Here, There, and Everywhere: The Wide Host Range and Geographic Distribution of Zoonotic Orthopoxviruses
Abstract
1. Poxvirus and Emerging Diseases
2. Orthopoxvirus
2.1. Monkeypox Virus
2.2. Cowpox Virus
2.3. Vaccinia Virus and Related Viruses
3. What Is Next for Monkeypox, Cowpox, and Vaccinia Viruses?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Zoonoses. Available online: https://www.who.int/topics/zoonoses/en/#:~:text=Azoonosisisanydisease,zoonoticinfectionsinnature (accessed on 24 October 2020).
- Woolhouse, M.E.J.; Gowtage-Sequeria, S. Host range and emerging and reemerging pathogens. Emerg. Infect. Dis. 2005. [Google Scholar] [CrossRef] [PubMed]
- Bird, B.H.; Mazet, J.A.K. Detection of Emerging Zoonotic Pathogens: An Integrated One Health Approach. Annu. Rev. Anim. Biosci. 2018. [Google Scholar] [CrossRef]
- Woolhouse, M.; Gaunt, E. Ecological origins of novel human pathogens. Crit. Rev. Microbiol. 2007, 33, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L.H.; Latham, S.M.; Woolhouse, M.E.J. Risk factors for human disease emergence. Philos. Trans. R. Soc. B Biol. Sci. 2001. [Google Scholar] [CrossRef]
- Fenner, F. Adventures with poxviruses of vertebrates. FEMS Microbiol. Rev. 2000, 24, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Moss, B. Poxviridae: The viruses and their replication. In Fields Virology; Fields, B.N., Knipe, D.M., Howley, P.M., Griffin, D.E., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; Volume 2, pp. 2126–2159. ISBN 9781451105636. [Google Scholar]
- Fenner, F. The Poxviruses. In Portraits of Viruses—A History of Virology; Gibbs, A., Ed.; Karger: Basil, Switzerland, 1988; pp. 1–23. [Google Scholar]
- Thèves, C.; Biagini, P.; Crubézy, E. The rediscovery of smallpox. Clin. Microbiol. Infect. 2014, 20, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Ladnyi, I.D.; Breman, J.G. Smallpox eradication: Progress and problems. Dev. Biol. Stand. 1978, 41, 281–290. [Google Scholar]
- Damon, I.K. Poxviruses. In Manual of Clinical Microbiology, 10th ed.; Versalovic, J., Carroll, K., Funke, G., Jorgensen, J., Landry, M., Warnock, D., Eds.; American Society of Microbiology: Washington, DC, USA, 2011; pp. 1647–1658. [Google Scholar]
- Lefkowitz, E.J.; Wang, C.; Upton, C. Poxviruses: Past, present and future. Virus Res. 2006, 117, 105–118. [Google Scholar] [CrossRef]
- Mahy, B.W.J. An overview on the use of a viral pathogen as a bioterrorism agent: Why smallpox? Antiviral Res. 2003, 57, 1–5. [Google Scholar] [CrossRef]
- Gubser, C.; Hué, S.; Kellam, P.; Smith, G.L. Poxvirus genomes: A phylogenetic analysis. J. Gen. Virol. 2004, 85, 105–117. [Google Scholar] [CrossRef]
- Essbauer, S.; Pfeffer, M.; Meyer, H. Zoonotic poxviruses. Vet. Microbiol. 2010, 140, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Shchelkunov, S.N. An Increasing Danger of Zoonotic Orthopoxvirus Infections. PLoS Pathog. 2013, 9, e1003756. [Google Scholar] [CrossRef] [PubMed]
- Khalafalla, A.I.; Abdelazim, F. Human and Dromedary Camel Infection. Vector-Borne Zoonotic Dis. 2017, 17, 281–284. [Google Scholar] [CrossRef]
- Vora, N.M.; Li, Y.; Ph, D.; Geleishvili, M.; Emerson, G.L.; Ph, D.; Khmaladze, E.; Maghlakelidze, G.; Navdarashvili, A. Human Infection with a Zoonotic Orthopoxvirus in the Country of Georgia. N. Engl. J. Med. 2016, 372, 1223–1230. [Google Scholar] [CrossRef] [PubMed]
- Doty, J.B.; Maghlakelidze, G.; Sikharulidze, I.; Tu, S.-L.; Morgan, C.N.; Mauldin, M.R.; Parkadze, O.; Kartskhia, N.; Turmanidze, M.; Matheny, A.M.; et al. Isolation and Characterization of Akhmeta Virus from Wild-Caught Rodents (Apodemus spp.) in Georgia. J. Virol. 2019, 93. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; Guagliardo, S.A.J.; Nakazawa, Y.J.; Doty, J.B.; Mauldin, M.R. Understanding orthopoxvirus host range and evolution: From the enigmatic to the usual suspects. Curr. Opin. Virol. 2018, 28, 108–115. [Google Scholar] [CrossRef]
- Woolhouse, M.E.J.; Taylor, L.H.; Haydon, D.T. Population biology of multihost pathogens. Science 2001, 292, 1109–1112. [Google Scholar] [CrossRef]
- Parrish, C.R.; Holmes, E.C.; Morens, D.M.; Park, E.-C.; Burke, D.S.; Calisher, C.H.; Laughlin, C.A.; Saif, L.J.; Daszak, P. Cross-Species Virus Transmission and the Emergence of New Epidemic Diseases. Microbiol. Mol. Biol. Rev. 2008. [Google Scholar] [CrossRef]
- Werden, S.J.; Rahman, M.M.; McFadden, G. Chapter 3 Poxvirus Host Range Genes. Adv. Virus Res. 2008, 71, 135–171. [Google Scholar] [CrossRef]
- Gallwitz, S.; Schutzbank, T.; Heberling, R.L.; Kalter, S.S.; Galpin, J.E. Smallpox: Residual antibody after vaccination. J. Clin. Microbiol. 2003, 41, 4068–4070. [Google Scholar] [CrossRef]
- Shchelkunov, S.N. Emergence and reemergence of smallpox: The need for development of a new generation smallpox vaccine. Vaccine 2011, 29, D49–D53. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.E.; Slifka, M.K. Retrospective analysis of monkeypox infection. Emerg. Infect. Dis. 2008, 14, 592. [Google Scholar] [CrossRef] [PubMed]
- Doshi, R.H.; Guagliardo, S.A.J.; Doty, J.B.; Babeaux, A.D.; Matheny, A.; Burgado, J.; Townsend, M.B.; Morgan, C.N.; Satheshkumar, P.S.; Ndakala, N. Epidemiologic and ecologic investigations of monkeypox, Likouala Department, Republic of the Congo, 2017. Emerg. Infect. Dis. 2019, 25, 273. [Google Scholar] [CrossRef] [PubMed]
- Huhn, G.D.; Bauer, A.M.; Yorita, K.; Graham, M.B.; Sejvar, J.; Likos, A.; Damon, I.K.; Reynolds, M.G.; Kuehnert, M.J. Clinical characteristics of human monkeypox, and risk factors for severe disease. Clin. Infect. Dis. 2005, 41, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Nakoune, E.; Lampaert, E.; Ndjapou, S.G.; Janssens, C.; Zuniga, I.; Van Herp, M.; Fongbia, J.P.; Koyazegbe, T.D.; Selekon, B.; Komoyo, G.F. A nosocomial outbreak of human monkeypox in the Central African Republic. Open forum Infect. Dis. 2017, 4, ofx168. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 48. 2017. Available online: https://apps.who.int/iris/bitstream/handle/10665/259557/OEW482504122017.pdf?sequence=1 (accessed on 25 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 52. 2017. Available online: https://apps.who.int/iris/bitstream/handle/10665/259794/OEW52-2329122017.pdf?sequence=1 (accessed on 25 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 01. 2020. Available online: https://apps.who.int/iris/bitstream/handle/10665/330353/OEW01-05012020.pdf (accessed on 26 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 37. 2020. Available online: https://apps.who.int/iris/bitstream/handle/10665/334303/OEW37-0713092020.pdf (accessed on 26 September 2020).
