Moisture-Limited Tree Growth for a Subtropical Himalayan Conifer Forest in Western Nepal
Abstract
1. Introduction
2. Materials and Methods
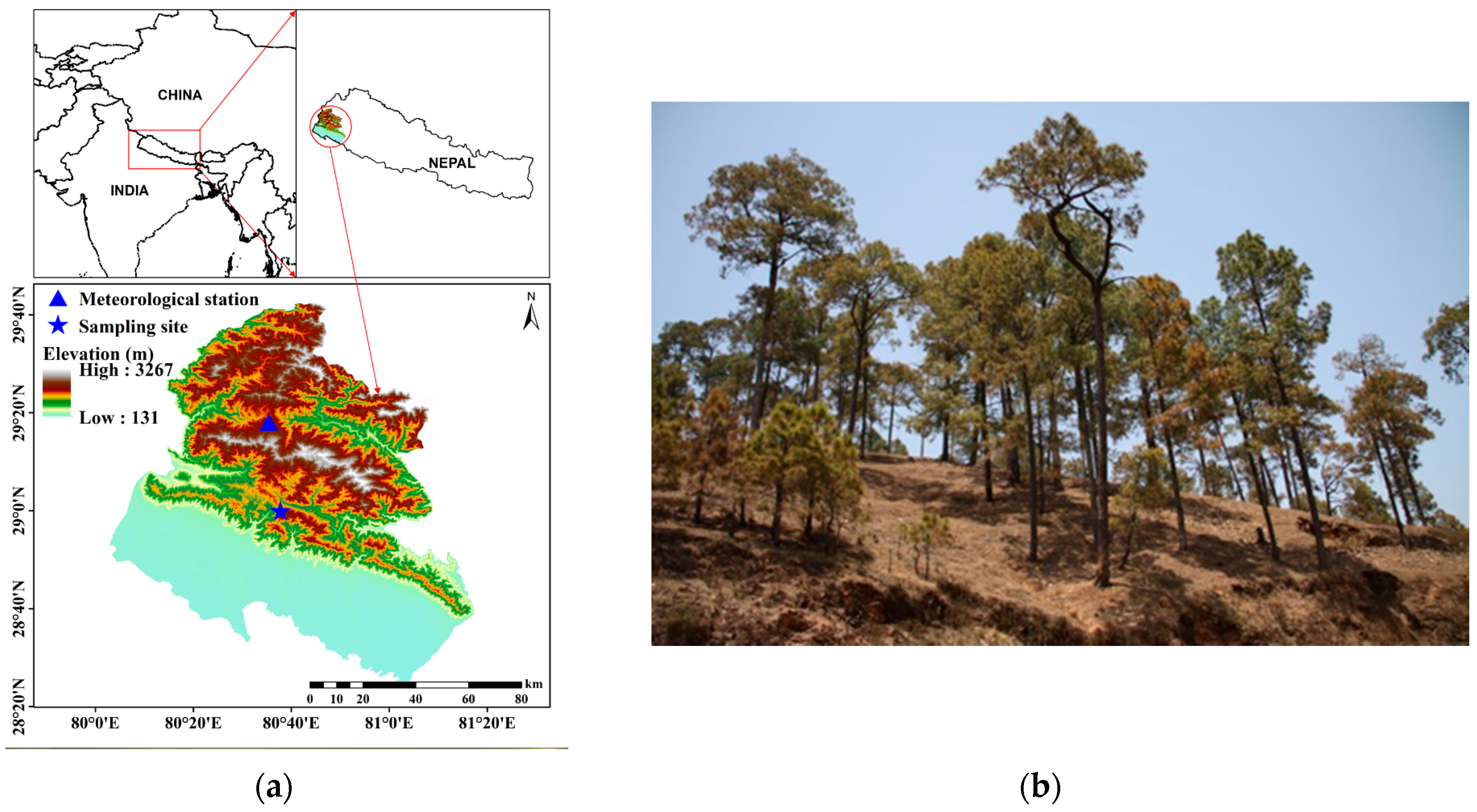
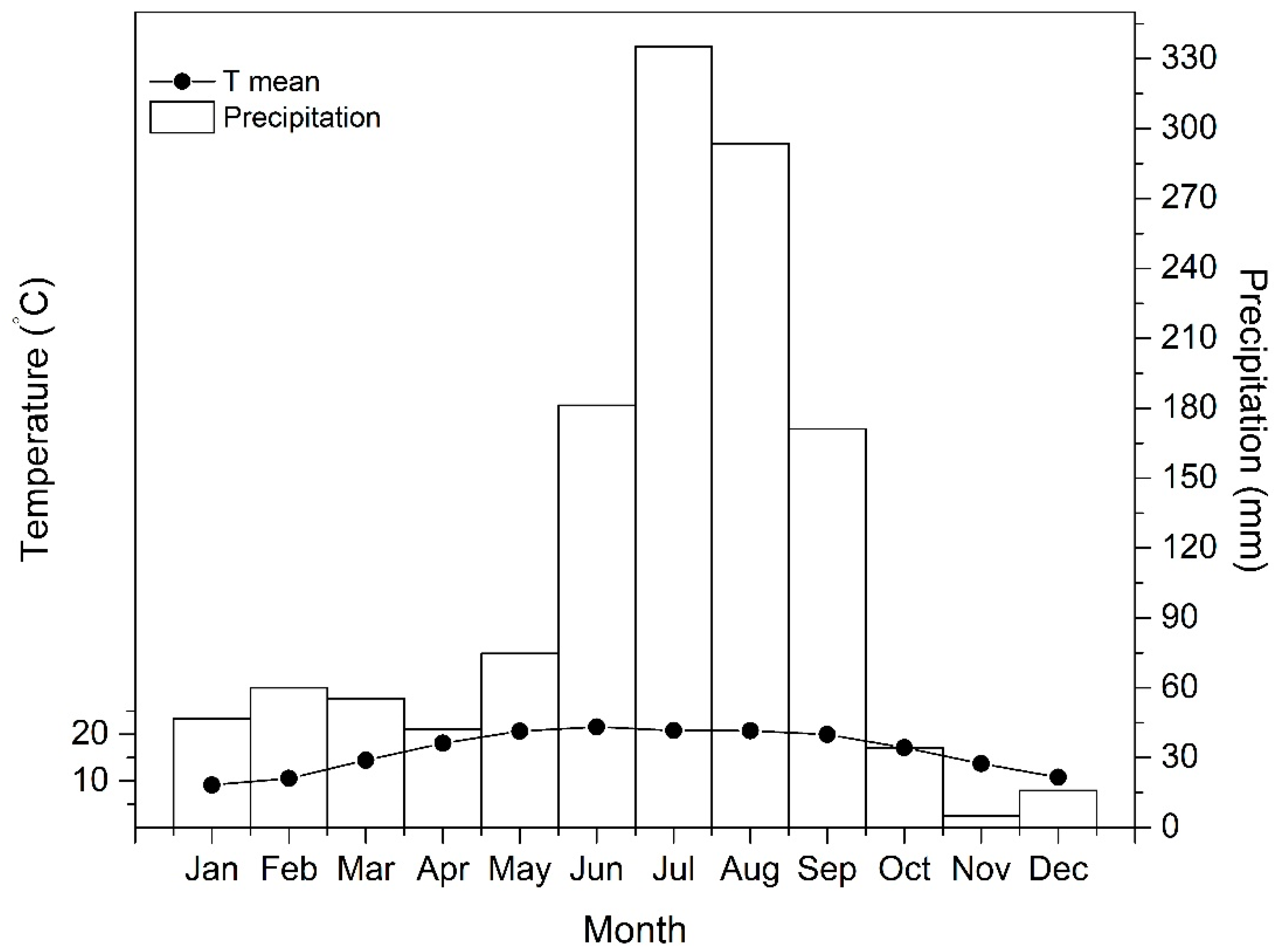
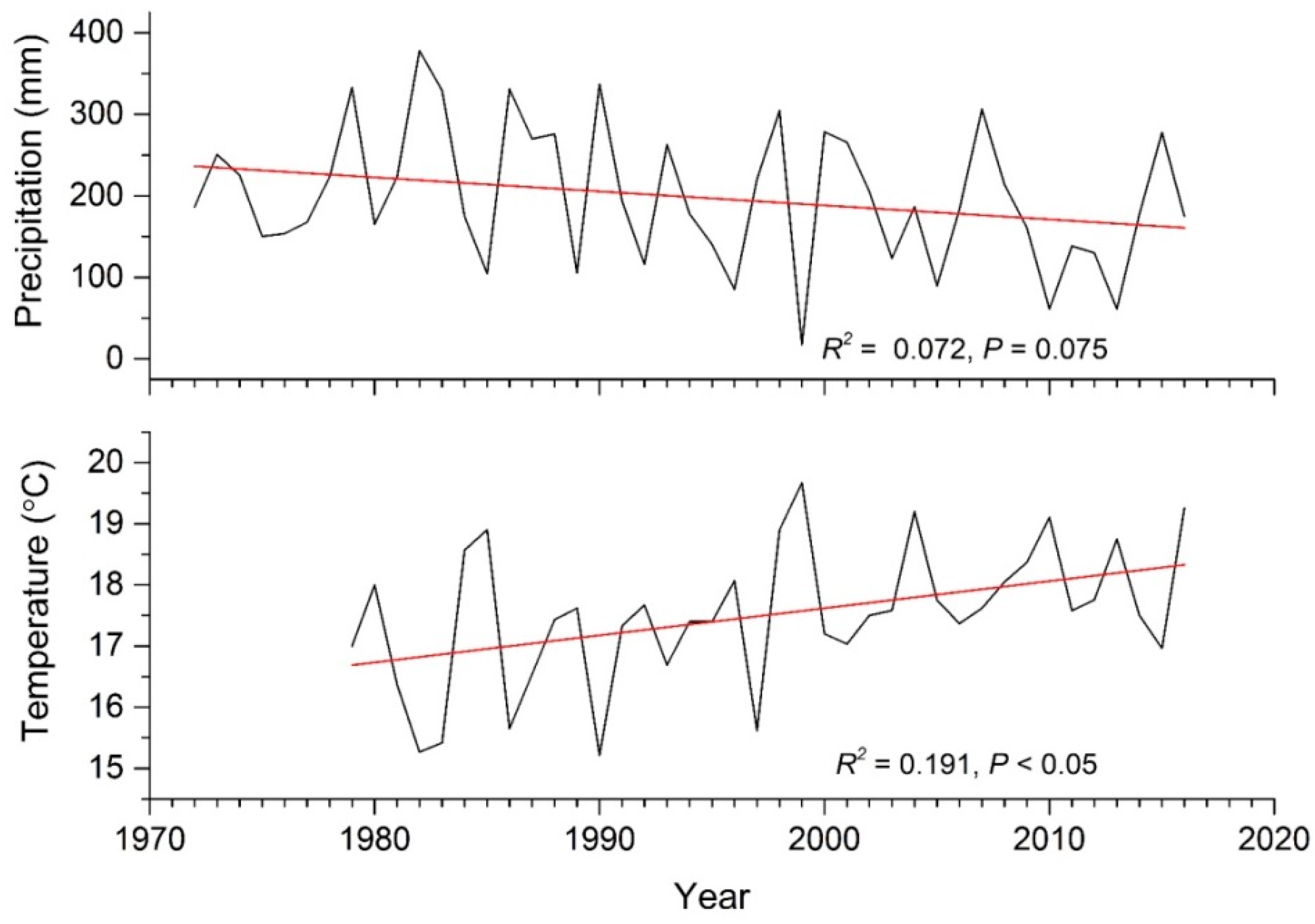
2.1. Study Area
2.2. Sample Collection and Dendrochronological Analyses
2.3. Climate-Growth Relationships
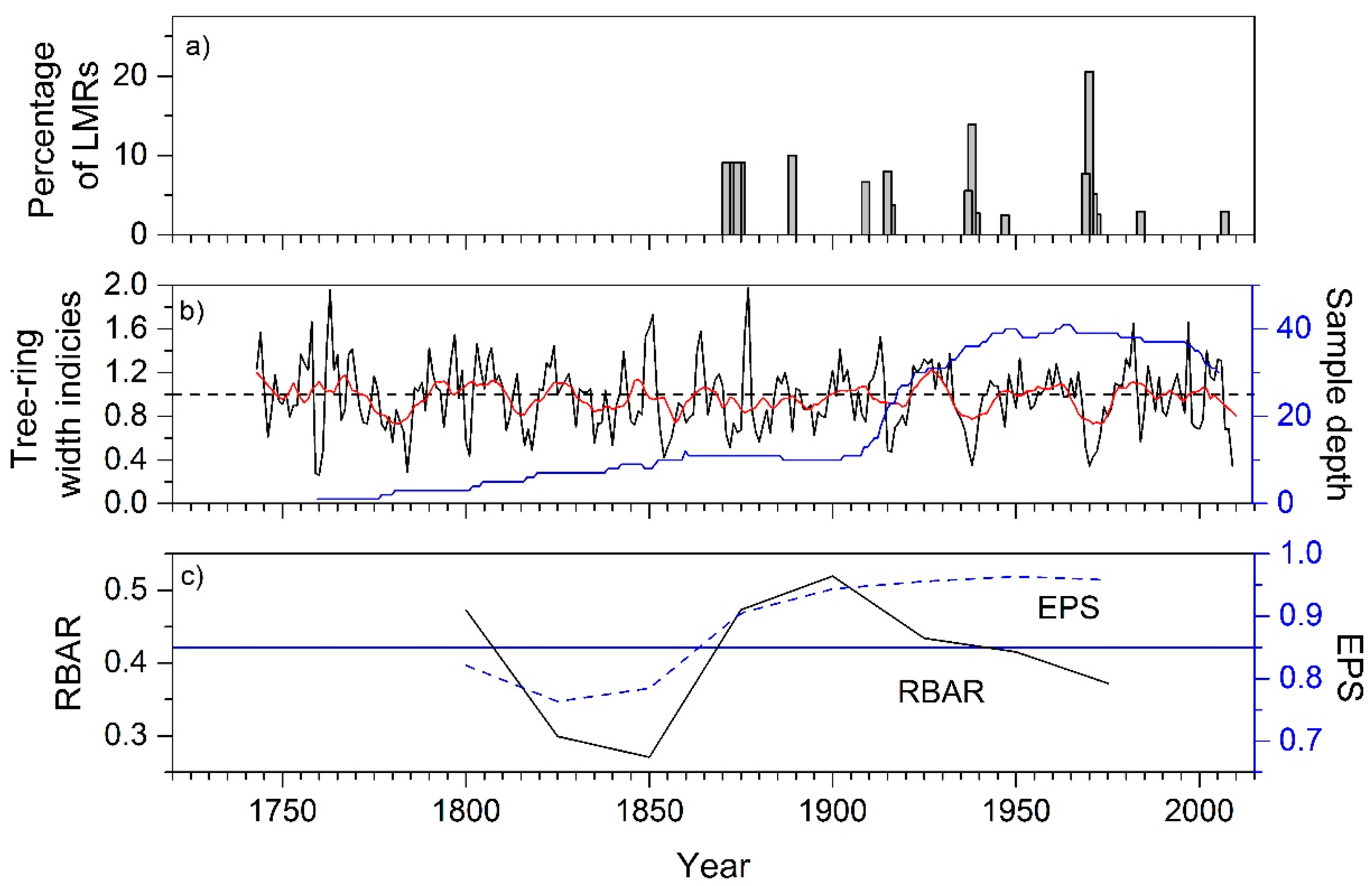
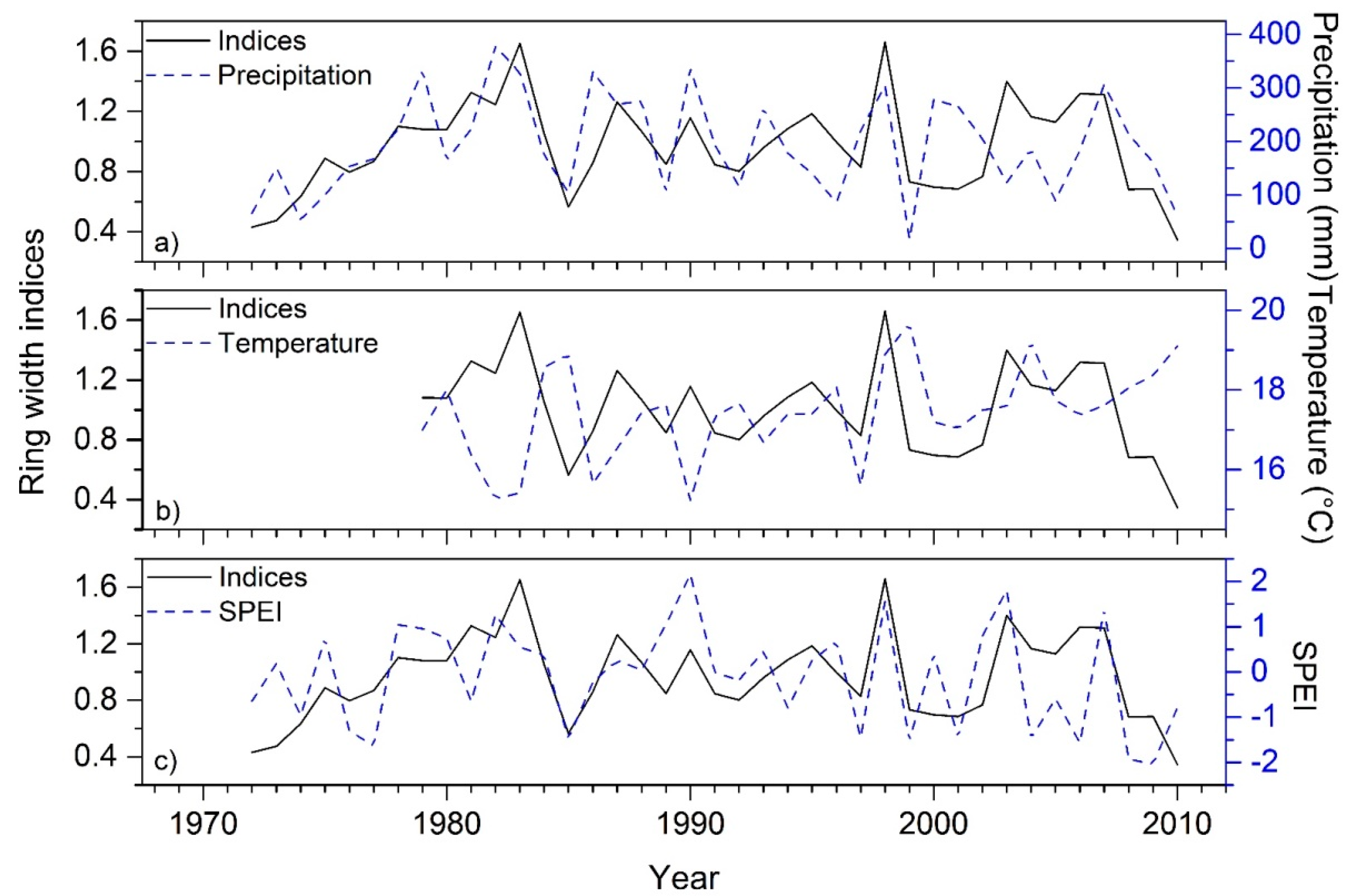
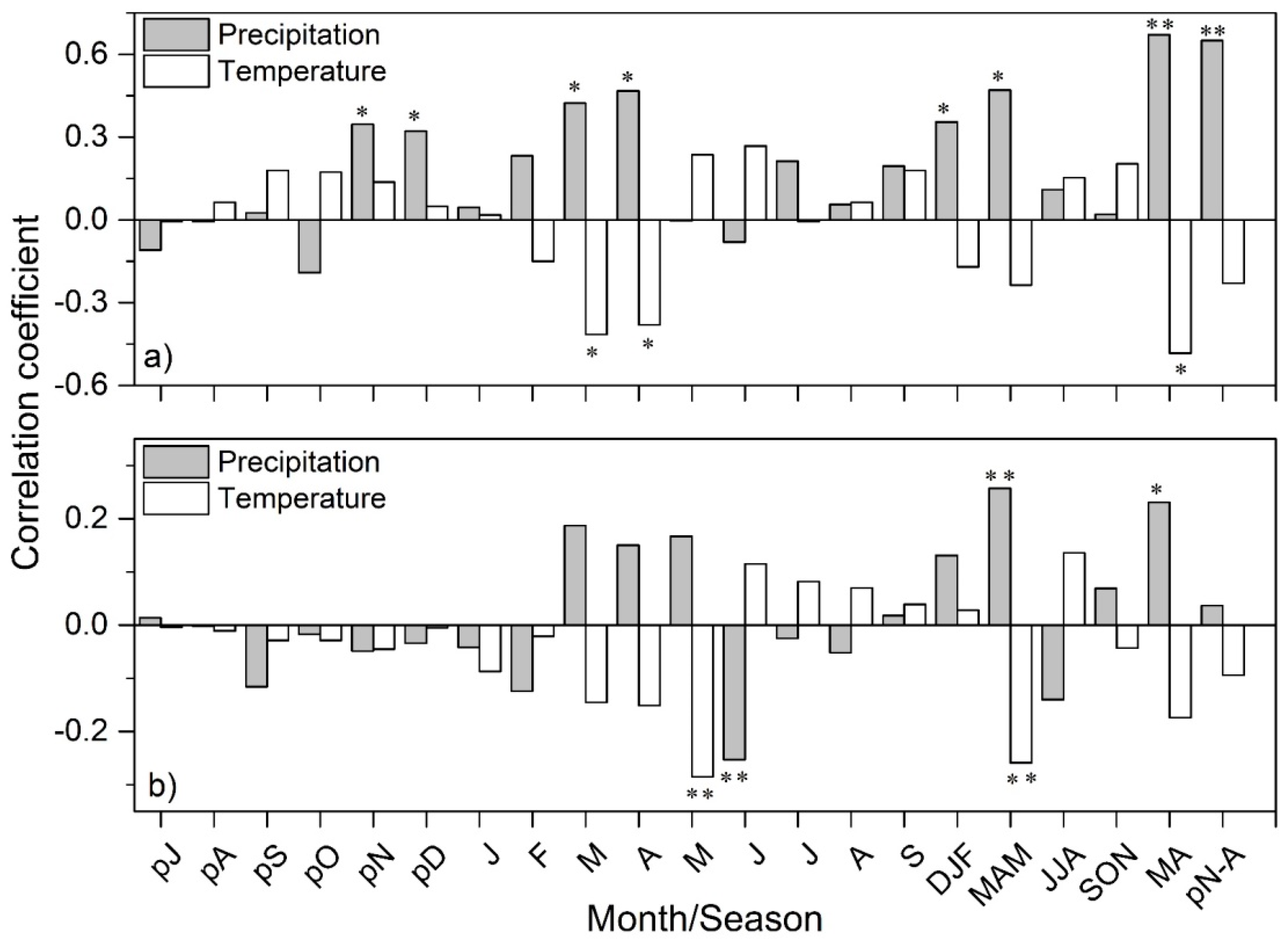
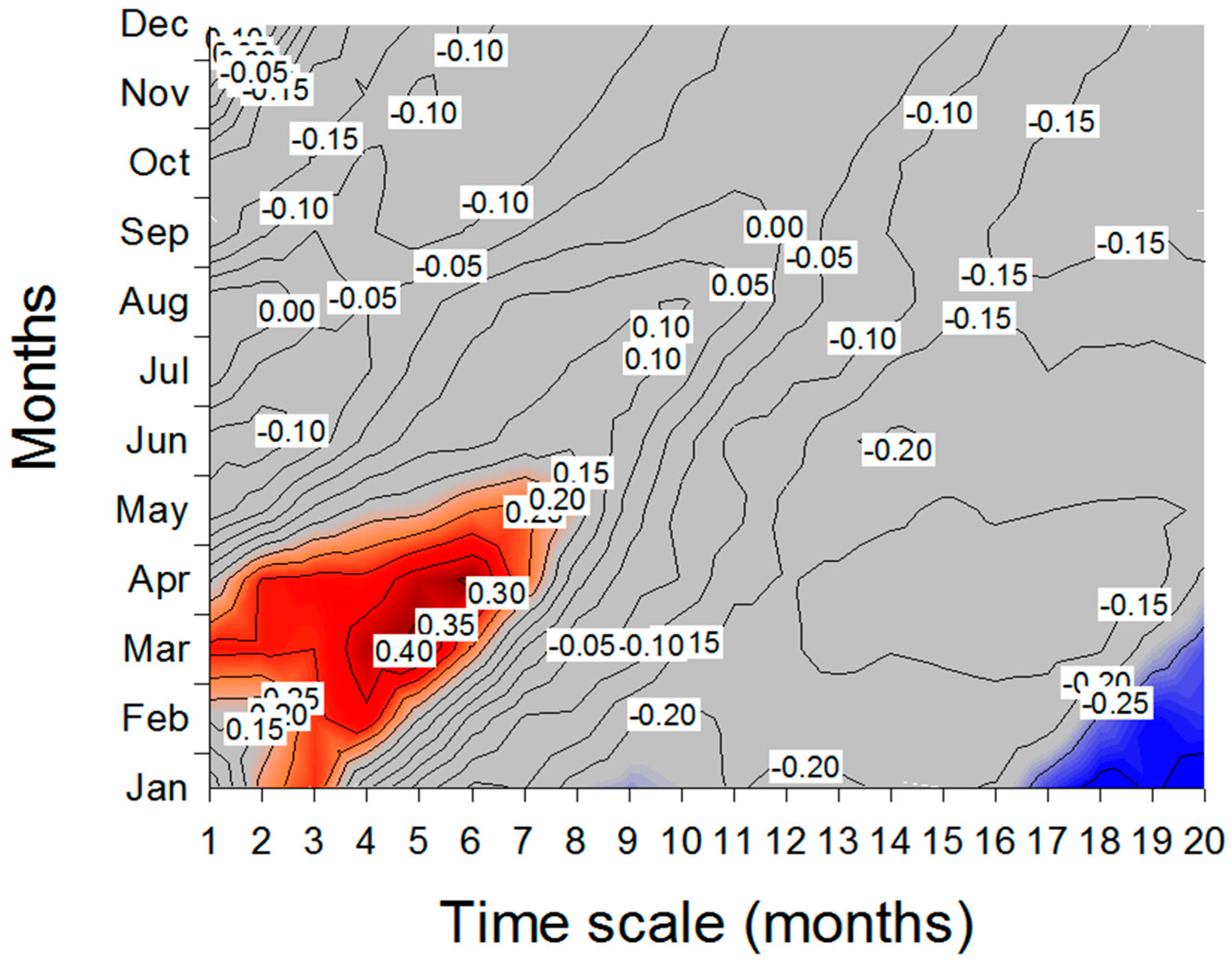
3. Results
3.1. Statistics of Chir Pine Site Chronology
3.2. Climate- and Drought-Growth Relationships
4. Discussion
4.1. Chir Pine Tree-Ring Chronology: Locally Missing Rings and Drought
