Effect of Tree Size Heterogeneity on the Overall Growth Trend of Trees in Coniferous Forests of the Tibetan Plateau
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Data Collection
2.2. Quantification of Tree Radial Growth Pattern and Its Overall Variability
2.3. Data Processing and Analysis
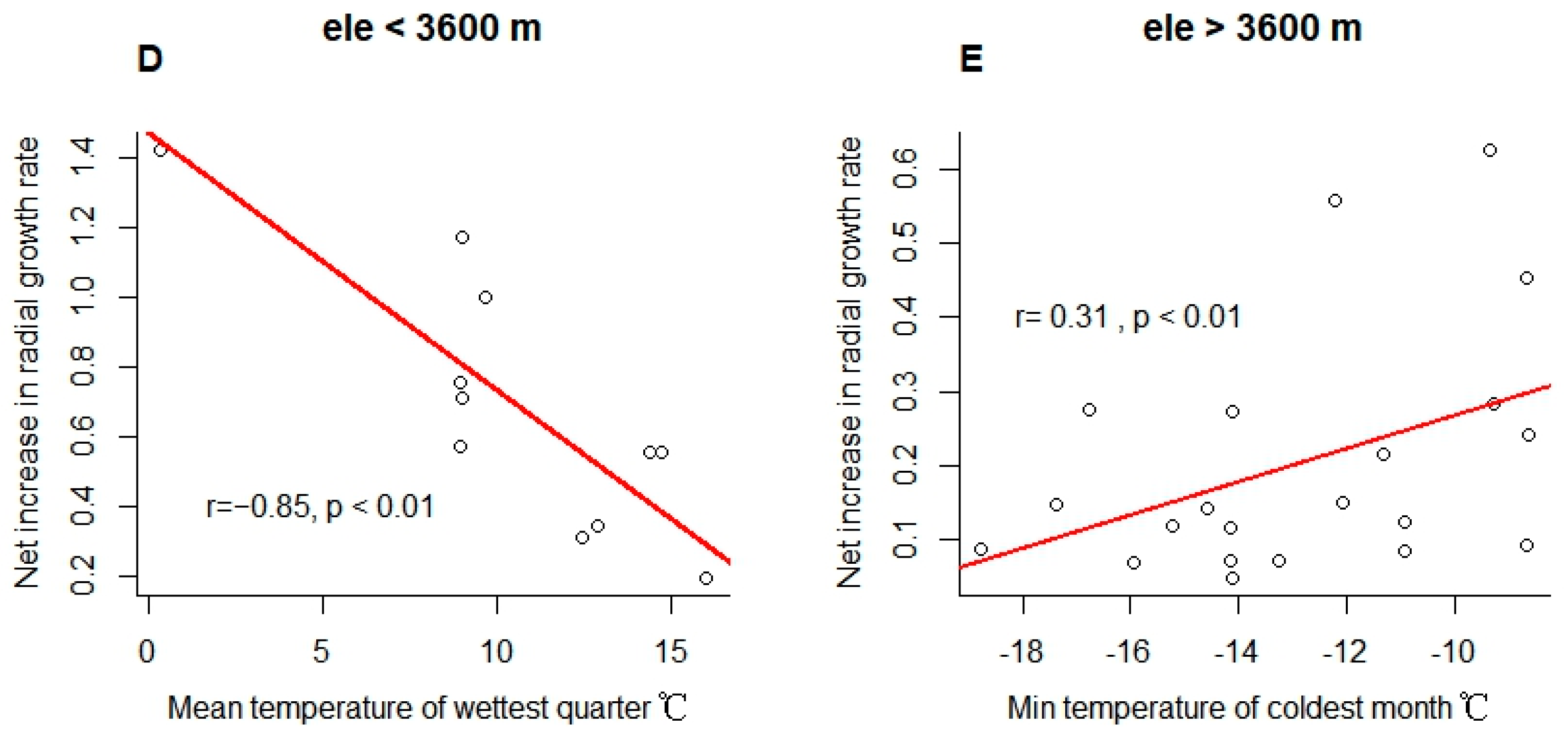
3. Results
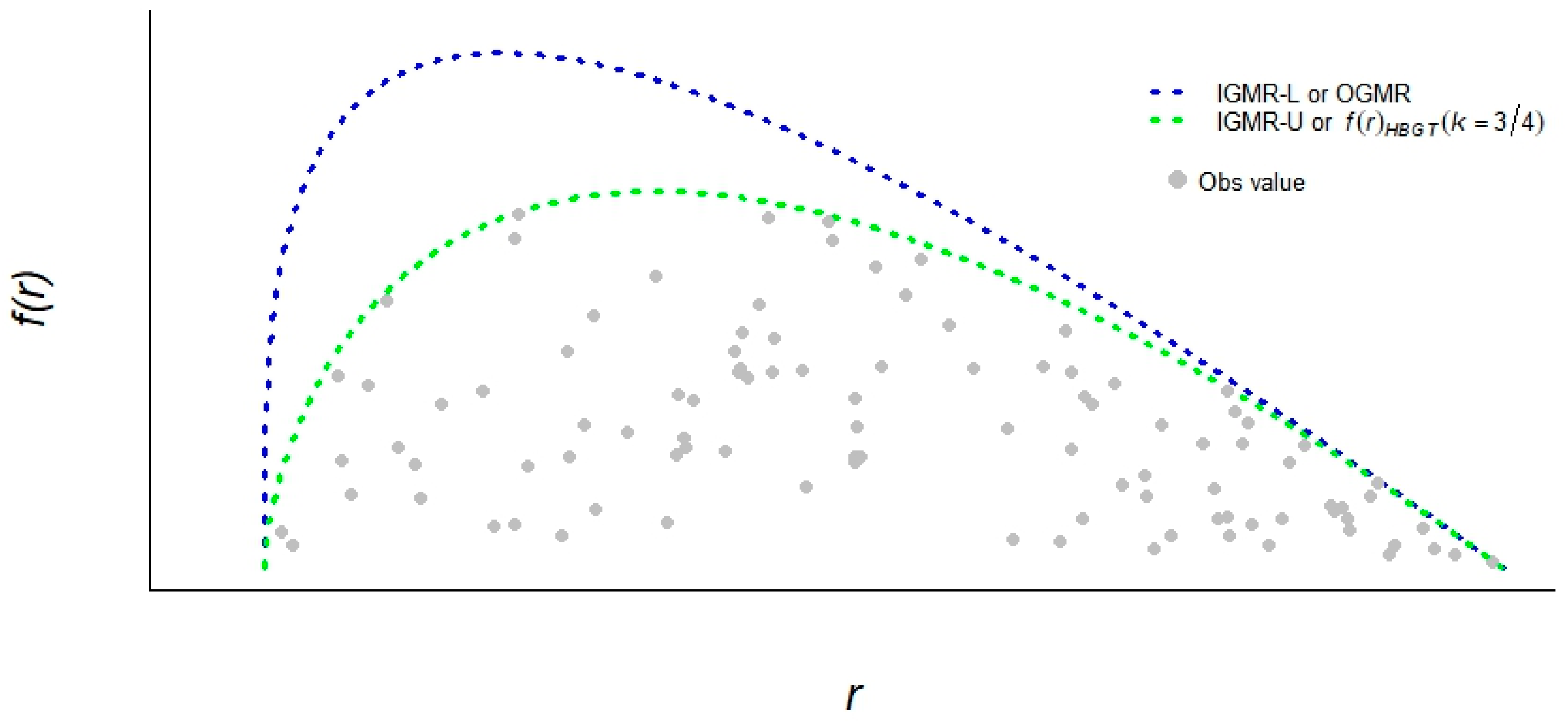
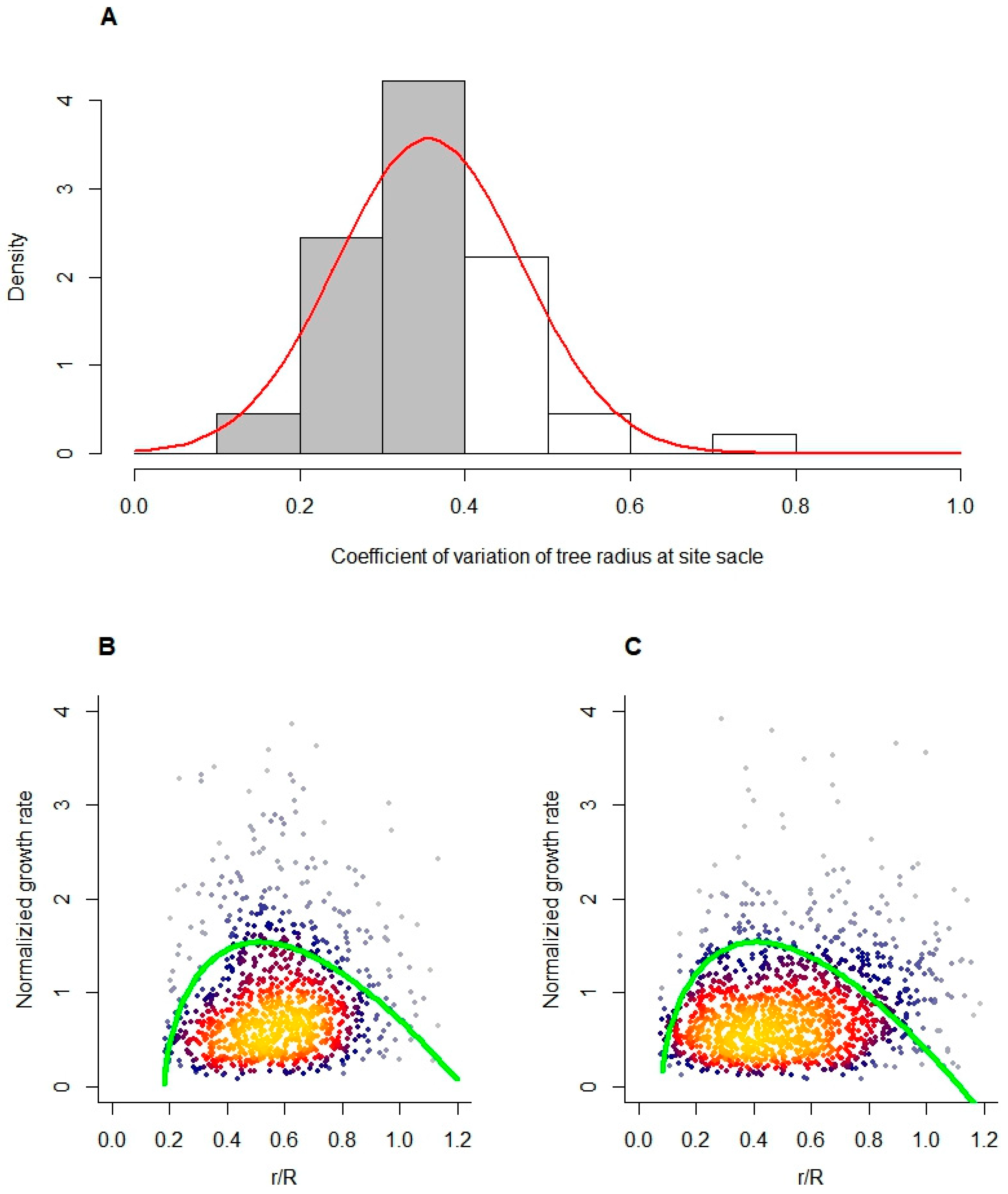
3.1. Tree Radial Growth Follows the IGMR-U
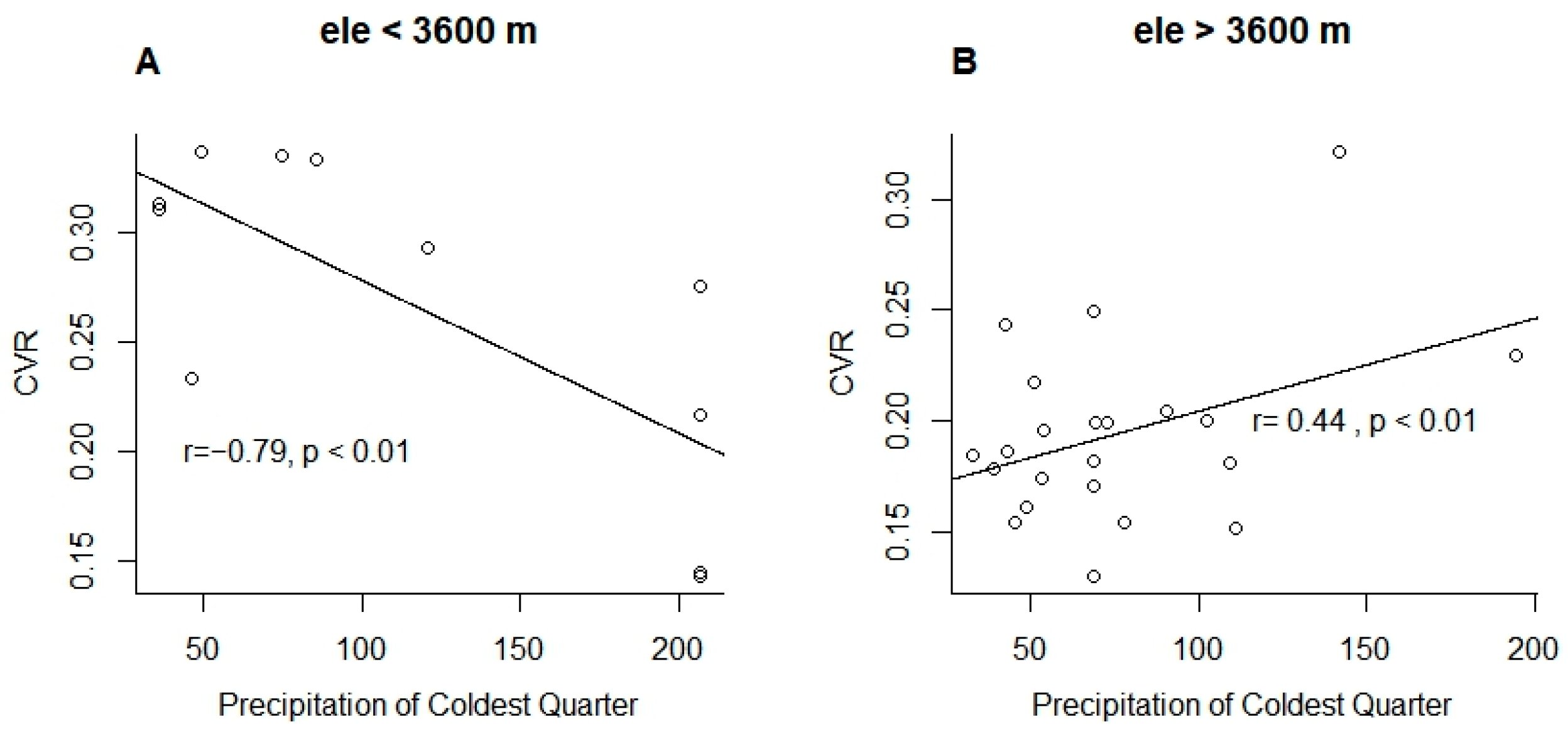
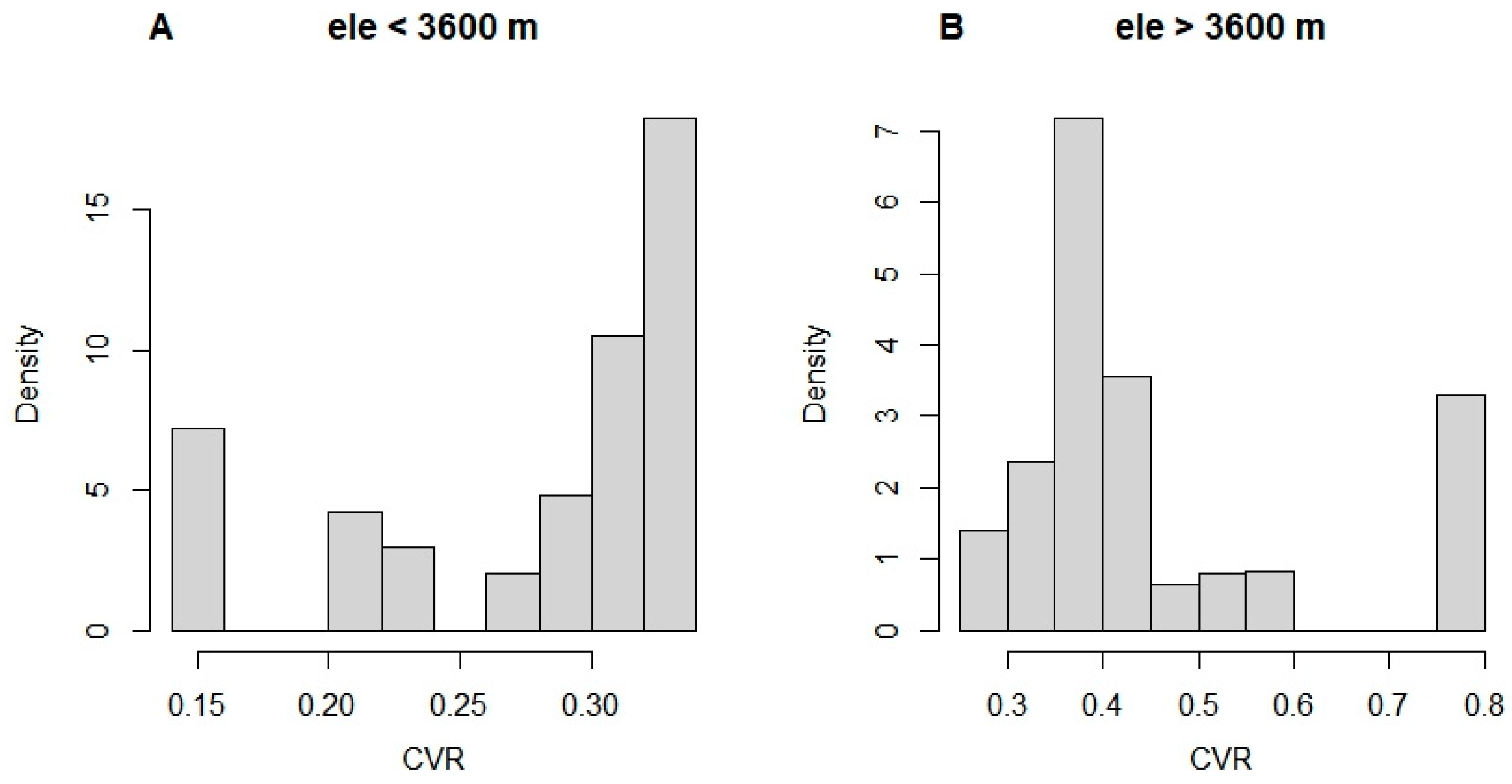
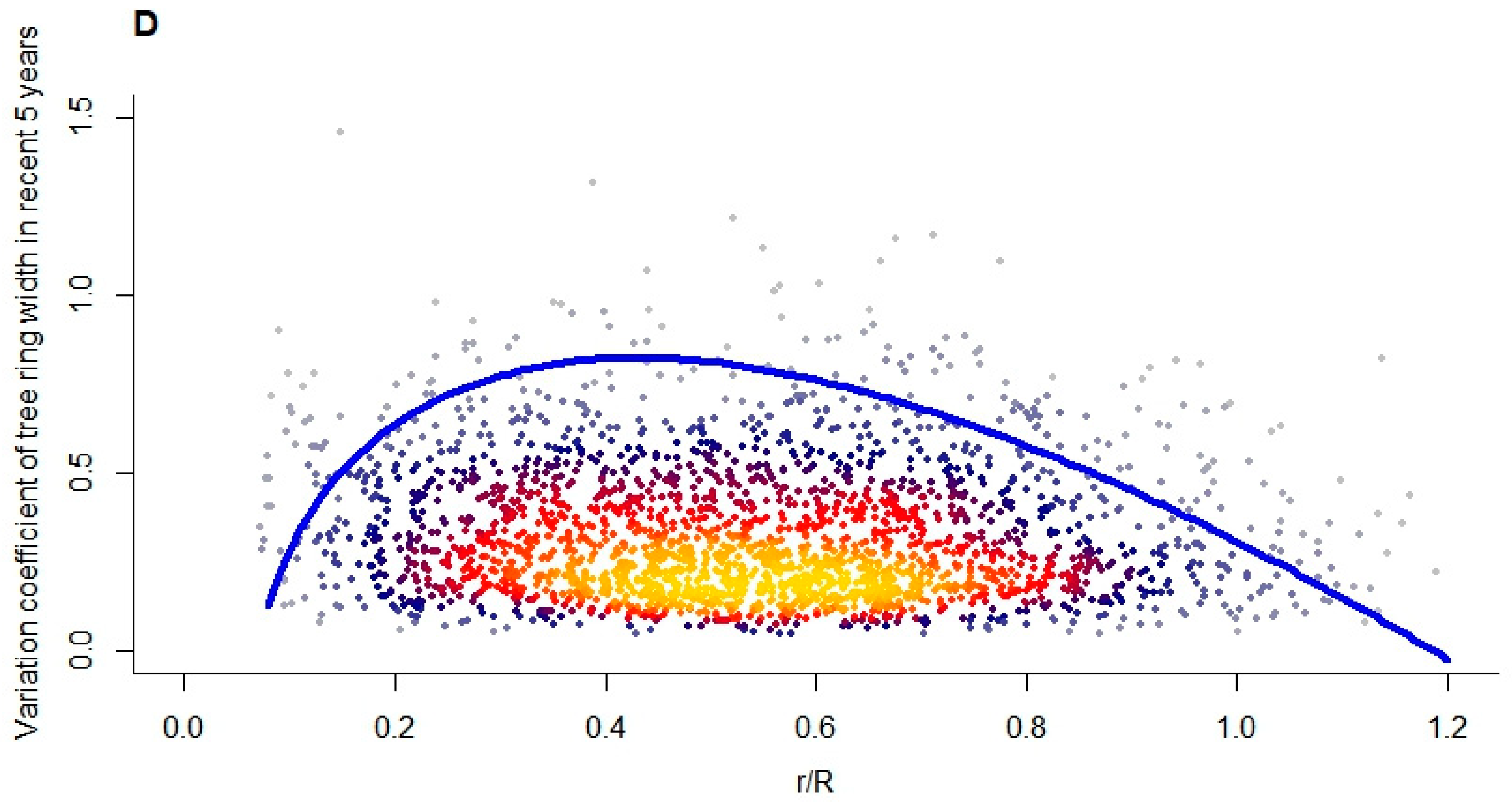
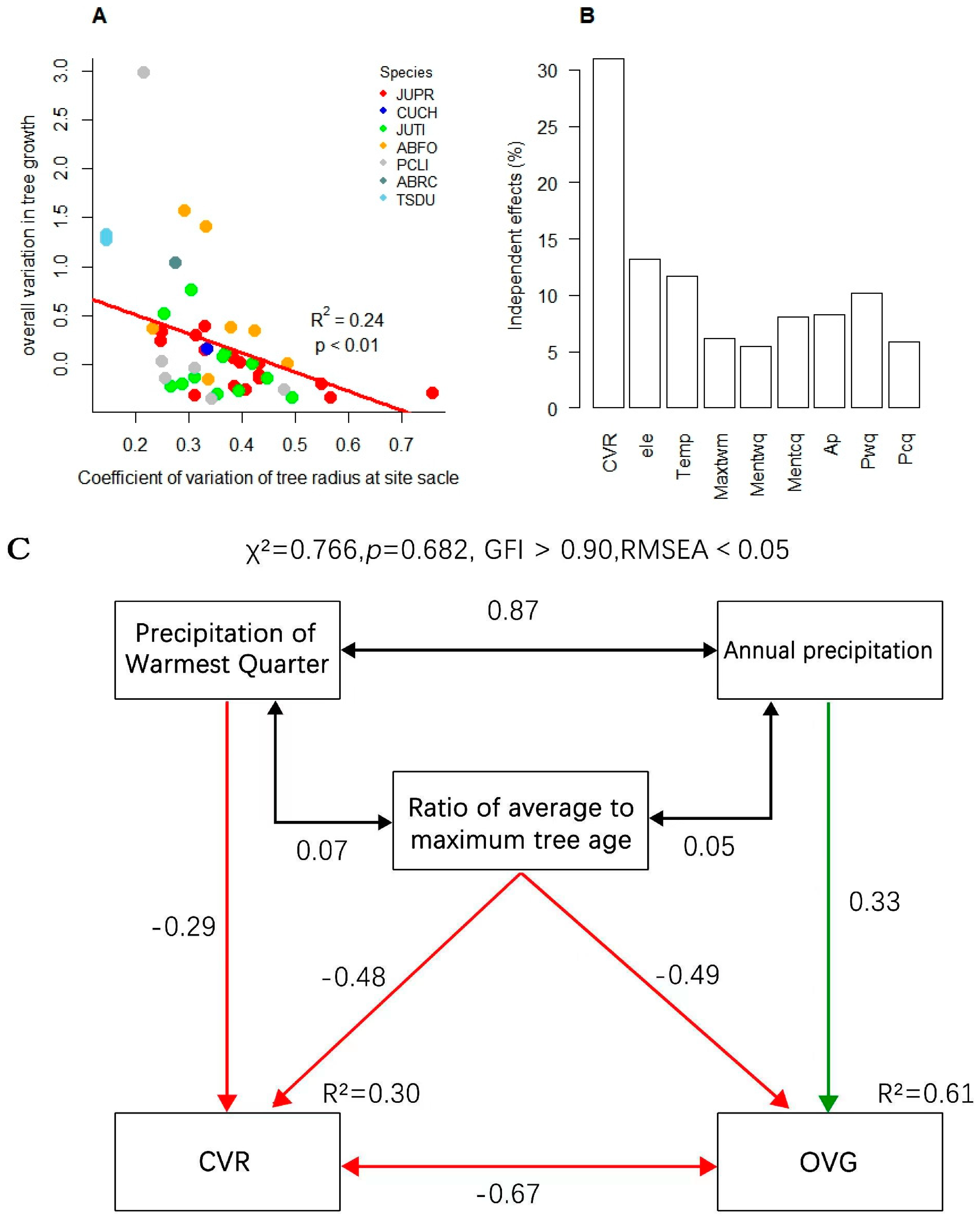
3.2. Effect of Tree Size Heterogeneity on Overall Growth Variability
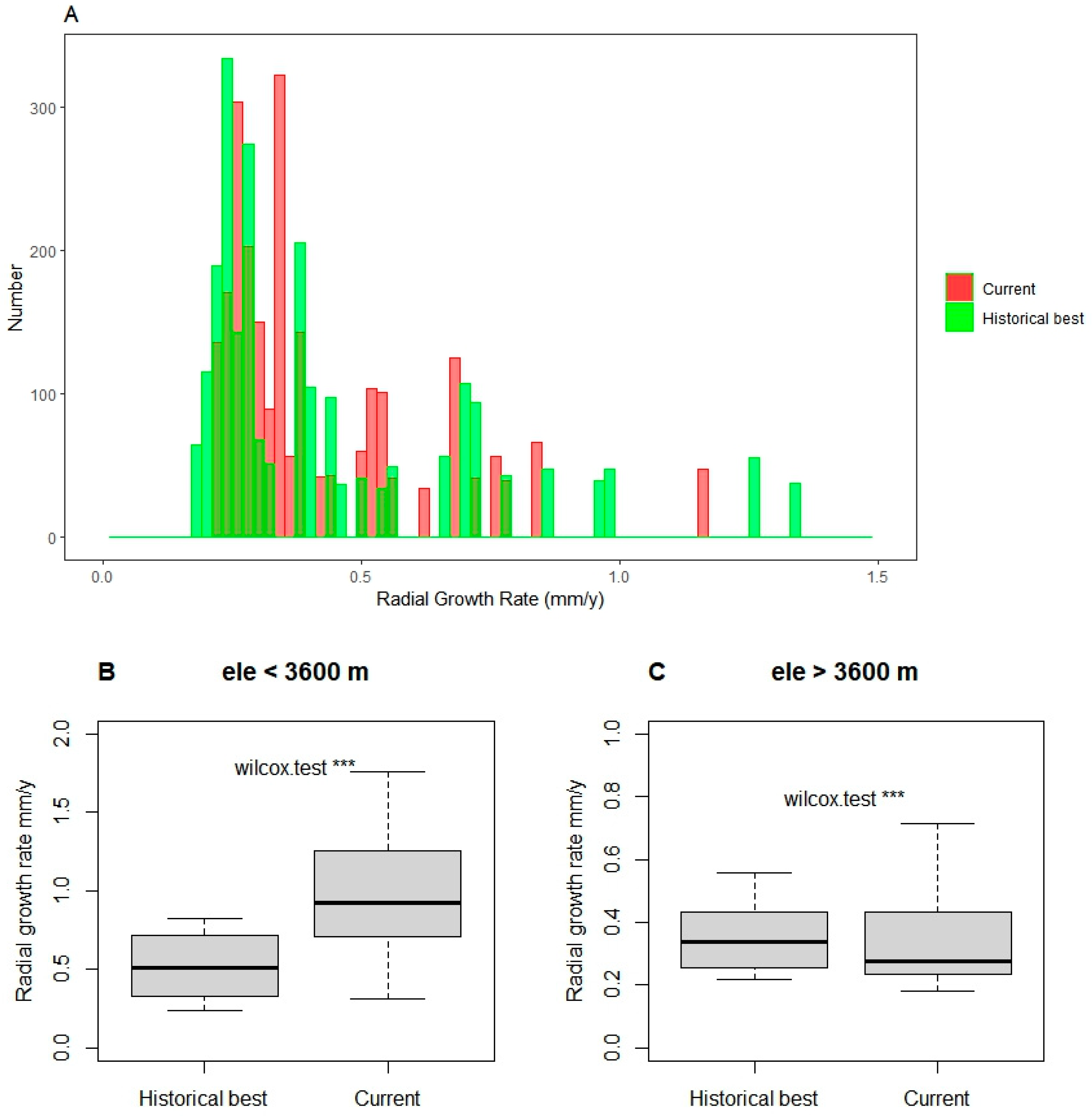
3.3. Tree Radial Growth Assessment
4. Discussion
4.1. Tree Size or Radius Constrains Its Radial Growth and Growth-Climate Sensitivity
4.2. Limited Influence of Climate on Tree Size Heterogeneity of Subalpine Forests
4.3. Forest Range Response to Tree Size Heterogeneity and Climate Change
4.4. Tree Growth Assessment Based on Generalized Metabolic Growth Theory
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Glossary
| Symbol | Meaning | Unit |
| f(r) | tree ring growth rate | mm/y |
| T | The formation time of unit tissue is primarily controlled genetically and by physiological activities, with the intrinsic or developmental growth rate independent of organism size | y |
| R | Tree maximum radius | mm |
| r | Tree current radius | mm |
| mr | Rate of maintenance respiration per unit of tissue | mg g−1 y−1 |
| gr | Cost of respiration needed to produce a unit of tissue | mg g−1 |
| b | Metabolic exponent, taken here as 0.75 | 1 |
| TGT | Total growth time | y |
| f(r)HGBT | Growth rate of historical best growth trajectory | mm/y |
| OVG | overall average growth variability | 1 |