- Coras, B.; Eßbauer, S.; Pfeffer, M.; Meyer, H.; Schröder, J.; Stolz, W.; Landthaler, M.; Vogt, T. Cowpox and a cat. Lancet 2005, 365, 446. [Google Scholar] [CrossRef]
- Ninove, L.; Domart, Y.; Vervel, C.; Voinot, C.; Salez, N.; Raoult, D.; Meyer, H.; Capek, I.; Zandotti, C.; Charrel, R.N. Cowpox virus transmission from pet rats to humans, France. Emerg. Infect. Dis. 2009, 15, 781–784. [Google Scholar] [CrossRef]
- Popova, A.Y.; Maksyutov, R.A.; Taranov, O.S.; Tregubchak, T.V.; Zaikovskaya, A.V.; Sergeev, A.A.; Vlashchenko, I.V.; Bodnev, S.A.; Ternovoi, V.A.; Alexandrova, N.S.; et al. Cowpox in a human, Russia, 2015. Epidemiol. Infect. 2017, 145, 755–759. [Google Scholar] [CrossRef]
- Nardin, C.; Dupond, A.S.; Pelletier, F.; Puzenat, E.; Aubin, F. Skin Lesions in a Child after Contact with a Domestic Rat. Clin. Infect. Dis. 2019, 68, 1063–1064. [Google Scholar] [CrossRef]
- Haddadeen, C.; Van Ouwerkerk, M.; Vicek, T.; Fityan, A. A case of cowpox virus infection in the UK occurring in a domestic cat and transmitted to the adult male owner. Br. J. Dermatol. 2020, 19319, 19319. [Google Scholar] [CrossRef]
- Wolfs, T.F.W.; Wagenaar, J.A.; Niesters, H.G.M.; Osterhaus, A.D.M.E. Rat-to-human transmission of cowpox infection. Emerg. Infect. Dis. 2002, 8, 1495. [Google Scholar] [CrossRef]
- Ducournau, C.; Ferrier-Rembert, A.; Ferraris, O.; Joffre, A.; Favier, A.-L.; Flusin, O.; Van Cauteren, D.; Kecir, K.; Auburtin, B.; Védy, S. Concomitant human infections with 2 cowpox virus strains in related cases, France, 2011. Emerg. Infect. Dis. 2013, 19, 1996. [Google Scholar] [CrossRef] [PubMed]
- Vogel, S.; Sárdy, M.; Glos, K.; Korting, H.C.; Ruzicka, T.; Wollenberg, A. The Munich outbreak of cutaneous cowpox infection: Transmission by infected pet rats. Acta Derm. Venereol. 2012, 92, 126–131. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, J.S.; Figueiredo, P.d.O.; Costa, G.B.; De Assis, F.L.; Drumond, B.P.; Da Fonseca, F.G.; Nogueira, M.L.; Kroon, E.G.; de Souza Trindade, G. Vaccinia virus natural infections in Brazil: The good, the bad, and the ugly. Viruses 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, G.; Balamurugan, V.; Prabhu, M.; Yogisharadhya, R.; Bora, D.P.; Gandhale, P.N.; Sankar, M.S.; Kulkarni, A.M.; Singh, R.K.; Bhanuprakash, V. Emerging and re-emerging zoonotic buffalopox infection: A severe outbreak in Kolhapur (Maharashtra), India. Vet Ital 2010, 46, 439–448. [Google Scholar] [PubMed]
- Singh, R.K.; Hosamani, M.; Balamurugan, V.; Bhanuprakash, V.; Rasool, T.J.; Yadav, M.P. Buffalopox: An emerging and re-emerging zoonosis. Anim. Heal. Res. Rev. 2007, 8, 105. [Google Scholar] [CrossRef] [PubMed]
- Gurav, Y.K.; Raut, C.G.; Yadav, P.D.; Tandale, B.V.; Sivaram, A.; Pore, M.D.; Basu, A.; Mourya, D.T.; Mishra, A.C. Buffalopox outbreak in humans and animals in Western Maharashtra, India. Prev. Vet. Med. 2011, 100, 242–247. [Google Scholar] [CrossRef]
- Silva-Fernandes, A.T.; Travassos, C.E.P.F.; Ferreira, J.M.S.; Abrahão, J.S.; de Oliveira Rocha, E.S.; Viana-Ferreira, F.; dos Santos, J.R.; Bonjardim, C.A.; Ferreira, P.C.P.; Kroon, E.G. Natural human infections with Vaccinia virus during bovine vaccinia outbreaks. J. Clin. Virol. 2009, 44, 308–313. [Google Scholar] [CrossRef]
- Nagasse-Sugahara, T.K.; Kisielius, J.J.; Ueda-Ito, M.; Curti, S.P.; Figueiredo, C.A.; Cruz, Á.S.; Silva, M.M.J.; Ramos, C.H.; Silva, M.C.C.; Sakurai, T. Human vaccinia-like virus outbreaks in Sao Paulo and Goias States, Brazil: Virus detection, isolation and identification. Rev. Inst. Med. Trop. Sao Paulo 2004, 46, 315–322. [Google Scholar] [CrossRef]
- Abrahão, J.S.; Campos, R.K.; Trindade, G.S.; da Fonseca, F.G.; Ferreira, P.C.P.; Kroon, E.G. Outbreak of severe zoonotic vaccinia virus infection, Southeastern Brazil. Emerg. Infect. Dis. 2015, 21, 695–698. [Google Scholar] [CrossRef]
- Oliveira, D.B.; Assis, F.L.; Ferreira, P.C.P.; Bonjardim, C.A.; de Souza Trindade, G.; Kroon, E.G.; Abrahão, J.S. Group 1 vaccinia virus zoonotic outbreak in Maranhao State, Brazil. Am. J. Trop. Med. Hyg. 2013, 89, 1142–1145. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.; Kurth, A.; Hessler, F.; Kramp, H.; Gokel, M.; Hoffmann, R.; Kuczka, A.; Nitsche, A. Cowpox virus infection in pet rat owners: Not always immediately recognized. Dtsch. Arztebl. Int. 2009, 106, 329. [Google Scholar] [CrossRef]
- Lu, B.; Cui, L.-B.; Gu, M.-H.; Shi, C.; Sun, C.-W.; Zhao, K.-C.; Bi, J.; Tan, Z.-M.; Guo, X.-L.; Huo, X. Outbreak of Vaccinia Virus Infection from Occupational Exposure, China, 2017. Emerg. Infect. Dis. 2019, 25, 1192. [Google Scholar] [CrossRef] [PubMed]
- Sklenovská, N.; Van Ranst, M. Emergence of Monkeypox as the Most Important Orthopoxvirus Infection in Humans. Front. Public Heal. 2018, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Balamurugan, V.; Bhanuprakash, V.; Venkatesan, G.; Hosamani, M. Emergence and reemergence of vaccinia-like viruses: Global scenario and perspectives. Indian J. Virol. 2012, 23, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Franco-Luiz, A.P.M.; Fagundes-Pereira, A.; Costa, G.B.; Alves, P.A.; Oliveira, D.B.; Bonjardim, C.A.; Ferreira, P.C.P.; de Souza Trindade, G.; Panei, C.J.; Galosi, C.M. Spread of vaccinia virus to cattle herds, Argentina, 2011. Emerg. Infect. Dis. 2014, 20, 1576. [Google Scholar] [CrossRef] [PubMed]
- Franco-Luiz, A.P.M.; Oliveira, D.B.; Pereira, A.F.; Gasparini, M.C.S.; Bonjardim, C.A.; Ferreira, P.C.P.; de Souza Trindade, G.; Puentes, R.; Furtado, A.; Abrahão, J.S. Detection of vaccinia virus in dairy cattle serum samples from 2009, Uruguay. Emerg. Infect. Dis. 2016, 22, 2174. [Google Scholar] [CrossRef]
- Usme-Ciro, J.A.; Paredes, A.; Walteros, D.M.; Tolosa-Pérez, E.N.; Laiton-Donato, K.; del Carmen Pinzón, M.; Petersen, B.W.; Gallardo-Romero, N.F.; Li, Y.; Wilkins, K. Detection and molecular characterization of zoonotic poxviruses circulating in the Amazon region of Colombia, 2014. Emerg. Infect. Dis. 2017, 23, 649. [Google Scholar] [CrossRef]
- Eder, I.; Vollmar, P.; Pfeffer, M.; Naether, P.; Rodloff, A.C.; Meyer, H. Two distinct clinical courses of human cowpox, Germany, 2015. Viruses 2017, 9, 375. [Google Scholar] [CrossRef]
- Likos, A.M.; Sammons, S.A.; Olson, V.A.; Frace, A.M.; Li, Y.; Olsen-rasmussen, M.; Davidson, W.; Galloway, R.; Khristova, M.L.; Reynolds, M.G.; et al. A tale of two clades: Monkeypox viruses. J. Gen. Virol. 2005, 86, 2661–2672. [Google Scholar] [CrossRef]
- Yinka-Ogunleye, A.; Aruna, O.; Dalhat, M.; Ogoina, D.; McCollum, A.; Disu, Y.; Mamadu, I.; Akinpelu, A.; Ahmad, A.; Burga, J.; et al. Outbreak of human monkeypox in Nigeria in 2017–18: A clinical and epidemiological report. Lancet Infect. Dis. 2019, 19, 872–879. [Google Scholar] [CrossRef]
- von Magnus, P.; Andersen, E.K.; Petersen, K.B.; Birch-Andersen, A. A pox-like disease in cynomolgus monkeys. Acta Pathol. Microbiol. Scand. 1959, 46, 156–176. [Google Scholar] [CrossRef]
- Parker, S.; Nuara, A.; Schultz, D.A. Human monkeypox: An emerging zoonotic disease. Future Microbiol. 2007, 2, 17–34. [Google Scholar] [CrossRef] [PubMed]
- Khodakevich, L.; Ježek, Z.; Kinzanzka, K. Isolation of monkeypox virus from wild squirrel infected in nature. Isol. Monkeypox Virus Wild Squirrel Infected Nat. 1986, 98–99. [Google Scholar] [CrossRef]
- Doty, J.B.; Malekani, J.M.; Kalemba, L.N.; Stanley, W.T.; Monroe, B.P.; Nakazawa, Y.U.; Id, M.R.M.; Bakambana, L.; Liyandja, T.; Liyandja, D.; et al. Assessing Monkeypox Virus Prevalence in Small Mammals at the Human—Animal Interface in the Democratic Republic of the Congo. Viruses 2017, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wachtman, L.; Mansfield, K. Viral diseases of nonhuman primates. Nonhum. Primates Biomed. Res. 2012, 1. [Google Scholar] [CrossRef]
