Comprehensive Analysis of HERV Transcriptome in HIV+ Cells: Absence of HML2 Activation and General Downregulation of Individual HERV Loci
Abstract
1. Introduction
2. Materials and Methods
2.1. RNA-Sequencing Dataset
2.2. HERV Reference Database
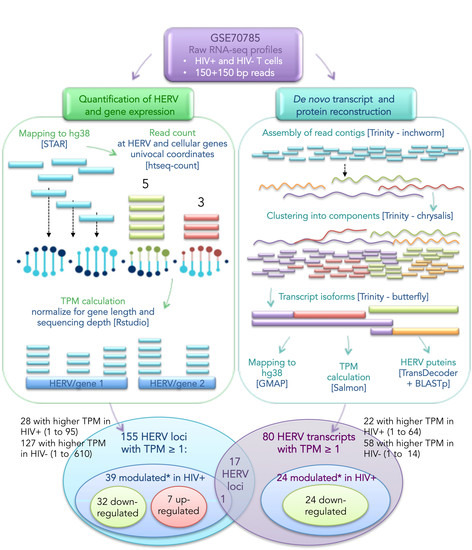
2.3. Bioinformatic Pipeline for HERV Expression Analysis
2.4. De Novo Trasnscript Reconstruction and Protein Prediction
3. Results
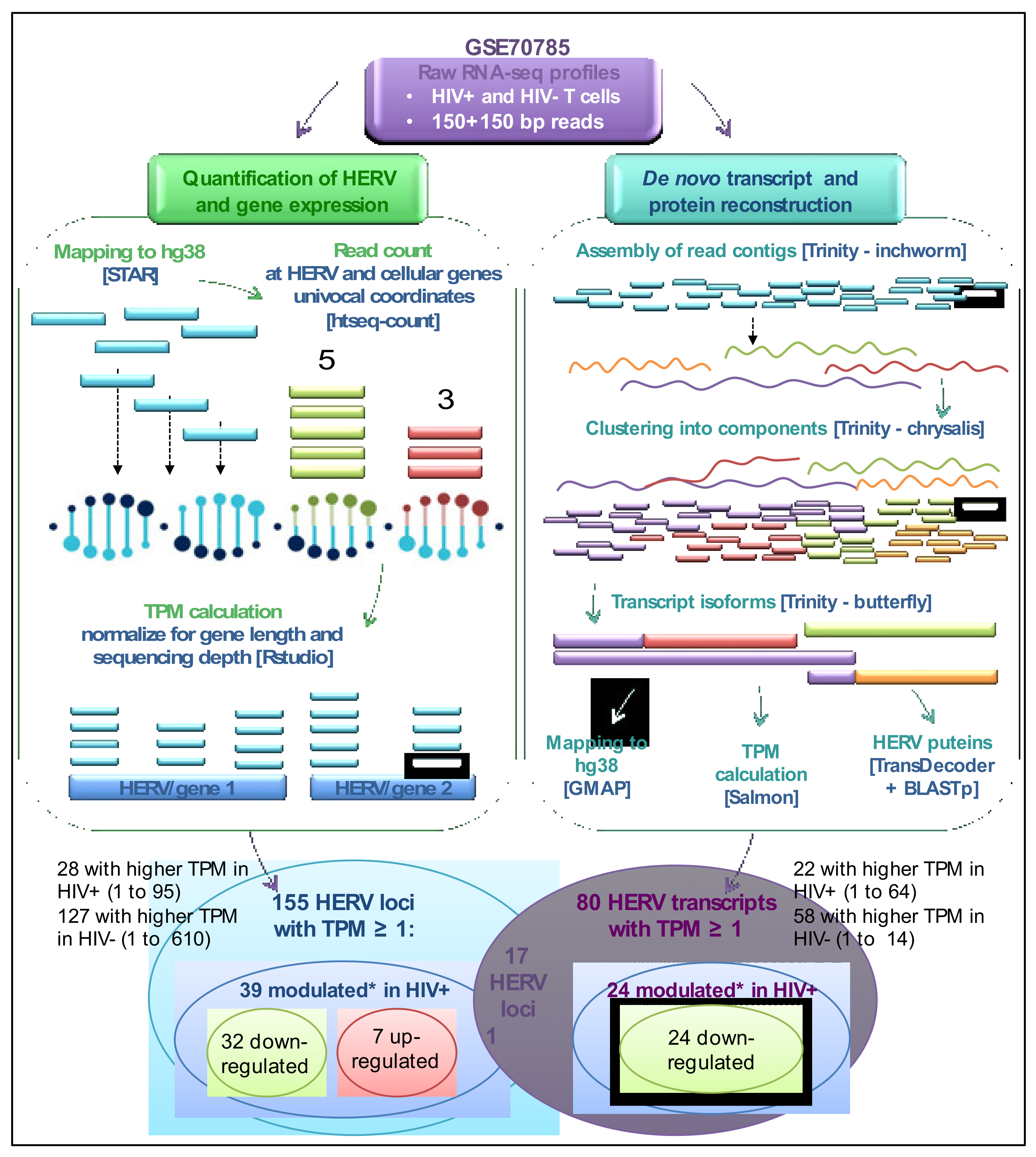
3.1. Landscape of HERV Expression in HIV-Infected and Uninfected T Cells
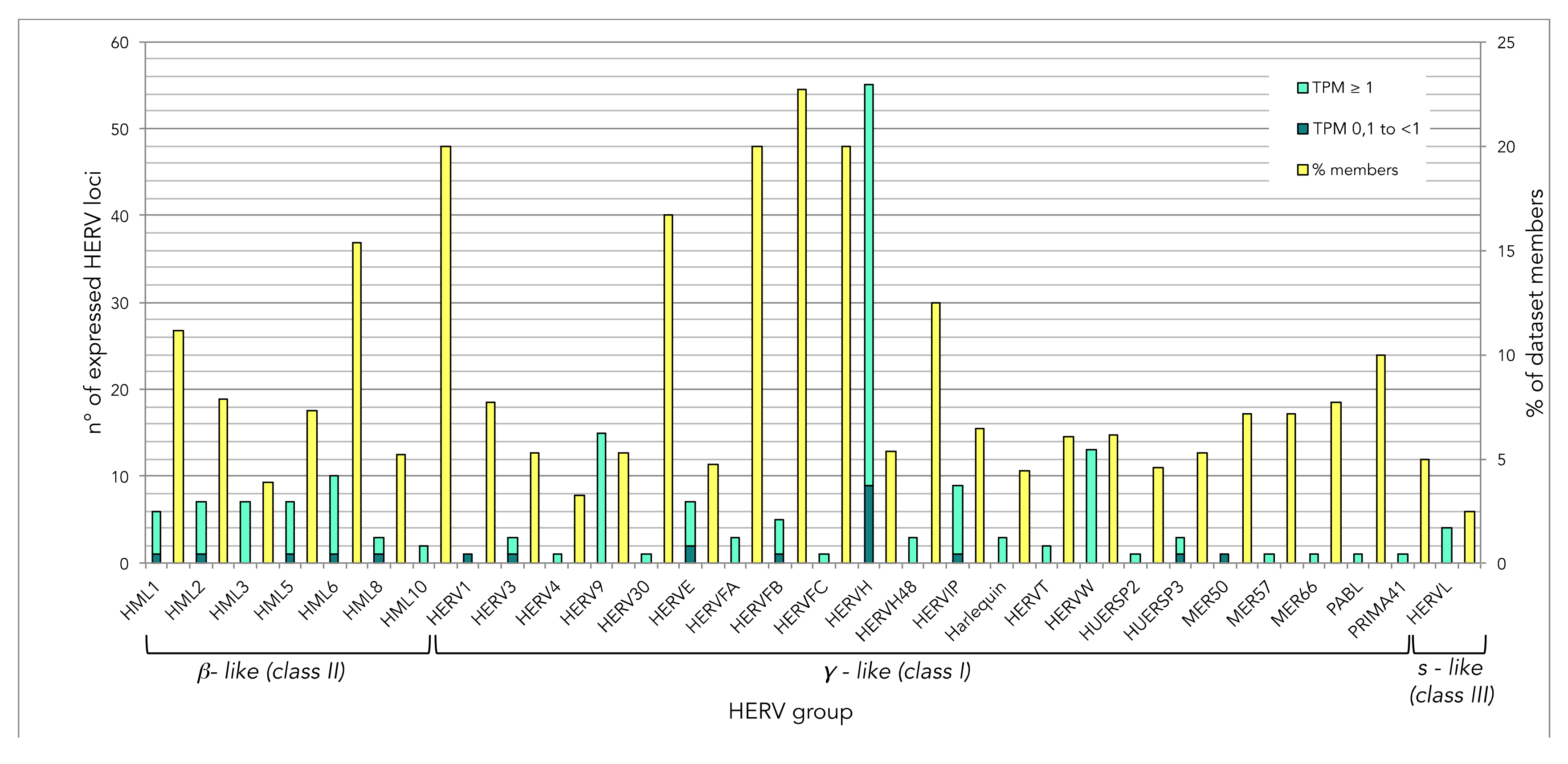
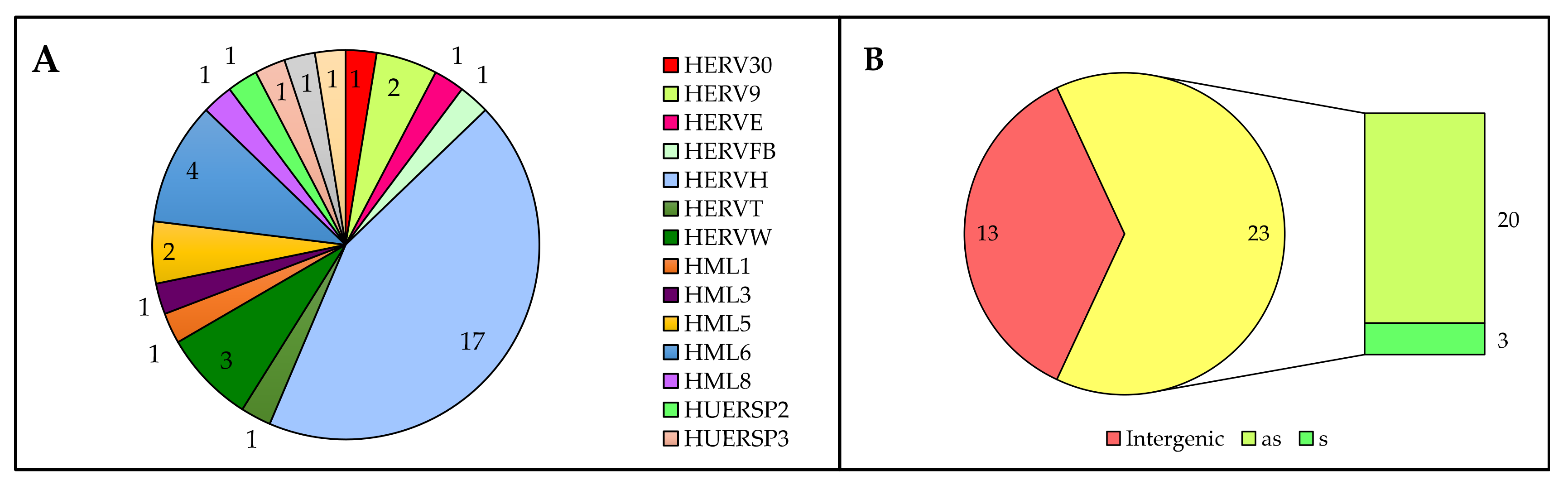
3.2. Analysis of Individual HERV Loci Modulated in the Presence of HIV Infection
3.3. Analysis of HERV-Derived Transcripts’ Expression
3.4. Identification of HERV-Derived Transcripts Modulated in the Presence of HIV Infection
3.5. Prediction of HERV-Encoded Putative Proteins
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grandi, N.; Pisano, M.P.; Tramontano, E. The emerging field of human endogenous retroviruses: Understanding their physiological role and contribution to diseases. Future Virol. 2019, 14, 441–444. [Google Scholar] [CrossRef]
- Grandi, N.; Tramontano, E. HERV envelope proteins: Physiological role and pathogenic potential in cancer and autoimmunity. Front. Microbiol. 2018, 9, 462. [Google Scholar] [CrossRef] [PubMed]
- Grandi, N.; Tramontano, E. Human endogenous retroviruses are ancient acquired elements still shaping innate immune responses. Front. Immunol. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Hurst, T.P.; Magiorkinis, G. Activation of the innate immune response by endogenous retroviruses. J. Gen. Virol. 2015, 96, 1207–1218. [Google Scholar] [CrossRef] [PubMed]
- Gürtler, C.; Bowie, A.G. Innate immune detection of microbial nucleic acids. Trends Microbiol. 2013, 21, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Melder, D.C.; Pankratz, V.S.; Federspiel, M.J. Evolutionary pressure of a receptor competitor selects different subgroup a avian leukosis virus escape variants with altered receptor interactions. J. Virol. 2003, 77, 10504–10514. [Google Scholar] [CrossRef]
- Spencer, T.E.; Mura, M.; Gray, C.A.; Griebel, P.J.; Palmarini, M. Receptor usage and fetal expression of ovine endogenous betaretroviruses: Implications for coevolution of endogenous and exogenous retroviruses. J. Virol. 2003, 77, 749–753. [Google Scholar] [CrossRef]
- Grandi, N.; Tramontano, E. Type W human endogenous retrovirus (HERV-W) integrations and their mobilization by L1 machinery: Contribution to the human transcriptome and impact on the host physiopathology. Viruses 2017, 9, 162. [Google Scholar] [CrossRef]
- Vargiu, L.; Rodriguez-Tomé, P.; Sperber, G.O.; Cadeddu, M.; Grandi, N.; Blikstad, V.; Tramontano, E.; Blomberg, J. Classification and characterization of human endogenous retroviruses; mosaic forms are common. Retrovirology 2016, 13, 7. [Google Scholar] [CrossRef]
