Influence of Hemlock Woolly Adelgid Infestation Levels on Water Stress in Eastern Hemlocks within the Great Smoky Mountains National Park, U.S.A.
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and Experimental Design
2.2. Sampling and Measurements
2.3. Data Analysis
3. Results and Discussion
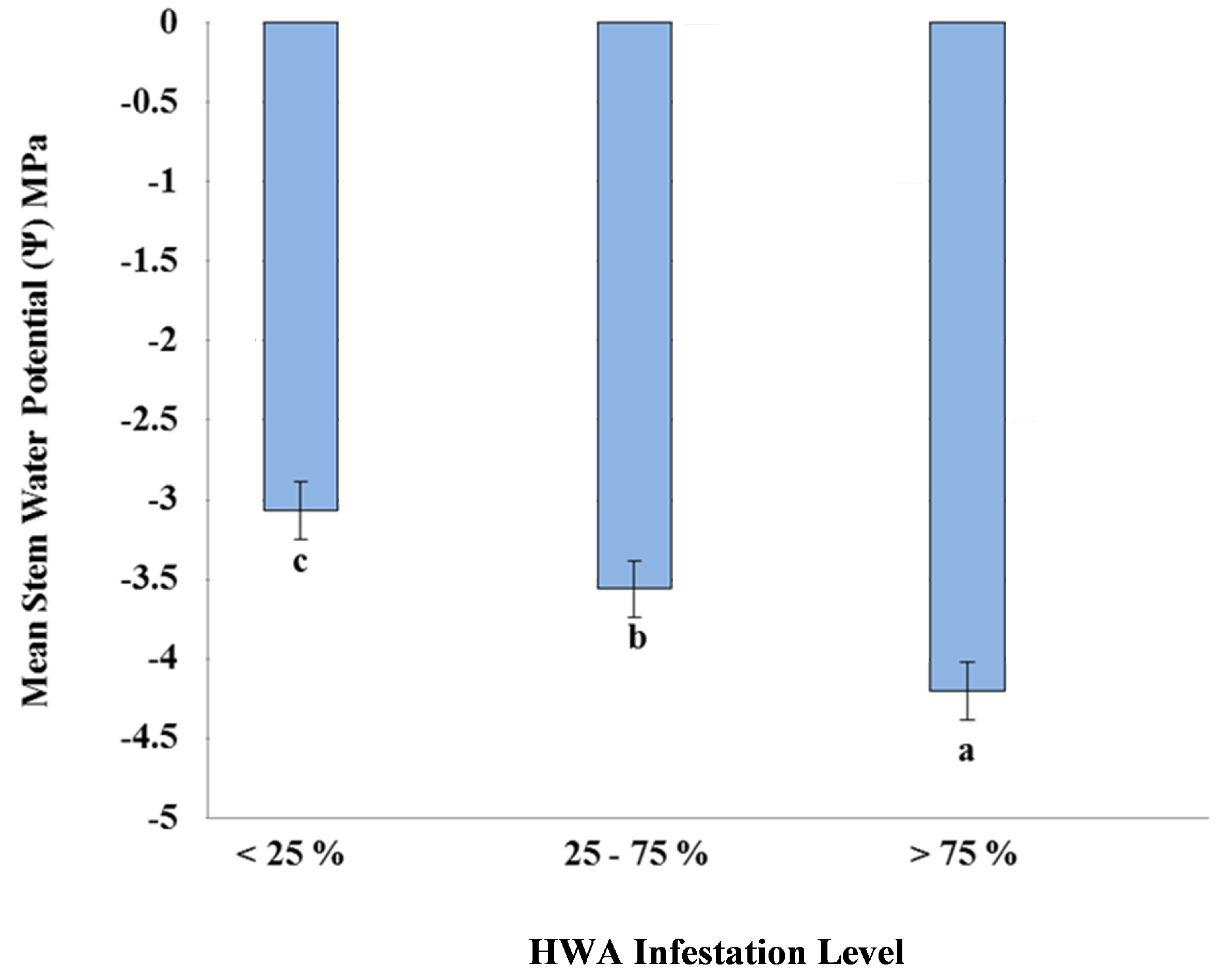
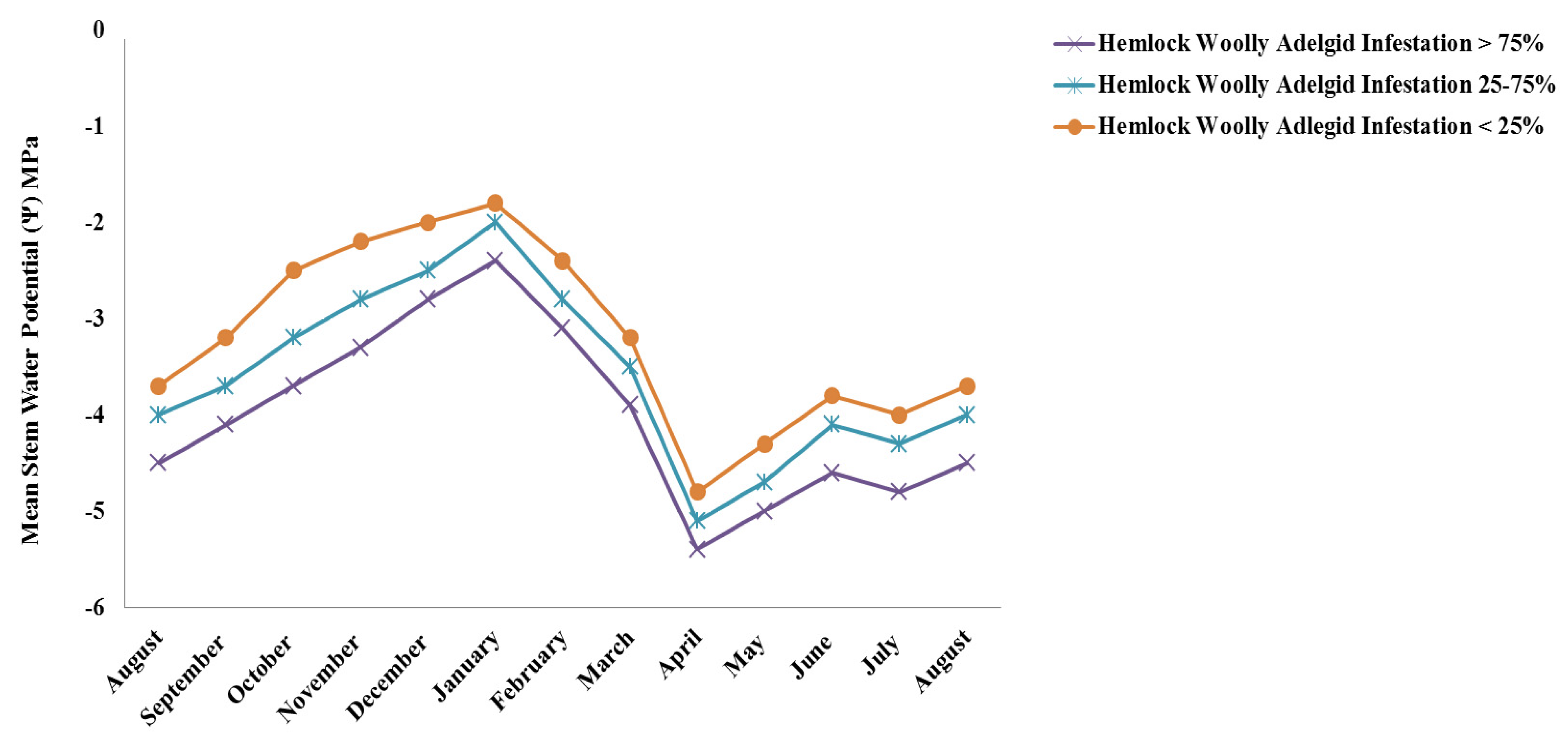
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cowles, R. Optimizing dosage and preventing leaching of imidaclorpid for management of Hemlock Woolly Adelgid in forests. For. Ecol. Manag. 2009, 257, 1026–1033. [Google Scholar] [CrossRef]
- Ford, C.R.; Vose, J.M. Tsuga canadensis (L.) Carr. mortality will impact hydrological processes in the southern Appalachian forest ecosystems. Ecol. Appl. 2007, 17, 1156–1167. [Google Scholar]
- Gonda-King, L.; Gómez, S.; Martin, J.L.; Orians, C.M.; Preisser, E. Tree responses to an invasive sap-feeding insect. Plant Ecol. 2014, 215, 297–304. [Google Scholar] [CrossRef]
- Ward, J.S. Restoration of damaged stands: Dealing with the after effects of hemlock woolly adelgid. In Proceedings of the Hemlock Woolly Adelgid in the Eastern United States Symposium, East Brunswick, NJ, USA, 5–7 February 2002; Onken, B., Reardon, R., Lashomb, J., Eds.; Rutgers University: New Brunswick, NJ, USA, 2002; pp. 118–126. [Google Scholar]
- Schmidt, T.L.; McWilliams, W.H. Status of eastern hemlock in the northern U.S. In Proceedings of the Regional Conference on Ecology and Management of Eastern Hemlock, Iron Mountain, MI, USA, 27–28 September 1995; Mroz, G., Martin, J., Eds.; University of Wisconsin: Madison, WI, USA, 1996; pp. 61–72. [Google Scholar]
- Buck, S.; Lambdin, P.; Paulsen, D.; Grant, J.; Saxton, A. Checklist of insect species associated with eastern hemlock in the Great Smoky Mountains National Park and environs. Tenn. Acad. Sci. 2005, 80, 1–10. [Google Scholar]
