Temporal Change in Aboveground Culms Carbon Stocks in the Moso Bamboo Forests and Its Driving Factors in Zhejiang Province, China
Abstract
1. Introduction
2. Materials and Methods
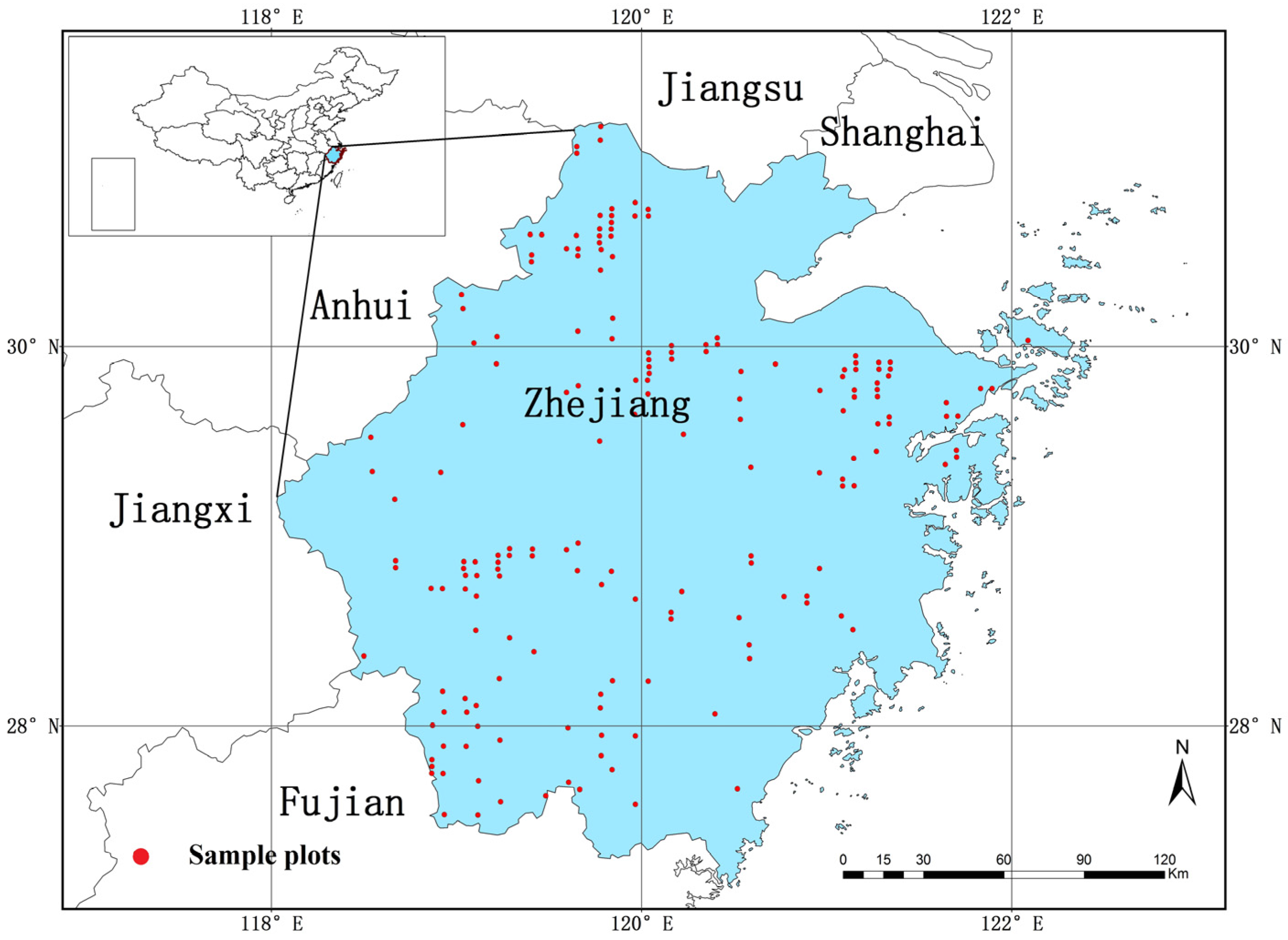
2.1. Research Area
2.2. Field Measurements and Carbon Stocks Calculations
2.3. Data Analysis
3. Results
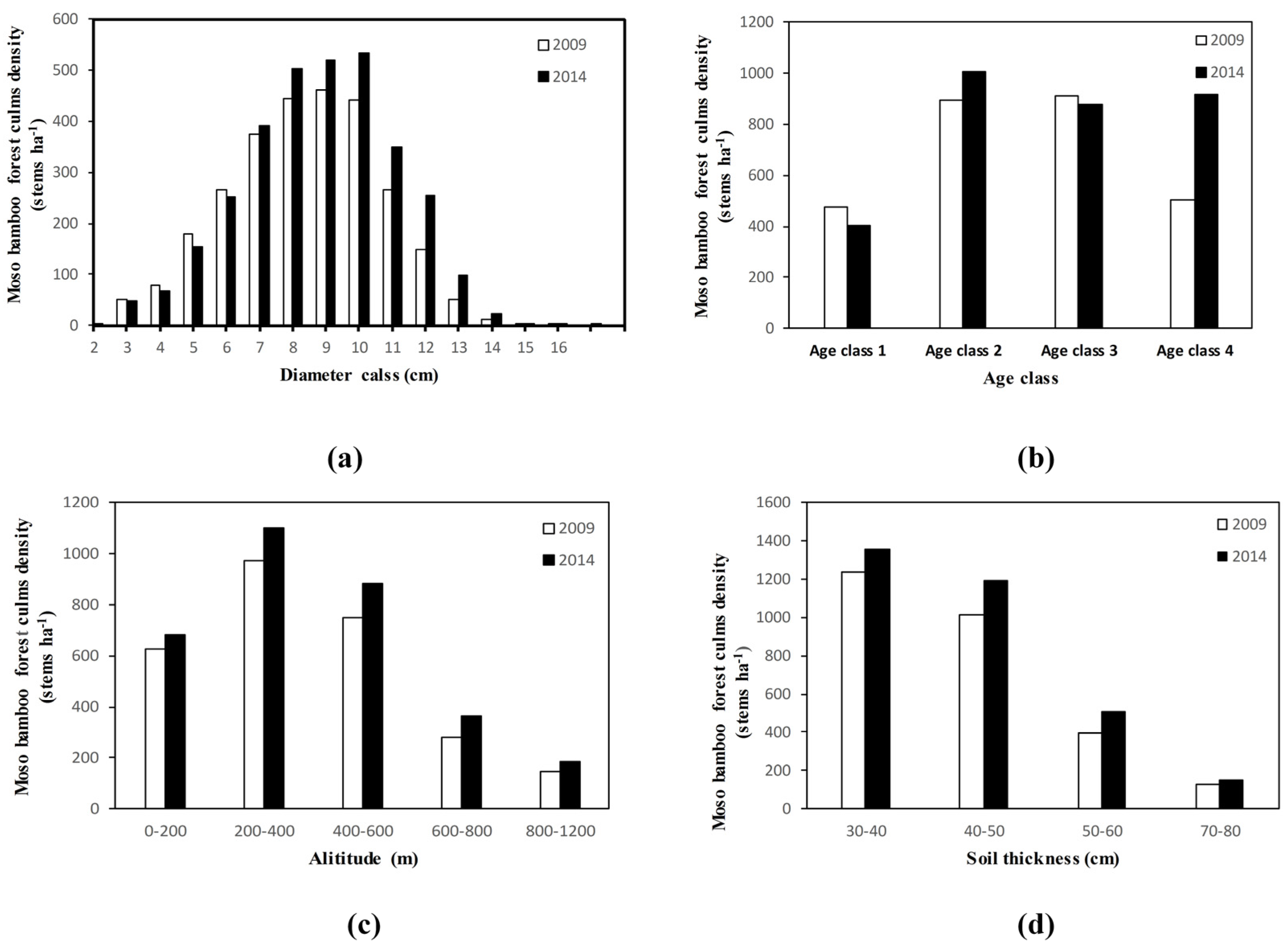
3.1. The Dynamic Distribution of Culms Density
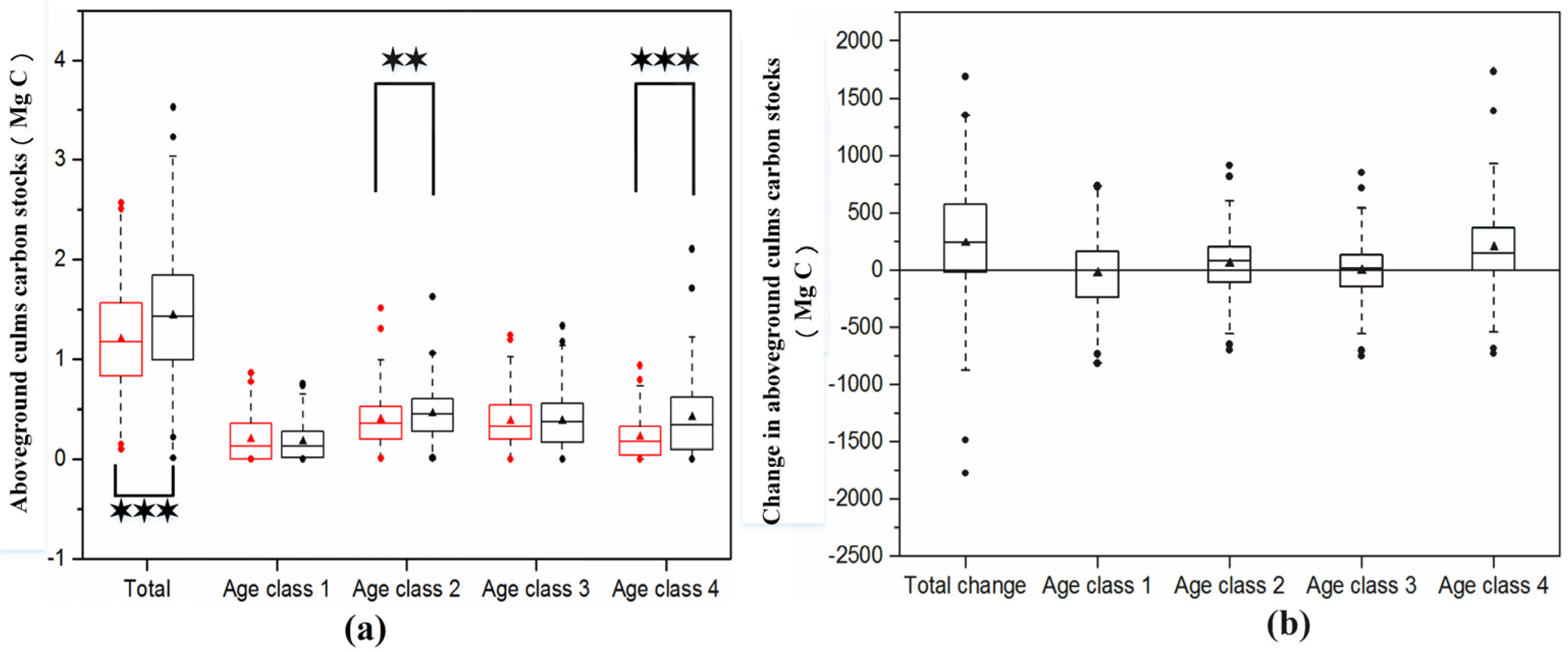
3.2. Change in Moso Bamboo Forest Carbon Stocks
3.3. Driving Factors of Carbon Stocks Change
4. Discussion
4.1. Change in Culms Density in the Moso Bamboo Forest
4.2. Large Carbon Sink Potential in the Moso Bamboo Forest
4.3. Implications for Regional Carbon Sequestration and Moso Bamboo Forest Management
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xu, B.; Pan, Y.; Plante, A.F.; Johnson, A.; Cole, J.; Birdsey, R. Decadal change of forest biomass carbon stocks and tree demography in the Delaware River Basin. For. Ecol. Manag. 2016, 374, 1–10. [Google Scholar] [CrossRef]
- Luyssaert, S.; Inglima, I.; Jung, M.; Richardson, A.D.; Reichstein, M.; Papale, D. CO2 balance of boreal, temperate, and tropical forestsderived from a global database. Glob. Chang. Biol. 2007, 13, 2509–2537. [Google Scholar] [CrossRef]
