Ecosystem Carbon Stock Loss after Land Use Change in Subtropical Forests in China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Land Use History
2.3. Estimation of Aboveground and Belowground Biomass
2.4. Soil Sampling and Analysis
2.5. Statistical Analysis
3. Results
3.1. Stand Characteristics of the SF and CFP Stand
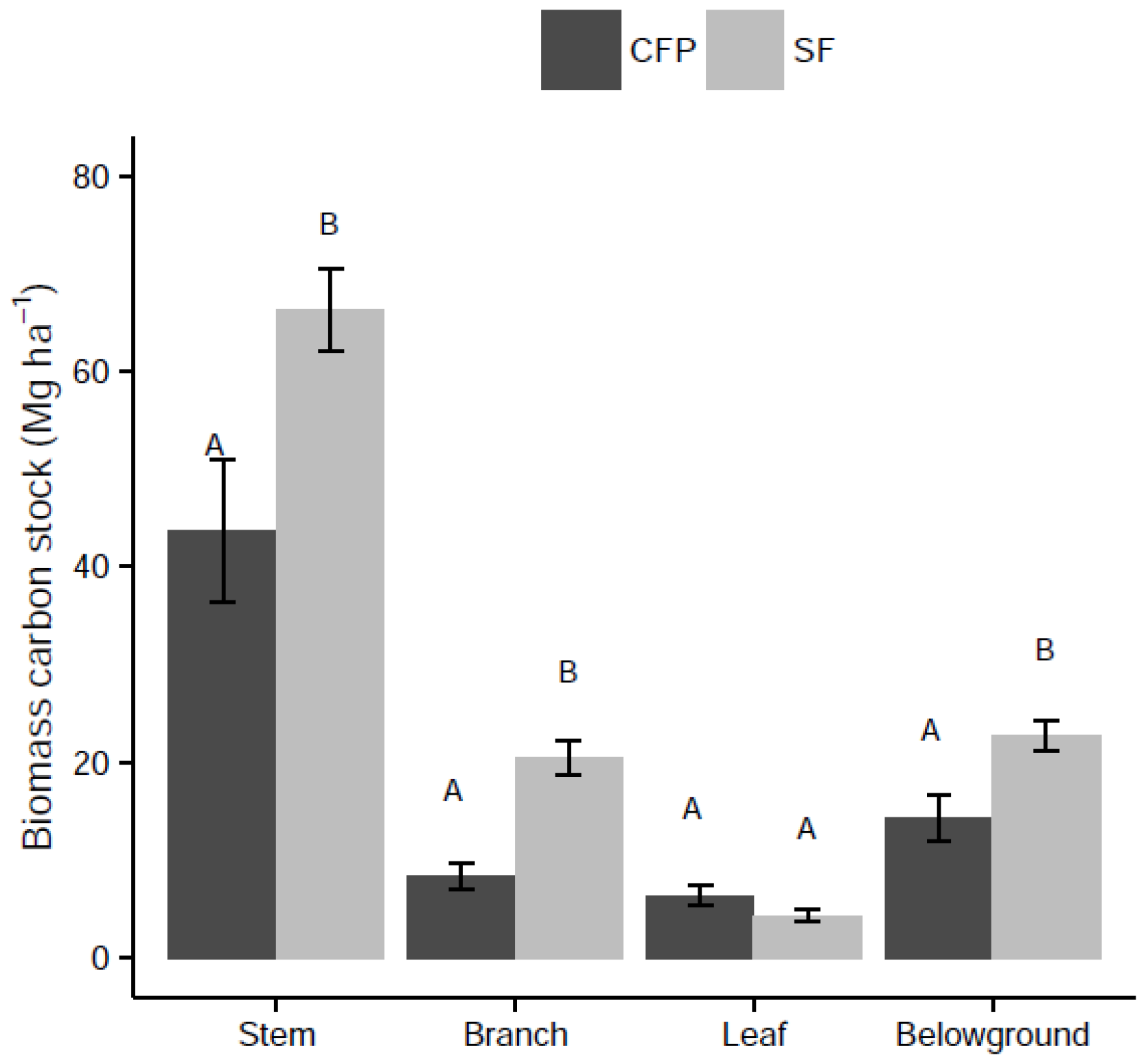
3.2. Effects of Land Use Change on Biomass C Stock
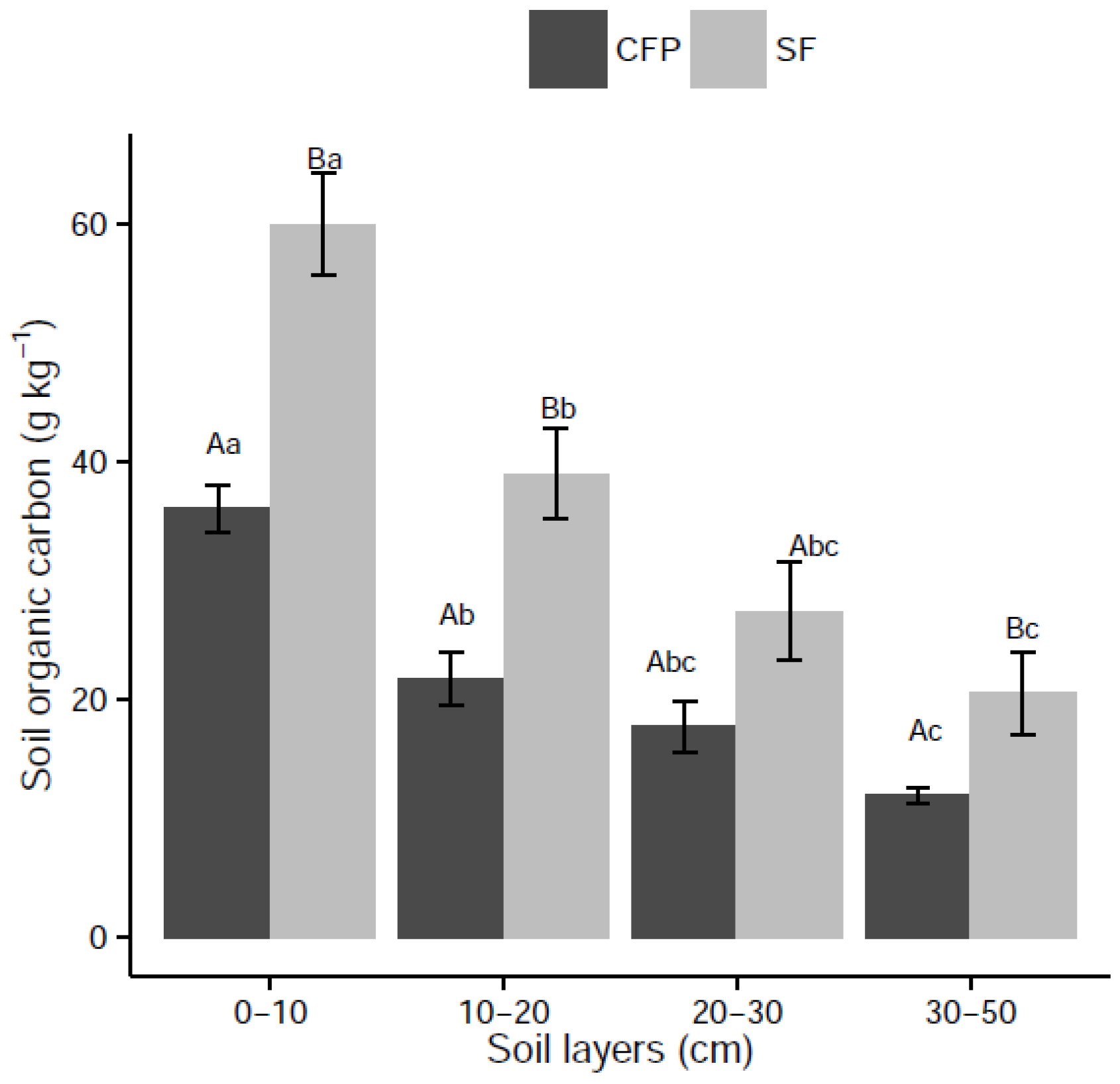
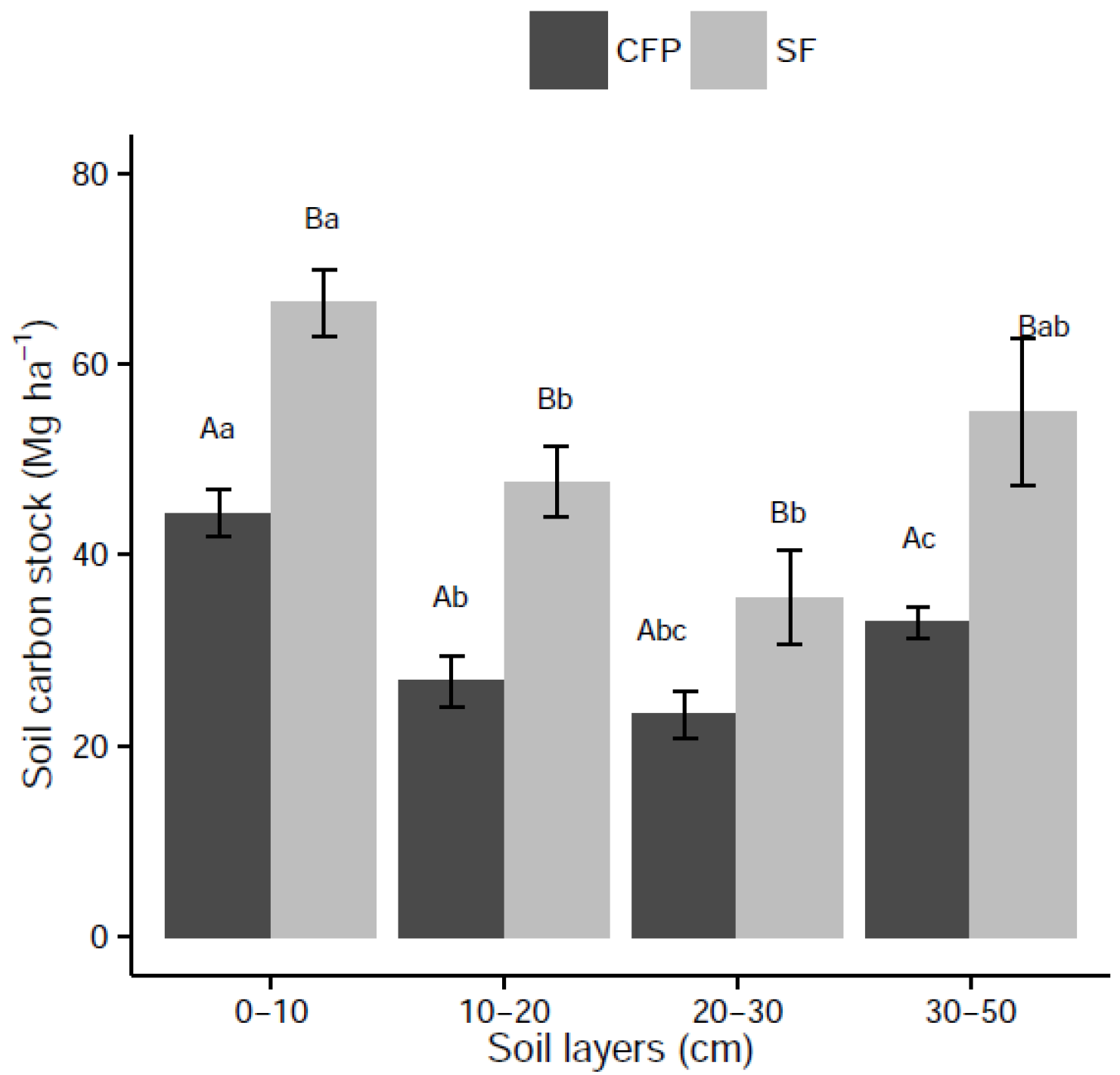
3.3. Effects of Land Use Change on Soil C Stock
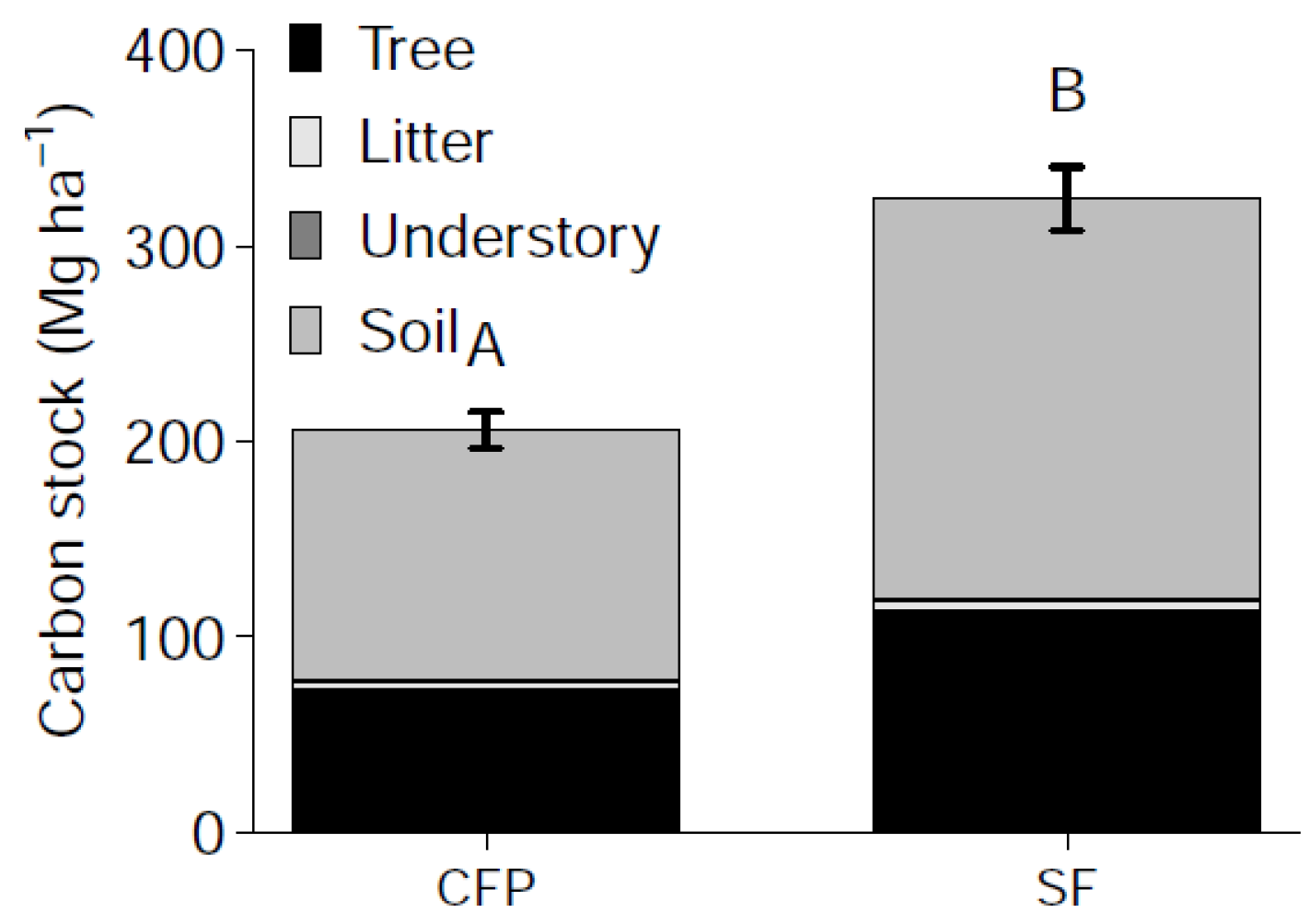
3.4. Effects of Land Use Change on Total Ecosystem C Stocks
4. Discussion
4.1. Land Use Change Decreased Biomass C
4.2. Land Use Change Reduced Soil C
4.3. Implication for Forest Management
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| C | carbon |
| SOC | soil organic carbon |
| CFP | Chinese fir plantation |
| SF | secondary forest |
| CO2 | carbon dioxide |
References
- Pan, Y.; Birdsey, R.A.; Fang, J.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G.; et al. A large and persistent carbon sink in the world’s forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef] [PubMed]
- IPCC. Climate Change 2007: Synthesis Report; Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2007; pp. 36–41. [Google Scholar]
- Houghton, R.A. Balancing the global carbon budget. Annu. Rev. Earth Planet. Sci. 2007, 35, 313–347. [Google Scholar] [CrossRef]
- Luo, Y.; Zhou, X. Soil Respiration and the Environment; Academic Press: San Diego, CA, USA, 2006; pp. 18–20. [Google Scholar]
- Bond-Lamberty, B.; Thomson, A. Temperature-associated increases in the global soil respiration record. Nature 2010, 464, 579–582. [Google Scholar] [CrossRef] [PubMed]
- Jobbágy, E.G.; Jackson, R.B. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol. Appl. 2000, 10, 423–436. [Google Scholar]
- Van der Werf, G.R.; Morton, D.C.; DeFries, R.S.; Olivier, J.G.J.; Kasibhatla, P.S.; Jackson, R.B.; Collatz, G.J.; Randerson, J.T. CO2 emissions from forest loss. Nat. Geosci. 2009, 2, 737–738. [Google Scholar] [CrossRef]
