Evaluation of HIV Transmission Clusters among Natives and Foreigners Living in Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Cohort Description
2.2. Study Population
2.3. Phylogenetic Analyses
2.4. Evaluation of HIV-1 Transmitted Drug Resistance
2.5. Statistical Analysis
2.6. Sequences Deposited to GenBank
3. Results
3.1. Patients’ Characteristics
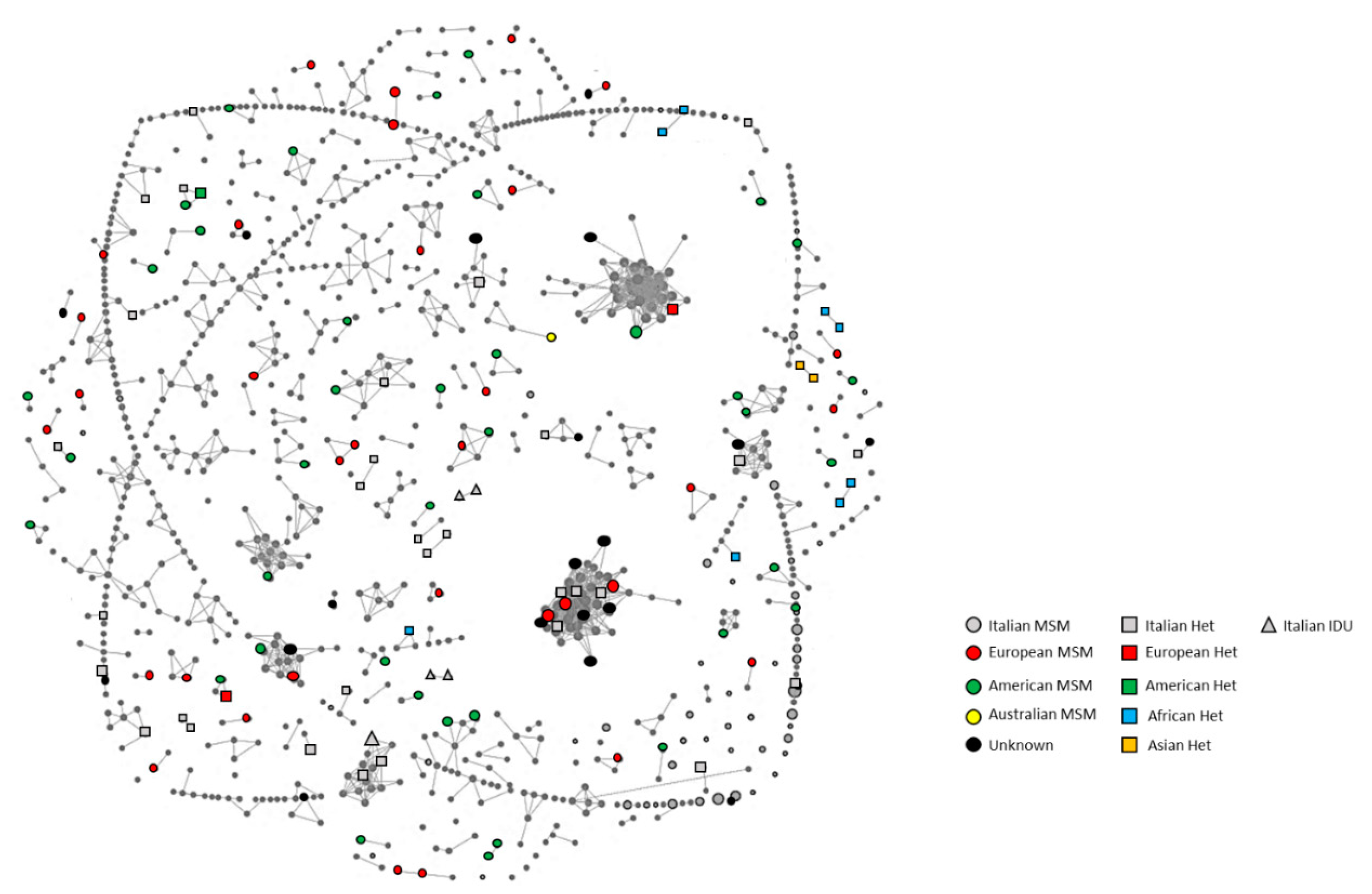
3.2. Cluster Identification
3.3. Epidemiological Characteristics and Factors Associated with MTCs
3.4. Characteristics of the Medium/Large Clusters
3.5. Characteristics of the Small Clusters
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The Joint United Nations Program on HIV/AIDS (UNAIDS). Available online: http://www.unaids.org/en (accessed on 28 April 2020).
- ECDC: Surveillance Report. HIV/AIDS Surveillance in Europe 2019 – 2018 Data. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/hiv-surveillance-report-2019.pdf (accessed on 28 April 2020).
- Regine, V.; Pugliese, L.; Boros, S.; Santaquilani, M.; Ferri, M.; Suligoi, B. Aggiornamento delle nuove diagnosi di infezione da HIV e dei casi di AIDS in Italia al 31 dicembre 2018. Notiziario ISS October 2019, 32:10. Available online: http://www.salute.gov.it/imgs/C_17_notizie_3963_0_file.pdf (accessed on 28 April 2020).
