Environmental Factors Effect on Stem Radial Variations of Picea crassifolia in Qilian Mountains, Northwestern China
Abstract
:1. Introduction
2. Materials and Methods
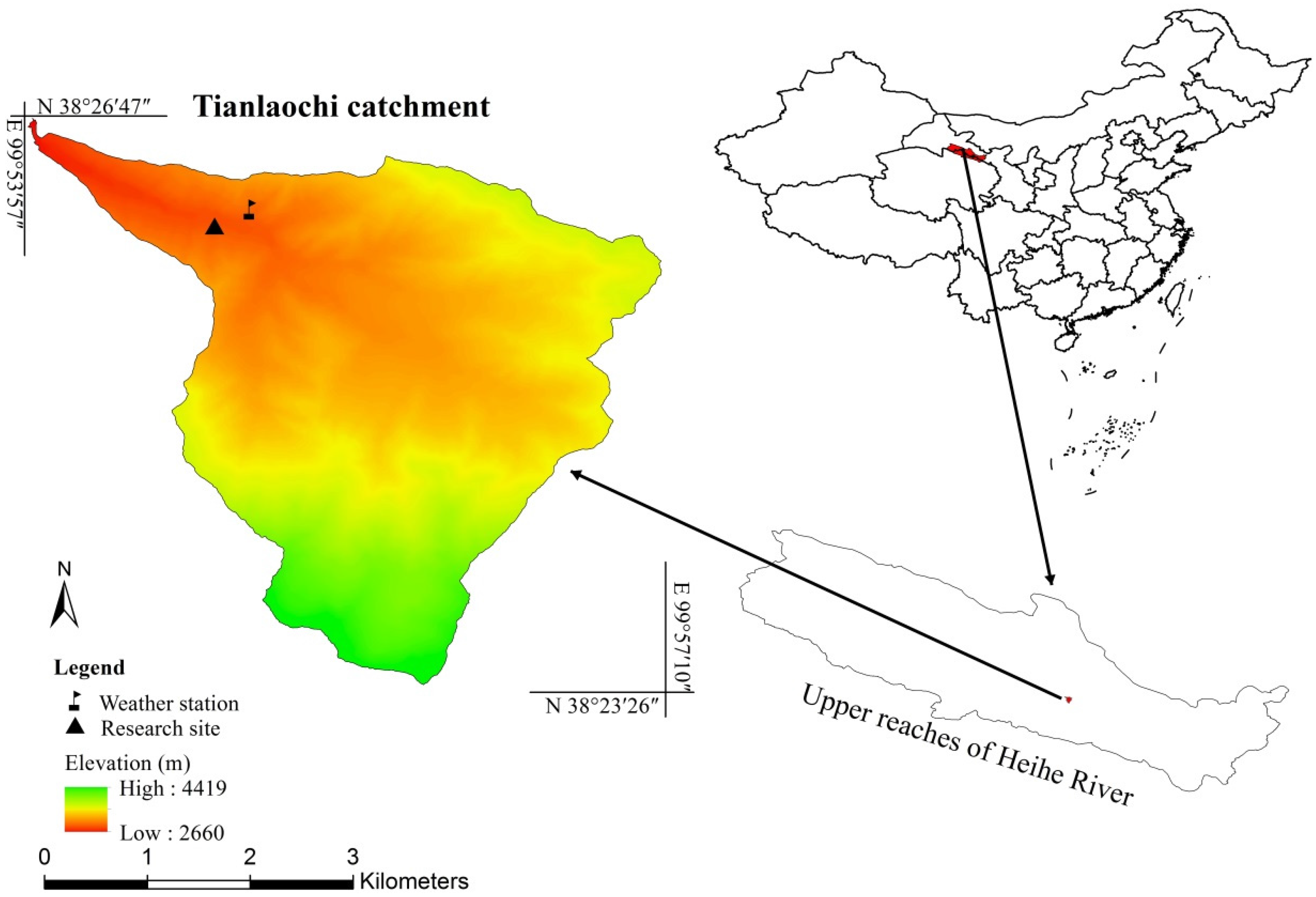
2.1. Study Area
2.2. Data Collection
2.2.1. Dendrometer Records
2.2.2. Environmental Data Collection
2.2.3. Micro-Core Sampling
2.3. Data Analysis
3. Results
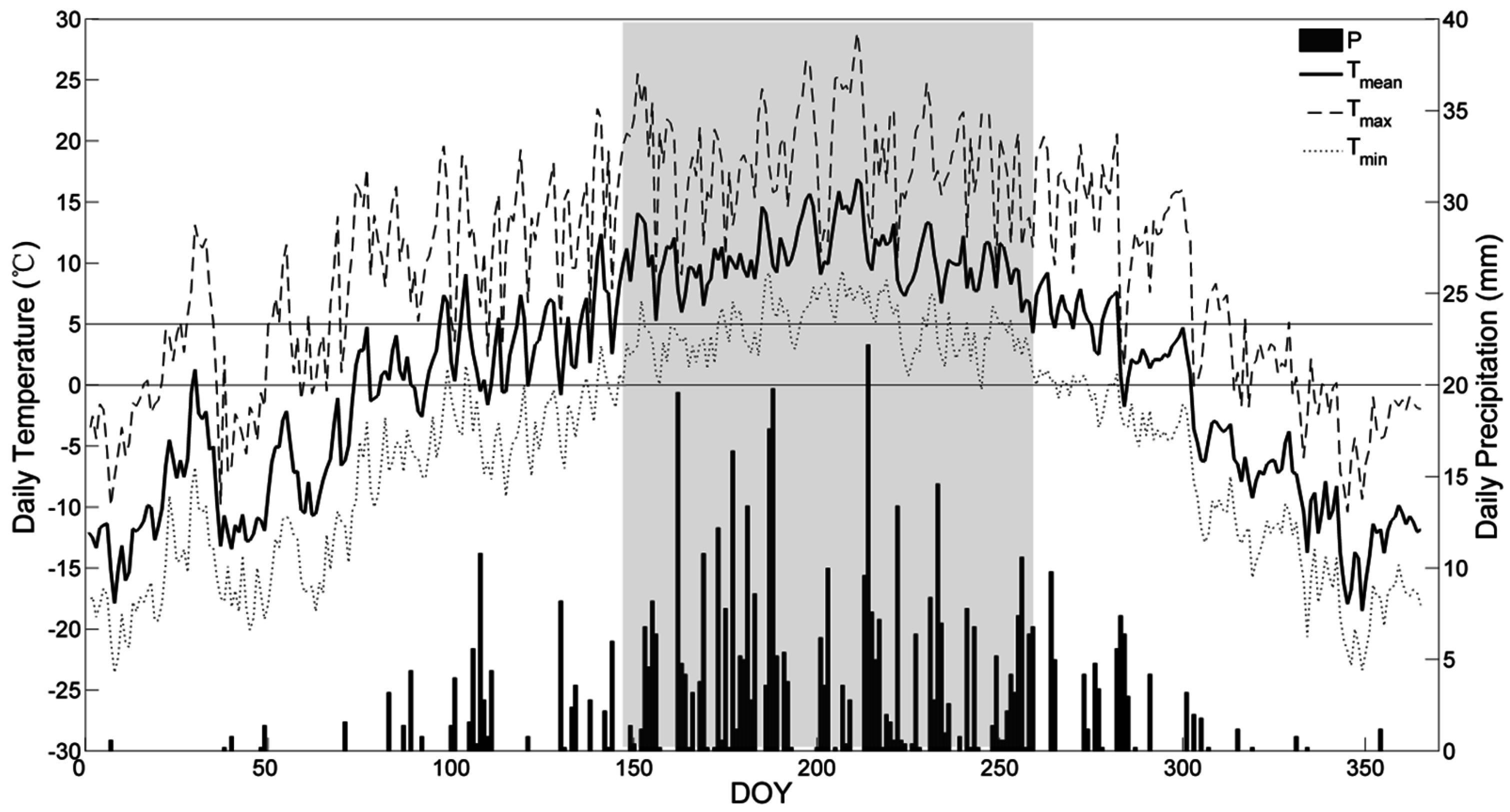
3.1. Variations of Environment Factors
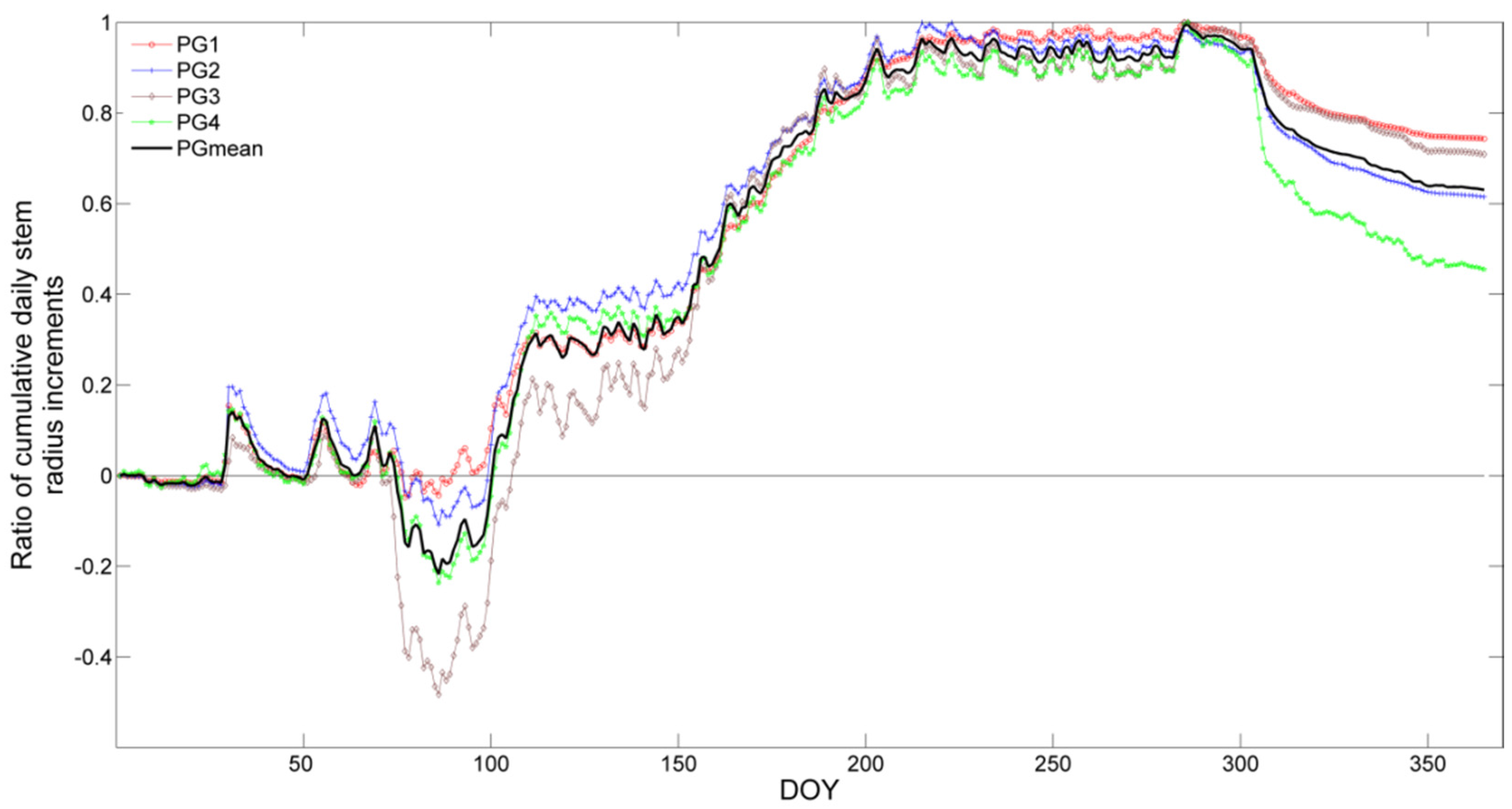
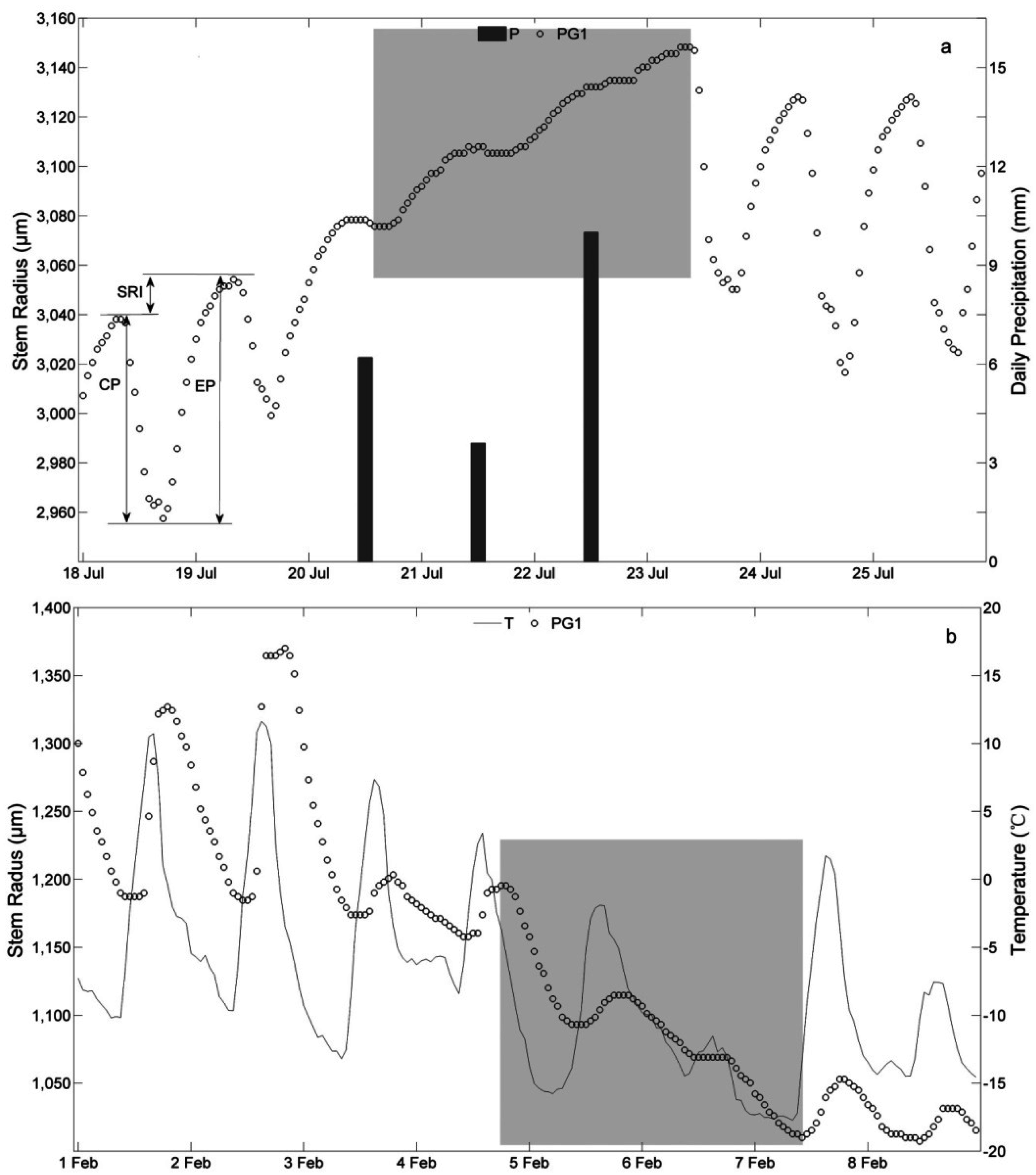
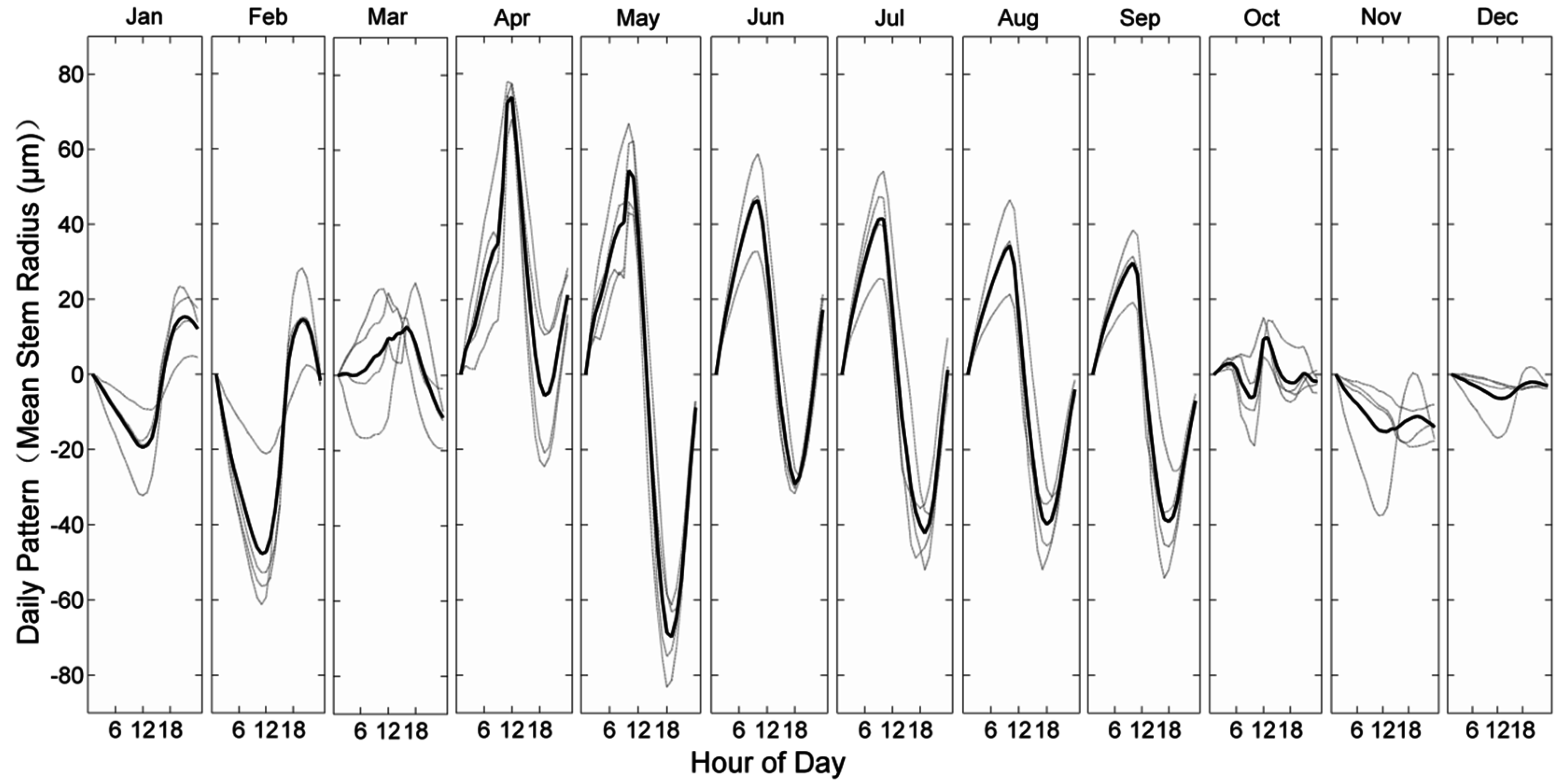
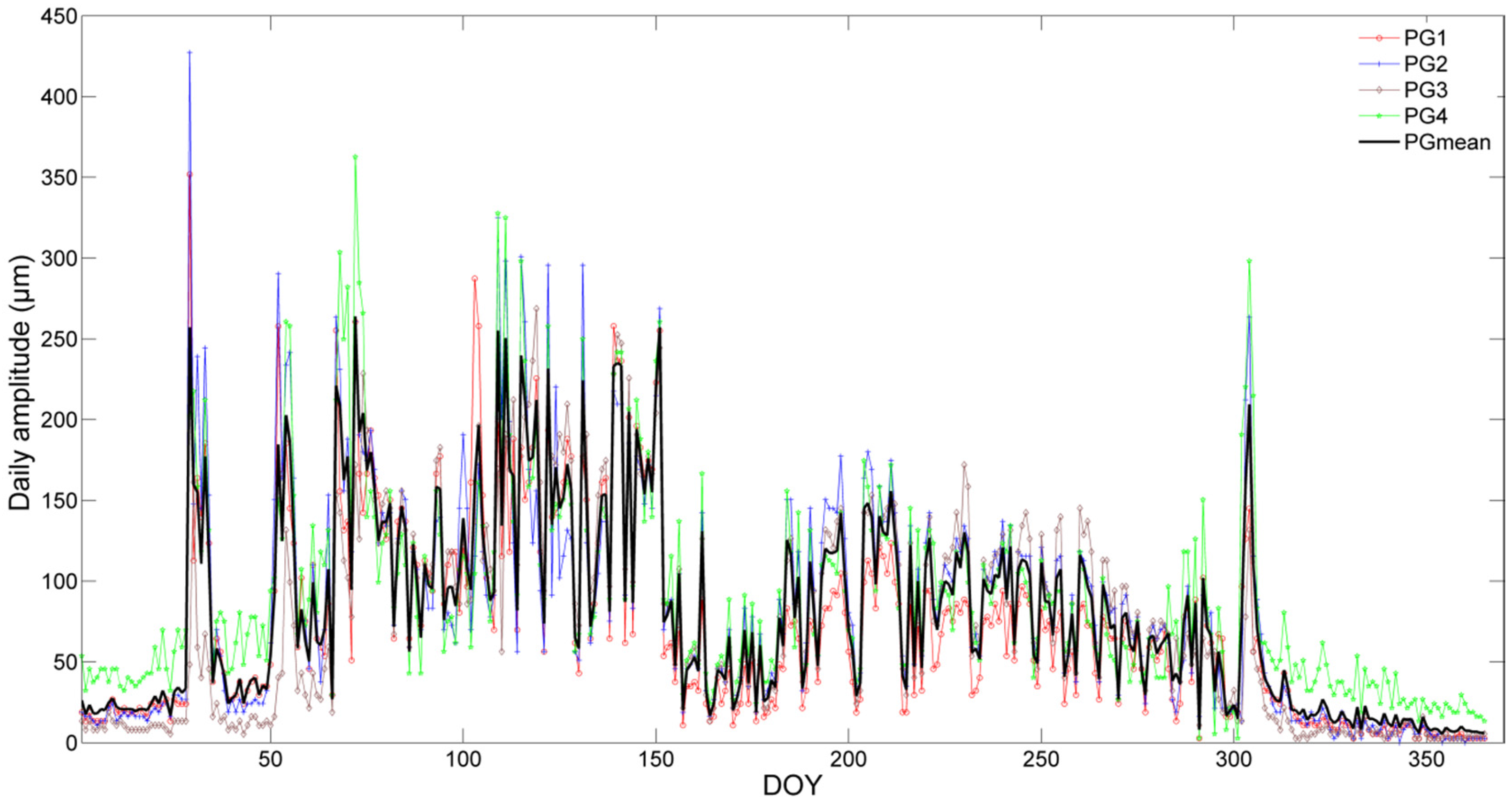
3.2. Stem Radial Variations
3.3. Determination of the Main Growing Period
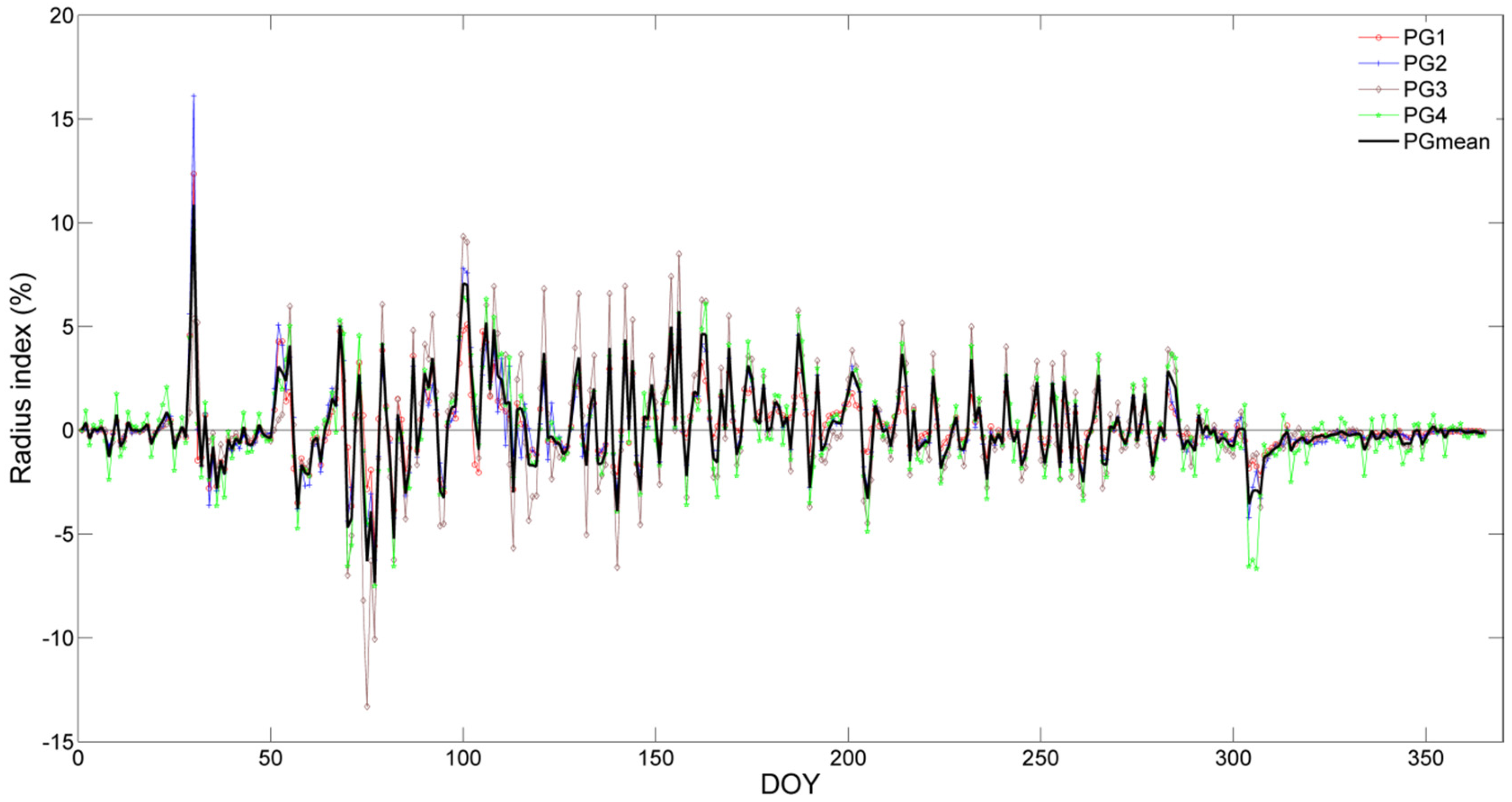
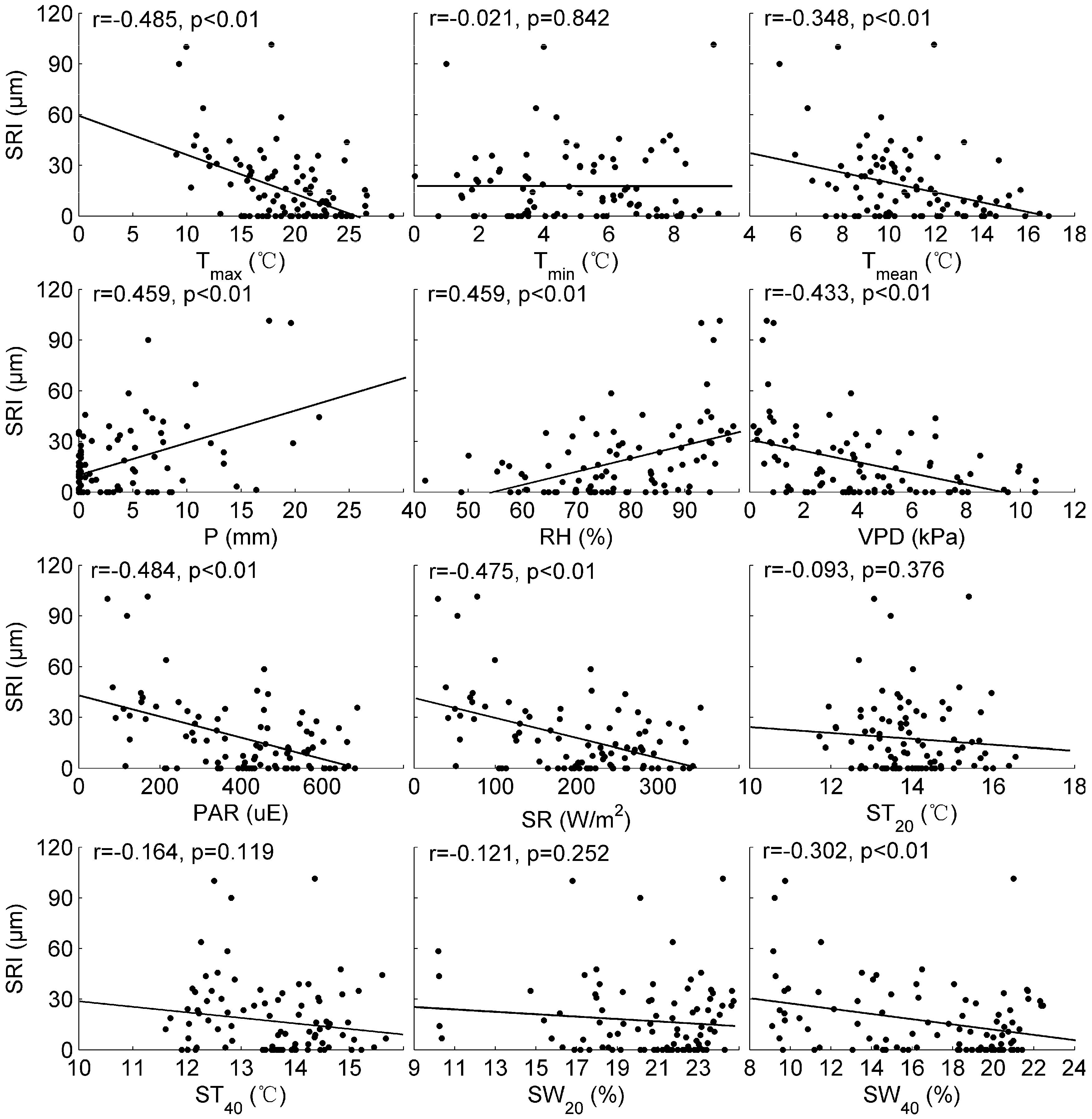
3.4. SRI and Its Relation to Environmental Factors
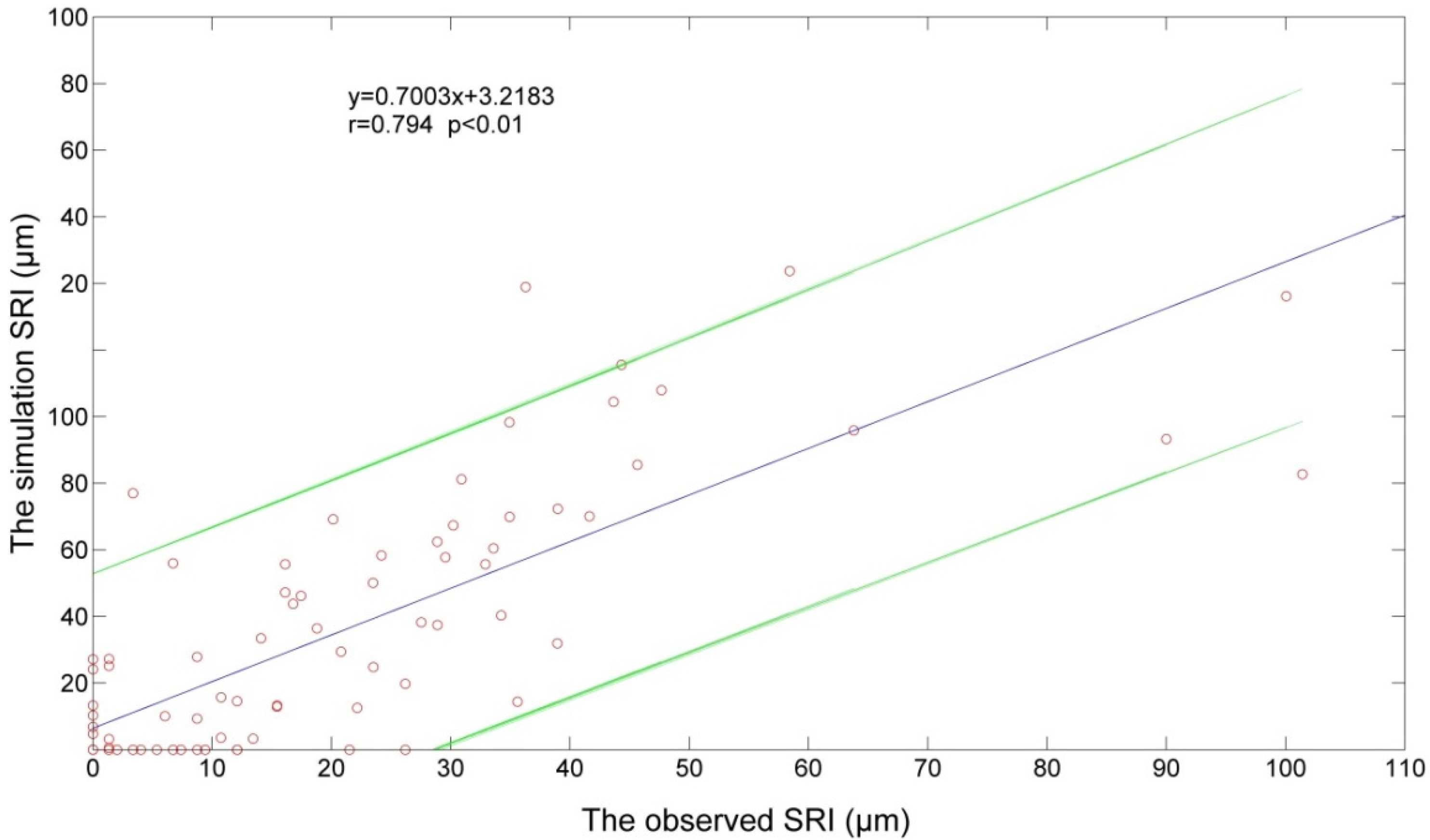
3.5. Model Development
4. Discussion
4.1. Stem Radius Variation
4.2. Determination of the Main Growing Period
4.3. The Relationship between SRI and Environmental Factors
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Y.; Guo, S.; Wang, J.; Hong, Y.; Bolin, X.; Duoyao, W. Estimation of forest ecosystem service value in the Qilian Mountains national nature reserve in Gansu of China. J. Desert Res. 2013, 33, 1905–1911. [Google Scholar]
- Tian, F.X.; Zhao, C.Y.; Feng, Z.D. Simulating evapotranspiration of Qinghai spruce (Picea crassifolia) forest in the Qilian Mountains, Northwestern China. J. Arid Environ. 2011, 75, 648–655. [Google Scholar] [CrossRef]
- Chang, X.; Zhao, W.; He, Z. Radial pattern of sap flow and response to microclimate and soil moisture in Qinghai spruce (Picea crassifolia) in the upper Heihe River basin of arid Northwestern China. Agric. For. Meteorol. 2014, 187, 14–21. [Google Scholar] [CrossRef]
- Peng, H.; Zhao, C.; Feng, Z.; Xu, Z.; Wang, C.; Zhao, Y. Canopy interception by a spruce forest in the upper reach of Heihe River basin, Northwestern China. Hydrol. Process 2014, 28, 1734–1741. [Google Scholar] [CrossRef]
- Zheng, X.; Zhao, C.; Peng, S.; Jian, S.; Liang, B.; Wang, X.; Yang, S.; Wang, C.; Peng, H.; Wang, Y. Soil CO2 efflux along an elevation gradient in Qinghai spruce forests in the upper reaches of the Heihe River, Northwest China. Environ. Earth Sci. 2013, 71, 2065–2076. [Google Scholar] [CrossRef]
- Peng, S.; Zhao, C.; Xu, Z. Modeling stem volume growth of Qinghai spruce (Picea crassifoliakom.) in Qilian Mountains of Northwest China. Scand. J. For. Res. 2015, 30, 1–9. [Google Scholar] [CrossRef]