- Ellis, C.K.; Carroll, D.S.; Lash, R.R.; Townsend Peterson, A.; Damon, I.K.; Malekani, J.; Formenty, P. Ecology and geography of human monkeypox case occurrences across Africa. J. Wildl. Dis. 2012, 48, 335–347. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Carroll, D.S.; Olson, V.A.; Hughes, C.; Galley, J.; Likos, A.; Montgomery, J.M.; Suu-Ire, R.; Kwasi, M.O.; Root, J.J.; et al. A silent enzootic of an orthopoxvirus in Ghana, West Africa: Evidence for multi-species involvement in the absence of widespread human disease. Am. J. Trop. Med. Hyg. 2010. [Google Scholar] [CrossRef]
- Fuller, T.; Thomassen, H.A.; Mulembakani, P.M.; Johnston, S.C.; Lloyd-Smith, J.O.; Kisalu, N.K.; Lutete, T.K.; Blumberg, S.; Fair, J.N.; Wolfe, N.D.; et al. Using remote sensing to map the risk of human monkeypox virus in the Congo basin. Ecohealth 2011, 8, 14–25. [Google Scholar] [CrossRef]
- Peters, J.C. An epizootic of monkey pox at Rotterdam Zoo. Int. Zoo Yearb. 1966, 6, 274–275. [Google Scholar] [CrossRef]
- Hutin, Y.J.; Williams, R.J.; Malfait, P.; Pebody, R.; Loparev, V.N.; Ropp, S.L.; Rodriguez, M.; Knight, J.C.; Tshioko, F.K.; Khan, A.S. Outbreak of human monkeypox, Democratic Republic of Congo, 1996 to 1997. Emerg. Infect. Dis. 2001, 7, 434. [Google Scholar] [CrossRef] [PubMed]
- Arita, I.; Henderson, D.A. Smallpox and monkeypox in non-human primates. Bull. World Health Organ. 1968, 39, 277. [Google Scholar] [PubMed]
- Marennikova, S.S.; Seluhina, E.M.; Mal’ceva, N.N.; Ladnyj, I.D. Poxviruses isolated from clinically ill and asymptomatically infected monkeys and a chimpanzee. Bull. World Health Organ. 1972, 46, 613–620. [Google Scholar]
- Gispen, R.; Brand Saathof, B.; Hekker, A.C. Monkeypox specific antibodies in human and simian sera from the Ivory Coast and Nigeria. Bull. World Health Organ. 1976, 53, 355–360. [Google Scholar] [PubMed]
- Marennikova, S.S.; Seluhina, E.M. Susceptibility of some rodent species to monkeypox virus, and course of the infection. Bull. World Health Organ. 1976, 53, 13–20. [Google Scholar] [PubMed]
- Falendysz, E.A.; Londoño-Navas, A.M.; Meteyer, C.U.; Pussini, N.; Lopera, J.G.; Osorio, J.E.; Rocke, T.E. Evaluation of monkeypox virus infection of black-tailed prairie dogs (Cynomys ludovicianus) using in vivo bioluminescent imaging. J. Wildl. Dis. 2014, 50, 524–536. [Google Scholar] [CrossRef]
- Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M.I. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. [Google Scholar] [CrossRef]
- Mucker, E.M.; Chapman, J.; Huzella, L.M.; Huggins, J.W.; Shamblin, J.; Robinson, C.G.; Hensley, L.E. Susceptibility of marmosets (Callithrix jacchus) to monkeypox virus: A low dose prospective model for monkeypox and smallpox disease. PLoS ONE 2015, 10, e0131742. [Google Scholar] [CrossRef]
- Reed, K.D.; Melski, J.W.; Graham, M.B.; Regnery, R.L.; Sotir, M.J.; Wegner, M.V.; Kazmierczak, J.J.; Stratman, E.J.; Li, Y.; Fairley, J.A.; et al. The Detection of Monkeypox in Humans in the Western Hemisphere. N. Engl. J. Med. 2004, 350, 342–350. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Update: Multistate Outbreak of Monkeypox—Illinois, Indiana, Missouri, Ohio, and Wisconsin. 2003. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5227a5.htm (accessed on 26 September 2020).
- Sklenovská, N. Monkeypox Virus. In Animal-Origin Viral Zoonoses—Livestock Diseases and Management; Malik, Y.S., Singh, R.K., Kuldeep, D., Eds.; Springer: Singapore, 2020; pp. 39–68. [Google Scholar]
- Ladnyj, I.D.; Ziegler, P.; Kima, E. A human infection caused by monkeypox virus in Basankusu Territory, Democratic Republic of the Congo. Bull. World Health Organ. 1972, 46, 593. [Google Scholar]
- Heymann, D.L.; Szczeniowski, M.; Esteves, K. Re-emergence of monkeypox in Africa: A review of the past six years. Br. Med. Bull. 1998, 54, 693–702. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Human monkeypox in Kasai Oriental, Democratic Republic of the Congo (former Zaire): Preliminary report of October, 1997 investigation. Wkly Epidemiol Rec. 1997, 72, 369–372. [Google Scholar]
- Petersen, E.; Kantele, A.; Koopmans, M.; Asogun, D.; Yinka-Ogunleye, A.; Ihekweazu, C.; Zumla, A. Human Monkeypox: Epidemiologic and Clinical Characteristics, Diagnosis, and Prevention. Infect. Dis. Clin. N. Am. 2019, 33, 1027–1043. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; Emerson, G.L.; Pukuta, E.; Karhemere, S.; Muyembe, J.J.; Bikindou, A.; McCollum, A.M.; Moses, C.; Wilkins, K.; Zhao, H.; et al. Short report: Detection of human monkeypox in the Republic of the Congo following intensive community education. Am. J. Trop. Med. Hyg. 2013, 88, 982–985. [Google Scholar] [CrossRef]
- Berthet, N.; Nakouné, E.; Whist, E.; Selekon, B.; Burguire, A.M.; Manuguerra, J.C.; Gessain, A.; Kazanji, M. Maculopapular lesions in the Central African Republic. Lancet 2011, 378, 1354. [Google Scholar] [CrossRef]
- McCollum, A.M.; Nakazawa, Y.; Ndongala, G.M.; Pukuta, E.; Karhemere, S.; Lushima, R.S.; Ilunga, B.K.; Kabamba, J.; Wilkins, K.; Gao, J.; et al. Case report: Human monkeypox in the kivus, a conflict region of the Democratic Republic of the Congo. Am. J. Trop. Med. Hyg. 2015, 93, 718–721. [Google Scholar] [CrossRef]
- Mwamba, D.K.; Kebela, B.I.; Shongo, R.L.; Pukuta, E.; Kayembe, N.J.M. Profil épidemiologique du monkeypox en RDC, 2010–2014. Ann. African Med. 2014, 8, 1855–1860. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). About Monkeypox. Available online: https://www.cdc.gov/poxvirus/monkeypox/about.html (accessed on 24 September 2020).
- Kalthan, E.; Dondo-Fongbia, J.P.; Yambele, S.; Dieu-Creer, L.R.; Zepio, R.; Pamatika, C.M. Twelve cases of monkeypox virus outbreak in Bangassou District (Central African Republic) in December 2015. Bull. Soc. Pathol. Exot. 2016, 109, 358–363. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Monkeypox in Central African Republic. Available online: https://www.who.int/csr/don/13-october-2016-monkeypox-caf/en/ (accessed on 25 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 35. 2017. Available online: https://apps.who.int/iris/bitstream/handle/10665/258888/OEW35-268192017.pdf?sequence=1 (accessed on 25 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 22. 2019. Available online: https://apps.who.int/iris/bitstream/handle/10665/325086/OEW22-270502062019.pdf (accessed on 25 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 11. 2020. Available online: https://apps.who.int/iris/bitstream/handle/10665/331451/OEW11-0915032020.pdf (accessed on 25 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 21. 2017. Available online: https://apps.who.int/iris/bitstream/handle/10665/255579/OEW21-202652017.pdf?sequence=1 (accessed on 25 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 08. 2018. Available online: https://apps.who.int/iris/bitstream/handle/10665/260335/OEW8-1723022018.pdf?sequence=1 (accessed on 26 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 39. 2018. Available online: https://apps.who.int/iris/bitstream/handle/10665/275136/OEW39-2228092018.pdf (accessed on 26 September 2020).
- World Health Organization (WHO). Weekly Bulletin on Outbreaks and Other Emergencies, Week 01. 2018. Available online: https://apps.who.int/iris/bitstream/handle/10665/278952/OEW01-29122018-04012019.pdf?sequence=1&isAllowed=y (accessed on 26 September 2020).
- Ministry of Health, State of Isreal—Monkeypox Patient Diagnosed. Available online: https://www.health.gov.il/English/News_and_Events/Spokespersons_Messages/Pages/12102018_1.aspx (accessed on 29 September 2020).