- Monno, L.; Brindicci, G.; Lo Caputo, S.; Punzi, G.; Scarabaggio, T.; Riva, C.; Di Bari, C.; Pierotti, P.; Saracino, A.; Lagioia, A.; et al. HIV-1 subtypes and circulating recombinant forms (CRFs) from HIV-infected patients residing in two regions of central and southern Italy. J. Med. Virol. 2005, 75, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Dauwe, K.; Mortier, V.; Schauvliege, M.; Van Den Heuvel, A.; Fransen, K.; Servais, J.Y.; Bercoff, D.P.; Seguin-Devaux, C.; Verhofstede, C. Characteristics and spread to the native population of HIV-1 non-B subtypes in two European countries with high migration rate. BMC Infect. Dis. 2015, 15, 524. [Google Scholar] [CrossRef] [PubMed]
- Fabeni, L.; Alteri, C.; Orchi, N.; Gori, C.; Bertoli, A.; Forbici, F.; Montella, F.; Pennica, A.; De Carli, G.; Giuliani, M.; et al. Recent transmission clustering of HIV-1 C and CRF17_BF strains characterized by NNRTI -Related mutations among newly diagnosed men in central Italy. PLoS ONE 2015, 10, e0135325. [Google Scholar] [CrossRef] [PubMed]
- Rossetti, B.; Di Giambenedetto, S.; Torti, C.; Postorino, M.C.; Punzi, G.; Saladini, F.; Gennari, W.; Borghi, V.; Monno, L.; Pignataro, A.R.; et al. Evolution of transmitted HIV-1 drug resistance and viral subtypes circulation in Italy from 2006 to 2016. HIV Med. 2018, 19, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Fabeni, L.; Alteri, C.; Berno, G.; Scutari, R.; Orchi, N.; De Carli, G.; Bertoli, A.; Carioti, L.; Gori, C.; Forbici, F.; et al. Characterisation of HIV-1 molecular transmission clusters among newly diagnosed individuals infected with non-B subtypes in Italy. Sex. Transm. Infect. 2019, 95, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Sagnelli, C.; Uberti-Foppa, C.; Bagaglio, S.; Cella, E.; Scolamacchia, V.; Hasson, H.; Salpietro, S.; Messina, E.; Morsica, G.; Angeletti, S.; et al. Molecular epidemiology of HIV-1 infection in immigrant population in northern Italy. Epidemiol. Infect. 2020, 148, e19. [Google Scholar] [CrossRef]
- Verhofstede, C.; Dauwe, K.; Fransen, K.; Van Laethem, K.; Van den Wijngaert, S.; Ruelle, J.; Delforge, M.L.; Vancutsem, E.; Vaira, D.; Stoffels, K.; et al. Phylogenetic analysis of the Belgian HIV-1 epidemic reveals that local transmission is almost exclusively driven by men having sex with men despite presence of large African migrant communities. Infect. Genet. Evol. 2018, 61, 36–44. [Google Scholar] [CrossRef]
- Brenner, B.G.; Roger, M.; Routy, J.P.; Moisi, D.; Ntemgwa, M.; Matte, C.; Baril, J.G.; Thomas, R.; Rouleau, D.; Bruneau, J.; et al. High rates of forward transmission events after acute/early HIV-1 infection. J. Infect. Dis. 2007, 195, 951–959. [Google Scholar] [CrossRef]
- Lai, A.; Bozzi, G.; Franzetti, M.; Binda, F.; Simonetti, F.R.; Micheli, V.; Meraviglia, P.; Corsi, P.; Bagnarelli, P.; De Luca, A.; et al. Phylogenetic analysis provides evidence of interactions between Italian heterosexual and South American homosexual males as the main source of national HIV-1 subtype C epidemics. J. Med. Virol. 2014, 86, 729–736. [Google Scholar] [CrossRef]
- Ragonnet-Cronin, M.; Lycett, S.J.; Hodcroft, E.B.; Hué, S.; Fearnhill, E.; Brown, A.E.; Delpech, V.; Dunn, D.; Leigh Brown, A.J.; United Kingdom HIV Drug Resistance Database. Transmission of Non-B HIV Subtypes in the United Kingdom Is Increasingly Driven by Large Non-Heterosexual Transmission Clusters. J. Infect. Dis. 2016, 213, 1410–1418. [Google Scholar] [CrossRef]
- Kostaki, E.G.; Soulie, C.; Visseaux, B.; Storto, A.; Charpentier, C.; Wirden, W.; Landman, R.; Katlama, C.; Calvez, V.; Descamps, D.; et al. Molecular Analysis Suggests Post-Migration HIV-1 Acquisition among Migrants in Paris. Virtual Conference on Retroviruses and Opportunistic Infections. Boston, Massachusetts, 2020. Abstract Number 921. Available online: https://www.croiconference.org/abstract/molecular-analysis-suggests-post-migration-hiv-1-acquisition-among-migrants-in-paris/ (accessed on 28 April 2020).
- Brenner, B.; Wainberg, M.A.; Roger, M. Phylogenetic inferences on HIV-1 transmission: Implications for the design of prevention and treatment interventions. AIDS 2013, 27, 1045–1057. [Google Scholar] [CrossRef] [PubMed]
- Beltrán, C.G.; Kostaki, E.G.; Carioti, L.; Alvarez, M.; Olalla, J.; Vidal Ampurdanes, M.C.; Montero, M.; García-Bujalance, S.; Blanco, J.R.; Rivero, M.; et al. HIV trace vs phylogenetic Analysis: Unraveling Transmission Clusters in Spain. Virtual Conference on Retroviruses and Opportunistic Infections. Boston, Massachusetts, 2020, Abstract Number 927. Available online: https://www.croiconference.org/abstract/hiv-trace-vs-phylogenetic-analysis-unraveling-transmission-clusters-in-spain/ (accessed on 28 April 2020).
- The Italian Cohort Naive Antiretroviral (ICONA) Foundation Study. Available online: http://www.fondazioneicona.org/ (accessed on 12 July 2020).
- Vourli, G.; Pharris, A.; Cazein, F.; Costagliola, D.; Dabis, F.; Del Amo, J.; Delpech, V.; Díaz, A.; Girardi, E.; Gourlay, A.; et al. Are European HIV cohort data within EuroCoord representative of the diagnosed HIV population? AIDS 2019, 33, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Fabeni, L.; Berno, G.; Fokam, J.; Bertoli, A.; Alteri, C.; Gori, C.; Forbici, F.; Takou, D.; Vergori, A.; Zaccarelli, M.; et al. Comparative Evaluation of Subtyping Tools for Surveillance of Newly Emerging HIV-1 Strains. J. Clin. Microbiol. 2017, 55, 2827–2837. [Google Scholar] [CrossRef]
- HIV-TRACE (Transmission Cluster Engine) Web Tool. Available online: http://hivtrace.datamonkey.org/hivtrace (accessed on 12 July 2020).