- Subramanian, R.P.; Wildschutte, J.H.; Russo, C.; Coffin, J.M. Identification, characterization, and comparative genomic distribution of the HERV-K (HML-2) group of human endogenous retroviruses. Retrovirology 2011, 8, 90. [Google Scholar] [CrossRef]
- Towler, E.M.; Gulnik, S.V.; Bhat, T.N.; Xie, D.; Gustschina, E.; Sumpter, T.R.; Robertson, N.; Jones, C.; Sauter, M.; Mueller-Lantzsch, N.; et al. Functional characterization of the protease of human endogenous retrovirus, K10: Can it complement HIV-1 protease? Biochemistry 1998, 37, 17137–17144. [Google Scholar] [CrossRef]
- Brinzevich, D.; Young, G.R.; Sebra, R.; Ayllon, J.; Maio, S.M.; Deikus, G.; Chen, B.K.; Fernandez-Sesma, A.; Simon, V.; Mulder, L.C.F. HIV-1 Interacts with human endogenous retrovirus K (HML-2) envelopes derived from human primary lymphocytes. J. Virol. 2014, 88, 6213–6223. [Google Scholar] [CrossRef] [PubMed]
- Monde, K.; Contreras-Galindo, R.; Kaplan, M.H.; Markovitz, D.M.; Ono, A. Human endogenous retrovirus K Gag coassembles with HIV-1 Gag and reduces the release efficiency and infectivity of HIV-1. J. Virol. 2012, 86, 11194–11208. [Google Scholar] [CrossRef] [PubMed]
- Bhat, R.K.; Rudnick, W.; Antony, J.M.; Maingat, F.; Ellestad, K.K.; Wheatley, B.M.; Tönjes, R.R.; Power, C. Human endogenous Retrovirus-K(II) envelope induction protects neurons during HIV/AIDS. PLoS ONE 2014, 9, e97984. [Google Scholar] [CrossRef] [PubMed]
- Terry, S.N.; Manganaro, L.; Cuesta-Dominguez, A.; Brinzevich, D.; Simon, V.; Mulder, L.C.F. Expression of HERV-K108 envelope interferes with HIV-1 production. Virology 2017, 509, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Lawoko, A.; Johansson, B.; Rabinayaran, D.; Pipkorn, R.; Blomberg, J. Increased immunoglobulin G, but not M, binding to endogenous retroviral antigens in HIV-1 infected persons. J. Med. Virol. 2000, 62, 435–444. [Google Scholar] [CrossRef]
- Garrison, K.E.; Jones, R.B.; Meiklejohn, D.A.; Anwar, N.; Ndhlovu, L.C.; Chapman, J.M.; Erickson, A.L.; Agrawal, A.; Spotts, G.; Hecht, F.M.; et al. T cell responses to human endogenous retroviruses in HIV-1 infection. PLoS Pathog. 2007, 3, e165. [Google Scholar] [CrossRef]
- SenGupta, D.; Tandon, R.; Vieira, R.G.S.; Ndhlovu, L.C.; Lown-Hecht, R.; Ormsby, C.E.; Loh, L.; Jones, R.B.; Garrison, K.E.; Martin, J.N.; et al. Strong human endogenous retrovirus-specific T cell responses are associated with control of HIV-1 in chronic infection. J. Virol. 2011, 85, 6977–6985. [Google Scholar] [CrossRef]
- Ormsby, C.E.; SenGupta, D.; Tandon, R.; Deeks, S.G.; Martin, J.N.; Jones, R.B.; Ostrowski, M.A.; Garrison, K.E.; Vázquez-Pérez, J.A.; Reyes-Terán, G.; et al. Human endogenous retrovirus expression is inversely associated with chronic immune activation in HIV-1 infection. PLoS ONE 2012, 7, 4–10. [Google Scholar] [CrossRef]
