Differences in Leaf Temperature between Lianas and Trees in the Neotropical Canopy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Species Selection and Field Design
2.3. Estimation of the Leaf Temperature
2.4. Estimations of Photosynthesis and Leaf Respiration
2.5. Data Analysis
3. Results
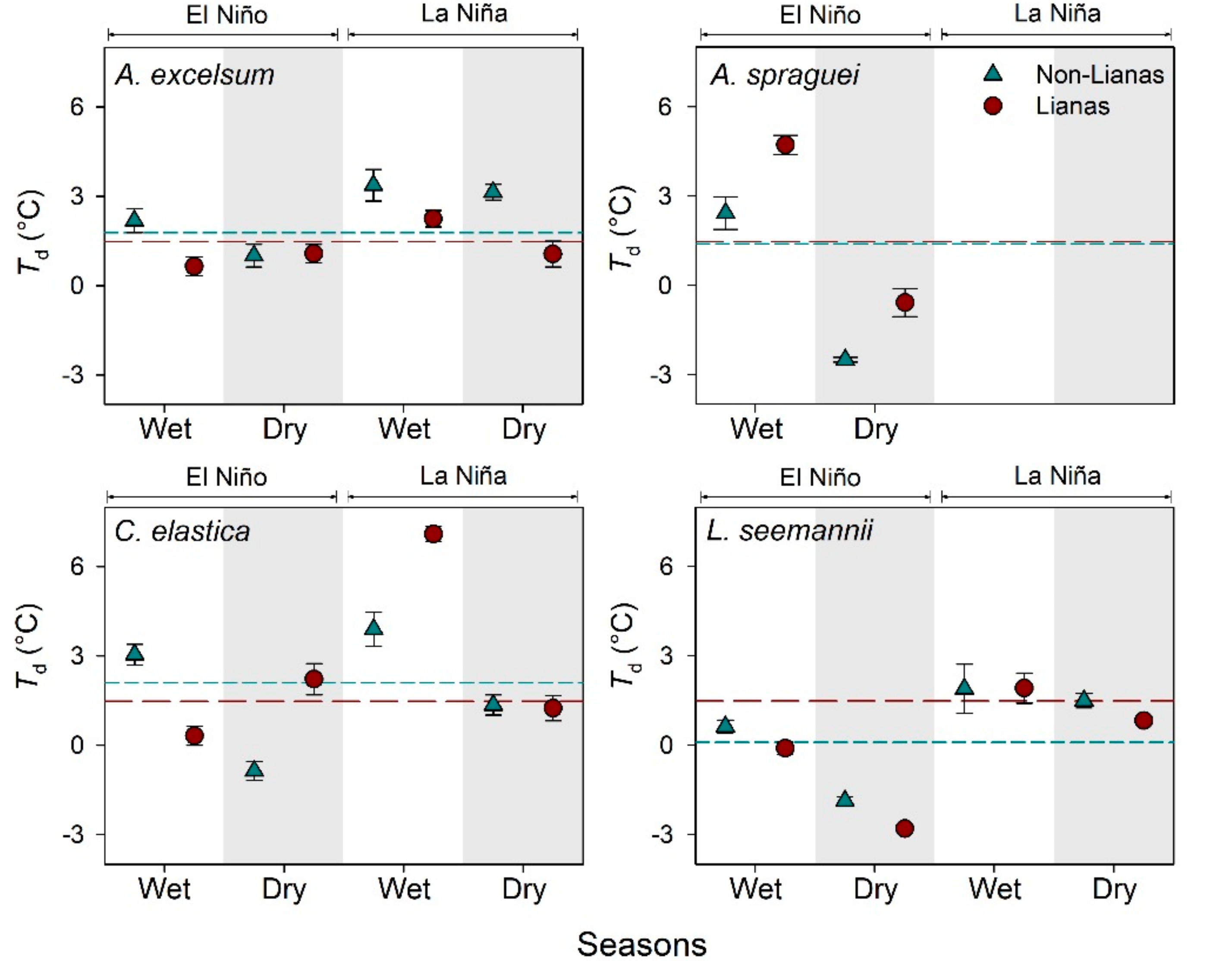
3.1. Leaf Temperature of Trees with and without Lianas
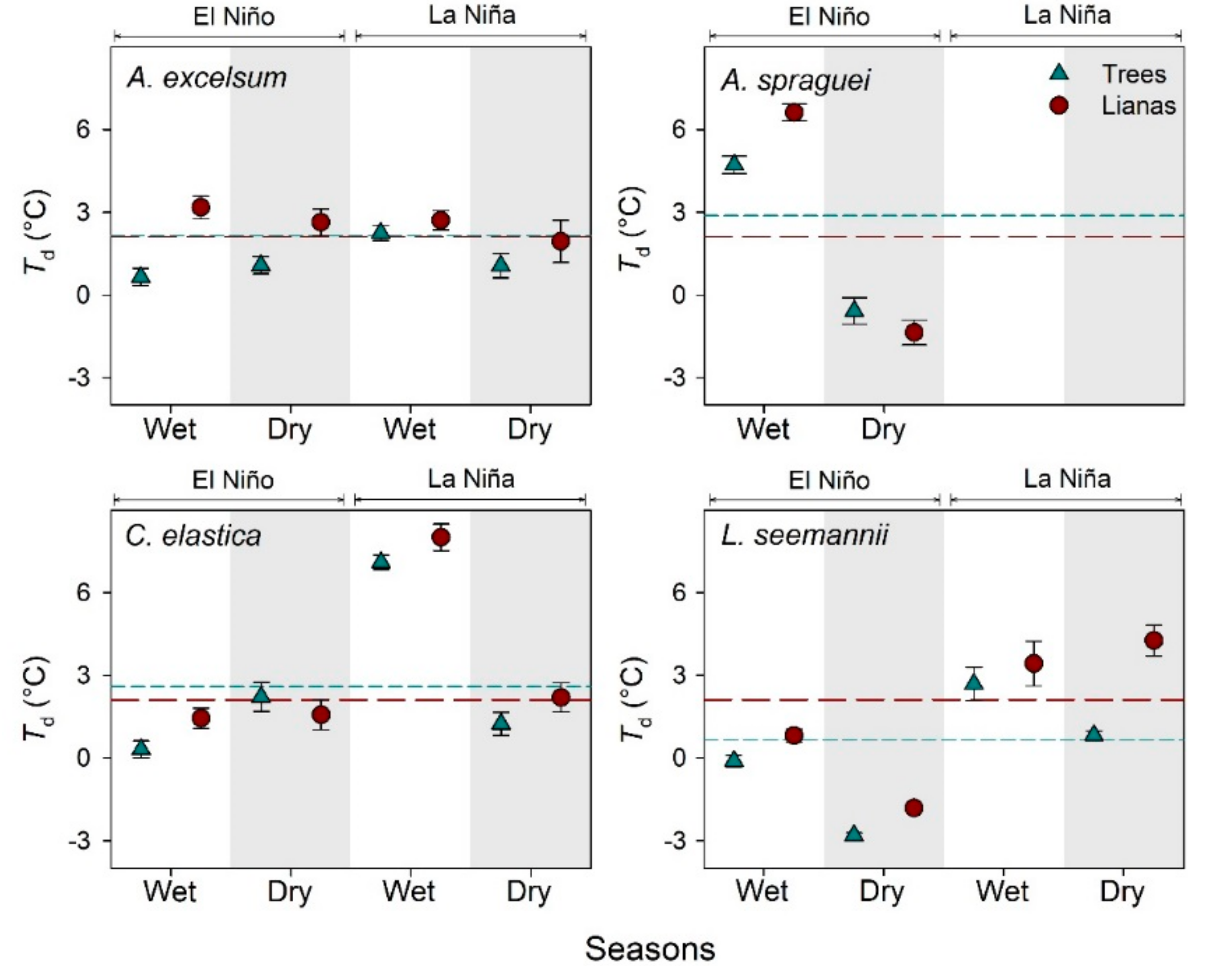
3.2. Leaf Temperature of Lianas and Their Host Trees
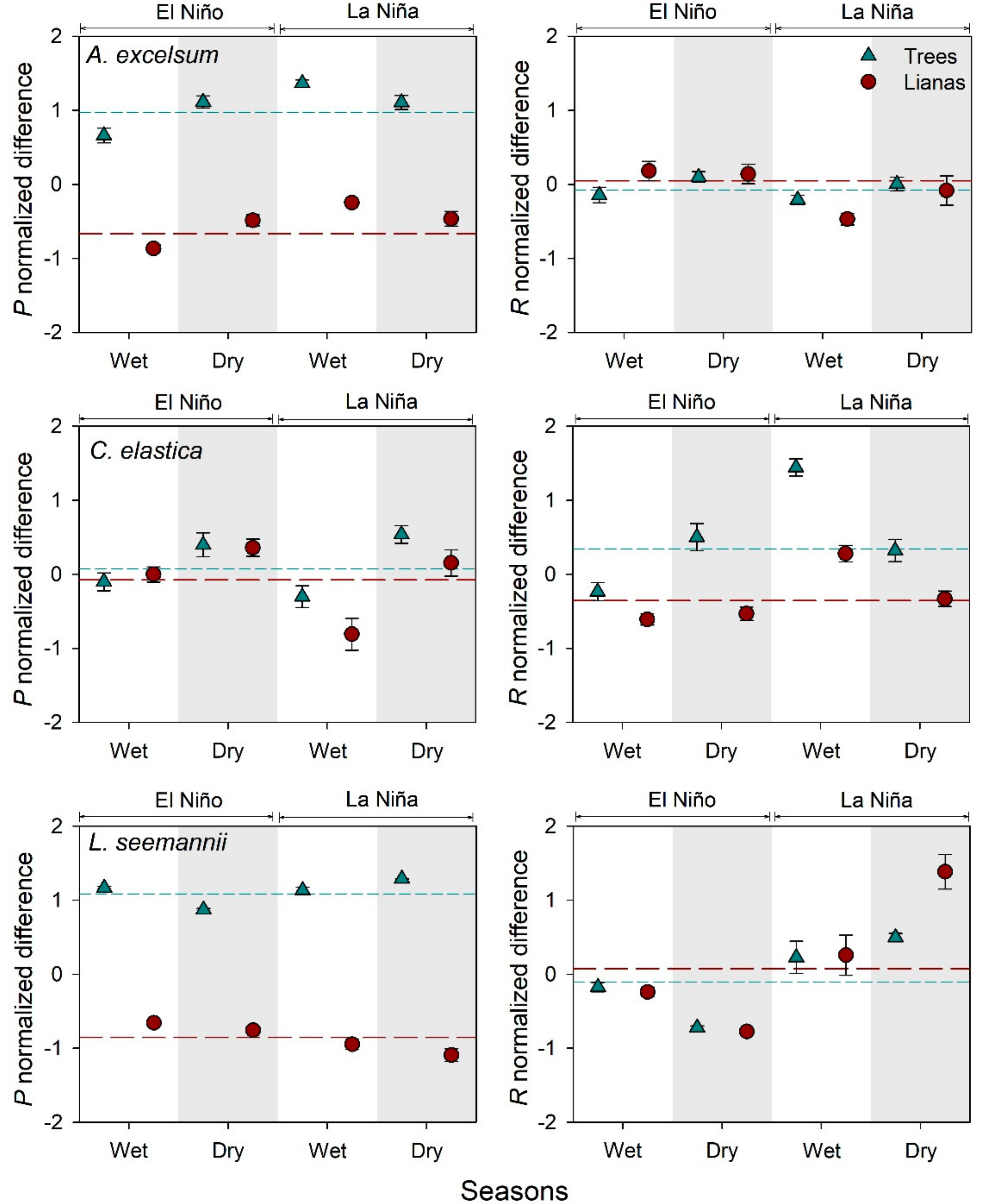
3.3. Comparisons of Photosynthesis and Leaf Respiration between Life Forms
4. Discussion
4.1. Leaf Temperature of Trees with and without Lianas
4.2. Leaf Temperature of Trees and Lianas
4.3. Effect of the Temperature on Photosynthesis and Respiration of Lianas And Trees
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rogers, A.; Medlyn, B.E.; Dukes, J.S.; Bonan, G.; von Caemmerer, S.; Dietze, M.C.; Kattge, J.; Leakey, A.D.B.; Mercado, L.M.; Niinemets, Ü.; et al. A roadmap for improving the representation of photosynthesis in Earth system models. New Phytol. 2017, 213, 22–42. [Google Scholar] [CrossRef] [PubMed]
- Leuzinger, S.; Körner, C. Tree species diversity affects canopy leaf temperatures in a mature temperate forest. Agric. For. Meteorol. 2007, 146, 29–37. [Google Scholar] [CrossRef]
- Jones, H.G. Use of thermography for quantitative studies of spatial and temporal variation of stomatal conductance over leaf surfaces. Plant Cell Environ. 1999, 22, 1043–1055. [Google Scholar] [CrossRef]
- Dai, Y.; Dickinson, R.E.; Wang, Y.-P. A two-big-leaf model for canopy temperature, photosynthesis, and stomatal conductance. J. Clim. 2004, 17, 2281–2299. [Google Scholar] [CrossRef]
