Lower Broadly Neutralizing Antibody Responses in Female Versus Male HIV-1 Infected Injecting Drug Users
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Study Population and Phenotype
2.3. Neutralization Assays
2.4. Statistical Analysis
2.5. Diversity Analysis
3. Results
3.1. Broadly Neutralizing Antibody Responses in Injecting Drug Users
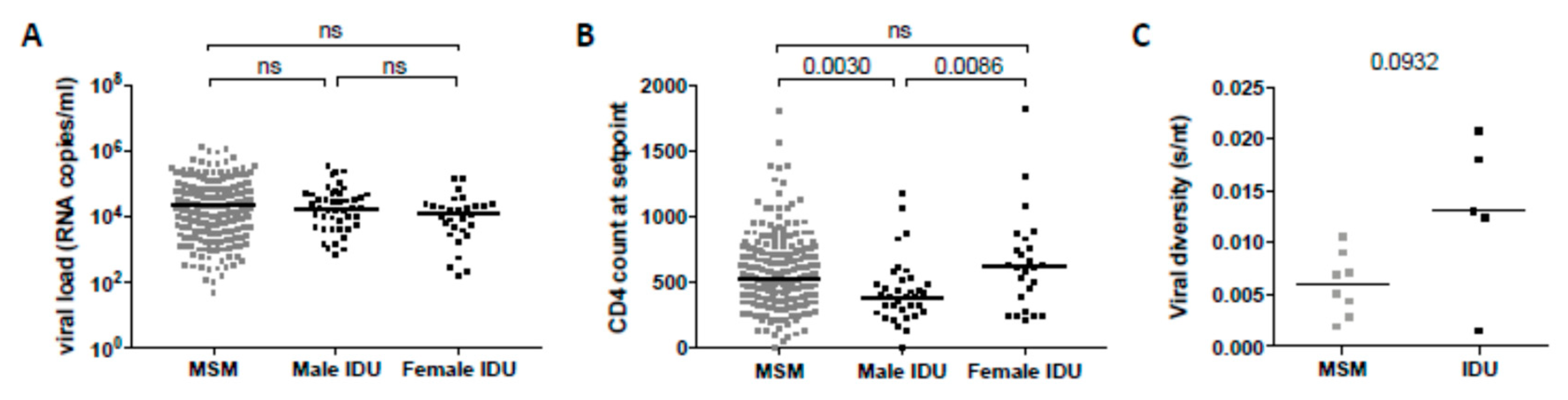
3.2. Clinical Factors Associated with the Development of Broadly Neutralizing Antibody Responses
3.3. Broadly Neutralizing Antibody Responses in the Swiss 4.5K Screen
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mascola, J.R.; Montefiori, D.C. The role of antibodies in HIV vaccines. Annu. Rev. Immunol. 2010, 28, 413–444. [Google Scholar] [CrossRef]
- McElrath, M.J.; Haynes, B.F. Induction of immunity to human immunodeficiency virus type-1 by vaccination. Immunity 2010, 33, 542–554. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.M.; Burton, D.R. Rational antibody-based HIV-1 vaccine design: Current approaches and future directions. Curr. Opin. Immunol. 2010, 22, 358–366. [Google Scholar] [CrossRef]
- Wei, X.; Decker, J.M.; Wang, S.; Hui, H.; Kappes, J.C.; Wu, X.; Salazar-Gonzalez, J.F.; Salazar, M.G.; Kilby, J.M.; Saag, M.S.; et al. Antibody neutralization and escape by HIV-1. Nature 2003, 422, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Richman, D.D.; Wrin, T.; Little, S.J.; Petropoulos, C.J. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc. Natl. Acad. Sci. USA 2003, 100, 4144–4149. [Google Scholar] [CrossRef] [Green Version]
- Euler, Z.; van Gils, M.J.; Bunnik, E.M.; Phung, P.; Schweighardt, B.; Wrin, T.; Schuitemaker, H. Cross-reactive neutralizing humoral immunity does not protect from HIV type 1 disease progression. J. Infect. Dis. 2010, 201, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Sather, D.N.; Armann, J.; Ching, L.K.; Mavrantoni, A.; Sellhorn, G.; Caldwell, Z.; Yu, X.; Wood, B.; Self, S.; Kalams, S.; et al. Factors associated with the development of cross-reactive neutralizing antibodies during human immunodeficiency virus type 1 infection. J. Virol. 2009, 83, 757–769. [Google Scholar] [CrossRef]
- Simek, M.D.; Rida, W.; Priddy, F.H.; Pung, P.; Carrow, E.; Laufer, D.S.; Lehrman, J.K.; Boaz, M.; Tarragona-Fiol, T.; Miiro, G.; et al. HIV-1 Elite Neutralizers: Individuals with Broad and Potent Neutralizing Activity Identified Using a High Throughput Neutralization Assay Together with an Analytical Selection Algorithm. J. Virol. 2009, 83, 7337–7348. [Google Scholar] [CrossRef]
