Genetic Variation of European Beech Populations and Their Progeny from Northeast Germany to Southwest Switzerland
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites and Plant Material
2.2. DNA Isolation
2.3. SSR Genotyping
2.4. SNP Genotyping
2.5. Data Analysis
3. Results
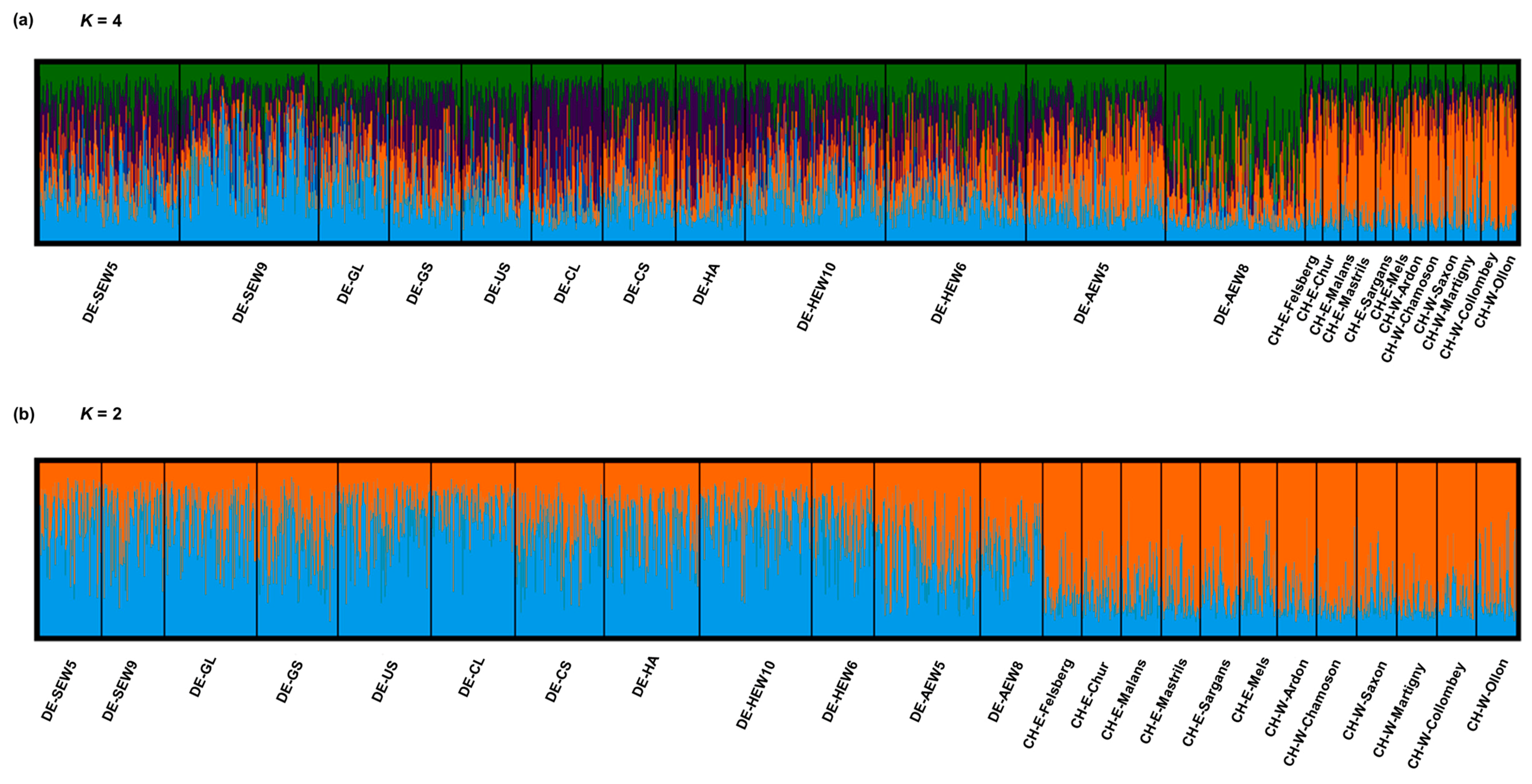
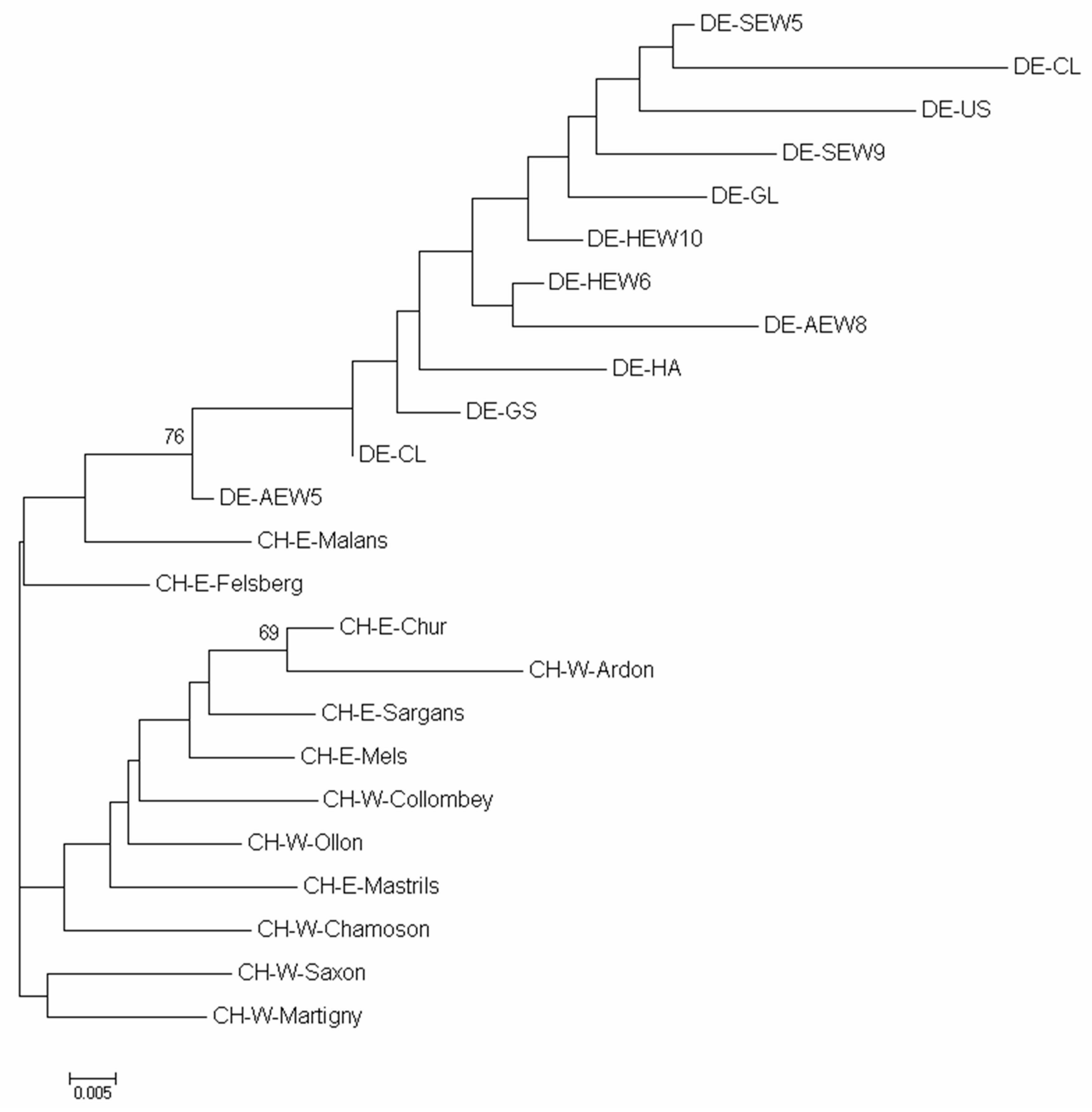
3.1. SSR Analysis
3.2. SNP Analysis
4. Discussion
4.1. Neutral Genetic Variation
4.2. Potentially Adaptive Genetic Variation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Menzel, A.; Fabian, P. Growing season extended in Europe. Nature 1999, 397, 659. [Google Scholar] [CrossRef]
- Bréda, N.; Huc, R.; Granier, A.; Dreyer, E. Temperate forest trees and stands under severe drought: A review of ecophysiological responses, adaptation processes and long-term consequences. Ann. For. Sci. 2006, 63, 625–644. [Google Scholar] [CrossRef]
- Bigler, C.; Gavin, D.G.; Gunning, C.; Veblen, T.T. Drought induces lagged tree mortality in a subalpine forest in the Rocky Mountains. Oikos 2007, 116, 1983–1994. [Google Scholar] [CrossRef]
- Van Mantgem, P.J.; Stephenson, N.L.; Byrne, J.C.; Daniels, L.D.; Franklin, J.F.; Fule, P.Z.; Harmon, M.E.; Larson, A.J.; Smith, J.M.; Taylor, A.H.; et al. Widespread increase of tree mortality rates in the western United States. Science 2009, 323, 521–524. [Google Scholar] [CrossRef] [PubMed]
- Pretzsch, H.; Biber, P.; Schutze, G.; Uhl, E.; Rotzer, T. Forest stand growth dynamics in Central Europe have accelerated since 1870. Nat. Commun. 2014, 5, 4967. [Google Scholar] [CrossRef] [PubMed]
- Jump, A.S.; Hunt, J.M.; Peñuelas, J. Rapid climate change-related growth decline at the southern range edge of Fagus sylvatica. Glob. Change Biol. 2006, 12, 2163–2174. [Google Scholar] [CrossRef]
- Piovesan, G.; Biondi, F.; Filippo, A.D.; Alessandrini, A.; Maugeri, M. Drought-driven growth reduction in old beech (Fagus sylvatica L.) forests of the central Apennines, Italy. Glob. Change Biol. 2008, 14, 1265–1281. [Google Scholar] [CrossRef]
- Charru, M.; Seynave, I.; Morneau, F.; Bontemps, J.D. Recent changes in forest productivity: An analysis of national forest inventory data for common beech (Fagus sylvatica L.) in north-eastern France. For. Ecol. Manag. 2010, 260, 864–874. [Google Scholar] [CrossRef]
- Knutzen, F.; Dulamsuren, C.; Meier, I.C.; Leuschner, C. Recent climate warming-related growth decline impairs European beech in the center of its distribution range. Ecosystems 2017, 20, 1494–1511. [Google Scholar] [CrossRef]
- Scharnweber, T.; Manthey, M.; Criegee, C.; Bauwe, A.; Schröder, C.; Wilmking, M. Drought matters—Declining precipitation influences growth of Fagus sylvatica L. and Quercus robur L. in north-eastern Germany. For. Ecol. Manag. 2011, 262, 947–961. [Google Scholar] [CrossRef]
- Zimmermann, J.; Hauck, M.; Dulamsuren, C.; Leuschner, C. Climate warming-related growth decline affects Fagus sylvatica, but not other broad-leaved tree species in Central European mixed forests. Ecosystems 2015, 18, 560–572. [Google Scholar] [CrossRef]