| c(ri) | current average growth rate over the past five years, for tree i. | mm/y |
| f(ri)HBGT | Estimated historical best growth rate for tree i | mm/y |
| f(r)c | average growth rate over the past five years | mm/y |
| rc | Statistical current diameter | mm |
| L | Statistical tree age | y |
| f(r)m | Statistical average tree ring growth rate | mm/y |
| k | Pending parameter | 1 |
| CVR | coefficient of variation of the tree radius | 1 |
Appendix A. Iterative Growth Model (IGM)
Appendix B


References
- Karadavut, U.; Kayis, S.; Okur, O. A Growth Curve Application to Compare Plant Heights and Dry Weights of Some Wheat Varieties. Am.-Eurasian J. Agric. Environ. Sci. 2008, 3, 888–892. [Google Scholar]
- Ryan, M.G.; Binkley, D.; Fownes, J.H. Age-Related Decline in Forest Productivity: Pattern and Process. In Advances in Ecological Research; Begon, M., Fitter, A.H., Eds.; Academic Press: Cambridge, MA, USA, 1997; Volume 27, pp. 213–262. [Google Scholar]
- Shi, P.-J.; Men, X.-Y.; Sandhu, H.S.; Chakraborty, A.; Li, B.-L.; Ou-Yang, F.; Sun, Y.-C.; Ge, F. The “general” ontogenetic growth model is inapplicable to crop growth. Ecol. Model. 2013, 266, 1–9. [Google Scholar] [CrossRef]
- Johnson, S.E.; Abrams, M.D. Age class, longevity and growth rate relationships: Protracted growth increases in old trees in the eastern United States. Tree Physiol. 2009, 29, 1317–1328. [Google Scholar] [CrossRef] [PubMed]
- Sillett, S.C.; Van Pelt, R.; Koch, G.W.; Ambrose, A.R.; Carroll, A.L.; Antoine, M.E.; Mifsud, B.M. Increasing wood production through old age in tall trees. For. Ecol. Manag. 2010, 259, 976–994. [Google Scholar] [CrossRef]
- Stephenson, N.L.; Das, A.J.; Condit, R.; Russo, S.E.; Baker, P.J.; Beckman, N.G.; Coomes, D.A.; Lines, E.R.; Morris, W.K.; Rüger, N.; et al. Rate of tree carbon accumulation increases continuously with tree size. Nature 2014, 507, 90–93. [Google Scholar] [CrossRef]