- van den Kerkhof, T.L.; Euler, Z.; van Gils, M.J.; Boeser-Nunnink, B.D.; Schuitemaker, H.; Sanders, R.W. Early development of broadly reactive HIV-1 neutralizing activity in elite neutralizers. AIDS 2014, 28, 1237–1240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Binley, J.M.; Lybarger, E.A.; Crooks, E.T.; Seaman, M.S.; Gray, E.; Davis, K.L.; Decker, J.M.; Wycuff, D.; Harris, L.; Hawkins, N.; et al. Profiling the specificity of neutralizing antibodies in a large panel of plasmas from patients chronically infected with human immunodeficiency virus type 1 subtypes B and C. J. Virol. 2008, 82, 11651–11668. [Google Scholar] [CrossRef] [PubMed]
- Doria-Rose, N.A.; Klein, R.M.; Daniels, M.G.; O’Dell, S.; Nason, M.; Lapedes, A.; Bhattacharya, T.; Migueles, S.A.; Wyatt, R.T.; Korber, B.T.; et al. Breadth of human immunodeficiency virus-specific neutralizing activity in sera: Clustering analysis and association with clinical variables. J. Virol. 2010, 84, 1631–1636. [Google Scholar] [CrossRef]
- Euler, Z.; van den Kerkhof, T.L.; van Gils, M.J.; Burger, J.A.; Edo-Matas, D.; Phung, P.; Wrin, T.; Schuitemaker, H. Longitudinal analysis of early HIV-1-specific neutralizing activity in an elite neutralizer and in five patients who developed cross-reactive neutralizing activity. J. Virol. 2012, 86, 2045–2055. [Google Scholar] [CrossRef]
- Hraber, P.; Seaman, M.S.; Bailer, R.T.; Mascola, J.R.; Montefiori, D.C.; Korber, B.T. Prevalence of broadly neutralizing antibody responses during chronic HIV-1 infection. AIDS 2014, 28, 163–169. [Google Scholar] [CrossRef] [Green Version]
- Landais, E.; Huang, X.; Havenar-Daughton, C.; Murrell, B.; Price, M.A.; Wickramasinghe, L.; Ramos, A.; Bian, C.B.; Simek, M.; Allen, S.; et al. Broadly Neutralizing Antibody Responses in a Large Longitudinal Sub-Saharan HIV Primary Infection Cohort. PLoS Pathog. 2016, 12, e1005369. [Google Scholar] [CrossRef]
- Rusert, P.; Kouyos, R.D.; Kadelka, C.; Ebner, H.; Schanz, M.; Huber, M.; Braun, D.L.; Hoze, N.; Scherrer, A.; Magnus, C.; et al. Determinants of HIV-1 broadly neutralizing antibody induction. Nat. Med. 2016, 22, 1260–1267. [Google Scholar] [CrossRef] [Green Version]
- Gray, E.S.; Madiga, M.C.; Hermanus, T.; Moore, P.L.; Wibmer, C.K.; Tumba, N.L.; Werner, L.; Mlisana, K.; Sibeko, S.; Williamson, C.; et al. The neutralization breadth of HIV-1 develops incrementally over four years and is associated with CD4+ T cell decline and high viral load during acute infection. J. Virol. 2011, 85, 4828–4840. [Google Scholar] [CrossRef]
- Mikell, I.; Sather, D.N.; Kalams, S.A.; Altfeld, M.; Alter, G.; Stamatatos, L. Characteristics of the earliest cross-neutralizing antibody response to HIV-1. PLoS Pathog. 2011, 7, e1001251. [Google Scholar] [CrossRef]
