1. Introduction
It is now widely accepted that global climate change is taking place as a result of human activity. Although the influence of a changing climate may be less severe in Scotland than that predicted for some regions of the world, there will be environmental and economic consequences including the potential impact on the commercially important forests of Sitka spruce (Picea sitchensis (Bong.) Carr.). This species dominates the forestry sector in Scotland accounting for over half of the total forest area of conifers of nearly one million hectares. It has long been favoured by the forestry industry due to its adaptability to a wide range of site types, relative ease of establishment, good growth rate, tolerance of exposure, and timber suited to a wide range of end uses such as construction, board manufacture and pulp. Not surprisingly, foresters and forest owners are increasingly concerned about the putative effects of climate change on Sitka spruce, and that decisions over the direction of future management cannot be delayed since the long timescales trees require to mature are well within the time periods of predicted change.
Sitka spruce has traditionally been managed in Scotland predominately as pure stands using clear-cutting and replanting that is simple to apply and effective at producing sawlogs in relatively short rotations of around 35–50 years. Foresters have long been aware that depending so heavily on one species managed as pure, even-aged stands has risks even before considering the potential consequences of climate change. Climate is a complex process and it can only be speculated at what changes in annual mean as well as seasonal variation in temperature, solar radiation (influenced by cloud cover), moisture and atmospheric CO
2 will have on the wider environment and how trees and forests will respond. Current evidence based on UK Climate Projections (UKCP09) suggests that by the end of the 21st century, the climate in Scotland will become warmer throughout the year with drier summers, particularly in eastern districts, and wetter winters [
1]. It has also been suggested that more frequent strong to storm force winds may occur particularly during the winter months [
2], increasing the risk of forest damage and timber loss [
3].
A warmer climate may improve growth of Sitka spruce where moisture and nutrients are not limiting [
4] and similar responses have been observed in other tree species and forest types [
5]. However, there are likely to be negative impacts on growth particularly in areas that become more prone to drought [
6]. A warmer climate may also lead to a greater susceptibility to biotic attack and there is already evidence that climate change is influencing the occurrence of pests and diseases leading to tree mortality [
7]. Higher atmospheric CO
2 concentration has generally been shown to have a positive impact on tree growth, but it remains uncertain whether rising CO
2 levels in the atmosphere will continue to cause increases in forest growth and carbon storage [
5]. Generally, climate models predict an increased risk of more extreme climatic events.
While climate change models may provide information on generalised future changes to the environment, they do not have the level of accuracy and precision required by foresters and other natural resource managers to make informed decisions [
8]. Indeed, there is absolutely no guarantee that any of these predictions will be realised and, given the scale of uncertainty, foresters will have to consider adaptive management strategies that maintain the ability to change silvicultural direction in forest stands that take account of the possibly that future climate change scenarios may not occur as predicted, or even happen at all. Nevertheless, managing for an uncertain future requires the “worse-case scenarios” suggested by climate change models to be taken into account, but at the same time not discounting the possibility that alternative patterns of change may occur. This review considers the potential impact of climate change on Sitka spruce forests in Scotland on future distribution of the species and potential risks of attack by pests and diseases. It examines silvicultural options that promote resilience in Sitka spruce stands and forests to potential climate change effects while maintaining management flexibility.
2. Geographic Distribution
Perhaps the key issue facing foresters is whether the tree species that support a large proportion of the timber processing sector are sufficiently adaptable to changing climatic conditions for their continued use. Given the long timescale required for new field trials to provide the necessary information, decisions on species and provenance suitability largely rely on observations made on stands currently growing under range of climatic conditions and matching these to a future climatic environment predicted for some other site [
9]. This information can be used along with climate change models to identify potentially suitable environmental conditions for particular species in the future [
10]. For non-native species such as Sitka spruce, information can also be gained by studying growth performance in their natural habitat.
Sitka spruce is native to the Pacific forests of North-West America forming a narrow coastal strip stretching from Alaska to California. The provenance of Sitka spruce grown in Scotland is almost entirely from Queen Charlotte Islands, Canada and is favoured due to its adaptability to the cool maritime conditions found here. Southern provenances, such as Washington and Oregon, have been found to be susceptible to autumn frost damage in comparison to Queen Charlotte Islands [
11], and although the incidence of frost is predicted to decline in the future, sufficient risks remain to continue with Queen Charlotte Islands as the preferred option [
2]. Sitka spruce, in common with most spruce species, requires a relatively high soil moisture status for good growth with an annual precipitation typically around 1000 mm or greater [
12]. Sitka spruce can grow reasonably well with an annual rainfall of as low a 700–800 mm [
13], but this would require consistent moisture availability particularly during the growing season and this cannot be guaranteed under predicted changes in the patterns of rainfall. The low annual rainfall currently experienced in parts of eastern Scotland already limits the planting of Sitka spruce and stem crack and tree death have been reported as a result of recent very dry summers [
14]. Where Sitka spruce is already showing signs of drought stress, then more drought tolerant species such as pines (
Pinus spp.) should be used to replace spruce, although even pines may suffer if the drought conditions become increasingly severe [
6]. Sitka spruce may also come under stress on shallow, poorly drained soils that may experience waterlogging in the winter then suffer an extended period of drought in the summer [
2].
