Abstract
Successful tree regeneration is essential for sustainable forest management, yet it can be limited by the interaction of harvesting effects and multiple ecological drivers. In northern hardwood forests, for example, there is uncertainty whether low-intensity selection harvesting techniques will result in adequate and desirable regeneration. Our research is part of a long-term study that tests the hypothesis that a silvicultural approach called “structural complexity enhancement” (SCE) can accelerate the development of late-successional forest structure and functions. Our objective is to understand the regeneration dynamics following three uneven-aged forestry treatments with high levels of retention: single-tree selection, group selection, and SCE. Regeneration density and diversity can be limited by differing treatment effects on or interactions among light availability, competitive environment, substrate, and herbivory. To explore these relationships, manipulations and controls were replicated across 2 ha treatment units at two Vermont sites. Forest inventory data were collected pre-harvest and periodically over 13 years post-harvest. We used mixed effects models with repeated measures to evaluate the effect of treatment on seedling and sapling density and diversity (Shannon–Weiner H’). The treatments were all successful in recruiting a sapling class with significantly greater sapling densities compared to the controls. However, undesirable and prolific beech (Fagus americana) sprouting dominates some patches in the understory of all the treatments, creating a high degree of spatial variability in the competitive environment for regeneration. Multivariate analyses suggest that while treatment had a dominant effect, other factors were influential in driving regeneration responses. These results indicate variants of uneven-aged systems that retain or enhance elements of stand structural complexity—including old-growth characteristics—can generally foster abundant regeneration of important late successional tree species depending on site conditions, but they may require beech control where beech sprouting inhibits desired regeneration.
1. Introduction
Forest management for old-growth characteristics provides an important opportunity to help mitigate the effects of climate change, while providing many social, economic, and ecological benefits. The approach is to manage for high biomass stand structures such as those often found in old-growth forests [1,2]. This could entail, for example, emulating—through various retention forestry techniques—the type of natural disturbances and stand development processes leading to the development of high biomass conditions [3,4]. Prior to European settlement, forests in the northeastern U.S. were dominated by relatively frequent, gap-forming and partial disturbances that created a finely patterned mosaic of successional conditions [5,6]. These were dominated by late successional/old-growth stand structures [7], providing higher levels of some ecosystem functions, in comparison to the younger, secondary forests that predominate today.
There are a number of old-growth attributes indicative of stand structural complexity in northern hardwoods that could be promoted through retention forestry [8,9,10,11]. These include greater availability of large downed and standing woody debris (snags), large trees, tip-up mounds, horizontal variation in stand density (e.g., gaps of varying sizes and shapes), vertically complex canopies, and advanced regeneration [2,12,13]. In managing for structural complexity, silviculturists might promote a variety of ecological functions, including habitat for late-successionally-associated wildlife, hydrologic regulation [14], and increased carbon storage potential [15,16,17]. Structural retention systems are of great interest as a means for providing a broad array of biodiversity and ecosystem services in managed forests [18,19,20], but successful regeneration outcomes are required for wider adoption by the forestry profession. For example, in the northern hardwood region of eastern North America, the increased canopy cover and shade associated with high levels of retention, particularly in the context of selection harvesting systems, pose challenges for regenerating economically and ecologically desirable tree species, even though many of these are shade tolerant [21]. Our research investigates the question of whether a silvicultural practice promoting old-growth characteristics, termed “Structural Complexity Enhancement (SCE),” can regenerate desirable tree species and establish a new cohort of saplings at sufficient densities to be sustainable in comparison to conventional uneven-aged prescriptions. SCE was specifically designed to accelerate the development of old-growth forest structure and function, targeting stand structure attributes derived from previous research on old-growth forests in the U.S. Northeast [8,12,22]. Effects on stand structure [6,9], elements of biodiversity [23,24,25], and carbon cycling [17] have been explored previously. Research has shown that Structural Complexity Enhancement can increase herbaceous understory plant diversity [24], terrestrial amphibian populations [23], and fungal species richness [25], while enhancing stand structural complexity and promoting late-successional functions such as carbon storage [9]. However, no study has tested the effect of Structural Complexity Enhancement on the regeneration abundance and diversity in northern hardwood forests. Additionally, we are interested in whether the regeneration responses are influenced by other sources of variability, such as herbivory, substrate, light intensity, and climate, that might interact with treatment effects.
Regeneration remains a key source of uncertainty in predicting the long-term viability and efficacy of structural complexity treatments (e.g., [26,27,28]), including old-growth silviculture [11,18]. Challenges to successful regeneration of desirable species in selection harvesting, including gap-based silviculture, go beyond the light environment and include seed availability, desirable substrate, and competition with both herbaceous cover and advanced regeneration [6]. Herbivory by white-tailed deer (Odocoileus virginianus), moose (Alces alces), and eastern cottontail rabbit (Sylvilagus floridanus) can strongly influence regeneration responses and redirect compositional development [29,30,31]. Moreover, tree regeneration in the Northeast is increasingly limited by competition with American beech (Fagus grandifolia) due to its high shade tolerance and vegetative sprouting response to beech bark disease (BBD, Nectria coccinea var. faginata), causing many [21] to be skeptical of the ability of single-tree selection harvesting systems to regenerate desirable shade tolerant (e.g., sugar maple (Acer saccharum) and intermediate tolerant (e.g., yellow birch (Betula alleghaniensis)) species. The vigorous root sprouting mechanism of beech leads to the formation of dense beech thickets [32]. Beech thickets alter stand species composition through competition for light and other resources, while providing a substrate for BBD to spread from older diseased trees to the sapling class [33]. Due to the uncertainty of beech regeneration effects on the understory competitive environment, we are particularly interested in the recruitment response of common beech associates, such as sugar maple and yellow birch. Regeneration of sugar maple and yellow birch following harvesting tends to be spatially and temporally variable and dependent on interactions among multiple factors. Harvest gaps, as well as soil scarification and deadwood availability, can positively influence yellow birch establishment [34,35]. Sugar maple regenerates well on rich soils with partial canopy cover. Acid deposition can substantially deplete soil calcium on poorly buffered soils and limit sugar maple growth in some areas, another factor that could favor beech over time [36].
Ability to accurately predict regeneration responses to structural retention remains highly limited in northern hardwood-conifer systems. The first objective of this project was to explore the potential of silvicultural techniques retaining and promoting old-growth structure to successfully regenerate desirable tree species. We defined successful as the establishment of seedlings (approx. 12,300/ha minimum as suggested by Nyland [37]) and the recruitment of seedlings into the sapling class. We defined desirable tree species diversity as non-declining species diversity, where the treatments do not negatively influence species diversity from the pre-treatment baseline. The second objective was to examine varying patterns of regeneration among species, specifically American beech and sugar maple. We defined desirable regeneration as including intermediate and shade-tolerant, commercially important (i.e., for wood products and syrup production) species (sugar maple, red maple (Acer rubrum), and yellow birch), at densities sufficient to ensure the development of a new, merchantable cohort. Red maple was included because it is increasingly tapped for maple syrup and utilized for a variety of innovative wood products [38,39]. The third objective was to explore relationships of interest based on a priori knowledge from the regeneration literature, including those that might be modified through silvicultural practices. Relationships of interest that influence regeneration dynamics included interactions between canopy cover, substrate composition, light intensity, herbivory, climate and silvicultural treatments.
2. Materials and Methods
2.1. Study Area
The Vermont Forest Ecosystem Management Demonstration Project (FEMDP) is a study testing the long-term stand dynamics, biodiversity responses, and ecosystem service outcomes of experimental treatments designed to emulate fine-scale natural disturbance effects and promote development of late-successional/old-growth characteristics. The two study areas are located in Vermont, USA at the Mount Mansfield State Forest (MMSF) and the University of Vermont’s Jericho Research Forest (JRF, Figure 1, Table 1).

Figure 1.
Regional map with locations of the two project study sites: Jericho Research Forest (A) and Mt. Mansfield State Forest (B). Also shown are treatment unit layout maps of the two study areas. Mansfield treatment manipulations: Units 1 and 8, Control; 2–3, Structural Complexity Enhancement 4–5, Single-Tree Selection; 6–7 Group Selection. Jericho manipulations: 1 and 4, Control; 2–3 Structural Complexity Enhancement.

Table 1.
Site characteristics of experimental treatment units located in the Mansfield and Jericho study areas of the Forest Ecosystem Management Demonstration Project Adapted from Ford and Keeton (in press).