- Jones, R.B.; Garrison, K.E.; Mujib, S.; Mihajlovic, V.; Aidarus, N.; Hunter, D.V.; Martin, E.; John, V.M.; Zhan, W.; Faruk, N.F.; et al. HERV-K—Specific T cells eliminate diverse HIV-1/2 and SIV primary isolates. J. Clin. Investig. 2012, 122, 4473–4489. [Google Scholar] [CrossRef]
- de Mulder, M.; SenGupta, D.; Deeks, S.G.; Martin, J.N.; Pilcher, C.D.; Hecht, F.M.; Sacha, J.B.; Nixon, D.F.; Michaud, H.-A. Anti-HERV-K (HML-2) capsid antibody responses in HIV elite controllers. Retrovirology 2017, 14, 41. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Galindo, R.; Kaplan, M.H.; Markovitz, D.M.; Lorenzo, E.; Yamamura, Y. Detection of HERV-K(HML-2) viral RNA in plasma of HIV type 1-infected individuals. AIDS Res. Hum. Retrovir. 2006, 22, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Galindo, R.; González, M.; Almodovar-Camacho, S.; González-Ramírez, S.; Lorenzo, E.; Yamamura, Y. A new Real-Time-RT-PCR for quantitation of human endogenous retroviruses type K (HERV-K) RNA load in plasma samples: Increased HERV-K RNA titers in HIV-1 patients with HAART non-suppressive regimens. J. Virol. Methods 2006, 136, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Galindo, R.; López, P.; Vélez, R.; Yamamura, Y. HIV-1 Infection Increases the Expression of Human Endogenous Retroviruses Type K (HERV-K) in Vitro. AIDS Res. Hum. Retrovir. 2007, 23, 116–122. [Google Scholar] [CrossRef]
- Gonzalez-Hernandez, M.J.; Swanson, M.D.; Contreras-Galindo, R.; Cookinham, S.; King, S.R.; Noel, R.J.; Kaplan, M.H.; Markovitz, D.M. Expression of human endogenous retrovirus type K (HML-2) is activated by the Tat protein of HIV-1. J. Virol. 2012, 86, 7790–7805. [Google Scholar] [CrossRef]
- Contreras-Galindo, R.; Kaplan, M.H.; Contreras-Galindo, A.C.; Gonzalez-Hernandez, M.J.; Ferlenghi, I.; Giusti, F.; Lorenzo, E.; Gitlin, S.D.; Dosik, M.H.; Yamamura, Y.; et al. Characterization of human endogenous retroviral elements in the blood of HIV-1-infected individuals. J. Virol. 2012, 86, 262–276. [Google Scholar] [CrossRef]
- Uleri, E.; Mei, A.; Mameli, G.; Poddighe, L.; Serra, C.; Dolei, A. HIV Tat acts on endogenous retroviruses of the W family and this occurs via Toll-like receptor4: Inference for neuroAIDS. AIDS 2014, 28, 2659–2670. [Google Scholar] [CrossRef]
- Esqueda, D.; Xu, F.; Moore, Y.; Yang, Z.; Huang, G.; Lennon, P.A.; Hu, P.C.; Dong, J. Lack of correlation between HERV-K expression and HIV-1 viral load in plasma specimens. Ann. Clin. Lab. Sci. 2013, 43, 122–125. [Google Scholar]
- Nali, L.H.S.; Oliveira, A.C.S.; Alves, D.O.; Caleiro, G.S.; Nunes, C.F.; Gerhardt, D.; Succi, R.C.M.; Romano, C.M.; Machado, D.M. Expression of human endogenous retrovirus K and W in babies. Arch. Virol. 2017, 162, 857–861. [Google Scholar] [CrossRef]