4.2. Climate- and Drought-Growth Associations: The Pivotal Role of Winter to Spring Conditions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- IPCC. Climate Change 2014—Impacts, Adaptation and Vulnerability: Regional Aspects; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Shrestha, A.B.; Bajracharya, S.R.; Sharma, A.R.; Duo, C.; Kulkarni, A. Observed trends and changes in daily temperature and precipitation extremes over the Koshi river basin 1975–2010. Int. J. Clim. 2017, 37, 1066–1083. [Google Scholar] [CrossRef]
- Yao, T.; Thompson, L.; Yang, W.; Yu, W.; Gao, Y.; Guo, X.; Yang, X.; Duan, K.; Zhao, H.; Xu, B.; et al. Different glacier status with atmospheric circulations in Tibetan Plateau and surroundings. Nat. Clim. Chang. 2012, 2, 663–667. [Google Scholar] [CrossRef]
- Cook, E.R.; Krusic, P.J.; Jones, P.D. Dendroclimatic signals in long tree-ring chronologies from the Himalayas of Nepal. Int. J. Clim. 2003, 23, 707–732. [Google Scholar] [CrossRef]
- Dawadi, B.; Liang, E.; Tian, L.; Devkota, L.P.; Yao, T. Pre-monsoon precipitation signal in tree rings of timberline Betula utilis in the central Himalayas. Quat. Int. 2013, 283, 72–77. [Google Scholar] [CrossRef]
- Liang, E.; Dawadi, B.; Pederson, N.; Eckstein, D. Is the growth of birch at the upper timberline in the Himalayas limited by moisture or by temperature? Ecology 2014, 95, 2453–2465. [Google Scholar] [CrossRef]
- Thapa, U.K.; Shah, S.K.; Gaire, N.P.; Bhuju, D.R. Spring temperatures in the far-western Nepal Himalaya since AD 1640 reconstructed from Picea smithiana tree-ring widths. Clim. Dyn. 2015, 45, 2069–2081. [Google Scholar] [CrossRef]
- Tiwari, A.; Fan, Z.X.; Jump, A.S.; Li, S.F.; Zhou, Z.K. Gradual expansion of moisture sensitive Abies spectabilis forest in the Trans-Himalayan zone of central Nepal associated with climate change. Dendrochronologia 2017, 41, 34–43. [Google Scholar] [CrossRef]
