Abstract
Forests on calcareous substrates constitute a large fraction of the vegetation in Puerto Rico. Plant growth on these substrates may be affected by nutrient deficiencies, mainly P and Fe, resulting from high pH and formation of insoluble compounds of these elements. The occurrence of these forests in humid and dry areas provides an opportunity to compare nutrient relations, water use efficiency, and N dynamics, using biogeochemical parameters. We selected sites under humid climate in the north, and dry climate in the southwest of Puerto Rico. Adult, healthy leaves of species with high importance values were collected at each site and analyzed for their elemental composition and the natural abundance of C and N isotopes. Calcium was the dominant cation in leaf tissues, explaining over 70% of the ash content variation, and Al and Ca concentration were positively correlated, excepting only two Al-accumulating species. Karst vegetation consistently showed high N/P ratios comparable to forests on P-poor soils. Dry karst sites had significantly higher δ13C and δ15N ratios. We conclude that forests on karst are mainly limited by P availability, and that mechanisms of nutrient uptake in the rhizosphere lead to linear correlations in the uptake of Ca and Al. Isotope ratios indicate higher water use efficiency, and predominant denitrification in dry karst forest sites.
1. Introduction
Limestone areas are widespread in the Caribbean under dry to subhumid climatic conditions. Those areas developed mostly during the middle Oligocene to the middle Pliocene and have been documented extensively in Jamaica, Cuba, Santo Domingo, and Puerto Rico [1,2,3,4,5,6]. Several forest types, from dry to moist, covering large areas of those geologic formations, have been described in detail in many locations [4,7,8,9].
In Puerto Rico, karst areas constitute about 27% of the territory and are separated as moist karst forming a northern belt crossing the island from east to west, and a dry karst formation in the southwest, under semiarid climate [4]. These formations have a similar geological age (Pliocene-Oligocene) but are under different rainfall regimes [10]. The limestone formations are covered throughout the island by sediments constituted by non-calcareous material that, in the northern karst belt, accumulate in depressions between haystack (mogotes) hills [10,11]. On top of the mogotes in the karst belt, or on the calcareous hills of the southern karst, this material may accumulate within crevices and fractures of the calcareous strata. Surface soils in karstic areas, when present, are highly heterogeneous, frequently shallow, and, in many cases, difficult to sample. Soils derived from the calcareous substrate itself are infrequent. On ridge tops and slopes, soil is almost inexistent, and substrate is a rocky, partially fragmented surface, covered with organic residues produced by the vegetation. Fragments of the carbonaceous rock and organic matter slide downhill and accumulate in the valleys [4,11,12]. On the valleys, true soils develop constituted by calcareous rocks fragments and the remaining material transported from volcaniclastic areas during the time when the carbonate rocks were under water (see [11] for an explanation of the composition of blanket sands). Assessment of nutrient availability for plants on the ridge top sites using soils as reference is therefore not a practical approach. The alternative is the analysis of plant organs, particularly leaves [13].
Nutrient availability for plant growth in soils derived from carbonate rocks is usually limited, particularly in P, as it tends to be immobilized as Ca compounds of low solubility [14]. High soil pH determines lower mobility of elements such as Mn and Fe, resulting in potential limitation for plant growth (Ca induced iron chlorosis). The nutrient relationships of plants from calcareous substrates have been mainly studied in temperate climates [15] and little is known from tropical vegetation [16].
Typical vegetation of moist karst forests has been described by Alvarez et al. [17] and Acevedo-Rodríguez and Axelrod [18] and that of the dry karst forest was studied by Murphy and Lugo [19], and documented in detail by Monsegur [20].
Our study took advantage of detailed phytosociological analysis of the northern karst vegetation undertaken by Chinea [21] and Aukema et al. [22]. In the latter, the authors quantified the composition of forest units according to species dominance relationships and geomorphological positions on the mogotes (top, slopes, valleys). This study was critical to the identification of tree assemblages on the top of mogotes. In addition, we used the vegetation analysis of Murphy and Lugo [19] and Molina and Lugo [23] for the Guánica dry forest in southern Puerto Rico. The dominant tree species in typical sites of northern and southern karts areas, as defined by their importance value, reveals the occurrence of common and restricted species. For example, Coccoloba diversifolia (Polygonaceae) and Gymnanthes lucida (Euphorbiaceae) occur as dominant species in both moist and dry karst, whereas Pisonia albida (Nyctaginaceae), Thouinia portoricensis (Sapindaceae), and Pictetia aculeata (Fabaceae) seem to be restricted to dry karst forests, and Lonchocarpus lancifolius (Fabaceae), and Prunus myrtifolia (Rosaceae) are restricted to the northern moist karst belt [24].
The objective of the present study was to provide a geochemical characterization of the karst areas in the island of Puerto Rico through the elemental analysis of substrate and plant leaves. Leaf analyses provide information on both the availability of specific elements in the soil solution, and reveal physiological properties such as exclusion or facilitation of element uptake at the root level [13]. We tested hypotheses related to the leaf elemental composition, substrate geochemistry, and environmental humidity: (1) Ca is the main component of leaf ash due to its expected high availability in the calcareous substrate; (2) Lower total P concentrations and high N to P ratios reflect relative limited availability of P, probably associated with the formation of insoluble Ca compounds; (3) Concentrations of heavy metals (Fe and Mn) and Al, are probably low compared to species from forests on non-calcareous substrates; (4) Environmental humidity determined by seasonality and magnitude of rainfall affect plant water use efficiency and rate of organic N mineralization in the substrate.