- van Gils, M.J.; Sanders, R.W. In vivo protection by broadly neutralizing HIV antibodies. Trends Microbiol. 2014, 22, 550–551. [Google Scholar] [CrossRef]
- Mascola, J.R.; Stiegler, G.; VanCott, T.C.; Katinger, H.; Carpenter, C.B.; Hanson, C.E.; Beary, H.; Hayes, D.; Frankel, S.S.; Birx, D.L.; et al. Protection of macaques against vaginal transmission of a pathogenic HIV-1/SIV chimeric virus by passive infusion of neutralizing antibodies. Nat. Med. 2000, 6, 207–210. [Google Scholar] [CrossRef]
- Burton, D.R.; Hessell, A.J.; Keele, B.F.; Klasse, P.J.; Ketas, T.A.; Moldt, B.; Dunlop, D.C.; Poignard, P.; Doyle, L.A.; Cavacini, L.; et al. Limited or no protection by weakly or nonneutralizing antibodies against vaginal SHIV challenge of macaques compared with a strongly neutralizing antibody. Proc. Natl. Acad. Sci. USA 2011, 108, 11181–11186. [Google Scholar] [CrossRef] [Green Version]
- Hessell, A.J.; Poignard, P.; Hunter, M.; Hangartner, L.; Tehrani, D.M.; Bleeker, W.K.; Parren, P.W.; Marx, P.A.; Burton, D.R. Effective, low-titer antibody protection against low-dose repeated mucosal SHIV challenge in macaques. Nat. Med. 2009, 15, 951–954. [Google Scholar] [CrossRef] [Green Version]
- Hessell, A.J.; Rakasz, E.G.; Poignard, P.; Hangartner, L.; Landucci, G.; Forthal, D.N.; Koff, W.C.; Watkins, D.I.; Burton, D.R. Broadly neutralizing human anti-HIV antibody 2G12 is effective in protection against mucosal SHIV challenge even at low serum neutralizing titers. PLoS Pathog. 2009, 5, e1000433. [Google Scholar] [CrossRef]
- Moldt, B.; Shibata-Koyama, M.; Rakasz, E.G.; Schultz, N.; Kanda, Y.; Dunlop, D.C.; Finstad, S.L.; Jin, C.; Landucci, G.; Alpert, M.D.; et al. A nonfucosylated variant of the anti-HIV-1 monoclonal antibody b12 has enhanced FcgammaRIIIa-mediated antiviral activity in vitro but does not improve protection against mucosal SHIV challenge in macaques. J. Virol. 2012, 86, 6189–6196. [Google Scholar] [CrossRef]
- Julg, B.; Liu, P.T.; Wagh, K.; Fischer, W.M.; Abbink, P.; Mercado, N.B.; Whitney, J.B.; Nkolola, J.P.; McMahan, K.; Tartaglia, L.J.; et al. Protection against a mixed SHIV challenge by a broadly neutralizing antibody cocktail. Sci. Transl. Med. 2017, 9, eaao4235. [Google Scholar] [CrossRef]
- Shingai, M.; Nishimura, Y.; Klein, F.; Mouquet, H.; Donau, O.K.; Plishka, R.; Buckler-White, A.; Seaman, M.; Piatak, M., Jr.; Lifson, J.D.; et al. Antibody-mediated immunotherapy of macaques chronically infected with SHIV suppresses viraemia. Nature 2013, 503, 277–280. [Google Scholar] [CrossRef]
- Caskey, M.; Klein, F.; Lorenzi, J.C.; Seaman, M.S.; West, A.P., Jr.; Buckley, N.; Kremer, G.; Nogueira, L.; Braunschweig, M.; Scheid, J.F.; et al. Viraemia suppressed in HIV-1-infected humans by broadly neutralizing antibody 3BNC117. Nature 2015, 522, 487–491. [Google Scholar] [CrossRef]