- Kelty, M.J. Productivity of New England hemlock/hardwood stands as affected by species composition and canopy structure. For. Ecol. Manag. 1989, 28, 237–257. [Google Scholar] [CrossRef]
- DeGraaf, R.M.; Yamasaki, M.; Leak, W.B.; Lanier, J.W. New England Wildlife: Management of Forested Habitats; Gen. Tech. Rep. NE-144; USA Department of Agriculture, Forest Service, Northeastern Forest Experiment Station: Radnor, PA, USA, 1992; p. 15. [Google Scholar]
- Snyder, C.; Young, J.; Smith, D.; Lemarie, D.; Ross, R.; Bennett, R. Stream Ecology Linked to Eastern Hemlock Decline in Delaware Water Gap National Recreation Area; USA Geological Survey: Kearneysville, WV, USA, 2004. Available online: http://www.lsc.usgs.gov/aeb/2048–03/dewa.asp (accessed on 20 April 2014).
- Lapin, B. The Impact of Hemlock Woolly Adelgid on Resources in the Lower Connecticut River Valley; Dept. of Agriculture, Forest Service, Northeastern Center for Forest Health Research: Hamden, CT, USA, 1994; p. 10. [Google Scholar]
- Tingley, M.W.; Orwig, D.; Field, R.; Motzkin, G. Avian response to removal of a forest dominant: consequences of hemlock woolly adelgid infestations. J. Biogeogr. 2002, 29, 1505–1516. [Google Scholar] [CrossRef]
- Dilling, C.I.; Lambdin, P.L.; Grant, J.F.; Buck, S.L. Insect guild structure associated with eastern hemlock in the southern Appalachians. Environ. Entomol. 2007, 36, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.A. An ecosystem unraveling? In Proceedings of the Hemlock Woolly Adelgid in the Eastern United States Symposium, East Brunswick, NJ, USA, 5–7 February 2002; Onken, B., Reardon, R., Lashomb, J., Eds.; Rutgers University: New Brunswick, NJ, USA, 2002; pp. 254–268. [Google Scholar]
- Young, R.F.; Shields, K.S.; Berlyn, G.P. Hemlock woolly adelgid (Homoptera: Adelgidae): Stylet bundle insertion and feeding sites. Ann. Entomol. Soc. Am. 1995, 88, 827–835. [Google Scholar] [CrossRef]