Both sites are mature (ca. 70–100 years), multi-aged northern hardwood-conifer forests with a documented history of timber management [9,24,40]. The Jericho Research Forest (44°26′43.70″N; 72°59′44.15″W) is located in the foothills of the Green Mountain Range, a northern extension of the Appalachian Range, at 200 to 250 m above sea level. Soils are Adams and Windsor loamy sands or sandy loams. The Mount Mansfield State Forest (44°30′23″ N; 72°50′11″ W) is on the western slopes of the Green Mountain Range. Elevations at the study area range from 470 to 660 m above sea level. Soils are primarily Peru extremely stony loams. The overstory at both sites is dominated by sugar maple, American beech, and yellow birch. At JRF, eastern hemlock (Tsuga canadensis) is also co-dominant, with minor components of red maple and northern red oak (Quercus rubra). Red spruce (Picea rubens) is a minor element of the canopy at MMSF [9].
2.2. Study Design
The FEMDP investigated three uneven-aged silvicultural techniques with high levels of post-harvest structure such as residual canopy cover, standing dead snags, and downed coarse woody debris. These were assigned to 2 ha treatment units in a randomized block design. At MMSF, the three treatments were all replicated twice; at JRF the SCE treatment was replicated twice. Both sites had two un-manipulated control units. To prevent soil damage and erosion, experimental manipulations (i.e., logging) were conducted in deep snow on frozen ground in the winter (January–February 2003). Marking guidelines specifically targeted retention of disease resistant beech, larger trees showing evidence of wildlife use (e.g., black bear (Ursus americanus) activity, cavity excavation, etc.), and tree species diversity.
Although all three treatments in the FEMDP shared structural retention as an objective, they differed in harvest approach, for example in the degree of uniform vs. variable density tree marking, silvicultural gap size and configuration, and amount of live and dead tree retention [6,9,41]. The treatments included two conventional uneven-aged prescriptions, single-tree selection (STS) and group selection (GS), which retain post-harvest structure at the high end of the range of retention parameters typical for selection harvesting in the region (Table 2). The modifications for single-tree selection set retention targets of 18.4 m2·ha−1 in the residual basal area, a maximum tree diameter of 60 cm at breast height (dbh, 1.37 m), and a q-factor of 1.3 to re-allocate the basal area into larger diameter classes. The group selection treatment followed the same diameter distribution-based prescription, but was applied through spatially aggregated harvesting. The group patches averaged 0.05 ha in size, a prescription intended to emulate the average size of canopy openings created by fine-scale natural disturbance in New England based on work summarized by Seymour et al. [42]. Group openings were variable in shape and some had light retention, consisting primarily of large snags and, occasionally, large diameter beech exhibiting resistance to beech bark disease (Nectria coccinea var. faginata). The treatment resulted in eight to nine groups per treatment unit, with the matrix between groups left unharvested [9].

Table 2.
Silvicultural prescription details for experimental treatment manipulations at the MMSF and JRF study areas. Listed in the table is the target BDq for each treatment The BDq is the residual basal area (B), maximum target diameter (D), and q-factor (q). The q-factor is equal to the ratio of the number trees in each successively larger size class. Adapted from Keeton (2006) and Ford and Keeton (in press).
The third treatment, SCE, employed a rotated sigmoid target (or post-harvest) diameter distribution (see [43,44]) defined by a high residual basal area (34 m2·ha−1), large maximum tree size (90 cm), and a variable q-factor applied to three portions of the diameter distribution (2.0 in the smallest size class, 1.1 in the medium size class, and 1.3 in the largest size class). The combined effect was a re-allocation of the basal area and growing space into larger diameter classes, while retaining abundant stems across all sizes and ages. Other structural objectives included vertically differentiated canopies, higher densities of large snags and downed logs, variable horizontal density including small (mean size of 0.02 ha) gaps, and recruitment of greater densities of large trees. The latter was achieved through partial or full crown release [8]. At one unit in each of the two study areas, trees were pulled (or pushed) over to create the tip-up mounds characteristic of old-growth northern hardwood forests.
2.3. Data Collection
Each 2 ha treatment unit contained five randomly placed 0.1 ha permanent sampling plots. The plots were buffered from edge effects through placement at least 15 m within the interior of unit boundaries and collectively represent 25% of each unit’s total area. For this study, we used one year of pre-treatment (2001) data, six years of post-harvest seedling data (2003, 2004, 2006, 2007, 2013, 2015), and three years of post-harvest sapling data (2003, 2004, 2015). Tree seedlings <1 m in height were identified and counted along two 1-m wide and 31.64 m long belt transects bisecting the 0.1 ha plots. Tree saplings >1 m in height and <5 cm dbh were identified and counted within a plot (0.02 ha) nested within the 0.1 ha plots. All live and dead trees >5 cm dbh within the 0.1 ha sampling plots were tagged, measured, and identified.
Additional variables hypothesized to influence seedling establishment and survival were inventoried in 2015. These included substrate and understory composition, light intensity, herbivory, and stand structure. Herbaceous and woody shrub cover by species was measured using an ocular estimate designed to ensure precision and consistency in estimation [45]. Understory plant cover data were measured with thirteen 1-m2 quadrats placed systematically along plot transects. The substrate data were inventoried using the same quadrats and estimation methodology as the understory data. The substrate data presented here for the purpose of assessing seed beds were categorized as fine litter, mineral soil, and coarse woody debris (>10 cm diameter). For each sapling inventoried in the 0.02 ha sapling plot, branches were assessed for the presence of deer or moose herbivory (browse). Saplings that were unequivocally browsed (i.e., torn, ragged) at any stem height (not distinguishing between moose and deer) were recorded as having the presence of browse [46].
To quantify light conditions in each treatment, hemispheric canopy photography was used to estimate the proportion of direct light (DSF) transmitted through the canopy. The camera was mounted on a tripod with a self-leveling mechanism set at 1-m height and two photographs were taken at the center of each plot. All analyses and photo selections were conducted by the same lab technician to minimize and systematize any error associated with selecting thresholds during analyses. The canopy photographs were processed and analyzed using HemiView canopy analysis software (Delta-T_Devices, Cambridge, UK).
The Palmer Drought Severity Index (PDSI) was obtained for U.S. climatological division 2 (western Vermont) from the Earth System Research Laboratory of the National Oceanic and Atmospheric Administration (NOAA; [47]). The PDSI parameters were compiled from weather stations and averaged to get a PDSI value at a spatial scale much larger than our study area, about 20,000 km2 or half the size of Vermont. Climate data from 2001 to 2013 were used as a coarse estimate of climatic conditions including relative dryness and drought potential. PDSI has been used previously to develop predictions of seedling success under different drought scenarios [48].
2.4. Statistical Analysis
Due to uneven treatment replication among the two study areas, which limited the strength of statistical inference, analyses were performed separately by site. Multivariate analyses were used to determine if treatment had an effect on trends in the regeneration response variables over time. We used Statistical Analysis Software (SAS Institute Inc.) to build a linear mixed effects model (LME) to model the diversity response (Shannon–Weiner H’) of seedling and saplings. Because seedling and sapling abundance estimates were not normally distributed, we used a generalized linear mixed model (GLIMMIX) to model total seedling and sapling densities, as well as sugar maple, red maple, yellow birch and American beech responses over time. To assess regeneration responses of those species (termed “desirable”) for which there is regional concern regarding regeneration success following selection harvesting, we combined sugar maple, red maple, and yellow birch regeneration into a single response variable, termed “SM_RM_YB”. GLIMMIX models use multivariate analyses to test for effects in non-normal data and are most appropriate for analyzing repeated measures [49,50]. We assumed a negative binomial distribution and a first order autoregressive covariance structure for the repeated measures. Treatment and year were modeled as fixed effects, while plots and units were modeled as random effects. Following the model design of Smith et al. [24], plots were nested within units. Using this method, the effective statistical sample size per treatment by site is 10 because plot data are pooled. The model output provided parameter estimates for the fixed effects and covariance estimates for the random effects, as well as a pairwise comparison test of the treatments at each time point to compare mean estimates between the treatments. Pre-treatment (2001) data for regeneration diversity and density were included in the model as a continuous covariate, to normalize data and allow for standardized comparisons among treatments across a range of inherent site variability. Percentage differences between pre-treatment, peak abundance, and thirteen years post-harvest were calculated following Littlefield and Keeton [51], using the Equation (1):
where VH is the post-harvest value and VB is the pre-treatment “baseline” value. Peak abundance occurred 4-years post-treatment for the seedling class and 13-years post-harvest for the sapling class.