- Meinzer, F.C.; Andrade, J.L.; Goldstein, G.; Holbrook, N.M.; Cavelier, J.; Jackson, P. Control of transpiration from the upper canopy of a tropical forest: The role of stomatal, boundary layer and hydraulic architecture components. Plant Cell Environ. 1997, 20, 1242–1252. [Google Scholar] [CrossRef]
- Vargas, G.; Cordero, R.A. Photosynthetic responses to temperature of two tropical rainforest tree species from Costa Rica. Trees 2013, 27, 1261–1270. [Google Scholar] [CrossRef]
- Slot, M.; Winter, K. In situ temperature response of photosynthesis of 42 tree and liana species in the canopy of two Panamanian lowland tropical forests with contrasting rainfall regimes. New Phytol. 2017, 214, 1103–1117. [Google Scholar] [CrossRef] [PubMed]
- Slot, M.; Garcia, M.N.; Winter, K. Temperature response of CO2 exchange in three tropical tree species. Funct. Plant Biol. 2016, 43, 468–478. [Google Scholar] [CrossRef]
- Slot, M.; Wright, S.J.; Kitajima, K. Foliar respiration and its temperature sensitivity in trees and lianas: In situ measurements in the upper canopy of a tropical forest. Tree Physiol. 2013, 33, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Slot, M.; Rey-Sánchez, C.; Winter, K.; Kitajima, K. Trait-based scaling of temperature-dependent foliar respiration in a species-rich tropical forest canopy. Funct. Ecol. 2014, 28, 1074–1086. [Google Scholar] [CrossRef]
- Lin, D.; Xia, J.; Wan, S. Climate warming and biomass accumulation of terrestrial plants: A meta-analysis. New Phytol. 2010, 188, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Durán, S.M.; Sánchez-Azofeifa, G.A.; Rios, R.S.; Gianoli, E. The relative importance of climate, stand variables and liana abundance for carbon storage in tropical forests. Glob. Ecol. Biogeogr. 2015, 24, 939–949. [Google Scholar] [CrossRef]
