Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) descriptions of infection and transmission have been increasing in companion animals in the past year. Although canine susceptibility is generally considered low, their role in the COVID-19 disease cycle remains unknown. In this study, we detected and sequenced a delta variant (AY.3) from a 12-year-old Collie living with owners that previously tested positive for SARS-CoV-2. It is unclear if the dogs’ symptoms were related to SARS-CoV-2 infection or underlying conditions. The whole genome sequence obtained from the dog sample had several unique consensus level changes not previously identified in a SARS-CoV-2 genome that may play a role in the rapid adaptation from humans to dogs. Within the spike coding region, 5/7 of the subconsensus variants identified in the dog sequence were also identified in the closest in-house human reference case. Taken together, the whole genome sequence, and phylogenetic and subconsensus variant analyses indicate the virus infecting the animal originated from a local outbreak cluster. The results of these analyses emphasize the importance of rapid detection and characterization of SARS-CoV-2 variants of concern in companion animals.
1. Introduction
In late 2019, a novel severe acute respiratory syndrome coronavirus (SARS-CoV-2) emerged in China and has since become one of the most economically impactful pandemics to date [1]. In the past year, multiple viral variants have been reported, causing various complications including higher rates of transmissibility [2,3,4], immune evasion [5,6,7,8,9], and diagnostic complications [10]. A subset of novel variants has been labeled variants of interest (VOI), on the basis of the preceding factors. VOIs include viruses that are phylogenetically classified into eta (B.1.525), iota (B.1.526), and kappa (B.1.617.1) lineages as well as one unclassified variant (B.1.617.3) [11]. The eta, iota, and kappa lineages were first identified in the United Kingdom, United States, and India, respectively. Following identification, many of these lineages have disseminated worldwide, further perpetuating case rates and clinical symptoms.
The rise of the pandemic and rapid evolution of SARS-CoV-2 started a race to develop a protective and efficacious vaccine, which resulted in development of multiple highly efficacious and safe vaccines [12,13,14]. To date, global vaccination rates are 30.7%, with moderately higher vaccination rates in the United States (54.7%) [15]. Despite the rapid vaccine development and administration efforts, a novel variant (delta lineage; B.1.617.2/AY), capable of evading the vaccinated immune system, originated in India and has since spread globally. Compared to other circulating strains, the delta variants have higher transmissibility and reduced neutralization in post-vaccinated individuals and have consequently been labeled ‘variants of concern’ (VOCs) [4,7]. VOCs are characterized by increased transmissibility, more severe disease outcomes, reduction in neutralization in vaccinated individuals, or failures in diagnostic detection [11]. SARS-CoV-2 variants are classified on the basis of genomic sequencing, specifically related to the Spike (S) protein coding region within the genome. To date, there are four SARS-CoV-2 lineages that are VOCs: alpha (B.1.1.7/Q) [16], beta (B.1.351.1,2,3) [17], delta, and gamma (P.1.) [18]. These lineages were first identified in the United Kingdom, South Africa, India, and Japan/Brazil, respectively.
SARS-CoV-2 is a member of the Betacoronavirus genus within the Coronaviridae family, of which there are five subgenera: Embecovirus, Hibecovirus, Merbecovirus, Nobecovirus, and Sarbecovirus [19]. SARS viruses are the current sole members of the Sarbecovirus subgenus. The SARS-CoV-2 genome is composed of ≈30 kb of positive, single-stranded RNA encoding 12 open reading frames of which the spike (S), membrane (M), nucleocapsid (N), and envelope (E) form the virion [19]. The S protein is the major antigenic protein, binding to the ACE2 host cell receptors for entry and is the focus of V and variant classifications [20,21].
Investigations into potential animal reservoirs of SARS-CoV-2, using the original virus from China, indicated cats and ferrets were permissible to the virus, while dogs, pigs, chickens, and ducks were markedly less susceptible to infection [22,23]. In particular, large cats such as tigers may become infected and demonstrate symptoms of disease [24]. While this is encouraging for pet-owners, studies using the delta variant have shown high levels of RNA and viral shedding from hamsters and Asiatic lions producing mild to moderate clinical signs [25,26]. Markedly, animals infected with the delta variant shed virus for up to 14 days post infection, four days longer than other strains [25]. Serological studies have detected circulating SARS-CoV-2 antibodies in canines, including in 43.9% of dogs in Croatia as well as a single dog in Italy [27,28]. Furthermore, molecular detection and sequencing identified an alpha variant (B.1.1.7) virus from an asymptomatic dog in Spain [29]. Since then, a total of 45 sequences have been obtained from dogs worldwide, 17 of which are in the United States (www.gisaid.org; accessed 25 August 2021). To the authors’ knowledge, no SARS-CoV-2 delta variant sequence from a canine host is currently available.