West and north Scotland is predicted to be wetter in the future and therefore least sensitive to drought and it has been suggested that forest expansion using Sitka spruce should be targeted in these areas [
6]. While climate models suggest a general warming of the atmosphere, there is an alternative scenario in which increased precipitation and rainfall frequency—predicted with continued climatic warming—may in fact cause in a
reduction in the temperature range in northern maritime regions due to increased cloud cover that may lead to increasing water-table levels and ultimately to an expansion of bog [
15], which in turn would limit where Sitka spruce can be potentially grown. Given the wide range of future climatic scenarios, it is only possible to speculate on the geographic shift where Sitka spruce may grow well.
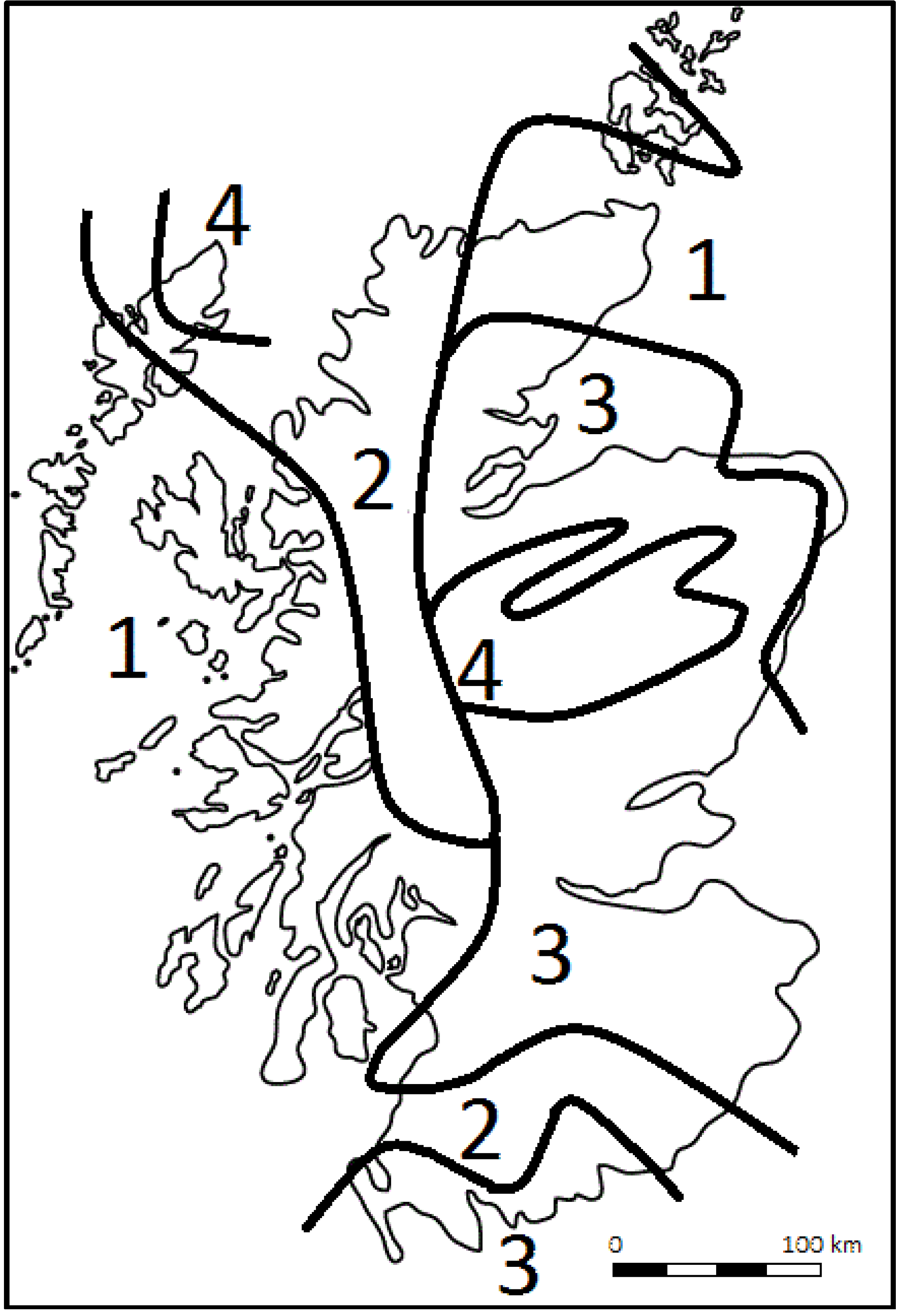
Figure 1 illustrates one scenario (based on UKCIP02 high emissions highlighting a more extreme case) for the predicted shift in geographic suitability of Sitka spruce in 2080 (based on Ray [
2]).