- Gaire, N.P.; Bhuju, D.R.; Koirala, M.; Shah, S.K.; Carrer, M.; Timilsena, R. Tree-ring based spring precipitation reconstruction in western Nepal Himalaya since AD 1840. Dendrochronologia 2017, 42, 21–30. [Google Scholar] [CrossRef]
- Panthi, S.; Bräuning, A.; Zhou, Z.K.; Fan, Z.X. Tree rings reveal recent intensified spring drought in the central Himalaya, Nepal. Glob. Planet. Chang. 2017, 157, 26–34. [Google Scholar] [CrossRef]
- Sohar, K.; Altman, J.; Lehečková, E.; Doležal, J. Growth-climate relationships of Himalayan conifers along elevational and latitudinal gradients. Int. J. Clim. 2017, 37, 2593–2605. [Google Scholar] [CrossRef]
- Yadav, R.R.; Misra, K.G.; Kotlia, B.S.; Upreti, N. Premonsoon precipitation variability in Kumaon Himalaya, India over a perspective of ~300 years. Quat. Int. 2014, 325, 213–219. [Google Scholar] [CrossRef]
- Bräuning, A. Tree-ring studies in the Dolpo-Himalaya (western Nepal). Tree Rings Archaeol. Climatol. Ecol. 2004, 2, 8–12. [Google Scholar]
- Bhattacharyya, A.; Chaudhary, V. Late-summer temperature reconstruction of the eastern Himalayan region based on tree-ring data of Abies densa. Arct. Antarct. Alp. Res. 2003, 35, 196–202. [Google Scholar] [CrossRef]
- Yadav, R.R.; Singh, J. Tree-ring analysis of Taxus baccata from the western Himalaya, India, and its dendroclimatic potential. Tree-Ring Res. 2002, 58, 23–29. [Google Scholar]
- Sano, M.; Ramesh, R.; Sheshshayee, M.; Sukumar, R. Increasing aridity over the past 223 years in the Nepal Himalaya inferred from a tree-ring δ18O chronology. Holocene 2012, 22, 809–817. [Google Scholar] [CrossRef]
- Shah, S.K.; Mehrotra, N. Tree-ring studies of Toona ciliata from subtropical wet hill forests of Kalimpong, eastern Himalaya. Dendrochronologia 2017, 46, 46–55. [Google Scholar] [CrossRef]
- Singh, N.D.; Venugopal, N. Cambial activity and annual rhythm of xylem production of Pinus kesiya royle ex. Gordon (pinaceae) in relation to phenology and climatic factors growing in sub-tropical wet forest of north east India. Flora 2011, 206, 198–204. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Shah, S.K. Tree-ring studies in India past appraisal, present status and future prospects. IAWA 2009, 30, 361–370. [Google Scholar] [CrossRef]
- DFRS. State of Nepal’s Forests; Ministry of Forests and Soil Conservation, Department of Forest Research and Survey: Kathmandu, Nepal, 2015.