Here, we document the detection and sequencing of an AY.3 virus from a 12-year-old Collie living with a SARS-CoV-2-infected owner. The animal was admitted to the Kansas State Veterinary Health Center (KSU VHC) for unrelated symptoms and tested positive for SARS-CoV-2 by qRT-PCR two days post-admission. Whole genome sequencing, phylogenetic and subconsenus analyses were performed on the sample confirming transmission from human to animal. These results suggest AY.3 transmission from humans to dogs can occur, potentially affecting the animal at high titers and emphasizes the importance of VOC studies.
2. Materials and Methods
2.1. Quantitative Reverse Transcription PCR (qRT-PCR) for the Detection of SARS-CoV-2 RNA
A nasal swab was collected from a 12-year-old male Collie and submitted to the Kansas State Veterinary Diagnostic Laboratory (KSVDL) for testing. At KSVDL, RNA was extracted from the sample using the QIAamp Viral RNA Mini kit following manufacturer’s specifications. The Path-ID Multiplex One-Step RT-PCR Kit was used in combination with the CDC N1 target primers (Table 1) to test for SARS-CoV-2 RNA. Prepared master mixes were run on an ABI-7500 machine. Results were reported as Ct values.

Table 1.
SARS-CoV-2 qRT-PCR primer sequences.
2.2. SARS-CoV-2 Whole Genome Sequencing
SARS-CoV-2 RNA was amplified from total nucleic acid (see above) using the QIAseq SARS-CoV-2 Primer panel (Qiagen). Primers were removed from the amplicon pools with a 0.9x AMPure XP bead cleanup (Beckman Coulter), diluted, and library prepped using the Nextera XT dual-indexing library prep kit (Illumina). All steps were performed following the manufacturers’ specifications. Libraries were run on an Illumina iSeq 100 using a 300-cycle v2 cartridge.
Raw reads were filtered (at or above Q30) and trimmed for quality and mapped to the Wuhan reference genome (Genbank# NC_045512), and then a consensus sequence was extracted for further analysis. All sequence manipulations were performed in CLC Genomics Workbench v21.0.4 using default parameters (Qiagen).
2.3. Subconsensus Variant Analysis
Subconsensus variants were extracted from the read mapping using CLC Genomics Workbench v 21.0.4 and the following parameters: quality score of 30, 20x coverage, forward/reverse ratio of >0.1, a frequency of 5%, and a significance of 5%. A t-test was used to compare subconsensus variants in Graphpad Prism v9.
2.4. Phylogenetic Analysis
SARS-CoV-2 sequence lineages were determined in Pangolin (https://pangolin.cog-uk.io/) and the Nextclade tool (https://clades.nextstrain.org/; accessed 25 August 2021) assigned clades and were used to perform the phylogenetic analysis. Phylogenetic analysis was performed on delta variant in-house sequences (Supplementary Table S1) obtained from Manhattan, KS. Sequences with >1% Ns in the S gene were excluded from the analysis, resulting in 30 full genomes in the dataset. The sequence alignment was created in mafft v 7.475 [30] using default parameters and was used to build a maximum likelihood tree in FastTree V2.1 [31] with a general time reversible (GTR) model. The best-fit model was determined to be GTR in Mega-X [32]. Trees were formatted in iTOL (https://itol.embl.de/; accessed 25 August 2021).
3. Results
3.1. Case Description
A 12-year-old male Collie was admitted to KSU VHC for collapsing following travel. The dog was diagnosed with a hemoabdomen secondary to a bleeding splenic mass after an abdominal FAST scan and abdomincentesis. Thoracic radiographs were also performed and showed a multilobar alveolar pulmonary pattern with mediastinal shift. Differentials included multilobar pneumonia, atelectasis, or multifocal pulmonary hemorrhage. A splenectomy was performed the day following admission, while in surgery, multifocal hepatic nodules were noted as well as a 5 × 4 cm mass on the left medial liver lobe. A complete liver lobectomy was performed to remove the mass. Pulmonary oximetry performed after surgery was between 88 and 90%, indicating poor perfusion, and therefore the dog was housed in an oxygen cage overnight. Thoracic radiographs were performed the next day, which showed pulmonary changes consistent with progressive aspiration pneumonia and mild cylindrical bronchiectasis. The animal was released 5 days after admission without the requirement for supplemental oxygen. The dog was found dead two days after discharge. No postmortem examination or ancillary testing was performed. The owner had tested positive for SARS-CoV-2 prior to the dog’s admission.