- Potter, K.M.; Jetton, R.M.; Bower, A.; Jacobs, D.F.; Man, G.; Hipkins, V.D.; Westwood, M. Banking on the future: Progress, challenges and opportunities for the genetic conservation of forest trees. New For. 2017, 48, 153–180. [Google Scholar] [CrossRef]
- Reed, D.H.; Frankham, R. Correlation between fitness and genetic diversity. Conserv. Biol. 2003, 17, 230–237. [Google Scholar] [CrossRef]
- Bilela, S.; Dounavi, A.; Fussi, B.; Konnert, M.; Holst, J.; Mayer, H.; Rennenberg, H.; Simon, J. Natural regeneration of Fagus sylvatica L. adapts with maturation to warmer and drier microclimatic conditions. For. Ecol. Manag. 2012, 275, 60–67. [Google Scholar] [CrossRef]
- Rajendra, K.C.; Seifert, S.; Prinz, K.; Gailing, O.; Finkeldey, R. Subtle human impacts on neutral genetic diversity and spatial patterns of genetic variation in European beech (Fagus sylvatica). For. Ecol. Manag. 2014, 319, 138–149. [Google Scholar] [CrossRef]
- Papageorgiou, A.C.; Vidalis, A.; Gailing, O.; Tsiripidis, I.; Hatziskakis, S.; Boutsios, S.; Galatsidas, S.; Finkeldey, R. Genetic variation of beech (Fagus sylvatica L.) in Rodopi (N.E. Greece). Eur. J. For. Res. 2007, 127, 81–88. [Google Scholar] [CrossRef]
- Müller, M.; Seifert, S.; Finkeldey, R. Comparison and confirmation of SNP-bud burst associations in European beech populations in Germany. Tree Genet. Genomes 2017, 13, 59. [Google Scholar] [CrossRef]
- Bell, D.M.; Clark, J.S. Seed predation and climate impacts on reproductive variation in temperate forests of the southeastern USA. Oecologia 2016, 180, 1223–1234. [Google Scholar] [CrossRef] [PubMed]
- Alizoti, P.G.; Kilimis, K.; Gallios, P. Temporal and spatial variation of flowering among Pinus nigra Arn. clones under changing climatic conditions. For. Ecol. Manag. 2010, 259, 786–797. [Google Scholar] [CrossRef]
- Ratnam, W.; Rajora, O.P.; Finkeldey, R.; Aravanopoulos, F.; Bouvet, J.-M.; Vaillancourt, R.E.; Kanashiro, M.; Fady, B.; Tomita, M.; Vinson, C. Genetic effects of forest management practices: Global synthesis and perspectives. For. Ecol. Manag. 2014, 333, 52–65. [Google Scholar] [CrossRef]
- Redmond, M.D.; Forcella, F.; Barger, N.N. Declines in pinyon pine cone production associated with regional warming. Ecosphere 2012, 3, 1–14. [Google Scholar] [CrossRef]
- Bell, D.M.; Bradford, J.B.; Lauenroth, W.K. Early indicators of change: Divergent climate envelopes between tree life stages imply range shifts in the western United States. Glob. Ecol. Biogeogr. 2014, 23, 168–180. [Google Scholar] [CrossRef]
- Clark, J.S.; Bell, D.M.; Hersh, M.H.; Nichols, L. Climate change vulnerability of forest biodiversity: Climate and competition tracking of demographic rates. Glob. Change Biol. 2011, 17, 1834–1849. [Google Scholar] [CrossRef]
- Peñuelas, J.; Ogaya, R.; Boada, M.; Jump, A.S. Migration, invasion and decline: Changes in recruitment and forest structure in a warming-linked shift of European beech forest in Catalonia (NE Spain). Ecography 2007, 30, 829–837. [Google Scholar] [CrossRef]
- Westergren, M.; Bozic, G.; Ferreira, A.; Kraigher, H. Insignificant effect of management using irregular shelterwood system on the genetic diversity of European beech (Fagus sylvatica L.): A case study of managed stand and old growth forest in Slovenia. For. Ecol. Manag. 2015, 335, 51–59. [Google Scholar] [CrossRef]