- Bhattacharyya, A.; LaMarche, V.C., Jr.; Hughes, M.K. Tree-ring chronologies from Nepal. Tree Ring Bull. 1992, 52, 59–66. [Google Scholar]
- Aryal, S.; Bhuju, D.R.; Kharal, D.K.; Gaire, N.P.; Dyola, N. Climatic upshot using growth pattern of Pinus roxburghii from western Nepal. Pak. J. Bot. 2018, 50, 579–588. [Google Scholar]
- Malla, S.B.; Rajbhandari, S.; Shrestha, T.; Adhikari, P.; Adhikari, S.; Shakya, P. Flora of Kathmandu Valley; Ministry of Forests and Soil Conservation-Department of Medicinal Plants: Kathmandu, Nepal, 1986; 963p. Available online: http://lib.icimod.org/record/2906 (accessed on 8 May 2018).
- Wang, S.Y.; Yoon, J.H.; Gillies, R.R.; Cho, C. What caused the winter drought in western Nepal during recent years? J. Clim. 2013, 26, 8241–8256. [Google Scholar] [CrossRef]
- Cook, E.R.; Kairiukstis, L.A. Methods of Dendrochronology: Applications in the Environmental Sciences; Kluwer: Dordrecht, The Netherlands, 1990. [Google Scholar]
- Holmes, R.L. Computer-assisted quality control in tree-ring dating and measurement. Tree Ring Bull. 1983, 43, 69–78. [Google Scholar]
- Cook, E.R. A Time Series Analysis Approach to Tree Ring Standardization. Ph.D. Thesis, University of Arizona, Arizona, AZ, USA, 1985. [Google Scholar]
- Wigley, T.M.; Briffa, K.R.; Jones, P.D. On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J. Clim. Appl. Meteorol. 1984, 23, 201–213. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I. A multi-scalar drought index sensitive to global warming: The Standardized Precipitation Evapotranspiration Index-SPEI. J. Clim. 2010, 23, 1696–1718. [Google Scholar] [CrossRef]
- Ram, S. Tree growth-climate relationships of conifer trees and reconstruction of summer season palmer drought severity index (PDSI) at Pahalgam in Srinagar, India. Quat. Int. 2012, 254, 152–158. [Google Scholar] [CrossRef]
- Liang, E.; Liu, W.; Ren, P.; Dawadi, B.; Eckstein, D. The alpine dwarf shrub Cassiope fastigiata in the Himalayas: Does it reflect site-specific climatic signals in its annual growth rings? Trees 2015, 29, 79–86. [Google Scholar] [CrossRef]
- Shah, S.K.; Shekhar, M.; Bhattacharyya, A. Anomalous distribution of Cedrus deodara and Pinus roxburghii in Parbati valley, Kullu, western Himalaya: An assessment in dendroecological perspective. Quat. Int. 2014, 325, 205–212. [Google Scholar] [CrossRef]
- Ram, S. Tree ring width variations over western Himalaya in India and its linkage with heat and aridity indices. Nat. Hazards 2018, 92, 635–645. [Google Scholar] [CrossRef]
- Li, J.; Shi, J.; Zhang, D.D.; Yang, B.; Fang, K.; Yue, P.H. Moisture increase in response to high-altitude warming evidenced by tree-rings on the southeastern Tibetan Plateau. Clim. Dyn. 2017, 48, 649–660. [Google Scholar] [CrossRef]
- Liang, E.; Lu, X.; Ren, P.; Li, X.; Zhu, L.; Eckstein, D. Annual increments of juniper dwarf shrubs above the tree line on the central Tibetan Plateau: A useful climatic proxy. Ann. Bot. 2012, 109, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.H.; Piao, S.; Zhao, H.; Jeong, S.J.; Wang, X.; Vitasse, Y.; Ciais, P.; Janssens, I.A. Unexpected role of winter precipitation in determining heat requirement for spring vegetation green-up at northern middle and high latitudes. Glob. Chang. Biol. 2014, 20, 3743–3755. [Google Scholar] [CrossRef] [PubMed]
- Cleaveland, M.K.; Stahle, D.W.; Therrell, M.D.; Villanueva-Diaz, J.; Burns, B.T. Tree-ring reconstructed winter precipitation and tropical teleconnections in Durango, Mexico. Clim. Chang. 2003, 59, 369–388. [Google Scholar] [CrossRef]
- Sarris, D.; Christodoulakis, D.; Koerner, C. Recent decline in precipitation and tree growth in the eastern Mediterranean. Glob. Chang. Biol. 2007, 13, 1187–1200. [Google Scholar] [CrossRef]
- Bräuning, A. Tree-ring evidence of ‘Little Ice Age’ glacier advances in southern Tibet. Holocene 2006, 16, 369–380. [Google Scholar] [CrossRef]
- Voltas, J.; Camarero, J.J.; Carulla, D.; Aguilera, M.; Ortiz, A.; Ferrio, J.P. A retrospective, dual-isotope approach reveals individual predispositions to winter-drought induced tree dieback in the southernmost distribution limit of Scots Pine. Plant Cell Environ. 2013, 36, 1435–1448. [Google Scholar] [CrossRef] [PubMed]
- Savva, Y.; Oleksyn, J.; Reich, P.B.; Tjoelker, M.G.; Vaganov, E.A.; Modrzynski, J. Interannual growth response of Norway spruce to climate along an altitudinal gradient in the Tatra Mountains, Poland. Trees 2006, 20, 735–746. [Google Scholar] [CrossRef]