3.2. Molecular Detection and Sequencing
The nasal swab sample collected from the dog following admission to the KSU VHC was tested and confirmed SARS-CoV-2-positive with a Ct of 12.17 two days after admission. The positive nucleic acid was prepped for whole genome sequencing immediately following qRT-PCR confirmation. A total of 1,458,751 reads mapped to the reference genome (Genbank# NC_045512) resulting in an average coverage of 6896x and was designated hCoV-19/dog/USA/KS-8074/2021 (GISAID # EPI_ISL_4253995). A complete SARS-CoV-2 coding region and partial 5′ and 3′UTRs was extracted from the deep sequencing data. The genome is 29,884 nucleotides in length with a GC content of 38.0% and encodes 12 open reading frames of the expected sizes. The genome is 99.96% identical to the next closest genome (an in-house sample, hCoV-19/USA/KS-KSU-2046/2021, GISAID# EPI_ISL_3693315) equating to 8 nucleotide differences. Of these nucleotide differences (4 in ORF1; 2 in S; 1 in M; 1 in N), three nonsynonymous (NS) changes occurred (1 in ORF1; 1 in S; 1 in N), none of which have been seen in delta viruses previously (Table 2).

Table 2.
Consensus changes in hCoV-19/dog/USA/KS-8074/2021 (GISAID # EPI_ISL_4253995) as compared to the closest reference hCoV-19/USA/KS-KSU-2046/2021 (GISAID# EPI_ISL_3693315). Nucleotide (NT) and amino acid (AA) changes were identified in ORF 1ab, the spike (S), the matrix (M), and the nucleocapsid (N) coding region only. AA position denoted only if the AA was changed from the reference.
3.3. Subconsensus Analysis
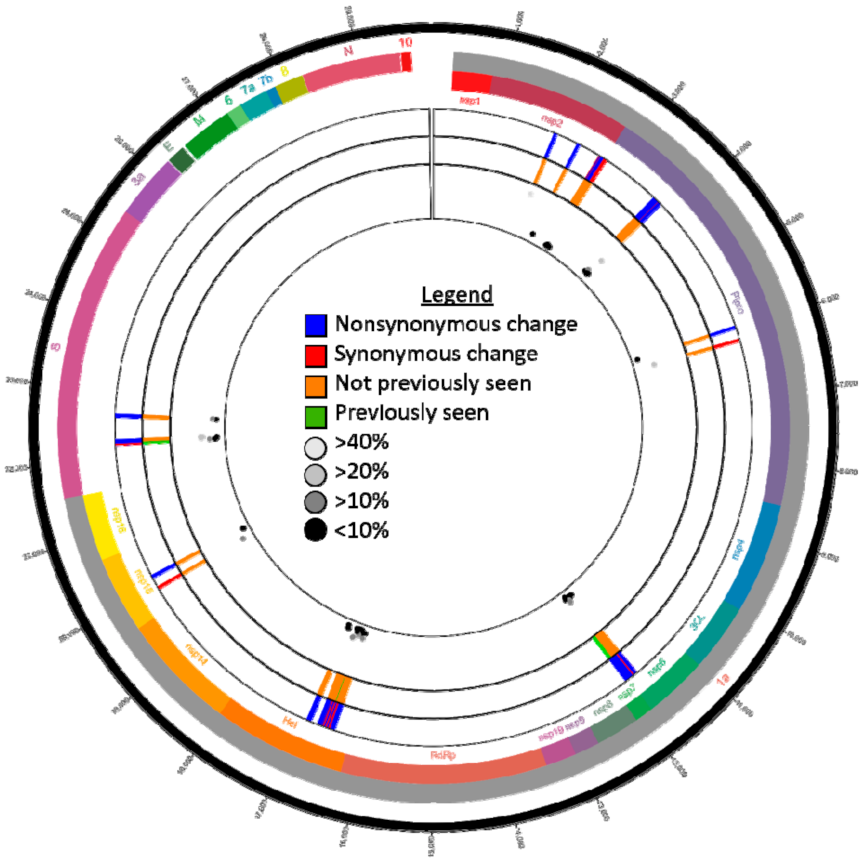
Subconsensus variants occurred in the ORF1ab and S coding regions only (Figure 1). In total, 71 subconsensus variants were identified, 8 were found in the S coding region (7 NS, and 1 Synonymous (S)) and 63 in the ORF1ab coding region (47NS and 16S). Of the spike nonsynonymous variants, 6/7 have never been identified at the consensus level using CoV-GLUE. The single spike variant identified in previous sequences was present at consensus level in the Wuhan reference genome. Interestingly, 5/7 of the NS variants identified in the S gene were also identified above 10% frequency in hCoV-19/USA/KS-KSU-2046/2021 (Figure 1). This trend was not reflected in the ORF 1ab data as only 1/47 NS subconsensus variants were also present in hCoV-19/USA/KS-KSU-2046/2021.