- Watson, R.T.; Noble, I.R.; Bolin, B.; Ravindranath, N.; Verardo, D.J.; Dokken, D.J. Land Use, Land-Use Change, and Forestry: A Special Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Fu, X.; Shao, M.; Wei, X.; Horton, R. Soil organic carbon and total nitrogen as affected by vegetation types in Northern Loess Plateau of China. Geoderma 2010, 155, 31–35. [Google Scholar] [CrossRef]
- Lozano-García, B.; Parras-Alcántara, L. Land use and management effects on carbon and nitrogen in Mediterranean Cambisols. Agric. Ecosyst. Environ. 2013, 179, 208–214. [Google Scholar]
- Guo, L.B.; Gifford, R.M. Soil carbon stocks and land use change: A meta analysis. Glob. Chang. Biol. 2002, 8, 345–360. [Google Scholar] [CrossRef]
- Kotowska, M.M.; Leuschner, C.; Triadiati, T.; Meriem, S.; Hertel, D. Quantifying above- and belowground biomass carbon loss with forest conversion in tropical lowlands of Sumatra (Indonesia). Glob. Chang. Biol. 2015, 21, 3620–3634. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Guo, Z.; Peng, C. Land use induced changes of organic carbon storage in soils of China. Glob. Chang. Biol. 2003, 9, 305–315. [Google Scholar] [CrossRef]
- Post, W.M.; Kwon, K.C. Soil carbon sequestration and land-use change: Processes and potential. Glob. Chang. Biol. 2000, 6, 317–327. [Google Scholar] [CrossRef]
- Wang, Q.K.; Wang, S.L.; Zhong, M.C. Ecosystem carbon storage and soil organic carbon stability in pure and mixed stands of Cunninghamia lanceolata and Michelia macclurei. Plant Soil 2013, 370, 295–304. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, J.; Chen, G.; Yin, Y.; Gao, R.; Lin, C. Effects of forest conversion on soil labile organic carbon fractions and aggregate stability in subtropical China. Plant Soil 2009, 323, 153–162. [Google Scholar] [CrossRef]
- Guan, F.; Tang, X.; Fan, S.; Zhao, J.; Peng, C. Changes in soil carbon and nitrogen stocks followed the conversion from secondary forest to Chinese fir and Moso bamboo plantations. Catena 2015, 133, 455–460. [Google Scholar] [CrossRef]