- Sandurska, E.; Ulaszewski, B.; Burczyk, J. Genetic insights into ecological succession from oak- (Quercus robur L.) to beech- (Fagus sylvatica L.) dominated forest stands. Acta Biol. Crac. S. Bot. 2017, 59, 59–69. [Google Scholar] [CrossRef]
- Konnert, M.; Ziehe, M.; Tröber, U.; Maurer, W.; Janßen, A.; Sander, T.; Hussendörfer, E.; Hertel, H. Genetic variation of beech (Fagus sylvatica L.) in Germany: Joint evaluation of genetic inventories from several federal states. Forst Holz 2000, 55, 403–408. [Google Scholar]
- Seifert, S. Variation of Candidate Genes Related to Climate Change in European Beech (Fagus sylvatica L.). Ph.D. Thesis, University of Göttingen, Göttingen, Germany, 2012. [Google Scholar]
- Müller, M.; Finkeldey, R. Genetic and adaptive trait variation in seedlings of European beech provenances from Northern Germany. Silvae Genet. 2017, 65, 65–73. [Google Scholar] [CrossRef]
- Cuervo-Alarcon, L.C.; Arend, M.; Müller, M.; Sperisen, C.; Finkeldey, R.; Krutovsky, K.V. Genetic variation and signatures of natural selection in populations of European beech (Fagus sylvatica L.) along precipitation gradients. Tree Genet. Genomes 2018. pending revisions. [Google Scholar]
- Cuervo-Alarcon, L.C.; Arend, M.; Müller, M.; Sperisen, C.; Finkeldey, R.; Krutovsky, K.V. Adaptation potential of European beech (Fagus sylvatica L.) to climate change: Linking genetic variability with drought related traits. Eur. J. For. Res. 2018. submitted. [Google Scholar]
- Holderegger, R.; Kamm, U.; Gugerli, F. Adaptive vs. neutral genetic diversity: Implications for landscape genetics. Landsc. Ecol. 2006, 21, 797–807. [Google Scholar] [CrossRef]
- Jahn, G. Temperate Deciduous Forests; Elsevier: Amsterdam, The Netherlands, 1991. [Google Scholar]
- Müller, M.; Seifert, S.; Finkeldey, R. A candidate gene-based association study reveals SNPs significantly associated with bud burst in European beech (Fagus sylvatica L.). Tree Genet. Genomes 2015, 11, 116. [Google Scholar] [CrossRef]
- Pastorelli, R.; Smulders, M.J.M.; Van’t Westende, W.P.C.; Vosman, B.; Giannini, R.; Vettori, C.; Vendramin, G.G. Characterization of microsatellite markers in Fagus sylvatica L. and Fagus orientalis Lipsky. Mol. Ecol. Notes 2003, 3, 76–78. [Google Scholar] [CrossRef]
- Vornam, B.; Decarli, N.; Gailing, O. Spatial distribution of genetic variation in a natural beech stand (Fagus sylvatica L.) based on microsatellite markers. Conserv. Genet. 2004, 5, 561–570. [Google Scholar] [CrossRef]
- Asuka, Y.; Tani, N.; Tsumura, Y.; Tomaru, N. Development and characterization of microsatellite markers for Fagus crenata Blume. Mol. Ecol. Notes 2004, 4, 101–103. [Google Scholar] [CrossRef]