Figure 1.
Sketch map of zones showing current and predicted suitability zones for Sitka spruce in Scotland by 2080. (1) remains largely suitable; (2) partially suitable becoming more suitable; (3) partially suitable becoming largely unsuitable; (4) remains largely unsuitable. Based on a simplified version of data presented in Ray [
2].
Figure 1.
Sketch map of zones showing current and predicted suitability zones for Sitka spruce in Scotland by 2080. (1) remains largely suitable; (2) partially suitable becoming more suitable; (3) partially suitable becoming largely unsuitable; (4) remains largely unsuitable. Based on a simplified version of data presented in Ray [
2].
Much of the Sitka spruce planted in Scotland in recent decades is selected stock derived from a long-term breeding programme with the aim of improving growth rate, stem form and wood density in comparison with the base population of Queen Charlotte Islands origin. Seed orchards were established using 40 superior genotypes, drawn from an initial selection of 1800 “plus” trees [
16], from which seed was derived for the production of forest plants. Since the 1990s, the process has been further refined with seed being produced from controlled crosses between selected parents from around 20 mother trees and a mixture of pollen from a similar number of unrelated father trees. In spite of concerns that the breeding programme may reduce genetic diversity and become less adaptable to potential climate change, trees produced from these crosses are likely to have similar levels of genetic diversity to trees produced from seed orchard seed and likely to be as diverse as unimproved collections from the North-West America [
17]. The breeding programme has been further refined to use “full sibling families” with a single known male and female tested parent [
16]. While there may be advantages in the superior performance of these trees, foresters need to be aware of the risks in reducing the genetic diversity that may result in spruce stands becoming less able to adapt to future climate change [
17]. Overall, the evidence suggests caution at significant changes to provenance choice of Sitka spruce and that the use of genetically improved planting stock should continue as long as the breeding programme takes account of the need to maintain genetic diversity within the population.
3. Susceptibility to Pests and Diseases
One of the greatest threats to the global forestry industry is the increase in attacks from pests and diseases thought to be associated with a changing climate, although international trading undoubtedly contributes to the risk of attack by alien invasive species. There is a high probability of pests and diseases becoming more predominant in Scotland where winters are milder and summers more prone to droughts increasing levels of stress [
18]. Already two major forest tree diseases of the genus
Dothistroma and
Phytophthora have affected large areas of forest in Scotland.
Dothistroma septosporum (Dorog.) has had a major impact on lodgepole pine (
Pinus contorta Dougl.) causing widespread death of this species. While the global spread of
Dothistroma septosporum is thought to be linked to a change in climate that promotes its spread [
19], the intensity of infection is thought be linked to the formation of large plantations of highly susceptible species [
7].
Species of
Phytophthora are causing major damage to tree species in many parts of the world. Over the last half century, observed increases in mean winter temperatures and a tendency for drier summers and wetter winters with heavier rainfall has favoured infection by several
Phytophthora species in Central Europe [
7].
Phytophtora ramorum (Werres, De Cock & Man in’t Veld) has already had a devastating effect on larches (
Larix species) wiping out large areas in south and west Scotland. A warmer climate is thought to favour the spread of pests and diseases northwards and in elevation that would have normally succumbed to winter cold. In addition, a changing climate may stress trees that then become more susceptible to native and introduced pests and diseases [
7]. To date, Sitka spruce has shown remarkable resilience to biotic damage. The great spruce bark beetle (
Dendroctonus micans Kug.) caused concern when first reported in Britain in 1982, but in spite of initial fears, damage has remained localised and has not become a major threat. However, warmer temperatures could promote its spread throughout the entire spruce range and that spruce stands subject to increasing drought stress, as is predicted for parts of eastern Scotland, may succumb to
Dendroctonus [
18].
The green spruce aphid (
Elatobium abietinum Walker) can cause significant defoliation of trees from time to time, but attacked trees appear to recover [
20]. Severe outbreaks of the green aphid have been linked to mild winters and can result in volume loss [
21]. Nevertheless, it cannot be ruled out that future milder winters may benefit the green spruce aphid and that attacks on Sitka spruce may have a greater impact on growth, particularly in eastern part of Scotland where drought stress is predicted to become more frequent [
18,
22]. There have been periodic outbreaks of the winter moth (
Operophtera brumata L.) on Sitka spruce in Scotland. These depend on the synchrony of the eggs hatching with the time of new spruce shoots flushing in spring on which the larva feed. Under climate warming, the emergence of the moth was predicted to occur earlier, but it appears that the date of budburst of Sitka spruce does not greatly change resulting in decreased synchrony between the two events [
23]. The authors suggest that synchrony between different types of temperature-driven phenologies may become poorer with climatic warming, although the moth may evolve to adapt to the asynchrony.
