Willingness to Participate and Associated Factors in a Zika Vaccine Trial in Indonesia: A Cross-Sectional Study
Abstract
1. Introduction
2. Material and Methods
2.1. Study Design and Study Participants
2.2. Study Instrument
2.3. Measures
2.3.1. Dependent Variable
2.3.2. Independent Variables
a. Sociodemographic Data
b. Attitude for Childhood Vaccination
c. Support to Zika Vaccine
d. The Role of Financial Compensation
2.4. Statistical Analysis
2.5. Ethical Consideration
3. Results
3.1. Participants’ Characteristics
3.2. Factors Associated with Willingness to Participate
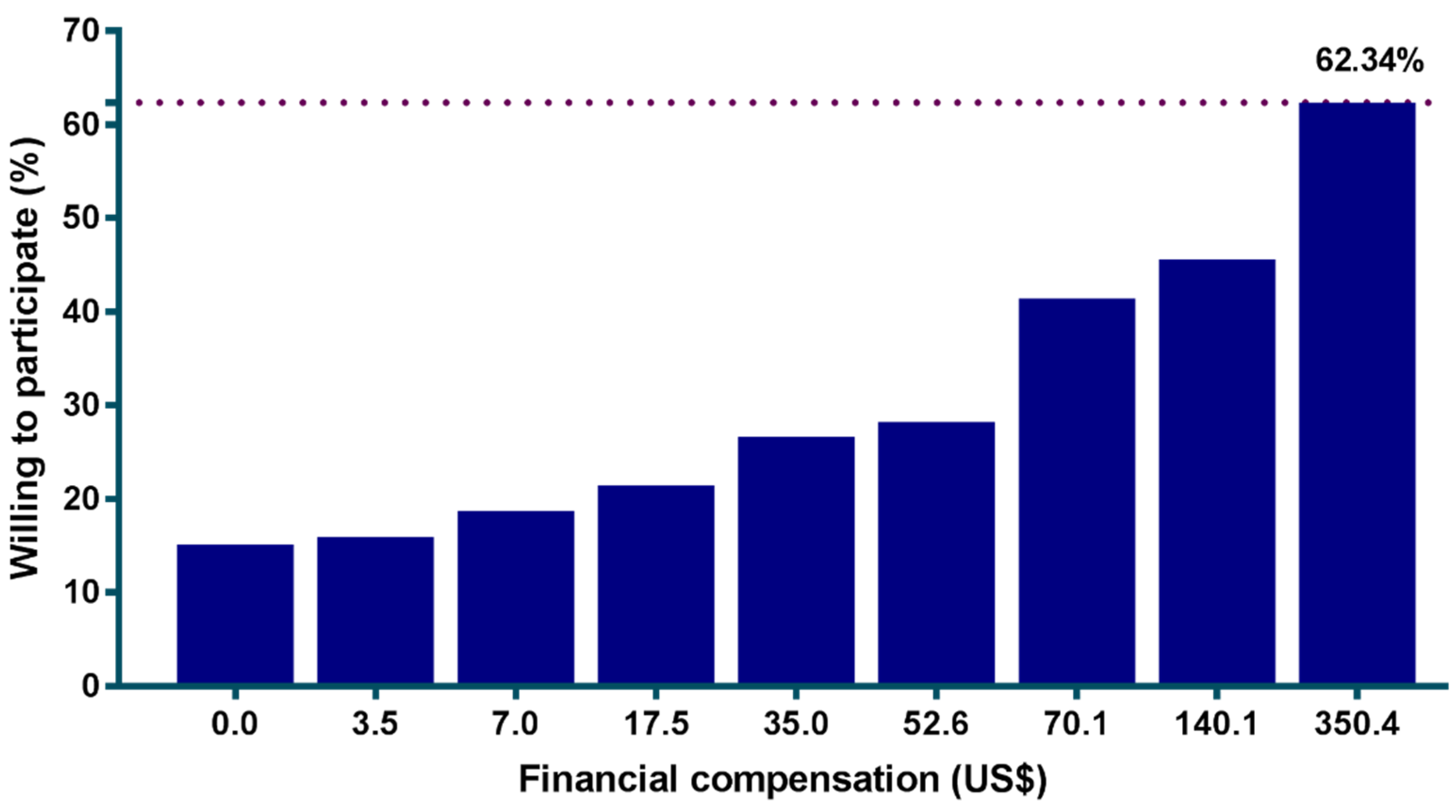
3.3. Financial Compensation and Willingness to Participate
4. Discussion
5. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Macnamara, F.N. Zika virus: A report on three cases of human infection during an epidemic of jaundice in Nigeria. Trans. R. Soc. Trop. Med. Hygiene 1954, 48, 139–145. [Google Scholar] [CrossRef]
- Duffy, M.R.; Chen, T.H.; Hancock, W.T.; Powers, A.M.; Kool, J.L.; Lanciotti, R.S.; Pretrick, M.; Marfel, M.; Holzbauer, S.; Dubray, C.; et al. Zika virus outbreak on Yap Island, Federated States of Micronesia. N. Engl. J. Med. 2009, 360, 2536–2543. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Zika Situation Report. Available online: http://www.who.int/emergencies/zika-virus/situation-report/10-march-2017/en/ (accessed on 1 January 2018).
