Direct Seeding of Pinus halepensis Mill. for Recovery of Burned Semi-Arid Forests: Implications for Post-Fire Management for Improving Natural Regeneration
Abstract
:1. Introduction
2. Materials and Methods
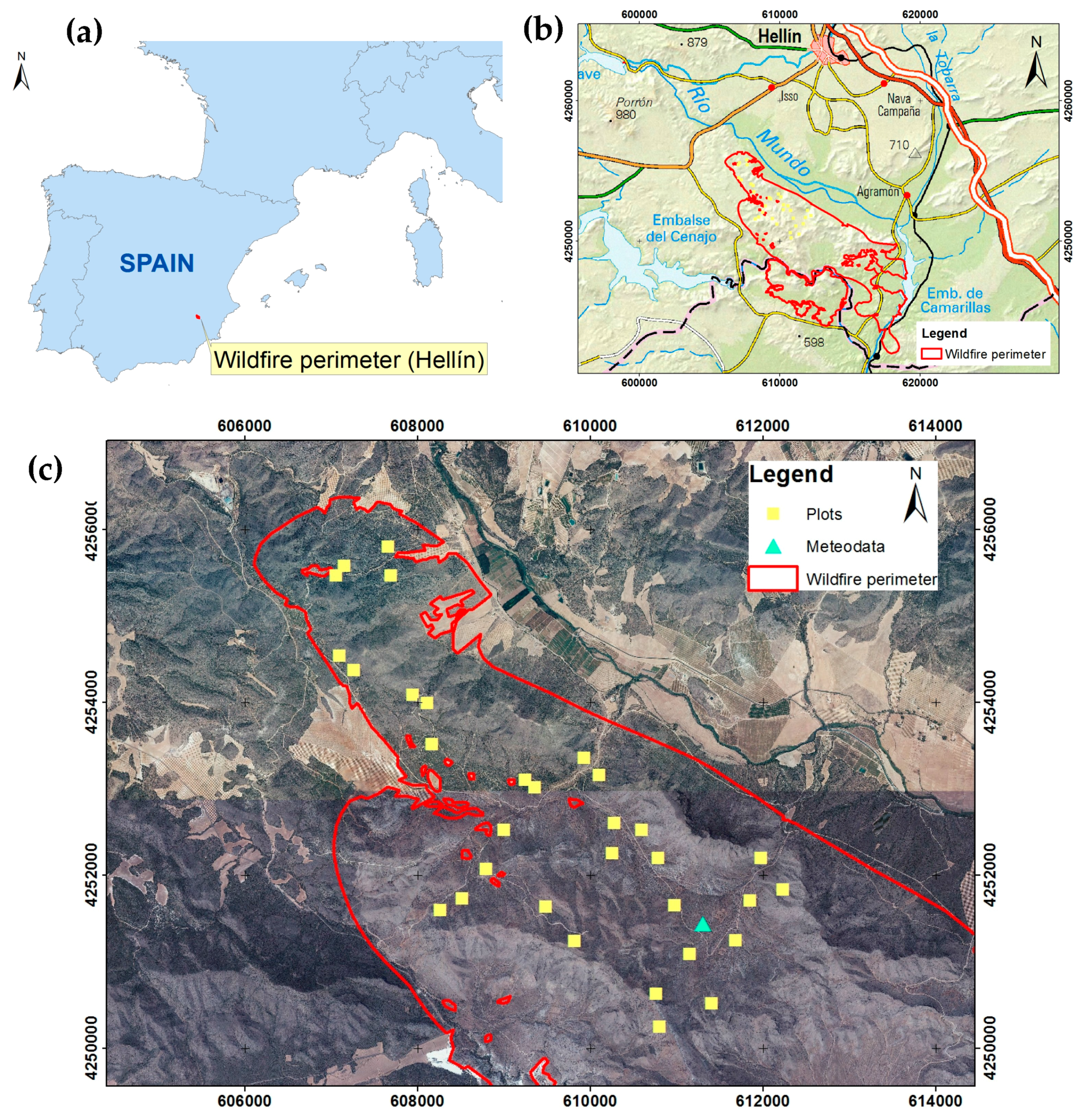
2.1. Study Area
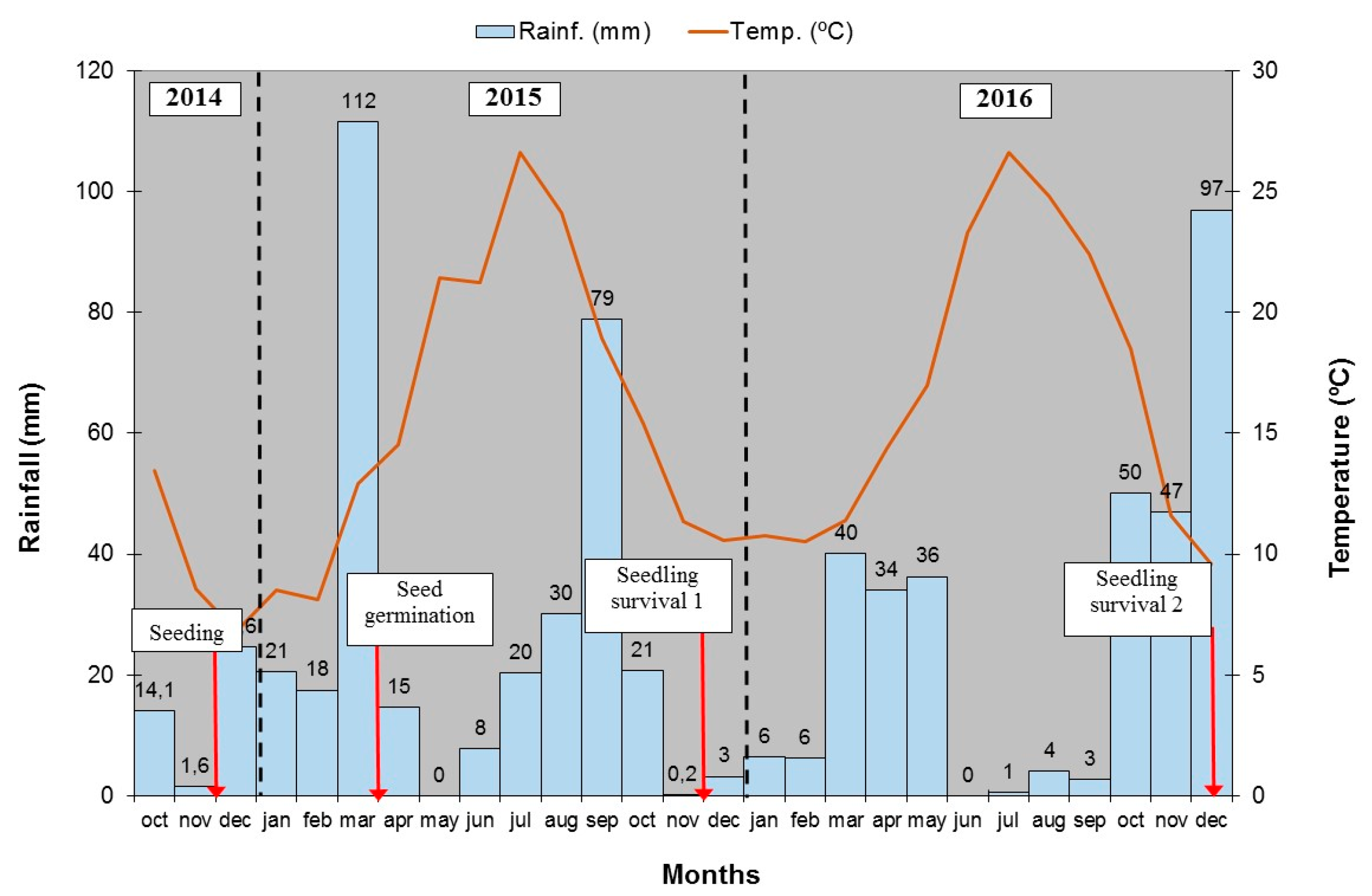
2.2. Experimental Design
2.3. Data Analysis
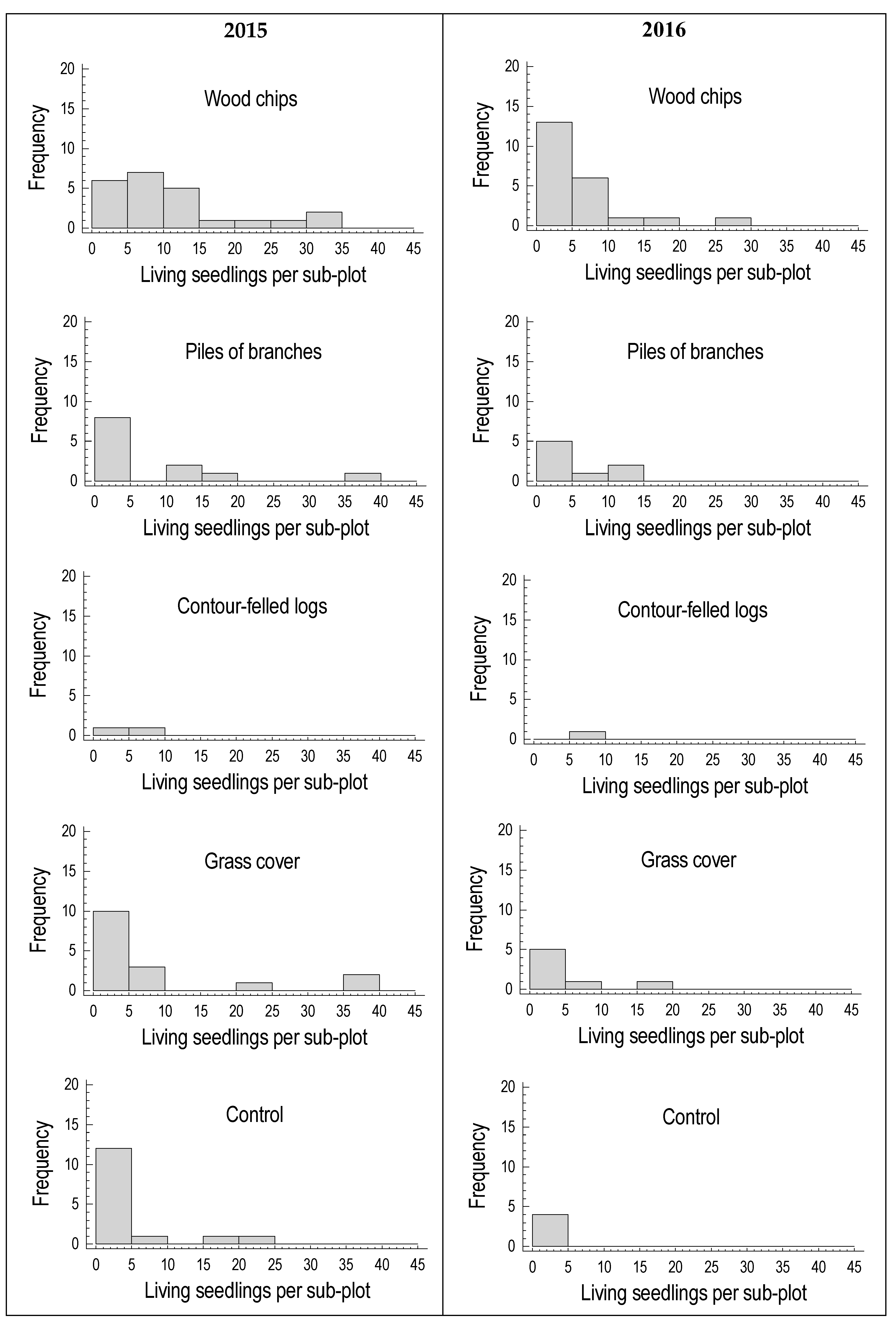
3. Results
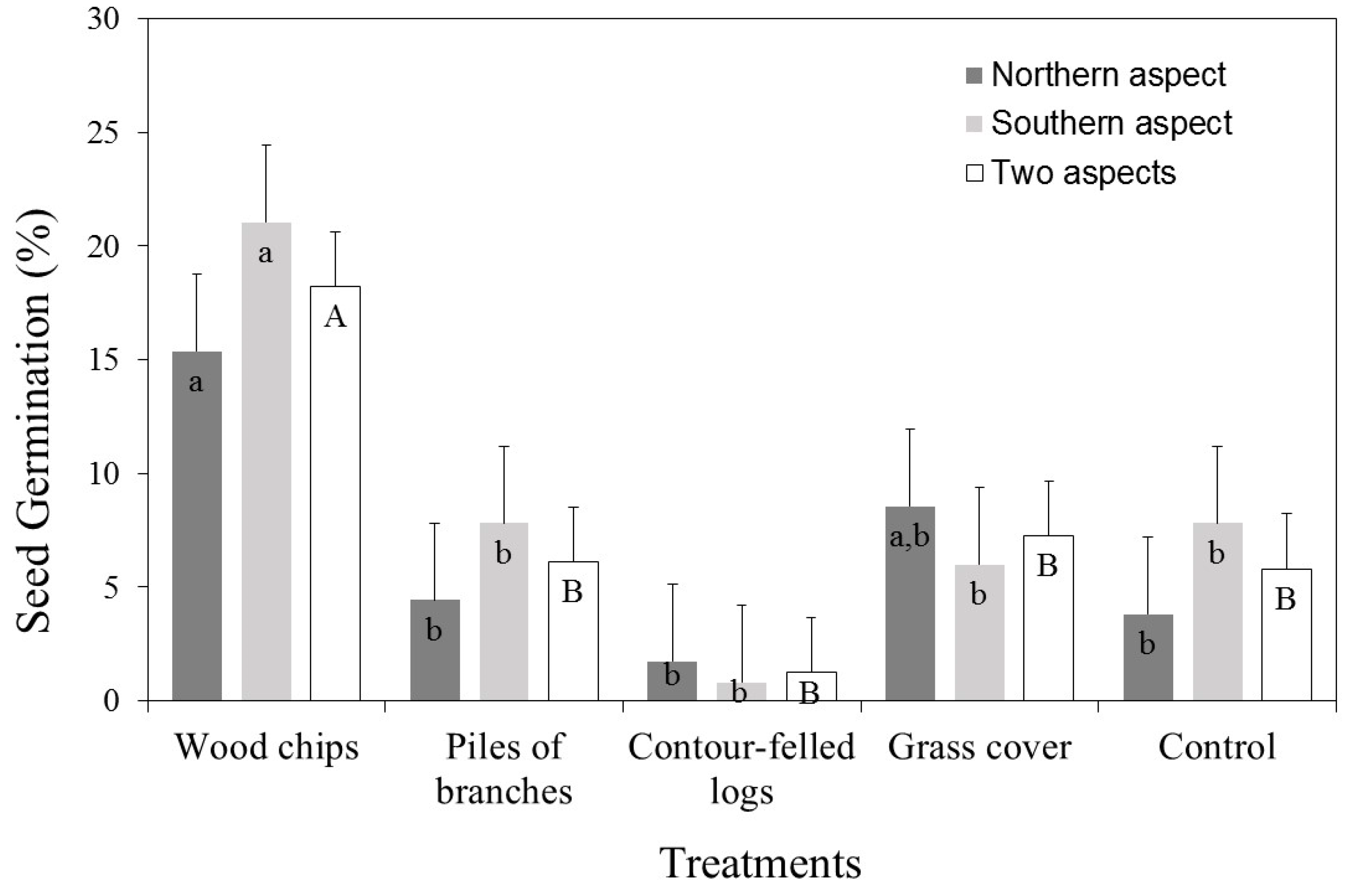
3.1. Seed Germination (%)
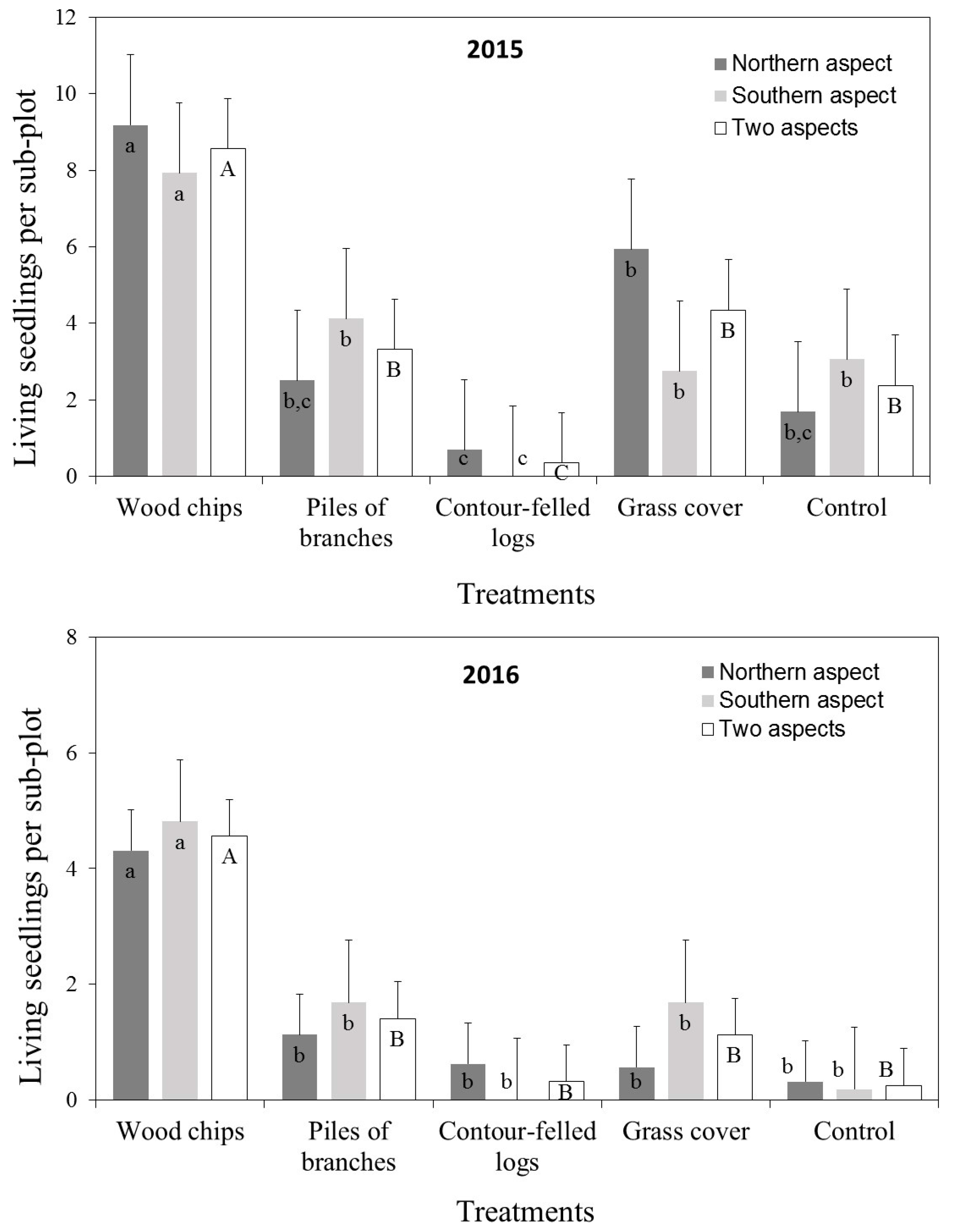
3.2. Seedling Survival
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thonicke, K.; Venevsky, S.; Sitch, S.; Cramer, W. The role of fire disturbance for global vegetation dynamics: Coupling fire into a dynamic global vegetation model. Glob. Ecol. Biogeogr. 2001, 10, 661–677. [Google Scholar] [CrossRef]
- Herranz, J.; Martinez Sanchez, J.; Marin, A.; Ferrandis, P. Postfire regeneration of Pinus halepensis Miller in a semi-arid area in Albacete Province (Southeastern Spain). Ecoscience 1997, 4, 86–90. [Google Scholar] [CrossRef]
- Pausas, J.; Llovet, J.; Rodrigo, A.; Vallejo, R. Are wildfires a disaster in the Mediterranean basin?—A review. Int. J. Wildland Fire 2008, 17, 713–723. [Google Scholar] [CrossRef]
- Broncano, M.; Rodrigo, A.; Retana, J. Post-dispersal seed predation in Pinus halepensis and consequences on seedling establishment after fire. Int. J. Wildland Fire 2008, 17, 407–414. [Google Scholar] [CrossRef]
- Ferrandis, P.; de las Heras, J.; Martinez-Sanchez, J.; Herranz, J. Influence of a low-intensity fire on a Pinus halepensis Mill. forest seed bank and its consequences on the early stages of plant succession. Isr. J. Plant Sci. 2001, 49, 105–114. [Google Scholar] [CrossRef]