- Sheil, D.; Eastaugh, C.S.; Vlam, M.; Zuidema, P.A.; Groenendijk, P.; Sleen, P.; Jay, A.; Vanclay, J. Does biomass growth increase in the largest trees? Flaws, fallacies and alternative analyses. Funct. Ecol. 2017, 31, 568–581. [Google Scholar] [CrossRef]
- Begović, K.; Schurman, J.S.; Svitok, M.; Pavlin, J.; Langbehn, T.; Svobodová, K.; Mikoláš, M.; Janda, P.; Synek, M.; Marchand, W.; et al. Large old trees increase growth under shifting climatic constraints: Aligning tree longevity and individual growth dynamics in primary mountain spruce forests. Glob. Chang. Biol. 2023, 29, 143–164. [Google Scholar] [CrossRef]
- Marshall, D.J.; White, C.R. Have We Outgrown the Existing Models of Growth? Trends Ecol. Evol. 2019, 34, 102–111. [Google Scholar] [CrossRef]
- Hérault, B.; Bachelot, B.; Poorter, L.; Rossi, V.; Bongers, F.; Chave, J.; Paine, C.E.T.; Wagner, F.; Baraloto, C. Functional traits shape ontogenetic growth trajectories of rain forest tree species. J. Ecol. 2011, 99, 1431–1440. [Google Scholar] [CrossRef]
- Yao, Y.; Shu, S.; Wang, W.; Liu, R.; Wang, Y.; Wang, X.; Zhang, S. Growth and carbon sequestration of poplar plantations on the Tibetan Plateau. Ecol. Indic. 2023, 147, 109930. [Google Scholar] [CrossRef]
- Zuo, W.; Moses, M.E.; West, G.B.; Hou, C.; Brown, J.H. A general model for effects of temperature on ectotherm ontogenetic growth and development. Proc. R. Soc. B Boil. Sci. 2012, 279, 1840–1846. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.; Zuo, W.; Moses, M.E.; Woodruff, W.H.; Brown, J.H.; West, G.B. Energy Uptake and Allocation During Ontogeny. Science 2008, 322, 736–739. [Google Scholar] [CrossRef] [PubMed]
- Huxley, J.S.; Teissier, G. Terminology of Relative Growth. Nature 1936, 137, 780–781. [Google Scholar] [CrossRef]