- Kosakovsky Pond, S.L.; Weaver, S.; Leigh Brown, A.J.; Wertheim, J.O. HIV-TRACE (TRAnsmission Cluster Engine): A Tool for Large Scale Molecular Epidemiology of HIV-1 and Other Rapidly Evolving Pathogens. Mol. Biol. Evol. 2018, 35, 1812–1819. [Google Scholar] [CrossRef]
- Wertheim, J.O.; Kosakovsky Pond, S.L.; Forgione, L.A.; Mehta, S.R.; Murrell, B.; Shah, S.; Smith, D.M.; Scheffler, K.; Torian, L.V. Social and Genetic Networks of HIV-1 Transmission in New York City. PLoS Pathog. 2017, 13, e1006000. [Google Scholar] [CrossRef] [PubMed]
- Hightower, G.K.; May, S.J.; Pérez-Santiago, J.; Pacold, M.E.; Wagner, G.A.; Little, S.J.; Richman, D.D.; Mehta, S.R.; Smith, D.M.; Pond, S.L. HIV-1 clade B pol evolution following primary infection. PloS ONE 2013, 8, e68188. [Google Scholar] [CrossRef]
- Poon, A.F.; Joy, J.B.; Woods, C.K.; Shurgold, S.; Colley, G.; Brumme, C.J.; Hogg, R.S.; Montaner, J.S.; Harrigan, P.R. The impact of clinical, demographic and risk factors on rates of HIV transmission: A population-based phylogenetic analysis in British Columbia, Canada. J. Infect. Dis. 2015, 211, 926–935. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef]
- Bennett, D.E.; Camacho, R.J.; Otelea, D.; Kuritzkes, D.R.; Fleury, H.; Kiuchi, M.; Heneine, W.; Kantor, R.; Jordan, M.R.; Schapiro, J.M.; et al. Drug resistance mutations for surveillance of transmitted HIV-1 drug-resistance: 2009 update. PLoS ONE 2009, 4, e4724. [Google Scholar] [CrossRef]
- Wensing, A.M.; Calvez, V.; Ceccherini-Silberstein, F.; Charpentier, C.; Günthard, H.F.; Paredes, R.; Shafer, R.W.; Richman, D.D. 2019 Update of the Drug Resistance Mutations in HIV-1. Top. Antivir. Med. 2019, 27, 111–121. [Google Scholar] [PubMed]
- Fakoya, I.; Álvarez-del Arco, D.; Woode-Owusu, M.; Monge, S.; Rivero-Montesdeoca, Y.; Delpech, V.; Rice, B.; Noori, T.; Pharris, A.; Amato-Gauci, A.J.; et al. A systematic review of post-migration acquisition of HIV among migrants from countries with generalised HIV epidemics living in Europe: Implications for effectively managing HIV prevention programmes and policy. BMC Public. Health. 2015, 15, 561. [Google Scholar] [CrossRef] [PubMed]
- Aldous, J.L.; Pond, S.K.; Poon, A.; Jain, S.; Qin, H.; Kahn, J.S.; Kitahata, M.; Rodriguez, B.; Dennis, A.M.; Boswell, S.L.; et al. Characterizing HIV transmission networks across the United States. Clin. Infect. Dis. 2012, 55, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Drescher, S.M.; von Wyl, V.; Yang, W.L.; Böni, J.; Yerly, S.; Shah, C.; Aubert, V.; Klimkait, T.; Taffé, P.; Furrer, H.; et al. Swiss HIV Cohort Study. Treatment-naive individuals are the major source of transmitted HIV-1 drug resistance in men who have sex with men in the Swiss HIV Cohort Study. Clin. Infect. Dis. 2014, 58, 285–294. [Google Scholar] [CrossRef]
- Brenner, B.G.; Ibanescu, R.I.; Hardy, I.; Stephens, D.; Otis, J.; Moodie, E.; Grossman, Z.; Vandamme, A.M.; Roger, M.; Wainberg, M.A. The Montreal PHI, SPOT cohorts. Large cluster outbreaks sustain the HIV epidemic among MSM in Quebec. AIDS 2017, 31, 707–717. [Google Scholar] [CrossRef]
- Lunar, M.M.; Mlakar, J.; Poljak, M. Increase in the numbers of HIV-1 non-B subtypes and potential recombinant forms circulating among Slovenian MSM in recent years. Virus Evol. 2017, 3 (Suppl. 1). [Google Scholar] [CrossRef]
- Patiño-Galindo, J.Á.; Torres-Puente, M.; Bracho, M.A.; Alastrué, I.; Juan, A.; Navarro, D.; Galindo, M.J.; Gimeno, C.; Ortega, E.; González-Candelas, F. Identification of a large, fast-expanding HIV-1 subtype B transmission cluster among MSM in Valencia, Spain. PLoS ONE 2017, 12, e0171062. [Google Scholar] [CrossRef]
- Parczewski, M.; Leszczyszyn-Pynka, M.; Witak-Jędra, M.; Szetela, B.; Gąsiorowski, J.; Knysz, B.; Bociąga-Jasik, M.; Skwara, P.; Grzeszczuk, A.; Jankowska, M.; et al. Expanding HIV-1 subtype B transmission networks among men who have sex with men in Poland. PLoS ONE 2017, 12, e0172473. [Google Scholar] [CrossRef]
- Stecher, M.; Chaillon, A.; Eberle, J.; Behrens, G.M.N.; Eis-Hübinger, A.M.; Lehmann, C.; Jablonka, A.; Bogner, J.; Fätkenheuer, G.; Spinner, C.D.; et al. Molecular Epidemiology of the HIV Epidemic in Three German Metropolitan Regions - Cologne/Bonn, Munich and Hannover, 1999-2016. Sci. Rep. 2018, 8, 6799. [Google Scholar] [CrossRef]