2. Materials and Methods
For the study of the nutritional relationships of tree species strictly associated with karstic substrates, we selected sites in the humid karst belt and in the semiarid south west karst area in Puerto Rico. The sampling was restricted to plants growing on the top of haystack hills in the northern karst belt, and on ridge sites and coastal carbonate pavement in the southwestern karst area.
We selected six mogote tops sites in the moist northern karst belt and two sites in the dry Guánica forest in southwestern Puerto Rico (Table 1). The former sites were located along a west-east line on the northern karst belt, on top of the Aymamon limestone type, whereas those on the dry karst areas are on the Ponce limestone type [10]. The northern karst belt receives, on average, annual rainfall amounts of 1000–1500 mm, whereas in the southern karst areas long-term annual rainfall averages lie below 1000 mm. This contrasting rainfall pattern is clearly exemplified by two stations, Utuado (158 m), located in the middle of the Aymamon karst [10] in the northern karst belt, and Ensenada (46 m), on the southwestern coast, representative for the rainfall regime of the southern karst belt (Figure 1).

Table 1.
Karst site locations (Lat, Long) and approximate altitude.
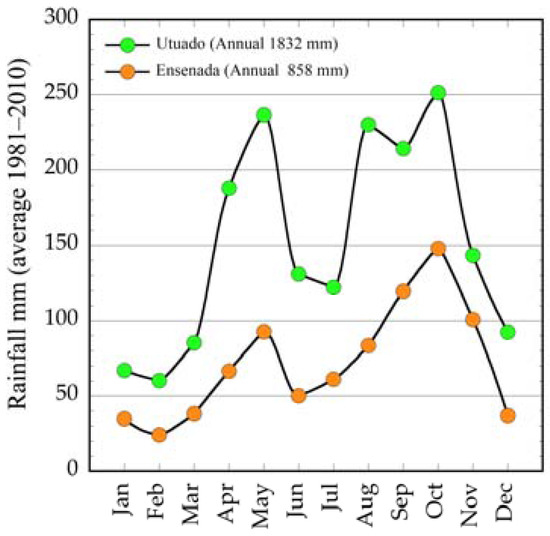
Figure 1.
Average rainfall pattern of humid and dry karst sites.
The species sampled at each site were those more common, with high importance values [16,19,22,23] (Table 2). The number of species varied from site to site due to different areal extension, and the number of tree species growing on the mogote top. In total, we sampled 30 species from the northern, moist sites, and 26 species from the dry sites, representing 29 families and 18 orders. Only five species were sampled from both humidity sites.

Table 2.
Species sampled at each site designated by name and municipality.
For elemental analyses, we sampled at each site healthy mature leaves, without visible herbivore consumption, of trees from the upper forest canopy. Leaves were dried in the laboratory in a ventilated oven at 65 °C (3–7 days) and ground to pass an 18-mesh screen. Rock fragments on the forest floor were collected from each site. In the laboratory, rock samples were dissolved in 2N HCl for elemental analysis.
Elemental composition of samples was determined (C, N, S, P, K, Ca, Mg, Al, Mn, Fe) using standard methodology of the laboratory at the International Institute of Tropical Forestry [25]. Briefly, in acid digested samples, C, N, and S were determined using a LECO elemental analyzer, whereas P and cations were measured using Inductively coupled plasma atomic emission spectroscopy (ICP-AES) techniques.
In addition, the natural abundance of C (δ13C) and N isotopes (δ15N) in leaves was determined at the Stable Isotope Laboratory of the University of Miami, Coral Gables, using mass spectrometric techniques. The δ13C values are used as indicators of long-term water use efficiency, because they are related to lower leaf conductance [26]. The leaf δ15N values are related to the natural abundance of N isotopes in the substrate. More positive values point to higher rates of mineralization of organic N in the soil [27].
Element concentrations and C and N isotopic ratios of all species per site were submitted to a one-way analysis of variance and post-hoc tests for comparison of site averages. Inter-element correlations were estimated for the whole set of species to establish commonalities among sites. All statistical analyses were performed using the JMP statistical program.
3. Results
3.1. Elemental Composition of the Substrate in Karst Areas in Puerto Rico
The analysis of the elemental composition of rocks from different karst types of the northern karst belt published by Monroe [12] indicate that nearly 83% is constituted by CaCO3, and that they contain small but significant amounts of Mg, Al, and Fe. The elements P and particularly K are found at concentrations below 1 mmol/kg. Rock fragments from the sites selected for vegetation sampling showed important differences in the proportion of minor elements. The sequence of elemental concentrations in the northern karst site confirmed the sequence Ca >> Mg > Al > Fe, corresponding to the average values from Monroe [12] (Figure 2). The samples from the southern karst had a similar elemental distribution, but Na was present in the Guánica samples at higher concentrations than any other site. Phosphorus concentration was at the same level in all samples (≈1 mmol/kg). Potassium reached concentrations around 10 mmol/kg in the southern samples, whereas it was always well below the 1 mmol/kg level in the northern karst samples.