- Shu, S.-M.; Zhu, W.-Z.; Kontsevich, G.; Zhao, Y.-Y.; Wang, W.-Z.; Zhao, X.-X.; Wang, X.-D. A discrete model of ontogenetic growth. Ecol. Model. 2021, 460, 109752. [Google Scholar] [CrossRef]
- Nepstad, D.C.; Tohver, I.M.; Ray, D.; Moutinho, P.; Cardinot, G. Mortality of large trees and lianas following experimental drought in an amazon forest. Ecology 2007, 88, 2259–2269. [Google Scholar] [CrossRef]
- Shu, S.-M.; Zhu, W.-Z.; Wang, W.-Z.; Jia, M.; Zhang, Y.-Y.; Sheng, Z.-L. Effects of tree size heterogeneity on carbon sink in old forests. For. Ecol. Manag. 2019, 432, 637–648. [Google Scholar] [CrossRef]
- O’Hara, K.L.; Ramage, B.S. Silviculture in an uncertain world: Utilizing multi-aged management systems to integrate disturbance. For. Int. J. For. Res. 2013, 86, 401–410. [Google Scholar] [CrossRef]
- Forrester, D.I. Linking forest growth with stand structure: Tree size inequality, tree growth or resource partitioning and the asymmetry of competition. For. Ecol. Manag. 2019, 447, 139–157. [Google Scholar] [CrossRef]
- Wang, W.; Jia, M.; Wang, G.; Zhu, W.; McDowell, N.G. Rapid warming forces contrasting growth trends of subalpine fir (Abies fabri) at higher- and lower-elevations in the eastern Tibetan Plateau. For. Ecol. Manag. 2017, 402, 135–144. [Google Scholar] [CrossRef]
- Pepin, N.; Bradley, R.S.; Diaz, H.F.; Baraer, M.; Caceres, E.B.; Forsythe, N.; Fowler, H.; Greenwood, G.; Hashmi, M.Z.; Liu, X.D.; et al. Elevation-dependent warming in mountain regions of the world. Nat. Clim. Chang. 2015, 5, 424–430. [Google Scholar] [CrossRef]
- Lyu, L.; Deng, X.; Zhang, Q.-B. Elevation Pattern in Growth Coherency on the Southeastern Tibetan Plateau. PLoS ONE 2016, 11, e0163201. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.-X.; Braeuning, A.; Cao, K.-F.; Zhu, S.-D. Growth-climate responses of high-elevation conifers in the central Hengduan Mountains, southwestern China. For. Ecol. Manag. 2009, 258, 306–313. [Google Scholar] [CrossRef]