- Zagas, T.; Ganatsas, P.; Tsitsoni, T.; Tsakaldimi, M. Post-fire regeneration of Pinus halepensis Mill. stands in the Sithonia Peninsula, Northern Greece. Plant Ecol. 2004, 171, 91–99. [Google Scholar] [CrossRef]
- Wagenbrenner, J.; MacDonald, L.; Rough, D. Effectiveness of three post-fire rehabilitation treatments in the Colorado Front Range. Hydrol. Process. 2006, 20, 2989–3006. [Google Scholar] [CrossRef]
- Robichaud, P.; Lillybridge, T.; Wagenbrenner, J. Effects of postfire seeding and fertilizing on hillslope erosion in north-central Washington, USA. Catena 2006, 67, 56–67. [Google Scholar] [CrossRef]
- Robichaud, P.R.; Beyers, J.L.; Neary, D.G. Rocky Mountain Research Station (Fort Collins, Colo.). In Evaluating the Effectiveness of Postfire Rehabilitation Treatments; U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2000; 85p. [Google Scholar]
- Bautista, S.; Bellot, J.; Vallejo, V. Mulching treatment for postfire soil conservation in a semiarid ecosystem. Arid Soil Res. Rehabilit. 1996, 10, 235–242. [Google Scholar] [CrossRef]
- Leon, J.; Echeverria, M.; Badia, D.; Marti, C.; Alvarez, C. Effectiveness of wood chips cover at reducing erosion in two contrasted burnt soils. Z. Geomorphol. 2013, 57, 27–37. [Google Scholar] [CrossRef]
- Badia, D.; Marti, C. Seeding and mulching treatments as conservation measures of two burned soils in the Central Ebro Valley, NE Spain. Arid Soil Res. Rehabil. 2000, 14, 219–232. [Google Scholar]
- Martinez-Sanchez, J.; Ferrandis, P.; de las Heras, J.; Herranz, J. Effect of burnt wood removal on the natural regeneration of Pinus halepensis after fire in a pine forest in Tus Valley (SE Spain). For. Ecol. Manag. 1999, 123, 1–10. [Google Scholar] [CrossRef]
