1208–1217. [Google Scholar] [CrossRef] [PubMed]
- Greber, U.F. Disassembly: The adenovirus. Rev. Med. Virol. 1998, 222, 213–222. [Google Scholar] [CrossRef]
- Chroboczek, J.; Bieber, F.; Jacrot, B. The sequence of the genome of adenovirus type 5 and its comparison with the genome of adenovirus type 2. Virology 1992, 186, 280–285. [Google Scholar] [CrossRef]
- El Bakkouri, M.; Seiradake, E.; Cusack, S.; Ruigrok, R.W.H.; Schoehn, G. Structure of the C-terminal head domain of the fowl adenovirus type 1 short fibre. Virology 2008, 378, 169–176. [Google Scholar] [CrossRef]
- Nemerow, G.R.; Stewart, P.L.; Reddy, V.S. Structure of human adenovirus. Curr. Opin. Virol. 2012, 2, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Stewart, P.L.; Burnett, R.M.; Cyrklaff, M.; Fuller, S.D. Image reconstruction reveals the complex molecular organization of adenovirus. Cell 1991, 67, 145–154. [Google Scholar] [CrossRef]
- Fabry, C.M.S.; Rosa-Calatrava, M.; Conway, J.F.; Zubieta, C.; Cusack, S.; Ruigrok, R.W.H.; Schoehn, G. A quasi-atomic model of human adenovirus type 5 capsid. EMBO J. 2005, 24, 1645–1654. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Jin, L.; Koh, S.B.S.; Atanasov, I.; Schein, S.; Wu, L.; Zhou, Z.H. Atomic structure of human adenovirus by Cryo-EM reveals interactions among protein networks. Science 2010, 329, 1038–1043. [Google Scholar] [CrossRef] [PubMed]
- Vassal-Stermann, E.; Effantin, G.; Zubieta, C.; Burmeister, W.; Iseni, F.; Wang, H.; Lieber, A.; Schoehn, G.; Fender, P. CryoEM structure of adenovirus type 3 fibre with desmoglein 2 shows an unusual mode of receptor engagement. Nat. Commun. 2019, 10, 1–7. [Google Scholar] [CrossRef]
- Martín, C.S. Latest insights on adenovirus structure and assembly. Viruses 2012, 4, 847–877. [Google Scholar] [CrossRef]
- Reddy, V.S.; Barry, M.A. Structural organization and protein-protein interactions in human adenovirus capsid. In Subcellular Biochemistry; Springer Science and Business Media: Berlin, Germany, 2021; Volume 96, pp. 503–518. [Google Scholar]
- Cao, C.; Dong, X.; Wu, X.; Wen, B.; Ji, G.; Cheng, L.; Liu, H. Conserved fiber-penton base interaction revealed by nearly atomic resolution cryo-electron microscopy of the structure of adenovirus provides insight into receptor interaction. J. Virol. 2012, 86, 12322–12329. [Google Scholar] [CrossRef]
- Zubieta, C.; Schoehn, G.; Chroboczek, J.; Cusack, S. The structure of the human adenovirus 2 penton. Mol. Cell 2005, 17, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Furcinitti, P.S.; van Oostrum, J.; Burnett, R.M. Adenovirus polypeptide IX revealed as capsid cement by difference images from electron microscopy and crystallography. EMBO J. 1989, 8, 3563–3570. [Google Scholar] [CrossRef] [PubMed]
- Wodrich, H.; Guan, T.; Cingolani, G.; von Seggern, D.; Nemerow, G.; Gerace, L. Switch from capsid protein import to adenovirus assembly by cleavage of nuclear transport signals. EMBO J. 2003, 22, 6245–6255. [Google Scholar] [CrossRef]
- San Martín, C. Transmission electron microscopy and the molecular structure of icosahedral viruses. Arch. Biochem. Biophys. 2015, 581, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Wu, L.; Sun, R.; Zhou, Z.H. Atomic structures of minor proteins VI and VII in human adenovirus. J. Virol. 2017, 91. [Google Scholar] [CrossRef]
- Russell, W.C. Adenoviruses: Update on structure and function. J. Gen. Virol. 2009, 90, 1–20. [Google Scholar] [CrossRef]
- Chatterjee, P.K.; Vayda, M.E.; Flint, S.J. Interactions among the three adenovirus core proteins. J. Virol. 1985, 55, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vargas, J.; Vaughan, R.C.; Houser, C.; Hastie, K.M.; Kao, C.C.; Nemerow, G.R. Isolation and characterization of the DNA and protein binding activities of adenovirus core protein V. J. Virol. 2014, 88, 9287–9296. [Google Scholar] [CrossRef]
- Bauer, M.; Gomez-Gonzalez, A.; Suomalainen, M.; Hemmi, S.; Greber, U.F. The E3 ubiquitin ligase Mind bomb 1 enhances nuclear import of viral DNA by inactivating a virion linchpin protein that suppresses exposure of virion pathogen-associated molecular patterns. bioRxiv 2020. [Google Scholar] [CrossRef]
- Wang, I.H.; Suomalainen, M.; Andriasyan, V.; Kilcher, S.; Mercer, J.; Neef, A.; Luedtke, N.W.; Greber, U.F. Tracking viral genomes in host cells at single-molecule resolution. Cell Host Microbe 2013, 14, 468–480. [Google Scholar] [CrossRef]
- Komatsu, T.; Dacheux, D.; Kreppel, F.; Nagata, K.; Wodrich, H. A method for visualization of incoming adenovirus chromatin complexes in fixed and living cells. PLoS ONE 2015, 10, e0137102. [Google Scholar] [CrossRef]
- Greber, U.F.; Suomalainen, M.; Stidwill, R.P.; Boucke, K.; Ebersold, M.W.; Helenius, A. The role of the nuclear pore complex in adenovirus DNA entry. EMBO J. 1997, 16, 5998–6007. [Google Scholar] [CrossRef]
- Karen, K.A.; Hearing, P. Adenovirus core protein VII protects the viral genome from a DNA damage response at early times after infection. J. Virol. 2011, 85, 4135–4142. [Google Scholar] [CrossRef] [PubMed]
- Avgousti, D.C.; Herrmann, C.; Kulej, K.; Pancholi, N.J.; Sekulic, N.; Petrescu, J.; Molden, R.C.; Blumenthal, D.; Paris, A.J.; Reyes, E.D.; et al. A core viral protein binds host nucleosomes to sequester immune danger signals. Nature 2016, 535, 173–177. [Google Scholar] [CrossRef]
- Avgousti, D.C.; della Fera, A.N.; Otter, C.J.; Herrmann, C.; Pancholi, N.J.; Weitzman, M.D. Adenovirus core protein VII downregulates the DNA damage response on the host genome. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Parks, R.J. Adenovirus protein IX: A new look at an old protein. Mol. Ther. 2005, 11, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Gorman, J.J.; Wallis, T.P.; Whelan, D.A.; Shaw, J.; Both, G.W. LH3, a “homologue” of the mastadenoviral E1B 55-kDa protein is a structural protein of atadenoviruses. Virology 2005, 342, 159–166. [Google Scholar] [CrossRef][Green Version]
- Colby, W.W.; Shenk, T. Adenovirus type 5 virions can be assembled in vivo in the absence of detectable polypeptide IX. J. Virol. 1981, 39, 977–980. [Google Scholar] [CrossRef]
- Ostapchuk, P.; Suomalainen, M.; Zheng, Y.; Boucke, K.; Greber, U.F.; Hearing, P. The adenovirus major core protein VII is dispensable for virion assembly but is essential for lytic infection. PLoS Pathog. 2017, 13, e1006455. [Google Scholar] [CrossRef]
- Cusack, S. Adenovirus complex structures. Curr. Opin. Struct. Biol. 2005, 15, 237–243. [Google Scholar] [CrossRef]
- Berk, A.J.; Lee, F.; Harrison, T.; Williams, J.; Sharp, P.A. Pre-early adenovirus 5 gene product regulates synthesis of early viral messenger RNAs. Cell 1979, 17, 935–944. [Google Scholar] [CrossRef]
- Zhang, W.; Imperiale, M.J. Requirement of the adenovirus IVa2 Protein for virus assembly. J. Virol. 2003, 77, 3586–3594. [Google Scholar] [CrossRef]
- Ahi, Y.S.; Mittal, S.K. Components of adenovirus genome packaging. Front. Microbiol. 2016, 7, 1–15. [Google Scholar] [CrossRef]
- Lehmberg, E.; Traina, J.A.; Chakel, J.A.; Chang, R.J.; Parkman, M.; McCaman, M.T.; Murakami, P.K.; Lahidji, V.; Nelson, J.W.; Hancock, W.S.; et al. Reversed-phase high-performance liquid chromatographic assay for the adenovirus type 5 proteome. J. Chromatogr. B Biomed. Sci. Appl. 1999, 732, 411–423. [Google Scholar] [CrossRef]
- Stewart, P.L.; Fuller, S.D.; Burnett, R.M. Difference imaging of adenovirus: Bridging the resolution gap between X-ray crystallography and electron microscopy. EMBO J. 1993, 12, 2589–2599. [Google Scholar] [CrossRef] [PubMed]
- Rancourt, C.; Tihanyi, K.; Bourbonniere, M.; Weber, J.M. Identification of active-site residues of the adenovirus endopeptidase. Proc. Natl. Acad. Sci. USA 1994, 91, 844–847. [Google Scholar] [CrossRef] [PubMed]
- Mangel, W.F.; Martín, C.S. Structure, function and dynamics in adenovirus maturation. Viruses 2014, 6, 4536–4570. [Google Scholar] [CrossRef]
- Perez-Berna, A.J.; Mangel, W.F.; McGrath, W.J.; Graziano, V.; Flint, J.; San Martin, C. Processing of the L1 52/55k protein by the adenovirus protease: A new substrate and new insights into virion maturation. J. Virol. 2014, 88, 1513–1524. [Google Scholar] [CrossRef] [PubMed]
- Cotten, M.; Weber, J.M. The adenovirus protease is required for virus entry into host cells. Virology 1995, 213, 494–502. [Google Scholar] [CrossRef]
- Pérez-Berná, A.J.; Marabini, R.; Scheres, S.H.W.; Menéndez-Conejero, R.; Dmitriev, I.P.; Curiel, D.T.; Mangel, W.F.; Flint, S.J.; Martín, C.S. Structure and uncoating of immature adenovirus. J. Mol. Biol. 2009, 392, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Wu, E.; Pache, L.; von Seggern, D.J.; Mullen, T.-M.; Mikyas, Y.; Stewart, P.L.; Nemerow, G.R. Flexibility of the adenovirus fiber is required for efficient receptor interaction. J. Virol. 2003, 77, 7225–7235. [Google Scholar] [CrossRef]
- Seki, T.; Dmitriev, I.; Kashentseva, E.; Takayama, K.; Rots, M.; Suzuki, K.; Curiel, D.T. Artificial extension of the adenovirus fiber shaft inhibits infectivity in coxsackievirus and adenovirus receptor-positive cell lines. J. Virol. 2002, 76, 1100–1108. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shayakhmetov, D.M.; Lieber, A. Dependence of adenovirus infectivity on length of the fiber shaft domain. J. Virol. 2000, 74, 10274–10286. [Google Scholar] [CrossRef]
- Fender, P.; Ruigrok, R.W.H.; Gout, E.; Buffet, S.; Chroboczek, J. Adenovirus dodecahedron, a new vector for human gene transfer. Nat. Biotechnol. 1997, 15, 52–56. [Google Scholar] [CrossRef]
- Fender, P.; Schoehn, G.; Foucaud-Gamen, J.; Gout, E.; Garcel, A.; Drouet, E.; Chroboczek, J. Adenovirus dodecahedron allows large multimeric protein transduction in human cells. J. Virol. 2003, 77, 4960–4964. [Google Scholar] [CrossRef]
- Pied, N.; Wodrich, H. Imaging the adenovirus infection cycle. FEBS Lett. 2019, 593, 3419–3448. [Google Scholar] [CrossRef]
- Nemerow, G.R.; Pache, L.; Reddy, V.; Stewart, P.L. Insights into adenovirus host cell interactions from structural studies. Virology 2009, 384, 380–388. [Google Scholar] [CrossRef]
- Kremer, E.J.; Nemerow, G.R. Adenovirus tales: From the cell surface to the nuclear pore complex. PLoS Pathog. 2015, 11, e1004821. [Google Scholar] [CrossRef] [PubMed]
- Philipson, L.; Lonberg-holm, K.; Pettersson, U.L.F. Virus-receptor interaction in an adenovirus system. J. Virol. 1968, 2, 1064–1075. [Google Scholar] [CrossRef]
- Bergelson, J.M.; Cunningham, J.A.; Droguett, G.; Kurt-Jones, E.A.; Krithivas, A.; Hong, J.S.; Horwitz, M.S.; Crowell, R.L.; Finberg, R.W. Isolation of a common receptor for coxsackie B viruses and adenoviruses 2 and 5. Science 1997, 275, 1320–1323. [Google Scholar] [CrossRef] [PubMed]
- Roelvink, P.W.; Lizonova, A.; Lee, J.G.M.; Li, Y.; Bergelson, J.M.; Finberg, R.W.; Brough, D.E.; Kovesdi, I.; Wickham, T.J. The coxsackievirus-adenovirus receptor protein can function as a cellular attachment protein for adenovirus serotypes from subgroups A, C, D, E, and F. J. Virol. 1998, 72, 7909–7915. [Google Scholar] [CrossRef] [PubMed]
- Mese, K.; Bunz, O.; Schellhorn, S.; Volkwein, W.; Jung, D.; Gao, J.; Zhang, W.; Baiker, A.; Ehrhardt, A. Identification of novel human adenovirus candidates using the coxsackievirus and adenovirus receptor for cell entry. Virol. J. 2020, 17, 1–7. [Google Scholar] [CrossRef]
- Gaggar, A.; Shayakhmetov, D.M.; Lieber, A. CD46 is a cellular receptor for group B adenoviruses. Nat. Med. 2003, 9, 1408–1412. [Google Scholar] [CrossRef]