- DeWalt, S.J.; Chave, J. Structure and biomass of four lowland Neotropical forests. Biotropica 2004, 36, 7–19. [Google Scholar] [CrossRef]
- Londré, R.; Schnitzer, S. The distribution of lianas and their change in abundance in temperate forests over the past 45 years. Ecology 2006, 87, 2973–2978. [Google Scholar] [CrossRef]
- Phillips, O.L.; Vasquez Martinez, R.; Arroyo, L.; Baker, T.R.; Killeen, T.; Lewis, S.L.; Malhi, Y.; Monteagudo Mendoza, A.; Neill, D.; Nunez Vargas, P.; et al. Increasing dominance of large lianas in Amazonian forests. Nature 2002, 418, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, S. A mechanistic explanation for global patterns of liana abundance and distribution. Am. Nat. 2005, 166, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, S. Increasing liana abundance in neotropical forests: Causes and consequences. In Ecology of Lianas; John Wiley & Sons, Ltd.: New York, NY, USA, 2015; pp. 451–464. ISBN 978-1-11-839240-9. [Google Scholar]
- Martínez-Izquierdo, L.; García, M.M.; Powers, J.S.; Schnitzer, S.A. Lianas suppress seedling growth and survival of 14 tree species in a Panamanian tropical forest. Ecology 2016, 97, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, S.A.; Carson, W.P. Lianas suppress tree regeneration and diversity in treefall gaps. Ecol. Lett. 2010, 13, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Durán, S.M.; Gianoli, E. Carbon stocks in tropical forests decrease with liana density. Biol. Lett. 2013, 9, 20130301. [Google Scholar] [CrossRef] [PubMed]
- Verbeeck, H.; Kearsley, E. The importance of including lianas in global vegetation models. Proc. Natl. Acad. Sci. USA 2016, 113, E4. [Google Scholar] [CrossRef] [PubMed]