Percentage difference = ((VH − VB)/((VH + VB)/2)) × 100
To test for the interaction among ecological variables having a possible influence on tree regeneration, we examined the 13-year post-harvest data set with another generalized linear mixed model. Based on the literature (for example, [52,53,54] and [41]), we chose variables that have been shown to influence regeneration dynamics. Predictor variables included in the model were percent leaf litter, percent browse, Direct Site Factor (DSF, obtained through analysis of hemispheric photographs), and Curtis relative-density (RD) structure index [55]. The model form for seedling density was as follows: seedling density~Percent_Browse + Percent_Fine_Litter + DSF + Curtis_RD + Site + Treatment + Site × Treatment. Collinearity diagnostics showed that covariates used in the model were not collinear, as measured by the variance inflation factor, which was 1.1 or less for all covariates. We chose DSF as a proxy for light conditions; it represents the proportion of direct solar radiation that reaches each plot, relative to that location with no sky obstructions (open canopy). The Curtis-relative density index is an aggregated stand structure index that integrates quadratic mean diameter and stem density. It is a good indicator of canopy retention as it depicts the total occupation of growing space based on tree density and size and can be used for uneven-aged management [55]. In a separate analysis, PDSI was analyzed with the Durbin-Watson statistical test which detects the presence of autocorrelation in the residuals from a time series regression analysis [56]. The Durbin-Watson test resulted in a statistical value which is significant if it falls between a given range of values. We tested the Vermont climate division 2 PDSI data against regeneration density and diversity by treatment separately.
3. Results
3.1. Objective 1: Regeneration Density and Diversity Responses
At Mount Mansfield State Forest (MMSF), the interaction between time and treatment was significant, indicating that the treatment effects changed in different patterns over time (Table 3). Analysis of total seedling density trends over time resulted in different patterns of regeneration by site (Figure 2). At Jericho Research Forest (JRF), the interaction between time and treatment was not significant (p = 0.1283) and SCE did not increase regeneration density significantly over background rates. At MMSF, the three harvested treatments had significantly more seedlings than the control for the first four years post-harvest (p < 0.0001). After 13 years, total seedling density was similar across all treatments. Seedling densities in the Control units were highly variable year to year, exhibiting pulses of recruitment and mortality, such that densities fluctuated from 2,925 seedlings/ha to 92,757 seedlings/ha. Regeneration dynamics in all the harvested treatments showed an initial pulse of recruitment in the first four years, with the percent difference of GS increasing by 103%, STS increasing by 113%, and SCE by 53% from pre-treatment densities in the seedling class. After thirteen years, GS decreased −33%, STS decreased −76%, and SCE decreased −17% from peak abundance to a similar mean seedling density.

Table 3.
Type 3 Tests of Fixed Effects results from the Generalized Linear Mixed Model with total seedling (A) and sapling (B) densities at Mt. Mansfield.
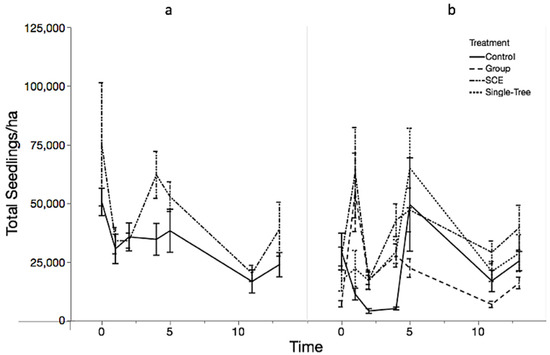
Figure 2.
Total seedlings/ha by treatment over time at JRF (a) and MMSF (b). Error bars represent ±1 standard error from the mean.
While total seedling densities initially increased and then declined to the background rate detected in the Control over the 13-year period, total sapling densities display the opposite trend (Table 4). At MMSF, total sapling densities did not increase significantly over the first two years post-treatment. However, after 13 years, all treatments were successful at regenerating and recruiting a new cohort into the sapling class, at densities significantly greater than the Control (Figure 3, Table 5). Sapling densities in the Control declined over the 13-year time series, while GS, STS, and SCE increased after the initial two-year time lag. Although not significantly different, GS had the highest mean (and standard error), followed by STS and then SCE. According to the percent difference equation, all harvested treatments increased from pre-treatment to year 13, with GS increasing by 79%, STS by 60%, and SCE by 4% at MMSF and 54% at JRF

Table 4.
Summary of seedling and sapling response thirteen years post-harvest at both sites. The table includes the mean of all species combined total stems/ha, minimum, and maximum values; mean beech stems/ha; SM_RM_YB = sugar maple, red maple, yellow birch combined means; and the Shannon–Weiner (H’) value. All means shown with ±1 standard error. N represents the number of plots within the treatments.
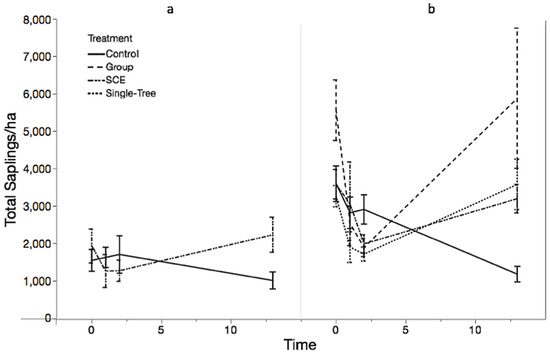
Figure 3.
Total saplings/ha by treatment over time at JRF (a) and MMSF (b). Error bars represent ±1 standard error.

Table 5.
Generalized Linear Mixed Model results from the pairwise comparison of Treatment × Time for total seedling (A) and sapling (B) densities at Mt. Mansfield. Estimate represents the transformed and adjusted stem counts (least squares mean). The outputs shown are immediately post-harvest (2003), and thirteen years post-harvest (2015).
At JRF, although the average sapling density was not significantly different between the Control and SCE at each time point (Time, p = 0.3665), the treatment × time fixed effect was significant (p = 0.0195) because the SCE treatment had an increase in sapling density while the Control had a decrease in sapling density over the 13-year time period. Pre-treatment sapling densities had a significant effect on these trends (p = 0.0079).
We found that there were significant differences in treatment effects on species diversity. At JRF, there was no significant treatment × time effect (p = 0.9373). However, both the Control and SCE increased in seedling diversity (H’), with SCE having slightly higher seedling diversity than the Control at all time intervals. At MMSF, there was a significant treatment × time effect (p = 0.0112) on seedling diversity. The GS treatment almost doubled in seedling diversity over the 13-year time series, while STS had an increase in the first four years and then after 13 years declined to the level immediately post-harvest. The SCE treatment increased seedling diversity from year 1 to year 2 following harvest, and then leveled off, remaining constant to year 13 post-treatment. The Control varied only slightly from year to year, remaining around the same level of seedling diversity and showing no treatment × time effect. In the first year post-harvest, seedling diversity was lowest in the GS, followed by SCE, STS, and Control. After thirteen years, SCE had the highest seedling diversity, followed by the Control, then GS and finally STS (Table 4). Additionally, pre-treatment seedling diversity (p = 0.0012) was highly correlated with the final seedling diversity, indicating that plots that were highly diverse before the treatment maintained high diversity for the following years.
Sapling diversity at JRF declined for both the SCE and Control treatments over the 13-year time series. Although there was no treatment effect, the pre-treatment sapling diversity had a significant influence on the resulting sapling diversity. At MMSF, the pre-treatment sapling diversity also had a significant effect on the resulting sapling diversity. The Control declined in sapling diversity (p = 0.0015) over the 13-year time series, indicating regeneration limitations occurred regardless of the harvest. GS units showed an initial increase in sapling diversity 2 years following the harvest and then declines significantly after 13 years post-harvest (p = 0.0042). SCE performed comparably to STS in regenerating and maintaining a diverse sapling cohort as demonstrated by the insignificant treatment × time effect, indicating that both treatments did not decline in sapling diversity as did the Control and GS units (see Table A1 for full species list).
3.2. Objective 2: Competitive Interactions Among Species
At JRF, beech seedlings had a significant treatment × time effect (p = 0.0216). The Control had a higher mean of beech seedlings for year 1 through year 11 post-harvest; however, the means were not significantly different between the treatments for the entire time series. At MMSF, there was a significant treatment × time effect (p < 0.0001). GS and SCE resulted in seedling recruitment responses that reached similar levels after 13 years, while STS and the Control had similar beech seedling densities from year to year. GS and SCE units had significantly more beech seedlings than STS and Control units, even though GS and SCE both showed decreasing beech seedling densities over the 13 years monitored.
At MMSF, there was a significant treatment × time effect (p = 0.0040) on beech sapling densities. Immediately post-harvest, SCE units had a greater beech sapling density than STS units (p = 0.0257); by year two, none of the treatment means were significantly different. However, after 13 years, the Control had significantly less beech saplings than GS (p = 0.0020) and SCE (p = 0.0244). Following the beech seedling trend at MMSF, GS and SCE displayed similar sapling densities while the Control and STS had similar sapling densities. SCE and STS treatments resulted in beech sapling densities that were not significantly different from each other 13 years post-harvest, while GS resulted in significantly more beech saplings than STS units (p = 0.0089).