- Zhao, M.; Xiang, W.; Peng, C.; Tian, D. Simulating age-related changes in carbon storage and allocation in a Chinese fir plantation growing in southern China using the 3-PG model. For. Ecol. Manag. 2009, 257, 1520–1531. [Google Scholar] [CrossRef]
- Zhang, X.Q.; Kirschbaum, M.U.F.; Hou, Z.H.; Guo, Z.H. Carbon stock changes in successive rotations of Chinese fir (Cunninghamia lanceolata (lamb) hook) plantations. For. Ecol. Manag. 2004, 202, 131–147. [Google Scholar] [CrossRef]
- Wu, Z.L. Chinese-Fir; China Forestry Publishing House: Beijing, China, 1984; p. 583. (In Chinese) [Google Scholar]
- State Forestry Administration. Report for Chinese Forest Resource—The 8th National Forest Inventory; China Forestry Publishing House: Beijing, China, 2014; p. 94. (In Chinese)
- Gelaw, A.M.; Singh, B.R.; Lal, R. Soil organic carbon and total nitrogen stocks under different land uses in a semi-arid watershed in Tigray, Northern Ethiopia. Agric. Ecosyst. Environ. 2014, 188, 256–263. [Google Scholar] [CrossRef]
- Cook, R.L.; Binkley, D.; Mendes, J.C.T.; Stape, J.L. Soil carbon stocks and forest biomass following conversion of pasture to broadleaf and conifer plantations in southeastern Brazil. For. Ecol. Manag. 2014, 324, 37–45. [Google Scholar] [CrossRef]
- Yang, Y.S.; Guo, J.F.; Chen, G.S.; Xie, J.S.; Gao, R.; Li, Z.; Jin, Z. Carbon and nitrogen pools in Chinese fir and evergreen broadleaved forests and changes associated with felling and burning in mid-subtropical China. For. Ecol. Manag. 2005, 216, 216–226. [Google Scholar] [CrossRef]
- Geng, T.S.; Wang, H.H. Research on the water and soil conservation in Shitai County of Anhui Province. J. Anhui Agric. Sci. 2011, 39, 451–452, 482. (In Chinese) [Google Scholar]
- Lu, C.M. Rock-soil geochemical features for Dashan Area, Shitai, Anhui. Geol. Anhui 2010, 20, 120–125. (In Chinese) [Google Scholar]