- Cauchemez, S.; Besnard, M.; Bompard, P.; Dub, T.; Guillemette-Artur, P.; Eyrolle-Guignot, D.; Salje, H.; Van Kerkhove, M.D.; Abadie, V.; Garel, C.; et al. Association between Zika virus and microcephaly in French Polynesia, 2013–2015: A retrospective study. Lancet 2016, 387, 2125–2132. [Google Scholar] [CrossRef]
- De Araujo, T.V.B.; Rodrigues, L.C.; Ximenes, R.A.D.A.; Miranda-Filho, D.D.B.; Montarroyos, U.R.; de Melo, A.P.L.; Valongueiro, S.; de Albuquerque, M.D.F.P.M.; Souza, W.V.; Braga, C.; et al. Association between Zika virus infection and microcephaly in Brazil, January to May, 2016: Preliminary report of a case-control study. Lancet Infect. Dis. 2016, 16, 1356–1363. [Google Scholar] [CrossRef]
- De Araujo, T.V.B.; Ximenes, R.A.D.A.; Miranda, D.D.; Souza, W.V.; Montarroyos, U.R.; de Melo, A.P.L.; Valongueiro, S.; de Albuquerque, M.D.P.M.; Braga, C.; Brandao, S.P.; et al. Association between microcephaly, Zika virus infection, and other risk factors in Brazil: Final report of a case-control study. Lancet Infect. Dis. 2018, 18, 328–336. [Google Scholar] [CrossRef]
- Schuler-Faccini, L.; Ribeiro, E.M.; Feitosa, I.M.L.; Horovitz, D.D.G.; Cavalcanti, D.P.; Pessoa, A.; Doriqui, M.J.R.; Neri, J.I.; Neto, J.M.D.; Wanderley, H.Y.C.; et al. Possible association between Zika virus Infection and microcephaly—Brazil, 2015. Morb. Mortal. Wkly. Rep. 2016, 65, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.J.; Oduyebo, T.; Brault, A.C.; Brooks, J.T.; Chung, K.-W.; Hills, S.; Kuehnert, M.J.; Mead, P.; Meaney-Delman, D.; Rabe, I. Modes of transmission of Zika virus. J. Infect. Dis. 2017, 216, S875–S883. [Google Scholar] [CrossRef] [PubMed]
- Leung, G.; Baird, R.W.; Druce, J.; Anstey, N.M. Zika virus infection in Australia following a monkey bite in Indonesia. Southeast Asian J. Trop. Med. Public Health 2015, 46, 460–464. [Google Scholar] [PubMed]
- World Health Organization (WHO). Fifth Meeting of the Emergency Committee under the International Health Regulations (2005) Regarding Microcephaly, Other Neurological Disorders and Zika virus. Available online: http://www.who.int/mediacentre/news/statements/2016/zikafifthec/en/ (accessed on 1 January 2018).
- World Health Organization (WHO). WHO/UNICEF Zika Virus (ZIKV) Vaccine Target Product Profile (TPP): Vaccine to Protect against Congenital Zika Syndrome for Use during an Emergency. Available online: http://www.who.int/immunization/research/development/WHO_UNICEF_Zikavac_TPP_Feb2017.pdf (accessed on 1 January 2018).