- Wang, H.; Li, Z.Y.; Liu, Y.; Persson, J.; Beyer, I.; Möller, T.; Koyuncu, D.; Drescher, M.R.; Strauss, R.; Zhang, X.B.; et al. Desmoglein 2 is a receptor for adenovirus serotypes 3, 7, 11 and 14. Nat. Med. 2011, 17, 96–104. [Google Scholar] [CrossRef]
- Arnberg, N.; Pring-Åkerblom, P.; Wadell, G. Adenovirus type 37 uses sialic acid as a cellular receptor on chang C cells. J. Virol. 2002, 76, 8834–8841. [Google Scholar] [CrossRef]
- Arnberg, N.; Kidd, A.H.; Edlund, K.; Nilsson, J.; Pring-Åkerblom, P.; Wadell, G. Adenovirus type 37 binds to cell surface sialic acid through a charge-dependent interaction. Virology 2002, 302, 33–43. [Google Scholar] [CrossRef]
- Arnberg, N.; Kidd, A.H.; Edlund, K.; Olfat, F.; Wadell, G. Initial interactions of subgenus D adenoviruses with A549 cellular receptors: Sialic acid versus αVintegrins. J. Virol. 2000, 74, 7691–7693. [Google Scholar] [CrossRef]
- Wu, H.; Curiel, D.T. Fiber-modified adenoviruses for targeted gene therapy. Methods Mol. Biol. 2008, 434, 113–132. [Google Scholar] [CrossRef]
- Noureddini, S.C.; Curiel, D.T. Genetic targeting strategies for adenovirus. Mol. Pharm. 2005, 2, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, M.; Both, G.W.; Banizs, B.; Tsuruta, Y.; Yamamoto, S.; Kawakami, Y.; Douglas, J.T.; Tani, K.; Curiel, D.T.; Glasgow, J.N. An adenovirus serotype 5 vector with fibers derived from ovine atadenovirus demonstrates CAR-independent tropism and unique biodistribution in mice. Virology 2006, 350, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Wickham, T.J.; Mathias, P.; Cheresh, D.A.; Nemerow, G.R. Integrins aJS and U& promote adenovirus internalization but not virus attachment. Cell 1993, 73, 309–319. [Google Scholar] [PubMed]
- Bai, M.; Harfe, B.; Freimuth, P. Mutations that alter an Arg-Gly-Asp (RGD) sequence in the adenovirus type 2 penton base protein abolish its cell-rounding activity and delay virus reproduction in flat cells. J. Virol. 1993, 67, 5198–5205. [Google Scholar] [CrossRef] [PubMed]
- Albinsson, B.; Kidd, A.H. Adenovirus type 41 lacks an RGD α(v)-integrin binding motif on the penton base and undergoes delayed uptake in A549 cells. Virus Res. 1999, 64, 125–136. [Google Scholar] [CrossRef]
- Chiu, C.Y.; Mathias, P.; Nemerow, G.R.; Stewart, P.L. Structure of adenovirus complexed with its internalization receptor, αvβ5 integrin. J. Virol. 1999, 73, 6759–6768. [Google Scholar] [CrossRef]
- Li, E.; Stupack, D.; Bokoch, G.M.; Nemerow, G.R. Adenovirus endocytosis requires actin cytoskeleton reorganization mediated by rho family GTPases. J. Virol. 1998, 72, 8806–8812. [Google Scholar] [CrossRef]
- Li, E.; Stupack, D.G.; Brown, S.L.; Klemke, R.; Schlaepfer, D.D.; Nemerow, G.R. Association of p130(CAS) with phosphatidylinositol-3-OH kinase mediates adenovirus cell entry. J. Biol. Chem. 2000, 275, 14729–14735. [Google Scholar] [CrossRef]
- Wang, K.; Huang, S.; Kapoor-Munshi, A.; Nemerow, G. Adenovirus internalization and infection require dynamin. J. Virol. 1998, 72, 3455–3458. [Google Scholar] [CrossRef] [PubMed]
- Meier, O.; Boucke, K.; Hammer, S.V.; Keller, S.; Stidwill, R.P.; Hemmi, S.; Greber, U.F. Adenovirus triggers macropinocytosis and endosomal leakage together with its clathrin-mediated uptake. J. Cell Biol. 2002, 158, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Lindert, S.; Silvestry, M.; Mullen, T.-M.; Nemerow, G.R.; Stewart, P.L. Cryo-electron microscopy structure of an adenovirus-integrin complex indicates conformational changes in both penton base and integrin. J. Virol. 2009, 83, 11491–11501. [Google Scholar] [CrossRef] [PubMed]
- Snijder, J.; Reddy, V.S.; May, E.R.; Roos, W.H.; Nemerow, G.R.; Wuite, G.J.L. Integrin and defensin modulate the mechanical properties of adenovirus. J. Virol. 2013, 87, 2756–2766. [Google Scholar] [CrossRef]
- Greber, U.F.; Willetts, M.; Webster, P.; Helenius, A. Stepwise dismantling of adenovirus 2 during entry into cells. Cell 1993, 75, 477–486. [Google Scholar] [CrossRef]
- Martin-Fernandez, M.; Longshaw, S.V.; Kirby, I.; Santis, G.; Tobin, M.J.; Clarke, D.T.; Jones, G.R. Adenovirus type-5 entry and disassembly followed in living cells by FRET, fluorescence anisotropy, and FLIM. Biophys. J. 2004, 87, 1316–1327. [Google Scholar] [CrossRef]
- Burckhardt, C.J.; Suomalainen, M.; Schoenenberger, P.; Boucke, K.; Hemmi, S.; Greber, U.F. Drifting motions of the adenovirus receptor CAR and immobile integrins initiate virus uncoating and membrane lytic protein exposure. Cell Host Microbe 2011, 10, 105–117. [Google Scholar] [CrossRef]
- Wiethoff, C.M.; Wodrich, H.; Gerace, L.; Nemerow, G.R. Adenovirus protein VI mediates membrane disruption following capsid disassembly. J. Virol. 2005, 79, 1992–2000. [Google Scholar] [CrossRef]
- Wodrich, H.; Henaff, D.; Jammart, B.; Segura-Morales, C.; Seelmeir, S.; Coux, O.; Ruzsics, Z.; Wiethoff, C.M.; Kremer, E.J. A capsid-encoded PPxY-motif facilitates adenovirus entry. PLoS Pathog. 2010, 6, e1000808. [Google Scholar] [CrossRef]
- Maier, O.; Galan, D.L.; Wodrich, H.; Wiethoff, C.M. An N-terminal domain of adenovirus protein VI fragments membranes by inducing positive membrane curvature. Virology 2010, 402, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Maier, O.; Wiethoff, C.M. N-terminal α-helix-independent membrane interactions facilitate adenovirus protein VI induction of membrane tubule formation. Virology 2010, 408, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Montespan, C.; Wiethoff, C.M.; Wodrich, H. A small viral PPxY peptide motif to control antiviral autophagy. J. Virol. 2017, 91, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Montespan, C.; Marvin, S.A.; Austin, S.; Burrage, A.M.; Roger, B.; Rayne, F.; Faure, M.; Campell, E.M.; Schneider, C.; Reimer, R.; et al. Multi-layered control of Galectin-8 mediated autophagy during adenovirus cell entry through a conserved PPxY motif in the viral capsid. PLoS Pathog. 2017, 13, e1006217. [Google Scholar] [CrossRef]
- Daussy, C.F.; Wodrich, H. “Repair me if you can”: Membrane damage, response, and control from the viral perspective. Cells 2020, 9, 2042. [Google Scholar] [CrossRef]
- Gastaldelli, M.; Imelli, N.; Boucke, K.; Amstutz, B.; Meier, O.; Greber, U.F. Infectious adenovirus type 2 transport through early but not late endosomes. Traffic 2008, 9, 2265–2278. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, N.; Leopold, P.L.; Hackett, N.R.; Ferris, B.; Worgall, S.; Falck-Pedersen, E.; Crystal, R.G. Fiber swap between adenovirus subgroups B and C alters intracellular trafficking of adenovirus gene transfer vectors. J. Virol. 1999, 73, 6056–6065. [Google Scholar] [CrossRef]
- Shayakhmetov, D.M.; Li, Z.-Y.; Ternovoi, V.; Gaggar, A.; Gharwan, H.; Lieber, A. The interaction between the fiber knob domain and the cellular attachment receptor determines the intracellular trafficking route of adenoviruses. J. Virol. 2003, 77, 3712–3723. [Google Scholar] [CrossRef] [PubMed]
- Teigler, J.E.; Kagan, J.C.; Barouch, D.H. Late endosomal trafficking of alternative serotype adenovirus vaccine vectors augments antiviral innate immunity. J. Virol. 2014, 88, 10354–10363. [Google Scholar] [CrossRef] [PubMed]
- Barlan, A.U.; Griffin, T.M.; Mcguire, K.A.; Wiethoff, C.M. Adenovirus membrane penetration activates the NLRP3 inflammasome. J. Virol. 2011, 85, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Bremner, K.; Scherer, J.; Yi, J. Adenovirus transport through a direct cytoplasmic dynein-hexon interaction. Cell Host 2009, 6, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Leopold, P.L.; Kreitzer, G.; Miyazawa, N.; Rempel, S.; Pfister, K.K.; Rodriguez-Boulan, E.; Crystal, R.G. Dynein- and microtubule-mediated translocation of adenovirus serotype 5 occurs after endosomal lysis. Hum. Gene Ther. 2000, 11, 151–165. [Google Scholar] [CrossRef]
- Suomalainen, M.; Nakano, M.Y.; Keller, S.; Boucke, K.; Stidwill, R.P.; Greber, U.F. Microtubule-dependent plus- and minus end-directed motilities are competing processes for nuclear targeting of adenovirus. J. Cell Biol. 1999, 144, 657–672. [Google Scholar] [CrossRef]
- Scherer, J.; Vallee, R.B. Conformational changes in the adenovirus hexon subunit responsible for regulating cytoplasmic dynein recruitment. J. Virol. 2015, 89, 1013–1023. [Google Scholar] [CrossRef]
- Kelkar, S.A.; Pfister, K.K.; Crystal, R.G.; Leopold, P.L. Cytoplasmic dynein mediates adenovirus binding to microtubules. J. Virol. 2004, 78, 10122–10132. [Google Scholar] [CrossRef]
- Zhou, J.; Scherer, J.; Yi, J.; Vallee, R.B. Role of kinesins in directed adenovirus transport and cytoplasmic exploration. PLoS Pathog. 2018, 14, e1007055. [Google Scholar] [CrossRef]
- Trotman, L.C.; Mosberger, N.; Fornerod, M.; Stidwill, R.P.; Greber, U.F. Import of adenovirus DNA involves the nuclear pore complex receptor CAN/Nup214 and histone H1. Nat. Cell Biol. 2001, 3, 1092–1100. [Google Scholar] [CrossRef]
- Cassany, A.; Ragues, J.; Guan, T.; Bégu, D.; Wodrich, H.; Kann, M.; Nemerow, G.R.; Gerace, L. Nuclear import of adenovirus DNA involves direct interaction of hexon with an N-terminal domain of the nucleoporin Nup214. J. Virol. 2015, 89, 1719–1730. [Google Scholar] [CrossRef]
- Strunze, S.; Engelke, M.F.; Wang, I.H.; Puntener, D.; Boucke, K.; Schleich, S.; Way, M.; Schoenenberger, P.; Burckhardt, C.J.; Greber, U.F. Kinesin-1-mediated capsid disassembly and disruption of the nuclear pore complex promote virus infection. Cell Host Microbe 2011, 10, 210–223. [Google Scholar] [CrossRef]
- Puntener, D.; Engelke, M.F.; Ruzsics, Z.; Strunze, S.; Wilhelm, C.; Greber, U.F. Stepwise loss of fluorescent core protein V from human adenovirus during entry into cells. J. Virol. 2011, 85, 481–496. [Google Scholar] [CrossRef]
- Crisostomo, L.; Soriano, A.M.; Mendez, M.; Graves, D.; Pelka, P. Temporal dynamics of adenovirus 5 gene expression in normal human cells. PLoS ONE 2019, 14, e0211192. [Google Scholar] [CrossRef] [PubMed]
- Thomas, G.P.; Mathews, M.B. DNA replication and the early to late transition in adenovirus infection. Cell 1980, 22, 523–533. [Google Scholar] [CrossRef]
- Nevins, J.R. Mechanism of activation of early viral transcription by the adenovirus E1A gene product. Cell 1981, 26, 213–220. [Google Scholar] [CrossRef]
- Flint, S.J. Regulation of adenovirus mrna formation. Adv. Virus Res. 1986, 31, 169–228. [Google Scholar] [CrossRef]
- Nevins, J.R. Regulation of early adenovirus gene expression. Microbiol. Rev. 1987, 51, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Farley, D.C.; Brown, J.L.; Leppard, K.N. Activation of the early-late switch in adenovirus type 5 major late transcription unit expression by L4 gene products. J. Virol. 2004, 78, 1782–1791. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, P.; Gonzalez, R.A. Formation of adenovirus DNA replication compartments. FEBS Lett. 2019, 593, 3518–3530. [Google Scholar] [CrossRef] [PubMed]
- Mondersert, G.; Tribouley, C.; Kedinger, C. Identification of a novel downstream bingind protein implicated in late-phase-specific activation of the adenovirus major late promoter. Nucleic Acids Res. 1992, 20, 3881–3889. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.R.; Ziff, E.B. Transcripts from the adenovirus-2 major late promoter yield a single early family of 3′ coterminal mRNAs and five late families. Cell 1980, 22, 905–916. [Google Scholar] [CrossRef]
- Weber, J.M.; Déry, C.V.; Amin Mirza, M.; Horvath, J. Adenovirus DNA synthesis is coupled to virus assembly. Virology 1985, 140, 351–359. [Google Scholar] [CrossRef]
