Light Energy Partitioning and Photoprotection in an Exotic Species (Salix Psammophila) Grown in a Semi-Arid Area of Northwestern China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Long-Term Monitoring of ChlF and Hydrometeorological Variables
2.3. ChlF-Parameter Calculation
2.4. Statistical Analyses
3. Results
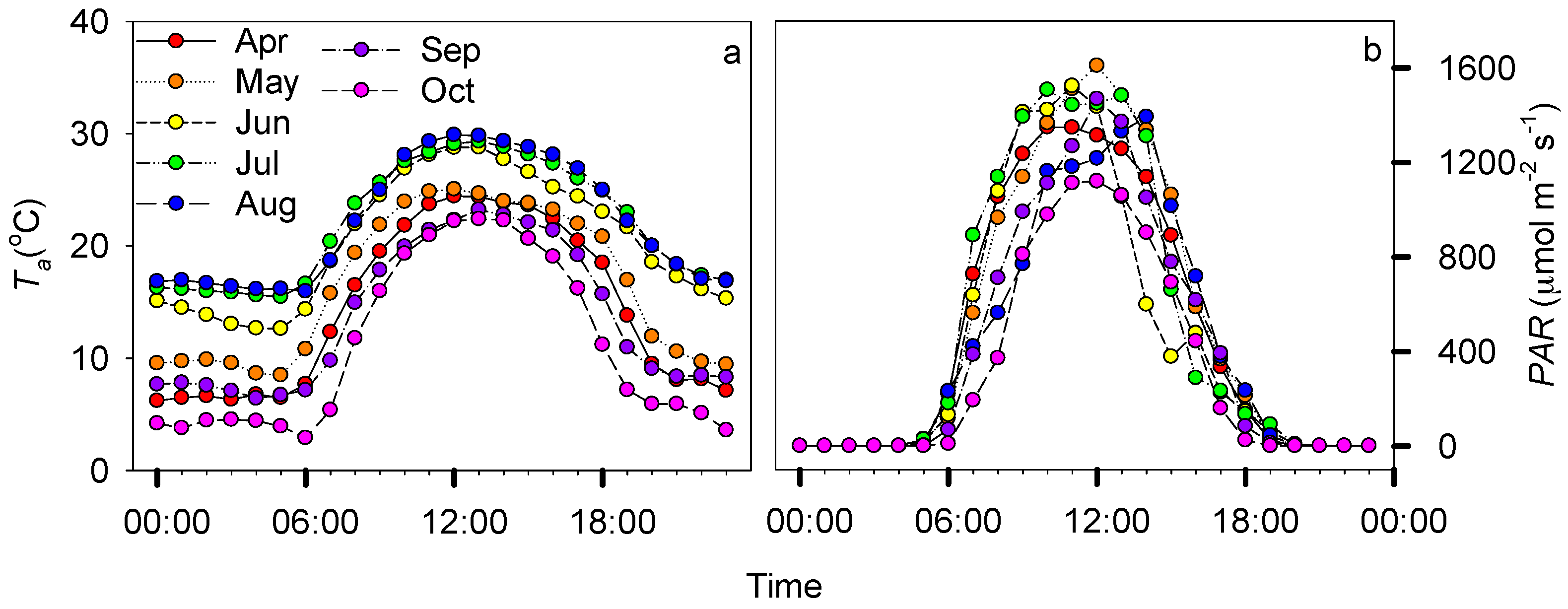
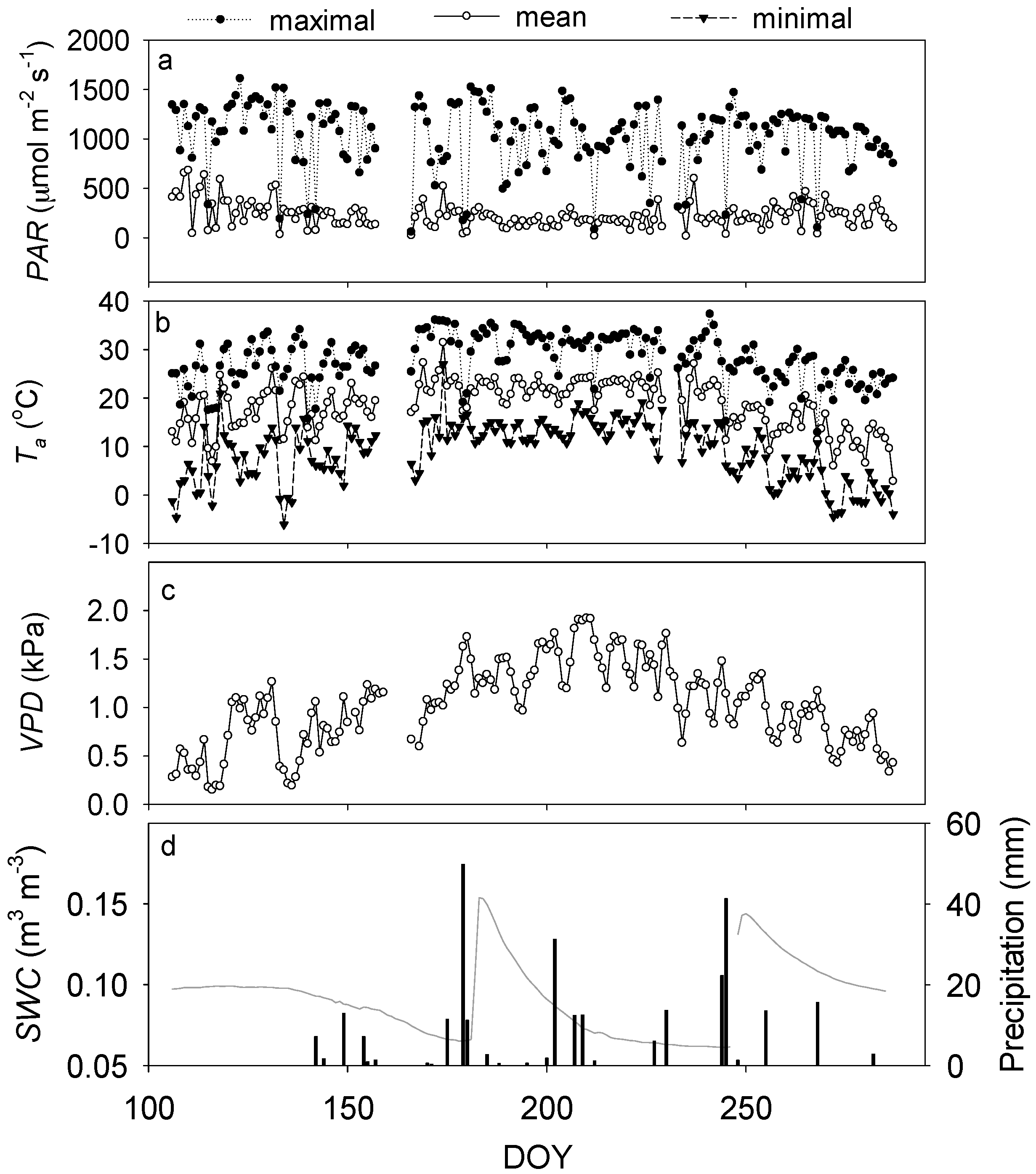
3.1. Environmental Dynamics
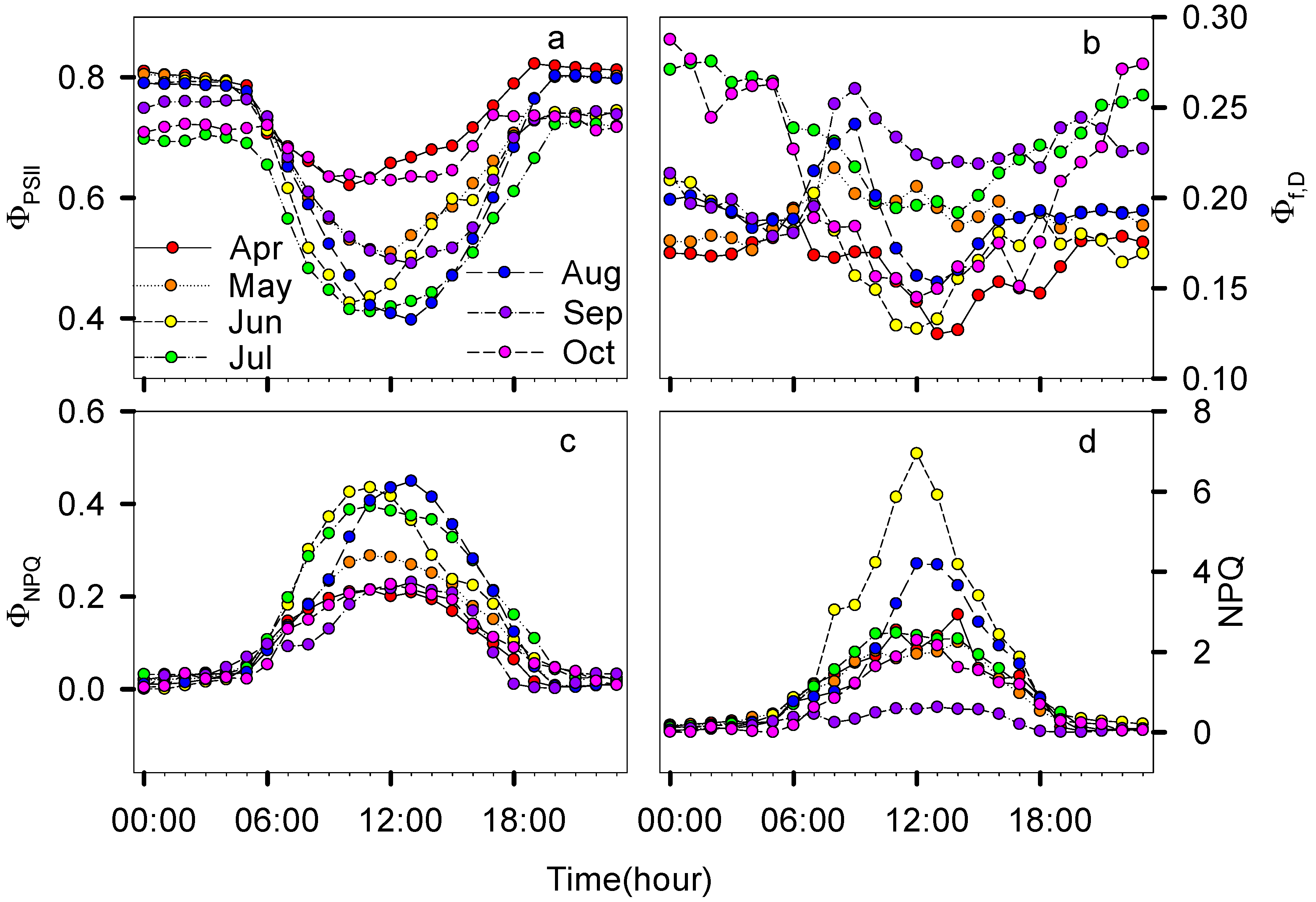
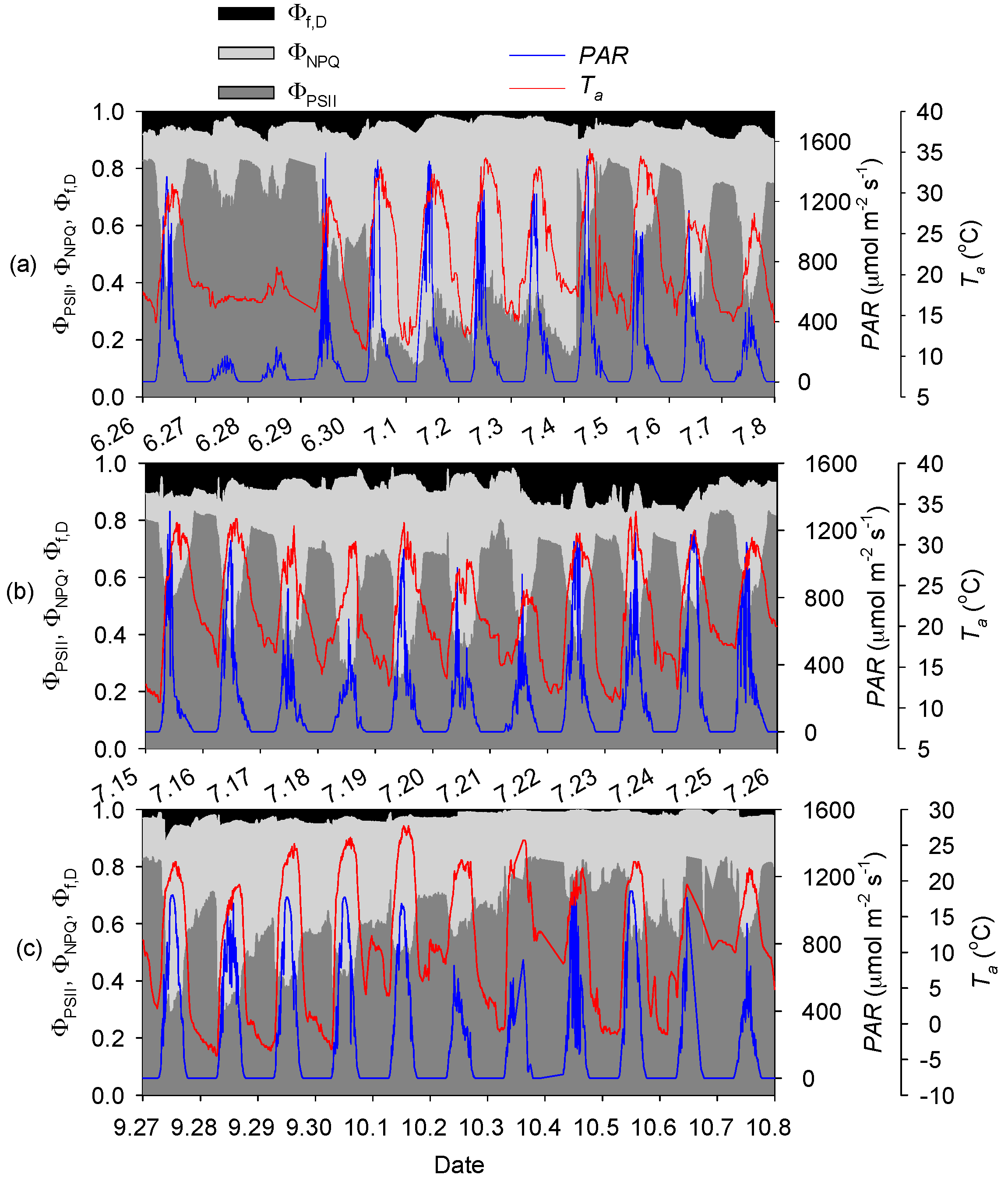
3.2. Seasonal and Diurnal Dynamics in Energy Partitioning Components
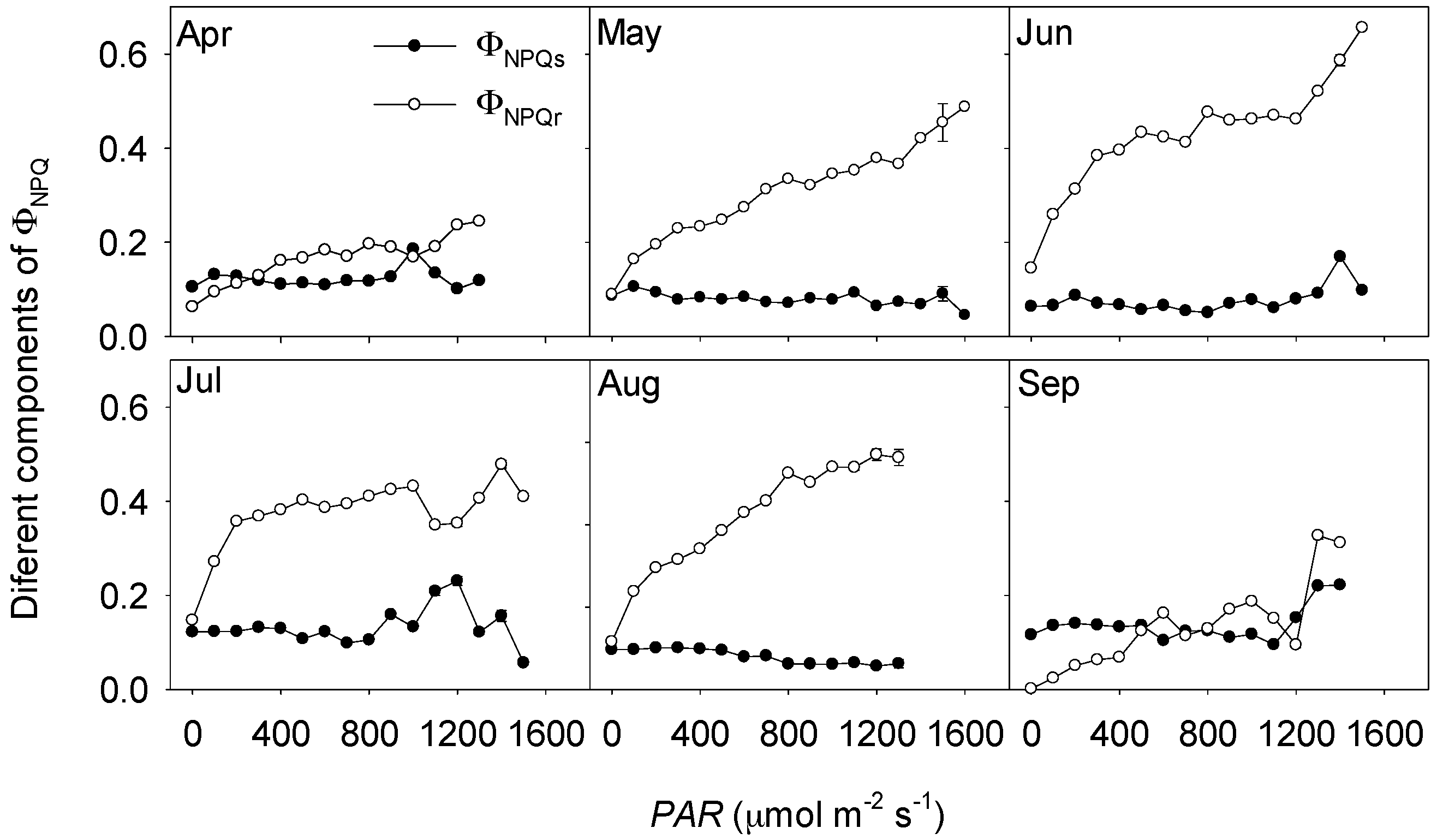
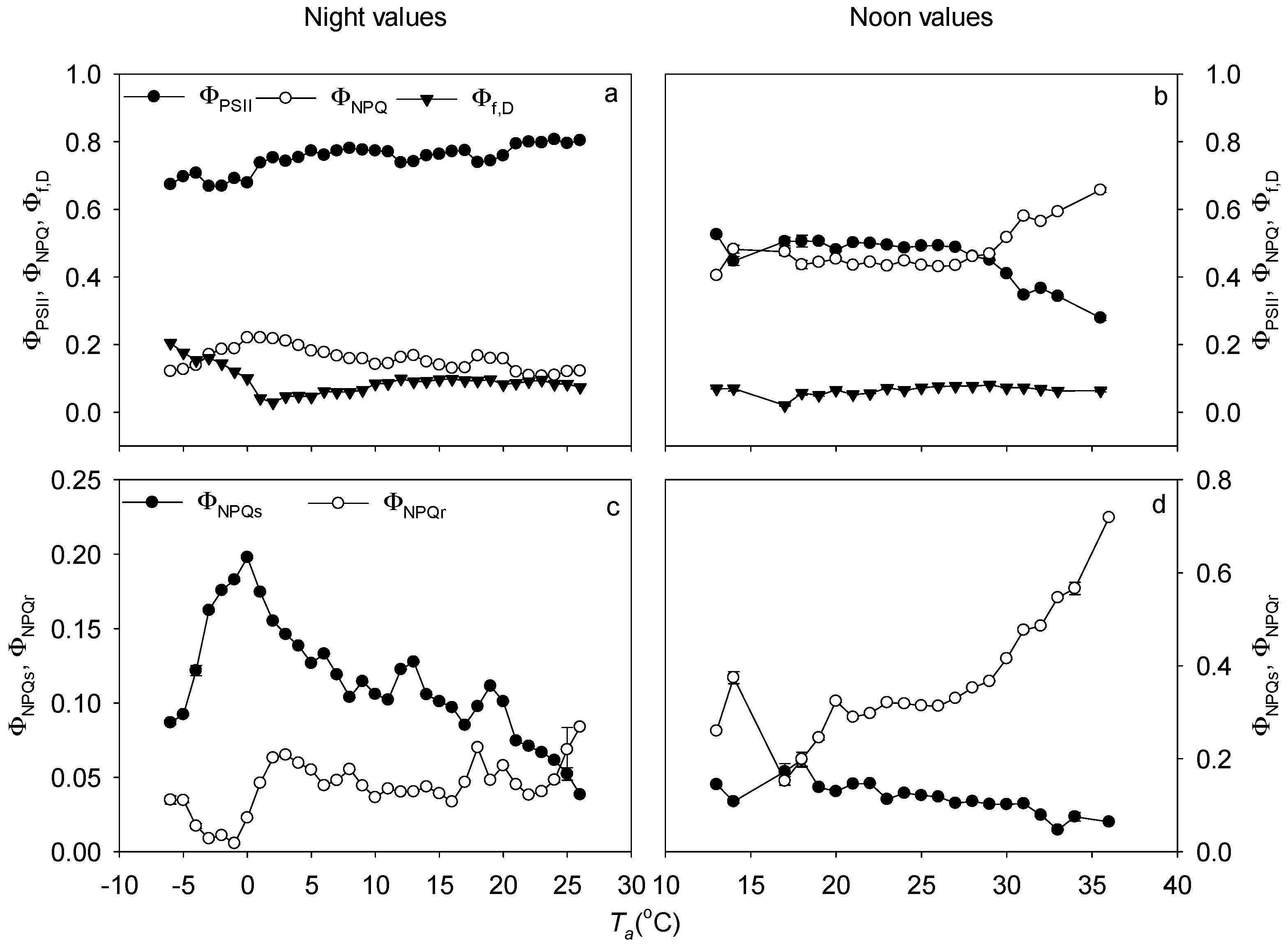
3.3. Non-Photochemical Quenching in Response to Solar Radation Stress
3.4. Non-Photochemical Quenching in Response to Temperature Stress
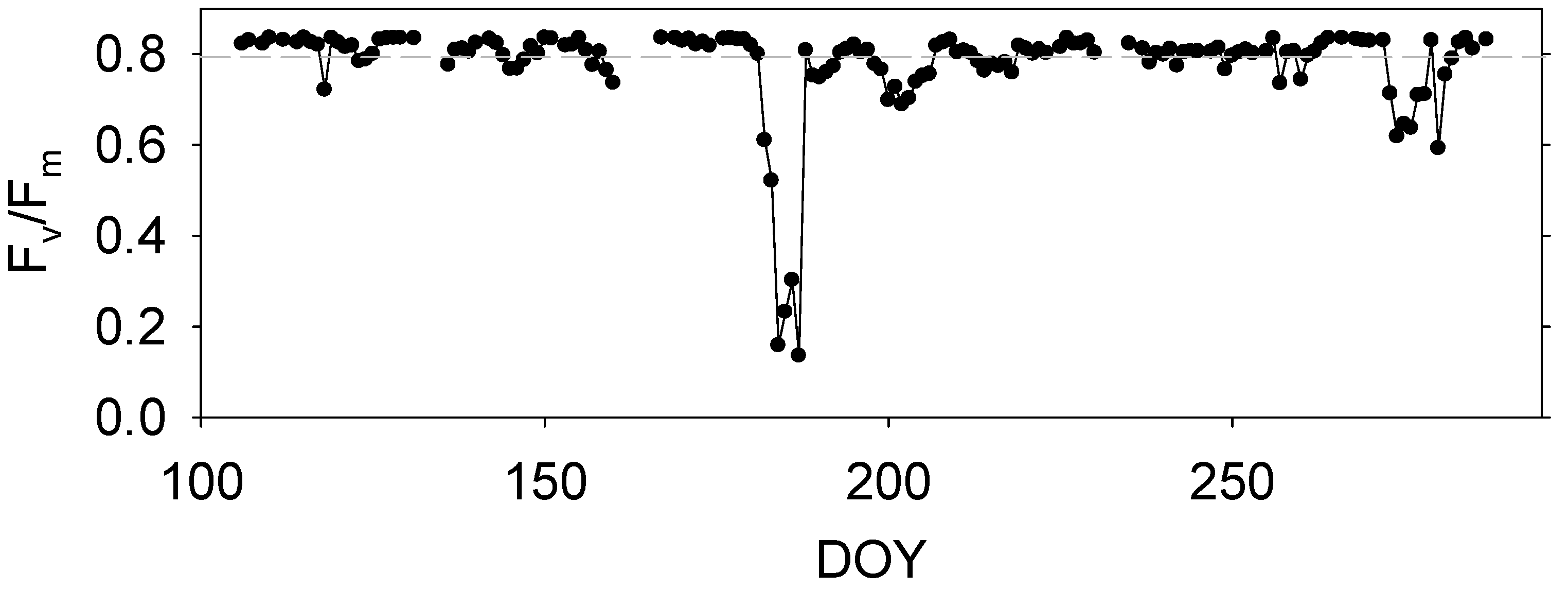
3.5. Seasonl Dynamics of Daily Fv/Fm
4. Discussion
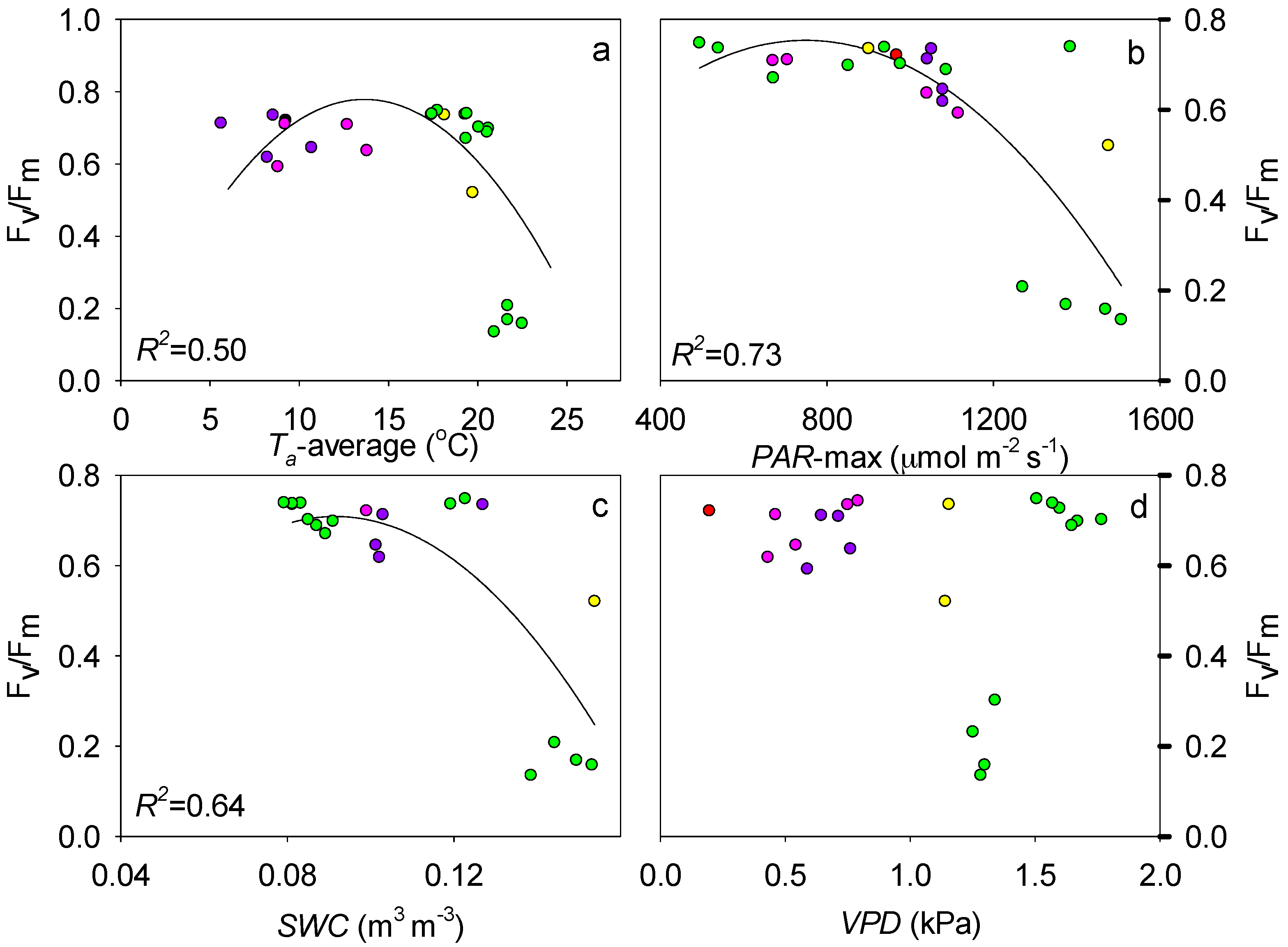
4.1. Effect of Abiotic Factors on Fv/Fm
4.2. Energy Partitioning in Response to Low Temperatures
4.3. Energy Partitioning in Response to High Temperatures
4.4. Energy Partitioning in Response to Solar Radiation
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ChlF | Chlorophyll a Flouresence |
| PSII | Photosystem II |
| LHCII | Light-harvesting complex II |
| RC | Reaction center |
| PsbS | Photoprotective protein |
| ROS | Reactive oxygen species |
| ∆pH | Transthylakoid pH gradient |
| Ft | ChlF at any time |
| Fm′ | Maximal ChlF in light |
| Fm | Maximal ChlF in dark |
| Fo | Original ChlF |
| FmR | Reference maximal fluorescence |
| Fv | Variable fluorescence |
| Fv/Fm | Maximal quantum yield of PSII photochemistry |
| Fv′/Fm′ | Effective photochemical efficiency |
| NPQ | Non-photochemical quenching |
| ΦPSII | Actual quantum yield of PSII photochemistry |
| ΦNPQ | Regulated thermal energy loss |
| Non-regulated thermal energy loss | |
| ΦNPQs | Sustained thermal dissipation |
| ΦNPQr | Rapidly reversible thermal dissipation |
| PAR | Photosynthetically active radiation |
| PAR-max | Maximal PAR |
| VPD | Vapor pressure Deficit |
| Ta | Air temperature |
| SWC | Soil water content |
References
- Oquist, G.; Huner, N.P. Photosynthesis of overwintering evergreen plants. Annu. Rev. Plant Biol. 2003, 54, 329–355. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Porcar-Castell, A.; Juurola, E.; Ensminger, I.; Berninger, F.; Hari, P.; Nikinmaa, E. Seasonal acclimation of photosystem II in Pinus sylvestris. II. Using the rate constants of sustained thermal energy dissipation and photochemistry to study the effect of the light environment. Tree Physiol. 2008, 28, 1483–1491. [Google Scholar] [CrossRef] [PubMed]