- Tang, X.; Lu, Y.; Fehrmann, L.; Forrester, D.I.; Guisasola-Rodríguez, R.; Pérez-Cruzado, C.; Kleinn, C. Estimation of stand-level aboveground biomass dynamics using tree ring analysis in a Chinese fir plantation in Shitai County, Anhui Province, China. New For. 2015, 47, 319–332. [Google Scholar] [CrossRef]
- Guisasola, R. Allometric Biomass Equations and Crown Architecture in Mixed-Species Forests of Subtropical China; Albert-Ludwigs Universität Freiburg: Freiburg, Germany, 2014. [Google Scholar]
- Luo, Y.J.; Wang, X.K.; Zhang, X.Q.; Booth, T.H.; Lu, F. Root: shoot ratios across China’s forests: Forest type and climatic effects. For. Ecol. Manag. 2012, 269, 19–25. [Google Scholar] [CrossRef]
- Fang, J.Y.; Wang, G.G.; Liu, G.H.; Xu, S.L. Forest biomass of China: An estimate based on the biomass-volume relationship. Ecol. Appl. 1998, 8, 1084–1091. [Google Scholar]
- Zhang, W.R.; Yang, G.Y.; Tu, X.N.; Zhang, P. Determination of Forest Soil Water-Physical Properties; China Standard Press: Beijing, China, 1999; Volume 1999, pp. 21–24. (In Chinese) [Google Scholar]
- Zhang, W.R.; Yang, G.Y.; Tu, X.N.; Zhang, P. Determinatlon of organic matter in forest soil and calculation carbon-nitrogen ratio. In Standards of Forest Soil Measurement; Zhang, W.R., Yang, G.Y., Tu, X.N., Zhang, P., Eds.; China Standard Press: Beijing, China, 1999; Volume 1999, pp. 105–108. (In Chinese) [Google Scholar]
- Zhang, L.H.; Xie, Z.K.; Zhao, R.F.; Wang, Y.J. The impact of land use change on soil organic carbon and labile organic carbon stocks in the Longzhong region of Loess Plateau. J. Arid Land 2012, 4, 241–250. [Google Scholar]
- Xie, Z.; Zhu, J.; Liu, G.; Cadisch, G.; Hasegawa, T.; Chen, C.; Sun, H.; Tang, H.; Zeng, Q. Soil organic carbon stocks in China and changes from 1980s to 2000s. Glob. Chang. Biol. 2007, 13, 1989–2007. [Google Scholar] [CrossRef]
- Team, R.C.R. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria. Available online: http://www.R-project.org/ (accessed on 6 March 2015).
- Liao, C.; Luo, Y.; Fang, C.; Li, B. Ecosystem carbon stock influenced by plantation practice: Implications for planting forests as a measure of climate change mitigation. PLoS ONE 2010, 5, e10867. [Google Scholar] [CrossRef] [PubMed]