- Pan, Y.; Birdsey, R.A.; Fang, J.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G. A large and persistent carbon sink in the world’ s forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Ciais, P.; Sabine, C.; Bala, G.; Bopp, L.; Brovkin, V.; Canadell, J. Carbon and Other Biogeochemical Cycles; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Li, B.; Song, Z.; Wang, H.; Li, Z.; Jiang, P.; Zhou, G. Lithological control on phytolith carbon sequestration in Moso bamboo forests. Sci. Rep. 2014, 4, 5262. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Liu, H.; Cae, S.; Yang, X.; Zhang, X. Phytolith carbon sequestration in global terrestrial biomes. Sci. Total Environ. 2017, 603–604, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Intergovernmental Panel on Climate Change (IPCC). Climate change 2007: The physical science basis. In Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007. [Google Scholar]
- Sabine, C.L.; Feely, R.A.; Gruber, N.; Key, R.M.; Lee, K.; Bullister, J.L.; Wanninkhof, R.; Wong, C.S.; Wallace, D.W.R.; Tilbrook, B.; et al. The oceanic sink for anthropogenic CO2. Science 2004, 305, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.G.; Zhang, X.Q.; Zhang, Y.P.; Booth, T.; He, X.H. Changes of carbon stocks in bamboo stands in China during 100 years. For. Ecol. Manag. 2009, 258, 1489–1496. [Google Scholar] [CrossRef]
- Zhou, G.M.; Meng, C.F.; Jiang, P.K.; Xu, Q.F. Review of Carbon Fixation in Bamboo Forests in China. Bot. Rev. 2011, 77, 262–270. [Google Scholar] [CrossRef]