- Peng, S.; Zhao, C.; Xu, Z.; Ashiq, M.W. Restoration and conservation potential of destroyed Qinghai spruce (Picea crassifolia) forests in the Qilian Mountains of Northwest China. Mitig. Adapt. Strateg. Glob. Chang. 2014, 21, 153–165. [Google Scholar] [CrossRef]
- Xu, Z.; Zhao, C.; Feng, Z. Species distribution models to estimate the deforested area of Picea crassifolia in arid region recently protected: Qilian Mts. National natural reserve (China). Pol. J. Ecol. 2012, 60, 515–524. [Google Scholar]
- Xu, Z.; Zhao, C.; Feng, Z.; Zhang, F.; Sher, H.; Wang, C.; Peng, H.; Wang, Y.; Zhao, Y.; Wang, Y.; et al. Estimating realized and potential carbon storage benefits from reforestation and afforestation under climate change: A case study of the Qinghai spruce forests in the Qilian Mountains, Northwestern China. Mitig. Adapt. Strateg. Glob. Chang. 2012, 18, 1257–1268. [Google Scholar] [CrossRef]
- Zhao, C.; Nan, Z.; Cheng, G.; Zhang, J.; Feng, Z. Gis-assisted modelling of the spatial distribution of Qinghai spruce (Picea crassifolia) in the Qilian Mountains, Northwestern China based on biophysical parameters. Ecol. Model. 2006, 191, 487–500. [Google Scholar] [CrossRef]
- Fu, B.; Wang, S.; Su, C.; Forsius, M. Linking ecosystem processes and ecosystem services. Curr. Opin. Environ. Sustain. 2013, 5, 4–10. [Google Scholar] [CrossRef]
- Liang, E.; Shao, X.; Eckstein, D.; Huang, L.; Liu, X. Topography- and species-dependent growth responses of Sabina przewalskii and Picea crassifolia to climate on the Northeast Tibetan Plateau. For. Ecol. Manag. 2006, 236, 268–277. [Google Scholar] [CrossRef]
- Zhang, Y.; Wilmking, M.; Gou, X. Changing relationships between tree growth and climate in Northwest China. Plant Ecol. 2008, 201, 39–50. [Google Scholar] [CrossRef]
- Zhang, Y.; Wilmking, M. Divergent growth responses and increasing temperature limitation of Qinghai spruce growth along an elevation gradient at the Northeast Tibet Plateau. For. Ecol. Manag. 2010, 260, 1076–1082. [Google Scholar] [CrossRef]
- Zhang, Y.; Shao, X.; Wilmking, M. Dynamic relationships between Picea crassifolia growth and climate at upper treeline in the Qilian Mts., Northeast Tibetan Plateau, China. Dendrochronologia 2011, 29, 185–199. [Google Scholar] [CrossRef]
- Wang, B.; Chen, T.; Wu, G.; Xu, G.; Zhang, Y.; Gao, H.; Zhang, Y.; Feng, Q. Qinghai spruce (Picea crassifolia) growth–climate response between lower and upper elevation gradient limits: A case study along a consistent slope in the mid-Qilian Mountains region. Environ. Earth Sci. 2016. [Google Scholar] [CrossRef]