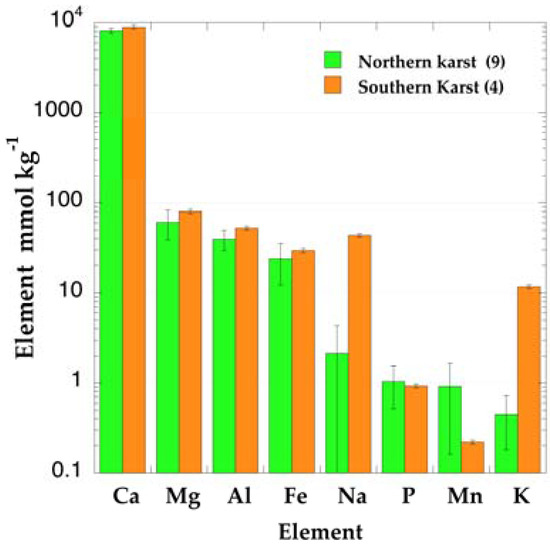
Figure 2.
Average elemental composition of karst rocks from northern and southern sites in Puerto Rico.
The presence of non-calcareous sediments in cracks, holes, and crevices of the karstic substrate raised the question about their potential as a source of nutrients for the vegetation. They have been characterized in the Dominican Republic as lateritic in nature, with high concentrations of Al and Fe [1]. We do not have information on the elemental composition of those sediments in Puerto Rico. However, we calculated sediment concentrations from a nutrient inventory study in the northern karst belt [28]. Both A and C horizons indicate much higher concentrations of Fe, Mn, and Al, and lower concentrations of Ca and Mg compared to those measured in calcareous rocks (Figure 3). The higher concentrations of Ca and Mg of the A horizon reveal the influence of in situ weathering of karst rocks. It appears then that these sediments may be a source of nutrients for the vegetation, and due to their clayey texture, they have much higher water retention and cation exchange capacities than the calcareous substrates.
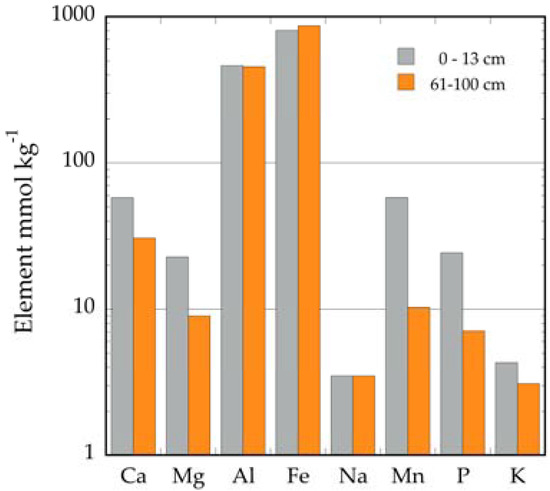
Figure 3.
Elemental composition of clay soils on karst in El Tallonal, Arecibo, Puerto Rico. Values for the A horizon (0–13 cm) and the C horizon (61–100 cm) were generated from [28].
3.2. Leaf Elemental Composition
The concentration of the main organic matter elements did not differ significantly between sites. The coefficients of variation were small for C (<10%), intermediate for N and P (10 to 100%), and large for S (>100%) (Table 3). The concentration of metallic elements differed between sites for K, Ca, Fe, Mn, and Na (Table 4). The coefficients of variation were intermediate for K, Mg, Ca, and Fe, large for Na, and very large for Al and Mn (>250%). In the southern karst, average K concentration was higher, whereas for Ca and Mn concentration averages were lower than the overall mean. The average Na concentration in the Guánica Dwarf forest site was much larger than the overall mean, probably because this site is located near the coast line, under the influence of sea salt spray, and Na may be present in the rooting substrate, as indicated by the composition of the limestone from Guánica (Figure 2).

Table 3.
Mean (±SE) concentrations of main elements of organic matter C in mol/kg, the rest in mmol/kg.

Table 4.
Mean (± SE) concentrations of ash (%) and metallic elements in leaves per site in mmol/kg.
Some species within the sampled set stand out, with concentrations of particular elements 2–3 times larger or smaller than the overall average (Table 5). These differences may be attributed to variations in element availability between sites, to differences in leaf age, or to actual physiological differences between species. Leaf age between samples of the same species at different sites cannot be discarded, as leaf sampling was conducted during several months; however, leaves collected were always fully expanded, healthy, and without visual signals of senescence or herbivory.

Table 5.
Maximum and minimum concentrations of elements (mmol/kg).
The design of the research was not oriented to differentiate species occurring at different sites; therefore, the data set is insufficient to conduct within species comparisons.
Elemental ratios between organic matter forming elements (C, N, S, P) do not differ significantly between sites (Table 6). However, K to Ca ratios were larger and the Ca to Mg ratios lower for the two sites on the southern karst in correspondence with the relative elemental concentration of those elements measured in the calcareous rocks. The other sites showed K to Ca ratios ranging from 0.14 to 0.47, revealing the high availability of Ca in the soil solution.

Table 6.
Molar elemental ratios of leaves from trees on calcareous substrates.