- Durand, J.; Bodenes, C.; Chancerel, E.; Frigerio, J.M.; Vendramin, G.; Sebastiani, F.; Buonamici, A.; Gailing, O.; Koelewijn, H.P.; Villani, F.; et al. A fast and cost-effective approach to develop and map EST-SSR markers: Oak as a case study. BMC Genom. 2010, 11, 570. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Seifert, S.; Finkeldey, R. Identification of SNPs in candidate genes potentially involved in bud burst in European beech (Fagus sylvatica L.). Silvae Genet. 2015, 64, 1–20. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GENALEX 6: Genetic analysis in Excel. Population genetic software for teaching and research. Mol. Ecol. Notes 2006, 6, 288–295. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GENAlEX 6.5: Genetic analysis in Excel. Population genetic software for teaching and research—An update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef] [PubMed]
- Meirmans, P.G.; Hedrick, P.W. Assessing population structure: F(ST) and related measures. Mol. Ecol. Resour. 2011, 11, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Nei, M. Molecular Evolutionary Genetics; Columbia University Press: New York, NY, USA, 1987. [Google Scholar]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2009. [Google Scholar]
- Goudet, J. FSTAT (Version 1.2): A computer program to calculate F-statistics. J. Hered. 1995, 86, 485–486. [Google Scholar] [CrossRef]
- Weir, B.S.; Cockerham, C.C. Estimating F-statistics for the analysis of population structure. Evolution 1984, 38, 1358–1370. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: http://www.R-project.org/ (accessed on 4 December 2017).
- Giraudoux, P. Pgirmess: Data Analysis in Ecology. R Package Version 1.6.7. Available online: https://CRAN.R-project.org/package=pgirmess (accessed on 6 December 2017).
- Rousset, F. Genepop’007: A complete re-implementation of the genepop software for Windows and Linux. Mol. Ecol. Resour. 2008, 8, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Hill, W.G. Estimation of effective population size from data on linkage disequilibrium. Genet. Res. 1981, 38, 209–216. [Google Scholar] [CrossRef]
- Waples, R.S. A bias correction for estimates of effective population size based on linkage disequilibrium at unlinked gene loci. Conserv. Genet. 2006, 7, 167. [Google Scholar] [CrossRef]
- Waples, R.S.; Do, C. Linkage disequilibrium estimates of contemporary Ne using highly variable genetic markers: A largely untapped resource for applied conservation and evolution. Evol. Appl. 2010, 3, 244–262. [Google Scholar] [CrossRef] [PubMed]
- Do, C.; Waples, R.S.; Peel, D.; Macbeth, G.M.; Tillett, B.J.; Ovenden, J.R. NeEstimator v2: Re-implementation of software for the estimation of contemporary effective population size (Ne) from genetic data. Mol. Ecol. Resour. 2014, 14, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [PubMed]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Earl, D.A.; vonHoldt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Kopelman, N.M.; Mayzel, J.; Jakobsson, M.; Rosenberg, N.A.; Mayrose, I. CLUMPAK: A program for identifying clustering modes and packaging population structure inferences across K. Mol. Ecol. Resour. 2015, 15, 1179–1191. [Google Scholar] [CrossRef] [PubMed]
- Antao, T.; Lopes, A.; Lopes, R.J.; Beja-Pereira, A.; Luikart, G. LOSITAN: A workbench to detect molecular adaptation based on a Fst-outlier method. BMC Bioinf. 2008, 9, 323. [Google Scholar] [CrossRef] [PubMed]
- Langella, O. Populations Version 1.2.32. Available online: http://www.bioinformatics.org/project/?group_id=84 (accessed on 24 November 2014).