- Slot, M.; Rey-Sánchez, C.; Gerber, S.; Lichstein, J.W.; Winter, K.; Kitajima, K. Thermal acclimation of leaf respiration of tropical trees and lianas: Response to experimental canopy warming, and consequences for tropical forest carbon balance. Glob. Chang. Biol. 2014, 20, 2915–2926. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ronderos, M.E.; Bohrer, G.; Sanchez-Azofeifa, A.; Powers, J.S.; Schnitzer, S.A. Contribution of lianas to plant area index and canopy structure in a Panamanian forest. Ecology 2016, 97, 3271–3277. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-J.; Cao, K.-F.; Schnitzer, S.A.; Fan, Z.-X.; Zhang, J.-L.; Bongers, F. Water-use advantage for lianas over trees in tropical seasonal forests. New Phytol. 2015, 205, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Ichihashi, R.; Chiu, C.-W.; Komatsu, H.; Kume, T.; Shinohara, Y.; Tateishi, M.; Tsuruta, K.; Otsuki, K. Contribution of lianas to community-level canopy transpiration in a warm-temperate forest. Funct. Ecol. 2017, 31, 1690–1699. [Google Scholar] [CrossRef]
- Lin, H.; Chen, Y.; Zhang, H.; Fu, P.; Fan, Z. Stronger cooling effects of transpiration and leaf physical traits of plants from a hot dry habitat than from a hot wet habitat. Funct. Ecol. 2017. [Google Scholar] [CrossRef]
- De Guzman, M.E.; Santiago, L.S.; Schnitzer, S.A.; Álvarez-Cansino, L. Trade-offs between water transport capacity and drought resistance in neotropical canopy liana and tree species. Tree Physiol. 2016, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.-Q.; Schnitzer, S.A.; Bongers, F. Seasonal differences in leaf-level physiology give lianas a competitive advantage over trees in a tropical seasonal forest. Oecologia 2009, 161, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.