- Caskey, M.; Schoofs, T.; Gruell, H.; Settler, A.; Karagounis, T.; Kreider, E.F.; Murrell, B.; Pfeifer, N.; Nogueira, L.; Oliveira, T.Y.; et al. Antibody 10-1074 suppresses viremia in HIV-1-infected individuals. Nat. Med. 2017, 23, 185–191. [Google Scholar] [CrossRef] [Green Version]
- Scheid, J.F.; Horwitz, J.A.; Bar-On, Y.; Kreider, E.F.; Lu, C.L.; Lorenzi, J.C.; Feldmann, A.; Braunschweig, M.; Nogueira, L.; Oliveira, T.; et al. HIV-1 antibody 3BNC117 suppresses viral rebound in humans during treatment interruption. Nature 2016, 535, 556–560. [Google Scholar] [CrossRef] [Green Version]
- Mendoza, P.; Gruell, H.; Nogueira, L.; Pai, J.A.; Butler, A.L.; Millard, K.; Lehmann, C.; Suarez, I.; Oliveira, T.Y.; Lorenzi, J.C.C.; et al. Combination therapy with anti-HIV-1 antibodies maintains viral suppression. Nature 2018, 561, 479–484. [Google Scholar] [CrossRef]
- Moore, P.L.; Williamson, C.; Morris, L. Virological features associated with the development of broadly neutralizing antibodies to HIV-1. Trends Microbiol. 2015, 23, 204–211. [Google Scholar] [CrossRef] [Green Version]
- van Gils, M.J.; Euler, Z.; Schweighardt, B.; Wrin, T.; Schuitemaker, H. Prevalence of cross-reactive HIV-1-neutralizing activity in HIV-1-infected patients with rapid or slow disease progression. AIDS 2009, 23, 2405–2414. [Google Scholar] [CrossRef] [Green Version]
- Borrow, P.; Moody, M.A. Immunologic characteristics of HIV-infected individuals who make broadly neutralizing antibodies. Immunol Rev. 2017, 275, 62–78. [Google Scholar] [CrossRef]
- Yamamoto, T.; Lynch, R.M.; Gautam, R.; Matus-Nicodemos, R.; Schmidt, S.D.; Boswell, K.L.; Darko, S.; Wong, P.; Sheng, Z.; Petrovas, C.; et al. Quality and quantity of TFH cells are critical for broad antibody development in SHIVAD8 infection. Sci. Transl. Med. 2015, 7, 298ra120. [Google Scholar] [CrossRef]
- Locci, M.; Havenar-Daughton, C.; Landais, E.; Wu, J.; Kroenke, M.A.; Arlehamn, C.L.; Su, L.F.; Cubas, R.; Davis, M.M.; Sette, A.; et al. Human circulating PD-1+CXCR3-CXCR5+ memory Tfh cells are highly functional and correlate with broadly neutralizing HIV antibody responses. Immunity 2013, 39, 758–769. [Google Scholar] [CrossRef]
- van den Kerkhof, T.L.; Feenstra, K.A.; Euler, Z.; van Gils, M.J.; Rijsdijk, L.W.; Boeser-Nunnink, B.D.; Heringa, J.; Schuitemaker, H.; Sanders, R.W. HIV-1 envelope glycoprotein signatures that correlate with the development of cross-reactive neutralizing activity. Retrovirology 2013, 10, 102. [Google Scholar] [CrossRef]