- Nei, M. Genetic distance between populations. Am. Nat. 1972, 106, 283–292. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Guichoux, E.; Lagache, L.; Wagner, S.; Chaumeil, P.; Leger, P.; Lepais, O.; Lepoittevin, C.; Malausa, T.; Revardel, E.; Salin, F.; et al. Current trends in microsatellite genotyping. Mol. Ecol. Resour. 2011, 11, 591–611. [Google Scholar] [CrossRef] [PubMed]
- Oddou-Muratorio, S.; Klein, E.K.; Vendramin, G.G.; Fady, B. Spatial vs. temporal effects on demographic and genetic structures: The roles of dispersal, masting and differential mortality on patterns of recruitment in Fagus sylvatica. Mol. Ecol. 2011, 20, 1997–2010. [Google Scholar] [CrossRef] [PubMed]
- Oddou-Muratorio, S.; Vendramin, G.G.; Buiteveld, J.; Fady, B. Population estimators or progeny tests: What is the best method to assess null allele frequencies at SSR loci? Conserv. Genet. 2009, 10, 1343–1347. [Google Scholar] [CrossRef]
- Magri, D. Patterns of post-glacial spread and the extent of glacial refugia of European beech (Fagus sylvatica). J. Biogeogr. 2008, 35, 450–463. [Google Scholar] [CrossRef]
- Magri, D.; Vendramin, G.G.; Comps, B.; Dupanloup, I.; Geburek, T.; Gömöry, D.; Latalowa, M.; Litt, T.; Paule, L.; Roure, J.M.; et al. A new scenario for the quaternary history of European beech populations: Palaeobotanical evidence and genetic consequences. New Phytol. 2006, 171, 199–221. [Google Scholar] [CrossRef] [PubMed]
- Pluess, A.R.; Weber, P. Drought-adaptation potential in Fagus sylvatica: Linking moisture availability with genetic diversity and dendrochronology. PLoS ONE 2012, 7, e33636. [Google Scholar] [CrossRef] [PubMed]
- Lalagüe, H.; Csilléry, K.; Oddou-Muratorio, S.; Safrana, J.; de Quattro, C.; Fady, B.; González-Martínez, S.C.; Vendramin, G.G. Nucleotide diversity and linkage disequilibrium at 58 stress response and phenology candidate genes in a European beech (Fagus sylvatica L.) population from southeastern France. Tree Genet. Genomes 2014, 10, 15–26. [Google Scholar] [CrossRef]
- Pluess, A.R.; Frank, A.; Heiri, C.; Lalague, H.; Vendramin, G.G.; Oddou-Muratorio, S. Genome-environment association study suggests local adaptation to climate at the regional scale in Fagus sylvatica. New Phytol. 2016, 210, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Eckert, A.J.; van Heerwaarden, J.; Wegrzyn, J.L.; Nelson, C.D.; Ross-Ibarra, J.; Gonzalez-Martinez, S.C.; Neale, D.B. Patterns of population structure and environmental associations to aridity across the range of loblolly pine (Pinus taeda L.; Pinaceae). Genetics 2010, 185, 969–982. [Google Scholar] [CrossRef] [PubMed]
- Chhatre, V.E.; Byram, T.D.; Neale, D.B.; Wegrzyn, J.L.; Krutovsky, K.V. Genetic structure and association mapping of adaptive and selective traits in the east Texas loblolly pine (Pinus taeda L.) breeding populations. Tree Genet. Genomes 2013, 9, 1161–1178. [Google Scholar] [CrossRef]
- Vidalis, A.; Curtu, A.L.; Finkeldey, R. Novel SNP development and analysis at a NADP+-specific IDH enzyme gene in a four species mixed oak forest. Plant Biol. 2013, 15, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Namroud, M.C.; Beaulieu, J.; Juge, N.; Laroche, J.; Bousquet, J. Scanning the genome for gene single nucleotide polymorphisms involved in adaptive population differentiation in white spruce. Mol. Ecol. 2008, 17, 3599–3613. [Google Scholar] [CrossRef] [PubMed]