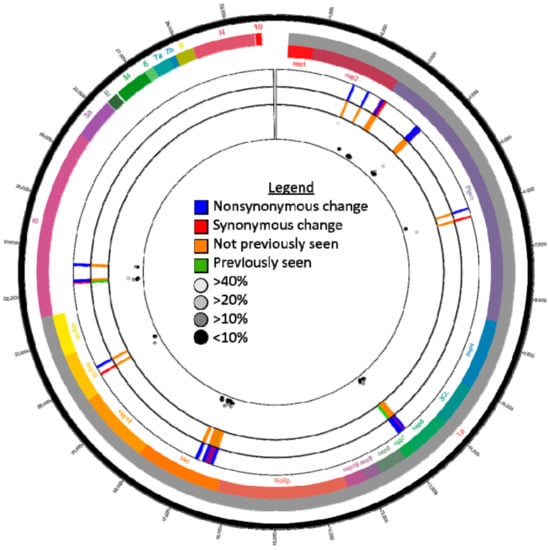
Figure 1.
Subconsensus variants identified in hCoV-19/dog/USA/KS-8074/2021 (GISAID # EPI_ISL_4253995) reads using the following conditions: quality score of 30, 20x coverage, forward/reverse ratio of >0.1, a frequency of 5%, and a significance of 5%. The inner scatter plot displays the frequencies of each variant while the two middle the two concentric circles directly surrounding the scatter plot indicate the subconsensus variant sites and metadata taken from CoV-GLUE (http://cov-glue.cvr.gla.ac.uk/; accessed 28 August 2021).
Subconsensus variants for the Kansas (human) batch of samples were analyzed for trends differing from those that were found in the hCoV-19/dog/USA/KS-8074/2021 sample. Interestingly, the nsp13 coding region within ORF 1b had a significantly greater (p = 0.001) number of subconsensus variants than the Kansas (human) group (37 versus an average of 4.1). No other gene-coding region had a significantly different number of variants than the dog sequence.
3.4. Phylogenetic Analysis and Comparison to Known Sequences
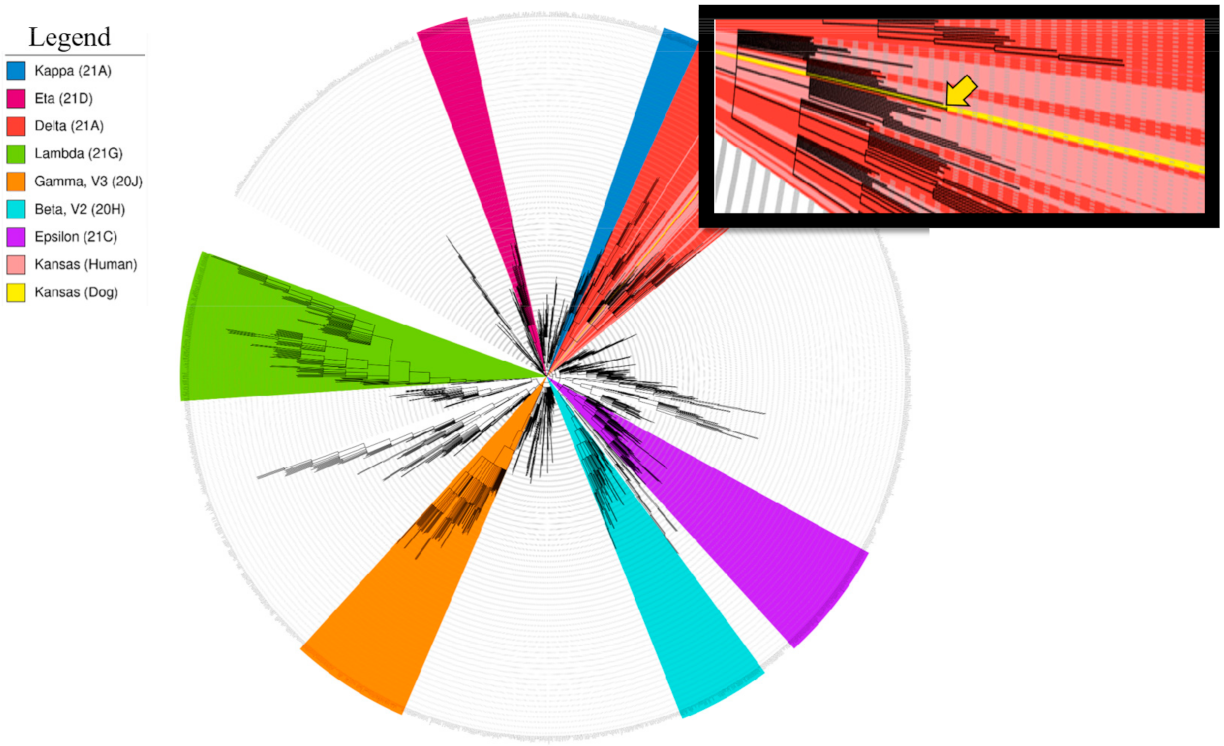
A phylogenetic tree was constructed from hCoV-19/dog/USA/KS-8074/2021 and the Kansas (human) delta variant references using the Nextclade bioinformatics pipeline (locally) (Figure 2). The resulting analysis confirmed hCoV-19/dog/USA/KS-8074/2021 is most similar to our in-house sequence, hCoV-19/USA/KS-KSU-2046/2021, previously collected from a 21-year-old woman over a month prior. All Kansas sequences (human and dog) had 31 total consensus mutations in common as compared to the reference genome from Wuhan (5′UTR, 2; ORF1ab, 13; S, 6; 3a, 1; E, 1; 7ab, 2; 8, 1; N, 4; 3′UTR, 1) ( Supplementary Table S2). Interestingly, the Kansas (human) sequences did not form a monophyletic clade reflecting infection from multiple sources. Kansas (human) sequences are interspersed in clades containing sequences from multiple states, suggesting the Kansas (human) sequences are a result of travel-related transmission events. The cluster of Kansas cases surrounding the dog sequence reflect a potential transmission cluster within the city.