- Howarth, J.F.; Durako, M.J. Diurnal variation in chlorophyll fluorescence of Thalassia testudinum seedlings in response to controlled salinity and light conditions. Mar. Biol. 2013, 160, 591–605. [Google Scholar] [CrossRef]
- Haque, M.S.; Kjaer, K.H.; Rosenqvist, E.; Sharma, D.K.; Ottosen, C.O. Heat stress and recovery of photosystem II efficiency in wheat (Triticum aestivum L.) cultivars acclimated to different growth temperatures. Environ. Exp. Bot. 2014, 99, 1–8. [Google Scholar] [CrossRef]
- Janka, E.; Körner, O.; Rosenqvist, E.; Ottosen, C.-O. Using the quantum yields of photosystem II and the rate of net photosynthesis to monitor high irradiance and temperature stress in chrysanthemum (Dendranthema grandiflora). Plant Physiol. Biochem. 2015, 90, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Zunzunegui, M.; Barradas, M.C.D.; Ain-Lhout, F.; Alvarez-Cansino, L.; Esquivias, M.; Novo, F.G. Seasonal physiological plasticity and recovery capacity after summer stress in Mediterranean scrub communities. Plant Ecol. 2011, 212, 127–142. [Google Scholar] [CrossRef]
- Wong, S.L.; Chen, C.W.; Huang, H.W.; Weng, J.H. Using combined measurements of gas exchange and chlorophyll fluorescence to investigate the photosynthetic light responses of plant species adapted to different light regimes. Photosynthetica 2012, 50, 206–214. [Google Scholar] [CrossRef]
- Oukarroum, A.; Strasser, R.J.; Schansker, G. Heat stress and the photosynthetic electron transport chain of the lichen Parmelina tiliacea (Hoffm.) Ach. in the dry and the wet state: Differences and similarities with the heat stress response of higher plants. Photosynth. Res. 2012, 111, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Melkonian, J.; Owens, T.G.; Wolfe, D.W. Gas exchange and co-regulation of photochemical and nonphotochemical quenching in bean during chilling at ambient and elevated carbon dioxide. Photosynth. Res. 2004, 79, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Chen, J.; Abd_Allah, E.; Wang, P.; Wang, G.; Hu, X.; Shi, J. Quantitative proteomics analysis reveals that S-nitrosoglutathione reductase (GSNOR) and nitric oxide signaling enhance poplar defense against chilling stress. Planta 2015, 242, 1361–1390. [Google Scholar] [CrossRef] [PubMed]
- Lavaud, J.; Rousseau, B.; Etienne, A.L. In diatoms, a transthylakoid proton gradient alone is not sufficient to induce a non-photochemical fluorescence quenching. FEBS Lett. 2002, 523, 163–166. [Google Scholar] [CrossRef]