Inter-Element Correlations
The correlation between leaf N and P was positive and significant, as usually reported for forest foliage throughout the world [29] (Figure 4). As expected, the Ash% was inversely correlated with C concentration (R2 adj = 0.57), but positively correlated with Ca (R2 adj = 0.72), Mg (R2 adj = 0.24), and Mn (R2 adj = 0.08). Clearly Ca and Mg were the main constituents of ash, explaining more than 80% of the variance.
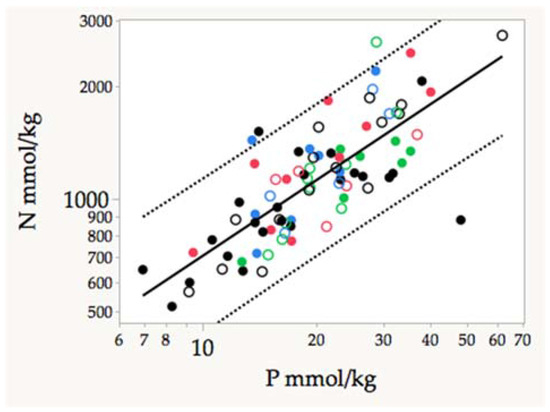
Figure 4.
Correlation between N and P concentrations in adult leaves from different karst forest species. Guánica forests: dwarf, Black; ridge, empty black; Guajataca, solid blue; Cambalache, empty blue; Tallonal solid green; Nevarez, empty green; Río Lajas, solid red; Hato Tejas, empty red. Log (N mmol/kg) = 5.011 + 0.672 × Log (P); Fratio = 120; F < p < 0.001; R2 adj = 0.592; n = 83.
We found an unexpected highly significant correlation between Ca and Al concentrations for all sites (Figure 5). Only two species departed strongly from this relationship, T. elaeagnoides and L. glaucifolius, both from the Guajataca site. This relationship deserves special attention because of its implications regarding the mechanisms of nutrient uptake from karstic complex substrates. Considering the low concentrations of Al, Fe, and Mn in the calcareous substrate in karst areas, it is not surprising that these elements are only a minor component of leaf ash of trees growing there. If clayey sediment accumulated in cracks and crevices of the rocky substrates on mogote tops was a source of those metallic elements, we expected higher concentrations of them in tree foliage, but that was not the case in our data set, with the exception of T. elaeagnoides, identified as a metal accumulator (Table 5).
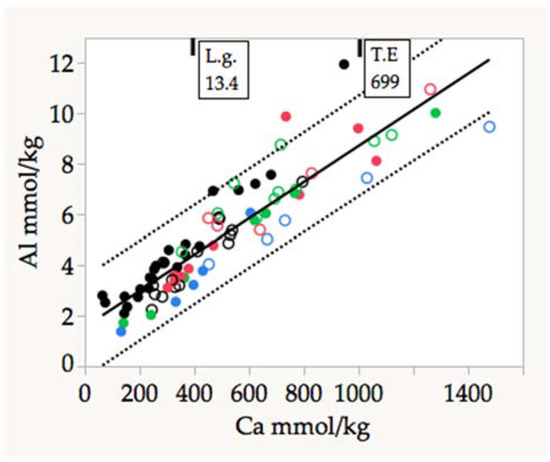
Figure 5.
Linear correlation between the concentrations of Ca and Al in adult leaves of karst tree species. The arrows indicate the values of Al accumulators which were not included in the regression. Al (mmol/kg) = 1.55 + 0.007 × Ca (mmol/kg). R2 adj = 0.825; F = 379, p < 0.0001, n = 81. Symbols are the same as in Figure 4.
3.3. Isotopic Ratios
The δ13C values did not differ significantly in the northern karst forest locations (Figure 6). The only outlier was represented by C. rosea, a tree with crassulacean acid metabolism [30] commonly occurring in the northern karst belt forests. It was excluded from average calculations. As expected, the two sites on the southern karst had significantly higher δ13C values, because of the drier conditions under which this vegetation grows. Average δ13C of leaves from humid karst was –30.83 ± 1.85‰ (n = 52), and from dry karst was −27.37 ± 1.16‰, the difference was highly significant (Tukey Honest Significant Difference test p = 0.01).
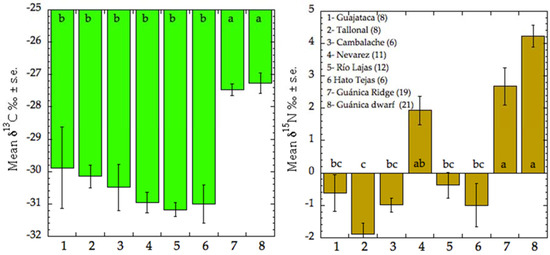
Figure 6.
Average ± SE. values of N and C isotopes in adult leaves from different karst forests in Puerto Rico. Columns with the same letter at the base indicate statistical similarity (Tukey Honest Significant Difference, p = 0.01).
The distribution of δ15N values varied significantly among sites, showing a more complex pattern than that of the carbon isotopes (Figure 6). The sites on southern karst and the Nevarez site showed positive values, whereas the rest of the sites had negative δ15N average values. Overall averages were positive in the dry karst, 3.49 ± 2.20‰ (n = 40), and slightly negative, −0.28 ± 1.82‰ (n = 51), in the moist karst forests. The degree of overlap was higher in the case of 15N, revealing the more complex regulation of this factor under natural conditions.