- Lagunas-Rangel, F.A.; Viveros-Sandoval, M.E.; Reyes-Sandoval, A. Current trends in Zika vaccine development. J. Virus Erad. 2017, 3, 124–127. [Google Scholar] [PubMed]
- Durbin, A.; Wilder-Smith, A. An update on Zika vaccine developments. Expert Rev. Vaccines 2017, 16, 781–787. [Google Scholar] [CrossRef] [PubMed]
- Poland, G.A.; Kennedy, R.B.; Ovsyannikova, I.G.; Palacios, R.; Ho, P.L.; Kalil, J. Development of vaccines against Zika virus. Lancet Infect. Dis. 2018, 18, E211–E219. [Google Scholar] [CrossRef]
- McDonald, A.M.; Knight, R.C.; Campbell, M.K.; Entwistle, V.A.; Grant, A.M.; Cook, J.A.; Elbourne, D.R.; Francis, D.; Garcia, J.; Roberts, I.; et al. What influences recruitment to randomised controlled trials? A review of trials funded by two UK funding agencies. Trials 2006, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Bower, P.; Wallace, P.; Ward, E.; Graffy, J.; Miller, J.; Delaney, B.; Kinmonth, A.L. Improving recruitment to health research in primary care. Fam. Pract. 2009, 26, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.A.; Neighbors, C.J.; Wasserman, M.R.; Armstrong, G.F.; Drnach, M.L.; Howie, S.L.; Hawthorne, T.L. Strategies and cost of recruitment of middle-aged and older unmarried women in a cancer screening study. Cancer Epidemiol. Biomark. Prev. 2007, 16, 2605–2614. [Google Scholar] [CrossRef] [PubMed]
- Frew, P.M.; Archibald, M.; Hixson, B.; del Rio, C. Socioecological influences on community involvement in HIV vaccine research. Vaccine 2011, 29, 6136–6143. [Google Scholar] [CrossRef] [PubMed]
- Detoc, M.; Gagneux-Brunon, A.; Lucht, F.; Botelho-Nevers, E. Barriers and motivations to volunteers’ participation in preventive vaccine trials: A systematic review. Expert Rev. Vaccines 2017, 16, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Walsh, E.; Sheridan, A. Factors affecting patient participation in clinical trials in Ireland: A narrative review. Contemp. Clin. Trials Commun. 2016, 3, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Anwar, S.; Bustaman, A.; Radiansyah, A.; Angraini, P.; Fasli, R.; Salwiyadi, S.; Bastian, R.A.; Oktiviyari, A.; Akmal, I.; et al. Community Willingness to Participate in a Dengue Study in Aceh Province, Indonesia. PLoS ONE 2016, 11, e0159139. [Google Scholar] [CrossRef] [PubMed]
- Opel, D.J.; Mangione-Smith, R.; Taylor, J.A.; Korfiatis, C.; Wiese, C.; Catz, S.; Martin, D.P. Development of a survey to identify vaccine-hesitant parents: The parent attitudes about childhood vaccines survey. Hum. Vaccines 2011, 7, 419–425. [Google Scholar] [CrossRef]
- Azizi, F.S.M.; Kew, Y.; Moy, F.M. Vaccine hesitancy among parents in a multi-ethnic country, Malaysia. Vaccine 2017, 35, 2955–2961. [Google Scholar] [CrossRef] [PubMed]
- Strelitz, B.; Gritton, J.; Klein, E.J.; Bradford, M.C.; Follmer, K.; Zerr, D.M.; Englund, J.A.; Opel, D.J. Parental vaccine hesitancy and acceptance of seasonal influenza vaccine in the pediatric emergency department. Vaccine 2015, 33, 1802–1807. [Google Scholar] [CrossRef] [PubMed]