- Ruban, A.V.; Berera, R.; Ilioaia, C.; Van Stokkum, I.H.; Kennis, J.T.; Pascal, A.A.; Van Amerongen, H.; Robert, B.; Horton, P.; Van Grondelle, R. Identification of a mechanism of photoprotective energy dissipation in higher plants. Nature 2007, 450, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.W.; Kuang, S.B.; Long, G.Q.; Yang, S.C.; Meng, Z.G.; Li, L.G.; Chen, Z.J.; Zhang, G.H. Photosynthesis, light energy partitioning, and photoprotection in the shade-demanding species Panax notoginseng under high and low level of growth irradiance. Funct. Plant Biol. 2016, 43, 479–491. [Google Scholar] [CrossRef]
- Yamori, W. Photosynthetic response to fluctuating environments and photoprotective strategies under abiotic stress. J. Plant Res. 2016, 129, 379–395. [Google Scholar] [CrossRef] [PubMed]
- Hallik, L.; Niinemets, Ü.; Kull, O. Photosynthetic acclimation to light in woody and herbaceous species: A comparison of leaf structure, pigment content and chlorophyll fluorescence characteristics measured in the field. Plant Biol. 2012, 14, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, L.; Furbank, R.T.; Chow, W.S. A simple alternative approach to assessing the fate of absorbed light energy using chlorophyll fluorescence. Photosynth. Res. 2004, 82, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Klughammer, C.; Schreiber, U. Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the Saturation Pulse method. PAM Appl. Notes 2008, 1, 201–247. [Google Scholar]
- Losciale, P.; Chow, W.S.; Corelli Grappadelli, L. Modulating the light environment with the peach ‘asymmetric orchard’: Effects on gas exchange performances, photoprotection, and photoinhibition. J. Exp. Bot. 2010, 61, 1177–1192. [Google Scholar] [CrossRef] [PubMed]
- Kramer, D.M.; Johnson, G.; Kiirats, O.; Edwards, G.E. New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth. Res. 2004, 79, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Adams, W.W. Photoprotection in an Ecological Context: The Remarkable Complexity of Thermal Energy Dissipation. New Phytol. 2006, 172, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Li, X.-P.; Niyogi, K.K. Non-photochemical quenching. A response to excess light energy. Plant Physiol. 2001, 125, 1558–1566. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, A.S.; Adams, W.W.; Demmig-Adams, B. Two forms of sustained xanthophyll cycle-dependent energy dissipation in overwintering Euonymus kiautschovicus. Plant Cell Environ. 1998, 21, 893–903. [Google Scholar] [CrossRef]
- Lloyd, J. Soil Temperature and Intermittent Frost Modulate the Rate of Recovery of Photosynthesis in Scots Pine under Simulated Spring Conditions. New Phytol. 2008, 177, 428. [Google Scholar]