- Tollefson, A.E.; Ryerse, J.S.; Scaria, A.; Hermiston, T.W.; Wold, W.S.M. The E3-11.6-kDa adenovirus death protein (ADP) is required for efficient cell death: Characterization of cells infected with adp mutants. Virology 1996, 220, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Tollefson, A.E.; Scaria, A.; Hermiston, T.W.; Ryerse, J.S.; Wold, L.J.; Wold, W.S. The adenovirus death protein (E3-11.6K) is required at very late stages of infection for efficient cell lysis and release of adenovirus from infected cells. J. Virol. 1996, 70, 2296–2306. [Google Scholar] [CrossRef]
- Weber, J. Genetic analysis of adenovirus type 2 III. Temperature sensitivity of processing viral proteins. J. Virol. 1976, 17, 462–471. [Google Scholar] [CrossRef]
- Greber, U.F.; Webster, P.; Weber, J.; Helenius, A. The role of the adenovirus protease in virus entry into cells. EMBO J. 1996, 15, 1766–1777. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, E.K.; Nemerow, G.R.; Smith, J.G. Direct evidence from single-cell analysis that human α-defensins block adenovirus uncoating to neutralize infection. J. Virol. 2010, 84, 4041–4049. [Google Scholar] [CrossRef]
- Worgall, S.; Wolff, G.; Falck-Pedersen, E.; Crystal, R.G. Innate immune mechanisms dominate elimination of adenoviral vectors following in vivo administration. Hum. Gene Ther. 1997, 8, 37–44. [Google Scholar] [CrossRef]
- Yang, Y.; Nunes, F.A.; Berencsi, K.; Furth, E.E.; Gönczöl, E.; Wilson, J.M. Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc. Natl. Acad. Sci. USA 1994, 91, 4407–4411. [Google Scholar] [CrossRef]
- Tatsis, N.; Ertl, H.C.J. Adenoviruses as vaccine vectors. Mol. Ther. 2004, 10, 616–629. [Google Scholar] [CrossRef] [PubMed]
- Li, J.X.; Hou, L.H.; Meng, F.Y.; Wu, S.P.; Hu, Y.M.; Liang, Q.; Chu, K.; Zhang, Z.; Xu, J.J.; Tang, R.; et al. Immunity duration of a recombinant adenovirus type-5 vector-based Ebola vaccine and a homologous prime-boost immunisation in healthy adults in China: Final report of a randomised, double-blind, placebo-controlled, phase 1 trial. Lancet Glob. Health 2017, 5, e324–e334. [Google Scholar] [CrossRef]
- Raper, S.E.; Chirmule, N.; Lee, F.S.; Wivel, N.A.; Bagg, A.; Gao, G.P.; Wilson, J.M.; Batshaw, M.L. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer. Mol. Genet. Metab. 2003, 80, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Schnell, M.A.; Zhang, Y.; Tazelaar, J.; Gao, G.P.; Yu, Q.C.; Qian, R.; Chen, S.J.; Varnavski, A.N.; LeClair, C.; Raper, S.E.; et al. Activation of innate immunity in nonhuman primates following intraportal administration of adenoviral vectors. Mol. Ther. 2001, 3, 708–722. [Google Scholar] [CrossRef] [PubMed]
- Hendrickx, R.; Stichling, N.; Koelen, J.; Kuryk, L.; Lipiec, A.; Greber, U.F. Innate immunity to adenovirus. Hum. Gene Ther. 2014, 25, 265–284. [Google Scholar] [CrossRef] [PubMed]
- Atasheva, S.; Shayakhmetov, D.M. Innate Immune Response to Adenovirus Vector Administration in Vivo, 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; ISBN 9780128002766. [Google Scholar]
- Atasheva, S.; Yao, J.; Shayakhmetov, D.M. Innate immunity to adenovirus: Lessons from mice. FEBS Lett. 2019, 593, 3461–3483. [Google Scholar] [CrossRef] [PubMed]
- Gregory, S.M.; Nazir, S.A.; Metcalf, J.P. Implications of the innate immune response to adenovirus and adenoviral vectors. Future Virol. 2011, 6, 357–374. [Google Scholar] [CrossRef]
- Borgland, S.L.; Bowen, G.P.; Wong, N.C.W.; Libermann, T.A.; Muruve, D.A. Adenovirus vector-induced expression of the C-X-C chemokine IP-10 is mediated through capsid-dependent activation of NF-κB. J. Virol. 2000, 74, 3941–3947. [Google Scholar] [CrossRef]
- Bowen, G.P.; Borgland, S.L.; Lam, M.; Libermann, T.A.; Wong, N.C.W.; Muruve, D.A. Adenovirus vector-induced inflammation: Capsid-dependent induction of the C-C chemokine RANTES requires NF-κB. Hum. Gene Ther. 2002, 13, 367–379. [Google Scholar] [CrossRef]
- Higginbotham, J.N.; Seth, P.; Blaese, R.M.; Ramsey, W.J. The release of inflammatory cytokines from human peripheral blood mononuclear cells in vitro following exposure to adenovirus variants and capsid. Hum. Gene Ther. 2002, 13, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Mccoy, R.D.; Roessler, B.J.; Huffnagle, G.A.R.Y.B.; Janich, S.L.; Laing, T.J.; Sevion, R.H. Pulmonary inflammation induced by incomplete or inactivated adenoviral particles. Hum. Gene Ther. 1995, 6, 1553–1560. [Google Scholar] [CrossRef] [PubMed]
- Muruve, D.A.; Barnes, M.J.; Stillman, I.E.; Libermann, T.A. Adenoviral gene therapy leads to rapid induction of multiple chemokines and acute neutrophil-dependent hepatic injury in vivo. Hum. Gene Ther. 1999, 10, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Muruve, D.A. The innate immune response to adenovirus vectors. Hum. Gene Ther. 2004, 15, 1157–1166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chirmule, N.; Gao, G.P.; Qian, R.; Croyle, M.; Joshi, B.; Tazelaar, J.; Wilson, J.M. Acute cytokine response to systemic adenoviral vectors in mice is mediated by dendritic cells and macrophages. Mol. Ther. 2001, 3, 697–707. [Google Scholar] [CrossRef] [PubMed]
- Tamanini, A.; Nicolis, E.; Bonizzato, A.; Bezzerri, V.; Melotti, P.; Assael, B.M.; Cabrini, G. Interaction of adenovirus type 5 fiber with the coxsackievirus and adenovirus receptor activates inflammatory response in human respiratory cells. J. Virol. 2006, 80, 11241–11254. [Google Scholar] [CrossRef] [PubMed]
- Farmer, C.; Morton, P.E.; Snippe, M.; Santis, G.; Parsons, M. Coxsackie adenovirus receptor (CAR) regulates integrin function through activation of p44/42 MAPK. Exp. Cell Res. 2009, 315, 2637–2647. [Google Scholar] [CrossRef]
- Iacobelli-Martinez, M.; Nepomuceno, R.R.; Connolly, J.; Nemerow, G.R. CD46-utilizing adenoviruses inhibit C/EBPβ-dependent expression of proinflammatory cytokines. J. Virol. 2005, 79, 11259–11268. [Google Scholar] [CrossRef]
- Di Paolo, N.C.; Miao, E.A.; Iwakura, Y.; Kaja, M.; Flavell, R.A.; Papayannopoulou, T.; Shayakhmetov, D.M. Virus sensing at the plasma membrane triggers interleukin-1α-mediated pro-inflammatory macrophage response in vivo. Thalia Stud. Lit. Humor 2010, 31, 110–121. [Google Scholar] [CrossRef]
- Suomalainen, M.; Nakano, M.Y.; Boucke, K.; Keller, S.; Greber, U.F. Adenovirus-activated PKA and p38/MAPK pathways boost microtubule-mediated nuclear targeting of virus. EMBO J. 2001, 20, 1310–1319. [Google Scholar] [CrossRef]
- Tibbles, L.A.; Spurrell, J.C.L.; Bowen, G.P.; Liu, Q.; Lam, M.; Zaiss, A.K.; Robbins, S.M.; Hollenberg, M.D.; Wickham, T.J.; Muruve, D.A. Activation of p38 and ERK signaling during adenovirus vector cell entry lead to expression of the C-X-C chemokine IP-10. J. Virol. 2002, 76, 1559–1568. [Google Scholar] [CrossRef]
- Li, E.; Stupack, D.; Klemke, R.; Cheresh, D.A.; Nemerow, G.R. Adenovirus endocytosis via αvintegrins requires phosphoinositide-3-OH kinase. J. Virol. 1998, 72, 2055–2061. [Google Scholar] [CrossRef]
- Philpott, N.J.; Nociari, M.; Elkon, K.B.; Falck-Pedersen, E. Adenovirus-induced maturation of dendritic cells through a PI3 kinase-mediated TNF-α induction pathway. Proc. Natl. Acad. Sci. USA 2004, 101, 6200–6205. [Google Scholar] [CrossRef]
- Zsengellér, Z.; Otake, K.; Hossain, S.-A.; Berclaz, P.-Y.; Trapnell, B.C. Internalization of adenovirus by alveolar macrophages initiates early proinflammatory signaling during acute respiratory tract infection. J. Virol. 2000, 74, 9655–9667. [Google Scholar] [CrossRef]
- Liu, Q.; Muruve, D.A. Molecular basis of the inflammatory response to adenovirus vectors. Gene Ther. 2003, 10, 935–940. [Google Scholar] [CrossRef]
- Cerullo, V.; Seiler, M.P.; Mane, V.; Brunetti-Pierri, N.; Clarke, C.; Bertin, T.K.; Rodgers, J.R.; Lee, B. Toll-like receptor 9 triggers an innate immune response to helper-dependent adenoviral vectors. Mol. Ther. 2007, 15, 378–385. [Google Scholar] [CrossRef]
- Fejer, G.; Drechsel, L.; Liese, J.; Schleicher, U.; Ruzsics, Z.; Imelli, N.; Greber, U.F.; Keck, S.; Hildenbrand, B.; Krug, A.; et al. Key role of splenic myeloid DCs in the IFN-αβ response to adenoviruses in vivo. PLoS Pathog. 2008, 4, e1000208. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Huang, X.; Yang, Y. Innate immune response to adenoviral vectors is mediated by both toll-like receptor-dependent and -independent pathways. J. Virol. 2007, 81, 3170–3180. [Google Scholar] [CrossRef]
- Stein, S.C.; Falck-Pedersen, E. Sensing adenovirus infection: Activation of interferon regulatory factor 3 in RAW 264.7 cells. J. Virol. 2012, 86, 4527–4537. [Google Scholar] [CrossRef] [PubMed]
- Nociari, M.; Ocheretina, O.; Murphy, M.; Falck-Pedersen, E. Adenovirus induction of IRF3 occurs through a binary trigger targeting jun N-terminal kinase and TBK1 kinase cascades and type I interferon autocrine signaling. J. Virol. 2009, 83, 4081–4091. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Muruve, D.A.; Pétrilli, V.; Zaiss, A.K.; White, L.R.; Clark, S.A.; Ross, P.J.; Parks, R.J.; Tschopp, J. The inflammasome recognizes cytosolic microbial and host DNA and triggers an innate immune response. Nature 2008, 452, 103–107. [Google Scholar] [CrossRef]
- Lam, E.; Falck-Pedersen, E. Unabated adenovirus replication following activation of the cGAS/STING-dependent antiviral response in human cells. J. Virol. 2014, 88, 14426–14439. [Google Scholar] [CrossRef]
- Pahl, J.H.W.; Verhoeven, D.H.J.; Kwappenberg, K.M.C.; Vellinga, J.; Lankester, A.C.; van Tol, M.J.D.; Schilham, M.W. Adenovirus type 35, but not type 5, stimulates NK cell activation via plasmacytoid dendritic cells and TLR9 signaling. Mol. Immunol. 2012, 51, 91–100. [Google Scholar] [CrossRef]
- Wohlfart, C.E.; Svensson, U.K.; Everitt, E. Interaction between HeLa cells and adenovirus type 2 virions neutralized by different antisera. J. Virol. 1985, 56, 896–903. [Google Scholar] [CrossRef]
- Watson, G.; Burdon, M.G.; Russell, W.C. An antigenic analysis of the adenovirus type 2 fibre polypeptide. J. Gen. Virol. 1988, 69, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xiang, Z.Q.; Li, Y.; Kurupati, R.K.; Jia, B.; Bian, A.; Zhou, D.M.; Hutnick, N.; Yuan, S.; Gray, C.; et al. Adenovirus-based vaccines: Comparison of vectors from three species of adenoviridae. J. Virol. 2010, 84, 10522–10532. [Google Scholar] [CrossRef]
- Roberts, D.M.; Nanda, A.; Havenga, M.J.E.; Abbink, P.; Lynch, D.M.; Ewald, B.A.; Liu, J.; Thorner, A.R.; Swanson, P.E.; Gorgone, D.A.; et al. Hexon-chimaeric adenovirus serotype 5 vectors circumvent pre-existing anti-vector immunity. Nature 2006, 441, 239–243. [Google Scholar] [CrossRef]
- Sumida, S.M.; Truitt, D.M.; Lemckert, A.A.C.; Vogels, R.; Custers, J.H.H.V.; Addo, M.M.; Lockman, S.; Peter, T.; Peyerl, F.W.; Kishko, M.G.; et al. Neutralizing antibodies to adenovirus serotype 5 vaccine vectors are directed primarily against the adenovirus hexon protein. J. Immunol. 2005, 174, 7179–7185. [Google Scholar] [CrossRef]
- Bradley, R.R.; Lynch, D.M.; Iampietro, M.J.; Borducchi, E.N.; Barouch, D.H. Adenovirus serotype 5 neutralizing antibodies target both hexon and fiber following vaccination and natural infection. J. Virol. 2012, 86, 625–629. [Google Scholar] [CrossRef]
- Ahi, S.Y.; Bangari, D.S.; Mittal, S.K. Adenoviral vector immunity: Its implications and circumvention strategies. Curr. Gene Ther. 2011, 11, 307–320. [Google Scholar] [CrossRef]
- Mast, T.C.; Kierstead, L.; Gupta, S.B.; Nikas, A.A.; Kallas, E.G.; Novitsky, V.; Mbewe, B.; Pitisuttithum, P.; Schechter, M.; Vardas, E.; et al. International epidemiology of human pre-existing adenovirus (Ad) type-5, type-6, type-26 and type-36 neutralizing antibodies: Correlates of high Ad5 titers and implications for potential HIV vaccine trials. Vaccine 2010, 28, 950–957. [Google Scholar] [CrossRef]