- Opel, D.J.; Taylor, J.A.; Zhou, C.; Catz, S.; Myaing, M.; Mangione-Smith, R. The Relationship between Parent Attitudes about Childhood Vaccines Survey Scores and Future Child Immunization Status a Validation Study. JAMA Pediatr. 2013, 167, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Opel, D.J.; Taylor, J.A.; Mangione-Smith, R.; Solomon, C.; Zhao, C.; Catz, S.; Martin, D. Validity and reliability of a survey to identify vaccine-hesitant parents. Vaccine 2011, 29, 6598–6605. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Anwar, S.; Bustaman, A.; Radiansyah, A.; Angraini, P.; Fasli, R.; Salwiyadi, S.; Bastian, R.A.; Oktiviyari, A.; Akmal, I.; et al. Modifiable determinants of attitude towards dengue vaccination among healthy inhabitants of Aceh, Indonesia: Findings from a community-based survey. Asian Pac. J. Trop. Med. 2016, 9, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Anwar, S.; Bustamam, A.; Radiansyah, A.; Angraini, P.; Fasli, R.; Salwiyadi, S.; Bastian, R.A.; Oktiviyari, A.; Akmal, I.; et al. Willingness to pay for a dengue vaccine and its associated determinants in Indonesia: A community-based, cross-sectional survey in Aceh. Acta Trop. 2017, 166, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Anwar, S.; Setiawan, A.M.; Sasmono, R.T.; Study, A.D. Dengue vaccine acceptance and associated factors in Indonesia: A community-based cross-sectional survey in Aceh. Vaccine 2016, 34, 3670–3675. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Rajamoorthy, Y.; Anwar, S.; Bustamam, A.; Radiansyah, A.; Angraini, P.; Fasli, R.; Salwiyadi, S.; Bastian, R.A.; Oktiviyari, A.; et al. Knowledge, attitude, and practice regarding dengue virus infection among inhabitants of Aceh, Indonesia: A cross-sectional study. BMC Infect. Dis. 2018, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Aletta, A.; Anwar, S.; Setiawan, A.M.; Maulana, R.; Wahyuniati, N.; Ramadana, M.R.; Haryanto, S.; Rodriguez-Morales, A.J.; Jamil, K.F. Healthcare workers’ knowledge towards Zika virus infection in Indonesia: A survey in Aceh. Asian Pac. J. Trop. Med. 2017, 10, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Alleta, A.; Anwar, S.; Setiawan, A.M.; Maulana, R.; Wahyuniati, N.; Ramadana, M.R.; Ikram, I.; Haryanto, S.; Jamil, K.F.; et al. Attitudes towards Zika virus infection among medical doctors in Aceh province, Indonesia. J. Infect. Public Health 2018, 11, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Suhud, U.; Syabai, N. Halal sex tourism in Indonesia: Understanding the motivation of young female host to marry with middle eastern male tourists. J. Econ. Sustain. Dev. 2014, 5, 91–94. [Google Scholar]
- Godskesen, T.; Hansson, M.G.; Nygren, P.; Nordin, K.; Kihlbom, U. Hope for a cure and altruism are the main motives behind participation in phase 3 clinical cancer trials. Eur. J. Cancer Care 2015, 24, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Limkakeng, A.; Phadtare, A.; Shah, J.; Vaghasia, M.; Wei, D.Y.; Shah, A.; Pietrobon, R. Willingness to participate in clinical trials among patients of Chinese heritage: A meta-synthesis. PLoS ONE 2013, 8, e51328. [Google Scholar] [CrossRef] [PubMed]
- McCann, S.K.; Campbell, M.K.; Entwistle, V.A. Reasons for participating in randomised controlled trials: Conditional altruism and considerations for self. Trials 2010, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Pulley, J.M.; Brace, M.M.; Bernard, G.R.; Masys, D.R. Attitudes and perceptions of patients towards methods of establishing a DNA biobank. Cell Tissue Bank 2008, 9, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Treweek, S.; Lockhart, P.; Pitkethly, M.; Cook, J.A.; Kjeldstrom, M.; Johansen, M.; Taskila, T.K.; Sullivan, F.M.; Wilson, S.; Jackson, C.; et al. Methods to improve recruitment to randomised controlled trials: Cochrane systematic review and meta-analysis. BMJ Open 2013, 3, e002360. [Google Scholar] [CrossRef] [PubMed]
- Lobato, L.; Bethony, J.M.; Pereira, F.B.; Grahek, S.L.; Diemert, D.; Gazzinelli, M.F. Impact of gender on the decision to participate in a clinical trial: A cross-sectional study. BMC Public Health 2014, 14, 1156. [Google Scholar] [CrossRef] [PubMed]