- Yen, T.M.; Lee, J.S. Comparing aboveground carbon sequestration between Moso bamboo (Phyllostachys heterocycla) and China fir (Cunninghamia lanceolata) forests based on the allometric model. For. Ecol. Manag. 2011, 261, 995–1002. [Google Scholar] [CrossRef]
- Mao, F.; Zhou, G.; Li, P.; Du, H.; Xu, X.; Shi, Y.; Mo, L.; Zhou, Y.; Tu, G. Optimizing selective cutting strategies for maximum carbon stocks and yield of Moso bamboo forest using BIOME-BGC model. J. Environ. Manag. 2017, 191, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Mao, F.; Li, P.; Zhou, G.; Du, H.; Xu, X.; Shi, Y.; Mo, L.; Zhou, Y.; Tu, G. Development of the BIOME-BGC model for the simulation of managed Moso bamboo forest ecosystems. J. Environ. Manag. 2016, 172, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.; Zhou, G.; Du, H.; Xu, X.; Shi, Y.; Lü, Y.; Zhou, Y.; Gu, C. Moso bamboo forest extraction and aboveground carbon storage estimation based on multi-source remotely sensed images. Int. J. Remote Sens. 2013, 34, 5351–5368. [Google Scholar] [CrossRef]
- Han, N.; Du, H.; Zhou, G.; Xu, X.; Cui, R.; Gu, C. Spatiotemporal heterogeneity of Moso bamboo aboveground carbon storage with Landsat Thematic Mapper images: A case study from Anji County, China. Int. J. Remote Sens. 2013, 34, 4917–4932. [Google Scholar] [CrossRef]