- Mauldin, M.R.; Mccollum, A.M.; Nakazawa, Y.J.; Mandra, A.; Whitehouse, E.R.; Davidson, W.; Zhao, H.; Gao, J.; Li, Y.; Doty, J.; et al. Exportation of Monkeypox Virus From the African Continent. J. Infect. Dis. 2020, 1–10. [Google Scholar] [CrossRef]
- Radonić, A.; Metzger, S.; Dabrowski, P.W.; Couacy-Hymann, E.; Schuenadel, L.; Kurth, A.; Mätz-Rensing, K.; Boesch, C.; Leendertz, F.H.; Nitsche, A. Fatal monkeypox in wild-living sooty mangabey, Côte d’Ivoire, 2012. Emerg. Infect. Dis. 2014, 20, 1009–1011. [Google Scholar] [CrossRef]
- Ligon, B.L. Monkeypox: A review of the history and emergence in the Western hemisphere. Semin. Pediatr. Infect. Dis. 2004, 15, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Hutson, C.L.; Lee, K.N.; Abel, J.; Carroll, D.S.; Montgomery, J.M.; Olson, V.A.; Li, Y.; Davidson, W.; Hughes, C.; Dillon, M.; et al. Monkeypox zoonotic associations: Insights from laboratory evaluation of animals associated with the multi-state US outbreak. Am. J. Trop. Med. Hyg. 2007, 76, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Ježek, Z.; Grab, B.; Szczeniowski, M.; Paluku, K.M.; Mutombo, M. Clinico-epidemiological features of monkeypox patients with an animal or human source of infection. Bull. World Health Organ. 1988, 66, 459. [Google Scholar] [PubMed]
- Kalthan, E.; Tenguere, J.; Ndjapou, S.G.; Koyazengbe, T.A.; Mbomba, J.; Marada, R.M.; Rombebe, P.; Yangueme, P.; Babamingui, M.; Sambella, A.; et al. Investigation of an outbreak of monkeypox in an area occupied by armed groups, Central African Republic. Med. Mal. Infect. 2018, 48, 263–268. [Google Scholar] [CrossRef]
- Laudisoit, A.; Baelo, P.; Mussaw Awazi, M.; VanHoutte, N.; VanHees, M.; Amundala, N.; Leirs, H. Biodiversity, Bushmeat and Monkeypox in the Democratic Republic of the Congo: Another viral threat upon larger cities? Trop. Med. Int. Heal. 2015, 1, 30–31. [Google Scholar]
- Kabuga, A.I.; El Zowalaty, M.E. A review of the monkeypox virus and a recent outbreak of skin rash disease in Nigeria. J. Med. Virol. 2019, 91, 533–540. [Google Scholar] [CrossRef]
- Mauldin, M.R.; Antwerpen, M.; Emerson, G.L.; Li, Y.; Zoeller, G.; Carroll, D.S.; Meyer, H. Cowpox virus: What’s in a name? Viruses 2017, 9, 1–15. [Google Scholar] [CrossRef]
- Baxby, D.; Bennett, M. Cowpox: A re-evaluation of the risks of human cowpox based on new epidemiological information. Arch. Virol. Suppl. 1997, 13, 1–12. [Google Scholar] [CrossRef]
- Gehring, H.; Mahnel, H.; Mayer, H. Kurze Mitteilungen: Elefantenpocken. Zent. Veterinärmedizin Reihe B 1972. [Google Scholar] [CrossRef]
- Mätz-Rensing, K.; Ellerbrok, H.; Ehlers, B.; Pauli, G.; Floto, A.; Alex, M.; Czerny, C.P.; Kaup, F.J. Fatal poxvirus outbreak in a colony of New World Monkeys. Vet. Pathol. 2006, 43, 212–218. [Google Scholar] [CrossRef]
- Martina, B.E.E.; Van Doornum, G.; Dorrestein, G.M.; Niesters, H.G.M.; Stittelaar, K.J.; Wolters, M.A.B.I.; Van Bolhuis, H.G.H.; Osterhaus, A.D.M.E. Cowpox virus transmission from rats to monkeys, the Netherlands. Emerg. Infect. Dis. 2006, 12, 1005–1007. [Google Scholar] [CrossRef] [PubMed]
- Girling, S.J.; Pizzi, R.; Cox, A.; Beard, P.M. Fatal cowpox virus infection in two squirrel monkeys (Saimiri sciureus). Vet. Rec. 2011, 169, 156. [Google Scholar] [CrossRef] [PubMed]
- Baxby, D.; Jones, D.M.; Ashton, D.G.; Thomsett, L.R. An outbreak of cowpox in captive cheetahs: Virological and epidemiological studies. J. Hyg. 1982, 89, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Marennikova, S.S.; Maltseva, N.N.; Korneeva, V.I.; Garanina, N.M. Outbreak of pox disease among carnivora (Felidae) and edentata. J. Infect. Dis. 1977, 135, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Willemse, A.; Egberink, H.F. Transmission of cowpox virus infection from domestic cat to man. Lancet 1985, 325, 1515. [Google Scholar] [CrossRef]
- Tryland, M.; Sandvik, T.; Hansen, H.; Haukenes, G.; Holtet, L.; Bennett, M.; Mehl, R.; Moens, U.; Olsvik, Ø.; Traavik, T. Characteristics of four cowpox virus isolates from Norway and Sweden. APMIS 1998. [Google Scholar] [CrossRef]
- Tryland, M.; Okeke, M.I.; af Segerstad, C.H.; Mörner, T.; Traavik, T.; Ryser-Degiorgis, M.P. Orthopoxvirus DNA in Eurasian Lynx, Sweden. Emerg. Infect. Dis. 2011, 17, 626–632. [Google Scholar] [CrossRef]
- Smith, K.C.; Bennett, M.; Garrett, D.C. Skin lesions caused by orthopoxvirus infection in a dog. J. Small Anim. Pract. 1999. [Google Scholar] [CrossRef]
- Chantrey, J.; Meyer, H.; Baxby, D.; Begon, M.; Bown, K.J.; Hazel, S.M.; Jones, T.; Montgomery, W.I.; Bennett, M. Cowpox: Reservoir hosts and geographic range. Epidemiol. Infect. 1999, 122, 455–460. [Google Scholar] [CrossRef]
- Hazel, S.M.; Bennett, M.; Chantrey, J.; Bown, K.; Cavanagh, R.; Jones, T.R.; Baxby, D.; Begon, M. A longitudinal study of an endemic disease in its wildlife reservoir: Cowpox and wild rodents. Epidemiol. Infect. 2000, 124, 551–562. [Google Scholar] [CrossRef]
- Campe, H.; Zimmermann, P.; Glos, K.; Bayer, M.; Bergemann, H.; Dreweck, C.; Graf, P.; Weber, B.K.; Meyer, H.; Büttner, M.; et al. Cowpox virus transmission from Pet Rats to humans, Germany. Emerg. Infect. Dis. 2009, 15, 777–780. [Google Scholar] [CrossRef]
- Marennikova, S.S.; Ladnyj, I.D.; Ogorodnikova, Z.I.; Shelukhina, E.M.; Maltseva, N.N. Identification and study of a poxvirus isolated from wild rodents in Turkmenia. Arch. Virol. 1978, 56, 7–14. [Google Scholar] [CrossRef]
- Essbauer, S.; Hartnack, S.; Misztela, K.; Kießling-tsalos, J. Patterns of Orthopox Virus Wild Rodent Host in South Germany. Vector-Borne Zoonotic Dis. 2009, 9, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, T.; Tryland, M.; Hansen, H.; Mehl, R.; Moens, U.; Olsvik, Ø.; Traavik, T. Naturally occurring orthopoxviruses: Potential for recombination with vaccine vectors. J. Clin. Microbiol. 1998, 36, 2542–2547. [Google Scholar] [CrossRef] [PubMed]
- Kik, M.J.L.; Liu, P.L.; Van Asten, J.A.M. Cowpoxvirus infection in the Patagonian cavy (Dolichotis patagonum) emerging disease in an educational animal park the first reported case. Vet. Q. 2006, 28, 42–44. [Google Scholar] [CrossRef]
- Tryland, M.; Sandvik, T.; Arnemo, J.M.; Stuve, G.; Olsvik, Ø.; Traavik, T. Antibodies against orthopoxviruses in wild carnivores from Fennoscandia. J. Wildl. Dis. 1998. [Google Scholar] [CrossRef] [PubMed]
- Boulanger, D.; Brochier, B.; Crouch, A.; Bennett, M.; Gaskell, R.M.; Baxby, D.; Pastoret, P.P. Comparison of the susceptibility of the red fox (Vulpes vulpes) to a vaccinia-rabies recombinant virus and to cowpox virus. Vaccine 1995. [Google Scholar] [CrossRef]
- Schaller, V.K.; Pilaski, J. Pocken bei Breitmaulnashornern (Ceratotheriilm s. simum) im Zoologischen Garten Münster. Zool. Gatten N.F 1979, 49, 169–184. [Google Scholar]
- Kurth, A.; Straube, M.; Kucska, A.; Dunsche, A.J.; Meyer, H.; Nitsche, A. Cowpox virus outbreak in banded mongooses (mungos mungo) and jaguarundis (Herpailurus yagouaroundi) with a time-delayed infection to humans. PLoS ONE 2009, 4, e6883. [Google Scholar] [CrossRef]
- Zwart, P.; Gispen, R.; Peters, J.C. Cowpox in okapis Okapia johnstoni at Rotterdam zoo. Br. Vet. J. 1971. [Google Scholar] [CrossRef]
- Franke, A.; Kershaw, O.; Jenckel, M.; König, L.; Beer, M.; Hoffmann, B.; Hoffmann, D. Fatal cowpox virus infection in an aborted foal. Vector-Borne Zoonotic Dis. 2016, 16, 431–433. [Google Scholar] [CrossRef] [PubMed]
- Wisser, J.; Pilaski, J.; Strauss, G.; Meyer, H.; Burck, G.; Truyen, U.; Rudolph, M.; Frölich, K. Cowpox virus infection causing stillbirth in an Asian elephant (Elphas maximus). Vet. Rec. 2001, 149, 244–246. [Google Scholar] [CrossRef] [PubMed]
- Hentschke, J.; Meyer, H.; Wittstatt, U.; Ochs, A.; Burkhardt, S.; Aue, A. Kuhpocken bei kanadischen bibern (castor fiver canadensis) und katzenbaren (ailurus fulgens). Tierarztl. Umsch. 1999, 54. [Google Scholar]
- Cardeti, G.; Brozzi, A.; Eleni, C.; Polici, N.; D’Alterio, G.; Carletti, F.; Scicluna, M.T.; Castilletti, C.; Capobianchi, M.R.; di Caro, A.; et al. Cowpox virus in Llama, Italy. Emerg. Infect. Dis. 2011, 17, 1513–1515. [Google Scholar] [CrossRef]
- Schuppel, K.F.; Menger, S.; Eulenberger, K.; Bernhard, A.; Pilaski, J. Kuhpocken Infektion bei Alpakas (Lama glama pacos). Verh ber Erkrg Zootiere 1997, 38, 259–264. [Google Scholar]
- Essbauer, S.; Meyer, H. Genus Orthopoxvirus: Cowpox virus. In Poxviruses. Birkhäuser Advances in Infectious Diseases; Mercer, A.A., Schmidt, A., Weber, O., Eds.; Birkhäuser: Basel, Switzerland, 2007. [Google Scholar]
- Stemmler, M.; Neubauer, H.; Meyer, H. Comparison of closely related orthopoxvirus isolates by random amplified polymorphic DNA and restriction fragment length polymorphism analysis. J. Vet. Med. Ser. B 2001. [Google Scholar] [CrossRef]
- Baxby, D. Is cowpox misnamed? A review of 10 human cases. Br. Med. J. 1977, 1, 1379–1381. [Google Scholar] [CrossRef]
- Mahnel, H.; Holejsovsky, J.; Bartak, P.; Czerny, C.P. Kongenitale “Ektromelie” bei Pelztieren durch Orthopoxvirus muris. Teirarztliche Prax. 1993, 21, 469–472. [Google Scholar]
- Vorou, R.M.; Papavassiliou, V.G.; Pierroutsakos, I.N. Cowpox virus infection: An emerging health threat. Curr. Opin. Infect. Dis. 2008, 21, 153–156. [Google Scholar] [CrossRef]
- Bennett, M.; Gaskell, C.J.; Baxbyt, D.; Gaskell, R.M.; Kelly, D.F.; Naidoot, J. Feline cowpox virus infection. J. Small Anim. Pract. 1990, 31, 167–173. [Google Scholar] [CrossRef]
- Thomsett, L.R.; Baxby, D.; Denham, E.M. Cowpox in the domestic cat. Vet. Rec. 1978, 103, 567. [Google Scholar] [CrossRef] [PubMed]
- Hinrichs, U.; Van De Poel, H.; Van Den Ingh, T.S.G.A.M. Necrotizing pneumonia in a cat caused by an orthopox virus. J. Comp. Pathol. 1999. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M.; Pfleghaar, S.; von Bomhard, D.; Kaaden, O.R.; Meyer, H. Retrospective investigation of feline cowpox in Germany. Vet. Rec. 2002, 150, 50–51. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.L. Poxviruses, Birkhäuser Advances in Infectious Diseases; Mercer, A.A., Schmidt, A., Weber, O., Eds.; Birkhäuser Verlag: Basel, Switzerland, 2007; ISBN 3-7643-7556-6. [Google Scholar]
- World Health Organization (WHO). The Global Eradication of Smallpox: Final Report of the Global Commission for the Certification of Smallpox Eradication, Geneva, December 1979. Available online: https://apps.who.int/iris/bitstream/handle/10665/39253/a41438.pdf?sequence=1&isAllowed=y (accessed on 30 September 2020).