- Cao, Z. Study on Nondestructive Measurement and Modeling Methods for Standing Tree Volume. Ph.D. Thesis, Beijing Forestry University, Beijing, China, 2015. [Google Scholar]
- Drew, D.M.; Downes, G.M. The use of precision dendrometers in research on daily stem size and wood property variation: A review. Dendrochronologia 2009, 27, 159–172. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, B.; Qin, C.; Shi, F. Research progress in monitoring and simulating stem radius growth: An overview. Sci. Cold Arid Reg. 2012, 4, 175–183. [Google Scholar]
- Deslauriers, A.; Morin, H.; Urbinati, C.; Carrer, M. Daily weather response of balsam fir (Abies balsamea (L.) Mill.) stem radius increment from dendrometer analysis in the boreal forests of Québec (Canada). Trees 2003, 17, 477–484. [Google Scholar] [CrossRef]
- Deslauriers, A.; Rossi, S.; Anfodillo, T. Dendrometer and intra-annual tree growth: What kind of information can be inferred? Dendrochronologia 2007, 25, 113–124. [Google Scholar] [CrossRef]
- Duchesne, L.; Houle, D.; D’Orangeville, L. Influence of climate on seasonal patterns of stem increment of balsam fir in a boreal forest of Québec, Canada. Agric. For. Meteorol. 2012, 162–163, 108–114. [Google Scholar] [CrossRef]
- Mäkinen, H.; Nöjd, P.; Saranpää, P. Seasonal changes in stem radius and production of new tracheids in Norway spruce. Tree Physiol. 2003, 23, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yang, B.; Deslauriers, A.; Bräuning, A. Intra-annual stem radial increment response of Qilian juniper to temperature and precipitation along an altitudinal gradient in Northwestern China. Trees 2014, 29, 25–34. [Google Scholar] [CrossRef]
- Korpela, M.; Mäkinen, H.; Nöjd, P.; Hollmén, J.; Sulkava, M. Automatic detection of onset and cessation of tree stem radius increase using dendrometer data. Neurocomputing 2010, 73, 2039–2046. [Google Scholar] [CrossRef]
- Kozlowski, T.T.; Winget, C.H. Diurnal and seasonal variation in radii of tree stems. Ecology 1964, 45, 149–155. [Google Scholar] [CrossRef]
- Herzog, K.M.; Häsler, R.; Thum, R. Diurnal changes in the radius of a subalpine Norway spruce stem: Their relation to the sap flow and their use to estimate transpiration. Trees 1995, 10, 94–101. [Google Scholar] [CrossRef]
- Offenthaler, I.; Hietz, P.; Richter, H. Wood diameter indicates diurnal and long-term patterns of xylem water potential in Norway spruce. Trees 2001, 15, 215–221. [Google Scholar] [CrossRef]