- Li, X.; Mao, F.; Du, H.; Zhou, G.; Xu, X.; Han, N.; Sun, S.; Gao, G.; Chen, L. Assimilating leaf area index of three typical types of subtropical forest in China from MODIS time series data based on the integrated ensemble Kalman filter and PROSAIL model. ISPRS. J. Photogram. Remote Sens. 2017, 126, 68–78. [Google Scholar] [CrossRef]
- Du, H.Q.; Zhou, G.M.; Fan, W.Y.; Ge, H.L.; Xu, X.J.; Shi, Y.J.; Fan, W.L. Spatial heterogeneity and carbon contribution of aboveground biomass of Moso bamboo by using geostatistical theory. Plant Ecol. 2009, 207, 131–139. [Google Scholar] [CrossRef]
- Gratani, L.; Crescente, M.F.; Varone, L.; Fabrini, G.; Digiulio, E. Growth pattern and photosynthetic activity of different bamboo species growing in the Botanical Garden of Rome. Flora 2008, 203, 77–84. [Google Scholar] [CrossRef]
- Li, Z.; Kobayashi, M. Plantation future of bamboo in China. J. For. Res. 2004, 15, 233–242. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). Global Forest Resources Assessment 2010: Main Report; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010. [Google Scholar]
- Guo, Q.R.; Yang, G.Y.; Du, T.Z.; Shi, J.M. Carbon character of Chinese bamboo forest. World Bamboo Rattan 2005, 3, 25–28. (In Chinese) [Google Scholar]
- Statse Forestry Administration of China. Forest Resources in China—The 8th National Forest Inventory; Statse Forestry Administration of China: Beijing, China, 2015.