- Peterson, D.; Dodson, E.; Harrod, R. Post-fire logging reduces surface woody fuels up to four decades following wildfire. For. Ecol. Manag. 2015, 338, 84–91. [Google Scholar] [CrossRef]
- Battaglia, M.A.; Rocca, M.E.; Rhoades, C.C.; Ryan, M.G. Surface fuel loadings within mulching treatments in Colorado coniferous forests. For. Ecol. Manag. 2010, 260, 1557–1566. [Google Scholar] [CrossRef]
- Miller, E.; Seastedt, T. Impacts of woodchip amendments and soil nutrient availability on understory vegetation establishment following thinning of a ponderosa pine forest. For. Ecol. Manag. 2009, 258, 263–272. [Google Scholar] [CrossRef]
- Adili, B.; El Aouni, M.; Balandier, P. Unravelling the influence of light, litter and understorey vegetation on Pinus pinea natural regeneration. Forestry 2013, 86, 297–304. [Google Scholar] [CrossRef]
- Lucas-Borja, M.E.; Fonseca, T.; Parresol, B.R.; Silva-Santos, P.; Garcia-Morote, F.A.; Tiscar-Oliver, P.A. Modelling Spanish black pine seedling emergence: Establishing management strategies for endangered forest areas. For. Ecol. Manag. 2011, 262, 195–202. [Google Scholar] [CrossRef]
- Del Cerro Barja, A.; Lucas-Borja, M.E.; Martinez Garcia, E.; Lopez Serrano, F.R.; Andres Abellan, M.; Garcia Morote, F.A.; Navarro Lopez, R. Influence of stand density and soil treatment on the Spanish black pine (Pinus nigra Arn. ssp. salzmannii) regeneration in Spain. Investig. Agrar. Sist. Recur. For. 2009, 18, 167–180. [Google Scholar] [CrossRef]
- Bertness, M.; Callaway, R. Positive interactions in communities. Trends Ecol. Evolut. 1994, 9, 191–193. [Google Scholar] [CrossRef]