- Hué, S.; Brown, A.E.; Ragonnet-Cronin, M.; Lycett, S.J.; Dunn, D.T.; Fearnhill, E.; Dolling, D.I.; Pozniak, A.; Pillay, D.; Delpech, V.C.; et al. UK Collaboration on HIV Drug Resistance and the Collaborative HIV, Anti-HIV Drug Resistance Network (CHAIN). Phylogenetic analyses reveal HIV-1 infections between men misclassified as heterosexual transmissions. AIDS 2014, 28, 1967–1975. [Google Scholar] [CrossRef] [PubMed]
- Van de Laar, T.J.; Bezemer, D.; van Laethem, K.; Vandewalle, G.; de Smet, A.; van Wijngaerden, E.; Claas, E.C.; van Sighem, A.I.; Vandamme, A.M.; Compernolle, V.; et al. Phylogenetic evidence for underreporting of male-to-male sex among human immunodeficiency virus-infected donors in the Netherlands and Flanders. Transfusion 2017, 57, 1235–1247. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Ruan, Y.; Yin, L.; Vermund, S.H.; Shepherd, B.E.; Shao, Y.; Qian, H.Z. Sex with women among men who have sex with men in China: Prevalence and sexual practices. AIDS Patient Care STDS 2013, 27, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Bruhn, C.A.W.; Audelin, A.M.; Helleberg, M.; Bjorn-Mortensen, K.; Obel, N.; Gerstoft, J.; Nielsen, C.; Melbye, M.; Medstrand, P.; Gilbert, M.T.P.; et al. The origin and emergence of an HIV-1 epidemic: From introduction to endemicity. AIDS 2014, 28, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Lai, A.; Riva, C.; Marconi, A.; Balestrieri, M.; Razzolini, F.; Meini, G.; Vicenti, I.; Rosi, A.; Saladini, F.; Caramma, I.; et al. Changing patterns in HIV-1 non-B clade prevalence and diversity in Italy over three decades. HIV Med. 2010, 11, 593–602. [Google Scholar] [CrossRef] [PubMed]
- García, F.; Pérez-Cachafeiro, S.; Guillot, V.; Alvarez, M.; Pérez-Romero, P.; Pérez-Elías, M.J.; Viciana, I.; Blanco, J.R.; López-Dieguez, M.; de Mendoza, C.; et al. Transmission of HIV drug resistance and non-B subtype distribution in the Spanish cohort of antiretroviral treatment naïve HIV-infected individuals (CoRIS). Antiviral. Res. 2011, 91, 150–153. [Google Scholar] [CrossRef]
- Patiño Galindo, J.A.; Torres-Puente, M.; Gimeno, C.; Ortega, E.; Navarro, D.; Galindo, M.J.; Navarro, L.; Navarro, V.; Juan, A.; Belda, J.; et al. CRIVIH. Expansion of the CRF19_cpx variant in Spain. J. Clin. Virol. 2015, 69, 146–149. [Google Scholar] [CrossRef]
- Esbjörnsson, J.; Mild, M.; Audelin, A.; Fonager, J.; Skar, H.; Bruun Jørgensen, L.; Liitsola, K.; Björkman, P.; Bratt, G.; Gisslén, M.; et al. SPREAD/ESAR Programme. HIV-1 transmission between MSM and heterosexuals, and increasing proportions of circulating recombinant forms in the Nordic Countries. Virus Evol. 2016, 2, vew010. [Google Scholar] [CrossRef]
- Chaillon, A.; Essat, A.; Frange, P.; Smith, D.M.; Delaugerre, C.; Barin, F.; Ghosn, J.; Pialoux, G.; Robineau, O.; Rouzioux, C.; et al. Spatiotemporal dynamics of HIV-1 transmission in France (1999-2014) and impact of targeted prevention strategies. Retrovirol. 2017, 14, 15. [Google Scholar] [CrossRef]
- Fabeni, L.; Alteri, C.; Di Carlo, D.; Orchi, N.; Carioti, L.; Bertoli, A.; Gori, C.; Forbici, F.; Continenza, F.; Maffongelli, G.; et al. Dynamics and phylogenetic relationships of HIV-1 transmitted drug resistance according to subtype in Italy over the years 2000–14. J. Antimicrob. Chemother. 2017, 72, 2837–2845. [Google Scholar] [CrossRef]
- Stecher, M.; Chaillon, A.; Stephan, C.; Wasmuth, J.C.; Eberle, J.; Behrens, G.; Roider, J.; Spinner, C.D.; Müller, M.C.; Knops, E.; et al. The Translational Platform HIV (TPHIV). Non-B Subtypes HIV 1 Infections in Germany before and after the European Migrant Crisis. Virtual Conference on Retroviruses and Opportunistic Infections. Boston, Massachusetts, 2020, Abstract Number 919. Available online: https://www.croiconference.org/abstract/non-b-subtype-hiv-infections-in-germany-before-and-after-the-european-migrant-crisis/ (accessed on 28 April 2020).
- Pineda-Peña, A.C.; Schrooten, Y.; Vinken, L.; Ferreira, F.; Li, G.; Trovão, N.S.; Khouri, R.; Derdelinckx, I.; De Munter, P.; Kücherer, C.; et al. Trends and predictors of transmitted drug resistance (TDR) and clusters with TDR in a local Belgian HIV-1 epidemic. PLoS ONE 2014, 9, e101738. [Google Scholar] [CrossRef]
- Haddad, M.; Stawiski, E.; Benhamida, J.; Coakley, E. Improved Genotypic Algorithm for Predicting Etravirine Susceptibility: Comprehensive List of Mutations Identified through Correlation with Matched Phenotype. 17th Conference on Retroviruses and Opportunistic Infections. San Francisco, California, 2010, Abstract Number 574. Available online: http://hivandhepatitis.com/2010_conference/croi/posters/Haddad.pdf (accessed on 28 April 2020).