- Busch, F.; Hüner, N.P.; Ensminger, I. Increased air temperature during simulated autumn conditions does not increase photosynthetic carbon gain but affects the dissipation of excess energy in seedlings of the evergreen conifer Jack pine. Plant Physiol. 2007, 143, 1242–1251. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.; Ren, R. Meridional and downward propagation of atmospheric circulation anomalies. Part I: Northern Hemisphere cold season variability. J. Atmos. Sci. 2007, 64, 1880–1901. [Google Scholar] [CrossRef]
- Chen, S.; Wang, J.; Xing, X.; Guo, J. Relationship between Abnormal OLR over Qinghai-Tibet Plateau and Air Temperature in Arid Region of Northwest China. J. Arid Meteorol. 2011, 3, 276–282. [Google Scholar]
- Chen, S.; Zheng, Y.; Lou, W.; Guo, Y. Changes in the First Frost Date from 1961 to 2009 in Northwest China. Resour. Sci. 2013, 35, 165–172. [Google Scholar]
- Saxe, H.; Ellsworth, D.S.; Heath, J. Tansley Review No. 98 Tree and Forest Functioning in an Enriched CO2 Atmosphere. New Phytol. 1998, 139, 395–436. [Google Scholar] [CrossRef]
- Sicher, R.C.; Bunce, J.A. Relationship of photosynthetic acclimation to changes of Rubisco activity in field-grown winter wheat and barley during growth in elevated carbon dioxide. Photosynth. Res. 1997, 52, 27–38. [Google Scholar] [CrossRef]
- Bunce, J.A. Direct and acclimatory responses of stomatal conductance to elevated carbon dioxide in four herbaceous crop species in the field. Glob. Chang. Biol. 2001, 7, 323–331. [Google Scholar] [CrossRef]
- Yamori, W.; Hikosaka, K.; Way, D.A. Temperature response of photosynthesis in C3, C4, and CAM plants: Temperature acclimation and temperature adaptation. Photosynth. Res. 2014, 119, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.B.; Hu, H.; Xu, K.; Li, Z.R.; Yang, Y.P. Flexible and reversible responses to different irradiance levels during photosynthetic acclimation of Cypripedium guttatum. J. Plant Physiol. 2007, 164, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Shen, Z.; Liu, Y.; Wang, L.; Hannaway, D.; Lu, H. Effects of shade treatments on the photosynthetic capacity, chlorophyll fluorescence, and chlorophyll content of Tetrastigma hemsleyanum Diels et Gilg. Environ. Exp. Bot. 2009, 65, 177–182. [Google Scholar] [CrossRef]
- Munson, S.M. Plant responses, climate pivot points, and trade-offs in water-limited ecosystems. Ecosphere 2013, 4, 1–15. [Google Scholar] [CrossRef]
- Maphangwa, K.W.; Musil, C.F.; Raitt, L.; Zedda, L. Experimental climate warming decreases photosynthetic efficiency of lichens in an arid South African ecosystem. Oecologia 2012, 169, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Osório, M.L.; Osório, J.; Romano, A. Photosynthesis, energy partitioning, and metabolic adjustments of the endangered Cistaceae species Tuberaria major under high temperature and drought. Photosynthetica 2013, 51, 75–84. [Google Scholar] [CrossRef]