- Balfour, L.; Corace, K.; Tasca, G.A.; Tremblay, C.; Routy, J.P.; Angel, J.B. Altruism motivates participation in a therapeutic HIV vaccine trial (CTN 173). AIDS Care 2010, 22, 1403–1409. [Google Scholar] [CrossRef] [PubMed]
- Bidad, N.; MacDonald, L.; Winters, Z.E.; Edwards, S.J.L.; Emson, M.; Griffin, C.L.; Bliss, J.; Horne, R. How informed is declared altruism in clinical trials? A qualitative interview study of patient decision-making about the QUEST trials (Quality of Life after Mastectomy and Breast Reconstruction). Trials 2016, 17, 431. [Google Scholar] [CrossRef] [PubMed]
- Setyobudihono, S.; Istiqomah, E.; Adiningsih, S. Husband Influences on Pregnant Women Who Following Consumption Iron Supplementation Program. Procedia Soc. Behav. Sci. 2016, 222, 160–168. [Google Scholar] [CrossRef]
- Pakpahan, D. Women and Culture in Indonesia: Images in Religion, Customary Law in the State. Lila-Asia Pac. Women Stud. J. 1996, 6, 7–11. [Google Scholar]
- Mystakidou, K.; Panagiotou, I.; Katsaragakis, S.; Tsilika, E.; Parpa, E. Ethical and practical challenges in implementing informed consent in HIV/AIDS clinical trials in developing or resource-limited countries. SAHARA J. 2009, 6, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Marshall, P.A.; Adebamowo, C.A.; Adeyemo, A.A.; Ogundiran, T.O.; Vekich, M.; Strenski, T.; Zhou, J.; Prewitt, T.E.; Cooper, R.S.; Rotimi, C.N. Voluntary participation and informed consent to international genetic research. Am. J. Public Health 2006, 96, 1989–1995. [Google Scholar] [CrossRef] [PubMed]
- Kufa, T.; Chihota, V.; Charalambous, S.; Verver, S.; Churchyard, G. Willingness to participate in trials and to be vaccinated with new tuberculosis vaccines in HIV-infected adults. Public Health Action 2013, 3, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Comis, R.L.; Miller, J.D.; Aldige, C.R.; Krebs, L.; Stoval, E. Public attitudes toward participation in cancer clinical trials. J. Clin. Oncol. 2003, 21, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.H.; Kim, E.J.; Jeong, S.H.; Park, G.L. Factors associated with willingness to participate in clinical trials: A nationwide survey study. BMC Public Health 2015, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Rikin, S.; Shea, S.; LaRussa, P.; Stockwell, M. Factors associated with willingness to participate in a vaccine clinical trial among elderly Hispanic patients. Contemp. Clin. Trials Commun. 2017, 7, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Mood, D.; Gadgeel, S.; Simon, M.S. An educational video to increase clinical trials enrollment among lung cancer patients. J. Thorac. Oncol. 2008, 3, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Evangeli, M.; Kafaar, Z.; Kagee, A.; Swartz, L.; Bullemor-Day, P. Does message framing predict willingness to participate in a hypothetical HIV vaccine trial: An application of Prospect Theory. AIDS Care 2013, 25, 910–914. [Google Scholar] [CrossRef] [PubMed]
- Grady, C. Payment of clinical research subjects. J. Clin. Investig. 2005, 115, 1681–1687. [Google Scholar] [CrossRef] [PubMed]
- Fry, C.L.; Ritter, A.; Baldwin, S.; Bowen, K.J.; Gardiner, P.; Holt, T.; Jenkinson, R.; Johnston, J. Paying research participants: A study of current practices in Australia. J. Med. Ethics 2005, 31, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Singer, E.; Couper, M.P. Do Incentives Exert Undue Influence on Survey Participation? Experimental Evidence. J. Empir. Res. Hum. Res. 2008, 3, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Afshar, R.; Sanavi, S.; Rajabi, M.R. Attitude and willingness of high school students toward organ donation. Saudi J. Kidney Dis. Transplant. 2012, 23, 929–933. [Google Scholar] [CrossRef]