-D.; Cao, K.-F. Hydraulic properties and photosynthetic rates in co-occurring lianas and trees in a seasonal tropical rainforest in southwestern China. Plant Ecol. 2009, 204, 295–304. [Google Scholar] [CrossRef]
- Bretfeld, M.; Ewers, B.E.; Hall, J.S. Plant water use responses along secondary forest succession during the 2015–2016 El Niño drought in Panama. New Phytol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, S.A.; van der Heijden, G.M.F.; Powers, J.S. Reply to Verbeeck and Kearsley: Addressing the challenges of including lianas in global vegetation models. Proc. Natl. Acad. Sci. USA 2016, 113, E5–E6. [Google Scholar] [CrossRef] [PubMed]
- Avalos, G.; Mulkey, S.S. Seasonal changes in liana cover in the upper canopy of a neotropical dry forest. Biotropica 1999, 31, 186–192. [Google Scholar] [CrossRef]
- Usamentiaga, R.; Venegas, P.; Guerediaga, J.; Vega, L.; Molleda, J.; Bulnes, F.G. Infrared thermography for temperature measurement and non-destructive testing. Sensors 2014, 14, 12305–12348. [Google Scholar] [CrossRef] [PubMed]
- López, A.; Molina-Aiz, F.D.; Valera, D.L.; Peña, A. Determining the emissivity of the leaves of nine horticultural crops by means of infrared thermography. Sci. Hortic. (Amsterdam) 2012, 137, 49–58. [Google Scholar] [CrossRef]
- Zuur, A.F.; Ieno, E.N.; Walker, N.J.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer Science and Business Media: New York, NY, USA, 2009. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. R Development Core Team nlme: Linear and Nonlinear Mixed Effects Models. Available online: http://cran.r-project.org/package=nlme (accessed on 30 May 2018).