- Tominaga, J.; Inafuku, S.; Coetzee, T.; Kawamitsu, Y. Diurnal regulation of photosynthesis in Jatropha curcas under drought during summer in a semi-arid region. Biomass Bioenergy 2014, 67, 279–287. [Google Scholar] [CrossRef]
- Song, L.; Chow, W.S.; Sun, L.; Li, C.; Peng, C. Acclimation of photosystem II to high temperature in two Wedelia species from different geographical origins: Implications for biological invasions upon global warming. J. Exp. Bot. 2010, 61, 4087–4096. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.; Zeng, X.; Liu, X.; Zhao, P. Effects of moderate high-temperature stress on photosynthesis in three saplings of the constructive tree species of subtropical forest. Plant Cell Environ. 2007, 27, 1283–1290. [Google Scholar]
- Sharkey, T.D. Effects of moderate heat stress on photosynthesis: Importance of thylakoid reactions, rubisco deactivation, reactive oxygen species, and thermotolerance provided by isoprene. Plant Cell Environ. 2005, 28, 269–277. [Google Scholar] [CrossRef]
- Yamane, Y.; Kashino, Y.; Koike, H.; Satoh, K. Effects of high temperatures on the photosynthetic systems in spinach: Oxygen-evolving activities, fluorescence characteristics and the denaturation process. Photosynth. Res. 1998, 57, 51–59. [Google Scholar] [CrossRef]
- Stasik, O.; Jones, H.G. Response of photosynthetic apparatus to moderate high temperature in contrasting wheat cultivars at different oxygen concentrations. J. Exp. Bot. 2007, 58, 2133–2143. [Google Scholar] [CrossRef] [PubMed]
- Ort, D.R. Impacts of chilling temperatures on photosynthesis in warm-climate plants. Trends Plant Sci. 2001, 6, 36. [Google Scholar]
- Jiang, G.; Zhu, G. Effects of natural high temperature and irradiatic on photosynthesis and related parameters in three arid sandy shrub species. Acta Phytoecol. Sin. 2000, 25, 525–531. [Google Scholar]
- Chen, G.; Zhao, W. Age structure and dynamics of Salix psammophila branches in southern edge of the Mu Us Sandy Land. J. Desert Res. 2015, 35, 1520–1526. [Google Scholar]
- Chen, X.; Duan, Z. Changes in soil physical and chemical properties during reversal of desertification in Yanchi County of Ningxia Hui autonomous region, China. Environ. Geol. 2009, 57, 975–985. [Google Scholar] [CrossRef]
- Wang, B.; Zha, T.; Jia, X.; Wu, B.; Zhang, Y.; Qin, S. Soil moisture modifies the response of soil respiration to temperature in a desert shrub ecosystem. Biogeosciences 2014, 11, 259–268. [Google Scholar] [CrossRef]