- Dang, H.; Zhang, Y.; Zhang, K.; Jiang, M.; Zhang, Q. Climate-growth relationships of subalpine fir (Abies fargesii) across the altitudinal range in the Shennongjia Mountains, central China. Clim. Chang. 2013, 117, 903–917. [Google Scholar] [CrossRef]
- Shi, S.; Liu, G.; Li, Z.; Ye, X. Elevation-dependent growth trends of forests as affected by climate warming in the southeastern Tibetan Plateau. For. Ecol. Manag. 2021, 498, 119551. [Google Scholar] [CrossRef]
- Thornley, J.H.M.; Cannell, M.G.R. Erratum: Modelling the Components of Plant Respiration: Representation and Realism. Ann. Bot. 2000, 85, 937. [Google Scholar] [CrossRef]
- Clarke, A. Energy Flow in Growth and Production. Trends Ecol. Evol. 2019, 34, 502–509. [Google Scholar] [CrossRef]
- Mori, S.; Yamaji, K.; Ishida, A.; Prokushkin, S.G.; Masyagina, O.V.; Hagihara, A.; Hoque, A.; Suwa, R.; Osawa, A.; Nishizono, T.; et al. Mixed-power scaling of whole-plant respiration from seedlings to giant trees. Proc. Natl. Acad. Sci. USA 2010, 107, 1447–1451. [Google Scholar] [CrossRef]
- West, G.B.; Brown, J.H.; Enquist, B.J. A general model for the structure and allometry of plant vascular systems. Nature 1999, 400, 664–667. [Google Scholar] [CrossRef]
- Cheng, D.-L.; Li, T.; Zhong, Q.-L.; Wang, G.-X. Scaling relationship between tree respiration rates and biomass. Biol. Lett. 2010, 6, 715–717. [Google Scholar] [CrossRef]
- Lai, J.; Zou, Y.; Zhang, J.; Peres-Neto, P.R. Generalizing hierarchical and variation partitioning in multiple regression and canonical analyses using the rdacca.hp R package. Methods Ecol. Evol. 2022, 13, 782–788. [Google Scholar] [CrossRef]
- Jackson, D.L. Revisiting Sample Size and Number of Parameter Estimates: Some Support for the N:q Hypothesis. Struct. Equ. Model. A Multidiscip. J. 2003, 10, 128–141. [Google Scholar] [CrossRef]