At JRF, seedling densities for SM_RM_YB were not significantly different between the Control and SCE (p = 0.0927). At MMSF, there was a significant treatment × time effect (p < 0.0001) on SM_RM_YB, as well as a significant effect from the pre-treatment SM_RM_YB densities (p = 0.0358). After 4 years, the Control units had significantly fewer SM_RM_YB seedlings than the other three treatments; however, by the following year the densities had stabilized at levels that were not significantly different from one another. After 11 years, the GS treatment had significantly fewer of these species in the seedling class than SCE and STS. Following the pattern of total seedling densities, by year 13, all treatment means were not significantly different from each other (Figure 4). Although insignificant, the harvested treatments all have more SM_RM_YB seedlings than the Control. STS had a higher density of SM_RM_YB seedlings than SCE, while SCE had a higher density than GS.
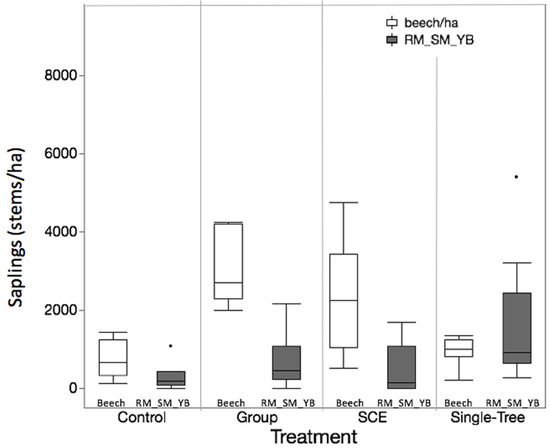
Figure 4.
MMSF treatments thirteen years post-harvest showing beech saplings (white) in comparison to RM_SM_YB (red maple, sugar maple, yellow birch combined) saplings (grey). (Tukey) outlier box plot showing data distribution.
3.3. Objective 3: Interactions with Herbivory, Substrate and Climate
While mean densities of seedlings were similar after 13 years across all the treatments, plot level data showed large spatial variation or patchiness within the treatments (Table 4). Across both sites, the two factors found to have the most significant effect on sapling recruitment were percent browse (herbivory) and percent fine litter substrate (Table 6).

Table 6.
Results from the Generalized Linear Mixed Model show that increasing browse and increasing fine litter substrate both resulted in a decline in sapling diversity (H’) and an increase in beech saplings. Direct Solar Fraction (DSF) and Curtis Relative-Density (RD) were not significant in the model.
Thirteen-years post-harvest, percent browse on saplings had a positive impact on seedling diversity (p = 0.0286) and a negative impact on sapling diversity (p = 0.0403). Increasing percent browse was positively correlated with the density of beech (p = 0.0512) and sugar maple saplings (p = 0.0443).
Percent fine litter had an influence on regeneration response similar to that of percent browse. Our data showed a positive relationship between fine litter substrate and beech sapling density (p = 0.0369), while there was a significantly negative relationship with sapling diversity (p = 0.0213).
Total seedling densities were not significantly correlated with any of the variables presenting possible non-treatment influences on regeneration dynamics. There was a significant treatment effect on total sapling densities (p = 0.0327), indicating that after light conditions (DSF), substrate (fine litter), herbivory, and Curtis-RD were accounted for in the model, the treatment still had the greatest effect on the amount of saplings (Table 6).
The Palmer Drought Severity Index (PDSI) values for northwestern Vermont indicated moist, non-drought conditions for most of the duration of the study. However, in the growing season (June–August) of 2001, the year the study was established, the PDSI was −1.927 which is categorized as a mild drought. This declined to a severe drought with a PDSI of −3.350 in the fall (Septeber–November). PDSI remained high with no drought until 2012 when there was a mild drought that decreased into an incipient (developing) drought in the spring of 2013.
In the Durban–Watson analysis, species diversity (H’) in the SCE treatment at JRF was significantly correlated with PDSI values indicative of high moisture, (DW statistic = 1.056, p = 0.05). None of the other treatments showed a correlation between PDSI and the diversity index. For all the treatments, the total density of seedlings was not significantly correlated with PDSI.
4. Discussion
Silvicultural techniques promoting the development of late-successional/old growth structure in northern hardwood-conifer forests have the potential to regenerate and establish a diverse new cohort of trees, including desirable species, such as sugar maple, red maple, and yellow birch, despite the relatively high levels of structural retention typical of selection harvesting systems. The effects of SCE were highly variable spatially, with successful regeneration in certain patches, while other areas were dominated by dense beech thickets. These results supported the hypothesis that SCE and other silvicultural approaches specifically intended to increase horizontal complexity in stand structure (see [57]), dependent on site conditions and other factors, have the potential to achieve regeneration levels close to or even greater than conventional uneven-aged practices, including selection systems employing larger group openings. They lend support to the efficacy of old-growth silviculture [18] for long-term sustainable management from a regeneration and recruitment perspective.
Yet, as with previous research on selection harvesting [58,59,60], regeneration success was clearly mixed and strongly affected by competition with beech sprouting especially. Regeneration of desirable species, in particular, ranged among patches and sites from absent or poor to very abundant.
4.1. Regeneration Response to Old-Growth Management
We found variants of uneven-aged silviculture systems designed to enhance structural complexity and old-growth characteristics adequately regenerate late-successional tree species, depending on site conditions and patch-scale controls. Although the seedling and sapling response was not as abundant as that from conventional even-aged systems, the total regeneration density was sufficient to reach full stocking according to the minimum threshold recommended by Nyland [37]. Regeneration dynamics in all the harvested treatments showed an initial pulse of recruitment in the seedling class. After thirteen years, all treatments returned to a similar mean seedling density. The decline from peak abundance (achieved in year 4 post-treatment) in seedling density can be attributed to a combination of factors, including partial canopy closure and reduced light availability, competition and density-dependent mortality, and recruitment into the sapling class [61]. The Control units at JRF had regeneration occurring in natural gaps thirteen-years post-harvest which could explain the lack of difference in regeneration between the Control and SCE treatments at that site. The treatments were all successful in recruiting a sapling class over the thirteen years post-harvest. However, 62% of the sapling layer was made up of dense beech (1843/ha on average) thickets after 13 years, a trend we found across all treatments. Beech thickets were most abundant in the GS treatment, intermediate in the SCE treatment, and least prevalent in the STS treatment.
Species diversity in the seedling class increased across all harvested treatments. Pre-treatment species diversity was as predictive as treatment type, suggesting a strong influence of initial site conditions and past stand development history on community composition. At the FEMDP sites, the spatially heterogeneous regeneration pattern for some species, such as sugar maple, red maple, and yellow birch, is clearly influenced both by light availability and competition with beech thickets, the latter being present in all treatments. Beech seedlings in all treatments showed a very large initial increase immediately post-harvest and then declined after a decade, most likely due to recruitment into the sapling class.
Previous studies have found that maple and beech tend to replace each other in stands uninfected with beach bark disease in the Upper Lake states, with saplings often occurring beneath the canopy of the other species [62,63]. Both beech and sugar maple have been found to outcompete one another following a disturbance or harvest, depending on site conditions. Sugar maple typically has faster growth rates than beech under higher light conditions in gaps (e.g., [64,65,66]). However, due to asexual reproduction through root sprouting and shared belowground nutritional supply, beech can often outcompete sugar maple in the understory, ultimately overtopping sexually reproduced maple [67,68,69]. In addition to outcompeting sugar maple, beech produces abundant beech leaf litter which contains leachate with phytotoxins that inhibit the development of sugar maple seedlings [70].
4.2. Sources of Variability in Regeneration Dynamics
Consistent with previous reports from northern hardwood forests, regeneration limitations in our study were linked to multiple factors including competition from dense beech understories and deer browse [71,72]. Adding the ecological variables into the linear mixed model showed that increasing herbivory and leaf litter were negatively associated with sapling diversity and positively associated with beech sapling densities. There was a positive association between herbivory and seedling diversity, found also in previous studies when deer populations are about 3–6 deer/km2 [73]. The positive impact of browse on seedling diversity could be a result of disproportionate browse on common, palatable species giving rare species seedlings a competitive advantage [74]. This effect may be short-lived as seedlings move into the sapling class and there is greater competition for resources. Although beech is palatable to deer, it is less preferentially browsed in comparison to yellow birch and sugar maple. Browse and litter substrate were positively associated with beech sapling density at the FEMDP sites. Browse pressure (deer, moose, and rabbit) was highest in GS (ranging from 80% to 97%), intermediate in SCE (43%–92%), and lowest in STS (45%–91%).