- Porcar-Castell, A. A high-resolution portrait of the annual dynamics of photochemical and non-photochemical quenching in needles of Pinus sylvestris. Physiol. Plant. 2011, 143, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Zha, T.S.; Wu, Y.J.; Jia, X.; Zhang, M.Y.; Bai, Y.J.; Liu, P.; Ma, J.Y.; Bourque, P.A.; Peltola, H. Diurnal response of effective quantum yield of PSII photochemistry to irradiance as an indicator of photosynthetic acclimation to stressed environments revealed in a xerophytic species. Ecol. Indicators 2017, 74, 191–197. [Google Scholar] [CrossRef]
- Cailly, A.; Rizza, F.; Genty, B.; Harbinson, J. Fate of excitation at PS II in leaves. The non-photochemical side. In Proceedings of the 10th FESPP Meeting, Florence, Italy, 9–13 September 1996. [Google Scholar]
- Mathur, S.; Agrawal, D.; Jajoo, A. Photosynthesis: Response to high temperature stress. J. Photchem. Photobiol. B Biol. 2014, 137, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Kornyeyev, D.; Hendrickson, L. Research note: Energy partitioning in photosystem II complexes subjected to photoinhibitory treatment. Funct. Plant Biol. 2007, 34, 214–220. [Google Scholar] [CrossRef]
- Neveux, J.; Dupouy, C.; Blanchot, J.; Le Bouteiller, A.; Landry, M.R.; Brown, S.L. Diel dynamics of chlorophylls in high-nutrient, low-chlorophyll waters of the equatorial Pacific (180°): Interactions of growth, grazing, physiological responses, and mixing. J. Geophys. Res. Oceans 2003, 108. [Google Scholar] [CrossRef]
- Su, L.; Dai, Z.; Li, S.; Xin, H. A novel system for evaluating drought–cold tolerance of grapevines using chlorophyll fluorescence. BMC Plant Biol. 2015, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Prieto, P.; Peñuelas, J.; Llusià, J.; Asensio, D.; Estiarte, M. Effects of long-term experimental night-time warming and drought on photosynthesis, Fv/Fm and stomatal conductance in the dominant species of a Mediterranean shrubland. Acta Physiol. Plant. 2009, 31, 729–739. [Google Scholar] [CrossRef]
- Longenberger, P.S.; Smith, C.W.; Duke, S.E.; McMichael, B.L. Evaluation of chlorophyll fluorescence as a tool for the identification of drought tolerance in upland cotton. Euphytica 2009, 166, 25. [Google Scholar] [CrossRef]
- Wang, Z.X.; Chen, L.; Ai, J.; Qin, H.Y.; Liu, Y.X.; Xu, P.L.; Jiao, Z.Q.; Zhao, Y.; Zhang, Q.T. Photosynthesis and activity of photosystem II in response to drought stress in Amur Grape (Vitis amurensis Rupr.). Photosynthetica 2012, 50, 189–196. [Google Scholar] [CrossRef]
- Huner, N.P.A.; Öquist, G.; Sarhan, F. Energy balance and acclimation to light and cold. Trends Plant Sci. 1998, 3, 224–230. [Google Scholar] [CrossRef]
- Sveshnikov, D.; Ensminger, I.; Ivanov, A.G.; Campbell, D.; Lloyd, J.; Funk, C.; Hüner, N.P.; Oquist, G. Excitation energy partitioning and quenching during cold acclimation in Scots pine. Tree Physiol. 2006, 26, 325. [Google Scholar] [CrossRef] [PubMed]