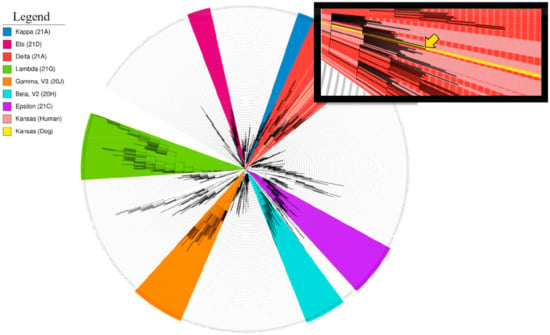
Figure 2.
Phylogeny of hCoV-19/dog/USA/KS-8074/2021. The phylogenetic tree was constructed using the Nextclade pipeline, run on a local server using a GTR model. The tree was modified in iTOL. SARS-CoV-2 lineages are illustrated using the following color scheme: kappa, blue; eta, maroon; delta, red; lambda, green; gamma, orange; beta, turquoise; epsilon, purple. The SARS-CoV-2 genomes obtained from people in Manhattan, KS, are indicated in light red. The SARS-CoV-2 dog genome is indicated in yellow in the main figure and with a yellow arrow in the inset. Variants of concern (VOCs) alpha, beta, gamma, epsilon, and delta lineages.
4. Discussion
To our knowledge, this is the first report of a dog infected with a SARS-CoV-2 delta variant. SARS-CoV-2 transmission from humans to pets has been documented, although infrequently. In this case, it was unclear as to whether the clinical signs post-operation were, in part, a result of the acute SARS-CoV-2 infection or whether the virus contributed to the animal’s death. Although canine susceptibility to SARS-CoV-2 alpha variants has been determined to be generally low [23,33], infectivity of the delta variant has not been tested in dogs. The low Ct value suggests an acute infection that could be a result of a compromised immune system associated with the hepatic and splenic masses, despite the congruence of some of the clinical signs to COVID-19 infection such as pneumonia, bronchiectasis, and lack of oxygenation. Adequate determination is no longer possible.
The high RNA load in the nasal swab suggests the dog may have been shedding virus, causing a risk of infection to surrounding susceptible individuals. To date, a universal protocol for quarantining SARS-CoV-2-positive animals has not been established. The lack of available guidelines is a risk to not only the health of the admitted animals but to clinic staff. Our results indicate steps should be taken to protect the health and environment of veterinary clinics to limit the potential for SARS-CoV-2 transmission.
The patient history coupled with our results confirm SARS-CoV-2 transmission from owner to pet. The owner tested positive prior to admission of the animal to KSU VHC. Transmission from infected owners to pets has been documented for the B.1.1.7 variant in the United States recently in a similar manner as we describe [33]. Phylogenetic and subconsensus analyses also indicate transmission occurred from a cluster of human cases in Manhattan starting more than a month prior to the dog’s positive SARS-CoV-2 test. This is particularly apparent in the S coding region as 5/7 of the NS variants identified in the gene-coding region were also found in the most similar in-house sequence collected over a month prior. Coupled with the high RNA load and potential to shed infectious SARS-CoV-2 virus, this confirms the high transmissibility of this viral lineage.
Coronavirus are known to rapidly evolve following or prior to jumps between species [34,35,36,37]. Our analyses indicate nonsynonymous changes are present in the ORF 1ab, S, and N genes. While these data may not conclude variant involvement in the jump between humans and dogs, the S gene is the target of the host immune system, suggesting the nonsynonymous mutation M1227L may be of particular importance for the delta variant’s rapid adaptation to dogs. The M1227L mutation is located in the S gene on the central hydrophobic portion in the transmembrane domain that functions to stabilize the trimeric structure used for membrane fusion [38,39]. Destabilization of the transmembrane domain within the S gene is associated with reduced viral infectivity and therefore efficiency of infection [40]. This study suggests the M1227L mutation may cause S protein destabilization that could contribute to the transmission of SARS-CoV-2 from humans to dogs.