- Arevalo, C.B.M.; Bhatti, J.S.; Chang, S.X.; Sidders, D. Ecosystem carbon stocks and distribution under different land-uses in north central Alberta, Canada. For. Ecol. Manag. 2009, 257, 1776–1785. [Google Scholar] [CrossRef]
- Tang, X. Estimation of Biomass, Volume and Growth of Subtropical Forests in Shitai County, China; Georg-August-University Göttingen: Göttingen, Germany, 2015. [Google Scholar]
- Chen, G.S.; Yang, Z.J.; Gao, R.; Xie, J.S.; Guo, J.F.; Huang, Z.Q.; Yang, Y.S. Carbon storage in a chronosequence of Chinese fir plantations in southern China. For. Ecol. Manag. 2013, 300, 68–76. [Google Scholar] [CrossRef]
- Acker, S.A.; Halpern, C.B.; Harmon, M.E.; Dyrness, C.T. Trends in bole biomass accumulation, net primary production and tree mortality in Pseudotsuga menziesii forests of contrasting age. Tree Physiol. 2002, 22, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, X.; Wang, J. Impact of land use change on profile distributions of soil organic carbon fractions in the Yanqi Basin. Catena 2014, 115, 79–84. [Google Scholar] [CrossRef]
- Lou, Y.; Xu, M.; Chen, X.; He, X.; Zhao, K. Stratification of soil organic C, N and C:N ratio as affected by conservation tillage in two maize fields of China. Catena 2012, 95, 124–130. [Google Scholar] [CrossRef]
- Ming, A.; Liu, S.; Nong, Y.; Cai, D.; Jia, H.; Huang, D.; Wang, Q.; Nong, Z. Comparison of carbon storage in juvenile monoculture and mixed plantation stands of three common broadleaved tree species in subtropical China. Acta Ecol. Sin. 2015, 35, 180–188. (In Chinese) [Google Scholar] [CrossRef]
- Hu, Y.L.; Zeng, D.H.; Fan, Z.P.; Chen, G.S.; Zhao, Q.; Pepper, D. Changes in ecosystem carbon stocks following grassland afforestation of semiarid sandy soil in the southeastern Keerqin Sandy Lands, China. J. Arid Environ. 2008, 72, 2193–2200. [Google Scholar] [CrossRef]
- Li, K.R.; Wang, S.Q.; Cao, M.K. Carbon stock in vegetation and soil in China. Sci. China Ser. D Earth Sci. 2003, 33, 72–80. (In Chinese) [Google Scholar]