- Young, G.R.; Terry, S.N.; Manganaro, L.; Cuesta-Dominguez, A.; Deikus, G.; Bernal-Rubio, D.; Campisi, L.; Fernandez-Sesma, A.; Sebra, R.; Simon, V.; et al. HIV-1 Infection of Primary CD4+ T Cells Regulates the Expression of Specific Human Endogenous Retrovirus HERV-K (HML-2) Elements. J. Virol. 2018, 92, e01507-17. [Google Scholar] [CrossRef]
- Gonzalez-Hernandez, M.J.; Cavalcoli, J.D.; Sartor, M.; Contreras-Galindo, R.; Meng, F.; Dai, M.; Dube, D.; Saha, K.; Gitlin, S.D.; Omenn, G.S.; et al. Regulation of the Human Endogenous Retrovirus K (HML-2) Transcriptome by the HIV-1 Tat Protein. J. Virol. 2014, 88, 8924–8935. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, N.; Maldarelli, F.; Mellors, J.; Coffin, J.M. HIV-1 infection leads to increased transcription of HERV-K (HML-2) proviruses in vivo but not to increased virion production. J. Virol. 2014, 88, 11108–11120. [Google Scholar] [CrossRef] [PubMed]
- Grandi, N.; Cadeddu, M.; Blomberg, J.; Tramontano, E. Contribution of type W human endogenous retroviruses to the human genome: Characterization of HERV—W proviral insertions and processed pseudogenes. Retrovirology 2016, 13, 67. [Google Scholar] [CrossRef] [PubMed]
- Grandi, N.; Cadeddu, M.; Pisano, M.P.; Esposito, F.; Blomberg, J.; Tramontano, E. Identification of a novel HERV-K(HML10): Comprehensive characterization and comparative analysis in non-human primates provide insights about HML10 proviruses structure and diffusion. Mob. DNA 2017, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Pisano, M.P.; Grandi, N.; Cadeddu, M.; Blomberg, J.; Tramontano, E. Comprehensive characterization of the HERV-K(HML-6): Overview of their structure, phylogeny and contribution to the human genome. J. Virol. 2019, 93, e00110-19. [Google Scholar] [CrossRef] [PubMed]
- Grandi, N.; Cadeddu, M.; Blomberg, J.; Mayer, J.; Tramontano, E. HERV-W group evolutionary history in non-human primates: Characterization of ERV-W orthologs in Catarrhini and related ERV groups in Platyrrhini. BMC Evol. Biol. 2018, 18, 1–14. [Google Scholar] [CrossRef]
- Grandi, N.; Pisano, M.P.; Demurtas, M.; Blomberg, J.; Magiorkinis, G.; Mayer, J.; Tramontano, E. Identification and characterization of ERV-W-like sequences in Platyrrhini species provides new insights into the evolutionary history of ERV-W in primates. Mob. DNA 2020, 11, 1–16. [Google Scholar] [CrossRef]
- Gupta, A.; Titus Brown, C.; Zheng, Y.H.; Adami, C. Differentially-expressed pseudogenes in HIV-1 infection. Viruses 2015, 7, 5191–5205. [Google Scholar] [CrossRef]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq-A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef]
- Harrow, J.; Frankish, A.; Gonzalez, J.M.; Tapanari, E.; Diekhans, M.; Kokocinski, F.; Aken, B.L.; Barrell, D.; Zadissa, A.; Searle, S.; et al. GENCODE: The reference human genome annotation for The ENCODE Project. Genome Res. 2012, 22, 1760–1774. [Google Scholar] [CrossRef] [PubMed]