We speculate that subconsensus variants may also contribute to the transmission between dogs and humans. The non-structural protein 13 in particular had a significantly greater number of subconsensus variants, yet no consensus level variants, in the dog sample as the reference database. This finding is curious considering the helicase is highly conserved across coronavirus family. The significance of this finding is not known.
Monitoring for the emergence of novel variants of SARS-CoV-2 is of critical public health importance. Our findings suggest monitoring may be appropriate and necessary in companion animals as well, especially those that reside in households that have a known SARS-CoV-2 infection. These measures will help to ensure the health of humans and animals in the continued effort to mitigate the risks of SARS-CoV-2.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/v13102104/s1, Table S1: SARS-CoV-2 sequences utilized in this study, Table S2: SARS-CoV-2 Kansas genomic changes. Nucleotide and amino acid changes are indicated.
Author Contributions
Conceptualization, L.N., K.A., J.B. and R.P.; methodology, R.P.; formal analysis, T.D., A.L. and R.P.; data curation, T.D., A.L. and R.P.; writing—original draft preparation, D.U. and R.P.; writing—review and editing, L.N., K.A., J.B., D.U. and R.P. All authors have read and agreed to the published version of the manuscript.
Funding
This work is/was supported by the USDA National Institute of Food and Agriculture, Animal Health and Disease Research project 1026878. Research reported in this publication was supported by the National Institute of General Medical Sciences (NIGMS) of the National Institutes of Health under award number P20GM130448. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Institutional Review Board Statement
The study was conducted according to protocols approved by Kansas State Universities Institutional Biosafety Committee (protocol code 1285; approval date: 27 July 2020).
Informed Consent Statement
Not applicable.
Data Availability Statement
The whole genome sequences for this study are available on GISAID. Deep sequence data for hCoV-19/dog/USA/KS-8074/2021 is available in NCBI’s Sequence Reads Archive under accession number: PRJNA764863, and in GISAID under accession: GISAID # EPI_ISL_4253995.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Davies, N.G.; Abbott, S.; Barnard, R.C.; Jarvis, C.I.; Kucharski, A.J.; Munday, J.D.; Pearson, C.A.B.; Russell, T.W.; Tully, D.C.; Washburne, A.D.; et al. Estimated Transmissibility and Impact of SARS-CoV-2 Lineage B.1.1.7 in England. Science 2021, 372, eabg3055. [Google Scholar] [CrossRef] [PubMed]
- Pearson, C.A.B.; Russell, T.W.; Davies, N.G.; Kucharski, A.J.; Edmunds, W.J.; Eggo, R.M. Estimates of Severity and Transmissibility of Novel South African SARS-CoV-2 Variant 501Y.V2. CMMID Repos. 2021, in press. [Google Scholar]
- Allen, H.; Vusirikala, A.; Flannagan, J.; Twohig, K.A.; Zaidi, A.; COG-UK Consortium; Groves, N.; Lopez-Bernal, J.; Harris, R.; Charlett, A.; et al. Increased Household Transmission of COVID-19 Cases Associated with SARS-CoV-2 Variant of Concern B.1.617.2: A National Casecontrol Study. Public Health Engl. 2021. [Google Scholar]
- Emary, K.R.W.; Golubchik, T.; Aley, P.K.; Ariani, C.V.; Angus, B.; Bibi, S.; Blane, B.; Bonsall, D.; Cicconi, P.; Charlton, S.; et al. Efficacy of ChAdOx1 NCoV-19 (AZD1222) Vaccine against SARS-CoV-2 Variant of Concern 202012/01 (B.1.1.7): An Exploratory Analysis of a Randomised Controlled Trial. Lancet 2021, 397, 1351–1362. [Google Scholar] [CrossRef]
- Wang, P.; Casner, R.G.; Nair, M.S.; Wang, M.; Yu, J.; Cerutti, G.; Liu, L.; Kwong, P.D.; Huang, Y.; Shapiro, L.; et al. Increased Resistance of SARS-CoV-2 Variant P.1 to Antibody Neutralization. Cell Host Microbe 2021, 29, 747–751.e4. [Google Scholar] [CrossRef]