- Downes, G.; Beadle, C.; Worledge, D. Daily stem growth patterns in irrigated Eucalyptus globulus and E. nitens in relation to climate. Trees 1999, 14, 102–111. [Google Scholar] [CrossRef]
- Tardif, J.; Flannigan, M.; Bergeron, Y. An analysis of the daily radial activity of 7 boreal tree species, Northwestern Québec. Environ. Monit. Assess. 2001, 67, 141–160. [Google Scholar] [CrossRef] [PubMed]
- Fedör Tatarinov, J.Č. Daily and seasonal variation of stem radius in oak. Ann. For. Sci. 1999, 56, 579–590. [Google Scholar] [CrossRef]
- Bouriaud, O.; Leban, J.M.; Bert, D.; Deleuze, C. Intra-annual variations in climate influence growth and wood density of Norway spruce. Tree Physiol. 2005, 25, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Turcotte, A.; Morin, H.; Krause, C.; Deslauriers, A.; Martel, M.T. The timing of spring rehydration and its relation with the onset of wood formation in black spruce. Agric. For. Meteorol. 2009, 149, 1403–1409. [Google Scholar] [CrossRef]
- Sulkava, M.; Mäkinen, H.; Nöjd, P.; Hollmén, J. Automatic detection of onset and cessation of tree stem radius increase using dendrometer data and CUSUM charts. In Proceedings of European Symposium on Time Series Prediction–ESTSP; Helsinki University of Technology: Helsinki, Finland, 2008; pp. 77–86. Available online: http://research.ics.aalto.fi/publications/sulkava/Sulkava2008b.pdf (accessed on 13th September 2016). [Google Scholar]
- Mäkinen, H.; Seo, J.-W.; Nöjd, P.; Schmitt, U.; Jalkanen, R. Seasonal dynamics of wood formation: A comparison between pinning, microcoring and dendrometer measurements. Eur. J. For. Res. 2008, 127, 235–245. [Google Scholar] [CrossRef]
- Camarero, J.J.; Olano, J.M.; Parras, A. Plastic bimodal xylogenesis in conifers from continental Mediterranean climates. New Phytol. 2010, 185, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.; Holtta, T.; Berninger, F.; Makinen, H.; Nojd, P.; Mencuccini, M.; Nikinmaa, E. Separating water-potential induced swelling and shrinking from measured radial stem variations reveals a cambial growth and osmotic concentration signal. Plant Cell Environ. 2016, 39, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Cocozza, C.; Palombo, C.; Tognetti, R.; La Porta, N.; Anichini, M.; Giovannelli, A.; Emiliani, G. Monitoring intra-annual dynamics of wood formation with microcores and dendrometers in Picea abies at two different altitudes. Tree Physiol. 2016, 36, 832–846. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Yuan, Y.J.; Wei, W.S.; Yu, S.L.; Fan, Z.A.; Zhang, R.B.; Zhang, T.W.; Li, Q.; Shang, H.M. Temperature reconstruction from tree-ring maximum latewood density of Qinghai spruce in middle Hexi Corridor, China. Theor. Appl. Climatol. 2011, 107, 633–643. [Google Scholar] [CrossRef]