Even although Sitka spruce has survived largely unscathed from attack by pests and diseases, the risk remains that currently unknown threats may appear [
24] with potentially devastating economic consequences given the scale of the processing industry that it supports. For example, a potential new threat to Sitka spruce is the European spruce bark beetle (
Ips typographus L.) that has killed living spruce trees during outbreaks in Continental Europe. Although there is no evidence that it has become established in Britain, with the advent of a warmer climate it could become a serious pest [
25]. In addition, abiotic impacts of climate change may affect biotic attacks if, for example, more dead wood is available. Milder winters may also see an increase in deer numbers placing greater pressure on the establishment phase. Depending so heavily on pure stands of Sitka spruce is increasingly seen as a risky strategy and that the use of alternative species in mixtures should be considered for at least part of the area of Sitka spruce forest to spread future risks of biotic attack.
4. Species Mixtures with Sitka Spruce
While Sitka spruce is well suited to the current climate in Scotland, greater use of alternative tree species is recommended to adapt these spruce forests to climate change [
26]. Mixed species stands are thought to be more resilient to the risks from global and regional climate change [
27] and are seen as being important in maintaining long-term forest health based on the premise that major disease or insect outbreaks do not appear to spread as easily as in pure stands [
28]. Indeed, large-scale planting of a few species may have exacerbated the spread of diseases [
29]. Mixed species stands may also be more resilient to abiotic damage than pure stands although objective evidence for this is limited. Pretzsch [
30] found that mixtures of Norway spruce (
Picea abies (L.) Karst.) and beech (
Fagus sylvatica L.) were more stable than pure spruce stands, possibly due to the ability of mixtures to recover more quickly from reductions in stand density through thinning or windthrow losses.
Potential alternatives to Sitka spruce are being actively considered in Scottish forests as a means of spreading the risk should Sitka spruce succumb to biotic attack [
18]. Much of the focus has been on identifying alternative species that would perform well in Scotland with the broad objective of reducing the dominance of Sitka spruce and to ensure a diversity of species to deal with shifting priorities in silvicultural practice and the threat of climate change [
31]. However, the forestry industry in Scotland will not give up on Sitka spruce without persuasive evidence that it is at serious risk from the effects of climate change. Part of the problem is that the silvicultural characteristics and timber properties of many of potential alternative species are poorly understood and that the necessary research to evaluate their properties may take decades to acquire. Rather than wait for more information on potential alternatives, a more pragmatic way forward would be to continue using Sitka spruce, but establish it in mixtures to ease uncertainties over future health and growth [
32]. It would allow time for foresters, timber processors and researchers to assess the silvicultural performance, wood properties and potential markets of the alternative species before committing to their use while “safeguarding” the short- and medium-term commercial value of the stands based on the tried and tested Sitka spruce. The decision on the suitability of the secondary species could be deferred until the first thinning window (typically around 20 years old for Sitka spruce) during which time growth performance, stem form and an early indication of timber quality could be evaluated. If the alternative species turns out to be unsuitable, it could be removed as thinnings leaving a pure Sitka spruce stand to go on to the full rotation. If on the other hand, if it performs well, the forester has the option of keeping it as part of a permanent mixture as a safeguard to future uncertainty over health and growth. Should the worst scenario happen and Sitka spruce succumbs to biotic attack, then at least there are alternative tree species to mitigate the economic loss of the spruce. Jacobsen and Thorsen [
32] analysed the effects of climate change on the economic value and optimal management of Norway spruce/Sitka spruce mixed stands in Denmark. Given the uncertainty of climate change and its impact on tree species, they showed that mixtures had a significant economic value advantage over pure species, and that this was the case when one of the species is less profitable. They also found that as long as uncertainty to future growth conditions remains, the longer it is worthwhile maintaining both tree species in the stand.
No other commercial tree species currently used in Scottish forestry is as adaptable as Sitka spruce to such a wide range of site conditions and therefore the “secondary” species in a mixed stand would likely become the limiting factor to the success of the mixture. The high growth rate, dense canopy and capacity of Sitka spruce to tolerate partial shade means that slower-growing, more light-demanding species will likely succumb relatively early in the rotation if left unthinned. In addition, mixtures with slower-growing species or species with less dense canopies, such as the larches and pines, may not provide sufficient competition to control the expansion of Sitka spruce canopies necessary to limit branch and knot size resulting in poor log and timber quality [
33,
34]. The object is to produce mixtures where neither species would dominate, even if managed under a no-thin regime, retaining the option to change the proportion of species within the mixture throughout the rotation. Alternate row (1:1) or 50:50 intimate mixtures would work where the growth rate of the two species is broadly similar and both are likely to remain within the canopy even where no thinning takes place. Where one species is less vigorous that the other with the risk of succumbing to competition, then a greater proportion of the secondary species, say as a 2:1 mixture, may be more suitable with the slower-growing species forming the double rows. Where the level of incompatibility between the species is higher and the stand is unlikely to be thinned, then a 3:1 mixture may be necessary to protect the weaker component. Intimate mixtures where both species are represented within rows are more complicated to establish than pure rows and less likely to be used for this reason. There are several potentially suitable species that would form compatible mixtures with Sitka spruce. Of these three of the most promising candidates form natural mixtures with Sitka spruce in its North American range.