- Deng, X.; Garcia-Knight, M.A.; Khalid, M.M.; Servellita, V.; Wang, C.; Morris, M.K.; Sotomayor-González, A.; Glasner, D.R.; Reyes, K.R.; Gliwa, A.S.; et al. Transmission, Infectivity, and Antibody Neutralization of an Emerging SARS-CoV-2 Variant in California Carrying a L452R Spike Protein Mutation. medRxiv 2021. [Google Scholar] [CrossRef]
- Wu, K.; Werner, A.P.; Moliva, J.I.; Koch, M.; Choi, A.; Stewart-Jones, G.B.E.; Bennett, H.; Boyoglu-Barnum, S.; Shi, W.; Graham, B.S.; et al. MRNA-1273 Vaccine Induces Neutralizing Antibodies against Spike Mutants from Global SARS-CoV-2 Variants. bioRxiv 2021. [Google Scholar] [CrossRef]
- Wang, P.; Nair, M.S.; Liu, L.; Iketani, S.; Luo, Y.; Guo, Y.; Wang, M.; Yu, J.; Zhang, B.; Kwong, P.D.; et al. Antibody Resistance of SARS-CoV-2 Variants B.1.351 and B.1.1.7. Nature 2021, 593, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Bal, A.; Destras, G.; Gaymard, A.; Stefic, K.; Marlet, J.; Eymieux, S.; Regue, H.; Semanas, Q.; d’Aubarede, C.; Billaud, G.; et al. Two-Step Strategy for the Identification of SARS-CoV-2 Variant of Concern 202012/01 and Other Variants with Spike Deletion H69-V70, France, August to December 2020. Eurosurveillance 2021, 26, 2100008. [Google Scholar] [CrossRef]
- CDC Coronavirus Disease 2019 (COVID-19). Available online: https://www.cdc.gov/coronavirus/2019-ncov/variants/variant-info.html (accessed on 21 September 2021).
- Cohen, J. Vaccine Designers Take First Shots at COVID-19. Science 2020, 368, 14–16. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Tenchov, R.; Smoot, J.; Liu, C.; Watkins, S.; Zhou, Q. A Comprehensive Review of the Global Efforts on COVID-19 Vaccine Development. ACS Cent. Sci. 2021, 7, 512–533. [Google Scholar] [CrossRef]
- Fontanet, A.; Autran, B.; Lina, B.; Kieny, M.P.; Karim, S.S.A.; Sridhar, D. SARS-CoV-2 Variants and Ending the COVID-19 Pandemic. Lancet 2021, 397, 952–954. [Google Scholar] [CrossRef]
- Ritchie, H.; Mathieu, E.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Ortiz-Ospina, E.; Hasell, J.; Macdonald, B.; Beltekian, D.; Roser, M. Coronavirus Pandemic (COVID-19). Our World Data. 2020. Available online: https://ourworldindata.org/coronavirus (accessed on 28 August 2021).
- Kirby, T. New Variant of SARS-CoV-2 in UK Causes Surge of COVID-19. Lancet Respir. Med. 2021, 9, e20–e21. [Google Scholar] [CrossRef]
- Happi, A.N.; Ugwu, C.A.; Happi, C.T. Tracking the Emergence of New SARS-CoV-2 Variants in South Africa. Nat. Med. 2021, 27, 372–373. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, C.; Bhattacharya, M.; Sharma, A.R.; Lee, S.-S.; Agoramoorthy, G. SARS-CoV-2 Brazil Variants in Latin America: More Serious Research Urgently Needed on Public Health and Vaccine Protection. Ann. Med. Surg. (Lond.) 2021, 66, 102428. [Google Scholar] [CrossRef]
- de Groot, R.J.; Cowley, J.A.; Enjuanes, L.; Faaberg, K.S.; Perlman, S.; Rottier, P.J.M.; Snijder, E.J.; Ziebuhr, J.; Gorbalenya, A.E. Nidovirales. In International Committee on Taxonomy of Viruses 9th Report; Elsevier: Oxford, UK, 2011. [Google Scholar]
- Sun, K.; Gu, L.; Ma, L.; Duan, Y. Atlas of ACE2 Gene Expression Reveals Novel Insights into Transmission of SARS-CoV-2. Heliyon 2021, 7, e05850. [Google Scholar] [CrossRef]
- Li, M.-Y.; Li, L.; Zhang, Y.; Wang, X.-S. Expression of the SARS-CoV-2 Cell Receptor Gene ACE2 in a Wide Variety of Human Tissues. Infect. Dis. Poverty 2020, 9, 45. [Google Scholar] [CrossRef]
- Poudel, U.; Subedi, D.; Pantha, S.; Dhakal, S. Animal Coronaviruses and Coronavirus Disease 2019: Lesson for One Health Approach. Open Vet. J. 2020, 10, 239–251. [Google Scholar] [CrossRef]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z.; et al. Susceptibility of Ferrets, Cats, Dogs, and Other Domesticated Animals to SARS–Coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [CrossRef] [Green Version]