- Domec, J.-C.; Rivera, L.N.; King, J.S.; Peszlen, I.; Hain, F.; Smith, B.; Frampton, J. Hemlock woolly adelgid (Adelges tsugae) infestation affects water and carbon relations of eastern hemlock (Tsuga canadensis) and Carolina hemlock (Tsuga caroliniana). New Phytol. 2013, 199, 452–463. [Google Scholar] [CrossRef]
- Jenkins, J.; Aber, J.D.; Canham, C.D. Hemlock woolly adelgid impacts on community structure and N cycling rates in eastern hemlock forests. Can. J. For. Res. 1999, 29, 630–645. [Google Scholar] [CrossRef]
- Nelson, L.A.; Dillaway, D.N.; Rieske, L.K. Effect of an exotic herbivore, Adelges tsugae, on photosynthesis of a highly susceptible Tsuga host, with notes on conspecifics. Arthropod Plant Interact. 2014, 8, 9–15. [Google Scholar] [CrossRef]
- McClure, M.S. Biological Control of Hemlock Woolly Adelgid in the Eastern United States; FHTET 2000-08; The USDA Forest Service, Forest Health Technology Enterprise Team: Morgantown, WV, USA, 2001; p. 10. [Google Scholar]
- Arthur, F.H.; Hain, F.P. Seasonal history of the balsam woolly adelgid (Homoptera: Adelgidae) in natural stands and plantations of Frasier fir. Econ. Entomol. 1985, 77, 1154–1158. [Google Scholar] [CrossRef]
- Dilling, C.I.; Lambdin, P.L.; Grant, J.F.; Rhea, J.R. Spatial and temporal distribution of imidacloprid in eastern hemlock in the southern Appalachians. Econ. Entomol. 2010, 103, 368–373. [Google Scholar] [CrossRef]
- Cowles, R.S.; Montgomery, M.E.; Cheah, C.A.S.-J. Activity and residues of imidacloprid applied to soil and tree trunks to control hemlock woolly adelgid (Hemiptera: Adelgidae) in forests. Econ. Entomol. 2006, 99, 1258–1267. [Google Scholar] [CrossRef]
- SAS Institute Inc. SAS User’s Guide, Statistics; SAS Institute Inc.: Cary, NC, USA, 2006. [Google Scholar]
- Tyree, M.T.; Sperry, J.S. Vulnerability of xylem to cavitation and embolism. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 19–36. [Google Scholar] [CrossRef]