- R Development Core Team. R: A language and environment for statistical computing. R Found. Stat. Comput. 2017, 1, 409. [Google Scholar]
- De Deurwaerder, H.; Hervé-Fernández, P.; Stahl, C.; Burban, B.; Petronelli, P.; Hoffman, B.; Bonal, D.; Boeckx, P.; Verbeeck, H. Liana and tree below-ground water competition—evidence for water resource partitioning during the dry season. Tree Physiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.D.; Schnitzer, S.A.; Muller-Landau, H.C.; Jongejans, E.; de Kroon, H.; Comita, L.S.; Hubbell, S.P.; Wright, S.J. Tree species vary widely in their tolerance for liana infestation: A case study of differential host response to generalist parasites. J. Ecol. 2018, 106, 781–794. [Google Scholar] [CrossRef]
- Sánchez-Azofeifa, G.A.; Rankine, C.; do Espirito Santo, M.M.; Fatland, R.; Garcia, M. Wireless sensing networks for environmental monitoring: Two case studies from tropical forests. In Proceedings of the IEEE Seventh International Conference on e-Science, Stockholm, Sweden, 5–8 December 2011; pp. 70–76. [Google Scholar]
- Leigh, A.; Sevanto, S.; Close, J.D.; Nicotra, A.B. The influence of leaf size and shape on leaf thermal dynamics: Does theory hold up under natural conditions? Plant Cell Environ. 2017, 40, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Medina, E.; Sobrado, M.; Herrera, R. Significance of leaf orientation for leaf temperature in an amazonian sclerophyll vegetation. Radiat. Environ. Biophys. 1978, 15, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Estrada, L.B.; Cano-Santana, Z.; Oyama, K. Variation in leaf trichomes of Wigandia urens: Environmental factors and physiological consequences. Tree Physiol. 2000, 20, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Rey-Sánchez, A.C.; Slot, M.; Posada, J.M.; Kitajima, K. Spatial and seasonal variation in leaf temperature within the canopy of a tropical forest. Clim. Res. 2016, 71, 75–89. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Schnitzer, S.A.; Zhang, Y.-J.; Fan, Z.-X.; Goldstein, G.; Tomlinson, K.W.; Lin, H.; Zhang, J.-L.; Cao, K.-F. Physiological regulation and efficient xylem water transport regulate diurnal water and carbon balances of tropical lianas. Funct. Ecol. 2017, 31, 306–317. [Google Scholar] [CrossRef]
- Prytz, G.; Futsaether, C.M.; Johnsson, A. Thermography studies of the spatial and temporal variability in stomatal conductance of Avena leaves during stable and oscillatory transpiration. New Phytol. 2003, 158, 249–258. [Google Scholar] [CrossRef]
- Sánchez-Azofeifa, G.A.; Castro, K.; Wright, S.J.; Gamon, J.; Kalacska, M.; Rivard, B.; Schnitzer, S.A.; Feng, J.L. Differences in leaf traits, leaf internal structure, and spectral reflectance between two communities of lianas and trees: Implications for remote sensing in tropical environments. Remote Sens. Environ. 2009, 113, 2076–2088. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Adams, W.W. Photoprotection and other responses of plants to high light stress. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1992, 43, 599–626. [Google Scholar] [CrossRef]
- Demmig-Adams, B. Survey of thermal energy dissipation and pigment composition in sun and shade leaves. Plant Cell Physiol. 1998, 39, 474–482. [Google Scholar] [CrossRef]
| Factors | Species | |||