- Zhang, Z.; Wu, X. Utilizing two Qinghai tree-ring chronologies to reconstruct and analyze local historical precipitation. Q. J. Appl. Meteorol. 1992, 3, 61–69. [Google Scholar]
- Gou, X.-H.; Chen, F.-H.; Yang, M.-X.; Peng, J.-F.; Qiang, W.-Y.; Tuo, C. Analysis of the tree-ring width chronology of Qilian Mountains at different elevation. Acta Ecol. Sin. 2004, 24, 172–176. [Google Scholar]
- Biondi, F.; Hartsough, P. Using automated point dendrometers to analyze tropical treeline stem growth at Nevado de Colima, Mexico. Sensors 2010, 10, 5827–5844. [Google Scholar] [CrossRef] [PubMed]
- Irvine, J.; Grace, J. Continuous measurements of water tensions in the xylem of trees based on the elastic properties of wood. Planta 1997, 202, 455–461. [Google Scholar] [CrossRef]
- Murray, F.W. On the computation of saturation vapor pressure. J. Appl. Meteorol. 1967, 6, 203–204. [Google Scholar] [CrossRef]
- Deslauriers, A.; Rossi, S.; Turcotte, A.; Morin, H.; Krause, C. A three-step procedure in SAS to analyze the time series from automatic dendrometers. Dendrochronologia 2011, 29, 151–161. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, B.; Deslauriers, A.; Qin, C.; He, M.; Shi, F.; Liu, J. Two phases of seasonal stem radius variations of Sabina przewalskii kom. in Northwestern China inferred from sub-diurnal shrinkage and expansion patterns. Trees 2012, 26, 1747–1757. [Google Scholar] [CrossRef]
- Zweifel, R.; Item, H.; Häsler, R. Link between diurnal stem radius changes and tree water relations. Tree Physiol. 2001, 21, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Nakai, T. Effect of the water status within a tree on tracheid morphogenesis in Cryptomeria japonica D. Don. Trees 1999, 14, 124–129. [Google Scholar]
- Sevanto, S.; Suni, T.; Pumpanen, J.; Grönholm, T.; Kolari, P.; Nikinmaa, E.; Hari, P.; Vesala, T. Wintertime photosynthesis and water uptake in a boreal forest. Tree Physiol. 2005, 26, 749–757. [Google Scholar] [CrossRef]
- Zweifel, R.; Häsler, R. Frost-induced reversible shrinkage of bark of mature subalpine conifers. Agric. For. Meteorol. 2000, 102, 213–222. [Google Scholar] [CrossRef]
- King, G.; Fonti, P.; Nievergelt, D.; Büntgen, U.; Frank, D. Climatic drivers of hourly to yearly tree radius variations along a 6 °C natural warming gradient. Agric. For. Meteorol. 2013, 168, 36–46. [Google Scholar] [CrossRef]