- Piantadosi, A.; Panteleeff, D.; Blish, C.A.; Baeten, J.M.; Jaoko, W.; McClelland, R.S.; Overbaugh, J. Breadth of neutralizing antibody response to human immunodeficiency virus type 1 is affected by factors early in infection but does not influence disease progression. J. Virol. 2009, 83, 10269–10274. [Google Scholar] [CrossRef]
- Richardson, S.I.; Chung, A.W.; Natarajan, H.; Mabvakure, B.; Mkhize, N.N.; Garrett, N.; Abdool Karim, S.; Moore, P.L.; Ackerman, M.E.; Alter, G.; et al. HIV-specific Fc effector function early in infection predicts the development of broadly neutralizing antibodies. PLoS Pathog. 2018, 14, e1006987. [Google Scholar] [CrossRef]
- Kadelka, C.; Liechti, T.; Ebner, H.; Schanz, M.; Rusert, P.; Friedrich, N.; Stiegeler, E.; Braun, D.L.; Huber, M.; Scherrer, A.U.; et al. Distinct, IgG1-driven antibody response landscapes demarcate individuals with broadly HIV-1 neutralizing activity. J. Exp. Med. 2018, 215, 1589–1608. [Google Scholar] [CrossRef] [Green Version]
- Euler, Z.; van Gils, M.J.; Boeser-Nunnink, B.D.; Schuitemaker, H.; van Manen, D. Genome-wide association study on the development of cross-reactive neutralizing antibodies in HIV-1 infected individuals. PLoS ONE 2013, 8, e54684. [Google Scholar] [CrossRef]
- Donahoe, R.M. Functional evidence for effects of drugs of abuse on infection and immune responses. Nida Res. Monogr. 1996, 162, 43–45. [Google Scholar]
- Donahoe, R.M.; Adler, M.W. Substance abuse and the immune system. Drug Alcohol Depend. 2001, 62, 109–110. [Google Scholar] [CrossRef]
- Donahoe, R.M.; Falek, A.; Madden, J.J.; Nicholson, J.K.; Bokos, P.; Gallegos, K.; Veit, R. Effects of cocaine and other drugs of abuse on immune function. Adv. Exp. Med. Biol. 1991, 288, 143–150. [Google Scholar]
- Zagon, I.S.; Donahue, R.N.; Bonneau, R.H.; McLaughlin, P.J. B lymphocyte proliferation is suppressed by the opioid growth factor-opioid growth factor receptor axis: Implication for the treatment of autoimmune diseases. Immunobiology 2011, 216, 173–183. [Google Scholar] [CrossRef]
- de Wolf, F.; Lange, J.M.; Houweling, J.T.; Coutinho, R.A.; Schellekens, P.T.; van der Noordaa, J.; Goudsmit, J. Numbers of CD4+ cells and the levels of core antigens of and antibodies to the human immunodeficiency virus as predictors of AIDS among seropositive homosexual men. J. Infect. Dis. 1988, 158, 615–622. [Google Scholar] [CrossRef]
- van Manen, D.; Kootstra, N.A.; Boeser-Nunnink, B.; Handulle, M.A.; van’t Wout, A.B.; Schuitemaker, H. Association of HLA-C and HCP5 gene regions with the clinical course of HIV-1 infection. AIDS 2009, 23, 19–28. [Google Scholar] [CrossRef] [Green Version]