- Bruno, J.; Stachowicz, J.; Bertness, M. Inclusion of facilitation into ecological theory. Trends Ecol. Evolut. 2003, 18, 119–125. [Google Scholar] [CrossRef]
- Gasque, M.; Garcia-Fayos, P. Interaction between Stipa tenacissima and Pinus halepensis: Consequences for reforestation and the dynamics of grass steppes in semi-arid Mediterranean areas. For. Ecol. Manag. 2004, 189, 251–261. [Google Scholar] [CrossRef]
- Gomez-Aparicio, L.; Zamora, R.; Gomez, J.; Hodar, J.; Castro, J.; Baraza, E. Applying plant facilitation to forest restoration: A meta-analysis of the use of shrubs as nurse plants. Ecol. Appl. 2004, 14, 1128–1138. [Google Scholar] [CrossRef] [Green Version]
- Pugnaire, F.; Armas, C.; Maestre, F. Positive plant interactions in the Iberian Southeast: Mechanisms, environmental gradients, and ecosystem function. J. Arid Environ. 2011, 75, 1310–1320. [Google Scholar] [CrossRef]
- Barnett, J.P. Direct Seeding Southern Pines: History and Status of a Technique Developed for Restoring Cutover Forests; General Technical Report SRS-GTR-187; USDA-Forest Service, Southern Research Station: Asheville, NC, USA, 2014. [Google Scholar]
- Duryea, M.L.; Dougherty, P.M. Forest Regeneration Manual; Kluwer Academic Publishers: Dordrecht, The Netherlands; Boston, MA, USA, 1991; 433p. [Google Scholar]
- Kozlowski, T. Physiological ecology of natural regeneration of harvested and disturbed forest stands: Implications for forest management. For. Ecol. Manag. 2002, 158, 195–221. [Google Scholar] [CrossRef]
- Urbieta, I.; Garcia, L.; Zavala, M.; Maranon, T. Mediterranean pine and oak distribution in Southern Spain: Is there a mismatch between regeneration and adult distribution? J. Veg. Sci. 2011, 22, 18–31. [Google Scholar] [CrossRef] [Green Version]