|---|---|---|---|---|
| A. excelsum | A. spraguei | C. elastica | L. seemannii | |
| Presence | 0.90 | 2.27 | 0.01 | 1.88 |
| (1, 14) | (1, 13) | (1, 13) | (1, 7) | |
| Season | 0.01 | 9.07 | 2.11 | 2.13 |
| (1, 14) | (1, 13) | (1, 13) | (1, 7) | |
| ENSO | 1.56 | - | 4.47 | 5.37 |
| (1, 14) | (1, 13) | (1, 7) | ||
| Presence*Season | 0.01 | 0.02 | 0.60 | 0.01 |
| (1, 14) | (1, 13) | (1, 13) | (1, 7) | |
| Presence*ENSO | 0.02 | - | 0.09 | 0.01 |
| (1, 14) | (1, 13) | (1, 7) | ||
| Season*ENSO | 0.02 | - | 1.14 | 2.45 |
| (1, 14) | (1, 13) | (1, 7) | ||
| Presence*Season*ENSO | 0.25 | - | 3.55 | 0.12 |
| (1, 14) | (1, 13) | (1, 7) | ||
| Factors | Species | |||
|---|---|---|---|---|
| A. excelsum | A. spraguei | C. elastica | L. seemannii | |
| Type | 22.22 *** | 12.96 *** | 10.04 ** | 192.32 *** |
| (1, 7) | (1, 2) | (1, 6) | (1, 4) | |
| Season | 0.02 | 9.06 | 0.40 | 1.12 |
| (1, 7) | (1, 2) | (1, 6) | (1, 4) | |
| ENSO | 0.15 | - | 2.46 | 1.87 |
| (1, 7) | (1, 6) | (1, 4) | ||
| Type*Season | 13.60 *** | 32.49 *** | 5.92 * | 16.22 *** |
| (1, 7) | (1, 2) | (1, 6) | (1, 4) | |
| Type*ENSO | 0.69 | - | 2.40 | 7.32 ** |
| (1, 7) | (1, 6) | (1, 4) | ||
| Season*ENSO | 0.01 | - | 2.46 | 2.40 |
| (1, 7) | (1, 6) | (1, 4) | ||
| Type*Season*ENSO | 1.09 | - | 11.88 *** | 0.44 |
| (1, 7) | (1, 6) | (1, 4) | ||
| Factors | Species or host tree | |||||
|---|---|---|---|---|---|---|
| Photosynthesis | Respiration | |||||
| A. excelsum | C. elastica | L. seemannii | A. excelsum | C. elastica | L. seemannii | |
| Type | 1133.01 *** | 11.42 *** | 3432.71 *** | 18.45 *** | 420.99 *** | 4.56 * |
| (1, 7) | (1, 6) | (1, 4) | (1, 7) | (1, 6) | (1, 4) | |
| Season | 0.49 | 1.03 | 2.49 | 0.05 | 0.01 | 0.02 |
| (1, 7) | (1, 6) | (1, 4) | (1, 7) | (1, 6) | (1, 4) | |
| ENSO | 0.97 | 0.37 | 3.39 | 0.09 | 1.51 | 0.19 |
| (1, 7) | (1, 6) | (1, 4) | (1, 7) | (1, 6) | (1, 4) | |
| Type*Season | 12.33 *** | 0.19 | 0.03 | 13.85 *** | 5.43 * | 0.15 |
| (1, 7) | (1, 6) | (1, 4) | (1, 7) | (1, 6) | (1, 4) | |
| Type*ENSO | 5.86 * | 3.87 * | 86.23 *** | 0.57 | 0.99 | 39.76 *** |
| (1, 7) | (1, 6) | (1, 4) | (1, 7) | (1, 6) | (1, 4) | |
| Season*ENSO | 1.69 | 0.41 | 0.34 | 0.17 | 1.29 | 2.71 |
| (1, 7) | (1, 6) | (1, 4) | (1, 7) | (1, 6) | (1, 4) | |
| Type*Season*ENSO | 4.97 * | 0.01 | 10.61 ** | 0.87 | 20.99 *** | 0.17 |
| (1, 7) | (1, 6) | (1, 4) | (1, 7) | (1, 6) | (1, 4) | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guzmán Q., J.A.; Sánchez-Azofeifa, G.A.; Rivard, B. Differences in Leaf Temperature between Lianas and Trees in the Neotropical Canopy. Forests 2018, 9, 307. https://doi.org/10.3390/f9060307
Guzmán Q. JA, Sánchez-Azofeifa GA, Rivard B. Differences in Leaf Temperature between Lianas and Trees in the Neotropical Canopy. Forests. 2018; 9(6):307. https://doi.org/10.3390/f9060307
Chicago/Turabian StyleGuzmán Q., J. Antonio, G. Arturo Sánchez-Azofeifa, and Benoit Rivard. 2018. "Differences in Leaf Temperature between Lianas and Trees in the Neotropical Canopy" Forests 9, no. 6: 307. https://doi.org/10.3390/f9060307
APA StyleGuzmán Q., J. A., Sánchez-Azofeifa, G. A., & Rivard, B. (2018). Differences in Leaf Temperature between Lianas and Trees in the Neotropical Canopy. Forests, 9(6), 307. https://doi.org/10.3390/f9060307