- Haddad, M.; Napolitano, L.A.; Paquet, A.C.; Evans, M.D.; Petropoulos, C.J.; Whitcomb, J.; Rimsky, L.; Vingerhoets, J.; Picchio, G.; Coakley, E. Impact of HIV-1 reverse transcriptase E138 mutations on rilpivirine drug susceptibility. International Workshop on HIV and Hepatitis Virus Drug Resistance and Curative Strategies. Los Cabos, Mexico. Antivir. Ther. 2011, 16 (Suppl. 1), A18. [Google Scholar]
- Tambuyzer, L.; Nijs, S.; Daems, B.; Picchio, G.; Vingerhoets, J. Effect of mutations at position E138 in HIV-1 reverse transcriptase on phenotypic susceptibility and virologic response to etravirine. J. Acquir. Immune. Defic. Syndr. 2011, 58, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Giannini, A.; Vicenti, I.; Materazzi, A.; Boccuto, A.; Dragoni, F.; Zazzi, M.; Saladini, F. The HIV-1 reverse transcriptase E138A natural polymorphism decreases the genetic barrier to resistance to etravirine in vitro. J. Antimicrob. Chemother. 2019, 74, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Bavaro, D.F.; Di Carlo, D.; Rossetti, B.; Bruzzone, B.; Vicenti, I.; Pontali, E.; Zoncada, A.; Lombardi, F.; Di Giambenedetto, S.; Borghi, V.; et al. Pretreatment HIV drug resistance and treatment failure in non-Italian HIV-1-infected patients enrolled in ARCA. Antivir. Ther. 2020. [Google Scholar] [CrossRef]
- Sluis-Cremer, N.; Jordan, M.R.; Huber, K.; Wallis, C.L.; Bertagnolio, S.; Mellors, J.W.; Parkin, N.T.; Harrigan, P.R. E138A in HIV-1 reverse transcriptase is more common in subtype C than B: Implications for rilpivirine use in resource-limited settings. Antiviral Res. 2014, 107, 31–34. [Google Scholar] [CrossRef]
- Rhee, S.Y.; Blanco, J.L.; Jordan, M.R.; Taylor, J.; Lemey, P.; Varghese, V.; Hamers, R.L.; Bertagnolio, S.; de Wit, T.F.; Aghokeng, A.F.; et al. Correction: Geographic and Temporal Trends in the Molecular Epidemiology and Genetic Mechanisms of Transmitted HIV-1 Drug Resistance: An Individual-Patient- and Sequence-Level Meta-Analysis. PLoS Med. 2015, 12, e1001845. [Google Scholar] [CrossRef]
- Machnowska, P.; Meixenberger, K.; Schmidt, D.; Jessen, H.; Hillenbrand, H.; Gunsenheimer-Bartmeyer, B.; Hamouda, O.; Kücherer, C.; Bannert, N.; German HIV-1 Seroconverter Study Group. Prevalence and persistence of transmitted drug resistance mutations in the German HIV-1 Seroconverter Study Cohort. PLoS ONE 2019, 14, e0209605. [Google Scholar] [CrossRef]
| Variables | Overall | Out of Cluster | In Cluster | p-Value a | Adjusted Model b | ||
|---|---|---|---|---|---|---|---|
| n = 3499 | 2773 (79.3%) | 726 (20.7%) | OR | (95% CI) | p-Value | ||
| Male gender, n (%) | 2872 (82.1%) | 2187 (78.9%) | 685 (94.3%) | <0.001 | - | - | - |
| Age, years, median (IQR) | 37 (30–45) | 38 (31–46) | 32 (27–40) | <0.001 | 0.65 | 0.59–0.72 | <0.001 |
| Mode of HIV transmission, n (%) | |||||||
| F heterosexual | 553 (15.8%) | 513 (18.5%) | 40 (5.5%) | <0.001 | 1.00 | - | - |
| F IVDU | 32 (0.9%) | 31 (1.1%) | 1 (0.1%) | 0.49 | 0.06–3.80 | 0.497 | |
| M heterosexual | 713 (20.4%) | 628 (22.7%) | 85 (11.7%) | 1.82 | 1.19–2.78 | 0.006 | |
| M IVDU | 161 (4.6%) | 145 (5.2%) | 16 (2.2%) | 1.52 | 0.80–2.90 | 0.204 | |
| MSM | 1789 (51.1%) | 1247 (45.0%) | 550 (75.8%) | 3.46 | 2.39–5.03 | <0.001 | |
| Other/unknown | 251 (7.2%) | 209 (7.5%) | 34 (4.7%) | 2.73 | 1.66–4.48 | <0.001 | |
| Nation of birth, n (%) | |||||||
| Italy | 2804 (80.1%) | 2160 (77.8%) | 644 (88.7%) | <0.001 | 1.00 | - | - |
| Africa | 219 (6.3%) | 212 (7.7%) | 7 (0.9%) | 0.18 | 0.08–0.39 | <0.001 | |
| Central and South America | 241 (6.9%) | 201 (7.3%) | 40 (5.5%) | 0.49 | 0.33–0.71 | 0.001 | |
| Europe | 187 (5.3%) | 159 (5.7%) | 28 (3.9%) | 0.29 | 0.08–0.97 | 0.045 | |