4. Discussion
4.1. Comparison between Humid and Dry Tropical Forests on Contrasting Soils
Comparison of the dry/moist karst leaf data with other assessments of leaf nutrient concentration in wet and seasonally dry tropical forests shows, in general, that karst forests in Puerto Rico are more limited by P than N, judging by their high N/P ratios, similar to those reported for P-poor forests in the Amazon basin (Table 7). A lower montane rain forest on volcanic soils in Puerto Rico (El Verde, Luquillo Experimental Forest) [31] has much higher concentrations of K and P, and lower N/P ratio than both the humid and dry karst leaves. Moreover, dry and humid karst leaves have quite similar average concentrations of Mg, P, and N, and large N/P ratios. Remarkably, dry karst leaves are richer in K, but poorer in Ca compared to humid karst leaves. Possible explanation for the high K may be the coastal location of the dry karst sites, whereas the higher Ca concentration in moist karst is probably associated with the prevailing humidity of the substrate, which leads to higher availability of soluble Ca.

Table 7.
Average leaf nutrient concentrations in a range of humid and dry tropical forests (mmol/kg).
The average values of C, N, and P from our data set are similar to those reported by Hättenschwiler et al. [33] for 45 tree species in a lowland wet neotropical forest. The range of variation of the species from karst forests (Table 3) is the same for C but much larger for N and P than that of the neotropical forest in French Guiana. The average N/P ratio of their data set amounts to 54, slightly lower than the average of our data set (58), but the C/N and C/P ratios are similar.
Absolute concentrations of N and P in karst forest species are also lower than those reported for many species from forests on P-poor oxisols in Amazonas by Townsend et al. [32], but the N/P ratio is quite similar, amounting to 58 on a molar basis. It may be concluded from these comparisons with moist and wet tropical forest sites that the karts forests from Puerto Rico are relatively limited by P.
Most seasonally dry forest soils in the neotropics are comparatively rich in Ca, but are as low in P as the karst forest reported here. Leaf P concentrations in seasonally dry forest in Chamela, Mexico [34], and a deciduous forest on calcareous outcrops in the cerrado of Goias in Brazil [35], are much higher than the values reported here (Table 7). Therefore, their average leaf N/P ratios are much lower than those measured in our karst forests.
A recent study from vegetation on karst in China slightly above 26° N reports surprisingly much higher P concentrations in leaves, and lower N/P values than those reported here for Puerto Rico [36]. Concentrations of the other elements are within the same range as those reported here (Table 7).
The influence of element availability in substrate is suggested by the fact that the species with higher K concentrations, and the mean of site values, correspond to the Guánica Ridge and Dwarf forests, in agreement with the results of rock analyses.
Physiological differences may be indicated for the case of Tetrazygia elaeagnoides, one of the few Melastomataceae tree species recorded for mogote tops in the northern karst belt [17,24]. This species is a strong Al accumulator, a characteristic common among the members of the Melastomataceae, but it is remarkable that it also showed the highest concentrations of Fe and Mn in the whole data set. In addition, the two legumes included in this data set, P. arboreum and L. glaucifolius, are among the species with highest concentrations of N.
Concentrations of heavy metals (Fe and Mn) in the Liu et al. study [36] were also about two times higher than in the present paper (2 vs. 0.7 mmol/kg). Data on Al in karst vegetation were not found in the available literature. Excluding the Al accumulators sampled in Guajataca site, concentrations reported here are below 10 mmol/kg (Table 4), much lower than values reported by Masunaga et al. [37] for non-accumulator trees in Sumatra rain forest (21.5).
4.2. The Ca/Al Relationship: Al Accumulators in Limestone Forests
In an extensive analysis of tree material in a tropical rain forest in West Sumatra, Masunaga et al. [37,38] were able to identify a wide variation in Al accumulation in tree foliage. They modified the criteria for defining Al-accumulator species using the 1 g/kg leaf concentration boundary (37 mmol/kg), introducing categories of accumulators between 1 and 3 g Al/kg (37–111 mmol/kg) and strong accumulators with >3 g Al/kg (>111 mmol/kg). For non-Al accumulators, Al was positively correlated with Ca (r = 0.62, n = 469) and Mg (r = 0.23). For strong accumulators, the correlation was not significant at the 1% level. For the non-accumulators, Masunaga et al. [37] indicate that the Al/Ca ratio averages 0.04 ± 0.02, whereas for our data set this ratio averaged0.011 ± 0.005, due to the much higher Ca concentration in leaf tissues (509 vs. 367 mmol Ca/kg).
The elemental concentration of leaves from rainforests on Ca-rich soils in West Sumatra is characterized by a large range of variation of most metals, particularly Al, Fe, and Mn, and includes several metal accumulator species [38]. The rainforest leaves of the West Sumatra forest have quite similar concentrations of P, S, K, Ca, and Mg, but much higher concentrations of Al, Fe, and Mn than the karst forest species reported here. These comparisons show that in spite of huge differences in rainfall, the main factor regulating concentration of cations in leaves is the elemental availability from soil, with the exception of metal accumulators. Aluminum accumulators are numerous in the West Sumatra data set [37], whereas only two tree species were detected in the karst forests studied.