- Parchman, T.L.; Jahner, J.P.; Uckele, K.A.; Galland, L.M.; Eckert, A.J. RADseq approaches and applications for forest tree genetics. Tree Genet. Genomes 2018, 14, 39. [Google Scholar] [CrossRef]
- Balkenhol, N.; Dudaniec, R.Y.; Krutovsky, K.V.; Johnson, J.S.; Cairns, D.M.; Segelbacher, G.; Selkoe, K.A.; von der Heyden, S.; Wang, I.J.; Selmoni, O.; et al. Landscape genomics: Understanding relationships between environmental heterogeneity and genomic characteristics of populations. In Population Genomics; Rajora, O., Ed.; Springer: Cham, Switzerland, 2017. [Google Scholar]
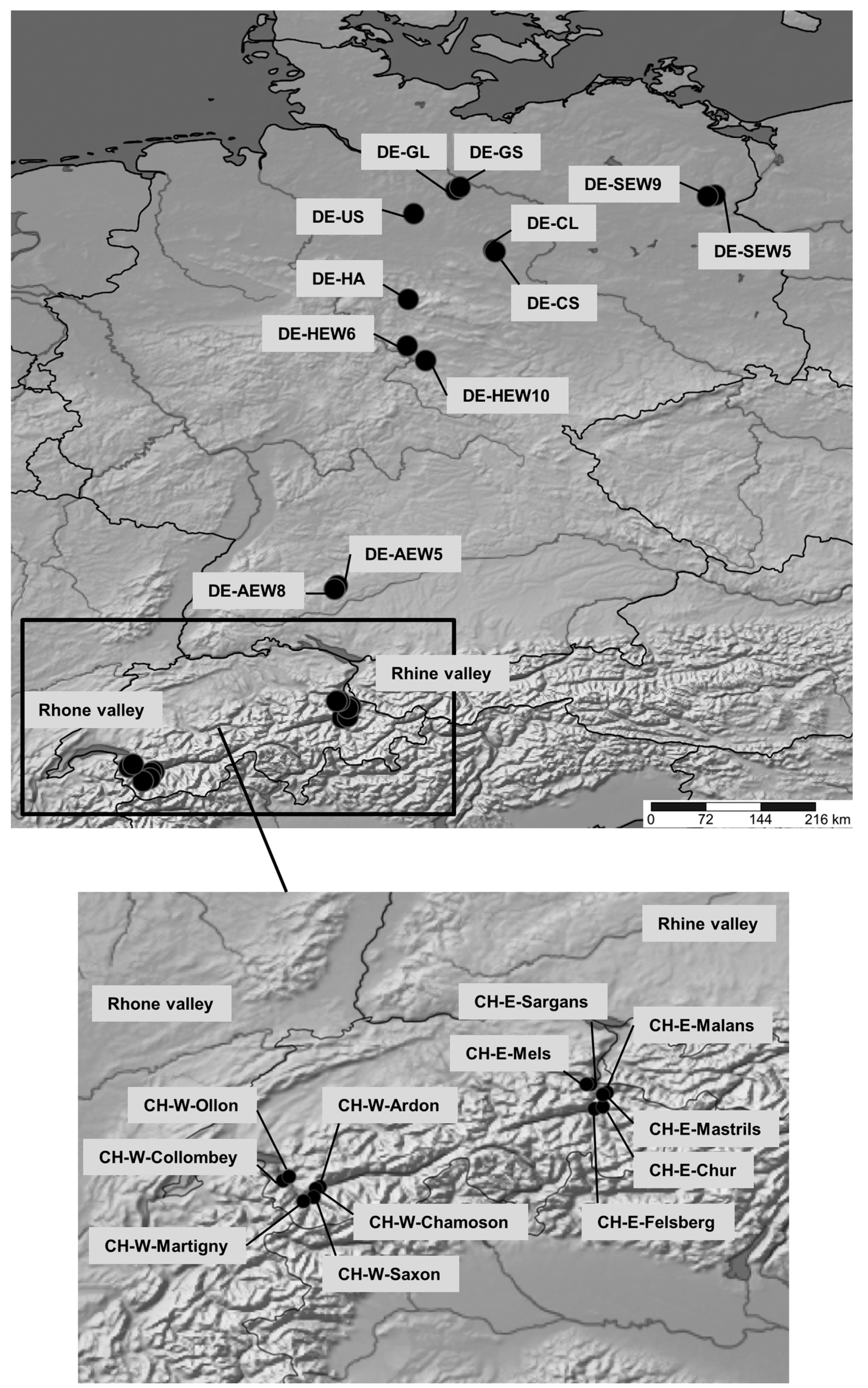
| Population | Location | Number | Altitude, m a.s.l. | Annual Temperature, °C | Mean Vegetation Season Temperature, °C | Mean Number of Summer Days a | Mean Number of Heat Days b | Mean annual Precipitation, mm | Mean Vegetation Season Precipitation, mm | EQ c | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Adult Trees | Seedlings | ||||||||||
| DE-SEW5 | 53°3′25.3″ N 13°53′7.3″ E | 200 | 200 | 64 | 8.3 | 15.38 | 33.4 | 5.6 | 532.1 | 273 | 32.9 |
| DE-SEW9 | 53°2′40.5″ N 13°48′36.4″ E | 198 | 200 | 79 | 8.3 | 15.38 | 33.4 | 5.6 | 532.1 | 273 | 32.9 |
| DE-GL | 53°07′ N 10°49′ E | 100 | 228 | 85 | 8.6 | 15.24 | 27.5 | 4.6 | 545.2 | 272 | 31.5 |
| DE-GS | 53°09′ N 10°52′ E | 103 | 144 | 85 | 8.6 | 15.24 | 27.5 | 4.6 | 545.2 | 272 | 31.5 |
| DE-US | 52°50′ N 10°19′ E | 100 | 211 | 117 | 8.1 | 14.58 | 26.8 | 4.8 | 800.5 | 352 | 20.6 |
| DE-CL | 52°24′ N 11°16′ E | 101 | 266 | 72 | 8.5 | 15.2 | 33.1 | 6.5 | 562.8 | 278 | 30.6 |
| DE-CS | 52°23′ N 11°17′ E | 104 | 230 | 75 | 8.5 | 15.2 | 33.1 | 6.5 | 562.8 | 278 | 30.6 |
| DE-HA | 51°49′ N 10°15′ E | 99 | 152 | 458 | 6.2 | 12.6 | 8 | 0.1 | 1326 | 548 | 10.9 |
| DE-HEW10 | 51°5′24″ N 10°27′44.8″ E | 200 | 200 | 378 | 7.5 | 14.06 | 18.7 | 1.7 | 662.6 | 306 | 24 |
| DE-HEW6 | 51°15′50″ N 10°14′27.4″ E | 200 | 200 | 435 | 7.5 | 14.06 | 18.7 | 1.7 | 662.6 | 306 | 24 |
| DE-AEW5 | 48°25′10.6″ N 9°24′52.9″ E | 198 | 200 | 788 | 6.5 | 13.14 | 20.5 | 1.6 | 963 | 477 | 16.1 |
| DE-AEW8 | 48°22′57.3″ N 9°22′56.6″ E | 199 | 200 | 766 | 6.5 | 13.14 | 20.5 | 1.6 | 963 | 477 | 16.1 |
| CH-E-Felsberg | 46°51′ N 9°28′ E | 25 | 62 | 725 | 9.2 | 15.8 | 40 | 5.4 | 798 | 430 | 22.6 |
| CH-E-Chur | 46°52′ N 9°32′ E | 25 | 63 | 750 | 9.2 | 15.8 | 40 | 5.4 | 798 | 430 | 22.6 |
| CH-E-Malans | 46°59′ N 9°34′ E | 25 | 64 | 650 | 9.3 | 16 | 36.6 | 3.5 | 1095 | 583 | 16.6 |
| CH-E-Mastrils | 46°58′ N 9°32′ E | 25 | 62 | 600 | 9.3 | 16 | 36.6 | 3.5 | 1095 | 583 | 16.6 |
| CH-E-Sargans | 47°3′ N 9°26′ E | 25 | 63 | 700 | 9.3 | 16 | 36.6 | 3.5 | 1318 | 647 | 13.8 |
| CH-E-Mels | 47°3′ N 9°24′ E | 25 | 60 | 700 | 9.3 | 16 | 36.6 | 3.5 | 1318 | 647 | 13.8 |
| CH-W-Ardon | 46°13′ N 7°14′ E | 25 | 63 | 800 | 9.2 | 16.5 | 55.3 | 10.8 | 598 | 233 | 31.9 |
| CH-W-Chamoson | 46°12′ N 7°12′ E | 25 | 64 | 800 | 9.2 | 16.5 | 55.3 | 10.8 | 598 | 233 | 31.9 |
| CH-W-Saxon | 46°8′ N 7°11′ E | 25 | 64 | 750 | 9.2 | 16.5 | 55.3 | 10.8 | 598 | 233 | 31.9 |
| CH-W-Martigny | 46°6′ N 7°6′ E | 25 | 64 | 600 | 9.2 | 16.5 | 55.3 | 10.8 | 843 | 319 | 22.7 |