- Wang, X.; Wang, T.; Xu, J.; Shen, Z.; Yang, Y.; Chen, A.; Wang, S.; Liang, E.; Piao, S. Enhanced habitat loss of the Himalayan endemic flora driven by warming-forced upslope tree expansion. Nat. Ecol. Evol. 2022, 6, 890–899. [Google Scholar] [CrossRef]
- Peltier, D.M.P.; Ogle, K. Tree growth sensitivity to climate is temporally variable. Ecol. Lett. 2020, 23, 1561–1572. [Google Scholar] [CrossRef] [PubMed]
- Wilmking, M.; Scharnweber, T.; van der Maaten-Theunissen, M.; van der Maaten, E. Reconciling the community with a concept—The uniformitarian principle in the dendro-sciences. Dendrochronologia 2017, 44, 211–214. [Google Scholar] [CrossRef]
- Wilmking, M.; van der Maaten-Theunissen, M.; van der Maaten, E.; Scharnweber, T.; Buras, A.; Biermann, C.; Gurskaya, M.; Hallinger, M.; Lange, J.; Shetti, R.; et al. Global assessment of relationships between climate and tree growth. Glob. Chang. Biol. 2020, 26, 3212–3220. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Xu, G.; Chen, T.; Liu, X.; Zhang, Y.; An, W.; Wang, W.; Fang, Z.-A.; Yu, S. Age-dependent tree-ring growth responses of Schrenk spruce (Picea schrenkiana) to climate—A case study in the Tianshan Mountain, China. Dendrochronologia 2013, 31, 318–326. [Google Scholar] [CrossRef]
- Sun, J.; Liu, Y. Age-independent climate-growth response of Chinese pine (Pinus tabulaeformis Carrière) in North China. Trees 2015, 29, 397–406. [Google Scholar] [CrossRef]
- Coomes, D.A.; Holdaway, R.J.; Kobe, R.K.; Lines, E.R.; Allen, R.B. A general integrative framework for modelling woody biomass production and carbon sequestration rates in forests. J. Ecol. 2012, 100, 42–64. [Google Scholar] [CrossRef]