- Henderson, D.A. Smallpox: The Death of a Disease: The Inside Story of Eradicating a Worldwide Killer; Prometheus Books: New York, NY, USA, 2009; ISBN 161592230X. [Google Scholar]
- Kroon, E.G.; Mota, B.E.F.; Abrahão, J.S.; da Fonseca, F.G.; de Souza Trindade, G. Zoonotic Brazilian Vaccinia virus: From field to therapy. Antiviral Res. 2011, 92, 150–163. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Laboratory Acquired Vaccinia Exposures and Infections—United States, 2005–2007. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5715a3.htm (accessed on 30 September 2020).
- Drexler, I.; Staib, C.; Sutter, G. Modified vaccinia virus Ankara as antigen delivery system: How can we best use its potential? Curr. Opin. Biotechnol. 2004, 15, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Meseda, C.A.; Garcia, A.D.; Kumar, A.; Mayer, A.E.; Manischewitz, J.; King, L.R.; Golding, H.; Merchlinsky, M.; Weir, J.P. Enhanced immunogenicity and protective effect conferred by vaccination with combinations of modified vaccinia virus Ankara and licensed smallpox vaccine Dryvax in a mouse model. Virology 2005, 339, 164–175. [Google Scholar] [CrossRef]
- Trindade, G.S.; Emerson, G.L.; Carroll, D.S.; Kroon, E.G.; Damon, I.K. Brazilian vaccinia viruses and their origins. Emerg. Infect. Dis. 2007, 13, 965. [Google Scholar] [CrossRef]
- Miranda, J.B.; Borges, I.A.; Campos, S.P.S.; Vieira, F.N.; De Ázara, T.M.F.; Marques, F.A.; Costa, G.B.; Luis, A.P.M.F.; De Oliveira, J.S.; Ferreira, P.C.P.; et al. Serologic and molecular evidence of vaccinia virus circulation among small mammals from different biomes, Brazil. Emerg. Infect. Dis. 2017, 23, 931–938. [Google Scholar] [CrossRef]
- Medaglia, M.L.G.; Moussatché, N.; Nitsche, A.; Dabrowski, P.W.; Li, Y.; Damon, I.K.; Lucas, C.G.O.; Arruda, L.B.; Damaso, C.R. Genomic Analysis, Phenotype, and Virulence of the Historical Brazilian Smallpox Vaccine Strain IOC: Implications for the Origins and Evolutionary Relationships of Vaccinia Virus. J. Virol. 2015, 89, 11909–11925. [Google Scholar] [CrossRef]
- Baxby, D.; Hill, B.J. Characteristics of a new poxvirus isolated from Indian buffaloes. Arch. Gesamte Virusforsch. 1971, 35, 70–79. [Google Scholar] [CrossRef]
- Bloch, B.; Lal, S.M. A study of the ultrastructure of the buffalo pox virus. Acta Pathol. Microbiol. Scand. Sect. B Microbiol. 1975, 83, 191–200. [Google Scholar] [CrossRef]
- Bera, B.C.; Shanmugasundaram, K.; Barua, S.; Anand, T.; Riyesh, T.; Vaid, R.K.; Virmani, N.; Bansal, M.; Shukla, B.N.; Malik, P. Sequence and phylogenetic analysis of host-range (E3L, K3L, and C7L) and structural protein (B5R) genes of buffalopox virus isolates from buffalo, cattle, and human in India. Virus Genes 2012, 45, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Eltom, K.H.; Samy, A.M.; Abd El Wahed, A.; Czerny, C.-P. Buffalopox Virus: An Emerging Virus in Livestock and Humans. Pathogens 2020, 9, 676. [Google Scholar] [CrossRef] [PubMed]
- Yadav, P.D.; Mauldin, M.R.; Nyayanit, D.A.; Albariño, C.G.; Sarkale, P.; Shete, A.; Guerrero, L.W.; Nakazawa, Y.; Nichol, S.T.; Mourya, D.T. Isolation and phylogenomic analysis of buffalopox virus from human and buffaloes in India. Virus Res. 2020, 277, 197836. [Google Scholar] [CrossRef]
- Goyal, T.; Varshney, A.; Bakshi, S.K.; Barua, S.; Bera, B.C.; Singh, R.K. Buffalo pox outbreak with atypical features: A word of caution and need for early intervention! Int. J. Dermatol. 2013, 52, 1224–1230. [Google Scholar] [CrossRef] [PubMed]
- Bhanuprakash, V.; Venkatesan, G.; Balamurugan, V.; Hosamani, M.; Yogisharadhya, R.; Chauhan, R.S.; Pande, A.; Mondal, B.; Singh, R.K. Pox outbreaks in sheep and goats at Makhdoom (Uttar Pradesh), India: Evidence of sheeppox virus infection in goats. Transbound. Emerg. Dis. 2010, 57, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Venkatesan, G.; Singh, R.K. Buffalopox Virus. In Animal-Origin Viral Zoonoses—Livestock Diseases and Management; Malik, Y.S., Singh, R.K., Kuldeep, D., Eds.; Springer: Singapore, 2020; pp. 145–162. [Google Scholar]
- Zafar, A.; Swanepoel, R.; Hewson, R.; Nizam, M.; Ahmed, A.; Husain, A.; Grobbelaar, A.; Bewley, K.; Mioulet, V.; Dowsett, B. Nosocomial buffalopoxvirus infection, Karachi, Pakistan. Emerg. Infect. Dis. 2007, 13, 902. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Bhat, P.P.; Mishra, B.P.; Singh, R.K. Biological transmissibility of buffalopox virus. J. Appl. Anim. Res. 1996, 9, 79–88. [Google Scholar] [CrossRef]
- Nalca, A.; Nichols, D.K. Rabbitpox: A model of airborne transmission of smallpox. J. Gen. Virol. 2011, 92, 31–35. [Google Scholar] [CrossRef]
- Christensen, L.R.; Bond, E.; Matanic, B. “Pock-less” rabbit pox. Lab. Anim. Care 1967, 17, 281. [Google Scholar]
- Fenner, F. The biological characters of several strains of vaccinia, cowpox and rabbitpox viruses. Virology 1958, 5, 502–529. [Google Scholar] [CrossRef]
- Dumbell, K.; Richardson, M. Virological investigations of specimens from buffaloes affected by buffalopox in Maharashtra State, India between 1985 and 1987. Arch. Virol. 1993, 128, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Leite, J.A.; Drumond, B.P.; Trindade, G.S.; Lobato, Z.I.P.; Da Fonseca, F.G.; Dos Santos, J.R.; Madureira, M.C.; Guedes, M.I.M.C.; Ferreira, J.M.S.; Bonjardim, C.A.; et al. Passatempo virus, a vaccinia virus strain, Brazil. Emerg. Infect. Dis. 2005, 11, 1935–1938. [Google Scholar] [CrossRef] [PubMed]
- Rivetti, A.V.; Guedes, M.I.M.C.; Rehfeld, I.S.; Oliveira, T.M.L.; Matos, A.C.D.; Abrahão, J.S.; Kroon, E.G.; Lobato, Z.I.P. Bovine vaccinia, a systemic infection: Evidence of fecal shedding, viremia and detection in lymphoid organs. Vet. Microbiol. 2013, 162, 103–111. [Google Scholar] [CrossRef]
- Matos, A.C.D.; Rehfeld, I.S.; Guedes, M.I.M.C.; Lobato, Z.I.P. Bovine vaccinia: Insights into the disease in cattle. Viruses 2018, 10, 120. [Google Scholar] [CrossRef]
- Damaso, C.R.A.; Esposito, J.J.; Condit, R.C.; Moussatché, N. An emergent poxvirus from humans and cattle in Rio de Janeiro State: Cantagalo virus may derive from Brazilian smallpox vaccine. Virology 2000, 277, 439–449. [Google Scholar] [CrossRef]