- Zaiss, A.K.; Machado, H.B.; Herschman, H.R. The influence of innate and pre-existing immunity on adenovirus therapy. J. Cell. Biochem. 2009, 108, 778–790. [Google Scholar] [CrossRef]
- Colloca, S.; Barnes, E.; Folgori, A.; Ammendola, V.; Capone, S.; Cirillo, A.; Siani, L.; Naddeo, M.; Grazioli, F.; Esposito, M.L.; et al. Generation and screening of a large collection of novel simian Adenovirus allows the identification of vaccine vectors inducing potent cellular immunity in humans. Sci. Transl. Med. 2012, 4, 115ra2. [Google Scholar] [CrossRef]
- Nébié, I.; Edwards, N.J.; Tiono, A.B.; Ewer, K.J.; Sanou, G.S.; Soulama, I.; Sanon, S.; Diarra, A.; Yaro, J.B.; Kangoye, D.; et al. Assessment of chimpanzee adenovirus serotype 63 neutralizing antibodies prior to evaluation of a candidate malaria vaccine regimen based on viral vectors. Clin. Vaccine Immunol. 2014, 21, 901–903. [Google Scholar] [CrossRef]
- De Santis, O.; Audran, R.; Pothin, E.; Warpelin-Decrausaz, L.; Vallotton, L.; Wuerzner, G.; Cochet, C.; Estoppey, D.; Steiner-Monard, V.; Lonchampt, S.; et al. Safety and immunogenicity of a chimpanzee adenovirus-vectored Ebola vaccine in healthy adults: A randomised, double-blind, placebo-controlled, dose-finding, phase 1/2a study. Lancet Infect. Dis. 2016, 16, 311–320. [Google Scholar] [CrossRef]
- Zhu, F.C.; Guan, X.H.; Li, Y.H.; Huang, J.Y.; Jiang, T.; Hou, L.H.; Li, J.X.; Yang, B.F.; Wang, L.; Wang, W.J.; et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2020, 396, 479–488. [Google Scholar] [CrossRef]
- Olive, M.; Eisenlohr, L.C.; Flomenberg, P. T-cell responses from healthy adults. Viral Immunol. 2001, 14, 403–413. [Google Scholar] [CrossRef]
- Flomenberg, P.; Piaskowski, V.; Truitt, R.L.; Casper, J.T. Characterization of human proliferative t cell responses to adenovirus. J. Infect. Dis. 1995, 171, 1090–1096. [Google Scholar] [CrossRef]
- Pédron, B.; Guérin, V.; Cordeiro, D.J.; Masmoudi, S.; Dalle, J.H.; Sterkers, G. Development of cytomegalovirus and adenovirus-specific memory CD4 T-cell functions from birth to adulthood. Pediatr. Res. 2011, 69, 106–111. [Google Scholar] [CrossRef]
- Flomenberg, P.; Piaskowski, V.; Truitt, R.L.; Casper, J.T. Human adenovirus-specific CD8+ T-cell responses are not inhibited by E3-19K in the presence of gamma interferon. J. Virol. 1996, 70, 6314–6322. [Google Scholar] [CrossRef] [PubMed]
- Leen, A.M.; Sili, U.; Savoldo, B.; Jewell, A.M.; Piedra, P.A.; Brenner, M.K.; Rooney, C.M. Fiber-modified adenoviruses generate subgroup cross-reactive, adenovirus-specific cytotoxic T lymphocytes for therapeutic applications. Blood 2004, 103, 1011–1019. [Google Scholar] [CrossRef]
- Smith, C.A.; Woodruff, L.S.; Rooney, C.; Kitchingman, G.R. Extensive cross-reactivity of cytotoxic T cells overview summary introduction. Hum. Gene Ther. 1998, 1427, 1419–1427. [Google Scholar] [CrossRef]
- Veltrop-Duits, L.A.; Heemskerk, B.; Sombroek, C.C.; van Vreeswijk, T.; Gubbels, S.; Toes, R.E.M.; Melief, C.J.M.; Franken, K.L.M.C.; Havenga, M.; van Tol, M.J.D.; et al. Human CD4+ T cells stimulated by conserved adenovirus 5 hexon peptides recognize cells infected with different species of human adenovirus. Eur. J. Immunol. 2006, 36, 2410–2423. [Google Scholar] [CrossRef]
- Zandvliet, M.L.; Falkenburg, J.h.F.; van Liempt, E.V.; Veltrop-Duits, L.A.; Lankester, A.C.; Kalpoe, J.S.; Kester, M.G.D.; van der Steen, D.M.; van Tol, M.J.; Willemze, R.; et al. Combined cd8+ and cd4+ adenovirus hexon-specific T cells associated with viral clearance after stem cell transplantation as treatment for adenovirus infection. Haematologica 2010, 95, 1943–1951. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.C.; Dayball, K.; Wan, Y.H.; Bramson, J. Detailedanalysis of the CD8+ T-cell response followingadenovirusvaccination. J. Virol. 2003, 77, 13407–13411. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.Y.; Millar, J.B.; Grinshtein, N.; Bassett, J.; Finn, J.; Bramson, J.L. T-cell immunity generated by recombinant adenovirus vaccines. Expert Rev. Vaccines 2007, 6, 347–356. [Google Scholar]
- Ouédraogo, A.; Tiono, A.B.; Kargougou, D.; Yaro, J.B.; Ouédraogo, E.; Kaboré, Y.; Kangoye, D.; Bougouma, E.C.; Gansane, A.; Henri, N.; et al. A phase 1b randomized, controlled, double-blinded dosage-escalation trial to evaluate the safety, reactogenicity and immunogenicity of an adenovirus type 35 based circumsporozoite malaria vaccine in burkinabe healthy adults 18 to 45 years of age. PLoS ONE 2013, 8, e78679. [Google Scholar] [CrossRef]
- Xiang, Z.Q.; Yang, Y.; Wilson, J.M.; Ertl, H.C.J. A replication-defective human adenovirus recombinant serves as a highly efficacious vaccine carrier. Virology 1996, 219, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Bassett, J.D.; Swift, S.L.; Bramson, J.L. Optimizing vaccine-induced CD8 + T-cell immunity: Focus on recombinant adenovirus vectors. Expert Rev. Vaccines 2011, 10, 1307–1319. [Google Scholar] [CrossRef] [PubMed]
- Tatsis, N.; Fitzgerald, J.C.; Reyes-Sandoval, A.; Harris-McCoy, K.C.; Hensley, S.E.; Zhou, D.; Lin, S.W.; Bian, A.; Zhi, Q.X.; Iparraguirre, A.; et al. Adenoviral vectors persist in vivo and maintain activated CD8+ T cells: Implications for their use as vaccines. Blood 2007, 110, 1916–1923. [Google Scholar] [CrossRef]
- Heemskerk, B.; Veltrop-Duits, L.A.; van Vreeswijk, T.; ten Dam, M.M.; Heidt, S.; Toes, R.E.M.; van Tol, M.J.D.; Schilham, M.W. extensive cross-reactivity of CD4+ adenovirus-specific T cells: Implications for immunotherapy and gene therapy. J. Virol. 2003, 77, 6562–6566. [Google Scholar] [CrossRef]
- Rock, K.L.; Reits, E.; Neefjes, J. Present yourself! By MHC class I and MHC class II molecules. Trends Immunol. 2016, 37, 724–737. [Google Scholar] [CrossRef]
- Münz, C. Autophagy beyond intracellular MHC class II antigen presentation. Trends Immunol. 2016, 37, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Cadwell, K. Crosstalk between autophagy and inflammatory signalling pathways: Balancing defence and homeostasis. Nat. Rev. Immunol. 2016, 16, 661–675. [Google Scholar] [CrossRef] [PubMed]
- Weidberg, H.; Elazar, Z. TBK1 mediates crosstalk between the innate immune response and autophagy. Sci. Signal. 2011, 4, 1–4. [Google Scholar] [CrossRef]
- Deretic, V.; Levine, B. Autophagy balances inflammation in innate immunity. Autophagy 2018, 14, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Parzych, K.R.; Klionsky, D.J. An overview of autophagy: Morphology, mechanism, and regulation. Antioxid. Redox Signal. 2014, 20, 460–473. [Google Scholar] [CrossRef]
- Deretic, V. Autophagy as an immune defense mechanism. Curr. Opin. Immunol. 2006, 18, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Münz, C. Autophagy proteins influence endocytosis for MHC restricted antigen presentation. Semin. Cancer Biol. 2020, 66, 110–115. [Google Scholar] [CrossRef]
- Nimmerjahn, F.; Milosevic, S.; Behrends, U.; Jaffee, E.M.; Pardoll, D.M.; Bornkamm, G.W.; Mautner, J. Major histocompatibility complex class II-restricted presentation of a cytosolic antigen by autophagy. Eur. J. Immunol. 2003, 33, 1250–1259. [Google Scholar] [CrossRef] [PubMed]
- Paludan, C.; Schmid, D.; Landthaler, M.; Vockerodt, M.; Kube, D.; Tuschl, T.; Münz, C. Endogenous MHC class II processing of a viral nuclear antigen after autophagy. Science 2005, 307, 593–596. [Google Scholar] [CrossRef]
- Leung, C.S.; Haigh, T.A.; Mackay, L.K.; Rickinson, A.B.; Taylor, G.S. Nuclear location of an endogenously expressed antigen, EBNA1, restricts access to macroautophagy and the range of CD4 epitope display. Proc. Natl. Acad. Sci. USA 2010, 107, 2165–2170. [Google Scholar] [CrossRef]
- Lee, H.K.; Mattei, L.M.; Steinberg, B.E.; Alberts, P.; Lee, Y.H.; Chervonsky, A.; Mizushima, N.; Grinstein, S.; Iwasaki, A. In vivo requirement for Atg5 in antigen presentation by dendritic cells. Immunity 2010, 32, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Comber, J.D.; Robinson, T.M.; Siciliano, N.A.; Snook, A.E.; Eisenlohr, L.C. Functional macroautophagy induction by influenza a virus without a contribution to major histocompatibility complex class II-restricted presentation. J. Virol. 2011, 85, 6453–6463. [Google Scholar] [CrossRef] [PubMed]
- Blanchet, F.P.; Moris, A.; Nikolic, D.S.; Lehmann, M.; Cardinaud, S.; Stalder, R.; Garcia, E.; Dinkins, C.; Leuba, F.; Wu, L.; et al. Human immunodeficiency virus-1 inhibition of immunoamphisomes in dendritic cells impairs early innate and adaptive immune responses. Immunity 2010, 32, 654–669. [Google Scholar] [CrossRef]
- Heckmann, B.L.; Green, D.R. LC3-associated phagocytosis at a glance. J. Cell Sci. 2019, 132, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Romao, S.; Gasser, N.; Becker, A.C.; Guhl, B.; Bajagic, M.; Vanoaica, D.; Ziegler, U.; Roesler, J.; Dengjel, J.; Reichenbach, J.; et al. Autophagy proteins stabilize pathogen-containing phagosomes for prolonged MHC II antigen processing. J. Cell Biol. 2013, 203, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.K.; Khanna, R. Autophagy mediates transporter associated with antigen processing-independent presentation of viral epitopes through MHC class I pathway. Blood 2012, 120, 994–1004. [Google Scholar] [CrossRef]
- Das, M.; Kaveri, S.V.; Bayry, J. Cross-presentation of antigens by dendritic cells: Role of autophagy. Oncotarget 2015, 6, 28527–28528. [Google Scholar] [CrossRef]
- Uhl, M.; Kepp, O.; Jusforgues-Saklani, H.; Vicencio, J.M.; Kroemer, G.; Albert, M.L. Autophagy within the antigen donor cell facilitates efficient antigen cross-priming of virus-specific CD8+ T cells. Cell Death Differ. 2009, 16, 991–1005. [Google Scholar] [CrossRef]
- Yi, Y.; Zhou, Z.; Shu, S.; Fang, Y.; Twitty, C.; Hilton, T.L.; Aung, S.; Urba, W.J.; Fox, B.A.; Hu, H.M.; et al. Autophagy-assisted antigen cross-presentation: Autophagosome as the argo of shared tumor-specifc antigens and DAMPs. Oncoimmunology 2012, 1, 976–978. [Google Scholar] [CrossRef]
- Crotzer, V.L.; Blum, J.S. Autophagy and its role in MHC-mediated antigen presentation. J. Immunol. 2009, 182, 3335–3341. [Google Scholar] [CrossRef]
- Windheim, M.; Hilgendorf, A.; Burgert, H.G. Immune evasion by adenovirus E3 proteins: Exploitation of intracellular trafficking pathways. Curr. Top. Microbiol. Immunol. 2004, 273, 29–85. [Google Scholar]
- Ginsberg, H.S. The ups and downs of adenovirus vectors. Bull. N. Y. Acad. Med. J. Urban. Health 1996, 73, 53–58. [Google Scholar]
- Bennett, E.M.; Bennink, J.R.; Yewdell, J.W.; Brodsky, F.M. Cutting edge: Adenovirus E19 has two mechanisms for affecting class I MHC expression. J. Immunol. 1999, 162, 5049–5052. [Google Scholar]
- Oliveira, E.R.A.; Bouvier, M. Immune evasion by adenoviruses: A window into host-virus adaptation. FEBS Lett. 2019, 593, 3496–3503. [Google Scholar] [CrossRef]
- Burgert, H.G. Subversion of the MHC class I antigen-presentation pathway by adenoviruses and herpes simplex viruses. Trends Microbiol. 1996, 4, 107–112. [Google Scholar] [CrossRef]
- Klein, S.R.; Jiang, H.; Hossain, M.B.; Fan, X.; Gumin, J.; Dong, A.; Alonso, M.M.; Gomez-Manzano, C.; Fueyo, J. Critical role of autophagy in the processing of adenovirus capsid-incorporated cancer-specific antigens. PLoS ONE 2016, 11, e0153814. [Google Scholar] [CrossRef] [PubMed]
- Neukirch, L.; Fougeroux, C.; Andersson, A.M.C.; Holst, P.J. The Potential of Adenoviral Vaccine Vectors with Altered Antigen Presentation Capabilities; Taylor & Francis: Milton Park, UK, 2020; Volume 19, ISBN 0000000275668. [Google Scholar]