- Pillet, M.; Joetzjer, E.; Belmin, C.; Chave, J.; Ciais, P.; Dourdain, A.; Evans, M.; Hérault, B.; Luyssaert, S.; Poulter, B. Disentangling competitive vs. climatic drivers of tropical forest mortality. J. Ecol. 2018, 106, 1165–1179. [Google Scholar] [CrossRef]
- Mencuccini, M.; Martinez-Vilalta, J.; Vanderklein, D.; Abdul-Hamid, H.; Korakaki, E.; Lee, S.; Michiels, B. Size-mediated ageing reduces vigour in trees: Size Reduces Vigour in Tall Trees. Ecol. Lett. 2005, 8, 1183–1190. [Google Scholar] [CrossRef]
- Vieira, J.; Nabais, C.; Campelo, F. Extreme Growth Increments Reveal Local and Regional Climatic Signals in Two Pinus pinaster Populations. Front. Plant Sci. 2021, 12, 658777. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, A.H.; Graumlich, L.J. Holocene dynamics of treeline forests in the sierra nevada. Ecology 1997, 78, 1199–1210. [Google Scholar] [CrossRef]
- Sigdel, S.R.; Wang, Y.; Camarero, J.J.; Zhu, H.; Liang, E.; Peñuelas, J. Moisture-mediated responsiveness of treeline shifts to global warming in the Himalayas. Glob. Chang. Biol. 2018, 24, 5549–5559. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Piao, S.; Ciais, P.; Myneni, R.B.; Chen, A.; Chevallier, F.; Dolman, A.J.; Janssens, I.A.; Peñuelas, J.; Zhang, G.; et al. Asymmetric effects of daytime and night-time warming on Northern Hemisphere vegetation. Nature 2013, 501, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Nehrbass-Ahles, C.; Babst, F.; Klesse, S.; Nötzli, M.; Bouriaud, O.; Neukom, R.; Dobbertin, M.; Frank, D. The influence of sampling design on tree-ring-based quantification of forest growth. Glob. Chang. Biol. 2014, 20, 2867–2885. [Google Scholar] [CrossRef]
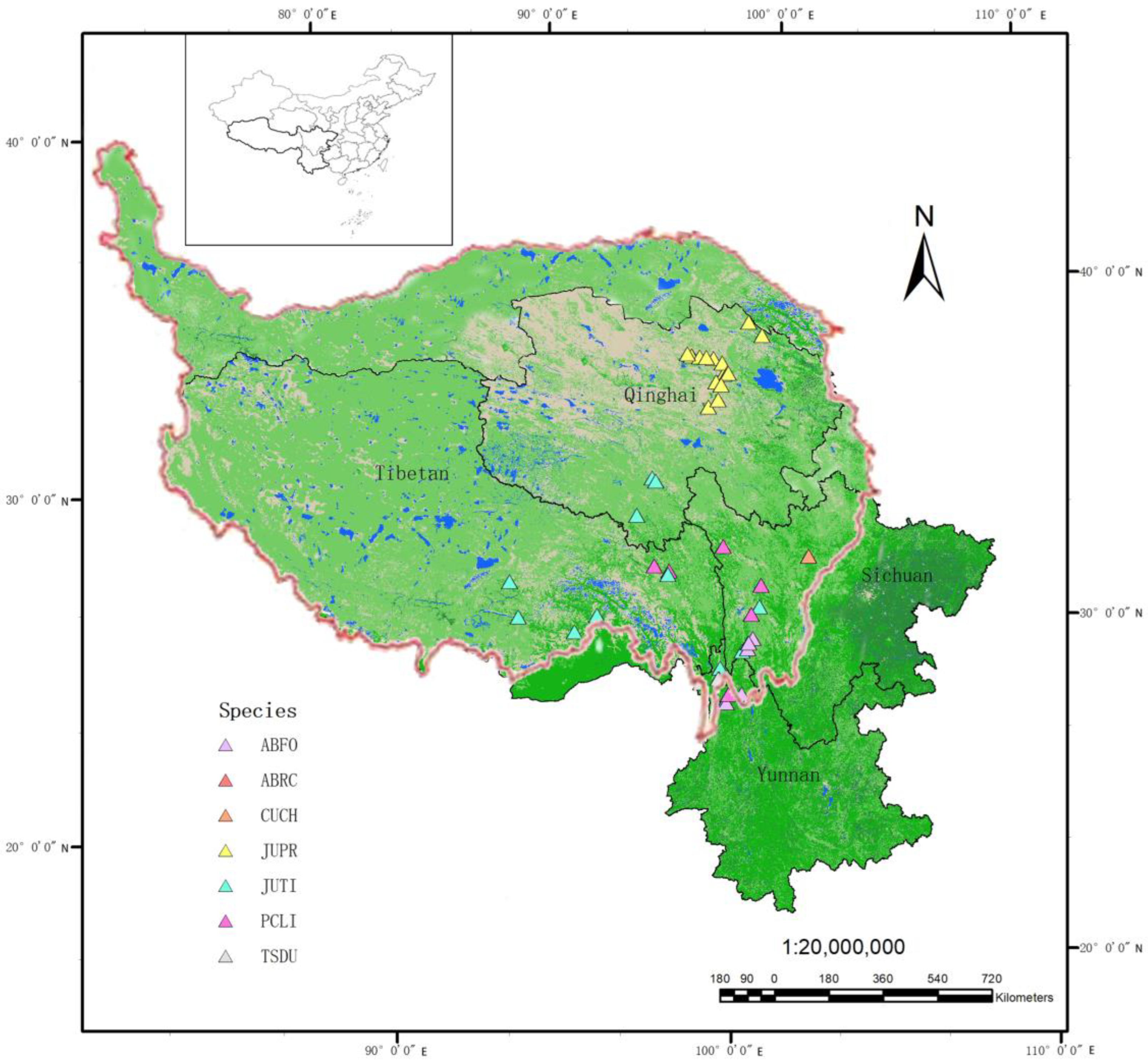
| Species Name | Abbreviated Name | Latitude | Longitude | Average Elevation (m) | Number of Sample Sites/Tree Cores | Species Composition | Age Structure | Average ± SD/Maximum DBH (mm) | Average ± SD/Maximum Age (y) |