| CH-W-Collombey | 46°16′ N 6°56′ E | 25 | 63 | 600 | 8.9 | 15.5 | 35.1 | 2 | 1032 | 492 | 17.4 |
| CH-W-Ollon | 46°18′ N 6°59′ E | 25 | 63 | 650 | 8.9 | 15.5 | 35.1 | 2 | 1032 | 492 | 17.4 |
| Name | Type | Gene |
|---|---|---|
| CP10_65 | Synonymous | Chloroplast chaperonin like |
| CP10_67 | Non-synonymous | |
| CP10_377 | Non-coding | |
| CP10_442 | Non-coding | |
| CP10_503 | Synonymous | |
| CP10_749 | Synonymous | |
| CP10_1317 | Non-coding | |
| CP10_1428 | Non-synonymous | |
| CysPro_118 | Synonymous | Cystein proteinase |
| CysPro_202 | Synonymous | |
| CysPro_728 | UTR | |
| CysPro_783 | UTR | |
| DAG_81 | UTR | DOF zinc finger protein |
| DAG_289 | Non-coding | |
| DAG_1059 | Synonymous | |
| His3C1_292 | Non-coding | Histone 3 |
| His3C2_104 | Synonymous | |
| His3C2_186 | Non-coding | |
| His3C2_260 | Synonymous | |
| NAC_854 | Non-synonymous | NAC transcription factor |
| NAC_962 | Synonymous | |
| NAC_1300 | UTR | |
| PP2C_315 | Non-synonymous | Protein phosphatase 2C |
| PP2C_391 | Synonymous | |
| PP2C_791 | Non-synonymous | |
| PP2C_941 | Non-coding | |
| PP2C_1200 | Synonymous |
| Population | Seedlings | Adults | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | AR | Ho | He | FIS | N | AR | Ho | He | FIS | |
| DE-SEW5 | 100 | 7.3 | 0.599 | 0.645 | 0.076 * | 200 | 6.6 | 0.636 | 0.646 | 0.018 |
| DE-SEW9 | 100 | 7.3 | 0.580 | 0.611 | 0.057 * | 198 | 6.1 | 0.620 | 0.616 | −0.005 |
| DE-GL | 148 | 7.6 | 0.610 | 0.633 | 0.041 * | 100 | 6.2 | 0.643 | 0.635 | −0.008 |
| DE-GS | 129 | 8.4 | 0.622 | 0.628 | 0.014 | 103 | 6.6 | 0.602 | 0.638 | 0.062 * |
| DE-US | 149 | 7.8 | 0.565 | 0.600 | 0.062 * | 100 | 6.2 | 0.594 | 0.595 | 0.005 |
| DE-CL | 134 | 7.7 | 0.596 | 0.603 | 0.016 | 101 | 6.5 | 0.581 | 0.599 | 0.036 |
| DE-CS | 142 | 8 | 0.631 | 0.635 | 0.009 | 104 | 6.2 | 0.599 | 0.619 | 0.036 |
| DE-HA | 152 | 8.2 | 0.627 | 0.638 | 0.020 | 99 | 6.4 | 0.612 | 0.630 | 0.033 |
| DE-HEW10 | 179 | 7.6 | 0.609 | 0.641 | 0.053 * | 200 | 6.1 | 0.607 | 0.626 | 0.033 |
| DE-HEW6 | 100 | 7.3 | 0.618 | 0.623 | 0.013 | 200 | 6.4 | 0.608 | 0.614 | 0.012 |
| DE-AEW5 | 169 | 7.9 | 0.585 | 0.625 | 0.066 * | 198 | 6.4 | 0.621 | 0.610 | −0.015 |
| DE-AEW8 | 100 | 7.2 | 0.589 | 0.600 | 0.023 | 199 | 6.1 | 0.627 | 0.621 | −0.006 |
| CH-E-Felsberg | 62 | 6.9 | 0.579 | 0.588 | 0.024 | 25 | 6.2 | 0.600 | 0.605 | 0.029 |
| CH-E-Chur | 63 | 6.9 | 0.591 | 0.592 | 0.010 | 25 | 5.2 | 0.547 | 0.547 | 0.022 |
| CH-E-Malans | 64 | 6.5 | 0.606 | 0.597 | −0.008 | 25 | 6.0 | 0.600 | 0.573 | −0.027 |
| CH-E-Mastrils | 62 | 6.3 | 0.573 | 0.576 | 0.012 | 25 | 5.4 | 0.573 | 0.560 | −0.004 |
| CH-E-Sargans | 63 | 6.9 | 0.626 | 0.602 | −0.032 | 25 | 6.1 | 0.596 | 0.604 | 0.035 |
| CH-E-Mels | 60 | 7.4 | 0.635 | 0.616 | −0.023 | 25 | 6.4 | 0.582 | 0.582 | 0.020 |
| CH-W-Ardon | 63 | 6.9 | 0.587 | 0.588 | 0.009 | 25 | 5.7 | 0.582 | 0.587 | 0.028 |
| CH-W-Chamoson | 64 | 5.4 | 0.580 | 0.579 | 0.007 | 25 | 5.4 | 0.591 | 0.578 | −0.002 |
| CH-W-Saxon | 64 | 6.7 | 0.583 | 0.592 | 0.023 | 25 | 5.4 | 0.573 | 0.595 | 0.057 |
| CH-W-Martigny | 64 | 6.6 | 0.604 | 0.589 | −0.017 | 25 | 5.2 | 0.573 | 0.581 | 0.033 |
| CH-W-Collombey | 63 | 7.2 | 0.563 | 0.590 | 0.054 | 25 | 6.0 | 0.604 | 0.603 | 0.018 |