4.1. Sitka Spruce/Western Hemlock
A review by Cameron and Mason [
35] highlighted the considerable potential of Sitka spruce/western hemlock (
Tsuga heterophylla (Raf.) Sarg.) mixtures. Western hemlock is found throughout the natural range of Sitka spruce where they can form dense mixed stands and achieve relatively high growth rates [
36]. One of the main features of western hemlock is its adaptability to a wide range of site types. It will grow on well drained brown earths through to wet gleys [
37]; soils that are relatively dry at one end of the range to the capacity to tolerate periods of waterlogging that the other [
38], and will tolerate nutrient poor soils better than many other fast-growing conifers [
39]. Western hemlock would work well in mixture with Sitka spruce over a range of sites, other than where exposure is severe. It may have a particular advantage where there is uncertainty over future rainfall patterns, such as in eastern districts of Scotland, where the use of a more drought tolerant species such as hemlock would provide a safe alternative should climatic conditions deteriorate to the detriment of Sitka spruce. Western hemlock is very shade tolerant and forms the sub-dominant species in mixture with Sitka spruce in natural forests [
40] negating the often held view that it will dominate spruce stands. In fact, studies have shown that Sitka spruce persists in natural mixed stands with western hemlock even with limited interventions [
41]. Stem and timber properties of both species seem to improve from being in a mixture [
35]. The strength properties of western hemlock timber have been described as somewhere between those of Sitka spruce and Douglas fir [
42] and the wood is suitable for a wide range of uses, such as construction and interior joinery [
43].
4.2. Sitka Spruce/Douglas Fir
Douglas fir (
Pseudotsuga menziesii (Mirb.) Franco) is a species that much is known about its silvicultural characteristics and timber properties under Scottish conditions. Douglas fir is associated with Sitka spruce in the southern part of the natural range of spruce in the north-west coast of North America [
36]. It is a fast-growing, semi-shade tolerant species that is capable of maintaining a position in the canopy with Sitka spruce. The timber is generally of high quality and out-performs Sitka spruce in stiffness and strength. However, Douglas fir is limited by its requirement for better quality, moderately fertile, and moist but well-drained mineral soils—not very common in sites typically associated with forestry in Scotland—and is not tolerant to high exposure [
44]. Nevertheless, where suitable conditions do exist, it should work well as a mixture with Sitka spruce.
4.3. Sitka Spruce/Grand Fir
Grand fir (
Abies grandis (Lamb.) Lindl.) is a fast-growing, shade tolerant species and is reasonably adaptable to a variety of sites although it does best when conditions are moist similar to those required by Sitka spruce. It is vulnerable to drought crack and is unsuitable in drier regions [
44]. There is limited information on its performance in mixture with Sitka spruce, but observations of mixed stands by the author shows that both species do well and neither dominates the other. However, initial growth of grand fir is slow and initially may not keep pace with the faster-establishing Sitka spruce. It also lacks tolerance to exposure, which restricts where it can be grown [
44]. Stem form of both species in mixture is good. The timber of grand fir is similar to Norway spruce, although with slightly lower stiffness, and can be used for a wide range of purposes.
4.4. Other Potential Alternatives
There are other species that have similar growth rates to Sitka spruce and may form reasonably stable mixtures. These include noble fir (
Abies procera Rehder), western red cedar (
Thuja plicata Don ex D. Don), and possibly coast redwood (
Sequoia sempervirens (D.Don) Endl.), but there are significant limitations with site adaptability and/or timber properties [
44] that may restrict their use. Slower-growing Norway spruce may work in mixture with Sitka spruce if regularly thinned; otherwise, Sitka spruce will likely dominate the stand. In addition, there is always the danger that a biotic attack may affect both spruces. Other possibilities have been suggested including
Abies amabilis (Doug. ex Forbes),
Cryptomeria japonica ((L.f.) Don) and
Chamaecyparis nootkatensis ((D. Don) Spach) [
31], but little is known about their performance in mixtures and timber properties. Significant use of any of the alternatives species suggested above would depend on forest nurseries sourcing seed of appropriate provenances and developing production methods to ensure sufficient planting stock was available, and this could take time to achieve. In addition, practically all of the species that could potentially form stable mixtures with Sitka spruce are more vulnerable to browsing damage by herbivores [
44], and as a consequence protection costs will almost certainly be higher than that for pure stands of spruce.