- Tang, D.; Kang, R.; Livesey, K.M.; Cheh, C.W.; Farkas, A.; Loughran, P.; Hoppe, G.; Bianchi, M.E.; Tracey, K.J.; Zeh, H.J.; et al. Endogenous HMGB1 regulates autophagy. J. Cell Biol. 2010, 190, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Degenhardt, K.; Mathew, R.; Beaudoin, B.; Bray, K.; Anderson, D.; Chen, G.; Mukherjee, C.; Shi, Y.; Gélinas, C.; Fan, Y.; et al. Autophagy promotes tumor cell survival and restricts necrosis, inflammation, and tumorigenesis. Cancer Cell 2006, 10, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Jiang, L.; Fu, X.; Wang, W.; Ma, J.; Tian, T.; Nan, K.; Liang, X. Cytoplasmic liver kinase B1 promotes the growth of human lung adenocarcinoma by enhancing autophagy. Cancer Sci. 2018, 109, 3055–3067. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Martis, P.C.; Excoffon, K.J.D.A. Adenovirus transduction: More complicated than receptor expression. Virology 2017, 502, 144–151. [Google Scholar] [CrossRef]
- Tritel, M.; Stoddard, A.M.; Flynn, B.J.; Darrah, P.A.; Wu, C.; Wille, U.; Shah, J.A.; Huang, Y.; Xu, L.; Betts, M.R.; et al. Prime-boost vaccination with HIV-1 gag protein and cytosine phosphate guanosine oligodeoxynucleotide, followed by adenovirus, induces sustained and robust humoral and cellular immune responses. J. Immunol. 2003, 171, 2538–2547. [Google Scholar] [CrossRef] [PubMed]
- Ertl, H.C. Viral vectors as vaccine carriers. Curr. Opin. Virol. 2016, 21, 1–8. [Google Scholar] [CrossRef]
- Ginn, S.L.; Amaya, A.K.; Alexander, I.E.; Edelstein, M.; Abedi, M.R. Gene therapy clinical trials worldwide to 2017: An update. J. Gene Med. 2018, 20, e3015. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Mese, K.; Bunz, O.; Ehrhardt, A. State-of-the-art human adenovirus vectorology for therapeutic approaches. FEBS Lett. 2019, 593, 3609–3622. [Google Scholar] [CrossRef]
- McGrory, W.J.; Bautista, D.S.; Graham, F.L. A simple technique for the rescue of early region I mutations into infectious human adenovirus type 5. Virology 1988, 163, 614–617. [Google Scholar] [CrossRef]
- Wold, W.S.M.; Doronin, K.; Toth, K.; Kuppuswamy, M.; Lichtenstein, D.L.; Tollefson, A.E. Immune responses to adenoviruses: Viral evasion mechanisms and their implications for the clinic. Curr. Opin. Immunol. 1999, 11, 380–386. [Google Scholar] [CrossRef]
- Crystal, R.G. Adenovirus: The first effective in vivo gene delivery vector. Hum. Gene Ther. 2014, 25, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Bett, A.J.; Prevec, L.; Graham, F.L. Packaging capacity and stability of human adenovirus type 5 vectors. J. Virol. 1993, 67, 5911–5921. [Google Scholar] [CrossRef]
- Bett, A.J.; Haddara, W.; Prevec, L.; Graham, F.L. An efficient and flexible system for construction of adenovirus vectors with insertions or deletions in early regions 1 and 3. Proc. Natl. Acad. Sci. USA 1994, 91, 8802–8806. [Google Scholar] [CrossRef]
- Akusjärvi, G. Proteins with transcription regulatory properties encoded by human adenoviruses. Trends Microbiol. 1993, 1, 163–170. [Google Scholar] [CrossRef]
- Graham, F.L.; Smiley, J.; Russell, W.C.; Nairn, R. Characteristics of a human cell line transformed by DNA from human adenovirus type 5. J. Gen. Virol. 1977, 36, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Shaw, G.; Morse, S.; Ararat, M.; Graham, F.L. Preferential transformation of human neuronal cells by human adenoviruses and the origin of HEK 293 cells. FASEB J. 2002, 16, 869–871. [Google Scholar] [CrossRef] [PubMed]
- Benihoud, K.; Yeh, P.; Perricaudet, M. Adenovirus vectors for gene delivery. Curr. Opin. Biotechnol. 1999, 10, 440–447. [Google Scholar] [CrossRef]
- Hehir, K.M.; Armentano, D.; Cardoza, L.M.; Choquette, T.L.; Berthelette, P.B.; White, G.A.; Couture, L.A.; Everton, M.B.; Keegan, J.; Martin, J.M.; et al. Molecular characterization of replication-competent variants of adenovirus vectors and genome modifications to prevent their occurrence. J. Virol. 1996, 70, 8459–8467. [Google Scholar] [CrossRef]
- Kreppel, F.; Hagedorn, C. Capsid and genome modification strategies to reduce the immunogenicity of adenoviral vectors. Int. J. Mol. Sci. 2021, 22, 2417. [Google Scholar] [CrossRef]
- Yang, Y.; Ertl, H.C.J.; Wilson, J.M. MHC class I-cestricted cytotoxic T lymphocytes to viral antigens destroy hepatocytes in mice infected with E1-deleted recombinant adenoviruses. Immunity 1994, 1, 433–442. [Google Scholar] [CrossRef]
- Engelhardt, J.F.; Litzky, L.; Wilson, J.M. Prolonged transgene expression in cotton rat lung with recombinant adenoviruses defective in E2a. Hum. Gene Ther. 1994, 5, 1217–1229. [Google Scholar] [CrossRef]
- Lusky, M.; Christ, M.; Rittner, K.; Dieterle, A.; Dreyer, D.; Mourot, B.; Schultz, H.; Stoeckel, F.; Pavirani, A.; Mehtali, M. In vitro and in vivo biology of recombinant adenovirus with E1, E1/E2A, or E1/E4 deleted. J. Virol. 1998, 72, 11. [Google Scholar] [CrossRef]
- Schaack, J. Induction and inhibition of innate inflammatory responses by adenovirus early region proteins. Viral Immunol. 2005, 18, 79–88. [Google Scholar] [CrossRef]
- Janssen, J.M.; Liu, J.; Skokan, J.; Gonçalves, M.A.F.V.; de Vries, A.A.F. Development of an AdEasy-based system to produce first- and second-generation adenoviral vectors with tropism for CAR- or CD46-positive cells. J. Gene Med. 2013, 15, 1–11. [Google Scholar] [CrossRef]
- Dedieu, J.F.; Vigne, E.; Torrent, C.; Jullien, C.; Mahfouz, I.; Caillaud, J.M.; Aubailly, N.; Orsini, C.; Guillaume, J.M.; Opolon, P.; et al. Long-term gene delivery into the livers of immunocompetent mice with E1/E4-defective adenoviruses. J. Virol. 1997, 71, 4626–4637. [Google Scholar] [CrossRef] [PubMed]
- Kochanek, S. High-capacity adenoviral vectors for gene transfer and somatic gene therapy. Hum. Gene Ther. 1999, 10, 2451–2459. [Google Scholar] [CrossRef] [PubMed]
- Segura, M.; Alba, R.; Bosch, A.; Chillon, M. Advances in helper-dependent adenoviral vector research. Curr. Gene Ther. 2008, 8, 222–235. [Google Scholar] [CrossRef]
- Brunetti-Pierri, N.; Ng, T.; Iannitti, D.; Cioffi, W.; Stapleton, G.; Law, M.; Breinholt, J.; Palmer, D.; Grove, N.; Rice, K.; et al. Transgene expression up to 7 years in nonhuman primates following hepatic transduction with helper-dependent adenoviral vectors. Hum. Gene Ther. 2013, 24, 761–765. [Google Scholar] [CrossRef] [PubMed]
- Sandig, V.; Youil, R.; Bett, A.J.; Franlin, L.L.; Oshima, M.; Maione, D.; Wang, F.; Metzker, M.L.; Savino, R.; Caskey, C.T. Optimization of the helper-dependent adenovirus system for production and potency in vivo. Proc. Natl. Acad. Sci. USA 2000, 97, 1002–1007. [Google Scholar] [CrossRef]
- Fisher, K.J.; Kelley, W.M.; Burda, J.F.; Wilson, J.M. A novel adenovirus-adeno-associated virus hybrid vector that displays efficient rescue and delivery of the AAV genome. Hum. Gene Ther. 1996, 7, 2079–2087. [Google Scholar] [CrossRef]
- Hardy, S.; Kitamura, M.; Harris-Stansil, T.; Dai, Y.; Phipps, M.L. Construction of adenovirus vectors through Cre-lox recombination. J. Virol. 1997, 71, 1842–1849. [Google Scholar] [CrossRef]
- Hartigan-O’Connor, D.; Amalfitano, A.; Chamberlain, J.S. Improved production of gutted adenovirus in cells expressing adenovirus preterminal protein and DNA polymerase. J. Virol. 1999, 73, 7835–7841. [Google Scholar] [CrossRef]
- Morral, N.; Parks, R.J.; Zhou, H.; Langston, C.; Schiedner, G.; Quinones, J.; Graham, F.L.; Kochanek, S.; Beaudet, A.L. High doses of a helper-dependent adenoviral vector yield supraphysiological levels of α1-antitrypsin with negligible toxicity. Hum. Gene Ther. 1998, 9, 2709–2716. [Google Scholar] [CrossRef]
- Schiedner, G.; Morral, N.; Parks, R.J.; Wu, Y.; Koopmans, S.C.; Langston, C.; Graham, F.L.; Beaudet, A.L.; Kochanek, S. Genomic DNA transfer with a high-capacity adenovirus vector results in improved in vivo gene expression and decreased toxicity. Nat. Genet. 1998, 18, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Brunetti-Pierri, N.; Palmer, D.J.; Mane, V.; Finegold, M.; Beaudet, A.L.; Ng, P. Increased hepatic transduction with reduced systemic dissemination and proinflammatory cytokines following hydrodynamic injection of helper-dependent adenoviral vectors. Mol. Ther. 2005, 12, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Brunetti-Pierri, N.; Liou, A.; Patel, P.; Palmer, D.; Grove, N.; Finegold, M.; Piccolo, P.; Donnachie, E.; Rice, K.; Beaudet, A.; et al. Balloon catheter delivery of helper-dependent adenoviral vector results in sustained, therapeutic hFIX expression in rhesus macaques. Mol. Ther. 2012, 20, 1863–1870. [Google Scholar] [CrossRef]
- Ehrhardt, A.; Xu, H.; Dillow, A.M.; Bellinger, D.A.; Nichols, T.C.; Kay, M.A. A gene-deleted adenoviral vector results in phenotypic correction of canine hemophilia B without liver toxicity or thrombocytopenia. Blood 2003, 102, 2403–2411. [Google Scholar] [CrossRef]
- Top, F.H.; Buescher, E.L.; Bancroft, W.H.; Russell, P.K. Immunization with live types 7 and 4 adenovirus vaccines.Ii. antibody response and protective effect against acute respiratory disease due to adenovirus type 7. J. Infect. Dis. 1971, 124, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Deal, C.; Pekosz, A.; Ketner, G. Prospects for oral replicating adenovirus-vectored vaccines. Vaccine 2013, 31, 3236–3243. [Google Scholar] [CrossRef]
- Radin, J.M.; Hawksworth, A.W.; Blair, P.J.; Faix, D.J.; Raman, R.; Russell, K.L.; Gray, G.C. Dramatic decline of respiratory illness among us military recruits after the renewed use of adenovirus vaccines. Clin. Infect. Dis. 2014, 59, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Broderick, M.; Myers, C.; Balansay, M.; Vo, S.; Osuna, A.; Russell, K. Adenovirus 4/7 vaccine’s effect on disease rates is associated with disappearance of adenovirus on building surfaces at a military recruit base. Mil. Med. 2017, 182, e2069–e2072. [Google Scholar] [CrossRef] [PubMed]
- Home—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ (accessed on 11 May 2021).
- Sullivan, N.J.; Sanchez, A.; Rollin, P.E.; Yang, Z.Y.; Nabel, G.J. Development of a preventive vaccine for Ebola virus infection in primates. Nature 2000, 408, 605–609. [Google Scholar] [CrossRef]
- Ewer, K.; Sebastian, S.; Spencer, A.J.; Gilbert, S.; Hill, A.V.S.; Lambe, T. Chimpanzee adenoviral vectors as vaccines for outbreak pathogens. Hum. Vaccines Immunother. 2017, 13, 3020–3032. [Google Scholar] [CrossRef]
- Priddy, F.H.; Brown, D.; Kublin, J.; Monahan, K.; Wright, D.P.; Lalezari, J.; Santiago, S.; Marmor, M.; Lally, M.; Novak, R.M.; et al. Safety and immunogenicity of a replication-incompetent adenovirus type 5 HIV-1 clade B gag/pol/nef vaccine in healthy adults. Clin. Infect. Dis. 2008, 46, 1769–1781. [Google Scholar] [CrossRef]
- Shiver, J.W.; Emini, E.A. Recent advances in the development of HIV-1 vaccines using replication-incompetent adenovirus vectors. Annu. Rev. Med. 2004, 55, 355–372. [Google Scholar] [CrossRef]
- Buchbinder, S.P.; Mehrotra, D.V.; Duerr, A.; Fitzgerald, D.W.; Mogg, R.; Li, D.; Gilbert, P.B.; Lama, J.R.; Marmor, M.; del Rio, C.; et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): A double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet 2008, 372, 1881–1893. [Google Scholar] [CrossRef]
- Cohen, J. AIDS research: Promising AIDS vaccine’s failure leaves field reeling. Science 2007, 318, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Press Release: Merck Halts Study of “Ineffective” HIV Vaccine|FierceBiotech. Available online: https://www.fiercebiotech.com/biotech/press-release-merck-halts-study-of-ineffective-hiv-vaccine (accessed on 11 May 2021).