- Wang, H.; Huang, Y.; Feng, Z.; Wang, S. C and N stocks under three plantation forest ecosystems of Chinese fir, Michelia macclurei and their mixture. Front. For. China 2007, 2, 251–259. [Google Scholar] [CrossRef]
- Bing, K.; Shirong, L.; Daoxiong, C.; Lihua, L. Characteristics of biomass, carbon accumulation and its spatial distribution in Cunninghamia lanceolata forest ecosystem in low subtropical area. Sci. Silvae Sin. 2009, 45, 147–153. (In Chinese) [Google Scholar]
- Xiao, F.M.; Fan, S.H.; Wang, S.L.; Xiong, C.Y.; Zhang, C.; Liu, S.P.; Zhang, J. Carbon storage and spatial distribution in Phyllostachy pubescens and Cunninghamia lanceolata plantation ecosystem. Acta Ecol. Sin. 2007, 27, 2794–2801. (In Chinese) [Google Scholar]
- Ji, Y.; Guo, K.; Ni, J.; Xu, X.; Wang, Z.; Wang, S. Current forest carbon stocks and carbon sequestration potential in Anhui Province, China. Chin. J. Plant Ecol. 2016, 40, 395–404. [Google Scholar]
- Huang, C.; Zhang, J.; Yang, W.; Zhang, G. Characteristics of carbon stock in artificial forest ecosystem in Sichuan Province of China. Chin. J. Appl. Ecol. 2008, 19, 1644–1650. (In Chinese) [Google Scholar]
- Wang, S.; Wang, X.; Ouyang, Z. Effects of land use, climate, topography and soil properties on regional soil organic carbon and total nitrogen in the Upstream Watershed of Miyun Reservoir, North China. J. Environ. Sci. 2012, 24, 387–395. [Google Scholar] [CrossRef]
- Chen, J.N.; Wang, S.L. Dynamic of soil carbon pool in Cunninghamia lanceolata plantation: Recent advances and future prospects. Guangxi For. Sci. 2007, 36, 147–151. (In Chinese) [Google Scholar]
- Ferré, C.; Comolli, R.; Leip, A.; Seufert, G. Forest conversion to poplar plantation in a Lombardy floodplain (Italy): Effects on soil organic carbon stock. Biogeosci. Discuss. 2014, 11, 9601–9627. [Google Scholar]
- Wang, Q.; Wang, S.; Yu, X. Decline of soil fertility during forest conversion of secondary forest to Chinese Fir plantations in subtropical China. Land Degrad. Dev. 2011, 22, 444–452. [Google Scholar] [CrossRef]