|---|---|---|---|---|---|---|---|---|---|
| Abies forestii | ABFO | 27.33–29.28 | 99.27–100.08 | 3521 | 7/345 | single species | Single/mixed age | 202 ± 94/386 | 261 ± 121/463 |
| Abies recurvata | ABRC | 28.04 | 99.02 | 3200 | 1/18 | - | Single age | 276 ± 76/394 | 272 ± 88/394 |
| Cupressus chengiana | CUCH | 31.78 | 101.9167 | 2500 | 1/39 | single species | - | 218 ± 73/330 | 210 ± 88/358 |
| Juniperus przewalskii | JUPR | 36.00–38.57 | 97.06–99.87 | 3741 | 16/1256 | - | Single/mixed age | 162 ± 80/294 | 552 ± 288/1046 |
| Juniperus tibetica | JUTI | 28.37–33.80 | 91.52–100.27 | 4136 | 12/549 | single species | Single/mixed age | 178 ± 81/323 | 407 ± 200/795 |
| Picea likiangensis | PCLI | 27.58–31.95 | 96.48–100.28 | 3520 | 6/195 | - | - | 212 ± 83/341 | 232 ± 102/439 |
| Tsuga dumosa | TSDU | 27.88–28.04 | 98.40–98.98 | 3125 | 2/63 | - | Single age | 310 ± 44/362 | 293 ± 82/460 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Shu, S.; Wang, X.; Chen, W. Effect of Tree Size Heterogeneity on the Overall Growth Trend of Trees in Coniferous Forests of the Tibetan Plateau. Forests 2023, 14, 1483. https://doi.org/10.3390/f14071483
Wang Y, Shu S, Wang X, Chen W. Effect of Tree Size Heterogeneity on the Overall Growth Trend of Trees in Coniferous Forests of the Tibetan Plateau. Forests. 2023; 14(7):1483. https://doi.org/10.3390/f14071483
Chicago/Turabian StyleWang, Yuelin, Shumiao Shu, Xiaodan Wang, and Wende Chen. 2023. "Effect of Tree Size Heterogeneity on the Overall Growth Trend of Trees in Coniferous Forests of the Tibetan Plateau" Forests 14, no. 7: 1483. https://doi.org/10.3390/f14071483
APA StyleWang, Y., Shu, S., Wang, X., & Chen, W. (2023). Effect of Tree Size Heterogeneity on the Overall Growth Trend of Trees in Coniferous Forests of the Tibetan Plateau. Forests, 14(7), 1483. https://doi.org/10.3390/f14071483