5. Improving Stand Stability
Climate change predictions of more frequent storm events [
2,
9] will have serious consequences for forestry in Scotland since endemic wind damage to forest stands is already a significant economic problem. Spruces tend to be shallow rooting and therefore developing a radially symmetrical structural root system is critical for stability. For this reason, cultivation methods in Scotland have shifted away from spaced-furrow ploughing, which tended to limit lateral root spread [
11,
45], to scarifying and mounding that encourage the radial spread of roots and improved tree stability [
46]. The risk of windthrow on exposed sites with shallow soils has led to the adoption of no-thinning regimes in many areas [
47] in an attempt to postpone the onset of damage. The prediction of more precipitation over the winter months in west and north of Scotland—likely to remain the focus of Sitka spruce forestry in the future—may increase the incidence of localised flooding and more site drainage may be necessary to prevent waterlogging and anaerobic conditions developing that would impede root growth.
Even where site conditions favour stable root systems, the choice of stand management can influence the risk of wind and snow damage. Natural pruning in Sitka spruce is quite slow and so a relatively close planting spacing is required (≤2 m between plants) to encourage early canopy closure and suppression of branches on the lower stem to limit the size of knots in the timber [
48]. However, maintaining closed canopy conditions restricts individual tree sway, necessary to stimulate structural root development and maintain an acceptable degree of stem taper, resulting in stands of trees with slender stems and restricted structural roots that are at risk of wind damage [
49]. Generally, the aim is to maintain a slenderness ratio (height/diameter) of less than 80 to maintain stability, but not less than 60 when the stem becomes too tapered for sawlogs.
Since the risk of storm damage is at its highest in newly thinned stands, selective thinning must start at an early age to encourage stem movement and the gradual development of stability features to improve resistance to wind damage. Foresters are often reluctant to thin early due to the small average tree size that may incur a financial loss [
48]. Delaying the first thinning to improve average tree size and volume output greatly increases instability when usually a heavier thinning takes place [
48]. The alternative is to adopt a no-thinning regime, relying on the mutual support of the trees within the stand to sustain continued height growth in the expectation that the stand will survive to some acceptable economic rotation age. However, trees become very slender in unthinned stands with shorter crowns that are highly susceptible to wind and snow damage, which when it does occur can be catastrophic with the loss of the entire stand [
48]. In a future of potentially more frequent winter storms, it is quite possible that these stands will blow down well before an acceptable threshold of timber production has been reached. Dense stands of suppressed trees are also more vulnerable to damage caused by drought [
50].
It will be increasingly necessary to intervene early by cutting racks for machine access and thinning to allow the trees to sway and develop stability features before they become too tall. To maintain sufficient mutual support, light low thinnings are recommended with a maximum removal of 30 percent of the trees at one time [
51] with first thinning and cutting of extraction racks taking place around 10 years after full canopy closure to allow sufficient suppression of the lower branches without the risk of the stems becoming too unstable [
48]. This may have to come at a cost given that early thinning interventions may not yield an income that will cover the cost of harvesting, but this must be seen as an investment in the future stand.
6. Developing Irregular Stand Structures
There has been considerable international interest in recent years in transforming predominately even-aged stands into irregular structures characterised by the presence of a wide range of tree ages and sizes growing within a relatively small space and where a certain level of canopy cover is maintained. Irregular forestry is seen as a useful silvicultural strategy to improve the resilience of forests to climate change [
52]. In fact, it was climate related damage to stands managed by uniform silvicultural systems that advanced the use of irregular forestry in the 19
th century as a result of the failure of the uniform system subject to storms [
53]. Irregular stand structures appear to be more resilient to abiotic damage than even-aged stands and lower damage levels have been observed in irregular stands following storms in comparison with regular even-aged stands [
54]. Dominant trees in irregular stands emerge earlier and develop deeper canopies than dominants in regular stands and, as a consequence, are exposed to bigger wind loads due to the greater crown size and over a longer period of time resulting in the development of stabilising features such as greater stem taper and stronger structural roots [
55]. Kenk and Guehne [
56] found that the bigger trees or dominants in irregular stands have a reasonable but not excessive stem taper (height/diameter ratio < 80) that imparts good stability. By contrast, trees grown in an even-aged stands experience periods of closed canopy between thinnings that restrict the development of these stabilising features leaving them more vulnerable to strong winds [
48]. Mason [
55] concluded that fully developed irregular stands in a ‘balanced’ condition would appear to be more resilient to wind damage than regular stands where exposure in moderate, but the difference is likely to be minimal where exposure is severe. Even where the balanced irregular condition exists, harvesting interventions have been found to cause a destabilising effect for a number of years [
54]. However, where damage does occur in heterogeneous stands, much of the advanced regeneration will remain limiting the loss of forest cover.