| Asia | 38 (1.1%) | 35 (1.3%) | 3 (0.4%) | 0.62 | 0.40–0.97 | 0.035 | |
| Other | 10 (0.3%) | 6 (0.2%) | 4 (0.6%) | 2.61 | 0.62–10.97 | 0.189 | |
| Education, n (%) | |||||||
| Primary school | 169 (4.8%) | 158 (5.7%) | 11 (1.5%) | <0.001 | 0.87 | 0.45–1.71 | 0.691 |
| Secondary school | 585 (16.7%) | 505 (18.2%) | 80 (11.0%) | 0.91 | 0.68–1.21 | 0.518 | |
| College/University | 1762 (50.4%) | 1329 (47.9%) | 433 (59.6%) | 1.00 | - | ||
| Unknown | 983 (28.1%) | 781 (28.2%) | 202 (27.8%) | 1.03 | 0.83–1.28 | 0.773 | |
| Employment, n (%) | |||||||
| Employed | 1476 (42.2%) | 1148 (41.4%) | 328 (45.2%) | <0.001 | 1.00 | - | - |
| Unemployed | 461 (13.2%) | 389 (14.0%) | 72 (9.9%) | 0.91 | 0.67–1.25 | 0.565 | |
| Self-employed | 526 (15.0%) | 413 (14.9%) | 113 (15.6) | 0.97 | 0.75–1.26 | 0.840 | |
| Student | 146 (4.2%) | 93 (3.4%) | 53 (7.3%) | 0.83 | 0.56–1.24 | 0.360 | |
| Housewife | 94 (2.7%) | 88 (3.2%) | 6 (0.8%) | 1.18 | 0.47–2.94 | 0.723 | |
| Other | 278 (7.9%) | 244 (8.8%) | 34 (4.7%) | 0.69 | 0.46–1.05 | 0.083 | |
| Unknown | 518 (14.8%) | 398 (14.3%) | 120 (16.5%) | 0.99 | 0.75–1.30 | 0.929 | |
| HIV RNA, copies/mL, n (%) | |||||||
| <1000 | 122 (3.5%) | 99 (3.6%) | 23 (3.2%) | 0.005 | 0.66 | 0.39–1.12 | 0.127 |
| 1000–10,000 | 559 (16.0%) | 445 (16.1%) | 114 (15.7%) | 0.81 | 0.61–1.08 | 0.147 | |
| 10,000–100,000 | 1470 (42.0%) | 1126 (40.6%) | 344 (47.4%) | 0.94 | 0.75–1.17 | 0.562 | |
| >100,000 | 1118 (32.0%) | 905 (32.6%) | 213 (29.3%) | 1.00 | - | ||
| Unknown | 230 (6.6%) | 198 (7.1%) | 32 (4.4%) | 0.76 | 0.38–1.55 | 0.451 | |
| CD4, cells/mm3, n (%) | |||||||
| ≤200 | 791 (22.6%) | 721 (26.0%) | 70 (9.6%) | <0.001 | 1.00 | - | - |
| 201–500 | 1485 (42.4%) | 1165 (42.0%) | 320 (44.1%) | 2.22 | 1.64–2.99 | <0.001 | |
| >500 | 1003 (28.7%) | 703 (25.4%) | 300 (41.3%) | 3.01 | 2.20–4.13 | <0.001 | |
| Unknown | 220 (6.3%) | 184 (6.6%) | 36 (5.0%) | 1.90 | 0.93–3.90 | 0.078 | |
| Year of diagnosis, median (IQR) | 2011 (2008–2014) | 2011 (2007–2014) | 2012 (2009–2014) | <0.001 | 1.09 | 1.06–1.11 | <0.001 |
| Subtype, n (%) | |||||||
| A1 | 104 (3.0%) | 85 (3.1%) | 19 (2.6%) | <0.001 | - | - | - |
| B | 2556 (73.1%) | 2038 (73.5%) | 518 (71.4%) | - | - | - | |
| C | 148 (4.2%) | 119 (4.3%) | 29 (4.0%) | - | - | - | |
| CRF02_AG | 187 (5.3%) | 141 (5.1%) | 46 (6.3%) | - | - | - | |
| CRF60_BC | 64 (1.8%) | 12 (0.4%) | 52 (7.2%) | - | - | - | |
| F1 | 179 (5.1%) | 157 (5.7%) | 22 (3.0%) | - | - | - | |
| Other | 261 (7.5%) | 221 (8.0%) | 40(5.5%) | - | - | - | |
| Transmitted drug resistance, n (%) | |||||||
| Any drug class | 501 (14.3) | 420 (15.1) | 81 (11.2) | 0.006 | - | - | - |
| NNRTI | 335 (9.6) | 281 (10.1) | 54 (7.4) | 0.028 | - | - | - |
| NRTI | 150 (4.3) | 134 (4.8) | 16 (2.2) | 0.002 | - | - | - |
| PI | 66 (1.9) | 53 (1.9) | 13 (1.8) | 0.832 | - | - | - |
| Cluster’s Characteristics | Small Clusters | Medium Clusters n = 36 | Large Clusters n = 6 | ||
|---|---|---|---|---|---|
| Overall | Clusters of 2 Persons | Clusters of 3 Persons | |||
| n = 186 | n = 156 | n = 30 | |||
| Sampling interval (min–max) | 2–3 | - | - | 4–9 | 10–52 |
| Age interval (min–max) | 19–65 | 19–65 | 20–56 | 18–53 | 18–58 |
| Mode of HIV transmission, n (%) | |||||
| Only MSM | 101 (54.3%) | 82 (52.5%) | 19 (63.3%) | 18 (50.0%) | - |
| Only heterosexual (F+M) | 13 (7.0%) | 13 (8.3%) | - | - | - |
| Only heterosexual (only F) | 4 (2.2%) | 4 (2.6%) | - | - | - |
| Only heterosexual (only M) | 5 (2.7%) | 5 (3.2%) | - | - | - |
| Only IVDU (F+M) | 1 (0.5%) | 1 (0.6%) | - | - | - |
| Only IVDU (only M) | 2 (1.1%) | 2 (1.3%) | - | - | - |
| Mixed (F+M) | 18 (9.7%) | 9 (5.8%) | 4 (13.3%) | 4 (11.1%) | 1 (16.7%) |
| Mixed (only M) | 42 (22.5%) | 40 (25.5%) | 7 (23.3%) | 14 (38.9%) | 5 (83.3%) |