- Deslauriers, A.; Rossi, S.; Anfodillo, T.; Saracino, A. Cambial phenology, wood formation and temperature thresholds in two contrasting years at high altitude in southern Italy. Tree Physiol. 2008, 28, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.R.; Park, W.K.; Singh, J.; Dubey, B. Do the western Himalayas defy global warming? Geophys. Res. Lett. 2004, 31, L17201. [Google Scholar] [CrossRef]
- Kobe, R.K.; Coates, K.D. Models of sapling mortality as a function of growth to characterize interspecific variation in shade tolerance of eight tree species of northwestern British Columbia. Can. J. For. Res. 1997, 27, 227–236. [Google Scholar] [CrossRef]
- Rollinson, C.R.; Kaye, M.W.; Canham, C.D. Interspecific variation in growth responses to climate and competition of five eastern tree species. Ecology 2016, 97, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Sigdel, M.; Ikeda, M. Spatial and temporal analysis of drought in Nepal using standardized precipitation index and its relationship with climate indices. J. Hydrol. Meteorol. 2010, 7, 59–74. [Google Scholar] [CrossRef]
- Gleason, K.E.; Bradford, J.B.; Bottero, A.; D’Amato, A.W.; Fraver, S.; Palik, B.J.; Battaglia, M.A.; Iverson, L.; Kenefic, L.; Kern, C.C. Competition amplifies drought stress in forests across broad climatic and compositional gradients. Ecosphere 2017, 8, e01849. [Google Scholar] [CrossRef]
- Martin-Benito, D.; Kint, V.; del Río, M.; Muys, B.; Cañellas, I. Growth responses of west-mediterranean pinus nigra to climate change are modulated by competition and productivity: Past trends and future perspectives. For. Ecol. Manag. 2011, 262, 1030–1040. [Google Scholar] [CrossRef]
- Linares, J.-C.; Delgado-Huertas, A.; Camarero, J.J.; Merino, J.; Carreira, J.A. Competition and drought limit the response of water-use efficiency to rising atmospheric carbon dioxide in the Mediterranean fir Abies pinsapo. Oecologia 2009, 161, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Liang, E.; Leuschner, C.; Dulamsuren, C.; Wagner, B.; Hauck, M. Global warming-related tree growth decline and mortality on the north-eastern Tibetan plateau. Clim. Chang. 2016, 134, 163–176. [Google Scholar] [CrossRef]
- Ren, P.; Rossi, S.; Camarero, J.J.; Ellison, A.M.; Liang, E.; Peñuelas, J. Critical temperature and precipitation thresholds for the onset of xylogenesis of Juniperus przewalskii in a semi-arid area of the north-eastern Tibetan Plateau. Ann. Bot. 2018, 121, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Zhang, Q.B.; Evans, M.N.; Lyu, L. Moisture dipole over the Tibetan Plateau during the past five and a half centuries. Nat. Commun. 2015, 6, 8062. [Google Scholar] [CrossRef] [PubMed]
- Bayramzadeh, V.; Zhu, H.; Lu, X.; Attarod, P.; Zhang, H.; Li, X.; Asad, F.; Liang, E. Temperature variability in northern Iran during the past 700 years. Sci. Bull. 2018, 63, 462–464. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Galván, J.D.; Sangüesa-Barreda, G.; Gutiérrez, E. Disparate effects of global-change drivers on mountain conifer forests: Warming-induced growth enhancement in young trees vs. CO2 fertilization in old trees from wet sites. Glob. Chang. Biol. 2015, 21, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Liang, E.; Liu, K.; Camarero, J.J. Species- and elevation-dependent growth responses to climate warming of mountain forests in the Qinling Mountains, central China. Forests 2018, 9, 248. [Google Scholar] [CrossRef]
| Variable (Units) | Values |
|---|---|
| No. trees/No. cores | 39/49 |
| Mean tree-ring width (mm) | 0.97 |
| AC1, first-order autocorrelation | 0.59 |
| Locally missing rings (%) | 0.71 |
| MS, mean sensitivity | 0.36 |
| RBAR, mean series inter-correlation | 0.45 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sigdel, S.R.; Dawadi, B.; Camarero, J.J.; Liang, E.; Leavitt, S.W. Moisture-Limited Tree Growth for a Subtropical Himalayan Conifer Forest in Western Nepal. Forests 2018, 9, 340. https://doi.org/10.3390/f9060340
Sigdel SR, Dawadi B, Camarero JJ, Liang E, Leavitt SW. Moisture-Limited Tree Growth for a Subtropical Himalayan Conifer Forest in Western Nepal. Forests. 2018; 9(6):340. https://doi.org/10.3390/f9060340
Chicago/Turabian StyleSigdel, Shalik Ram, Binod Dawadi, J. Julio Camarero, Eryuan Liang, and Steven W. Leavitt. 2018. "Moisture-Limited Tree Growth for a Subtropical Himalayan Conifer Forest in Western Nepal" Forests 9, no. 6: 340. https://doi.org/10.3390/f9060340
APA StyleSigdel, S. R., Dawadi, B., Camarero, J. J., Liang, E., & Leavitt, S. W. (2018). Moisture-Limited Tree Growth for a Subtropical Himalayan Conifer Forest in Western Nepal. Forests, 9(6), 340. https://doi.org/10.3390/f9060340