While resilience to abiotic damage may be an important feature in established irregular stands, the transformation process from an even-aged structure to the balanced irregular condition is fraught with difficulties. An intermediate shade tolerant species like Sitka spruce requires regular and substantial openings of the canopy to maintain growth of the seedlings and saplings [
41,
57,
58] causing periodic destabilisation of the canopy and increased risk of wind damage. Mason and Kerr [
59] highlighted the risks of attempting a transformation in high windthrow risk stands recommending more sheltered locations with soils that encourage good root development. The authors also pointed out the difficulties in achieving successful natural regeneration on more fertile soils due to potential vegetation competition. Together, these factors will undoubtedly limit the areas where transformations of Sitka spruce into irregular structures may be feasible. Where transformations of even-aged stands of Sitka spruce are possible, introducing alternative shade tolerant species into stands is thought to improve the resilience of the irregular structure to damage [
60].
7. Limiting the Size of the Growing Stock
The area of older forests in Europe has increased in recent decades while the proportion of younger forests has declined [
61]. This has coincided with an increased frequency of very damaging storm events in Europe that has resulted in high losses of timber volume that is in part attributed to the retention of very high standing volumes [
62] likely to comprise of older stands of taller trees that are more susceptible to damage [
61,
63]. There is now a growing body of opinion that limiting the size of the growing stock in both regular and irregular forests would restrict the scale of damage caused by storm events [
61,
64].
An effective way of managing the proportion of older stands of taller trees in regular forests is by “normalising” the age-class structure to create an approximately balanced distribution of stands from newly established to those with mature trees. Since storm events have the greatest impact on stands with the oldest and most likely tallest trees [
63], younger stands of smaller trees may avoid significant damaged and so at least part of the financial investment in the forest would be safeguarded. By spreading the stands of different ages throughout the forest area, the damage would also be distributed limiting the visual and environmental impact. Limiting the maximum tree size through shortening rotation ages would also reduce the potential impact of abiotic damage although this will result in a proportionally greater amount of juvenile wood in logs [
65]. In Scotland, substantial areas of Sitka spruce were planted over a relatively short time period, particularly in the 1960s and 70s, resulting in the creation of large even-aged blocks of forest. While there has been a programme of creating a better age-class distribution in these stands, the focus has been primarily on improving the visual appearance of plantation forests and not in terms of protecting the growing stock. Creating more diverse age-class range of stands in established forests is often hampered by wind damaging adjacent compartments to recently felling coupes resulting in relatively large areas being cleared and restocked at the one time. Hence, the importance of developing stability in spruce stands from an early age.
Limiting the size of the growing stock of irregular stands has been discussed as a means of limiting the economic loss to forests if damaging events occur [
64]. This could be achieved by limiting the proportion of larger trees—those at greatest risk of damage—and increasing the cycle of harvesting interventions to maintain a modest standing volume or “capital at risk” [
66]. It has been argued that past management of irregular montane forests has been too cautious leading to a build-up of the growing stock [
67] leading to increased vulnerability to damage [
64]. Maintaining a relatively open canopy, through lowering the harvesting diameter and shorter cutting cycles, could suit the semi-shade tolerant Sitka spruce within irregular stand structures to promote the establishment and growth of young seedlings and saplings. While the diameter distribution of irregular structures will be altered after a damaging event, the smaller diameter groups within the stand (advanced regeneration, saplings and possibly young pole stage trees) would quickly respond to the additional light limiting the overall impact on the forest.
8. Conclusions
The lack of certainty of the potential impacts of climate change on the commercially important Sitka spruce forests in Scotland suggests a pragmatic approach to future management that avoids over-reaction while developing sufficient flexibility to allow change in direction at any point in the future.
Table 1 summarises the key adaptive strategies recommended to protect the economic viability of Sitka spruce forests in Scotland in response to climate change and sets out developments in policy required to advance these strategies. Where Sitka spruce is showing clear signs of environmental stress or damage, such as evidence of increasing drought stress in some parts of eastern Scotland [
14], then it may have to be replaced by more drought tolerant species. Otherwise, Sitka spruce should continue to be planted where it currently grows well, but preferably in mixed species stands where this is silviculturally feasible to increase resilience to particularly biotic damage. The key is to choose the correct species to mix with Sitka spruce to ensure good growth and development of both species even if the stand remains unthinned. Western hemlock, Douglas fir and grand fir all form mixtures with Sitka spruce in their natural range in North America, have good growth rates, are capable of maintaining their position in the canopy with Sitka spruce on appropriate sites, and all produce timber of acceptable performance. While there are other potential alternative species that could be used in mixtures, the lack of information on their silvicultural characteristics and timber performance in mixed stands limits their use until more research findings are available. Several potential disadvantages of mixed species stands highlighted in
Table 1 will need to be addressed and an ongoing research programme will be vital to support the changes proposed. More immediate issues include the current lack of availability of planting stock of alternative species in forest nurseries and problems of the greater vulnerability of these species to browsing damage leading to greater establishment costs.