- Jung, C.; Lowdermilk, W. Forestry. In Agroindustries and Forestry; Daniels, C.A., Menzies, N.K., Eds.; Cambridge University Press: Cambridge, UK, 1996; pp. 540–667. [Google Scholar]
- Li, P.; Zhou, G.; Du, H.; Lu, D.; Mo, L.; Xu, X.; Shi, Y.; Zhou, Y. Current and potential carbon stocks in Moso bamboo forests in China. J. Environ. Manag. 2015, 156, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.M. Carbon Storage, Fixation and Distribution in Moso bamboo (Phyllostachy pubescens) Stands Ecosystem; Zhejiang University: Hangzhou, China, 2006. [Google Scholar]
- Fu, J.H. Chinese Moso bamboo: Its importance. Bamboo 2001, 22, 5–7. [Google Scholar]
- Yen, T.M. Culm height development, biomass accumulation and carbon storage in an initial growth stage for a fast-growing Moso bamboo (Phyllostachy pubescens). Bot.Stud. 2016, 57, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zeng, Q.; Wu, Z.; Zhou, G.; Chen, B. Estimation of amount of carbon pool in natural tropical forest of China. For. Res. 1998, 11, 156–162. (In Chinese) [Google Scholar]
- Komatsu, H.; Onozawa, Y.; Kume, T.; Tsuruta, K.; Shinohara, Y.; Otsuki, K. Canopy conductance for a Moso bamboo (Phyllostachys pubescens) forest in western Japan. Agric. For. Meteorol. 2012, 156, 111–120. [Google Scholar] [CrossRef]
- Zhou, B.; Fu, M.; Xie, J.; Yang, X.; Li, Z. Ecological functions of bamboo forest: Research and application. J. For. Res. 2005, 16, 143–147. (In Chinese) [Google Scholar]
- Anonymous. Healing degraded land. INBAR Mag. 1997, 5, 40–45. [Google Scholar]
- Zhou, G.; Xu, X.; Du, H.; Ge, H.; Shi, Y.; Zhou, Y. Estimating aboveground carbon of Moso bamboo forests using the k nearest neighbors technique and satellite imagery. Photogramm. Eng. Remote Sens. 2011, 77, 1123–1131. [Google Scholar] [CrossRef]
- Du, H.Q.; Zhou, G.M.; Ge, H.L.; Fan, W.Y.; Xu, X.J.; Fan, W.L.; Shi, Y.J. Satellite-based carbon stock estimation for bamboo forest with a non-linear partial least square regression technique. Int. J. Remote Sens. 2012, 33, 1917–1933. [Google Scholar] [CrossRef]
- Mao, F.; Du, H.; Zhou, G.; Li, X.; Xu, X.; Li, P.; Sun, S. Coupled LAI assimilation and BEPS model for analyzing the spatiotemporal pattern and heterogeneity of carbon fluxes of the bamboo forest in Zhejiang Province, China. Agric. For. Meteorol. 2017, 242, 96–108. [Google Scholar] [CrossRef]
- Soil Survey Staff of United States Department of Agriculture (USDA). Soil Taxonomy: A Basic System of Soil Classification for Making and Interpreting Soil Survey; Agriculture Handbook 1999, (No. 436.); Soil Survey Staff of United States Department of Agriculture (USDA): Washington, DC, USA, 1999.
- Department of Forestry of Zhejiang Province. Announcement of Value of Forest Resources and Ecological Functions of Zhejiang Province in 2014; Department of Forestry of Zhejiang Province: Zhejiang, China, 2015.