- Tyree, M.T.; Sperry, J.S. Do woody plants operate near the point of catastrophic xylem dysfunction caused by dynamic water stress? Answers from a model. Plant Phys. 1988, 88, 574–580. [Google Scholar] [CrossRef]
- Gómez, S.; Orians, C.; Preisser, E. Exotic herbivores on a shared native host: Tissue quality after individual, simultaneous, and sequential attack. Oecologia 2012, 169, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Nuckolls, A.E.; Wurzburger, N.; Ford, C.R.; Hendrick, R.L.; Vose, J.M.; Kloeppel, B.D. Hemlock declines rapidly with hemlock woolly adelgid infestation: Impacts on the carbon cycle of southern Appalachian forests. Ecosystems 2009, 12, 179–190. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coots, C.; Lambdin, P.; Franklin, J.; Grant, J.; Rhea, R. Influence of Hemlock Woolly Adelgid Infestation Levels on Water Stress in Eastern Hemlocks within the Great Smoky Mountains National Park, U.S.A. Forests 2015, 6, 271-279. https://doi.org/10.3390/f6010271
Coots C, Lambdin P, Franklin J, Grant J, Rhea R. Influence of Hemlock Woolly Adelgid Infestation Levels on Water Stress in Eastern Hemlocks within the Great Smoky Mountains National Park, U.S.A. Forests. 2015; 6(1):271-279. https://doi.org/10.3390/f6010271
Chicago/Turabian StyleCoots, Carla, Paris Lambdin, Jennifer Franklin, Jerome Grant, and Rusty Rhea. 2015. "Influence of Hemlock Woolly Adelgid Infestation Levels on Water Stress in Eastern Hemlocks within the Great Smoky Mountains National Park, U.S.A." Forests 6, no. 1: 271-279. https://doi.org/10.3390/f6010271
APA StyleCoots, C., Lambdin, P., Franklin, J., Grant, J., & Rhea, R. (2015). Influence of Hemlock Woolly Adelgid Infestation Levels on Water Stress in Eastern Hemlocks within the Great Smoky Mountains National Park, U.S.A. Forests, 6(1), 271-279. https://doi.org/10.3390/f6010271