- Rstudio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2016. [Google Scholar]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644. [Google Scholar] [CrossRef] [PubMed]
- Haas, B.J.; Papanicolaou, A.; Yassour, M.; Grabherr, M.; Blood, P.D.; Bowden, J.; Couger, M.B.; Eccles, D.; Li, B.; Lieber, M.; et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 2013, 8, 1494–1512. [Google Scholar] [CrossRef] [PubMed]
- Patro, R.; Duggal, G.; Love, M.I.; Irizarry, R.A.; Kingsford, C. Salmon: Fast and bias-aware quantification of transcript expression using dual-phase inference. Nat. Methods 2017, 14, 417. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.D.; Watanabe, C.K. GMAP: A genomic mapping and alignment program for mRNA and EST sequences. Bioinformatics 2005, 21, 1859–1875. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Sperber, G.; Airola, T.; Jern, P.; Blomberg, J. Automated recognition of retroviral sequences in genomic data—RetroTector©. Nucleic Acids Res. 2007, 35, 4964–4976. [Google Scholar] [CrossRef]
- Thorvaldsdóttir, H.; Robinson, J.T.; Mesirov, J.P. Integrative Genomics Viewer (IGV): High-performance genomics data visualization and exploration. Brief. Bioinform. 2013, 14, 178–192. [Google Scholar] [CrossRef]
- Peng, X.; Sova, P.; Green, R.R.; Thomas, M.J.; Korth, M.J.; Proll, S.; Xu, J.; Cheng, Y.; Yi, K.; Chen, L.; et al. Deep sequencing of HIV-infected cells: Insights into nascent transcription and host-directed therapy. J. Virol. 2014, 88, 8768–8782. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, J.; White, K.P. RNA-seq differential expression studies: More sequence or more replication? Bioinformatics 2014, 30, 301–304. [Google Scholar] [CrossRef]
- Kent, W.J. BLAT—The BLAST-like alignment tool. Genome Res. 2002, 12, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Hubley, R.; Finn, R.D.; Clements, J.; Eddy, S.R.; Jones, T.A.; Bao, W.; Smit, A.F.A.; Wheeler, T.J. The Dfam database of repetitive DNA families. Nucleic Acids Res. 2016, 44, D81–D89. [Google Scholar] [CrossRef] [PubMed]
- Marchler-Bauer, A.; Bo, Y.; Han, L.; He, J.; Lanczycki, C.J.; Lu, S.; Chitsaz, F.; Derbyshire, M.K.; Geer, R.C.; Gonzales, N.R.; et al. CDD/SPARCLE: Functional classification of proteins via subfamily domain architectures. Nucleic Acids Res. 2017, 45, D200–D203. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Galindo, R.; Almodóvar-Camacho, S.; González-Ramírez, S.; Lorenzo, E.; Yamamura, Y. Comparative longitudinal studies of HERV-K and HIV-1 RNA titers in HIV-1-infected patients receiving successful versus unsuccessful highly active antiretroviral therapy. AIDS Res. Hum. Retrovir. 2007, 23, 1083–1086. [Google Scholar] [CrossRef] [PubMed]