- De Souza Trindade, G.; Da Fonseca, F.G.; Marques, J.T.; Nogueira, M.L.; Mendes, L.C.N.; Borges, A.S.; Peiró, J.R.; Pituco, E.M.; Bonjardim, C.A.; Ferreira, P.C.P.; et al. Araçatuba virus: A vaccinia-like virus associated with infection in humans and cattle. Emerg. Infect. Dis. 2003, 9, 155–160. [Google Scholar] [CrossRef]
- Campos, R.K.; Brum, M.C.S.; Nogueira, C.E.W.; Drumond, B.P.; Alves, P.A.; Siqueira-Lima, L.; Assis, F.L.; Trindade, G.S.; Bonjardim, C.A.; Ferreira, P.C.; et al. Assessing the variability of Brazilian Vaccinia virus isolates from a horse exanthematic lesion: Coinfection with distinct viruses. Arch. Virol. 2011, 156, 275–283. [Google Scholar] [CrossRef]
- Lima, M.T.; Oliveira, G.P.; Assis, F.L.; de Oliveira, D.B.; Vaz, S.M.; de Souza Trindade, G.; Abrahão, J.S.; Kroon, E.G. Ocular vaccinia infection in dairy worker, Brazil. Emerg. Infect. Dis. 2018, 24, 161. [Google Scholar] [CrossRef]
- Trindade, G.d.S.; Emerson, G.L.; Sammons, S.; Frace, M.; Govil, D.; Mota, B.E.F.; Abrahão, J.S.; de Assis, F.L.; Olsen-Rasmussen, M.; Goldsmith, C.S.; et al. Serro 2 virus highlights the fundamental genomic and biological features of a natural vaccinia virus infecting humans. Viruses 2016, 8, 328. [Google Scholar] [CrossRef]
- Drumond, B.P.; Leite, J.A.; da Fonseca, F.G.; Bonjardim, C.A.; Ferreira, P.C.P.; Kroon, E.G. Brazilian Vaccinia virus strains are genetically divergent and differ from the Lister vaccine strain. Microbes Infect. 2008, 10, 185–197. [Google Scholar] [CrossRef] [PubMed]
- de Souza Trindade, G.; Li, Y.; Olson, V.A.; Emerson, G.; Regnery, R.L.; da Fonseca, F.G.; Damon, I. Real-time PCR assay to identify variants of Vaccinia virus: Implications for the diagnosis of bovine vaccinia in Brazil. J. Virol. Methods 2008, 152, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Costa, G.B.; Miranda, J.B.; Almeida, G.G.; Silva de Oliveira, J.; Pinheiro, M.S.; Gonçalves, S.A.; Pimenta Dos Reis, J.K.; Gonçalves, R.; Ferreira, P.C.P.; Bonjardim, C.A.; et al. Detection of Vaccinia Virus in Urban Domestic Cats, Brazil. Emerg. Infect. Dis. 2017, 23, 360–362. [Google Scholar] [CrossRef] [PubMed]
- Dutra, L.A.L.; de Freitas Almeida, G.M.; Oliveira, G.P.; Abrahão, J.S.; Kroon, E.G.; Trindade, G.d.S. Molecular evidence of Orthopoxvirus DNA in capybara (Hydrochoerus hydrochaeris) stool samples. Arch. Virol. 2017, 162, 439–448. [Google Scholar] [CrossRef]
- Brum, M.C.S.; dos Anjos, B.L.; Nogueira, C.E.W.; Amaral, L.A.; Weiblen, R.; Flores, E.F. An outbreak of orthopoxvirus-associated disease in horses in southern Brazil. J. Vet. Diagn. Investig. 2010, 22, 143–147. [Google Scholar] [CrossRef]
- Abrahão, J.S.; de Souza Trindade, G.; Pereira-Oliveira, G.; de Oliveira Figueiredo, P.; Costa, G.; Moreira Franco-Luiz, A.P.; Lopes Assis, F.; Bretas de Oliveira, D.; Mattos Paim, L.R.; de Araújo Oliveira, C.E.; et al. Detection of Vaccinia virus during an outbreak of exanthemous oral lesions in Brazilian equids. Equine Vet. J. 2017. [Google Scholar] [CrossRef]
- Lima, M.T.; Oliveira, G.P.; Afonso, J.A.B.; Souto, R.J.C.; De Mendonça, C.L.; Dantas, A.F.M.; Abrahao, J.S.; Kroon, E.G. An update on the known host range of the brazilian vaccinia virus: An outbreak in Buffalo Calves. Front. Microbiol. 2019, 9, 3327. [Google Scholar] [CrossRef]
- Abrahão, J.S.; Guedes, M.I.M.; Trindade, G.S.; Fonseca, F.G.; Campos, R.K.; Mota, B.F.; Lobato, Z.I.P.; Silva-Fernandes, A.T.; Rodrigues, G.O.L.; Lima, L.S.; et al. One more piece in the VACV ecological puzzle: Could peridomestic rodents be the link between wildlife and bovine vaccinia outbreaks in Brazil? PLoS ONE 2009, 4, e7428. [Google Scholar] [CrossRef]
- Martins da Costa, P.S.P.; Oliveira, J.S.; Domingos, I.J.d.S.; e Silva, P.H.B.; Dutra, A.G.S.; Amaral, C.D.; Abrahão, J.S.; Richini Pereira, V.B.; Kroon, E.G.; Barbosa Costa, G.; et al. Circulation of vaccinia virus in southern and south-eastern wildlife, Brazil. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Costa, G.B.; de Almeida, L.R.; Cerqueira, A.G.R.; Mesquita, W.U.; de Oliveira, J.S.; Miranda, J.B.; Saraiva-Silva, A.T.; Abrahão, J.S.; Drumond, B.P.; Kroon, E.G.; et al. Vaccinia virus among domestic dogs and wild coatis, Brazil, 2013–2015. Emerg. Infect. Dis. 2018, 24, 2338–2342. [Google Scholar] [CrossRef]
- Assis, F.L.; Borges, I.A.; Peregrino Ferreira, P.C.; Bonjardim, C.A.; de Souza Trindade, G.; Portela Lobato, Z.I.; Maldonado Guedes, M.I.; Mesquita, V.; Kroon, E.G.; Abrahão, J.S. Group 2 vaccinia virus, Brazil. Emerg. Infect. Dis. 2012. [Google Scholar] [CrossRef]
- Peres, M.G.; Barros, C.B.; Appolinário, C.M.; Antunes, J.M.A.P.; Mioni, M.S.R.; Bacchiega, T.S.; Allendorf, S.D.; Vicente, A.F.; Fonseca, C.R.; Megid, J. Dogs and opossums positive for vaccinia virus during outbreak affecting cattle and humans, São Paulo State, Brazil. Emerg. Infect. Dis. 2016, 22, 271–273. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.M.S.; Drumond, B.P.; Guedes, M.I.M.C.; Pascoal-Xavier, M.A.; Almeida-Leite, C.M.; Arantes, R.M.E.; Mota, B.E.F.; Abrahão, J.S.; Alves, P.A.; Oliveira, F.M.; et al. Virulence in murine model shows the existence of two distinct populations of Brazilian Vaccinia virus strains. PLoS ONE 2008, 3, e3043. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, G.; Assis, F.; Almeida, G.; Albarnaz, J.; Lima, M.; Andrade, A.C.; Calixto, R.; Oliveira, C.; Neto, J.D.; Trindade, G.; et al. From lesions to viral clones: Biological and molecular diversity amongst autochthonous Brazilian Vaccinia virus. Viruses 2015, 7, 1218–1237. [Google Scholar] [CrossRef] [PubMed]
- Abrahão, J.S.; Oliveira, T.M.L.; Campos, R.K.; Madureira, M.C.; Kroon, E.G.; Lobato, Z.I.P. Bovine vaccinia outbreaks: Detection and isolation of vaccinia virus in milk samples. Foodborne Pathog. Dis. 2009, 6, 1141–1146. [Google Scholar] [CrossRef] [PubMed]
- Rehfeld, I.S.; Matos, A.C.D.; Guedes, M.I.M.C.; Costa, A.G.; Fraiha, A.L.S.; Lobato, Z.I.P. Subclinical bovine vaccinia: An important risk factor in the epidemiology of this zoonosis in cattle. Res. Vet. Sci. 2017, 114, 233–235. [Google Scholar] [CrossRef] [PubMed]
- Abrahão, J.S.; Silva-Fernandes, A.T.; Lima, L.S.; Campos, R.K.; Guedes, M.I.M.C.; Cota, M.M.G.; Assis, F.L.; Borges, I.A.; Souza-Júnior, M.F.; Lobato, Z.I.P.; et al. Vaccinia virus infection in monkeys, Brazilian Amazon. Emerg. Infect. Dis. 2010, 16, 976–979. [Google Scholar] [CrossRef] [PubMed]