Why is there such a clear relationship between Ca and Al concentrations in non-Al accumulators, both in rain and moist and dry karst forests? We suggest that root activity in calcareous substrate includes the secretion of organic acids in the rhizosphere, leading to the dissolution of the carbonatic rocks, thereby releasing Ca and other metals present, as well as P. This is one of the specific mechanisms described to counteract low Fe availability in calcareous soils [14]. Organic acids may act as chelating agents facilitating Fe uptake, but also the uptake of other metallic cations present in the soil solution. In addition to the high Al-Ca correlation, our data set shows that Ca is linearly, but more weakly, related to the concentrations of Mg and Fe (p = 0.05), but no relationship was detected with Mn.
4.3. Isotopic Relationships
The significant higher values of δ13C in both dry karst forest sites were expected. The restrictions in water supply determine long-term reductions in stomatal conductance leading to reduction in 13C discrimination in the photosynthetic process [26].
Variation of N isotopic values under natural conditions can be associated with intrinsic biological characteristics of the species involved or with ecosystems nutrient cycling properties:
- (a)
- Occurrence of mycorrhiza may decrease values compared to non-mycorrhizal plants [39,40].
- (b)
- Symbiotic N2 fixation (as in legumes-Rhizobium association) results in lower values because this process does not discriminate against the heavier N isotope [41].
- (c)
- Conditions inhibiting denitrification processes (water saturated soils) lead to more negative values in vegetation, the opposite effect occurs in drier areas where denitrification operates freely [42,43].
- (d)
- High levels of N availability and P limitation lead to higher values [40,44].
There are few studies on mycorrhizal associations in woody vegetation on calcareous substrates in the Caribbean. However, a recent report concluded that seasonal rainfall has a strong influence on the diversity of mycorrhizal fungi but not on infectivity [45]. Therefore, we may assume that mycorrhiza is not responsible for the differences observed. Furthermore, legumes represent only a small fraction of the species analyzed (2 out of 56). Finally, P deficiencies assessed through the N to P ratios were similar for moist and dry sites. The main factor determining lower leaf δ15N values in the moist north karst species compared to the drier southern karst region seems to be the difference in rainfall. In mogote tops, however, humidity conditions may be highly heterogeneous due to low water retention capacity of calcareous substrates, and the random occurrence of rock cracks and crevices where water may be retained.
5. Conclusions
- Karst vegetation in Puerto Rico appears to be P limited based both on lower P concentrations and high N/P ratios in leaves. The latter are similar to those of rain forests on P poor soils.
- Source of cations originate both from calcareous rocks (Ca and Mg) and allochthonous sediments from volcaniclastic rocks (Al and Fe).
- Calcium is the predominant cation in leaf tissue and its concentration explains more than 70% of the variation in ash content.
- The leaf concentrations of K and Na are larger in the dry karst sites, probably because of their coastal location. Calcium was higher in moist karst sites, probably due the larger availability of soluble Ca in the substrate.
- The tree species of both moist and dry karst forest accumulate small amounts of Al, which is linearly correlated with the total accumulation of Ca in leaf tissue. This correlation may derive from the process of element solubilization in the rhizosphere driven by the secretion of organic acids.
- The δ13C values of leaf tissue are significantly higher in the dry karst forest, revealing the limitations of water supply.
- The δ15N values of leaf tissue were, with one exception, significantly lower in moist karst sites. The probable cause is that denitrification tends to predominate in drier environments.
Acknowledgments
Field work and chemical analyses were conducted using IITF facilities. Alberto Rodríguez (formerly at IITF) provided assistance during field work, and Marcos Caraballo (University of Puerto Rico) helped with the species identification. Edwin López Soto from IITF lab revised the methods section and the whole data set.
Author Contributions
E.M., E.C. and A.E.L. conceived the project; E.M. and E.C. conducted the field sampling; E.M. conducted the statistical analyses and wrote the paper with contributions from E.C. and A.E.L.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Goldich, S.S.; Bergquist, H.R. Aluminous Lateritic Soil of the Sierra de Bahoruco Area Dominican Republic, W. I.; Bulletin 953-C; United States Department of the Interior, Geological Survey, United States Government Printing Office: Washington, DC, USA, 1947.
- Asprey, G.F.; Robbins, R.G. The vegetation of Jamaica. Ecol. Monogr. 1953, 23, 359–412. [Google Scholar] [CrossRef]
- Lötschert, W. Die Übereinstimmung von geologischer Unterlage und Vegetation in der Sierra de los Organos (Westkuba). Ber. Deutsch. Bot. Ges. 1958, 71, 55. [Google Scholar]
- Lugo, A.E.; Miranda Castro, L.; Vale, A.; López, T.M.; Hernández Prieto, E.; García Martinó, A.; Puente Rolón, A.R.; Tossas, A.G.; McFarlane, D.A.; Miller, T.; et al. Puerto Rican Karst—A Vital Resource; General Technical Report WO-65; USDA Forest Service: Washington, DC, USA, 2001.