- Hurry, V.M.; Strand, A.; Tobiaeson, M.; Gardestrom, P.; Oquist, G. Cold Hardening of Spring and Winter Wheat and Rape Results in Differential Effects on Growth, Carbon Metabolism, and Carbohydrate Content. Plant Physiol. 1995, 109, 697. [Google Scholar] [CrossRef] [PubMed]
- Nath, K.; Jajoo, A.; Poudyal, R.S.; Timilsina, R.; Park, Y.S.; Aro, E.M.; Nam, H.G.; Lee, C.H. Towards a critical understanding of the photosystem II repair mechanism and its regulation during stress conditions. FEBS Lett. 2013, 587, 3372–3381. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gong, B.; Xu, K. Interaction of nitric oxide and polyamines involves antioxidants and physiological strategies against chilling-induced oxidative damage in Zingiber officinale Roscoe. Sci. Hortic. 2014, 170, 237–248. [Google Scholar] [CrossRef]
- Lindfors, L.; Hölttä, T.; Lintunen, A.; Porcar-Castell, A.; Nikinmaa, E.; Juurola, E. Dynamics of leaf gas exchange, chlorophyll fluorescence and stem diameter changes during freezing and thawing of Scots pine seedlings. Tree Physiol. 2015, 35, 1314. [Google Scholar] [CrossRef] [PubMed]
- Gulías, J.; Cifre, J.; Jonasson, S.; Medrano, H.; Flexas, J. Seasonal and inter-annual variations of gas exchange in thirteen woody species along a climatic gradient in the Mediterranean island of Mallorca. Flora 2009, 204, 169–181. [Google Scholar] [CrossRef]
- Havaux, M.; Niyogi, K.K. The violaxanthin cycle protects plants from photooxidative damage by more than one mechanism. Proc. Natl. Acad. Sci. USA 1999, 96, 8762–8767. [Google Scholar] [CrossRef] [PubMed]
- Li, X.P.; Müller-Moulé, P.; Gilmore, A.M.; Niyogi, K.K. PsbS-Dependent Enhancement of Feedback De-Excitation Protects Photosystem II from Photoinhibition. Proc. Natl. Acad. Sci. USA 2002, 99, 15222. [Google Scholar] [CrossRef] [PubMed]
- Niyogi, K.K.; Li, X.P.; Rosenberg, V.; Jung, H.S. Is PsbS the site of non-photochemical quenching in photosynthesis? J. Exp. Bot. 2005, 56, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Song, L.Y.; Li, C.H.; Peng, S.L. Elevated CO2 increases energy-use efficiency of invasive wedelia trilobata over its indigenous congenger. Biol. Invasions 2010, 12, 1221–1230. [Google Scholar] [CrossRef]
- Rapparini, F.; Neri, L.; Mihailova, G.; Petkova, S.; Georgieva, K. Growth irradiance affects the photoprotective mechanisms of the resurrection angiosperm Haberlea rhodopensis Friv. in response to desiccation and rehydration at morphological, physiological and biochemical levels. Environ. Exp. Bot. 2015, 113, 67–79. [Google Scholar] [CrossRef]
- Aro, E.M.; Virgin, I.; Andersson, B. Photoinhibition of Photosystem II. Inactivation, protein damage and turnover. Biochim. Biophys. Acta Bioenerg. 1993, 1143, 113. [Google Scholar] [CrossRef]