- State Forestry Administration of China. Main Technical Regulations for Continuous Inventory of National Forest Resources; Department of Forestry Resources Management of the State Forestry Administration of China: Beijing, China, 2003; pp. 36–44.
- Yen, T.M. Comparing aboveground structure and aboveground carbon storage of an age series of Moso bamboo forests subjected to different management strategies. J. For. Res. 2014, 20, 1–8. [Google Scholar] [CrossRef]
- Liu, E.B.; Shi, Y.J.; Li, Y.F.; Zhou, G.M. Carbon sequestration potential of Moso bamboo forest in Zhejiang province based on the non-spatial structure. Sci. Silv. Sin. 2012, 48, 9–14. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Vienna, Austria, 2015. Available online: http://www.Rproject.org (accessed on 21 May 2017).
- Qiu, F.G. The on-year and off-year of Phyllostachys pubescens forests and their control. J. Bamboo Res. 1984, 3, 62–69. [Google Scholar]
- Wang, K.; Bao, Q. Phytohormone content and distribution in Phyllostachys pubescens in shooting period. J. Bamboo Res. 1989, 8, 35–46. (In Chinese) [Google Scholar]
- Li, R.; Zhong, Z.; Werger, M.J.A. The relationship between the clonal growth and the density of adult shoots with different leaf age in Phyllostachys pubescens mazel. Acta Phytoecol. Sin. 1997, 21, 545–550. (In Chinese) [Google Scholar]
- Wu, L.; Xiao, J. Study on dynamic characteristics of eudogenous phytohormone in on-and-off year bamboo (Phyllostachys heterocycles var. pubescens) grove. J. Bamboo Res. 1998, 17, 24–30. (In Chinese) [Google Scholar]
- Wu, J.; Zhou, G.; Xu, Q.; Yang, F. Spatial distribution of nutrition element and its relationship with soil nutrients in different years of Phyllostachys pubescens. Sci. Silv. Sin. 2005, 41, 171–174. (In Chinese) [Google Scholar]
- Zhou, G.M.; Wu, J.S.; Jiang, P.K. Effects of different management models on carbon storage in Phyllostachys pubescens forests. J. Beijing For. Univ. 2006, 28, 51–55. (In Chinese) [Google Scholar]
- Wen, G.S.; Zhang, L.Y.; Zhang, R.M.; Cao, Z.H.; Zhou, G.M.; Huang, H. Temporal and spatial dynamics of carbon fixation by Moso bamboo (Phyllostachys pubescens) in subtropical China. Bot. Rev. 2011, 77, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Ji, B.Y.; Tao, J.X.; Zhang, G.J.; Du, Q.; Yao, H.W.; Xu, J. Zhejiang Province’s forest vegetation biomass assessment for guaranteed accuracy. J. Zhejiang Agric. For. Univ. 2012, 29, 328–334. [Google Scholar]