- Díaz del Olmo, F.; Cámara Artigas, R. Karst Tropical de Colinas, Tipología y Evolución en el Pliocuaternario en República Dominicana; Flor, G., Ed.; Actas de la XI Reunión Nacional de Cuaternario, XI Reunión Nacional de Cuaternario: Oviedo, Spain, 2003; pp. 123–129. [Google Scholar]
- Day, M. Human interaction with Caribbean karst landscapes: Past, present and future. Acta Carsologica 2010, 39, 137–146. [Google Scholar] [CrossRef]
- Kelly, D.L.; Tanner, E.V.J.; Kapos, V.; Dickinson, T.A.; Goodfriend, G.A.; Fairbairn, P. Jamaican Limestone Forests: Floristics, structure and environment of three examples along a rainfall gradient. J. Trop. Ecol. 1988, 4, 121–156. [Google Scholar] [CrossRef]
- Borhidi, A. Dry coastal ecosystems of Cuba. In Ecosystems of the World. Dry Coastal Ecosystems Africa, America, Asia and Oceania; van der Maarel, E., Ed.; Elsevier: Amsterdam, The Netherlands, 1993; Volume 2, pp. 423–452. [Google Scholar]
- Areces-Mallea, A.E.; Weakley, A.S.; Li, X.; Sayre, R.G.; Parrish, J.D.; Tipton, C.V.; Bouche, T. A Guide to Caribbean Vegetation Types: Preliminary Classification System and Descriptions; Panagopoulos, N., Ed.; The Nature Conservancy International Headquarters: Washington, DC, USA, 1999; p. 166. [Google Scholar]
- Alemán-Gonzalez, W.B. Karst Map of Puerto Rico; US Geological Survey: Reston, VA, USA, 2010.
- Briggs, R.P. The blanket sands of northern Puerto Rico. In Proceedings of the Transactions of the 3rd Caribbean Geology Conference, Kingston, Jamaica, 2–11 April 1962; Jamaica Geological Survey Publication: Kingston, Jamaica, 1966; pp. 60–69. [Google Scholar]
- Monroe, W.H. The Karst Landforms of Puerto Rico; U.S. Geological Survey Professional Paper 899; Government Printing Office: Washington, DC, USA, 1976; p. 69.
- Mengel, K.; Kirkby, E.A. Principles of Plant Nutrition, 5th ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; p. 849. [Google Scholar]
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: Cambridge, MA, USA, 1995. [Google Scholar]
- Kinzel, H. Influence of limestone, silicates and soil pH on vegetation. In Physiological Plant Ecology III. Responses to Chemical and Biological Environment; Lange, O.L., Nobel, P.S., Osmond, C.B., Ziegler, H., Eds.; Encyclopedia of Plant Physiology; Springer: Berlin, Germany, 1983; Volume 12, pp. 201–244. [Google Scholar]
- Medina, E.; Cuevas, E.; Molina, S.; Lugo, A.E.; Ramos, O. Structural variability and species diversity of a dwarf Caribbean dry forest. Caribb. J. Sci. 2012, 46, 203–215. [Google Scholar] [CrossRef]
- Alvarez Ruiz, M.; Acevedo Rodriguez, P.; Vazquez, M. Quantitative description of the structure and diversity of the vegetation in the Limestone Forest of Rio Abajo, Arecibo-Utuado, Puerto Rico. Acta Cient. 1997, 11, 21–66. [Google Scholar]
- Acevedo-Rodríguez, P.; Axelrod, F.S. Annotated checklist for the tracheophytes of Río Abajo forest reserve, Puerto Rico. Caribb. J. Sci. 1999, 35, 265–285. [Google Scholar]
- Murphy, P.G.; Lugo, A.E. Structure and biomass of a subtropical dry forest in Puerto Rico. Biotropica 1986, 18, 89–96. [Google Scholar] [CrossRef]
- Monsegur-Rivera, O.A. Vascular Flora of the Guánica dry Forest, Puerto Rico. Master’s Thesis, University of Puerto Rico, Mayagüez Campus, San Juan, Puerto Rico, 2009. [Google Scholar]
- Chinea, J.D. The Forest Vegetation of the Limestone Hills of Northern Puerto Rico. Ph.D. Thesis, Cornell University, Ithaca, NY, USA, 1980. [Google Scholar]
- Aukema, J.E.; Carlo, T.A.; Collazo, J.A. Landscape assessment of tree communities in the northern karst region of Puerto Rico. Plant Ecol. 2007, 189, 101–115. [Google Scholar] [CrossRef]
- Molina Colon, S.; Lugo, A.E. Recovery of a subtropical dry forest after abandonment of different land uses. Biotropica 2006, 38, 354–364. [Google Scholar] [CrossRef]
- Axelrod, F.S. A Systematic Vademecum to the Vascular Plants of Puerto Rico; Botanical Research Institute of Texas: Fort Worth, TX, USA, 2011. [Google Scholar]
- Sánchez, M.J.; Lopez, E.; Lugo, A.E. Chemical and Physical Analysis of Selected Plants and Soils from Puerto Rico (1981–2000); Gen. Tech. Rep. GTR-IITF-45; U.S. Department of Agriculture, Forest Service, International Institute of Tropical Forestry: Rio Piedras, PR, USA, 2015; p. 85.