- Harro, C.; Sun, X.; Stek, J.E.; Leavitt, R.Y.; Mehrotra, D.V.; Wang, F.; Bett, A.J.; Casimiro, D.R.; Shiver, J.W.; DiNubile, M.J.; et al. Safety and immunogenicity of the merck adenovirus serotype 5 (MRKAd5) and MRKAd6 human immunodeficiency virus type 1 trigene vaccines alone and in combination in healthy adults. Clin. Vaccine Immunol. 2009, 16, 1285–1292. [Google Scholar] [CrossRef] [PubMed]
- Harro, C.D.; Robertson, M.N.; Lally, M.A.; O’Neill, L.D.; Edupuganti, S.; Goepfert, P.A.; Mulligan, M.J.; Priddy, F.H.; Dubey, S.A.; Kierstead, L.S.; et al. Safety and immunogenicity of adenovirus-vectored near-consensus HIV type 1 clade B gag vaccines in healthy adults. AIDS Res. Hum. Retroviruses 2009, 25, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Patterson, L.J. The “STEP-Wise” Future of adenovirus-based HIV vaccines. Curr. Med. Chem. 2011, 18, 3981–3986. [Google Scholar] [CrossRef]
- Duerr, A.; Huang, Y.; Buchbinder, S.; Coombs, R.W.; Sanchez, J.; del Rio, C.; Casapia, M.; Santiago, S.; Gilbert, P.; Corey, L.; et al. Extended follow-up confirms early vaccine-enhanced risk of HIV acquisition and demonstrates waning effect over time among participants in a randomized trial of recombinant adenovirus HIV vaccine (Step Study). J. Infect. Dis. 2012, 206, 258–266. [Google Scholar] [CrossRef]
- Koblin, B.A.; Mayer, K.H.; Noonan, E.; Wang, C.Y.; Marmor, M.; Sanchez, J.; Brown, S.J.; Robertson, M.N.; Buchbinder, S.P. Sexual risk behaviors, circumcision status, and preexisting immunity to adenovirus type 5 among men who have sex with men participating in a randomized HIV-1 vaccine efficacy trial: Step study. J. Acquir. Immune Defic. Syndr. 2012, 60, 405–413. [Google Scholar] [CrossRef]
- Curlin, M.E.; Cassis-Ghavami, F.; Magaret, A.S.; Spies, G.A.; Duerr, A.; Celum, C.L.; Sanchez, J.L.; Margolick, J.B.; Detels, R.; McElrath, M.J.; et al. Serological immunity to adenovirus serotype 5 is not associated with risk of HIV infection: A case-control study. AIDS 2011, 25, 153–158. [Google Scholar] [CrossRef]
- Stephenson, K.E.; Hural, J.; Buchbinder, S.P.; Sinangil, F.; Barouch, D.H. Preexisting adenovirus seropositivity is not associated with increased HIV-1 acquisition in three HIV-1 vaccine efficacy trials. J. Infect. Dis. 2012, 205, 1806–1810. [Google Scholar] [CrossRef][Green Version]
- Zak, D.E.; Andersen-Nissen, E.; Peterson, E.R.; Sato, A.; Hamilton, M.K.; Borgerding, J.; Krishnamurty, A.T.; Chang, J.T.; Adams, D.J.; Hensley, T.R.; et al. Merck Ad5/HIV induces broad innate immune activation that predicts CD8+ T-cell responses but is attenuated by preexisting Ad5 immunity. Proc. Natl. Acad. Sci. USA 2012, 109, E3503–E3512. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Duerr, A.; Frahm, N.; Zhang, L.; Moodie, Z.; de Rosa, S.; Mcelrath, M.J.; Gilbert, P.B. Immune-correlates analysis of an HIV-1 vaccine efficacy trial reveals an association of nonspecific interferon-C secretion with increased HIV-1 infection risk: A cohort- Based modeling study. PLoS ONE 2014, 9, e108631. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, S.C. Adenovirus-vectored Ebola vaccines. Expert Rev. Vaccines 2015, 14, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
- Vanderzanden, L.; Bray, M.; Fuller, D.; Roberts, T.; Custer, D.; Spik, K.; Jahrling, P.; Huggins, J.; Schmaljohn, A.; Schmaljohn, C. DNA vaccines expressing either the GP or NP genes of Ebola virus protect mice from lethal challenge. Virology 1998, 246, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, N.J.; Geisbert, T.W.; Gelsbert, J.B.; Xu, L.; Yang, Z.Y.; Roederer, M.; Koup, R.A.; Jahrling, P.B.; Nabel, G.J. Accelerated vaccination for Ebola virus haemorrhagic fever in non-human primates. Nature 2003, 424, 681–684. [Google Scholar] [CrossRef]
- Sullivan, N.J.; Geisbert, T.W.; Geisbert, J.B.; Shedlock, D.J.; Xu, L.; Lamoreaux, L.; Custers, J.H.H.V.; Popernack, P.M.; Yang, Z.Y.; Pau, M.G.; et al. Immune protection of nonhuman primates against Ebola virus with single low-dose adenovirus vectors encoding modified GPs. PLoS Med. 2006, 3, e177. [Google Scholar] [CrossRef]
- Ledgerwood, J.E.; Costner, P.; Desai, N.; Holman, L.; Enama, M.E.; Yamshchikov, G.; Mulangu, S.; Hu, Z.; Andrews, C.A.; Sheets, R.A.; et al. A replication defective recombinant Ad5 vaccine expressing Ebola virus GP is safe and immunogenic in healthy adults. Vaccine 2010, 29, 304–313. [Google Scholar] [CrossRef]
- Quinn, K.M.; da Costa, A.; Yamamoto, A.; Berry, D.; Lindsay, R.W.B.; Darrah, P.A.; Wang, L.; Cheng, C.; Kong, W.-P.; Gall, J.G.D.; et al. Comparative analysis of the magnitude, quality, phenotype, and protective capacity of simian immunodeficiency virus gag-specific CD8 + T cells following human-, simian-, and chimpanzee-derived recombinant adenoviral vector immunization. J. Immunol. 2013, 190, 2720–2735. [Google Scholar] [CrossRef]
- Zhu, F.C.; Hou, L.H.; Li, J.X.; Wu, S.P.; Liu, P.; Zhang, G.R.; Hu, Y.M.; Meng, F.Y.; Xu, J.J.; Tang, R.; et al. Safety and immunogenicity of a novel recombinant adenovirus type-5 vector-based Ebola vaccine in healthy adults in China: Preliminary report of a randomised, double-blind, placebo-controlled, phase 1 trial. Lancet 2015, 385, 2272–2279. [Google Scholar] [CrossRef]
- Wu, S.; Kroeker, A.; Wong, G.; He, S.; Hou, L.; Audet, J.; Wei, H.; Zhang, Z.; Fernando, L.; Soule, G.; et al. An adenovirus vaccine expressing Ebola virus variant makona glycoprotein is efficacious in Guinea pigs and nonhuman primates. J. Infect. Dis. 2016, 214, S326–S332. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhang, Z.; Gao, H.; Li, Y.; Hou, L.; Yao, H.; Wu, S.; Liu, J.; Wang, L.; Zhai, Y.; et al. Open-label phase I clinical trial of Ad5-EBOV in Africans in China. Hum. Vaccines Immunother. 2017, 13, 2078–2085. [Google Scholar] [CrossRef]
- Geisbert, T.W.; Bailey, M.; Hensley, L.; Asiedu, C.; Geisbert, J.; Stanley, D.; Honko, A.; Johnson, J.; Mulangu, S.; Pau, M.G.; et al. Recombinant adenovirus serotype 26 (Ad26) and Ad35 vaccine vectors bypass immunity to Ad5 and protect nonhuman primates against ebolavirus challenge. J. Virol. 2011, 85, 4222–4233. [Google Scholar] [CrossRef] [PubMed]
- Milligan, I.D.; Gibani, M.M.; Sewell, R.; Clutterbuck, E.A.; Campbell, D.; Plested, E.; Nuthall, E.; Voysey, M.; Silva-Reyes, L.; McElrath, M.J.; et al. Safety and immunogenicity of novel adenovirus type 26-and modified vaccinia Ankara-vectored Ebola vaccines: A randomized clinical trial. JAMA J. Am. Med. Assoc. 2016, 315, 1610–1623. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, R.; Agrawal, B. Adenoviral vector-based vaccines and gene therapies: Current status and future prospects. In Adenoviruses; IntechOpen: London, UK, 2019. [Google Scholar]
- Matz, K.M.; Marzi, A.; Feldmann, H. Ebola vaccine trials: Progress in vaccine safety and immunogenicity. Expert Rev. Vaccines 2019, 18, 1229–1242. [Google Scholar] [CrossRef] [PubMed]
- Kobinger, G.P.; Feldmann, H.; Zhi, Y.; Schumer, G.; Gao, G.; Feldmann, F.; Jones, S.; Wilson, J.M. Chimpanzee adenovirus vaccine protects against Zaire Ebola virus. Virology 2006, 346, 394–401. [Google Scholar] [CrossRef]
- Stanley, D.A.; Honko, A.N.; Asiedu, C.; Trefry, J.C.; Lau-Kilby, A.W.; Johnson, J.C.; Hensley, L.; Ammendola, V.; Abbate, A.; Grazioli, F.; et al. Chimpanzee adenovirus vaccine generates acute and durable protective immunity against ebolavirus challenge. Nat. Med. 2014, 20, 1126–1129. [Google Scholar] [CrossRef]
- Ledgerwood, J.E.; DeZure, A.D.; Stanley, D.A.; Coates, E.E.; Novik, L.; Enama, M.E.; Berkowitz, N.M.; Hu, Z.; Joshi, G.; Ploquin, A.; et al. Chimpanzee adenovirus vector ebola vaccine. N. Engl. J. Med. 2017, 376, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Ewer, K.; Rampling, T.; Venkatraman, N.; Bowyer, G.; Wright, D.; Lambe, T.; Imoukhuede, E.B.; Payne, R.; Fehling, S.K.; Strecker, T.; et al. A monovalent chimpanzee adenovirus Ebola vaccine boosted with MVA. N. Engl. J. Med. 2016, 374, 1635–1646. [Google Scholar] [CrossRef]
- Kennedy, S.B.; Bolay, F.; Kieh, M.; Grandits, G.; Badio, M.; Ballou, R.; Eckes, R.; Feinberg, M.; Follmann, D.; Grund, B.; et al. Phase 2 placebo-controlled trial of two vaccines to prevent Ebola in Liberia. N. Engl. J. Med. 2017, 377, 1438–1447. [Google Scholar] [CrossRef] [PubMed]
- Mu, J.; Jeyanathan, M.; Shaler, C.R.; Horvath, C.; Damjanovic, D.; Zganiacz, A.; Kugathasan, K.; McCormick, S.; Xing, Z. Respiratory mucosal immunization with adenovirus gene transfer vector induces helper CD4 T cell-independent protective immunity. J. Gene Med. 2010, 12, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Forbes, E.K.; Sander, C.; Ronan, E.O.; McShane, H.; Hill, A.V.S.; Beverley, P.C.L.; Tchilian, E.Z. Multifunctional, high-level cytokine-producing Th1 cells in the lung, but not spleen, correlate with protection against mycobacterium tuberculosis aerosol challenge in mice. J. Immunol. 2008, 181, 4955–4964. [Google Scholar] [CrossRef]
- Wang, J.; Thorson, L.; Stokes, R.W.; Santosuosso, M.; Huygen, K.; Zganiacz, A.; Hitt, M.; Xing, Z. Single mucosal, but not parenteral, immunization with recombinant adenoviral-based vaccine provides potent protection from pulmonary tuberculosis. J. Immunol. 2004, 173, 6357–6365. [Google Scholar] [CrossRef]
- Smaill, F.; Jeyanathan, M.; Smieja, M.; Medina, M.F.; Thanthrige-Don, N.; Zganiacz, A.; Yin, C.; Heriazon, A.; Damjanovic, D.; Puri, L.; et al. A human type 5 adenovirus-based tuberculosis vaccine induces robust T cell responses in humans despite preexisting anti-adenovirus immunity. Sci. Transl. Med. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Raviprakash, K.; Wang, D.; Ewing, D.; Holman, D.H.; Block, K.; Woraratanadharm, J.; Chen, L.; Hayes, C.; Dong, J.Y.; Porter, K. A tetravalent dengue vaccine based on a complex adenovirus vector provides significant protection in rhesus monkeys against all four serotypes of dengue virus. J. Virol. 2008, 82, 6927–6934. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, S.; Khanna, N.; Swaminathan, S. Replication-defective adenoviral vaccine vector for the induction of immune responses to dengue virus type 2. J. Virol. 2003, 77, 12907–12913. [Google Scholar] [CrossRef]
- Khanam, S.; Pilankatta, R.; Khanna, N.; Swaminathan, S. An adenovirus type 5 (AdV5) vector encoding an envelope domain III-based tetravalent antigen elicits immune responses against all four dengue viruses in the presence of prior AdV5 immunity. Vaccine 2009, 27, 6011–6021. [Google Scholar] [CrossRef]
- Hung, P.P.; Chanda, P.K.; Natuk, R.J.; Mason, B.B.; Chengalvala, M.; Bhat, B.M.; Molnar-Kimber, K.L.; Dheer, S.K.; Morin, J.E.; Mizutani, S.; et al. Adenovirus vaccine strains genetically engineered to express HIV-1 or HBV antigens for use as live recombinant vaccines. Nat. Immun. Cell Growth Regul. 1990, 9, 160–164. [Google Scholar]
- Lubeck, M.D.; Davis, A.R.; Chengalvala, M.; Natuk, R.J.; Morin, J.E.; Molnar-Kimber, K.; Mason, B.B.; Bhat, B.M.; Mizutani, S.; Hung, P.P.; et al. Immunogenicity and efficacy testing in chimpanzees of an oral hepatitis B vaccine based on live recombinant adenovirus. Proc. Natl. Acad. Sci. USA 1989, 86, 6763–6767. [Google Scholar] [CrossRef]
- Makimura, M.; Miyake, S.; Akino, N.; Takamori, K.; Matsuura, Y.; Miyamura, T.; Saito, I. Induction of antibodies against structural proteins of hepatitis C virus in mice using recombinant adenovirus. Vaccine 1996, 14, 28–34. [Google Scholar] [CrossRef]
- Park, S.H.; Yang, S.H.; Lee, C.G.; Youn, J.W.; Chang, J.; Sung, Y.C. Efficient induction of T helper 1 CD4+ T-cell responses to hepatitis C virus core and E2 by a DNA prime-adenovirus boost. Vaccine 2003, 21, 4555–4564. [Google Scholar] [CrossRef]
- Matsui, M.; Moriya, O.; Akatsuka, T. Enhanced induction of hepatitis C virus-specific cytotoxic T lymphocytes and protective efficacy in mice by DNA vaccination followed by adenovirus boosting in combination with the interleukin-12 expression plasmid. Vaccine 2003, 21, 1629–1639. [Google Scholar] [CrossRef]
- Arribillaga, L.; de Cerio, A.L.D.; Sarobe, P.; Casares, N.; Gorraiz, M.; Vales, A.; Bruna-Romero, O.; Borrás-Cuesta, F.; Paranhos-Baccala, G.; Prieto, J.; et al. Vaccination with an adenoviral vector encoding hepatitis C virus (HCV) NS3 protein protects against infection with HCV-recombinant vaccinia virus. Vaccine 2002, 21, 202–210. [Google Scholar] [CrossRef]
- Seong, Y.R.; Choi, S.; Lim, J.S.; Lee, C.H.; Lee, C.K.; Im, D.S. Immunogenicity of the E1E2 proteins of hepatitis C virus expressed by recombinant adenoviruses. Vaccine 2001, 19, 2955–2964. [Google Scholar] [CrossRef]
- Prevec, L.; Campbell, J.B.; Christie, B.S.; Belbeck, L.; Graham, F.L. A recombinant human adenovirus vaccine against rabies. J. Infect. Dis. 1990, 161, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Vos, A.; Neubert, A.; Pommerening, E.; Müller, T.; Döhner, L.; Neubert, L.; Hughes, K. Immunogenicity of an E1-deleted recombinant human adenovirus against rabies by different routes of administration. J. Gen. Virol. 2001, 82, 2191–2197. [Google Scholar] [CrossRef] [PubMed]
- Bouet-Cararo, C.; Contreras, V.; Fournier, A.; Jallet, C.; Guibert, J.M.; Dubois, E.; Thiery, R.; Bréard, E.; Tordo, N.; Richardson, J.; et al. Canine adenoviruses elicit both humoral and cell-mediated immune responses against rabies following immunisation of sheep. Vaccine 2011, 29, 1304–1310. [Google Scholar] [CrossRef]
- Xiang, Z.Q.; Greenberg, L.; Ertl, H.C.; Rupprecht, C.E. Protection of non-human primates against rabies with an adenovirus recombinant vaccine. Virology 2014, 450–451, 243–249. [Google Scholar] [CrossRef]
- Van Kampen, K.R.; Shi, Z.; Gao, P.; Zhang, J.; Foster, K.W.; Chen, D.T.; Marks, D.; Elmets, C.A.; Tang, D.C.C. Safety and immunogenicity of adenovirus-vectored nasal and epicutaneous influenza vaccines in humans. Vaccine 2005, 23, 1029–1036. [Google Scholar] [CrossRef]
- Peters, W.; Brandl, J.R.; Lindbloom, J.D.; Martinez, C.J.; Scallan, C.D.; Trager, G.R.; Tingley, D.W.; Kabongo, M.L.; Tucker, S.N. Oral administration of an adenovirus vector encoding both an avian influenza A hemagglutinin and a TLR3 ligand induces antigen specific granzyme B and IFN-γ T cell responses in humans. Vaccine 2013, 31, 1752–1758. [Google Scholar] [CrossRef]
- Gurwith, M.; Lock, M.; Taylor, E.M.; Ishioka, G.; Alexander, J.; Mayall, T.; Ervin, J.E.; Greenberg, R.N.; Strout, C.; Treanor, J.J.; et al. Safety and immunogenicity of an oral, replicating adenovirus serotype 4 vector vaccine for H5N1 influenza: A randomised, double-blind, placebo-controlled, phase 1 study. Lancet Infect. Dis. 2013, 13, 238–250. [Google Scholar] [CrossRef]
- Gao, W.; Soloff, A.C.; Lu, X.; Montecalvo, A.; Nguyen, D.C.; Matsuoka, Y.; Robbins, P.D.; Swayne, D.E.; Donis, R.O.; Katz, J.M.; et al. Protection of mice and poultry from lethal H5N1 avian influenza virus through adenovirus-based immunization. J. Virol. 2006, 80, 1959–1964. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhou, D. Adenoviral vector-based strategies against infectious disease and cancer. Hum. Vaccines Immunother. 2016, 12, 2064–2074. [Google Scholar] [CrossRef] [PubMed]
- Ebola Virus Disease Democratic Republic of Congo: External Situation Report 94/2019. Available online: https://www.who.int/publications/i/item/10665-332654 (accessed on 12 May 2021).