- Carnicer, J.; Coll, M.; Pons, X.; Ninyerola, M.; Vayreda, J.; Penuelas, J. Large-scale recruitment limitation in Mediterranean pines: The role of Quercus ilex and forest successional advance as key regional drivers. Glob. Ecol. Biogeogr. 2014, 23, 371–384. [Google Scholar] [CrossRef]
- Tiscar, P.; Linares, J. Large-scale regeneration patterns of Pinus nigra subsp. salzmannii: Poor evidence of increasing facilitation across a drought gradient. Forests 2014, 5, 1–20. [Google Scholar] [CrossRef]
- Breton, V.; Crosaz, Y.; Rey, F. Effects of wood chip amendments on the revegetation performance of plant species on eroded marly terrains in a Mediterranean mountainous climate (Southern Alps, France). Solid Earth 2016, 7, 599–610. [Google Scholar] [CrossRef]
- Lucas-Borja, M.E.; Candel-Perez, D.; Garcia Morote, F.A.; Onkelinx, T.; Tiscar, P.A.; Balandier, P. Pinus nigra Arn. ssp. salzmannii seedling recruitment is affected by stand basal area, shrub cover and climate interactions. Ann. For. Sci. 2016, 73, 649–656. [Google Scholar]
- Valladares, F.; Pearcy, R. Drought can be more critical in the shade than in the sun: A field study of carbon gain and photo-inhibition in a Californian shrub during a dry El Niño year. Plant Cell Environ. 2002, 25, 749–759. [Google Scholar] [CrossRef]
- Daskalakou, E.; Thanos, C. Postfire regeneration of Aleppo pine—The temporal pattern of seedling recruitment. Plant Ecol. 2004, 171, 81–89. [Google Scholar] [CrossRef]