- Ma, X.; Yang, Y.; Lin, K.; Liu, A.; He, Z. Effect of different ground clearance on Chinese fir plantation ecosystems. Acta Ecol. Sin. 1996, 17, 176–183. [Google Scholar]
- Lugo, A.; Brown, S. Management of tropical soils as sinks or sources of atmospheric carbon. Plant Soil 1993, 149, 27–41. [Google Scholar] [CrossRef]
- Meng, X. Forest Mensuration, 3rd ed.; China Forestry Publishing House: Beijing, China, 2006. [Google Scholar]
- Liu, S.L.; Fu, B.J.; Chen, L.D.; Lu, Y.H.; Ma, K.M. Effects of land use changes on soil properties in Wolong Nature Reserve. Geogr. Res. Aust. 2002, 21, 682–688. (In Chinese) [Google Scholar]
| Species | Components | Model | R2adj | n | RMSE |
|---|---|---|---|---|---|
| Cunninghamia lanceolata | Stem | 0.0202 × (DBH2·H)0.967 | 0.9764 | 12 | 0.0179 |
| Branch | 0.0118 × DBH3.742 × H−1.603 | 0.9722 | 12 | 0.0294 | |
| Leaf | 0.0319 × DBH3.207 × H−1.511 | 0.9522 | 12 | 0.0363 | |
| LA | 0.1635 × DBH3.2532 × H−1.5334 | 0.9500 | 12 | 0.0351 | |
| Castanopsis sclerophylla | Stem | 0.0240 × (DBH2·H)1.0123 | 0.9795 | 24 | 0.0213 |
| Branch | 0.0471 × DBH2.187 | 0.8889 | 24 | 0.0863 | |
| Leaf | 0.102 × DBH1.873 × H−0.731 | 0.8138 | 24 | 0.0735 | |
| LA | 0.4040 × DBH1.4193 | 0.7501 | 24 | 0.0969 |
| Stand | SF | CFP | F Values | p Values |
|---|---|---|---|---|
| N (trees·ha−1) | 930 ± 73 a | 1444 ± 342 a | 2.163 | 0.18 |
| DBH (cm) | 22.3 ± 0.3 a | 18.5 ± 0.8 b | 20.49 | 0.002 |
| H (m) | 12.0 ± 0.2 a | 12.4 ± 0.4 a | 0.686 | 0.432 |
| BA (m2·ha−1) | 31.5 ± 2.1 a | 33.1 ± 6.5 a | 0.053 | 0.824 |
| Volume (m3·ha−1) | 219.5 ± 14.8 a | 205.0 ± 37.5 a | 0.129 | 0.728 |
| LAI (m2·m−2) | 7.4 ± 0.5 a | 5.3 ± 1.0 a | 3.538 | 0.097 |
| Bulk density (g·cm−3) | 1.11 ± 0.03 a | 1.23 ± 0.01 b | 10.737 | 0.011 |
| Nitrogen (mg·g−1) | 2.10 ± 0.54 a | 1.33 ± 0.10 a | 1.929 | 0.202 |
| Porosity (%) | 37.48 ± 1.99 a | 42.78 ± 1.90 a | 3.604 | 0.094 |
| Soil type | Alfisol | Alfisol | 2.163 | 0.18 |
| Soil texture | Silty clay loam | Silty clay loam | 20.49 | 0.002 |
| Forest Type | Location | Species | Mean Age (Years) | Soil Depth (cm) | Soil/Ecosystem C | Ref. |