- van den Hoek, J.A.; Coutinho, R.A.; van Haastrecht, H.J.; van Zadelhoff, A.W.; Goudsmit, J. Prevalence and risk factors of HIV infections among drug users and drug-using prostitutes in Amsterdam. AIDS 1988, 2, 55–60. [Google Scholar] [CrossRef]
- Boom, R.; Sol, C.J.; Heijtink, R.; Wertheim-van Dillen, P.M.; van der Noordaa, J. Rapid purification of hepatitis B virus DNA from serum. J. Clin. Microbiol. 1991, 29, 1804–1811. [Google Scholar]
- Giorgi, E.E.; Funkhouser, B.; Athreya, G.; Perelson, A.S.; Korber, B.T.; Bhattacharya, T. Estimating time since infection in early homogeneous HIV-1 samples using a poisson model. Bmc Bioinform. 2010, 11, 532. [Google Scholar] [CrossRef]
- Tully, D.C.; Ogilvie, C.B.; Batorsky, R.E.; Bean, D.J.; Power, K.A.; Ghebremichael, M.; Bedard, H.E.; Gladden, A.D.; Seese, A.M.; Amero, M.A.; et al. Differences in the Selection Bottleneck between Modes of Sexual Transmission Influence the Genetic Composition of the HIV-1 Founder Virus. PLoS Pathog. 2016, 12, e1005619. [Google Scholar] [CrossRef]
- Keele, B.F.; Giorgi, E.E.; Salazar-Gonzalez, J.F.; Decker, J.M.; Pham, K.T.; Salazar, M.G.; Sun, C.; Grayson, T.; Wang, S.; Li, H.; et al. Identification and characterization of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc. Natl. Acad. Sci. USA 2008, 105, 7552–7557. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.Y.; Giorgi, E.E.; Keele, B.F.; Gaschen, B.; Athreya, G.S.; Salazar-Gonzalez, J.F.; Pham, K.T.; Goepfert, P.A.; Kilby, J.M.; Saag, M.S.; et al. Modeling sequence evolution in acute HIV-1 infection. J. Biol. 2009, 261, 341–360. [Google Scholar] [CrossRef]
- Prins, M.; Veugelers, P.J. Comparison of progression and non-progression in injecting drug users and homosexual men with documented dates of HIV-1 seroconversion. European Seroconverter Study and the Tricontinental Seroconverter Study. AIDS 1997, 11, 621–631. [Google Scholar] [CrossRef]
- Gilbert, P.; Wang, M.; Wrin, T.; Petropoulos, C.; Gurwith, M.; Sinangil, F.; D’Souza, P.; Rodriguez-Chavez, I.R.; DeCamp, A.; Giganti, M.; et al. Magnitude and breadth of a nonprotective neutralizing antibody response in an efficacy trial of a candidate HIV-1 gp120 vaccine. J. Infect. Dis. 2010, 202, 595–605. [Google Scholar] [CrossRef]
- Bandawe, G.P.; Moore, P.L.; Werner, L.; Gray, E.S.; Sheward, D.J.; Madiga, M.; Nofemela, A.; Thebus, R.; Marais, J.C.; Maboko, L.; et al. Differences in HIV type 1 neutralization breadth in 2 geographically distinct cohorts in Africa. J. Infect. Dis. 2015, 211, 1461–1466. [Google Scholar] [CrossRef]