- Devine, W.D.; Harrington, C.A. Factors affecting diurnal stem contraction in young Douglas-fir. Agric. For. Meteorol. 2011, 151, 414–419. [Google Scholar] [CrossRef]
- Zweifel, R.; Häsler, R. Dynamics of water storage in mature subalpine Picea abies: Tempotal and spatial patterns of change in stem radius. Tree Physiol. 2001, 21, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Deslauriers, A.; Anfodillo, T. Assessment of cambial activity and xylogenesis by microsampling tree species: An example at the alpine timberline. IAWA J. 2006, 27, 383–394. [Google Scholar] [CrossRef]
- Bäucker, E.; Bues, C.-T.; Vogel, M. Radial growth dynamics of spruce (Picea abies) measured by micro-cores. IAWA J. 1998, 19, 301–309. [Google Scholar] [CrossRef]
- Körner, C. A re-assessment of high elevation treeline positions and their explanation. Oecologia 1998, 115, 445–459. [Google Scholar] [CrossRef]
- Rossi, S.; Deslauriers, A.; Anfodillo, T.; Carraro, V. Evidence of threshold temperatures for xylogenesis in conifers at high altitudes. Oecologia 2007, 152, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Deslauriers, A.; Griçar, J.; Seo, J.-W.; Rathgeber, C.B.K.; Anfodillo, T.; Morin, H.; Levanic, T.; Oven, P.; Jalkanen, R. Critical temperatures for xylogenesis in conifers of cold climates. Glob. Ecol. Biogeogr. 2008, 17, 696–707. [Google Scholar] [CrossRef]
- Lenz, A.; Hoch, G.; Körner, C. Early season temperature controls cambial activity and total tree ring width at the alpine treeline. Plant Ecol. Divers. 2013, 6, 365–375. [Google Scholar] [CrossRef]
- Proseus, T.E.; Boyer, J.S. Turgor pressure moves polysaccharides into growing cell walls of Chara corallina. Ann. Bot. 2005, 95, 967–979. [Google Scholar] [CrossRef] [PubMed]
- Zweifel, R.; Zimmermann, L.; Zeugin, F.; Newbery, D.M. Intra-annual radial growth and water relations of trees: Implications towards a growth mechanism. J. Exp. Bot. 2006, 57, 1445–1459. [Google Scholar] [CrossRef] [PubMed]
- Köcher, P.; Horna, V.; Leuschner, C. Environmental control of daily stem growth patterns in five temperate broad-leaved tree species. Tree Physiol. 2012, 32, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Liang, E.; Eckstein, D.; Liu, H. Climate-growth relationships of relict Pinus tabulaeformis at the northern limit of its natural distribution in Northern China. J. Veg. Sci. 2008, 19, 393–406. [Google Scholar] [CrossRef]