- Kettis-Lindblad, A.; Ring, L.; Viberth, E.; Hansson, M.G. Genetic research and donation of tissue samples to biobanks. What do potential sample donors in the Swedish general public think? Eur. J. Public Health 2006, 16, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Harapan, H.; Mudatsir, M.; Yufika, A.; Nawawi, Y.; Wahyuniati, N.; Anwar, S.; Yusri, F.; Haryanti, N.; Wijayanti, N.P.; Rizal, R.; et al. Community acceptance and willingness-to-pay for a hypothetical Zika vaccine: A cross-sectional study in Indonesia. Vaccine 2018, sumbitted. [Google Scholar]
- Olowookere, S.A.; Abioye-Kuteyi, E.A.; Adekanle, O. Willingness to participate in Ebola viral disease vaccine trials and receive vaccination by health workers in a tertiary hospital in Ile-Ife, Southwest Nigeria. Vaccine 2016, 34, 5758–5761. [Google Scholar] [CrossRef] [PubMed]
- Raheja, D.; Davila, E.P.; Johnson, E.T.; Deovic, R.; Paine, M.; Rouphael, N. Willingness to Participate in Vaccine-Related Clinical Trials among Older Adults. Int. J. Environ. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Shen, X.; Guo, R.; Liu, B.; Zhu, L.; Wang, J.; Zhang, L.; Sun, J.; Zhang, X.; Xu, J. Willingness to participate in HIV therapeutic vaccine trials among HIV-infected patients on ART in China. PLoS ONE 2014, 9, e111321. [Google Scholar] [CrossRef] [PubMed]
| Variable | n (%) |
|---|---|
| Age group (years) | |
| 20–29 (R) | 230 (24.1) |
| 30–39 | 469 (49.1) |
| 40–49 | 189 (19.8) |
| More than 50 | 68 (7.1) |
| Gender | |
| Male (R) | 129 (13.5) |
| Female | 827 (86.5) |
| Educational attainment | |
| Primary school (R) | 42 (4.4) |
| Junior high school | 106 (11.1) |
| Senior high school | 361 (37.8) |
| Diploma certificate | 223 (23.3) |
| University graduate | 224 (23.4) |
| Employment status | |
| Unemployment (R) | 471 (49.3) |
| Employee | 485 (50.7) |
| Types of workplace | |
| Non–healthcare sector (R) | 260 (53.6) |
| Healthcare sector | 225 (46.4) |
| Monthly income (IDR) | |
| Less than 3 million (R) | 523 (54.7) |
| 3–5 million | 328 (34.3) |
| More than 5 million | 105 (10.9) |
| Number of children | |
| The first pregnancy (R) | 31 (3.2) |
| 1–2 | 564 (59.0) |
| 3–5 | 337 (35.3) |
| More than 5 | 24 (2.5) |
| Have heard about Zika | |
| No (R) | 704 (73.6) |
| Yes | 252 (26.4) |
| Attitude towards childhood vaccination | |
| Poor (R) | 152 (15.9) |
| Good | 804 (84.1) |
| Vaccination behaviour sub-domain | |
| Poor (R) | 368 (39.8) |
| Good | 557 (60.2) |
| General attitude sub-domain | |
| Poor (R) | 69 (7.2) |
| Good | 887 (92.8) |
| Vaccine safety and efficacy sub-domain | |
| Poor (R) | 589 (61.6) |
| Good | 367 (38.4) |
| Acceptance for Zika Vaccine | |
| Unwilling (R) | 199 (20.8) |
| Willing | 757 (79.2) |
| Variable | Willing to ParticipateYes/No | Univariate | Multivariate | ||
|---|---|---|---|---|---|
| OR (95% CI) | p-Value | aOR (95% CI) | p-Value | ||
| Age group (years) | |||||
| 20–29 (R) | 32/198 | 1 | 1 | ||
| 30–39 | 61/408 | 0.93 (0.58–1.47) | 0.740 | 1.29 (0.77–2.15) | 0.328 |
| 40–49 | 30/159 | 1.17 (0.68–2.00) | 0.574 | 1.83 (0.97–3.44) | 0.061 |
| More than 50 | 21/47 | 2.77 (1.46–5.22) | 0.002 | 5.00 (2.37–10.53) | <0.001 |
| Gender | |||||
| Male (R) | 11/118 | 1 | 1 | ||