- Yen, T.M.; Wang, C.T. Assessing carbon storage and carbon sequestration for natural forests, man-made forests, and bamboo forests in Taiwan. Int. J. Sustain. Dev. World Ecol. 2013, 20, 455–460. [Google Scholar] [CrossRef]
- Jiang, P.K.; Meng, C.F.; Zhou, G.M.; Xu, Q.F. Comparative study of carbon storage in different forest stands in subtropical china. Bot. Rev. 2011, 77, 242–251. [Google Scholar] [CrossRef]
- Zheng, H.; Ouyang, Z.Y.; Xu, W.H.; Wang, X.K.; Miao, H.; Li, X.Q.; Tian, Y.X. Variation of carbon storage by different reforestation types in the hilly red soil region of southern china. For. Ecol. Manag. 2008, 255, 1113–1121. [Google Scholar] [CrossRef]
- Lou, Y.P.; Wu, L.R.; Shao, D.F.; Yan, Z.W. Effect of long term management of pure Phyllostachys pubescens stands on soil fertility. For. Res. 1997, 10, 125–129. (In Chinese) [Google Scholar]
- Zhang, C.X.; Ding, X.C. Studies on yield of Phyllost Achys heterocycla var. Pubescens stands. J. Bamboo Res. 1997, 16, 31–36. (In Chinese) [Google Scholar]
- Zheng, Y.S.; Hong, W. A study on age structure model of bamboo stand and its application. Sci. Silv. Sin. 1998, 34, 32–39. (In Chinese) [Google Scholar]
- Zheng, Y.S.; Hong, W.; Chen, L.G. Study on the density structure for bamboo forest management. Sci. Silv. Sin. 1998, 34, 5–10. (In Chinese) [Google Scholar]
- Wu, B.S. A preliminary analysis of the relationship between the underground structure of a Phyllostachys pubescens forest and its yield. J. Bamboo Res. 1984, 3, 49–58. (In Chinese) [Google Scholar]
- He, L.; He, X.Y. Rational age structure and growth regularity during shooting period of Phyllostachys pubescens forest for shoot. J. Bamboo Res. 1993, 12, 52–56. (In Chinese) [Google Scholar]
- Zhou, G.M.; Jiang, P.K.; Mo, L.F. Bamboo: A possible approach to the control of global warming. Int. J. Nonlinear Sci. Numer. Simul. 2009, 10, 547–550. [Google Scholar]
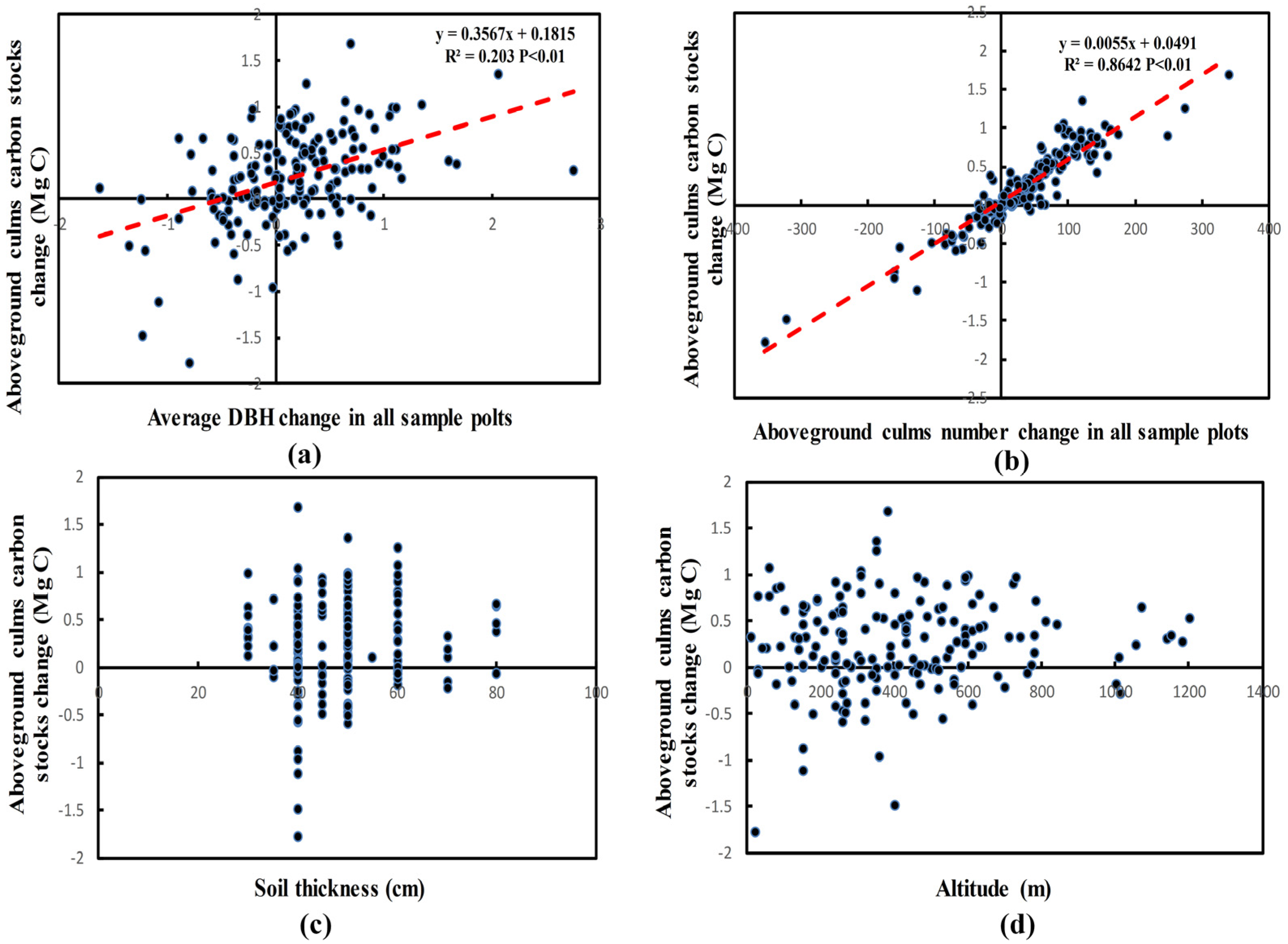
| Variable | Slope | Intercept | R2 | p |
|---|---|---|---|---|
| change in culms density | 6.34 | 20.47 | 0.864 | <0.01 ** |
| DBH change | 1757.15 | −33.89 | 0.203 | <0.01 ** |
| geomorphology | −8747.85 | 37,206.47 | 0.008 | 0.26 |