- Peres, M.G.; Bacchiega, T.S.; Appolinário, C.M.; Vicente, A.F.; De Souza Ribeiro Mioni, M.; Ribeiro, B.L.D.; Fonseca, C.R.S.; Pelícia, V.C.; Ferreira, F.; Oliveira, G.P.; et al. Vaccinia virus in blood samples of humans, domestic and wild mammals in Brazil. Viruses 2018, 10, 42. [Google Scholar] [CrossRef]
- Peres, M.G.; Bacchiega, T.S.; Appolinário, C.M.; Vicente, A.F.; Allendorf, S.D.; Antunes, J.M.A.P.; Moreira, S.A.; Legatti, E.; Fonseca, C.R.; Pituco, E.M.; et al. Serological study of vaccinia virus reservoirs in areas with and without official reports of outbreaks in cattle and humans in São Paulo, Brazil. Arch. Virol. 2013, 158, 2433–2441. [Google Scholar] [CrossRef]
- Barbosa, A.V.; Medaglia, M.L.G.; Soares, H.S.; Quixabeira-Santos, J.C.; Gennari, S.M.; Damaso, C.R. Presence of neutralizing antibodies to orthopoxvirus in capybaras (hydrochoerus hydrochaeris) in Brazil. J. Infect. Dev. Ctries. 2014, 8, 1646–1649. [Google Scholar] [CrossRef]
- de Assis, F.L.; Pereira, G.; Oliveira, C.; Rodrigues, G.O.L.; Cotta, M.M.G.; Silva-Fernandes, A.T.; Ferreira, P.C.P.; Bonjardim, C.A.; de Souza Trindade, G.; Kroon, E.G.; et al. Serologic evidence of orthopoxvirus infection in buffaloes, Brazil. Emerg. Infect. Dis. 2012, 18, 698–700. [Google Scholar] [CrossRef] [PubMed]
- Franco-Luiz, A.P.M.; Fagundes Pereira, A.; de Oliveira, C.H.S.; Barbosa, J.D.; Oliveira, D.B.; Bonjardim, C.A.; Ferreira, P.C.P.; de Souza Trindade, G.; Abrahão, J.S.; Kroon, E.G. The detection of Vaccinia virus confirms the high circulation of Orthopoxvirus in buffaloes living in geographical isolation, Marajó Island, Brazilian Amazon. Comp. Immunol. Microbiol. Infect. Dis. 2016. [Google Scholar] [CrossRef]
- Siqueira Ferreira, J.M.; Abrahão, J.S.; Drumond, B.P.; Oliveira, F.M.; Alves, P.A.; Pascoal-Xavier, M.A.; Lobato, Z.I.P.; Bonjardim, C.A.; Peregrino Ferreira, P.C.; Kroon, E.G. Vaccinia virus: Shedding and horizontal transmission in a murine model. J. Gen. Virol. 2008, 89, 2986–2991. [Google Scholar] [CrossRef] [PubMed]
- Costa, G.B.; Lavergne, A.; Darcissac, E.; Lacoste, V.; Drumond, B.P.; Abrahão, J.S.; Kroon, E.G.; de Thoisy, B.; de Souza Trindade, G. Absence of vaccinia virus detection in a remote region of the Northern Amazon forests, 2005–2015. Arch. Virol. 2017, 162, 2369–2373. [Google Scholar] [CrossRef] [PubMed]
- Abrahão, J.S.; de Souza Trindade, G.; Ferreira, J.M.S.; Campos, R.K.; Bonjardim, C.A.; Ferreira, P.C.P.; Kroon, E.G. Long-lasting stability of Vaccinia virus strains in murine feces: Implications for virus circulation and environmental maintenance. Arch. Virol. 2009. [Google Scholar] [CrossRef] [PubMed]
- Fassbender, P.; Zange, S.; Ibrahim, S.; Zoeller, G.; Herbstreit, F.; Meyer, H. Generalized cowpox virus infection in a patient with HIV, Germany, 2012. Emerg. Infect. Dis. 2016, 22, 553. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, J.S.; Costa, G.B.; Franco Luiz, A.P.M.; Leite, J.A.; Bonjardim, C.A.; Abrahão, J.S.; Drumond, B.P.; Kroon, E.G.; Trindade, G.d.S. Cross-sectional study involving healthcare professionals in a Vaccinia virus endemic area. Vaccine 2017, 35, 3281–3285. [Google Scholar] [CrossRef] [PubMed]
- Lobato, Z.I.P.; Trindade, G.S.; Frois, M.C.M.; Ribeiro, E.B.T.; Dias, G.R.C.; Teixeira, B.M.; Lima, F.A.; Almeida, G.M.F.; Kroon, E.G. Outbreak of exantemal disease caused by Vaccinia virus in human and cattle in Zona da Mata region, Minas Gerais. Arq. Bras. Med. Vet. e Zootec. 2005. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Doty, J.B.; McCollum, A.M.; Olson, V.A.; Nakazawa, Y. Monkeypox re-emergence in Africa: A call to expand the concept and practice of One Health. Expert Rev. Anti. Infect. Ther. 2019, 17, 129–139. [Google Scholar] [CrossRef]
- Hassell, J.M.; Begon, M.; Ward, M.J.; Fèvre, E.M. Urbanization and Disease Emergence: Dynamics at the Wildlife–Livestock–Human Interface. Trends Ecol. Evol. 2017, 32, 55–67. [Google Scholar] [CrossRef]
- Oliveira, G.P.; Fernandes, A.T.S.; De Assis, F.L.; Alves, P.A.; Luiz, A.P.M.F.; Figueiredo, L.B.; De Almeida, C.M.C.; Travassos, C.E.P.F.; De Souza Trindade, G.; Abrahão, J.S.; et al. Short report: Intrafamilial transmission of Vaccinia virus during a bovine vaccinia outbreak in Brazil: A new insight in viral transmission chain. Am. J. Trop. Med. Hyg. 2014. [Google Scholar] [CrossRef]
- Laiton-Donato, K.; Ávila-Robayo, P.; Páez-Martinez, A.; Benjumea-Nieto, P.; Usme-Ciro, J.A.; Pinzón-Nariño, N.; Giraldo, I.; Torres-Castellanos, D.; Nakazawa, Y.; Patel, N.; et al. Progressive vaccinia acquired through zoonotic transmission in a patient with HIV/AIDS, Colombia. Emerg. Infect. Dis. 2020, 26, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.; Abubakar, I.; Ihekweazu, C.; Heymann, D.; Ntoumi, F.; Blumberg, L.; Asogun, D.; Mukonka, V.; Lule, S.A.; Bates, M.; et al. Monkeypox—Enhancing public health preparedness for an emerging lethal human zoonotic epidemic threat in the wake of the smallpox post-eradication era. Int. J. Infect. Dis. 2019, 78, 78–84. [Google Scholar] [CrossRef] [PubMed]
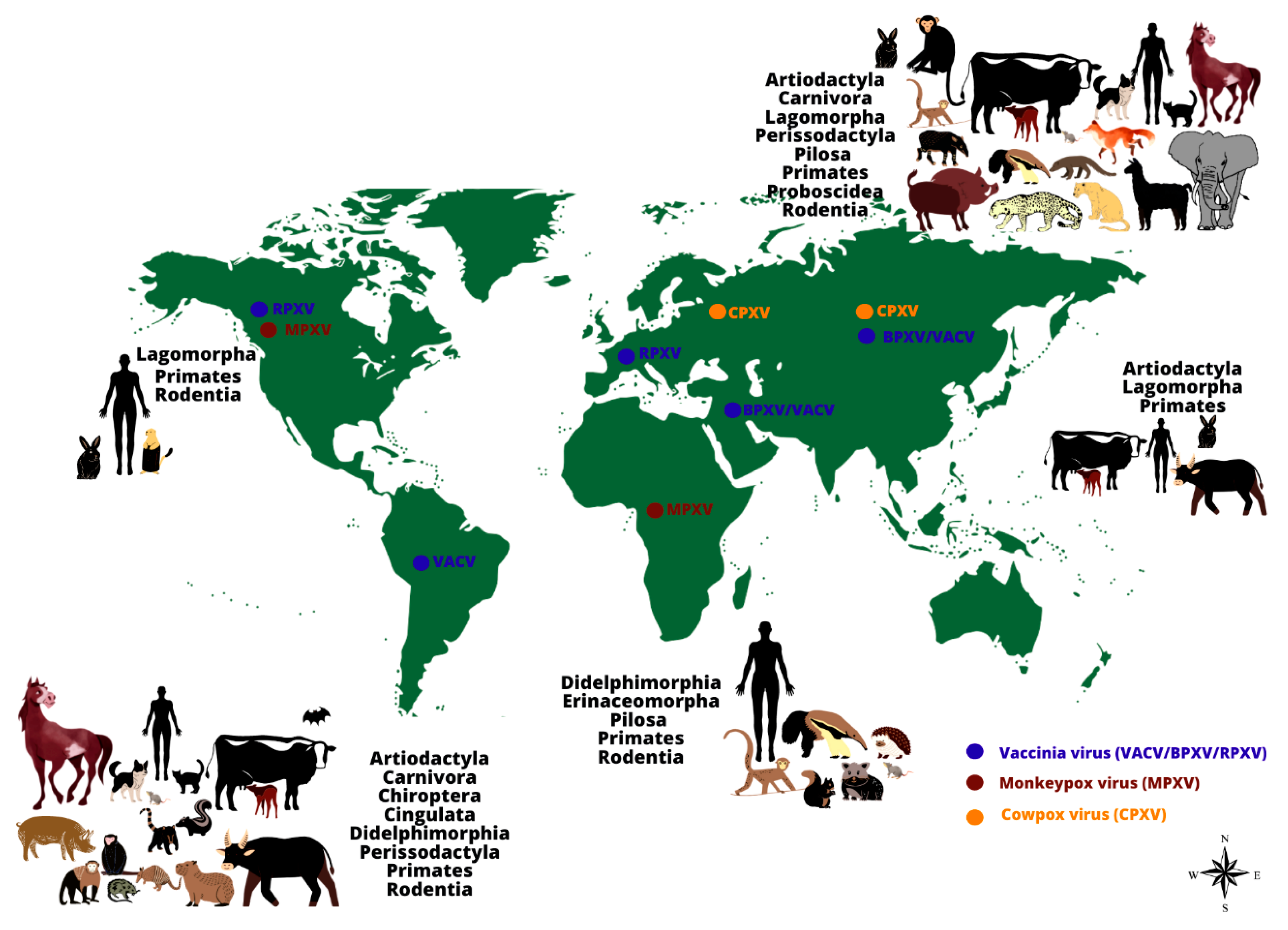
| Order/Family | Species | Method of Investigation * | Association to Human Infection ** |
|---|---|---|---|
| Primates/ Hominidae | Humans (Homo sapiens) | viral isolation | yes |
| Orangutans (Pongo pygmaeus) | viral isolation | yes | |
| Chimpanzees (Pan troglodytes) | viral isolation | no | |
| Primates/ Cercopithecidae | Sooty mangabeys (Cercocebus atys) | PCR/ viral isolation | no |
| Cynomolgus monkeys (Macaca fascicularis) | viral isolation | yes | |
| Primates/ Callithrichidae | White-tufted marmosets (Callithrix jacchus) | Lab. Infec. | no |
| Rodentia/Chinchillidae | Rabbits (Oryctolagus cuniculus) | Lab. Infec. | no |
| Rodentia/Muridae | Inbred mouses (Mus musculus) | Lab. Infec. | no |
| Rodentia/Cricetidae | hamsters | Lab. Infec. | no |
| Rodentia/Nesomyidae | Giant-pouched rats (Cricetomys sp.) | PCR/ viral isolation | no |
| Rodentia/Gliridae | African dormices (Graphiurus sp.) | PCR/ viral isolation | no |
| Rodentia/Sciuridae | Rope squirrels (Funisciurus sp.) | PCR/ viral isolation | yes |