In the FEMDP experiment, the SCE treatment initially increased coarse woody debris (CWD) volumes by 140% [9], though these later declined due to decay [37]. In addition to mineral soil, partially decayed softwood coarse woody debris is a preferred seedbed for yellow birch establishment and is a substrate that provides a competitive advantage for birch by altering the sapling morphology (i.e., multilayered crowns) for improved shade-tolerance [35,75]. Beech control in the treatment units could allow established yellow birch saplings to grow into merchantable size classes, such as on the CWD substrate.
4.3. Effects of Climate Variability and Drought
Although we did not see a significant treatment effect on seedling diversity at JRF, there was a relationship between climate and SCE seedling diversity. This implies that there may be an influence of drought stress, as measured by PDSI, on regeneration trends in the structural complexity enhancement treatment [48]. Mild spring drought conditions in recent years could influence early seedling establishment, potentially causing a decline in regeneration diversity. In the winter of 2012, 2013, and 2015 the PDSI indicated an incipient drought, which could relate to lower amounts of precipitations in the form of snow. A decline in snow depth can lead to decreased seedling survival due to increased browse and soil freezing stresses [76,77]. Increased sapling mortality has also been associated with root exposure due to declining snow pack [78].
Climate change models predict that increased temperatures and higher drought deficits will influence regeneration dynamics in the eastern U.S. [79,80,81]. Some models suggest that now dominant northern hardwood and conifer species may decline due to water deficits [82]. However, sugar maple, red maple, yellow birch, and American beech, surprisingly, showed enhanced seedling survival with increasing water deficit between 25 mm and 625 mm across their entire range [81]. These may be transient effects; if conditions become too dry or too wet seedling mortality may increase [83,84]. However, maintaining tree species diversity, as the FEMDP treatments did, at stand and landscape scales is an important strategy conferring ecosystem resilience to climate related stresses [19].
5. Management Implications
Our research suggests that management for old-growth characteristics in northern hardwood-conifer forests can promote adequate regeneration; however, site specific modifications are recommended for successful recruitment of species other than beech. Some silviculturists recommend patch- or clear-cutting to prevent prolific beech regeneration [21]. However, in our study, beech sprouting is most abundant in the group selection openings, even though advanced beech regeneration was cut (or cleaned) at the time of harvest, suggesting that more aggressive beech control would be needed for a desirable regeneration outcome. Based on our results, we suggest that low-intensity selection systems can regenerate at desirable densities [37] but may require beech control to sustain a diversity of intermediate- and shade-tolerant species. All the treatments resulted in total seedling densities above the minimum recommended by regional silvicultural guides. If regenerating commercially valuable species is the primary management objective, alternative silvicultural systems may be preferable. However, where timber is integrated with other management objectives, such as late-successional wildlife habitat, non-timber forest products, riparian functionality, or carbon storage, the regeneration densities and mixed composition resulting from selection systems that enhance structural complexity might be considered acceptable.
Removal of beech saplings alone may not be sufficient to promote sugar maple seedling establishment under selection harvesting [66]. Precutting of beech saplings and repeated cleaning entries post-harvest are potential applications of beech control [52]. Repeated cutting of beech before overstory harvest can still promote beech suckering; however, beech regeneration may remain shorter than sugar maple and yellow birch [52]. As an alternative, several studies have tested limited herbicide treatments (e.g., [85,86]) and found them to be highly effective. Broad range application of glyphosate or triclopyr in the summer months is the most efficient and most effective treatment suggested by Nyland et al. [52] based on a literature review of experimental treatments to eliminate understory interferences. Glyphosate treatments were also found to be successful at maintaining desirable species such as sugar maple and yellow birch, while minimizing American beech in a northern Maine study [87]. However, herbicide application carries risks that some managers may find unacceptable. Combined beech removal and deer exclusion, though expensive and thus often not practical, offers another means for improving regeneration outcomes [88].
Gap size can strongly influence regeneration dynamics [89]. For example, a study of northern hardwoods in Minnesota found that gaps greater than 0.02 ha had the highest regeneration densities [90]. Another long-term silvicultural study found better sugar maple establishment and survival under single-tree selection in comparison to large and medium sized group-selection openings, while yellow birch seedlings followed the reverse trend, with higher densities in larger gaps compared to smaller gaps [61]. However, these study sites were uninfected with beech bark disease. Canopy gaps increase light levels which is thought to increase species diversity [91]. However, other sources of variability may lead to unanticipated regeneration responses to gap-based silviculture [54,61,90]. These include seed availability, presence or absence of a seedbank, and seed predation [6]. As a consequence, the regeneration objective remains challenging. However, in a review of gap-based silviculture, Kern et al. [6] suggest moving towards a continuum of gap sizes, shapes, and within-gap retention, emulating the complexity associated with partial disturbances. We support this suggestion based on our findings, in which spatially complex regeneration responses likely reflected, in part, the varied light environments created by canopy openings that differed in size among the treatments. Therefore, we recommend utilizing a range of gap sizes, with smaller gaps on better quality sites and larger gaps incorporating beech control on poorer sites.
Managing forests for old-growth and late-successional characteristics is an option that some managers are considering that would provide a broader range of habitat conditions and ecosystem service co-benefits approaches [9,11,18,92]. Our research shows that SCE, like other retention-based systems, is sustainable from a regeneration perspective and thus provides a viable approach that could be integrated into holistic forest management.
Acknowledgments
This research would not have been possible without support from the Northeastern States Research Cooperative. Prior support for the FEMDP was provided by the Vermont Monitoring Cooperative, the USDA McIntire-Stennis Forest Research program, and the USDA National Research Initiative. Thanks especially to Timothy Perkins, and Anthony D’Amato for extensive input on this paper. Valuable reviews were also provided by Garrett Meigs, Sarah Ford, Andrea Urbano, and the UVM Carbon Dynamics Laboratory. Finally, we wish to thank Alan Howard for assistance with statistical modeling and analysis.
Author Contributions