- Montefiori, D.C.; Metch, B.; McElrath, M.J.; Self, S.; Weinhold, K.J.; Corey, L.; Network, H.I.V.V.T. Demographic factors that influence the neutralizing antibody response in recipients of recombinant HIV-1 gp120 vaccines. J. Infect. Dis. 2004, 190, 1962–1969. [Google Scholar] [CrossRef]
- Sajadi, M.M.; Guan, Y.; DeVico, A.L.; Seaman, M.S.; Hossain, M.; Lewis, G.K.; Redfield, R.R. Correlation between circulating HIV-1 RNA and broad HIV-1 neutralizing antibody activity. J. Acquir. Immune Defic. Syndr 2011, 57, 9–15. [Google Scholar] [CrossRef] [Green Version]
- Collaboration, C. Differences in CD4 cell counts at seroconversion and decline among 5739 HIV-1-infected individuals with well-estimated dates of seroconversion. J. Acquir. Immune Defic. Syndr. 2003, 34, 76–83. [Google Scholar] [CrossRef]
- Fischinger, S.; Boudreau, C.M.; Butler, A.L.; Streeck, H.; Alter, G. Sex differences in vaccine-induced humoral immunity. Semin Immunopathol. 2018, 41, 239–249. [Google Scholar] [CrossRef]
- Pauthner, M.; Havenar-Daughton, C.; Sok, D.; Nkolola, J.P.; Bastidas, R.; Boopathy, A.V.; Carnathan, D.G.; Chandrashekar, A.; Cirelli, K.M.; Cottrell, C.A.; et al. Elicitation of Robust Tier 2 Neutralizing Antibody Responses in Nonhuman Primates by HIV Envelope Trimer Immunization Using Optimized Approaches. Immunity 2017, 46, 1073–1088. [Google Scholar] [CrossRef]
- Bar, K.J.; Li, H.; Chamberland, A.; Tremblay, C.; Routy, J.P.; Grayson, T.; Sun, C.; Wang, S.; Learn, G.H.; Morgan, C.J.; et al. Wide variation in the multiplicity of HIV-1 infection among injection drug users. J. Virol. 2010, 84, 6241–6247. [Google Scholar] [CrossRef]
- Masharsky, A.E.; Dukhovlinova, E.N.; Verevochkin, S.V.; Toussova, O.V.; Skochilov, R.V.; Anderson, J.A.; Hoffman, I.; Cohen, M.S.; Swanstrom, R.; Kozlov, A.P. A substantial transmission bottleneck among newly and recently HIV-1-infected injection drug users in St Petersburg, Russia. J. Infect. Dis. 2010, 201, 1697–1702. [Google Scholar] [CrossRef]
- Cortez, V.; Odem-Davis, K.; McClelland, R.S.; Jaoko, W.; Overbaugh, J. HIV-1 superinfection in women broadens and strengthens the neutralizing antibody response. PLoS Pathog. 2012, 8, e1002611. [Google Scholar] [CrossRef]
- Williams, K.L.; Wang, B.; Arenz, D.; Williams, J.A.; Dingens, A.S.; Cortez, V.; Simonich, C.A.; Rainwater, S.; Lehman, D.A.; Lee, K.K.; et al. Superinfection Drives HIV Neutralizing Antibody Responses from Several B Cell Lineages that Contribute to a Polyclonal Repertoire. Cell Rep. 2018, 23, 682–691. [Google Scholar] [CrossRef]
- Cornelissen, M.; Euler, Z.; van den Kerkhof, T.L.; van Gils, M.J.; Boeser-Nunnink, B.D.; Kootstra, N.A.; Zorgdrager, F.; Schuitemaker, H.; Prins, J.M.; Sanders, R.W.; et al. The Neutralizing Antibody Response in an Individual with Triple HIV-1 Infection Remains Directed at the First Infecting Subtype. Aids Res. Hum. Retrovir. 2016, 32, 1135–1142. [Google Scholar] [CrossRef]