- Vincendeau, M.; Göttesdorfer, I.; Schreml, J.M.H.; Wetie, A.G.N.; Mayer, J.; Greenwood, A.D.; Helfer, M.; Kramer, S.; Seifarth, W.; Hadian, K.; et al. Modulation of human endogenous retrovirus (HERV) transcription during persistent and de novo HIV-1 infection. Retrovirology 2015, 12, 27. [Google Scholar] [CrossRef]
- Gray, L.R.; Jackson, R.E.; Jackson, P.E.H.; Bekiranov, S.; Rekosh, D.; Hammarskjöld, M.L. HIV—1 Rev interacts with HERV—K RcREs present in the human genome and promotes export of unspliced HERV—K proviral RNA. Retrovirology 2019, 16, 40. [Google Scholar] [CrossRef]
- Ledda, A.; Caocci, G.; Spinicci, G.; Cocco, E.; Mamusa, A.; La Nasa, G. Two new cases of acute promyelocytic leukemia following mitoxantrone treatment in patients with multiple sclerosis. Leukemia 2006, 20, 2217–2218. [Google Scholar] [CrossRef][Green Version]
- Seifarth, W.; Frank, O.; Zeilfelder, U.; Spiess, B.; Greenwood, A.D.; Hehlmann, R.; Leib-Mösch, C. Comprehensive analysis of human endogenous retrovirus transcriptional activity in human tissues with a retrovirus-specific microarray. J. Virol. 2005, 79, 341–352. [Google Scholar] [CrossRef]

| ID 1 | Group | TPM HIV- | TPM HIV+ | FC | Coordinates (hg38) | St. | Colocalized Genes 2 | TPM HIV- | TPM HIV+ | FC |
|---|---|---|---|---|---|---|---|---|---|---|
| HERV loci upregulated in HIV+ cells | ||||||||||
| 4594 | HERVH | 0.12 | 1.45 | 11.9 | 19:5548576-5553212 | + | ||||
| 3166 | HERVH | 0.12 | 1.07 | 8.9 | 9:131547214-131551936 | + | PRRT1B (+) | 0.04 | 0.21 | 4.9 |
| 2307 | HERV30 | 0.35 | 1.90 | 5.4 | 6:118617102-118626802 | + | CEP85L (-) | 0.34 | 0.49 | 1.4 |
| 2082 | HERV9 | 0.25 | 1.38 | 5.4 | 6:16455730-16464625 | + | ATXN1 (-) | 0.04 | 0.03 | 1.2 |
| 4457 | HML6 | 0.31 | 1.56 | 4.9 | 17:43872528-43874324 | + | ||||
| 1744 | HERVH | 2.91 | 10.11 | 3.5 | 5:702471-708501 | + | ||||
| 3112 | PABL | 0.45 | 1.32 | 3.0 | 9:94265146-94270221 | - | ZNF169 (+) | 0.01 | 0.01 | - |
| HERV loci downregulated in HIV+ cells | ||||||||||
| 691 | HML6 | 1.97 | 0.10 | 19.2 | 2:136071818-136077261 | - | ||||
| 5407 | HML6 | 2.29 | 0.17 | 13.1 | X:57102981-57109396 | + | ||||
| 2658 | HERVH | 2.52 | 0.21 | 12.1 | 7:143105117-143110504 | + | ||||
| 4741 | HERVH | 22.77 | 2.16 | 10.5 | 19:46102204-46109974 | - | ||||
| 2476 | HML3 | 3.21 | 0.32 | 10.1 | 7:43859699-43864985 | - | ||||
| 3788 | HERVH | 3.39 | 0.39 | 8.8 | 12:48111344-48115676 | - | PFKM (+) | 8.66 | 4.88 | 1.8 |
| 6q27b | HERVW | 4.33 | 0.55 | 7.9 | 6:166891083-166898253 | + | AL159163.1 (-) | 0.00 | 0.00 | - |
| 4511 | MER57 | 1.07 | 0.15 | 7.1 | 18:13700682-13708050 | + | FAM210A (-) | 0.11 | 0.07 | 1.5 |
| 5387 | HML6 | 41.38 | 6.53 | 6.3 | X:53159114-53163826 | - | ||||
| 3637 | HUERSP2 | 2.23 | 0.35 | 6.3 | 11:108198480-108204806 | + | ||||
| 933 | HERVH | 3.55 | 0.58 | 6.1 | 3:44344658-44350393 | - | TCAIM (+) | 0.28 | 0.17 | 1.6 |
| 957 | HERV9 | 1.78 | 0.29 | 6.1 | 3:51742261-51747980 | + | ||||
| 5815 | HERVFB | 39.41 | 6.51 | 6.1 | 1:8921052-8927320 | + | ||||