- Ripullone, F.; Guerrieri, M.R.; Nole’, A.; Magnani, F.; Borghetti, M. Stomatal conductance and leaf water potential responses to hydraulic conductance variation in Pinus pinaster seedlings. Trees 2007, 21, 371–378. [Google Scholar] [CrossRef]
- Michelot, A.; Simard, S.; Rathgeber, C.; Dufrene, E.; Damesin, C. Comparing the intra-annual wood formation of three european species (Fagus sylvatica, Quercus petraea and Pinus sylvestris) as related to leaf phenology and non-structural carbohydrate dynamics. Tree Physiol. 2012, 32, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Wang, B.-Q.; Dong, M.-Y.; Huang, Y.-M.; Wang, M.-C.; Wang, B. Response of daily stem radial growth of Platycladus orientalis to environmental factors in a semi-arid area of North China. Trees 2014, 29, 87–96. [Google Scholar] [CrossRef]
- Paoletti, E.; Bytnerowicz, A.; Andersen, C.; Augustaitis, A.; Ferretti, M.; Grulke, N.; Gunthardt-Goerg, M.S.; Innes, J.; Johnson, D.; Karnosky, D.; et al. Impacts of air pollution and climate change on forest ecosystems–emerging research needs. Sci. World J. 2007, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Matyssek, R.; Bytnerowicz, A.; Karlsson, P.E.; Paoletti, E.; Sanz, M.; Schaub, M.; Wieser, G. Promoting the O3 flux concept for European forest trees. Environ. Pollut. 2007, 146, 587–607. [Google Scholar] [CrossRef] [PubMed]
- Buker, P.; Feng, Z.; Uddling, J.; Briolat, A.; Alonso, R.; Braun, S.; Elvira, S.; Gerosa, G.; Karlsson, P.E.; Le Thiec, D.; et al. New flux based dose-response relationships for ozone for European forest tree species. Environ. Pollut. 2015, 206, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Pretzsch, H.; Dieler, J.; Matyssek, R.; Wipfler, P. Tree and stand growth of mature Norway spruce and European beech under long-term ozone fumigation. Environ. Pollut. 2010, 158, 1061–1070. [Google Scholar] [CrossRef] [PubMed]
- Assis, P.I.; Alonso, R.; Meirelles, S.T.; Moraes, R.M. DO3SE model applicability and O3 flux performance compared to AOT40 for an O3-sensitive tropical tree species (Psidium guajava L. ‘Paluma’). Environ. Sci. Pollut. Res. Int. 2015, 22, 10873–10881. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, J.; Mauzerall, D.L.; Emmons, L.K.; Walters, S.; Horowitz, L.W.; Tao, S. Effects of trans-eurasian transport of air pollutants on surface ozone concentrations over Western China. J. Geophys. Res. Atmos. 2014, 119, 12338–12354. [Google Scholar] [CrossRef]
- Duchesne, L.; Houle, D. Modelling day-to-day stem diameter variation and annual growth of balsam fir (Abies balsamea (L.) Mill.) from daily climate. For. Ecol. Manag. 2011, 262, 863–872. [Google Scholar] [CrossRef]