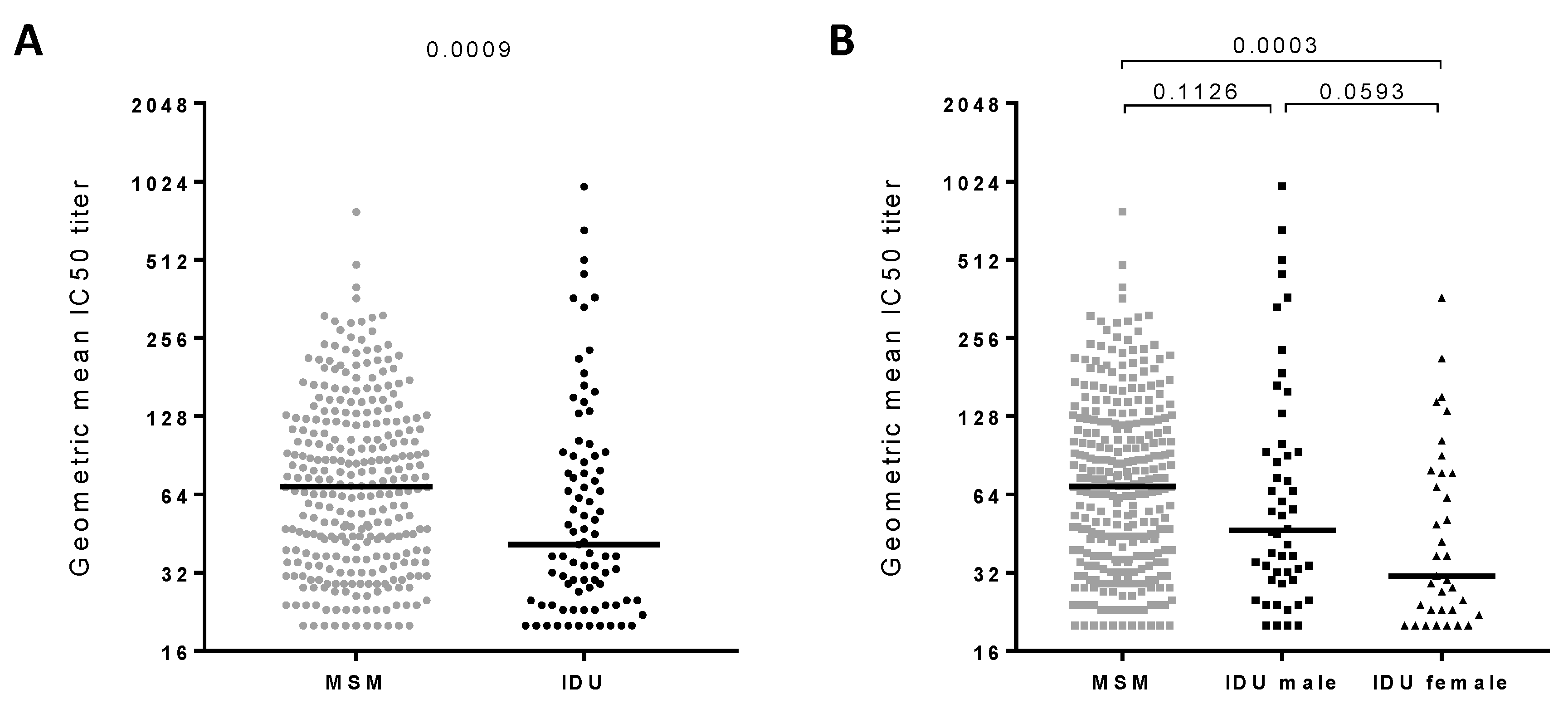
| N | % Individuals with bNAb Responses (≥ 4 Viruses Neutralized with IC50 > 100) | % Elite Neutralizers (GMT > 500) | |||
|---|---|---|---|---|---|
| MSM | 299 | 27% | p = 0.1567 | 0.3% | p = 0.0354 |
| IDU | 85 | 19% | 3.5% | ||
| IDU-Male | 50 | 20% | p = 0.7839 | 6.0% | p = 0.2647 |
| IDU-Female | 35 | 17% | 0.0% | ||
| Univariate Analysis | Multivariate Analysis* | |||
|---|---|---|---|---|
| p-Value | Effect | p-Value | Effect | |
| Gender and route of transmission | 0.002 | −0.16 | 0.001 | −0.12 |
| CD4+ T cell count at setpoint | 0.002 | −0.16 | 0.017 | −0.13 |
| Viral load at setpoint | 0.011 | 1.34 | 0.127 | 0.09 |
| Univariable Analysis | Multivariable Analysis | ||||
|---|---|---|---|---|---|
| p-Value | Effect | p-Value | Effect | ||
| Risk Group and Sex | MSM | 1.5 × 10−5 | −1.16 | 0.004 | −0.77 |
| IDU-Male | reference | reference | |||
| IDU-Female | 0.007 | −1.06 | 0.006 | −1.04 | |
| CD4+ T cell count | 0.005 | −0.31 | 0.100 | −0.19 | |
| Viral load | 0.019 | 0.33 | 4.5 × 10−4 | 0.51 | |
| Infection Time | Group 1 | reference | reference | ||
| Group 3 | 2.3 × 10−26 | 3.40 | 2.8 × 10−27 | 3.44 | |
| Group 5 | 3.1 × 10−33 | 3.72 | 2.1 × 10−32 | 3.76 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
EULER, Z.; VAN DEN KERKHOF, T.L.; KOUYOS, R.D.; TULLY, D.C.; ALLEN, T.M.; TRKOLA, A.; SANDERS, R.W.; SCHUITEMAKER, H.; VAN GILS, M.J. Lower Broadly Neutralizing Antibody Responses in Female Versus Male HIV-1 Infected Injecting Drug Users. Viruses 2019, 11, 384. https://doi.org/10.3390/v11040384
EULER Z, VAN DEN KERKHOF TL, KOUYOS RD, TULLY DC, ALLEN TM, TRKOLA A, SANDERS RW, SCHUITEMAKER H, VAN GILS MJ. Lower Broadly Neutralizing Antibody Responses in Female Versus Male HIV-1 Infected Injecting Drug Users. Viruses. 2019; 11(4):384. https://doi.org/10.3390/v11040384
Chicago/Turabian StyleEULER, Zelda, Tom L. VAN DEN KERKHOF, Roger D. KOUYOS, Damien C. TULLY, Todd M. ALLEN, Alexandra TRKOLA, Rogier W. SANDERS, Hanneke SCHUITEMAKER, and Marit J. VAN GILS. 2019. "Lower Broadly Neutralizing Antibody Responses in Female Versus Male HIV-1 Infected Injecting Drug Users" Viruses 11, no. 4: 384. https://doi.org/10.3390/v11040384
APA StyleEULER, Z., VAN DEN KERKHOF, T. L., KOUYOS, R. D., TULLY, D. C., ALLEN, T. M., TRKOLA, A., SANDERS, R. W., SCHUITEMAKER, H., & VAN GILS, M. J. (2019). Lower Broadly Neutralizing Antibody Responses in Female Versus Male HIV-1 Infected Injecting Drug Users. Viruses, 11(4), 384. https://doi.org/10.3390/v11040384