Longer term issues such as developing markets for a greater range of timber species will need to be addressed through research and greater engagement with timber processors.
The prediction of stronger wind events in the future will require earlier and more frequent silvicultural interventions to improve stand stability. Early cutting of extraction racks and first thinning should take place to allow stability features to develop—such as stronger roots and better stem taper—while the trees are relatively young and not at risk of damage through opening the canopy. While the current market for small roundwood in Scotland is good, particularly driven by the growing demand for biomass for energy, forest owners may have to accept early first thinnings at a financial loss as a necessary investment to develop future stand stability. Financial incentives through, for example, the provision of early thinning grants may be necessary to encourage foresters to carry out these vital early interventions. Subsequent thinning should aim to maintain around half tree height as living crown and never allow the canopy depth to fall below one third tree height in older stands [
48]. This is based on the view that the wind force on a larger crown area will place greater stresses on the stem and root system over a long period of time resulting in greater root strength and less slender stems. While irregular forest stands are widely believed to be more resilient to a changing climate, it is unlikely that significant areas of even-aged Sitka spruce stands in Scotland can be transformed into irregular structures due to high exposure or inappropriate site types [
59].
Table 1.
Potential advantages and disadvantages of adaptive strategies to improve the resilience of Sitka spruce in response to climate change, and suggested policy developments to enable their introduction.
Table 1.
Potential advantages and disadvantages of adaptive strategies to improve the resilience of Sitka spruce in response to climate change, and suggested policy developments to enable their introduction.
| Adaptive Strategy | Potential Advantages | Potential Disadvantages | Suggested Policy Developments |
|---|
| Species mixtures with Sitka spruce | Spreads risk of potential attack by pests and diseases. | Reluctance by industry to commit to untested species with timber of currently limited market value. | Establish programme of research into the silvicultural requirements and wood properties of alternative species. |
| Improves stability of mixed stands in comparison with pure stands. | Limited number of species that are silviculturally compatible with Sitka spruce. | Require greater engagement between researchers and foresters to convince them of the need to use mixtures. |
| Lack of availability of tree seedlings of the secondary species from forest nurseries. |
| Maintains Sitka spruce as the main commercial species. | Thinning stands to maintain a mixed species composition may not be feasible in very exposed areas. | Require greater engagement between the forestry industry and government to consider funding streams to support the planting of mixtures and their subsequent protection. |
| Improves biodiversity of forest stands in comparison with pure stands. | Alternative species likely to suffer greater browsing damage by herbivores than Sitka spruce leading to greater establishment and protection costs. | Require greater engagement between the growing sector, researchers and processing industries to consider development of alternative markets. |
| Greater diversity of products at harvesting will require more sorting and increase harvesting costs. |
| Management to improve stand stability | Improves of long-term stand stability through early cutting of extraction racks and thinning. | Early harvesting interventions may incur a financial loss and deter foresters from undertaking these operations. | Engagement between industry and government to support the establishment of grants to cover early loss-making thinning operations. |
| Developing irregular structures | Creates greater resilience to abiotic damage in comparison with even-aged stands. | Time constraints and costs in achieving irregular structures through lost production during the transformation period. | Need for more research and setting up of demonstration stands to show foresters where and how transformations are carried out. |
| Improves biodiversity through the creation of a vertically diverse habitat. | Lack of knowledge of the process of transformation Sitka spruce dominated stands. |
| Limited range of sites where transformations may be possible. |
| Limiting the size of the growing stock | Reduces the area of older stands of taller trees that are more susceptible to damage. | Creating a more diverse range of stand age-classes in established uniform forests is complicated by wind damage to stands adjacent to recently felled coupes. | (As above for management to improve stand stability). |
| Reduces the potential impact of abiotic damage by limiting average tree size through shortening rotation age. | Greater proportion of juvenile wood within logs possible by limiting tree size through shortening rotations. | Need for more research into possible alternative sawing patterns to separate the juvenile and mature wood. |
Forest structures comprising a limited age-class distribution—typical of many Sitka spruce forests in Scotland—should be avoided since they are generally believed to be more vulnerable to storm damage [
62]. Instead, foresters should aim to achieve a ‘mosaic’ of stands of different age classes broadly following the traditional concept of the normal forest. Storm damage in these forests would most likely be limited to the stands with the taller and older trees whereas the younger stands, particularly if they have been thinned correctly, should survive largely intact lessening the economic loss to the forest owner. The effect on the forest environment will be less since a substantial part of the forest area will remain. It is also suggested that the size of the growing stock within a forest should also be limited to minimise the economic loss in the event on a damaging event by avoiding the retention of large areas of older stands.