|---|---|---|---|---|---|---|
| P | Global | Global forests | 30 | 100 | 51% | [36] |
| NF | Global | Global forests | - | 100 | 45% | [36] |
| P | Template China | Pinus sylvestris var. mongolica | 15 | 60 | 75% | [44] |
| P | Template China | Populus simonii | 15 | 60 | 37% | [44] |
| MPNF | China | Deciduous forests | - | 100 | 69% | [45] |
| MPNF | China | Evergreen forests | - | 100 | 48% | [45] |
| P | Subtropical China | Erythrophleum fordii | 8 | 100 | 74% | [43] |
| P | Subtropical China | Castanopsis hystrix | 8 | 100 | 79% | [43] |
| P | Subtropical China | Mytilaria laosensis | 8 | 100 | 58% | [43] |
| P | Subtropical China | Mixed E. fordii, C. hytrix and M. laosensis | 8 | 100 | 63% | [43] |
| P | Subtropical China | Cunninghamia lanceolata | 22 | 100 | 53% | [46] |
| P | Subtropical China | Cunninghamia lanceolata | 13 | 60 | 74% | [47] |
| P | Subtropical China | Cunninghamia lanceolata | 15 | 60 | 62% | [48] |
| MPNF | Subtropical China | Subtropical forests | - | 100 | 44% | [49] |
| MPNF | Subtropical China | Subtropical forests | - | 100 | 88% | [50] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, S.; Guan, F.; Xu, X.; Forrester, D.I.; Ma, W.; Tang, X. Ecosystem Carbon Stock Loss after Land Use Change in Subtropical Forests in China. Forests 2016, 7, 142. https://doi.org/10.3390/f7070142
Fan S, Guan F, Xu X, Forrester DI, Ma W, Tang X. Ecosystem Carbon Stock Loss after Land Use Change in Subtropical Forests in China. Forests. 2016; 7(7):142. https://doi.org/10.3390/f7070142
Chicago/Turabian StyleFan, Shaohui, Fengying Guan, Xingliang Xu, David I. Forrester, Wu Ma, and Xiaolu Tang. 2016. "Ecosystem Carbon Stock Loss after Land Use Change in Subtropical Forests in China" Forests 7, no. 7: 142. https://doi.org/10.3390/f7070142
APA StyleFan, S., Guan, F., Xu, X., Forrester, D. I., Ma, W., & Tang, X. (2016). Ecosystem Carbon Stock Loss after Land Use Change in Subtropical Forests in China. Forests, 7(7), 142. https://doi.org/10.3390/f7070142