- Aro, E.M.; Suorsa, M.; Rokka, A.; Allahverdiyeva, Y.; Paakkarinen, V.; Saleem, A.; Battchikova, N.; Rintamäki, E. Dynamics of photosystem II: A proteomic approach to thylakoid protein complexes. J. Exp. Bot. 2005, 56, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Badger, M.R. Photoprotection in plants: A new light on photosystem II damage. Trends Plant Sci. 2011, 16, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Kudoh, H.; Sonoike, K. Irreversible damage to photosystem I by chilling in the light: Cause of the degradation of chlorophyll after returning to normal growth temperature. Planta 2002, 215, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Ksas, B.; Becuwe, N.; Chevalier, A.; Havaux, M. Plant tolerance to excess light energy and photooxidative damage relies on plastoquinone biosynthesis. Sci. Rep. 2015, 5, 10919. [Google Scholar] [CrossRef] [PubMed]

| Process Yield | Equation | No. | Equation Source |
|---|---|---|---|
| Maximal quantum yield of PSII photochemistry | 1 | [49] | |
| Actual quantum yield of PSII photochemistry | 2 | [5,9,17,52] | |
| Non-regulated thermal energy loss | 3 | [17] | |
| Regulated thermal energy loss | 4 | [49,51] | |
| Sustained thermal dissipation | 5 | [3] | |
| Rapidly reversible thermal dissipation | 6 | [17,53] | |
| Non-photochemical quenching coefficient | 7 | [12,13] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, Y.; Wu, J.; Tian, Y.; Zha, T.; Jia, X.; Bourque, C.P.-A.; Wu, Y.; Bai, Y.; Ma, J.; Zhang, M. Light Energy Partitioning and Photoprotection in an Exotic Species (Salix Psammophila) Grown in a Semi-Arid Area of Northwestern China. Forests 2018, 9, 341. https://doi.org/10.3390/f9060341
Han Y, Wu J, Tian Y, Zha T, Jia X, Bourque CP-A, Wu Y, Bai Y, Ma J, Zhang M. Light Energy Partitioning and Photoprotection in an Exotic Species (Salix Psammophila) Grown in a Semi-Arid Area of Northwestern China. Forests. 2018; 9(6):341. https://doi.org/10.3390/f9060341
Chicago/Turabian StyleHan, Yini, Juying Wu, Yun Tian, Tianshan Zha, Xin Jia, Charles P.-A. Bourque, Yajuan Wu, Yujie Bai, Jingyong Ma, and Mingyan Zhang. 2018. "Light Energy Partitioning and Photoprotection in an Exotic Species (Salix Psammophila) Grown in a Semi-Arid Area of Northwestern China" Forests 9, no. 6: 341. https://doi.org/10.3390/f9060341
APA StyleHan, Y., Wu, J., Tian, Y., Zha, T., Jia, X., Bourque, C. P.-A., Wu, Y., Bai, Y., Ma, J., & Zhang, M. (2018). Light Energy Partitioning and Photoprotection in an Exotic Species (Salix Psammophila) Grown in a Semi-Arid Area of Northwestern China. Forests, 9(6), 341. https://doi.org/10.3390/f9060341