| Nationality, n (%) | |||||
| Only Italians | 137 73.7%) | 122 (78.2%) | 15 (50.0%) | 24 (66.7%) | 2 (33.3%) |
| Italians and migrants | 43 (23.1%) | 28 (18.0%) | 15 (50.0%) | 12 (33.3%) | 4 (66.7%) |
| Only migrants | 6 (3.2%) | 6 (3.8%) | - | - | - |
| Subtype, n (%) | |||||
| A1 | 3 (1.6%) | 2 (1.2%) | 1 (3.3%) | 2 (5.6%) | - |
| B | 149 (80.1%) | 125 (80.1%) | 24 (80.0%) | 25 (69.3%) | 3 (50.0%) |
| C | 5 (2.7%) | 4 (2.6%) | 1 (3.3%) | 2 (5.6%) | 1 (16.7%) |
| CRF02_AG | 5 (2.7%) | 3 (1.9%) | 2 (6.7%) | 5 (13.9%) | 1 (16.7%) |
| CRF60_BC | - | - | - | - | 1 (16.7%) |
| F1 | 11 (5.9%) | 11 (7.1%) | - | - | - |
| Other | 13 (7.0%) | 11 (7.1%) | 2 (6.7%) | 2 (5.6%) | - |
| Resistance, n (%) | |||||
| None | 150 (80.7) | 123 (78.8) | 27 (90.0) | 31 (86.1) | 5 (83.3) |
| Any drug class | 36 (19.3) | 33 (21.2) | 3 (10.0) | 5 (13.9) | 1 (16.7) |
| NNRTI | 24 (12.9) | 22 (14.1) | 2 (6.7) | 3 (8.3) | 1 (16.7) |
| NRTI | 6 (3.2) | 5 (3.2) | 1 (3.3) | 1 (2.8) | 0 (0.0) |
| PI | 7 (3.8) | 7 (4.5) | 0 (0.0) | 1 (2.8) | 0 (0.0) |
| Medium MTCs (4–9 Sequences) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ID | Subtype | Cluster Size a n | Sampling Interval Year | HIV Diagnosis Interval Year | Migrants n (%) | Migrants’ Nationality n | Age, Years Median (IQR) | Risk Factor n | Genetic Distance Mean (SE) | Resistance |
| 1 | B | 4 | 2006–2007 | 2006–2007 | 0 (0.0) | - | 46 (44–46) | 4 MSM | 0.008 (0.002) | No |
| 2 | B | 4 | 2007–2012 | 2004–2012 | 0 (0.0) | - | 46 (42–50) | 1 F Het, 2 M Het, 1 M Unk | 0.010 (0.002) | No |
| 3 | B | 4 | 2008–2014 | 2008–2014 | 0 (0.0) | - | 52 (50–53) | 4 MSM | 0.008 (0.002) | No |
| 4 | B | 4 | 2008–2016 | 2008–2016 | 0 (0.0) | - | 39 (37–41) | 3 MSM, 1 M Unk | 0.006 (0.002) | No |
| 5 | B | 4 | 2009–2012 | 2009–2012 | 0 (0.0) | - | 54 (51–56) | 3 MSM, 1 F Het | 0.009 (0.002) | No |
| 6 | B | 4 | 2009–2016 | 2009–2016 | 0 (0.0) | - | 36 (33–41) | 3 MSM, 1 M Unk | 0.009 (0.002) | Yes (n = 4; RT: E138A) |
| 7 | B | 4 | 2010–2011 | 2010 | 1 (25.0) | 1 NA | 50 (46–52) | 4 MSM | 0.004 (0.002) | No |
| 8 | B | 4 | 2011–2013 | 2011–2013 | 0 (0.0) | - | 37 (35–38) | 4 MSM | 0.008 (0.002) | No |
| 9 | B | 4 | 2011–2017 | 2011–2017 | 0 (0.0) | - | 53 (48–56) | 4 MSM | 0.007 (0.002) | No |
| 10 | B | 4 | 2013–2014 | 2010–2014 | 0 (0.0) | - | 41 (34–48) | 4 MSM | 0.006 (0.002) | No |
| 11 | B | 4 | 2013–2018 | 2013–2018 | 1 (25.0) | 1 EE | 41 (37–43) | 3 MSM, 1 M Het | 0.008 (0.002) | No |
| 12 | B | 4 | 2016 | 2015–2016 | 0 (0.0) | - | 38 (28–49) | 4 MSM | 0.004 (0.001) | Yes (n = 1; PR: I85V) |
| 13 | B | 5 | 2007–2012 | 2007–2011 | 1 (20.0) | 1 NA | 38 (37–45) | 5 MSM | 0.006 (0.001) | No |
| 14 | B | 5 | 2008–2014 | 2008–2014 | 0 (0.0) | - | 36 (36–37) | 5 MSM | 0.011 (0.002) | No |
| 15 | B | 5 | 2010–2012 | 2009–2012 | 2 (40.0) | 1 CA, 1 EE | 41 (37–43) | 5 MSM | 0.008 (0.002) | No |
| 16 | B | 5 | 2011–2017 | 2008–2017 | 0 (0.0) | - | 38 (37–45) | 4 MSM, 1 M Het | 0.009 (0.002) | No |
| 17 | B | 5 | 2011–2017 | 2011–2017 | 0 (0.0) | - | 33 (32–35) | 5 MSM | 0.009 (0.002) | Yes (n = 5; RT: K101E) |
| 18 | B | 5 | 2013–2018 | 2013–2018 | 1 (20.0) | 1 SA | 38 (36–43) | 3 MSM, 2 M Het | 0.011 (0.002) | No |
| 19 | B | 6 | 2007–2011 | 2006–2011 | 0 (0.0) | - | 41 (36–48) | 6 MSM | 0.010 (0.002) | No |
| 20 | B | 6 | 2007–2011 | 2007–2011 | 0 (0.0) | - | 42 (37–45) | 5 MSM, 1 M Unk | 0.011 (0.002) | No |
| 21 | B | 6 | 2010–2014 | 2010–2014 | 0 (0.0) | - | 37 (35–43) | 5 MSM, 1 M Het | 0.011 (0.002) | No |
| 22 | B | 7 | 2006–2016 | 2006–2009 | 1 (14.3) | 1 SA | 43 (39–48) | 7 MSM | 0.010 (0.002) | No |
| 23 | B | 7 | 2009–2014 | 2008–2014 | 2 (28.6) | 2 SA | 51 (40–53) | 7 MSM | 0.011 (0.002) | No |