- Broncano, M.; Riba, M.; Retana, J. Seed germination and seedling performance of two Mediterranean tree species, holm oak (Quercus ilex L.) and Aleppo pine (Pinus halepensis Mill.): A multifactor experimental approach. Plant Ecol. 1998, 138, 17–26. [Google Scholar] [CrossRef]
- Eugenio, M.; Lloret, F. Fire recurrence effects on the structure and composition of Mediterranean Pinus halepensis communities in Catalonia (Northeast Iberian Peninsula). Ecoscience 2004, 11, 446–454. [Google Scholar] [CrossRef]
- Lucas-Borja, M.E.; Fonseca, T.; Linares, J.C.; Garcia Morote, F.A.; Lopez Serrano, F.R. Does the recruitment pattern of Spanish black pine (Pinus nigra Arn. ssp. salzmannii) change the regeneration niche over the early life cycle of individuals? For. Ecol. Manag. 2012, 284, 93–99. [Google Scholar] [CrossRef]
- Maestre, F.; Valladares, F.; Reynolds, J. Is the change of plant-plant interactions with abiotic stress predictable? A meta-analysis of field results in arid environments. J. Ecol. 2005, 93, 748–757. [Google Scholar] [CrossRef]
- Rivas Martínez, S. Mapa de Series de Vegetación de España: 1:400.000; ICONA: Madrid, Spain, 1987. [Google Scholar]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World map of the Köppen-Geiger climate classification. Meteorol. Z. 2006, 15, 259–263. [Google Scholar] [CrossRef]
- Instituto Geológico y Minero de España; García Domingo, A. Isso. Material Cartográfico: Mapa Geológico de España E. 1:50.000, 1st ed.; Ministerio de Industria y Energía, Servicio de Publicaciones: Madrid, Spain, 1986. [Google Scholar]
- United States Department of Agriculture Natural Resources Conservation Service. Soil Taxonomy: A Basic System of Soil Classification for Making and Interpreting Soil Surveys, 2nd ed.; U.S. Department of Agriculture: Washington, DC, USA, 1999; 869p.