- Der Li, Y.; Chi, W.Y.; Su, J.H.; Ferrall, L.; Hung, C.F.; Wu, T.C. Coronavirus vaccine development: From SARS and MERS to COVID-19. J. Biomed. Sci. 2020, 27, 104. [Google Scholar]
- Dai, L.; Gao, G.F. Viral targets for vaccines against COVID-19. Nat. Rev. Immunol. 2021, 21, 73–82. [Google Scholar] [CrossRef]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef]
- Zhu, F.C.; Li, Y.H.; Guan, X.H.; Hou, L.H.; Wang, W.J.; Li, J.X.; Wu, S.P.; Wang, B.S.; Wang, Z.; Wang, L.; et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: A dose-escalation, open-label, non-randomised, first-in-human trial. Lancet 2020, 395, 1845–1854. [Google Scholar] [CrossRef]
- Mercado, N.B.; Zahn, R.; Wegmann, F.; Loos, C.; Chandrashekar, A.; Yu, J.; Liu, J.; Peter, L.; McMahan, K.; Tostanoski, L.H.; et al. Single-shot Ad26 vaccine protects against SARS-CoV-2 in rhesus macaques. Nature 2020, 586, 583–588. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Lambe, T.; Spencer, A.; Belij-Rammerstorfer, S.; Purushotham, J.N.; Port, J.R.; Avanzato, V.A.; Bushmaker, T.; Flaxman, A.; Ulaszewska, M.; et al. ChAdOx1 nCoV-19 vaccine prevents SARS-CoV-2 pneumonia in rhesus macaques. Nature 2020, 586, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Graham, S.P.; McLean, R.K.; Spencer, A.J.; Belij-Rammerstorfer, S.; Wright, D.; Ulaszewska, M.; Edwards, J.C.; Hayes, J.W.P.; Martini, V.; Thakur, N.; et al. Evaluation of the immunogenicity of prime-boost vaccination with the replication-deficient viral vectored COVID-19 vaccine candidate ChAdOx1 nCoV-19. Vaccines 2020, 5. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatullin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G.; et al. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: Two open, non-randomised phase 1/2 studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Shcheblyakov, D.V.; Tukhvatulin, A.I.; Zubkova, O.V.; Dzharullaeva, A.S.; Kovyrshina, A.V.; Lubenets, N.L.; Grousova, D.M.; Erokhova, A.S.; et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: An interim analysis of a randomised controlled phase 3 trial in Russia. Lancet 2021, 397, 671–681. [Google Scholar] [CrossRef]
- Lanzi, A.; Youssef, G.B.; Perricaudet, M.; Benihoud, K. Anti-adenovirus humoral responses influence on the efficacy of vaccines based on epitope display on adenovirus capsid. Vaccine 2011, 29, 1463–1471. [Google Scholar] [CrossRef] [PubMed]
- Vujadinovic, M.; Vellinga, J. Progress in adenoviral capsid-display vaccines. Biomedicines 2018, 6, 81. [Google Scholar] [CrossRef]
- Barouch, D.H.; Kik, S.V.; Weverling, G.J.; Dilan, R.; King, S.L.; Maxfield, L.F.; Clark, S.; Ng’ang’a, D.; Brandariz, K.L.; Abbink, P.; et al. International seroepidemiology of adenovirus serotypes 5, 26, 35, and 48 in pediatric and adult populations. Vaccine 2011, 29, 5203–5209. [Google Scholar] [CrossRef]
- Thorner, A.R.; Lemckert, A.A.C.; Goudsmit, J.; Lynch, D.M.; Ewald, B.A.; Denholtz, M.; Havenga, M.J.E.; Barouch, D.H. Immunogenicity of heterologous recombinant adenovirus prime-boost vaccine regimens is enhanced by circumventing vector cross-reactivity. J. Virol. 2006, 80, 12009–12016. [Google Scholar] [CrossRef]
- Wang, D.; Chen, L.; Ding, Y.; Zhang, J.; Hua, J.; Geng, Q.; Ya, X.; Zeng, S.; Wu, J.; Jiang, Y.; et al. Viral etiology of medically attended influenza-like illnesses in children less than five years old in Suzhou, China, 2011–2014. J. Med. Virol. 2016, 88, 1334–1340. [Google Scholar] [CrossRef]
- Ma, J.; Duffy, M.R.; Deng, L.; Dakin, R.S.; Uil, T.; Custers, J.; Kelly, S.M.; McVey, J.H.; Nicklin, S.A.; Baker, A.H. Manipulating adenovirus hexon hypervariable loops dictates immune neutralisation and coagulation factor X-dependent cell interaction in vitro and in vivo. PLoS Pathog. 2015, 11, e1004673. [Google Scholar] [CrossRef]
- Zhang, S.; Huang, W.; Zhou, X.; Zhao, Q.; Wang, Q.; Jia, B. Seroprevalence of neutralizing antibodies to human adenoviruses type-5 and type-26 and chimpanzee adenovirus type-68 in healthy Chinese adults. J. Med. Virol. 2013, 85, 1077–1084. [Google Scholar] [CrossRef]
- McCoy, K.; Tatsis, N.; Korioth-Schmitz, B.; Lasaro, M.O.; Hensley, S.E.; Lin, S.-W.; Li, Y.; Giles-Davis, W.; Cun, A.; Zhou, D.; et al. Effect of preexisting immunity to adenovirus human serotype 5 antigens on the immune responses of nonhuman primates to vaccine regimens based on human- or chimpanzee-derived adenovirus vectors. J. Virol. 2007, 81, 6594–6604. [Google Scholar] [CrossRef]
- Moffatt, S.; Hays, J.; Hogenesch, H.; Mittal, S.K. Circumvention of vector-specific neutralizing antibody response by alternating use of human and non-human adenoviruses: Implications in gene therapy. Virology 2000, 272, 159–167. [Google Scholar] [CrossRef]
- Hammond, J.M.; McCoy, R.J.; Jansen, E.S.; Morrissy, C.J.; Hodgson, A.L.M.; Johnson, M.A. Vaccination with a single dose of a recombinant porcine adenovirus expressing the classical swine fever virus gp55 (E2) gene protects pigs against classical swine fever. Vaccine 2000, 18, 1040–1050. [Google Scholar] [CrossRef]
- Wüest, T.; Both, G.W.; Prince, A.M.; Hofmann, C.; Löser, P. Recombinant ovine atadenovirus induces a strong and sustained T cell response against the hepatitis C virus NS3 antigen in mice. Vaccine 2004, 22, 2717–2721. [Google Scholar] [CrossRef]
- Barlan, A.U.; Danthi, P.; Wiethoff, C.M. Lysosomal localization and mechanism of membrane penetration influence nonenveloped virus activation of the NLRP3 inflammasome. Virology 2011, 412, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Defer, C.; Belin, M.T.; Caillet-Boudin, M.L.; Boulanger, P. Human adenovirus-host cell interactions: Comparative study with members of subgroups B and C. J. Virol. 1990, 64, 3661–3673. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, N.; Crystal, R.G.; Leopold, P.L. Adenovirus serotype 7 retention in a late endosomal compartment prior to cytosol escape is modulated by fiber protein. J. Virol. 2001, 75, 1387–1400. [Google Scholar] [CrossRef]
- Iacobelli-Martinez, M.; Nemerow, G.R. Preferential activation of toll-like receptor nine by CD46-utilizing adenoviruses. J. Virol. 2007, 81, 1305–1312. [Google Scholar] [CrossRef]
- Campden, R.I.; Zhang, Y. The role of lysosomal cysteine cathepsins in NLRP3 inflammasome activation. Arch. Biochem. Biophys. 2019, 670, 32–42. [Google Scholar] [CrossRef]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 inflammasome: An overview of mechanisms of activation and regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [PubMed]
- Bossy-Wetzel, E.; Newmeyer, D.D.; Green, D.R. Mitochondrial cytochrome c release in apoptosis occurs upstream of DEVD-specific caspase activation and independently of mitochondrial transmembrane depolarization. EMBO J. 1998, 17, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Kupgan, G.; Hentges, D.C.; Muschinske, N.J.; Picking, W.D.; Picking, W.L.; Ramsey, J.D. The effect of fiber truncations on the stability of adenovirus type 5. Mol. Biotechnol. 2014, 56, 979–991. [Google Scholar] [CrossRef] [PubMed]
- Wiethoff, C.M.; Nemerow, G.R. Adenovirus membrane penetration: Tickling the tail of a sleeping dragon. Virology 2015, 479–480, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Nociari, M.; Ocheretina, O.; Schoggins, J.W.; Falck-Pedersen, E. Sensing infection by adenovirus: Toll-like receptor-independent viral DNA recognition signals activation of the interferon regulatory factor 3 master regulator. J. Virol. 2007, 81, 4145–4157. [Google Scholar] [CrossRef] [PubMed]