| 24 | B | 9 | 2010–2016 | 2010–2014 | 0 (0.0) | - | 40 (34–52) | 7 MSM, 1 M Het, 1 M Unk | 0.009 (0.002) | No |
| 25 | B | 9 | 2017–2018 | 2017–2018 | 0 (0.0) | - | 34 (31–41) | 8 MSM, 1 M Unk | 0.006 (0.001) | No |
| 26 | A1 | 4 | 2013–2015 | 2013–2015 | 0 (0.0) | - | 47 (45–50) | 4 MSM | 0.010 (0.002) | No |
| 27 | A1 | 8 | 2014–2016 | 2013–2016 | 1 (12.5) | 1 WE | 37 (33–39) | 8 MSM | 0.008 (0.002) | No |
| 28 | C | 4 | 2007–2011 | 2006–2011 | 1 (25.0) | 1 SA | 46 (44–47) | 3 MSM, 1 M Het | 0.007 (0.002) | No |
| 29 | C | 4 | 2012–2016 | 2012–2016 | 0 (0.0) | - | 50 (45–52) | 3 M Het, 1 M MSM | 0.009 (0.002) | No |
| 30 | CRF02_AG | 4 | 2009–2013 | 2009–2012 | 0 (0.0) | - | 35 (35–37) | 4 MSM | 0.007 (0.002) | No |
| 31 | CRF02_AG | 4 | 2009–2013 | 2009–2013 | 0 (0.0) | - | 38 (38–40) | 2 MSM, 1 F Het, 1 M Unk | 0.004 (0.001) | No |
| 32 | CRF02_AG | 4 | 2014 | 2014 | 1 (25.0) | 1 EE | 34 (30–39) | 3 MSM, 1 M Het | 0.009 (0.002) | Yes (n = 1; RT: E138A) |
| 33 | CRF02_AG | 5 | 2013–2014 | 2012–2014 | 0 (0.0) | - | 34 (33–35) | 4 MSM, 1 M Unk | 0.006 (0.002) | No |
| 34 | CRF02_AG | 7 | 2010–2017 | 2010–2016 | 0 (0.0) | - | 41 (36–51) | 6 MSM, 1 M Het | 0.005 (0.001) | No |
| 35 | CRF12_BF | 5 | 2014–2015 | 2013–2015 | 1 (20.0) | 1 SA | 34 (27–34) | 2 F Het; 2 M Het, 1 M Unk | 0.010 (0.002) | Yes (n = 3; RT: A62V) |
| 36 | CRF20_BG | 7 | 2013–2017 | 2013–2017 | 1 (14.3) | 1 Aus | 33 (32–37) | 7 MSM | 0.005 (0.001) | No |
| Large MTCs (≥10 Sequences) | ||||||||||
| 1 | B | 14 | 2008–2016 | 2008–2016 | 0 (0.0) | - | 40 (34–49) | 11 MSM, 2 M Het, 1 M IVDU | 0.010 (0.001) | No |
| 2 | B | 19 | 2007–2015 | 2007–2015 | 2 (10.5) | 1 EE, 1 SA | 37 (32–43) | 18 MSM, 1 M Unk | 0.016 (0.002) | No |
| 3 | B | 35 | 2009–2017 | 2009–2017 | 2 (5.7) | 1 WE, 1 SA | 34 (33–43) | 33 MSM, 1 M Het, 1 M Unk | 0.013 (0.002) | No |
| 4 | C | 10 | 2011–2016 | 2011–2016 | 0 (0.0) | - | 39 (35–42) | 8 MSM, 1 M Het, 1 M Unk | 0.009 (0.001) | No |
| 5 | CRF02_AG | 10 | 2006–2016 | 2006–2016 | 1 (10.0) | 1 CA | 42 (35–46) | 9 MSM, 1 M Het | 0.011 (0.002) | No |
| 6 | CRF60_BC | 52 | 2008–2018 | 2008–2018 | 5 (9.6) | 4 EE, 1 Unk | 34 (31–37) | 41 MSM, 2 F Het, 3 M Het, 6 M Unk | 0.012 (0.001) | Yes (n = 1; RT: E138A) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fabeni, L.; Santoro, M.M.; Lorenzini, P.; Rusconi, S.; Gianotti, N.; Costantini, A.; Sarmati, L.; Antinori, A.; Ceccherini-Silberstein, F.; d’Arminio Monforte, A.; et al. Evaluation of HIV Transmission Clusters among Natives and Foreigners Living in Italy. Viruses 2020, 12, 791. https://doi.org/10.3390/v12080791
Fabeni L, Santoro MM, Lorenzini P, Rusconi S, Gianotti N, Costantini A, Sarmati L, Antinori A, Ceccherini-Silberstein F, d’Arminio Monforte A, et al. Evaluation of HIV Transmission Clusters among Natives and Foreigners Living in Italy. Viruses. 2020; 12(8):791. https://doi.org/10.3390/v12080791
Chicago/Turabian StyleFabeni, Lavinia, Maria Mercedes Santoro, Patrizia Lorenzini, Stefano Rusconi, Nicola Gianotti, Andrea Costantini, Loredana Sarmati, Andrea Antinori, Francesca Ceccherini-Silberstein, Antonella d’Arminio Monforte, and et al. 2020. "Evaluation of HIV Transmission Clusters among Natives and Foreigners Living in Italy" Viruses 12, no. 8: 791. https://doi.org/10.3390/v12080791
APA StyleFabeni, L., Santoro, M. M., Lorenzini, P., Rusconi, S., Gianotti, N., Costantini, A., Sarmati, L., Antinori, A., Ceccherini-Silberstein, F., d’Arminio Monforte, A., Saracino, A., Girardi, E., & on behalf of the Icona Foundation Study Cohort. (2020). Evaluation of HIV Transmission Clusters among Natives and Foreigners Living in Italy. Viruses, 12(8), 791. https://doi.org/10.3390/v12080791