| altitude | 15.79 | −6318.05 | 0.008 | 0.24 |
| aspect | −3197.53 | 14,415.89 | 0.004 | 0.4 |
| slope | −12,567 | 359,006 | 0.000 | 0.95 |
| slope position | 8650.8 | −26,385 | 0.005 | 0.35 |
| canopy density | −40,590 | 30,302 | 0.008 | 0.25 |
| soil thickness | 464.7 | −21,963 | 0.01 | 0.20 |
| Study Area | Year | Aboveground Carbon Stocks (Mg C ha−1) | Total Aboveground Culms Carbon Stocks (Tg C) | Mean Aboveground Carbon Sequestration (Mg C ha−1 Year−1) | Aboveground Carbon Sequestration (Tg C Year−1) | Forest Type |
|---|---|---|---|---|---|---|
| Zhejiang Province | 2004 | 13.00 [39] | / | / | / | Moso bamboo forest |
| Zhejiang Province | 2009 | 15.01 (this study) | 10.37 [48] 10.75 (this study) | / | / | Moso bamboo forest |
| Zhejiang Province | 2014 | 17.96 (this study) | 14.11 (this study) | / | / | Moso bamboo forest |
| Zhejiang Province | 2009–2014 | / | / | 0.59 (this study) | 0.42 (this study) | Moso bamboo forest |
| Zhejiang Province | 2011–2014 | / | / | / | 1.74 [34] | Bamboo forest |
| Central Taiwan | 2004–2007 | 40.6 [11] | / | 8.13 [11] | / | Moso bamboo plantation |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, L.; Shi, Y.; Zhou, G.; Xu, X.; Liu, E.; Zhou, Y.; Li, C.; Fang, H.; Deng, X. Temporal Change in Aboveground Culms Carbon Stocks in the Moso Bamboo Forests and Its Driving Factors in Zhejiang Province, China. Forests 2017, 8, 371. https://doi.org/10.3390/f8100371
Xu L, Shi Y, Zhou G, Xu X, Liu E, Zhou Y, Li C, Fang H, Deng X. Temporal Change in Aboveground Culms Carbon Stocks in the Moso Bamboo Forests and Its Driving Factors in Zhejiang Province, China. Forests. 2017; 8(10):371. https://doi.org/10.3390/f8100371
Chicago/Turabian StyleXu, Lin, Yongjun Shi, Guomo Zhou, Xiaojun Xu, Enbing Liu, Yufeng Zhou, Chong Li, Huiyun Fang, and Xu Deng. 2017. "Temporal Change in Aboveground Culms Carbon Stocks in the Moso Bamboo Forests and Its Driving Factors in Zhejiang Province, China" Forests 8, no. 10: 371. https://doi.org/10.3390/f8100371
APA StyleXu, L., Shi, Y., Zhou, G., Xu, X., Liu, E., Zhou, Y., Li, C., Fang, H., & Deng, X. (2017). Temporal Change in Aboveground Culms Carbon Stocks in the Moso Bamboo Forests and Its Driving Factors in Zhejiang Province, China. Forests, 8(10), 371. https://doi.org/10.3390/f8100371