| Female | 133/694 | 2.06 (1.08–3.92) | 0.029 | 2.20 (1.11–4.37) | 0.025 |
| Educational attainment | |||||
| Primary school (R) | 4/38 | 1 | 1 | ||
| Junior high school | 24/82 | 2.78 (0.90–8.58) | 0.075 | 2.88 (0.90–9.25) | 0.075 |
| Senior high school | 56/305 | 1.74 (0.60–5.08) | 0.308 | 1.95 (0.64–5.97) | 0.240 |
| Diploma certificate | 23/200 | 1.09 (0.36–3.34) | 0.877 | 1.14 (0.34–3.83) | 0.827 |
| University graduate | 37/187 | 1.88 (0.63–5.59) | 0.256 | 1.84 (0.56–6.05) | 0.317 |
| Employment status | |||||
| Unemployment (R) | 79/392 | 1 | 1 | ||
| Employee | 65/420 | 0.77 (0.54–1.10) | 0.146 | 0.78 (0.49–1.26) | 0.313 |
| Types of workplace | |||||
| Non–healthcare sector(R) | 34/226 | 1 | – | ||
| Healthcare sector | 31/194 | 1.06 (0.63–1.79) | 0.821 | ||
| Monthly income (IDR) | |||||
| Less than 3 million (R) | 77/446 | 1 | – | ||
| 3–5 million | 53/275 | 1.12 (0.76–1.63) | 0.571 | ||
| More than 5 million | 14/91 | 0.89 (0.48–1.64) | 0.712 | ||
| Number of children | |||||
| The first pregnancy (R) | 7/24 | 1 | 1 | ||
| 1–2 | 87/477 | 0.63 (0.26–1.50) | 0.292 | 0.46 (0.18–1.18) | 0.107 |
| 3–5 | 45/292 | 0.53 (0.22–1.30) | 0.164 | 0.28 (0.10–0.79) | 0.016 |
| More than 5 | 5/19 | 0.90 (0.25–3.30) | 0.876 | 0.38 (0.09–1.60) | 0.187 |
| Have heard about Zika | |||||
| No (R) | 86/618 | 1 | 1 | ||
| Yes | 58/194 | 2.15 (1.48–3.11) | <0.001 | 2.41 (1.59–3.65) | <0.001 |
| Attitude towards childhood vaccination | |||||
| Poor (R) | 18/134 | 1 | 1 | ||
| Good | 126/678 | 1.38 (0.82–2.35) | 0.228 | 1.08 (0.60–1.93) | 0.802 |
| Vaccination behaviour sub-domain | |||||
| Poor (R) | 44/324 | 1 | |||
| Good | 93/464 | 1.48 (1.00–2.17) | 0.048 | – | |
| General attitude sub-domain | |||||
| Poor (R) | 9/60 | 1 | |||
| Good | 135/752 | 1.20 (0.58–2.47) | 0.627 | – | |
| Vaccine safety and efficacy sub-domain | |||||
| Poor (R) | 87/502 | 1 | |||
| Good | 57/310 | 1.06 (0.74–1.53) | 0.749 | – | |
| Acceptance for Zika Vaccine | |||||
| Unwilling (R) | 20/179 | 1 | 1 | ||
| Willing | 124/633 | 1.75 (1.06–2.89) | 0.028 | 1.53 (0.88–2.67) | 0.128 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harapan, H.; Mudatsir, M.; Yufika, A.; Nawawi, Y.; Wahyuniati, N.; Anwar, S.; Yusri, F.; Haryanti, N.; Wijayanti, N.P.; Rizal, R.; et al. Willingness to Participate and Associated Factors in a Zika Vaccine Trial in Indonesia: A Cross-Sectional Study. Viruses 2018, 10, 648. https://doi.org/10.3390/v10110648
Harapan H, Mudatsir M, Yufika A, Nawawi Y, Wahyuniati N, Anwar S, Yusri F, Haryanti N, Wijayanti NP, Rizal R, et al. Willingness to Participate and Associated Factors in a Zika Vaccine Trial in Indonesia: A Cross-Sectional Study. Viruses. 2018; 10(11):648. https://doi.org/10.3390/v10110648
Chicago/Turabian StyleHarapan, Harapan, Mudatsir Mudatsir, Amanda Yufika, Yusuf Nawawi, Nur Wahyuniati, Samsul Anwar, Fitria Yusri, Novi Haryanti, Nanda Putri Wijayanti, Rizal Rizal, and et al. 2018. "Willingness to Participate and Associated Factors in a Zika Vaccine Trial in Indonesia: A Cross-Sectional Study" Viruses 10, no. 11: 648. https://doi.org/10.3390/v10110648
APA StyleHarapan, H., Mudatsir, M., Yufika, A., Nawawi, Y., Wahyuniati, N., Anwar, S., Yusri, F., Haryanti, N., Wijayanti, N. P., Rizal, R., Fitriani, D., Maulida, N. F., Syahriza, M., Ikram, I., Fandoko, T. P., Syahadah, M., Asrizal, F. W., Jamil, K. F., Rajamoorthy, Y., ... Imrie, A. (2018). Willingness to Participate and Associated Factors in a Zika Vaccine Trial in Indonesia: A Cross-Sectional Study. Viruses, 10(11), 648. https://doi.org/10.3390/v10110648