The study was originally designed and implemented by W.S.K., who also coordinated early stages of data collection. Latter stages of data collection, as well as the data analysis, were conducted by A.J.G. Both authors contributed to writing and editing the paper.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix

Table A1.
Seedling and sapling densities thirteen years post-harvest by species, treatment means (±1 standard error). In Group, Control, SCE the n = 10, in Single-Tree the n = 9.
| Site | JRF | MMSF | |||||
|---|---|---|---|---|---|---|---|
| Treatment | Control | SCE | Control | Group | SCE | Single-Tree | |
| Seedling | |||||||
| Striped maple | 727 ± 421 | 1471 ± 424 | 885 ± 220 | 269 ± 82 | 617 ± 267 | 422 ± 115 | |
| Red maple | 9899 ± 3826 | 22,359 ± 9863 | 32 ± 32 | 95 ± 63 | 1439 ± 1184 | 53 ± 37 | |
| Sugar maple | 3052 ± 1278 | 4206 ± 2084 | 15,591 ± 4930 | 4096 ± 2357 | 18,770 ± 9065 | 19,889 ± 8417 | |
| Mountain maple | 0 ± 0 | 0 ± 0 | 395 ± 195 | 127 ± 66 | 300 ± 166 | 158 ± 83 | |
| Yellow birch | 4254 ± 2298 | 1407 ± 838 | 6120 ± 1301 | 7005 ± 1390 | 9614 ± 1953 | 6378 ± 1665 | |
| Sweet birch | 0 ± 0 | 0 ± 0 | 0 ± 0 | 47 ± 47 | 0 ± 0 | 0 ± 0 | |
| Paper birch | 0 ± 0 | 0 ± 0 | 63 ± 35 | 79 ± 26 | 79 ± 49 | 88 ± 60 | |
| Bitternut hickory | 0 ± 0 | 0 ± 0 | 364 ± 313 | 237 ± 237 | 1059 ± 621 | 35 ± 35 | |
| American beech | 3004 ± 1382 | 1850 ± 464 | 1265 ± 294 | 3447 ± 553 | 6578 ± 1983 | 1125 ± 289 | |
| White ash | 901 ± 329 | 2325 ± 720 | 32 ± 21 | 348 ± 196 | 680 ± 236 | 141 ± 81 | |
| Hophornbeam | 1344 ± 680 | 2625 ± 1078 | 16 ± 16 | 0 ± 0 | 0 ± 0 | 0 ± 0 | |
| Red spruce | 0 ± 0 | 0 ± 0 | 411 ± 177 | 174 ± 60 | 158 ± 91 | 193 ± 51 | |
| White pine | 32 ± 21 | 79 ± 35 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | |
| Pin cherry | 0 ± 0 | 16 ± 16 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | |
| Black cherry | 0 ± 0 | 0 ± 0 | 32 ± 21 | 16 ± 16 | 32 ± 32 | 105 ± 46 | |
| Red oak | 63 ± 35 | 1502 ± 346 | 0 ± 0 | 0 ± 0 | 47 ± 34 | 0 ± 0 | |
| Eastern hemlock | 411 ± 283 | 885 ± 385 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | |
| Sapling | |||||||
| Striped maple | 20 ± 14 | 296 ± 84 | 80 ± 36 | 176 ± 75 | 396 ± 90 | 720 ± 127 | |
| Sugar maple | 80 ± 58 | 4 ± 4 | 56 ± 25 | 4 ± 4 | 116 ± 58 | 98 ± 46 | |
| Mountain maple | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 18 ± 18 | |
| Yellow birch | 12 ± 12 | 12 ± 9 | 236 ± 93 | 648 ± 205 | 360 ± 160 | 1564 ± 559 | |
| Sweet birch | 0 ± 0 | 0 ± 0 | 0 ± 0 | 8 ± 5 | 0 ± 0 | 0 ± 0 | |
| American beech | 768 ± 238 | 1596 ± 501 | 736 ± 144 | 4788 ± 1831 | 2240 ± 433 | 964 ± 117 | |
| White pine | 8 ± 8 | 12 ± 12 | 0 ± 0 | 8 ± 5 | 16 ± 11 | 0 ± 0 | |
| Hophornbeam | 8 ± 8 | 244 ± 192 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | |
| Red spruce | 0 ± 0 | 0 ± 0 | 96 ± 36 | 272 ± 68 | 100 ± 26 | 244 ± 74 | |
| Black cherry | 0 ± 0 | 0 ± 0 | 0 ± 0 | 8 ± 8 | 0 ± 0 | 0 ± 0 | |
| Eastern hemlock | 140 ± 68 | 96 ± 46 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | |
References
- Nunery, J.S.; Keeton, W.S. Forest Carbon Storage in the Northeastern United States: Net Effects of Harvesting Frequency, Post-Harvest Retention, and Wood Products. For. Ecol. Manag. 2010, 259, 1363–1375. [Google Scholar] [CrossRef]
- Burrascano, S.; Keeton, W.S.; Sabatini, F.M.; Blasi, C. Commonality and Variability in the Structural Attributes of Moist Temperate Old-Growth Forests: A Global Review. For. Ecol. Manag. 2013, 291, 458–479. [Google Scholar] [CrossRef]
- Franklin, J.F.; Lindenmayer, D.B.; MacMahon, J.A.; McKee, A.; Magnuson, J.; Perry, D.A.; Waide, R.; Foster, D. Threads of Continuity. Conserv. Pract. 2000, 1, 8–17. [Google Scholar] [CrossRef]
- North, M.P.; Keeton, W.S. Emulating Natural Disturbance Regimes: An Emerging Approach for Sustainable Forest Management. In Patterns and Processes in Forest Landscapes; Lafortezza, R., Sanesi, G., Chen, J., Crow, T.R., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 341–372. [Google Scholar]
- Cogbill, C.V.; Burk, J.; Motzkin, G. The Forests of Presettlement New England, USA: Spatial and Compositional Patterns Based on Town Proprietor Surveys. J. Biogeogr. 2002, 29, 1279–1304. [Google Scholar] [CrossRef]
- Kern, C.; Burton, J.I.; Raymond, P.; D’Amato, A.W.; Keeton, W.S.; Royo, A.A.; Walters, M.B.; Webster, C.R.; Willis, J.L. Challenges Facing Gap-Based Silviculture and Possible Solutions for Mesic Northern Forests in North America. Forestry 2017, 90, 4–17. [Google Scholar] [CrossRef]
- Lorimer, C.G.; White, A.S. Scale and Frequency of Natural Disturbances in the Northeastern US: Implications for Early Successional Forest Habitats and Regional Age Distributions. For. Ecol. Manag. 2003, 185, 41–64. [Google Scholar] [CrossRef]
- Singer, M.T.; Lorimer, C.G. Crown Release as a Potential Old-Growth Restoration Approach in Northern Hardwoods. Can. J. For. Res. 1997, 27, 1222–1232. [Google Scholar] [CrossRef]
- Keeton, W.S. Managing for Late-Successional/Old-Growth Characteristics in Northern Hardwood-Conifer Forests. For. Ecol. Manag. 2006, 235, 129–142. [Google Scholar] [CrossRef]
- Hanson, J.J.; Lorimer, C.G.; Halpin, C.R.; Palik, B.J. Ecological Forestry in an Uneven-Aged, Late-Successional Forest: Simulated Effects of Contrasting Treatments on Structure and Yield. For. Ecol. Manag. 2012, 270, 94–107. [Google Scholar] [CrossRef]
- Fassnacht, K.S.; Bronson, D.R.; Palik, B.J.; D’Amato, A.W.; Lorimer, C.G.; Martin, K.J. Accelerating the Development of Old-Growth Characteristics in Second-Growth Northern Hardwoods; General Technical Report; U.S. Department of Agriculture, Forest Service, Northern Research Station: Newtown Square, PA, USA, 2015.
- McGee, G.G.; Leopold, D.J.; Nyland, R.D. Structural Characteristics of Old-growth, Maturing, and Partially Cut Northern Hardwood Forests. Ecol. Appl. 1999, 9, 1316–1329. [Google Scholar] [CrossRef]
- Keeton, W.S.; Kraft, C.E.; Warren, D.R. Mature and Old-Growth Riparian Forests: Structure, Dynamics, and Effects on Adirondack Stream Habitats. Ecol. Appl. 2007, 17, 852–868. [Google Scholar] [CrossRef] [PubMed]
- Wirth, C.; Messier, C.; Bergeron, Y.; Frank, D.; Fankhänel, A. Old-Growth Forest Definitions: A Pragmatic View. In Old-Growth Forests; Springer: Berlin/Heidelberg, Germany, 2009; pp. 11–33. [Google Scholar]
- Keeton, W.S.; Whitman, A.A.; McGee, G.C.; Goodale, C.L. Late-Successional Biomass Development in Northern Hardwood-Conifer Forests of the Northeastern United States. For. Sci. 2011, 57, 489–505. [Google Scholar]
- McGarvey, J.C.; Thompson, J.R.; Epstein, H.E.; Shugart, H.H. Carbon Storage in Old-growth Forests of the Mid-Atlantic: Toward Better Understanding the Eastern Forest Carbon Sink. Ecology 2015, 96, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Ford, S.E.; Keeton, W.S. Enhanced Carbon Storage through Management for Old-Growth Characteristics in Northern Hardwood-Conifer Forests. Ecosphere 2016, in press. [Google Scholar]
- Bauhus, J.; Puettmann, K.; Messier, C. Silviculture for Old-Growth Attributes. For. Ecol. Manag. 2009, 258, 525–537. [Google Scholar] [CrossRef]
- D’Amato, A.W.; Bradford, J.B.; Fraver, S.; Palik, B.J. Forest Management for Mitigation and Adaptation to Climate Change: Insights from Long-Term Silviculture Experiments. For. Ecol. Manag. 2011, 262, 803–816. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; Franklin, J.F.; Lõhmus, A.; Baker, S.C.; Bauhus, J.; Beese, W.; Brodie, A.; Kiehl, B.; Kouki, J.; Pastur, G.M.; et al. A Major Shift to the Retention Approach for Forestry Can Help Resolve Some Global Forest Sustainability Issues: Retention Forestry for Sustainable Forests. Conserv. Lett. 2012, 5, 421–431. [Google Scholar] [CrossRef]
- Leak, W.B.; Yamasaki, M.; Holleran, R. Silvicultural Guide for Northern Hardwoods in the Northeast; USDA: Washington, DC, USA, 2014.