- Hoshmand, H.R. Design of Experiments for Agriculture and the Natural Sciences Second Edition, 2nd ed.; Champan & Hall/CRC: New York, NY, USA, 2006; p. 437. [Google Scholar]
- Neter, J.; Kutner, M.; Wasserman, W.; Nachtsheim, C. Applied Linear Statistical Models, 4th ed.; McGraw-Hill-Irwin: Chicago, IL, USA, 1996. [Google Scholar]
- Wennstrom, U.; Bergsten, U.; Nilsson, J. Seedling establishment and growth after direct seeding with Pinus sylvestris: Effects of seed type, seed origin, and seeding year. Silva Fenn. 2007, 41, 299–314. [Google Scholar] [CrossRef]
- Sabin, T.E.; Stafford, S.C. Assessing the Need for Transformation of Response Variables; Volume Special Publication 20; Forestry Publications Office, Oregon State University: Corvallis, OR, USA, 1990. [Google Scholar]
- Rhoades, C.; Battaglia, M.; Rocca, M.; Ryan, M. Short- and medium-term effects of fuel reduction mulch treatments on soil nitrogen availability in Colorado conifer forests. For. Ecol. Manag. 2012, 276, 231–238. [Google Scholar] [CrossRef]
- Wolk, B.; Rocca, M. Thinning and chipping small-diameter ponderosa pine changes understory plant communities on the Colorado Front Range. For. Ecol. Manag. 2009, 257, 85–95. [Google Scholar] [CrossRef]
- Castro, J.; Zamora, R.; Hodar, J.; Gomez, J. Use of shrubs as nurse plants: A new technique for reforestation in Mediterranean mountains. Restor. Ecol. 2002, 10, 297–305. [Google Scholar] [CrossRef]
- Trabaud, L.; Michels, C.; Grosman, J. Recovery of burnt Pinus-halepensis Mill. forests. 2. Pine reconstitution after wildfire. For. Ecol. Manag. 1985, 13, 167–179. [Google Scholar] [CrossRef]
- Pausas, J.; Ouadah, N.; Ferran, A.; Gimeno, T.; Vallejo, R. Fire severity and seedling establishment in Pinus halepensis woodlands, eastern Iberian Peninsula. Plant Ecol. 2003, 169, 205–213. [Google Scholar] [CrossRef]
- Daskalakou, E.; Thanos, C. Aleppo pine (Pinus halepensis) postfire regeneration: The role of canopy and soil seed banks. Int. J. Wildland Fire 1996, 6, 59–66. [Google Scholar] [CrossRef]
- Hedo, J.; Lucas-Borja, M.; Wic, C.; Andres-Abellan, M.; de Las Heras, J. Soil microbiological properties and enzymatic activities of long-term post-fire recovery in dry and semiarid Aleppo pine (Pinus halepensis M.) forest stands. Solid Earth 2015, 6, 243–252. [Google Scholar] [CrossRef]
- Bochet, E. The fate of seeds in the soil: A review of the influence of overland flow on seed removal and its consequences for the vegetation of arid and semiarid patchy ecosystems. Soil 2015, 1, 131–146. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Seeds: Ecology, Biogeography, and Evolution of Dormancy and Germination; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
- Garcia Morote, F.A.; Lopez Serrano, F.R.; Andres, M.; Rubio, E.; Gonzalez Jimenez, J.L.; de las Heras, J. Allometries, biomass stocks and biomass allocation in the thermophilic Spanish juniper woodlands of Southern Spain. For. Ecol. Manag. 2012, 270, 85–93. [Google Scholar] [CrossRef]
- Smith, T.; Huston, M. A theory of the spatial and temporal dynamics of plant-communities. Vegetatio 1989, 83, 49–69. [Google Scholar] [CrossRef]
- Aranda, I.; Pardos, M.; Puertolas, J.; Jimenez, M.; Pardos, J. Water-use efficiency in cork oak (Quercus suber) is modified by the interaction of water and light availabilities. Tree Physiol. 2007, 27, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, T.; Sullivan, D.; Lindgren, P.; Ransome, D. If we build habitat, will they come? Woody debris structures and conservation of forest mammals. J. Mammal. 2012, 93, 1456–1468. [Google Scholar] [CrossRef]
- Sutherland, E.; Dickman, C. Mechanisms of recovery after fire by rodents in the Australian environment: A review. Wildl. Res. 1999, 26, 405–419. [Google Scholar] [CrossRef]
- Doherty, T.; Davis, R.; van Etten, E.; Collier, N.; Krawiec, J. Response of a shrubland mammal and reptile community to a history of landscape-scale wildfire. Int. J. Wildland Fire 2015, 24, 534–543. [Google Scholar] [CrossRef]