| No. | Height (m) | DBH (cm) | Age (Year) | Crown Diameter (m) |
|---|---|---|---|---|
| PG 1 | 13.6 | 23.40 | 101 | 4.43 |
| PG 2 | 14.4 | 27.34 | 126 | 5.21 |
| PG 3 | 22.8 | 37.50 | 130 | 4.69 |
| PG 4 | 19.0 | 37.11 | 163 | 4.55 |
| M 1 | 20.5 | 34.38 | 139 | 4.78 |
| M 2 | 22.1 | 33.10 | 125 | 4.59 |
| M 3 | 14.0 | 28.58 | 123 | 4.32 |
| M 4 | 15.9 | 28.23 | 118 | 4.07 |
| Phases | DOY | Temperature Characteristics | Amplitude (μm) | Absolute Values of RI (%) |
|---|---|---|---|---|
| Strong variation phase | 51–147 260–302 | Tmax > 5 °C Tmin < 0 °C Tmean > 0 °C | 114.32 ± 5.10 | 1.86 ± 0.13 |
| Weak variation phase | 1–50 303–365 | Tmax < 5 °C Tmin < 0 °C Tmean < 0 °C | 33.08 ± 3.94 | 0.71 ± 0.11 |
| Moderate variation phase | 148–259 | Tmax > 5 °C Tmin > 0 °C Tmean > 5 °C | 84.75 ± 4.07 | 1.44 ± 0.11 |
| Model Summary | |||||
|---|---|---|---|---|---|
| R | R Square | Adjusted R Square | Std. Error of the Estimate | p-Value | Durbin-Watson |
| 0.808 | 0.653 | 0.633 | 17.367 | <0.01 | 2.05 |
| Coefficients | |||||
| Variable | B | Std. Error | t | p-Value | VIF |
| Constant | 31.930 | 32.023 | 0.997 | 0.322 | |
| Tmax | −1.391 | 0.854 | −1.630 | 0.107 | 4.37 |
| RH | 1.496 | 0.276 | 5.419 | <0.01 | 3.81 |
| PAR | 0.052 | 0.022 | 2.375 | 0.020 | 3.92 |
| RH(t − 2) | −1.441 | 0.159 | −9.075 | <0.01 | 1.35 |
| SWC40(t − 2) | −1.213 | 0.506 | −2.397 | 0.019 | 1.41 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Zhang, F.; Yuan, L.; Wang, Q.; Zheng, K.; Zhao, C. Environmental Factors Effect on Stem Radial Variations of Picea crassifolia in Qilian Mountains, Northwestern China. Forests 2016, 7, 210. https://doi.org/10.3390/f7100210
Wang W, Zhang F, Yuan L, Wang Q, Zheng K, Zhao C. Environmental Factors Effect on Stem Radial Variations of Picea crassifolia in Qilian Mountains, Northwestern China. Forests. 2016; 7(10):210. https://doi.org/10.3390/f7100210
Chicago/Turabian StyleWang, Wenbin, Fen Zhang, Liming Yuan, Qingtao Wang, Kai Zheng, and Chuanyan Zhao. 2016. "Environmental Factors Effect on Stem Radial Variations of Picea crassifolia in Qilian Mountains, Northwestern China" Forests 7, no. 10: 210. https://doi.org/10.3390/f7100210
APA StyleWang, W., Zhang, F., Yuan, L., Wang, Q., Zheng, K., & Zhao, C. (2016). Environmental Factors Effect on Stem Radial Variations of Picea crassifolia in Qilian Mountains, Northwestern China. Forests, 7(10), 210. https://doi.org/10.3390/f7100210