- Ziegler, S.S. Disturbance Regimes of Hemlock-Dominated Old-Growth Forests in Northern New York, USA. Can. J. For. Res. 2002, 32, 2106–2115. [Google Scholar] [CrossRef]
- McKenny, H.C.; Keeton, W.S.; Donovan, T.M. Effects of Structural Complexity Enhancement on Eastern Red-Backed Salamander (Plethodon cinereus) Populations in Northern Hardwood Forests. For. Ecol. Manag. 2006, 230, 186–196. [Google Scholar] [CrossRef]
- Smith, K.J.; Keeton, W.S.; Twery, M.J.; Tobi, D.R. Understory Plant Responses to Uneven-Aged Forestry Alternatives in Northern Hardwood-Conifer Forests. Can. J. For. Res. 2008, 38, 1303–1318. [Google Scholar] [CrossRef]
- Dove, N.C.; Keeton, W.S. Structural Complexity Enhancement Increases Fungal Species Richness in Northern Hardwood Forests. Fungal Ecol. 2015, 13, 181–192. [Google Scholar] [CrossRef]
- Price, D.T.; Zimmermann, N.E.; van der Meer, P.J.; Lexer, M.J.; Leadley, P.; Jorritsma, I.T.M.; Schaber, J.; Clark, D.F.; Lasch, P.; McNulty, S.; et al. Regeneration in Gap Models: Priority Issues for Studying Forest Responses to Climate Change. Clim. Chang. 2011, 51, 475–508. [Google Scholar] [CrossRef]
- Dodson, E.K.; Burton, J.I.; Puettmann, K.J. Multiscale Controls on Natural Regeneration Dynamics after Partial Overstory Removal in Douglas-Fir Forests in Western Oregon, USA. For. Sci. 2014, 60, 953–961. [Google Scholar] [CrossRef]
- Kneeshaw, D.; Bergeron, Y. Applying Knowledge of Natural Disturbance Regimes to Develop an Ecosystem Management Approach in Forestry. In Ecological Forest Management Handbook; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Augustine, D.J.; McNaughton, S.J. Ungulate Effects on the Functional Species Composition of Plant Communities: Herbivore Selectivity and Plant Tolerance. J. Wildl. Manag. 1998, 62, 1165–1183. [Google Scholar] [CrossRef]
- Collins, B. Ground Layer Competition and Herbivory Effects on Cherrybark Oak (Quercus pagoda Raf.) Regeneration in Experimental Canopy Gaps. J. Torrey Bot. Soc. 2003, 130, 147–157. [Google Scholar] [CrossRef]
- Andreozzi, H.A.; Pekins, P.J.; Langlais, M.L. Impact of Moose Browsing on Forest Regeneration in Northeast Vermont. Alces J. Devoted Biol. Manag. Moose 2014, 50, 67–79. [Google Scholar]
- Houston, D.R. Beech Bark Disease—The Aftermath Forests Are Structured for a New Outbreak. J. For. 1975, 73, 660–663. [Google Scholar]
- Giencke, L.M.; Dovčiak, M.; Mountrakis, G.; Cale, J.A.; Mitchell, M.J. Beech Bark Disease: Spatial Patterns of Thicket Formation and Disease Spread in an Aftermath Forest in the Northeastern United States. Can. J. For. Res. 2014, 44, 1042–1050. [Google Scholar] [CrossRef]
- Gauthier, M.-M.; Lambert, M.-C.; Bédard, S. Effects of Harvest Gap Size, Soil Scarification, and Vegetation Control on Regeneration Dynamics in Sugar Maple-Yellow Birch Stands. For. Sci. 2016, 62, 237–246. [Google Scholar] [CrossRef]
- Lambert, J.-B.; Ameztegui, A.; Delagrange, S.; Messier, C. Birch and Conifer Deadwood Favour Early Establishment and Shade Tolerance in Yellow Birch Juveniles Growing in Sugar Maple Dominated Stands. Can. J. For. Res. 2016, 46, 114–121. [Google Scholar] [CrossRef]
- Huggett, B.A.; Schaberg, P.G.; Hawley, G.J.; Eagar, C. Long-Term Calcium Addition Increases Growth Release, Wound Closure, and Health of Sugar Maple (Acer saccharum) Trees at the Hubbard Brook Experimental Forest. Can. J. For. Res. 2007, 37, 1692–1700. [Google Scholar] [CrossRef]
- Nyland, R.D. Silviculture: Concepts and Applications; McGraw-Hill Series in Forest Resources; Waveland Press: Long Grove, IL, USA, 2007. [Google Scholar]
- Blankenhorn, P.R.; Labosky, P., Jr.; Janowiak, J.J.; Manbeck, H. The Development of Preservation Treatment Recommendations for Red Oak and Red Maple Glued-Laminated Timber Bridge Members. For. Prod. J. 1999, 49, 87. [Google Scholar]
- Wang, X.; Ross, R.J.; Green, D.W.; Brashaw, B.; Englund, K.; Wolcott, M. Stress Wave Sorting of Red Maple Logs for Structural Quality. Wood Sci. Technol. 2004, 37, 531–537. [Google Scholar] [CrossRef]
- Hannah, P.R. Species Composition and Dynamics in Two Hardwood Stands in Vermont: A Disturbance History. For. Ecol. Manag. 1999, 120, 105–116. [Google Scholar] [CrossRef]
- D’Amato, A.W.; Catanzaro, P.F.; Fletcher, L.S. Early Regeneration and Structural Responses to Patch Selection and Structural Retention in Second-Growth Northern Hardwoods. For. Sci. 2015, 61, 183–189. [Google Scholar] [CrossRef]
- Seymour, R.S.; White, A.S.; deMaynadier, P.G. Natural Disturbance Regimes in Northeastern North America—Evaluating Silvicultural Systems Using Natural Scales and Frequencies. For. Ecol. Manag. 2002, 155, 357–367. [Google Scholar] [CrossRef]
- Goff, F.G.; West, D. Canopy-Understory Interaction Effects on Forest Population Structure. For. Sci. 1975, 21, 98–108. [Google Scholar]
- Goodburn, J.M.; Lorimer, C.G. Population Structure in Old-Growth and Managed Northern Hardwoods: An Examination of the Balanced Diameter Distribution Concept. For. Ecol. Manag. 1999, 118, 11–29. [Google Scholar] [CrossRef]
- Peet, R.K.; Wentworth, T.R.; White, P.S. A Flexible, Multipurpose Method for Recording Vegetation Composition and Structure. Castanea 1998, 63, 262–274. [Google Scholar]
- Faison, E.K.; Motzkin, G.; Foster, D.R.; McDonald, J.E. Moose Foraging in the Temperate Forests of Southern New England. Northeast. Nat. 2010, 17, 1–18. [Google Scholar] [CrossRef]
- Create a monthly or seasonal time series of climate variables. Available online: https://www.esrl.noaa.gov/psd/data/timeseries/ (accessed on 1 May 2016).
- Zwolinski, J.; Donald, D.; Van Laar, A.; Groenewald, W. Regeneration Procedures of Pinus radiata in the Southern Cape Province: Part V: Post Planting Mortality and Growth of Trees in Response to the Experimental Treatments and Planting Site Environment. S. Afr. For. J. 1994, 168, 7–21. [Google Scholar]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.-S.S. Generalized Linear Mixed Models: A Practical Guide for Ecology and Evolution. Trends Ecol. Evol. 2009, 24, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Zuur, A.F.; Ieno, E.N.; Smith, G.M. Analysing Ecological Data. Statistics for Biology and Health; Springer: New York, NY, USA; London, UK, 2007. [Google Scholar]
- Littlefield, C.E.; Keeton, W.S. Bioenergy Harvesting Impacts on Ecologically Important Stand Structure and Habitat Characteristics. Ecol. Appl. 2012, 22, 1892–1909. [Google Scholar] [CrossRef] [PubMed]
- Nyland, R.D.; Bashant, A.L.; Bohn, K.K.; Verostek, J.M. Interference to Hardwood Regeneration in Northeastern North America: Controlling Effects of American Beech, Striped Maple, and Hobblebush. North. J. Appl. For. 2006, 23, 122–132. [Google Scholar]
- Ward, J.; Worthley, T.E.; Smallidge, P.J.; Bennet, K.P. Northeastern Forest Regeneration Handbook; USDA: Newtown Square, PA, USA, 2013.