- Saracino, A.; Pacella, R.; Leone, V.; Borghetti, M. Seed dispersal and changing seed characteristics in a Pinus halepensis Mill. forest after fire. Plant Ecol. 1997, 130, 13–19. [Google Scholar] [CrossRef]
- Castro, J.; Zamora, R.; Hodar, J.; Gomez, J.; Gomez-Aparicio, L. Benefits of using shrubs as nurse plants for reforestation in Mediterranean mountains: A 4-year study. Restor. Ecol. 2004, 12, 352–358. [Google Scholar] [CrossRef] [Green Version]
- Gomez-Aparicio, L.; Zamora, R.; Castro, J.; Hodar, J. Facilitation of tree saplings by nurse plants: Microhabitat amelioration or protection against herbivores? J. Veg. Sci. 2008, 19, 161–172. [Google Scholar] [CrossRef]
- Michalet, R.; Brooker, R.; Cavieres, L.; Kikvidze, Z.; Lortie, C.; Pugnaire, F.; Valiente-Banuet, A.; Callaway, R. Do biotic interactions shape both sides of the humped-back model of species richness in plant communities? Ecol. Lett. 2006, 9, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Garcia, E.; Ordonez, C.; Bravo, F. Effects of shrub and canopy cover on the relative growth rate of Pinus pinaster Ait. seedlings of different sizes. Ann. For. Sci. 2011, 68, 337–346. [Google Scholar] [CrossRef]

| Soil Parameter | North Slopes | South Slopes |
|---|---|---|
| Sand (%) | 48.5 ± 1.96 | 52.1 ± 2.0 |
| Silt (%) | 16.7 ± 0.6 | 13.9 ± 0.6 |
| Clay (%) | 34.8 ± 1.7 | 33.9 ± 1.8 |
| pH (1:2.5) | 8.3 ± 0.3 | 8.4 ± 0.2 |
| Organic matter (%) | 4.32 ± 0.23 | 3.91 ± 0.24 |
| C/N | 13 | 10 |
| Total C (%) | 2.50 ± 0.14 | 2.27 ± 0.14 |
| Total nitrogen (%) | 0.16 ± 0.011 | 0.18 ± 0.012 |
| Available phosphorus (ppm) | 3.56 ± 0.41 | 4.23 ± 0.42 |
| Source | Sum of Squares | d.f. | Mean Square | F-Ratio | p-Value |
|---|---|---|---|---|---|
| Aspect | 0.063 | 1 | 0.063 | 0.59 | 0.448 |
| Plot (Aspect) | 3.19 | 30 | 0.11 | 1.22 | 0.226 |
| Treatment | 2.63 | 4 | 0.66 | 7.55 | 0.000 |
| Aspect × Treatment | 0.15 | 4 | 0.039 | 0.44 | 0.778 |
| Residual | 10.46 | 120 | 0.087 | ||
| Total (corrected) | 16.5 | 159 |
| Effects | Two Years | Autumn 2015 | Autumn 2016 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| d.f. | F | p | d.f. | F | p | d.f. | F | p | |
| Year | 1 | 18.1 | 0.00 | - | - | - | - | - | - |
| Year × Treatment | 4 | 1.6 | 0.18 | - | - | - | - | - | - |
| Aspect | 1 | 0.2 | 0.66 | 1 | 0.5 | 0.49 | 1 | 0.0 | 0.95 |
| Plot (Aspect) | 30 | 4.3 | 0.00 | 30 | 2.3 | 0.00 | 30 | 1.9 | 0.01 |
| Treatment | 4 | 30.8 | 0.00 | 4 | 13.5 | 0.00 | 4 | 17 | 0.00 |
| Aspect × Treatment | 4 | 0.4 | 0.79 | 4 | 0.1 | 0.99 | 4 | 0.6 | 0.66 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Morote, F.A.; Martínez-García, E.; Andrés-Abellán, M.; Caballero, E.R.; Miettinen, H.; López-Serrano, F.R. Direct Seeding of Pinus halepensis Mill. for Recovery of Burned Semi-Arid Forests: Implications for Post-Fire Management for Improving Natural Regeneration. Forests 2017, 8, 353. https://doi.org/10.3390/f8090353
García-Morote FA, Martínez-García E, Andrés-Abellán M, Caballero ER, Miettinen H, López-Serrano FR. Direct Seeding of Pinus halepensis Mill. for Recovery of Burned Semi-Arid Forests: Implications for Post-Fire Management for Improving Natural Regeneration. Forests. 2017; 8(9):353. https://doi.org/10.3390/f8090353
Chicago/Turabian StyleGarcía-Morote, Francisco Antonio, Eduardo Martínez-García, Manuela Andrés-Abellán, Eva Rubio Caballero, Heli Miettinen, and Francisco Ramón López-Serrano. 2017. "Direct Seeding of Pinus halepensis Mill. for Recovery of Burned Semi-Arid Forests: Implications for Post-Fire Management for Improving Natural Regeneration" Forests 8, no. 9: 353. https://doi.org/10.3390/f8090353
APA StyleGarcía-Morote, F. A., Martínez-García, E., Andrés-Abellán, M., Caballero, E. R., Miettinen, H., & López-Serrano, F. R. (2017). Direct Seeding of Pinus halepensis Mill. for Recovery of Burned Semi-Arid Forests: Implications for Post-Fire Management for Improving Natural Regeneration. Forests, 8(9), 353. https://doi.org/10.3390/f8090353