- Farquhar, G.D.; Hubick, K.T.; Condon, A.G.; Richards, R.A. Carbon isotope fractionation and plant water use efficiency. In Stable Isotopes in Ecological Research; Rundel, P.W., Ehleringer, J.R., Nagy, K.A., Eds.; Ecological Studies; Springer: New York, NY, USA, 1989; Volume 68, pp. 21–40. [Google Scholar]
- Handley, L.L.; Raven, J.A. The use of natural abundance of nitrogen isotopes in plant physiology and ecology. Plant Cell Environ. 1992, 15, 965–985. [Google Scholar] [CrossRef]
- Viera Martínez, C.A.; Abelleira Martínez, O.J.; Lugo, A.E. Estructura y química del suelo en un bosque de Castilla elastica en el carso del norte de Puerto Rico: Resultados de una calicata. Acta Cient. 2008, 22, 29–35. [Google Scholar]
- McGroddy, M.E.; Daufresne, T.; Hedin, L.O. Scaling of C:N:P stoichiometry in forests worldwide: Implications of terrestrial Redfield-type ratios. Ecology 2004, 85, 2390–2401. [Google Scholar] [CrossRef]
- Tinoco-Ojanguren, C.; Vázquez-Yánes, C. Especies CAM in la selva húmeda tropical de Los Tuxtlas, Veracruz. Bol. Soc. Bot. Mex. 1983, 45, 150–153. [Google Scholar]
- Ovington, J.D.; Olson, J.S. Biomass and Chemical Content of EI Verde Lower Montane Rain Forest Plants. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde, Puerto Rico; Odum, H.T., Pigeon, R.F., Eds.; National Technical Information Service: Springfield, VA, USA, 1970; Volume H-2, pp. H53–H75. [Google Scholar]
- Townsend, A.R.; Cleveland, C.C.; Asner, G.P.; Bustamante, M.M.C. Controls over foliar N:P ratios in tropical rain forests. Ecology 2007, 88, 107–118. [Google Scholar] [CrossRef]
- Hättenschwiler, S.; Aeschlimann, B.; Coûteaux, M.-M.; Roy, J.; Bonal, D. High variation in foliage and leaf litter chemistry among 45 tree species of a neotropical rainforest community. New Phytol. 2008, 179, 165–175. [Google Scholar]
- Rentería, L.Y.; Jaramillo, V.J. Rainfall drives leaf traits and leaf nutrient resorption in a tropical dry forest in Mexico. Oecologia 2011, 165, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Rossatto, D.R.; Alvim Carvalho, F.; Haridasan, M. Soil and leaf nutrient content of tree species support deciduous forests on limestone outcrops as a eutrophic ecosystem. Acta Bot. Bras. 2015, 29, 231–238. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y.; Guo, K.; Wang, S.; Yang, Y. Concentrations and resorption patterns of 13 nutrients in different plant functional types in the karst region of south-western China. Ann. Bot. 2014, 113, 873–885. [Google Scholar] [CrossRef] [PubMed]
- Masunaga, T.; Kubota, D.; Hotta, M.; Wakatsuki, T. Mineral composition of leaves and bark in aluminum accumulators in a tropical rain forest in Indonesia. Soil Sci. Plant Nutr. 1998, 44, 347–358. [Google Scholar] [CrossRef]
- Masunaga, T.; Kubota, D.; Hotta, M.; Wakatsuki, T. Nutritional characteristics of mineral elements in tree species of tropical rain forest, west Sumatra, Indonesia. Soil Sci. Plant Nutr. 1997, 43, 405–418. [Google Scholar] [CrossRef]
- Högberg, P. 15N natural abundance in soil-plant systems. New Phytol. 1997, 137, 179–203. [Google Scholar] [CrossRef]
- Craine, J.M.; Elmore, A.J.; Aidar, M.P.M.; Bustamante, M.; Dawson, T.E.; Hobbie, E.A.; Kahmen, A.; Mack, M.C.; McLauchlan, K.K.; Michelsen, A.; et al. Global patterns of foliar nitrogen isotopes and their relationships with climate, mycorrhizal fungi, foliar nutrient concentrations, and nitrogen availability. New Phytol. 2009, 183, 980–992. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.; Kohl, D.H. N2-fixation in field settings: Estimations based on natural 15N abundance. Aust. J. Plant Physiol. 1986, 13, 699–756. [Google Scholar] [CrossRef]
- Martinelli, L.A.; Piccolo, M.C.; Townsend, A.R.; Vitousek, P.M.; Cuevas, E.; Mcdowell, W.; Robertson, G.P.; Santos, O.C.; Treseder, K. Nitrogen stable isotopic composition of leaves and soil: Tropical versus temperate forests. Biogeochemistry 1999, 46, 45–65. [Google Scholar] [CrossRef]
- Medina, E.; Izaguirre, M.L. N2-fixation in tropical American savannas evaluated by the natural abundance of 15N in plant tissues and soil organic matter. Trop. Ecol. 2004, 45, 87–95. [Google Scholar]
- Nardoto, G.B.; Ometto, J.P.H.B.; Ehleringer, J.R.; Higuchi, N.; Bustamante, M.M.; Martinelli, L.A. Understanding the influences of spatial patterns on N availability within the Brazilian Amazon forest. Ecosystems 2008, 11, 1234–1246. [Google Scholar] [CrossRef]
- Guadarrama, P.; Castillo, S.; Ramos-Zapata, J.A.; Hernández-Cuevas, L.V.; Camargo-Ricalde, S.L. Arbuscular mycorrhizal fungal communities in changing environments: The effects of seasonality and anthropogenic disturbance in a seasonal dry forest. Pedobiologia 2014, 57, 87–95. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).