- Kern, C.; Reich, P.B.; Montgomery, R.A.; Strong, T.F. Do Deer and Shrubs Override Canopy Gap Size Effects on Growth and Survival of Yellow Birch, Northern Red Oak, Eastern White Pine, and Eastern Hemlock Seedlings? For. Ecol. Manag. 2012, 267, 134–143. [Google Scholar] [CrossRef]
- Curtis, R.O. Notes: A Simple Index of Stand Density for Douglas-Fir. For. Sci. 1982, 28, 92–94. [Google Scholar]
- Durbin, J.; Watson, G. Testing for Serial Correlation in Least Squares Regression, III. Biometrika 1971, 58, 1–19. [Google Scholar] [CrossRef]
- Franklin, J.F.; Spies, T.A.; van Pelt, R.; Carey, A.B.; Thornburgh, D.A.; Berg, D.R.; Lindenmayer, D.B.; Harmon, M.E.; Keeton, W.S.; Shaw, D.C.; et al. Disturbances and Structural Development of Natural Forest Ecosystems with Silvicultural Implications, Using Douglas-Fir Forests as an Example. For. Ecol. Manag. 2002, 155, 399–423. [Google Scholar] [CrossRef]
- Mader, S.F.; Nyland, R.D. Six-Year Response of Northern Hardwoods to the Selection System. North. J. Appl. For. 1984, 1, 87–91. [Google Scholar]
- Jones, R.H.; Nyland, R.D.; Raynal, D.J. Response of American Beech Regeneration to Selection Cutting of Northern Hardwoods in New York. North. J. Appl. For. 1989, 6, 34–36. [Google Scholar]
- Donoso, P.J.; Nyland, R.D.; Zhang, L. Growth of Saplings after Selection Cutting in Northern Hardwoods. North. J. Appl. For. 2000, 17, 149–152. [Google Scholar]
- Poznanovic, S.K.; Webster, C.R.; Bump, J.K. Maintaining Mid-Tolerant Tree Species with Uneven-Aged Forest Management: 9-Year Results from a Novel Group-Selection Experiment. Forestry 2013, 86, 555–567. [Google Scholar] [CrossRef]
- Woods, K.D. Reciprocal Replacement and the Maintenance of Codominance in a Beech-Maple Forest. Oikos 1979, 33, 31–39. [Google Scholar] [CrossRef]
- Whittaker, R.; Levin, S. The Role of Mosaic Phenomena in Natural Communities. Theor. Popul. Biol. 1977, 12, 117–139. [Google Scholar] [CrossRef]
- McClure, J.W.; Lee, T.D.; Leak, W.B. Gap Capture in Northern Hardwoods: Patterns of Establishment and Height Growth in Four Species. For. Ecol. Manag. 2000, 127, 181–189. [Google Scholar] [CrossRef]
- Ricard, J.-P.; Messier, C.; Delagrange, S.; Beaudet, M. Do Understory Sapling Respond to Both Light and below-Ground Competition? A Field Experiment in a North-Eastern American Hardwood Forest and a Literature Review. Ann. For. Sci. 2003, 60, 749–756. [Google Scholar] [CrossRef]
- Nolet, P. Coexistence et Sylviculture de L’érable À Sucre et Du Hêtre À Grandes Feuilles Dans Un Contexte de Changements Globaux; Université du Québec à Montréal: Montréal, QC, Canada, 2016. [Google Scholar]
- Beaudet, M.; Messier, C. Growth and Morphological Responses of Yellow Birch, Sugar Maple, and Beech Seedlings Growing under a Natural Light Gradient. Can. J. For. Res. 1998, 28, 1007–1015. [Google Scholar] [CrossRef]
- Beaudet, M.; Messier, C.; Paré, D.; Brisson, J.; Bergeron, Y. Possible Mechanisms of Sugar Maple Regeneration Failure and Replacement by Beech in the Boisé-Des-Muir Old-Growth Forest, Québec. Écoscience 1999, 6, 264–271. [Google Scholar] [CrossRef]
- Gasser, D.; Messier, C.; Beaudet, M.; Lechowicz, M.J. Sugar Maple and Yellow Birch Regeneration in Response to Canopy Opening, Liming and Vegetation Control in a Temperate Deciduous Forest of Quebec. For. Ecol. Manag. 2010, 259, 2006–2014. [Google Scholar] [CrossRef]
- Hane, E.N.; Hamburg, S.P.; Barber, A.L.; Plaut, J.A. Phytotoxicity of American Beech Leaf Leachate to Sugar Maple Seedlings in a Greenhouse Experiment. Can. J. For. Res. 2003, 33, 814–821. [Google Scholar] [CrossRef]
- Sage, R.W.; Porter, W.F.; Underwood, H.B. Windows of Opportunity: White-Tailed Deer and the Dynamics of Northern Hardwood Forests of the Northeastern US. J. Nat. Conserv. 2003, 10, 213–220. [Google Scholar] [CrossRef]
- Horsley, S.B.; Stout, S.L.; DeCalesta, D.S. White-Tailed Deer Impact on the Vegetation Dynamics of a Northern Hardwood Forest. Ecol. Appl. 2003, 13, 98–118. [Google Scholar] [CrossRef]
- Healy, W.M. Influence of Deer on the Structure and Composition of Oak Forests in Central Massachusetts. In The Science of Overabundance: Deer Ecology and Population Management; Smithsonian Institution Press: Washington, DC, USA, 1997; pp. 249–266. [Google Scholar]
- Paine, C.E.T.; Beck, H. Seed Predation by Neotropical Rain Forest Mammals Increases Diversity in Seedling Recruitment. Ecology 2007, 88, 3076–3087. [Google Scholar] [CrossRef] [PubMed]
- Marx, L.M.; Walters, M.B. Effects of Nitrogen Supply and Wood Species on Tsuga canadensis and Betula alleghaniensis Seedling Growth on Decaying Wood. Can. J. For. Res. 2006, 36, 2873–2884. [Google Scholar] [CrossRef]
- Decker, K.L.M.; Wang, D.; Waite, C.; Scherbatskoy, T. Snow Removal and Ambient Air Temperature Effects on Forest Soil Temperatures in Northern Vermont. Soil Sci. Soc. Am. J. 2003, 67, 1234. [Google Scholar] [CrossRef]
- Christenson, L.M.; Mitchell, M.J.; Groffman, P.M.; Lovett, G.M. Cascading Effects of Climate Change on Forest Ecosystems: Biogeochemical Links Between Trees and Moose in the Northeast USA. Ecosystems 2013, 17, 442–457. [Google Scholar] [CrossRef]
- Drescher, M.; Thomas, S.C. Snow Cover Manipulations Alter Survival of Early Life Stages of Cold-Temperate Tree Species. Oikos 2013, 122, 541–554. [Google Scholar] [CrossRef]
- Millar, C.I.; Stephenson, N.L.; Stephens, S.L. Climate Change and Forests of the Future: Managing in the Face of Uncertainty. Ecol. Appl. 2007, 17, 2145–2151. [Google Scholar] [CrossRef] [PubMed]
- Rustad, L.; Campbell, J.; Dukes, J.S.; Huntington, T.; Lambert, K.F.; Mohan, J.; Rodenhouse, N. Changing Climate, Changing Forests: The Impacts of Climate Change on Forests of the Northeastern United States and Eastern Canada; USDA: Washington, DC, USA, 2012.
- Canham, C.D.; Murphy, L. The Demography of Tree Species Response to Climate: Seedling Recruitment and Survival. Ecosphere 2016, 7, e01424. [Google Scholar] [CrossRef]
- Iverson, L.; Prasad, A.; Matthews, S. Modeling Potential Climate Change Impacts on the Trees of the Northeastern United States. Mitig. Adapt. Strateg. Glob. Chang. 2008, 13, 487–516. [Google Scholar] [CrossRef]
- Fisichelli, N.A.; Frelich, L.E.; Reich, P.B. Climate and Interrelated Tree Regeneration Drivers in Mixed Temperate–Boreal Forests. Landsc. Ecol. 2013, 28, 149–159. [Google Scholar] [CrossRef]
- Fisichelli, N.; Wright, A.; Rice, K.; Mau, A.; Buschena, C.; Reich, P.B. First-Year Seedlings and Climate Change: Species-Specific Responses of 15 North American Tree Species. Oikos 2014, 123, 1331–1340. [Google Scholar] [CrossRef]
- Mallett, A.L. Management of Understory American Beech by Manual and Chemical Control Methods; State University of New York, College of Environmental Science and Forestry: Syracuse, NY, USA, 2002. [Google Scholar]
- Kochenderfer, J.D.; Zedaker, S.M.; Johnson, J.E.; Smith, D.W.; Miller, G.W. Herbicide Hardwood Crop Tree Release in Central West Virginia. North. J. Appl. For. 2001, 18, 46–54. [Google Scholar]
- Nelson, A.S.; Wagner, R.G. Improving the Composition of Beech-Dominated Northern Hardwood Understories in Northern Maine. North. J. Appl. For. 2011, 28, 186–193. [Google Scholar]
- Forrester, J.A.; Lorimer, C.G.; Dyer, J.H.; Gower, S.T.; Mladenoff, D.J. Response of Tree Regeneration to Experimental Gap Creation and Deer Herbivory in North Temperate Forests. For. Ecol. Manag. 2014, 329, 137–147. [Google Scholar] [CrossRef]
- Arseneault, J.E.; Saunders, M.R.; Seymour, R.S.; Wagner, R.G. First Decadal Response to Treatment in a Disturbance-Based Silviculture Experiment in Maine. For. Ecol. Manag. 2011, 262, 404–412. [Google Scholar] [CrossRef]
- Bolton, N.W.; D’Amato, A.W. Regeneration Responses to Gap Size and Coarse Woody Debris within Natural Disturbance-Based Silvicultural Systems in Northeastern Minnesota, USA. For. Ecol. Manag. 2011, 262, 1215–1222. [Google Scholar] [CrossRef]
- Kern, C.; Montgomery, R.A.; Reich, P.B.; Strong, T.F. Harvest-Created Canopy Gaps Increase Species and Functional Trait Diversity of the Forest Ground-Layer Community. For. Sci. 2014, 60, 335–344. [Google Scholar] [CrossRef]
- Harmon, M.E.; Ferrell, W.K.; Franklin, J.F. Effects on Carbon Storage of Conversion of Old-Growth Forests to Young Forests. Science 1990, 247, 699–703. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).