- Favier, A.L.; Burmeister, W.P.; Chroboczek, J. Unique physicochemical properties of human enteric Ad41 responsible for its survival and replication in the gastrointestinal tract. Virology 2004, 322, 93–104. [Google Scholar] [CrossRef] [PubMed]
- V’kovski, P.; Gultom, M.; Kelly, J.N.; Steiner, S.; Russeil, J.; Mangeat, B.; Cora, E.; Pezoldt, J.; Holwerda, M.; Kratzel, A.; et al. Disparate temperature-dependent virus-host dynamics for SARS-CoV-2 and SARS-CoV in the human respiratory epithelium. PLoS Biol. 2021, 19, e3001158. [Google Scholar] [CrossRef]
- Brown, M.; Wilson-Friesen, H.L.; Doane, F. A block in release of progeny virus and a high particle-to-infectious unit ratio contribute to poor growth of enteric adenovirus types 40 and 41 in cell culture. J. Virol. 1992, 66, 3198–3205. [Google Scholar] [CrossRef]
- Schoggins, J.W.; Nociari, M.; Philpott, N.; Falck-Pedersen, E. Influence of fiber detargeting on adenovirus-mediated innate and adaptive immune activation. J. Virol. 2005, 79, 11627–11637. [Google Scholar] [CrossRef]
- Yu, X.; Veesler, D.; Campbell, M.G.; Barry, M.E.; Asturias, F.J.; Barry, M.A.; Reddy, V.S. Cryo-EM structure of human adenovirus D26 reveals the conservation of structural organization among human adenoviruses. Sci. Adv. 2017, 3, e1602670. [Google Scholar] [CrossRef]
- Abbink, P.; Lemckert, A.A.C.; Ewald, B.A.; Lynch, D.M.; Denholtz, M.; Smits, S.; Holterman, L.; Damen, I.; Vogels, R.; Thorner, A.R.; et al. Comparative seroprevalence and immunogenicity of six rare serotype recombinant adenovirus vaccine vectors from subgroups B and D. J. Virol. 2007, 81, 4654–4663. [Google Scholar] [CrossRef]
- Peruzzi, D.; Dharmapuri, S.; Cirillo, A.; Bruni, B.E.; Nicosia, A.; Cortese, R.; Colloca, S.; Ciliberto, G.; la Monica, N.; Aurisicchio, L. A novel Chimpanzee serotype-based adenoviral vector as delivery tool for cancer vaccines. Vaccine 2009, 27, 1293–1300. [Google Scholar] [CrossRef]
- Roy, S.; Gao, G.; Clawson, D.S.; Vandenberghe, L.H.; Farina, S.F.; Wilson, J.M. Complete nucleotide sequences and genome organization of four chimpanzee adenoviruses. Virology 2004, 324, 361–372. [Google Scholar] [CrossRef]
- Dicks, M.D.J.; Spencer, A.J.; Coughlan, L.; Bauza, K.; Gilbert, S.C.; Hill, A.V.S.; Cottingham, M.G. Differential immunogenicity between HAdV-5 and chimpanzee adenovirus vector ChAdOx1 is independent of fiber and penton RGD loop sequences in mice. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
- Kremer, E.J. CAR chasing: Canine adenovirus vectors—All bite and no bark? J. Gene Med. 2004, 6, S139–S151. [Google Scholar] [CrossRef] [PubMed]
- Soudais, C.; Laplace-Builhe, C.; Kissa, K.; Kremer, E.J. Preferential transduction of neurons by canine adenovirus vectors and their efficient retrograde transport in vivo. FASEB J. 2001, 15, 2283–2285. [Google Scholar] [CrossRef] [PubMed]
- Perreau, M.; Mennechet, F.; Serratrice, N.; Glasgow, J.N.; Curiel, D.T.; Wodrich, H.; Kremer, E.J. Contrasting effects of human, canine, and hybrid adenovirus vectors on the phenotypical and functional maturation of human dendritic cells: Implications for clinical efficacy. J. Virol. 2007, 81, 3272–3284. [Google Scholar] [CrossRef]
- Soudais, C.; Boutin, S.; Kremer, E.J. Characterization of cis-acting sequences involved in canine adenovirus packaging. Mol. Ther. 2001, 3, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.A.; Russell, W.C. Adenovirus protein-protein interactions: Molecular parameters governing the binding of protein VI to hexon and the activation of the adenovirus 23K protease. J. Gen. Virol. 1995, 76, 1959–1969. [Google Scholar] [CrossRef] [PubMed]
- Hernando-Pérez, M.; Martín-González, N.; Pérez-Illana, M.; Suomalainen, M.; Condezo, G.N.; Ostapchuk, P.; Gallardo, J.; Menéndez, M.; Greber, U.F.; Hearing, P.; et al. Dynamic competition for hexon binding between core protein VII and lytic protein VI promotes adenovirus maturation and entry. Proc. Natl. Acad. Sci. USA 2020, 117, 13699–13707. [Google Scholar] [CrossRef]
- Moyer, C.L.; Besser, E.S.; Nemerow, G.R. A single maturation cleavage site in adenovirus impacts cell entry and capsid assembly. J. Virol. 2016, 90, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Van Rosmalen, M.G.M.; Nemerow, G.R.; Wuite, G.J.L.; Roos, W.H. A single point mutation in precursor protein VI doubles the mechanical strength of human adenovirus. J. Biol. Phys. 2018, 44, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Moyer, C.L.; Nemerow, G.R. Disulfide-bond formation by a single cysteine mutation in adenovirus protein VI impairs capsid release and membrane lysis. Virology 2012, 428, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.; Schellenberger, P.; Vasishtan, D.; Aknin, C.; Austin, S.; Dacheux, D.; Rayne, F.; Siebert, A.; Ruzsics, Z.; Gruenewald, K.; et al. The amphipathic helix of adenovirus capsid protein VI contributes to penton release and postentry sorting. J. Virol. 2015, 89, 2121–2135. [Google Scholar] [CrossRef] [PubMed]
- Moyer, C.L.; Wiethoff, C.M.; Maier, O.; Smith, J.G.; Nemerow, G.R. Functional genetic and biophysical analyses of membrane disruption by human adenovirus. J. Virol. 2011, 85, 2631–2641. [Google Scholar] [CrossRef]
- Takahashi, H. Antigen presentation in vaccine development. Comp. Immunol. Microbiol. Infect. Dis. 2003, 26, 309–328. [Google Scholar] [CrossRef]
- Maier, O.; Marvin, S.A.; Wodrich, H.; Campbell, E.M.; Wiethoff, C.M. Spatiotemporal dynamics of adenovirus membrane rupture and endosomal escape. J. Virol. 2012, 86, 10821–10828. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Gómez-Sintes, R.; Boya, P. Lysosomal membrane permeabilization and cell death. Traffic 2018, 19, 918–931. [Google Scholar] [CrossRef]

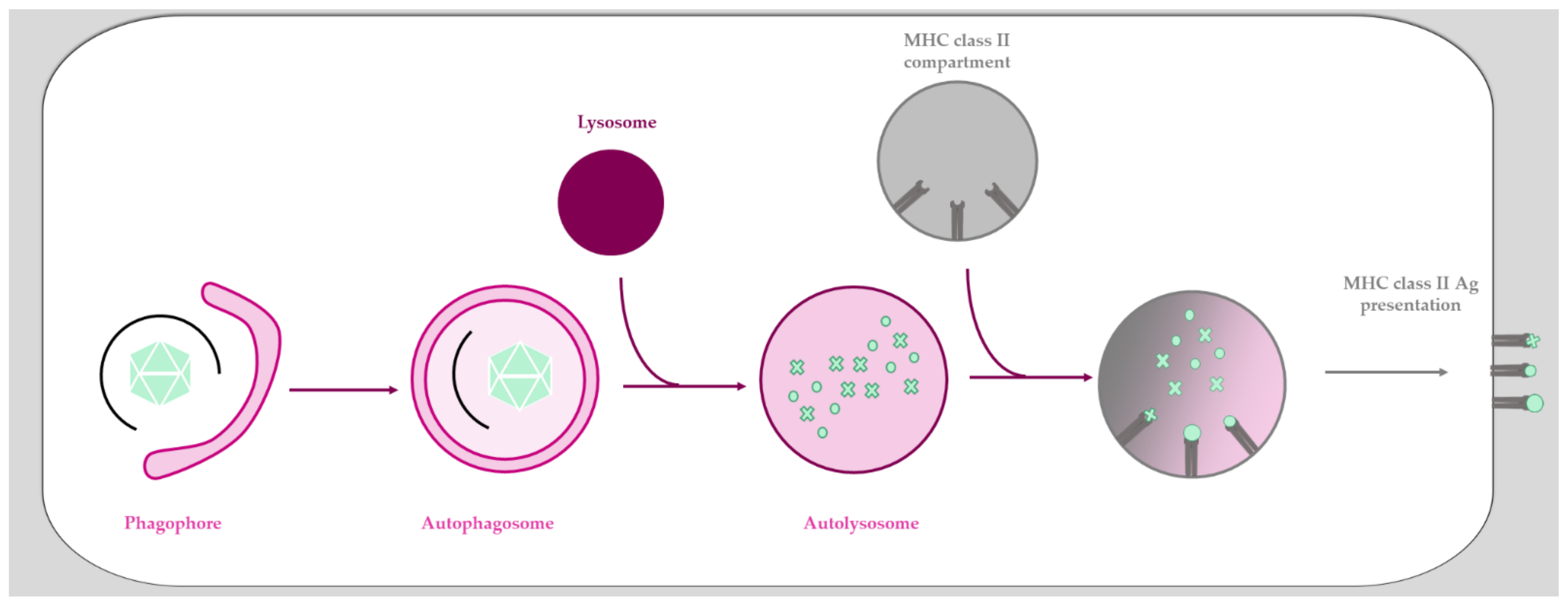
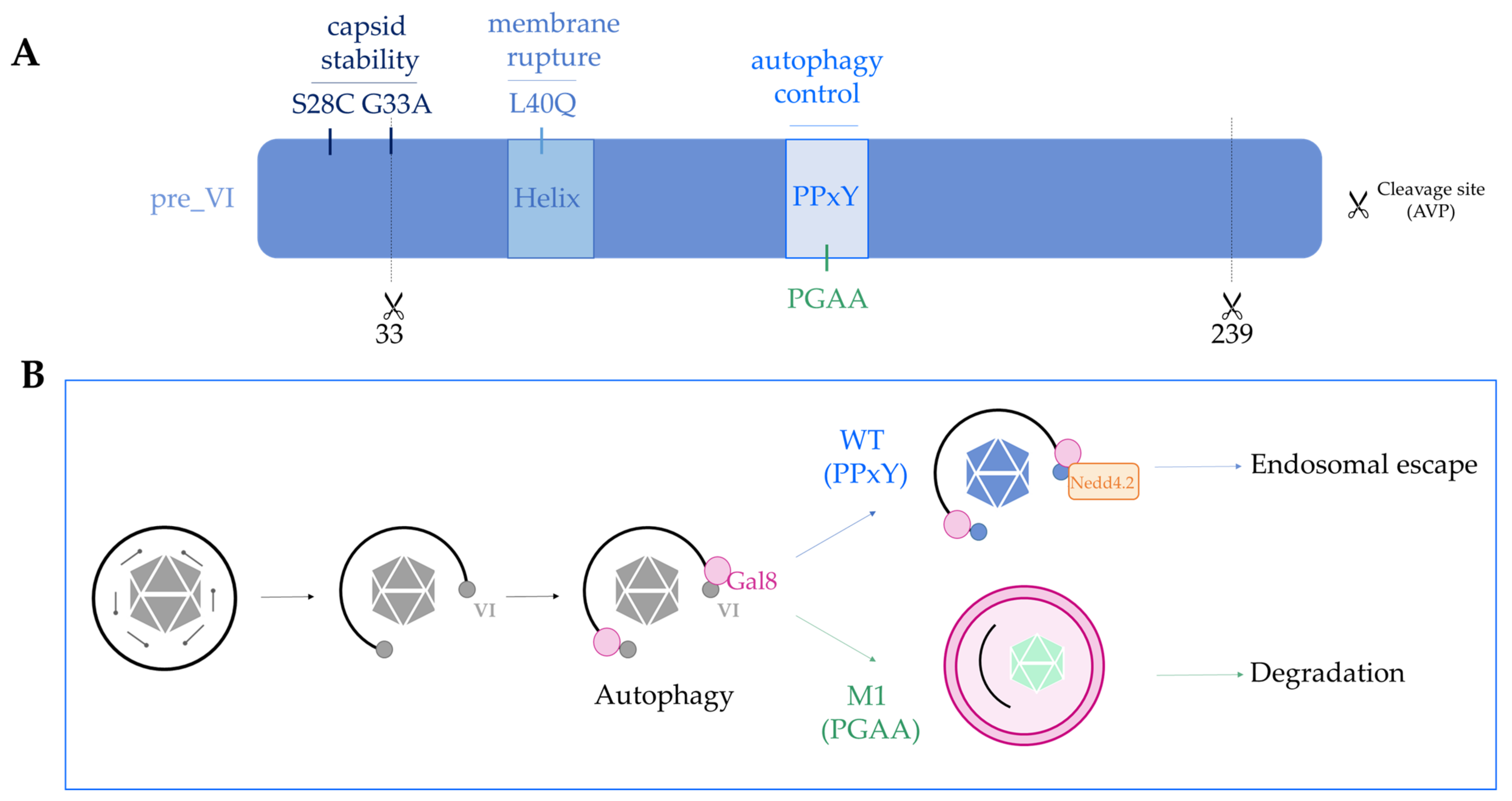
| Species | Type | Tropism | Receptor | Remarks |
|---|---|---|---|---|
| A | 12, 18 and 31 | Intestinal | CAR | |
| B | B1 : 3, 7, 16, 21 and 50 B2 : 11, 14, 34 and 35 | Respiratory and ocular | CD46/DSG-2 | |
| C | 1, 2, 5 and 6 | Respiratory | CAR | Ad5 : used as vaccine against SARS-CoV-2 and in clinical trial against Ebola and HIV-1 (but prematurely stopped) |
| D | 8-10, 13, 15, 17, 19, 20, 22-30, 32, 33, 36-39, 42-49 | Ocular and intestinal | CAR | Ad26 : used vaccine against Ebola and SARS-CoV-2 |
| E | 4 | Respiratory and ocular | CAR | |
| F | 40,41 | Intestinal | CAR | Oral vaccine |
| G | 52 | Intestinal | CAR/sialic acid | |
| Non-human (Chimpanzee) (Canine) | ChAd3 ChAdOx1 CAV-2 | — | CAR | ChAd3 : used as a vaccine in a clinical trial against Ebola ChAdOx1 : used as a vaccine against SARS-CoV-2 CAV-2 : can target neuronal cells |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daussy, C.F.; Pied, N.; Wodrich, H. Understanding Post Entry Sorting of Adenovirus Capsids; A Chance to Change Vaccine Vector Properties. Viruses 2021, 13, 1221. https://doi.org/10.3390/v13071221
Daussy CF, Pied N, Wodrich H. Understanding Post Entry Sorting of Adenovirus Capsids; A Chance to Change Vaccine Vector Properties. Viruses. 2021; 13(7):1221. https://doi.org/10.3390/v13071221
Chicago/Turabian StyleDaussy, Coralie F., Noémie Pied, and Harald Wodrich. 2021. "Understanding Post Entry Sorting of Adenovirus Capsids; A Chance to Change Vaccine Vector Properties" Viruses 13, no. 7: 1221. https://doi.org/10.3390/v13071221
APA StyleDaussy, C. F., Pied, N., & Wodrich, H. (2021). Understanding Post Entry Sorting of Adenovirus Capsids; A Chance to Change Vaccine Vector Properties. Viruses, 13(7), 1221. https://doi.org/10.3390/v13071221