| 4329 | HERVH | 1.56 | 0.29 | 5.4 | 15:101072487-101078293 | + | LRRK1 (+) | 0.18 | 0.17 | 1.08 |
| 3436 | HUERSP3 | 5.73 | 1.16 | 5.0 | 11:34897477-34907140 | + | APIP (-) | 0.68 | 0.52 | 1.3 |
| 1113 | HERVH | 3.24 | 0.70 | 4.6 | 3:128959818-128965400 | + | KIAA1257 (-) | 1.83 | 1.46 | 1.3 |
| 4374 | HERVT | 13.98 | 3.07 | 4.5 | 16:35267618-35277076 | - | ||||
| 1407 | HERVH | 8.37 | 1.84 | 4.5 | 4:54020079-54025546 | + | CHIC2 (-) | 0.05 | 0.00 | - |
| 5356 | HML8 | 2.70 | 0.59 | 4.5 | X:46486517-46490287 | - | KRBOX4 (+) | 0.07 | 0.06 | 1.2 |
| 2334 | HERVH | 18.93 | 4.45 | 4.3 | 6:130192311-130198222 | + | SAMD3 (-) | 0.23 | 0.16 | 1.4 |
| 3887 | HML5 | 1.85 | 0.46 | 4.0 | 12:100159365-100165461 | - | GOLGA2P5 (-) | 0.78 | 0.23 | 3.5 |
| 18p11.21 | HERVW | 1.95 | 0.48 | 4.0 | 18:4681680-4692409 | + | ||||
| Xp11.21 | HERVW | 2.47 | 0.63 | 3.9 | X:7699169-7704068 | - | ||||
| 2219 | HML1 | 5.49 | 1.45 | 3.8 | 6:78854118-78858747 | + | ||||
| 1549 | HML5 | 1.75 | 0.46 | 3.8 | 4:106109837-106114668 | + | TBCK (-) | 2.58 | 1.56 | 1.7 |
| 4475 | HERVH | 1.16 | 0.32 | 3.6 | 17:55467773-55476512 | + | SMIM36 (-) | 0.00 | 0.00 | - |
| 3370 | HERVH | 4.52 | 1.29 | 3.5 | 10:120844932-120850561 | + | WDR11-AS1 (-) | 0.29 | 0.14 | 2.1 |
| 1975 | HERVH | 14.08 | 4.34 | 3.2 | 5:124966686-124972226 | - | LINC02240 (+) | 0.01 | 0.01 | - |
| 3701 | HERVH | 8.68 | 2.68 | 3.2 | 12:9962437-9968690 | - | CLEC12A (+) | 0.20 | 0.13 | 1.6 |
| 5705 | HERVH | 7.88 | 2.53 | 3.1 | X:131712025-131717764 | + | FIRRE (-) | 0.00 | 0.00 | - |
| 3544 | HERVE | 2.09 | 0.69 | 3.0 | 11:71886034-71890084 | + | AP002495.1 (-) | 0.04 | 0.00 | - |
| 4610 | HERVH | 1.96 | 0.64 | 3.0 | 19:9635997-9642066 | - | ZNF561-AS1 (+) | 0.21 | 0.54 | 2.6 |
| HERV Locus Information | Transcript Description | Transcript in HIV- Cells | Transcript in HIV+ Cells | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| ID 1 | Group | Coloc. Genes | TPM HIV- | TPM HIV+ | Trinity ID | nt | FC | read n° | TPM | read n° | TPM |
| 19q13.2a | HERV-W | ZNF780A | 83.78 | 94.62 | DN62359_c2_g9_i3 * | 378 | 3.6 | 18 | 1.29 | 5 | 0.35 |
| 4444 | HERV-E | SLFN12L | 214.46 | 74.94 | DN67670_c4_g2_i6 | 420 | 3.3 | 77 | 4.56 | 22 | 1.37 |
| DN67943_c3_g5_i5 | 512 | 3.3 | 91 | 3.84 | 26 | 1.15 | |||||
| DN67943_c3_g2_i1 | 889 | 3.1 | 106 | 1.93 | 33 | 0.63 | |||||
| DN59238_c1_g1_i11 | 264 | 3.5 | 15 | 2.18 | 4 | 0.62 | |||||
| DN60987_c8_g9_i1 | 442 | 3.1 | 75 | 4.07 | 23 | 1.31 | |||||
| DN67799_c2_g4_i2 | 431 | 3.8 | 23 | 1.29 | 6 | 0.34 | |||||
| DN61824_c3_g12_i1 | 372 | 3.5 | 60 | 4.39 | 16 | 1.24 | |||||
| DN67943_c3_g5_i8 | 629 | 5.1 | 60 | 1.82 | 11 | 0.36 | |||||
| DN57253_c4_g1_i1 | 257 | 12.3 | 13 | 2.01 | 1 | 0.16 | |||||
| DN64887_c2_g1_i10 | 339 | 5.7 | 99 | 8.71 | 16 | 1.52 | |||||
| DN67670_c4_g1_i22 | 1025 | 4.4 | 112 | 1.69 | 24 | 0.38 | |||||