| CH-W-Ollon | 63 | 6.8 | 0.607 | 0.591 | −0.019 | 25 | 5.3 | 0.573 | 0.542 | −0.037 |
| Mean | 98 | 7.2 | 0.599 | 0.608 | 0.020 | 88 | 6.0 | 0.598 | 0.600 | 0.016 |
| Population | GST |
|---|---|
| DE-SEW5 | 0.0022 * |
| DE-SEW9 | 0.0013 * |
| DE-GL | 0.0020 * |
| DE-GS | 0.0005 |
| DE-US | 0.0019 * |
| DE-CL | 0.0005 |
| DE-CS | 0.0004 |
| DE-HA | 0.0025 * |
| DE-HEW10 | 0.0021 * |
| DE-HEW6 | 0.0011 * |
| DE-AEW5 | 0.0016 * |
| DE-AEW8 | 0.0059 * |
| CH-E-Felsberg | 0.0001 |
| CH-E-Chur | 0.0033 |
| CH-E-Malans | 0.0007 |
| CH-E-Mastrils | 0.0015 |
| CH-E-Sargans | 0.0011 |
| CH-E-Mels | 0.0011 |
| CH-W-Ardon | 0.0010 |
| CH-W-Chamoson | 0.0050 * |
| CH-W-Saxon | 0.0000 |
| CH-W-Martigny | 0.0025 |
| CH-W-Collombey | 0.0017 |
| CH-W-Ollon | 0.0008 |
| Mean | 0.0017 |
| Population | N | Ho | He | FIS |
|---|---|---|---|---|
| DE-SEW5 | 200 | 0.266 | 0.258 | −0.027 |
| DE-SEW9 | 200 | 0.259 | 0.253 | −0.023 |
| DE-GL | 228 | 0.273 | 0.279 | 0.025 |
| DE-GS | 144 | 0.269 | 0.270 | 0.008 |
| DE-US | 211 | 0.280 | 0.284 | 0.015 |
| DE-CL | 266 | 0.246 | 0.248 | 0.011 |
| DE-CS | 230 | 0.273 | 0.278 | 0.020 |
| DE-HA | 123 | 0.270 | 0.278 | 0.033 |
| DE-HEW10 | 200 | 0.248 | 0.249 | 0.006 |
| DE-HEW6 | 200 | 0.271 | 0.277 | 0.023 |
| DE-AEW5 | 200 | 0.281 | 0.277 | −0.012 |
| DE-AEW8 | 200 | 0.254 | 0.275 | 0.078 * |
| CH-E-Felsberg | 62 | 0.290 | 0.278 | −0.036 |
| CH-E-Chur | 63 | 0.288 | 0.278 | −0.029 |
| CH-E-Malans | 64 | 0.242 | 0.246 | 0.024 |
| CH-E-Mastrils | 62 | 0.269 | 0.271 | 0.016 |
| CH-E-Sargans | 63 | 0.282 | 0.273 | −0.028 |
| CH-E-Mels | 60 | 0.244 | 0.252 | 0.042 |
| CH-W-Ardon | 63 | 0.276 | 0.250 | −0.095 |
| CH-W-Chamoson | 64 | 0.279 | 0.288 | 0.037 |
| CH-W-Saxon | 64 | 0.192 | 0.226 | 0.156 * |
| CH-W-Martigny | 64 | 0.231 | 0.233 | 0.015 |
| CH-W-Collombey | 63 | 0.249 | 0.252 | 0.019 |
| CH-W-Ollon | 63 | 0.249 | 0.241 | −0.025 |
| Mean | 131.5 | 0.262 | 0.263 | 0.011 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Müller, M.; Cuervo-Alarcon, L.; Gailing, O.; K.C., R.; Chhetri, M.S.; Seifert, S.; Arend, M.; Krutovsky, K.V.; Finkeldey, R. Genetic Variation of European Beech Populations and Their Progeny from Northeast Germany to Southwest Switzerland. Forests 2018, 9, 469. https://doi.org/10.3390/f9080469
Müller M, Cuervo-Alarcon L, Gailing O, K.C. R, Chhetri MS, Seifert S, Arend M, Krutovsky KV, Finkeldey R. Genetic Variation of European Beech Populations and Their Progeny from Northeast Germany to Southwest Switzerland. Forests. 2018; 9(8):469. https://doi.org/10.3390/f9080469
Chicago/Turabian StyleMüller, Markus, Laura Cuervo-Alarcon, Oliver Gailing, Rajendra K.C., Meena Suyal Chhetri, Sarah Seifert, Matthias Arend, Konstantin V. Krutovsky, and Reiner Finkeldey. 2018. "Genetic Variation of European Beech Populations and Their Progeny from Northeast Germany to Southwest Switzerland" Forests 9, no. 8: 469. https://doi.org/10.3390/f9080469
APA StyleMüller, M., Cuervo-Alarcon, L., Gailing, O., K.C., R., Chhetri, M. S., Seifert, S., Arend, M., Krutovsky, K. V., & Finkeldey, R. (2018). Genetic Variation of European Beech Populations and Their Progeny from Northeast Germany to Southwest Switzerland. Forests, 9(8), 469. https://doi.org/10.3390/f9080469