- McAloose, D.; Laverack, M.; Wang, L.; Killian, M.L.; Caserta, L.C.; Yuan, F.; Mitchell, P.K.; Queen, K.; Mauldin, M.R.; Cronk, B.D.; et al. From People to Panthera: Natural SARS-CoV-2 Infection in Tigers and Lions at the Bronx Zoo. mBio 2020, 11, e02220-20. [Google Scholar] [CrossRef]
- Mohandas, S.; Yadav, P.D.; Shete, A.; Nyayanit, D.; Sapkal, G.; Lole, K.; Gupta, N. SARS-CoV-2 Delta Variant Pathogenesis and Host Response in Syrian Hamsters. Viruses 2021, 13, 1773. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Kumar, N.; Bhatia, S.; Aasdev, A.; Kanniappan, S.; Thayasekhar, A.; Gopinadhan, A.; Silambarasan, R.; Sreekumar, C.; Dubey, C.K.; et al. Natural Infection of SARS-CoV-2 Delta Variant in Asiatic Lions (Panthera Leo Persica) in India. Emerg. Infect. Dis. 2021, in press. [Google Scholar] [CrossRef]
- Stevanovic, V.; Tabain, I.; Vilibic-Cavlek, T.; Mauric Maljkovic, M.; Benvin, I.; Hruskar, Z.; Kovac, S.; Smit, I.; Miletic, G.; Hadina, S.; et al. The Emergence of SARS-CoV-2 within the Dog Population in Croatia: Host Factors and Clinical Outcome. Viruses 2021, 13, 1430. [Google Scholar] [CrossRef]
- Klaus, J.; Zini, E.; Hartmann, K.; Egberink, H.; Kipar, A.; Bergmann, M.; Palizzotto, C.; Zhao, S.; Rossi, F.; Franco, V.; et al. SARS-CoV-2 Infection in Dogs and Cats from Southern Germany and Northern Italy during the First Wave of the COVID-19 Pandemic. Viruses 2021, 13, 1453. [Google Scholar] [CrossRef]
- Barroso-Arévalo, S.; Rivera, B.; Domínguez, L.; Sánchez-Vizcaíno, J.M. First Detection of SARS-CoV-2 B.1.1.7 Variant of Concern in an Asymptomatic Dog in Spain. Viruses 2021, 13, 1379. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [Green Version]
- Price, M.N.; Dehal, P.S.; Arkin, A.P. FastTree 2—Approximately Maximum-Likelihood Trees for Large Alignments. PLoS ONE 2010, 5, e9490. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Hamer, S.A.; Ghai, R.R.; Zecca, I.B.; Auckland, L.D.; Roundy, C.M.; Davila, E.; Busselman, R.E.; Tang, W.; Pauvolid-Corrêa, A.; Killian, M.L.; et al. SARS-CoV-2 B.1.1.7 Variant of Concern Detected in a Pet Dog and Cat after Exposure to a Person with COVID-19, USA. Transbound. Emerg. Dis. 2021. [Google Scholar] [CrossRef]
- Dimonte, S.; Babakir-Mina, M.; Hama-Soor, T.; Ali, S. Genetic Variation and Evolution of the 2019 Novel Coronavirus. PHG 2021, 24, 54–66. [Google Scholar] [CrossRef]
- Chakraborty, C.; Sharma, A.R.; Bhattacharya, M.; Agoramoorthy, G.; Lee, S.-S. Evolution, Mode of Transmission, and Mutational Landscape of Newly Emerging SARS-CoV-2 Variants. mBio 2021, 12, e0114021. [Google Scholar] [CrossRef]
- Jo, W.K.; Drosten, C.; Drexler, J.F. The Evolutionary Dynamics of Endemic Human Coronaviruses. Virus Evol. 2021, 7. [Google Scholar] [CrossRef] [PubMed]
- Dhama, K.; Patel, S.K.; Sharun, K.; Pathak, M.; Tiwari, R.; Yatoo, M.I.; Malik, Y.S.; Sah, R.; Rabaan, A.A.; Panwar, P.K.; et al. SARS-CoV-2 Jumping the Species Barrier: Zoonotic Lessons from SARS, MERS and Recent Advances to Combat This Pandemic Virus. Travel Med. Infect. Dis. 2020, 37, 101830. [Google Scholar] [CrossRef] [PubMed]
- Xia, X. Domains and Functions of Spike Protein in SARS-CoV-2 in the Context of Vaccine Design. Viruses 2021, 13, 109. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yang, C.; Xu, X.; Xu, W.; Liu, S. Structural and Functional Properties of SARS-CoV-2 Spike Protein: Potential Antivirus Drug Development for COVID-19. Acta Pharmacol. Sin. 2020, 41, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Broer, R.; Boson, B.; Spaan, W.; Cosset, F.-L.; Corver, J. Important Role for the Transmembrane Domain of Severe Acute Respiratory Syndrome Coronavirus Spike Protein during Entry. J. Virol. 2006, 80, 1302–1310. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).