| 2384 | HERV-FA | TMEM181 | 124.57 | 58.95 | DN59786_c3_g6_i2 | 260 | 6.1 | 13 | 1.96 | 2 | 0.32 |
| DN59236_c4_g1_i1 | 229 | 8.5 | 9 | 1.81 | 1 | 0.21 | |||||
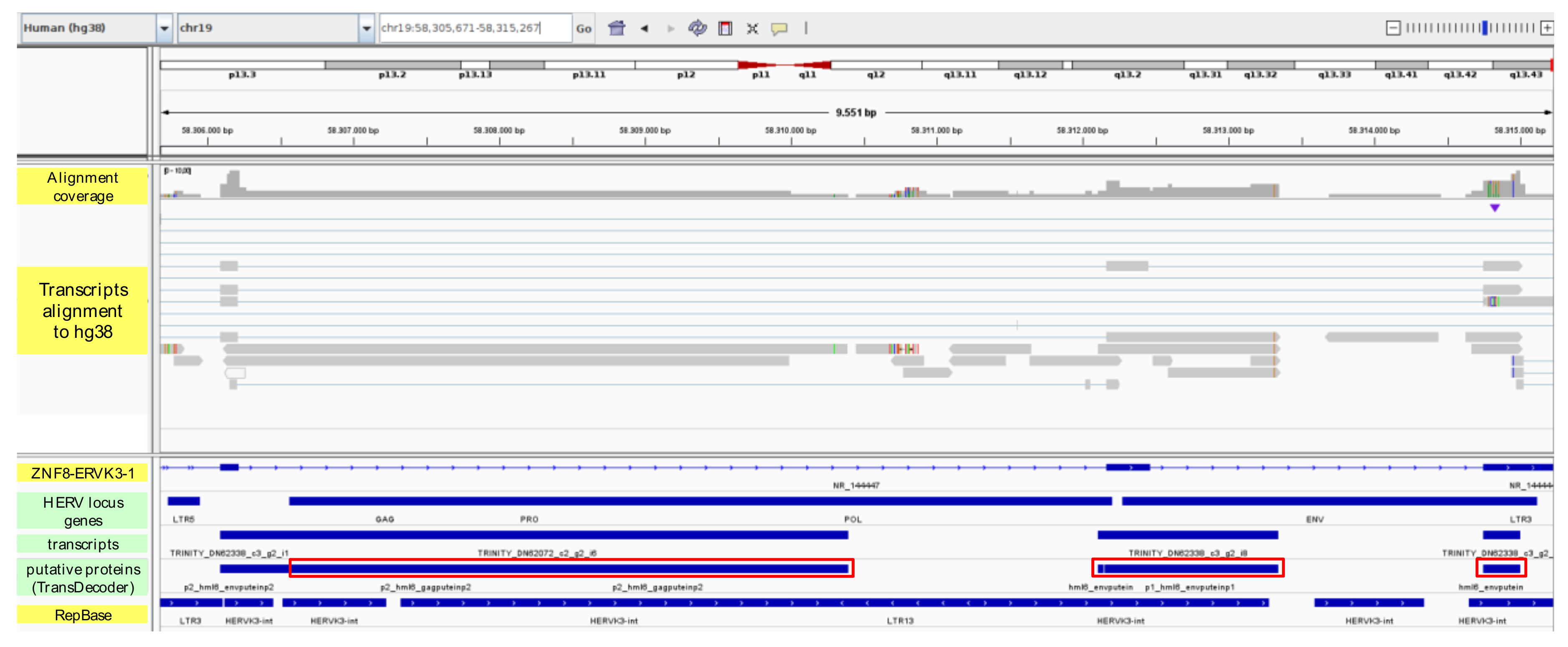
| 4796 | HML6 | ZNF8 - ERVK3-1 | 22.68 | 28.20 | DN62338_c3_g2_i2 | 838 | 9.5 | 58 | 1.14 | 6 | 0.12 |
| 4618 | HERVE | ZNF700 | 23.27 | 8.57 | DN62411_c1_g3_i5 | 513 | 4.5 | 24 | 1.01 | 5 | 0.22 |
| 3651 | HERVH | DSCAML1 | 31.76 | 14.32 | DN60751_c2_g1_i13 | 340 | 4.6 | 13 | 1.14 | 3 | 0.25 |
| 14q32.11 | HERVW | DGLUCY | 14.06 | 7.88 | DN64353_c11_g2_i6 * | 1431 | 3.3 | 157 | 1.54 | 45 | 0.47 |
| 3955 | HERVH | - | 6.18 | 4.33 | DN61830_c1_g1_i9 | 324 | 3.7 | 16 | 1.49 | 4 | 0.40 |
| 2334 | HERVH | SAMD3 | 18.93 | 4.45 | DN67393_c0_g6_i1 | 229 | 4.7 | 10 | 2.01 | 2 | 0.43 |
| DN60366_c3_g1_i11 | 369 | 5.4 | 17 | 1.27 | 3 | 0.24 | |||||
| HERV Locus ID1 | HERV Group | Colocalized Gene | Transcript ID | HIV- TPM | HIV+ TPM | Protein Match | Q Size | Q Start | Q End |
|---|---|---|---|---|---|---|---|---|---|
| 6074 | HML5 | - | DN57819_c2_g11_i2 | 0.95 | 0.56 | hml5_gagputein | 108 | 0 | 96 |
| 3651 | HERV-H | DSCAML1 | DN60751_c2_g1_i13 | 1.14 | 0.25 | HERVH_ polputein | 112 | 25 | 108 |
| 4796 | HML6 | ZNF8 - ERVK3-1 | DN62072_c2_g2_i6 | 2.42 | 1.68 | hml6_gagputein | 188 | 6 | 134 |
| DN62338_c3_g2_i2 | 1.14 | 0.12 | hml6_envputein | 99 | 1 | 35 | |||
| DN62338_c3_g2_i6 | 41.79 | 63.59 | hml6_envputein | 109 | 49 | 97 | |||
| DN62338_c3_g2_i8 | 2.29 | 2.30 | hml6_envputein | 157 | 56 | 157 | |||
| 4655 | HML3 | ZNF493 | DN62722_c3_g4_i3 | 0.12 | 0.99 | hml3_polputein | 193 | 6 | 155 |
| 4444 | HERV-E | SLFN12L | DN67943_c3_g2_i1 | 1.93 | 0.63 | harlequin_envputein | 132 | 0 | 107 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grandi, N.; Pisano, M.P.; Scognamiglio, S.; Pessiu, E.; Tramontano, E. Comprehensive Analysis of HERV Transcriptome in HIV+ Cells: Absence of HML2 Activation and General Downregulation of Individual HERV Loci. Viruses 2020, 12, 481. https://doi.org/10.3390/v12040481
Grandi N, Pisano MP, Scognamiglio S, Pessiu E, Tramontano E. Comprehensive Analysis of HERV Transcriptome in HIV+ Cells: Absence of HML2 Activation and General Downregulation of Individual HERV Loci. Viruses. 2020; 12(4):481. https://doi.org/10.3390/v12040481
Chicago/Turabian StyleGrandi, Nicole, Maria Paola Pisano, Sante Scognamiglio, Eleonora Pessiu, and Enzo Tramontano. 2020. "Comprehensive Analysis of HERV Transcriptome in HIV+ Cells: Absence of HML2 Activation and General Downregulation of Individual HERV Loci" Viruses 12, no. 4: 481. https://doi.org/10.3390/v12040481
APA StyleGrandi, N., Pisano, M. P., Scognamiglio, S., Pessiu, E., & Tramontano, E. (2020). Comprehensive Analysis of HERV Transcriptome in HIV+ Cells: Absence of HML2 Activation and General Downregulation of Individual HERV Loci. Viruses, 12(4), 481. https://doi.org/10.3390/v12040481