| Black-tailed prairie dogs (Cynomys ludovicianus) | PCR | yes | |
| Woodchucks (Marmota monax) | PCR/ viral isolation | no | |
| Rodentia/ Dipodidae | Jerboas (Jaculus sp.) | PCR/ viral isolation | no |
| Rodentia/Hystricidae | Porcupines (Atherurus africanus) | PCR/ viral isolation | no |
| Pilosa/Macroscelididae | Ant-eaters (Myrmecophaga tridactyla) | viral Isolation | no |
| Didelphimorphia/ Didelphidae | Southern opossums (Didelphis marsupialis) | PCR/ viral isolation | no |
| Shot-tailed opossums (Monodelphis domestica) | PCR/ viral isolation | no | |
| Erinaceomorpha/ Erinaceidae | African hedgehogs (Atelerix sp.) | PCR/ viral isolation | no |
| Order/Family | Species | Method of Investigation * | Association to Human Infection ** |
|---|---|---|---|
| Primates/Hominidae | Humans (Homo sapiens) | virus isolation | no |
| Primates/Callithrichidae | White-tufted marmosets (Callithrix jacchus) | virus isolation | no |
| Primates/Cercopithecidae | Barbary macaques (Macaca sylvanus) | virus isolation | no |
| Cynomolgus macaques (Macaca fascicularis) | Lab. Infec. | no | |
| Rhesus macaques (Macaca mulata) | Lab. Infec. | no | |
| Carnivora/Felidae | Domestic cats (Felis catus) | virus isolation | yes |
| Cheetahs (Acinonyx jubatus) | virus isolation | yes | |
| Lions (Panthera leo) | virus isolation | no | |
| Pumas (Felis concolor) | virus isolation | no | |
| Black panthers (Panthera padus) | virus isolation | no | |
| Jaguarundis (Herpailurus yaguarondi) | virus isolation | no | |
| Jaguares (Felis onca) | virus isolation | no | |
| Carnivora/Canidae | Dogs (Canis lupus familiaris) | virus isolation | no |
| Foxes (Vulpes vulpes) | Lab. Infec. | no | |
| Carnivora/Herpestidae | Banded mongooses (Mungos mungo) | virus isolation | no |
| Carnivora/Ailuridae | Bearcats (Aiulurus fulgens) | virus isolation | no |
| Perissodactyla/ Rhinocerotidae | Black rhinoceros (Diceros bicornis) | virus isolation | no |
| White rhinoceros (Ceratotherium s. simum) | virus isolation | no | |
| Perissodactyla/Equidae | Horses (Equus caballus) | virus isolation | no |
| Artiodactyla/Bovidae | Cows (Bos taurus) | virus isolation | yes |
| Artiodactyla/Giraffidae | Okapis (Okapia johnstoni) | virus isolation | no |
| Artiodactyla/Camelidae | Lamas (Lama glama sp.) | virus isolation | no |
| Rodentia/Arvicolidae | Field voles (Microtus agrestis.) | virus isolation | no |
| Rodentia/Muridae | Brown rats (Rattus norvegicus) | virus isolation | yes |
| Giant gerbils (Rhombomys opimus) | virus isolation | no | |
| Rodentia/Cricetidae | Root voles (Microtus oeconomus) | virus isolation | no |
| Rodentia/Caviidae | Patagonian cavys (Dolichotis patagonum) | PCR | no |
| Rodentia/Castoridae | Beavers (Castor fibor canadensis) | virus isolation | no |
| Rodentia/Sciuridae | Ground squirrels (Citellus fulvus) | virus isolation | no |
| Proboscidea/ Elephantidae | Asian elephants (Elephas maximus) | virus isolation | yes |
| African elephants (Loxodonta africana) | virus isolation | no |
| Order/Family | Species | Method of Investigation * | Association to Human Infection ** |
|---|---|---|---|
| Artiodactyla/Bovidae | domestic buffaloes (Bubalus bubalis) | PCR/ Viral isolation | yes |
| Artiodactyla/Bovidae | cattle/cows (Bos taurus) | PCR/ Viral isolation | yes |
| Primates/Hominidae | Humans (Homo sapiens) | PCR/ Viral isolation | yes |
| Primates/Cebidae | Capuchin monkeys (Sapajus apella) | PCR | no |
| Primates/Atelidae | Black-howler monkeys (Alouatta caraya) | PCR | no |
| Didelphimorphia/Didelphidae | Black-eared possums (Didelphis aurita) | PCR | no |
| White-eared possums (Didelphis albiventris) | PCR | no | |
| Wooly-cuycas (Caluromys philander) | PCR | no | |
| Carnivora/Procyonidae | Ring-tailed coatis (Nasua nasua) | PCR | no |
| Carnivora/Felidae | Domestic cats (Felis catus) | PCR | no |
| Carnivora/Canidae | Domestic dogs (Canis familiaris) | PCR | no |
| Cingulata/Chlamyphoridae | Armadillos (Euphractus sexcintus) | PCR | no |
| Perissodactyla/Equidae | Horses (Equus ferus caballus) | PCR/ Viral isolation | yes |
| Donkeys (Equus africanus sp.) | PCR | yes | |
| Mules (Equus mulus) | PCR | yes | |
| Chiroptera/Molossidae | Black-molossus bats (Molossus rufus) | PCR | no |
| Broad-eared bats (Eumops perotis) | PCR | no | |
| Lagomorpha/Leporidae | Rabbits + | PCR | yes |
| Rodentia/Cricetidae | (Oryzomys spp.) | PCR/ Viral isolation | no |
| Black-footed colilargos (Oligoryzomys nigripes) | PCR | no | |
| Yellow pygmy rice rats (Oligoryzomys flavenscens) | PCR | no | |
| Rat-headed rice rats (Sooretamys angouya) | PCR | no | |
| Vesper mouses (Calomys spp.) | PCR | no | |
| Grass mouses (Akodon spp.) | PCR | no | |
| Hairy-tailed Bolo Mouses (Necromys Lasiurus) | PCR | no | |
| Bush mouses (Cerradomys subflavus) | PCR | no | |
| Rodentia/Echimyidae | Hairy Atlantic spiny rats (Trinomys setosus) | PCR | no |
| Rodentia/Muridae | Inbred-mouses (Mus musculus) | PCR/ Viral isolation | yes |
| Black-mouses (Rattus rattus) | PCR | no | |
| Rodentia/Caviidae | Capybaras (Hydrochoerus hydrochaeris) | PCR | no |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, N.I.O.; de Oliveira, J.S.; Kroon, E.G.; Trindade, G.d.S.; Drumond, B.P. Here, There, and Everywhere: The Wide Host Range and Geographic Distribution of Zoonotic Orthopoxviruses. Viruses 2021, 13, 43. https://doi.org/10.3390/v13010043
Silva NIO, de Oliveira JS, Kroon EG, Trindade GdS, Drumond BP. Here, There, and Everywhere: The Wide Host Range and Geographic Distribution of Zoonotic Orthopoxviruses. Viruses. 2021; 13(1):43. https://doi.org/10.3390/v13010043
Chicago/Turabian StyleSilva, Natalia Ingrid Oliveira, Jaqueline Silva de Oliveira, Erna Geessien Kroon, Giliane de Souza Trindade, and Betânia Paiva Drumond. 2021. "Here, There, and Everywhere: The Wide Host Range and Geographic Distribution of Zoonotic Orthopoxviruses" Viruses 13, no. 1: 43. https://doi.org/10.3390/v13010043
APA StyleSilva, N. I. O., de Oliveira, J. S., Kroon, E. G., Trindade, G. d. S., & Drumond, B. P. (2021). Here, There, and Everywhere: The Wide Host Range and Geographic Distribution of Zoonotic Orthopoxviruses. Viruses, 13(1), 43. https://doi.org/10.3390/v13010043






