TiO2-WO3 Loaded onto Wood Surface for Photocatalytic Degradation of Formaldehyde
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
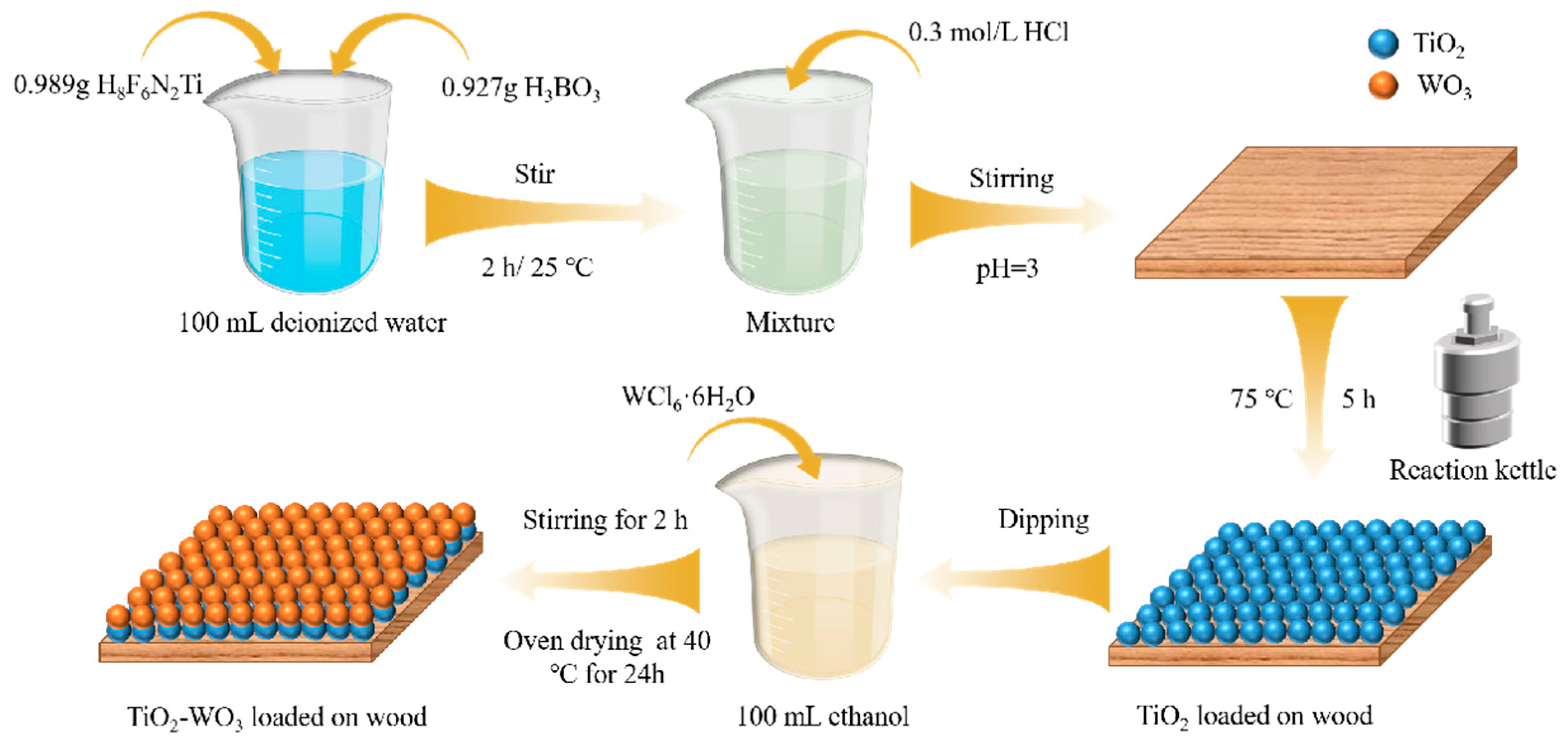
2.2. Preparation of TiO2 Loaded onto Wood
2.3. Preparation of TiO2-WO3 Loaded onto Wood
2.4. Characterization
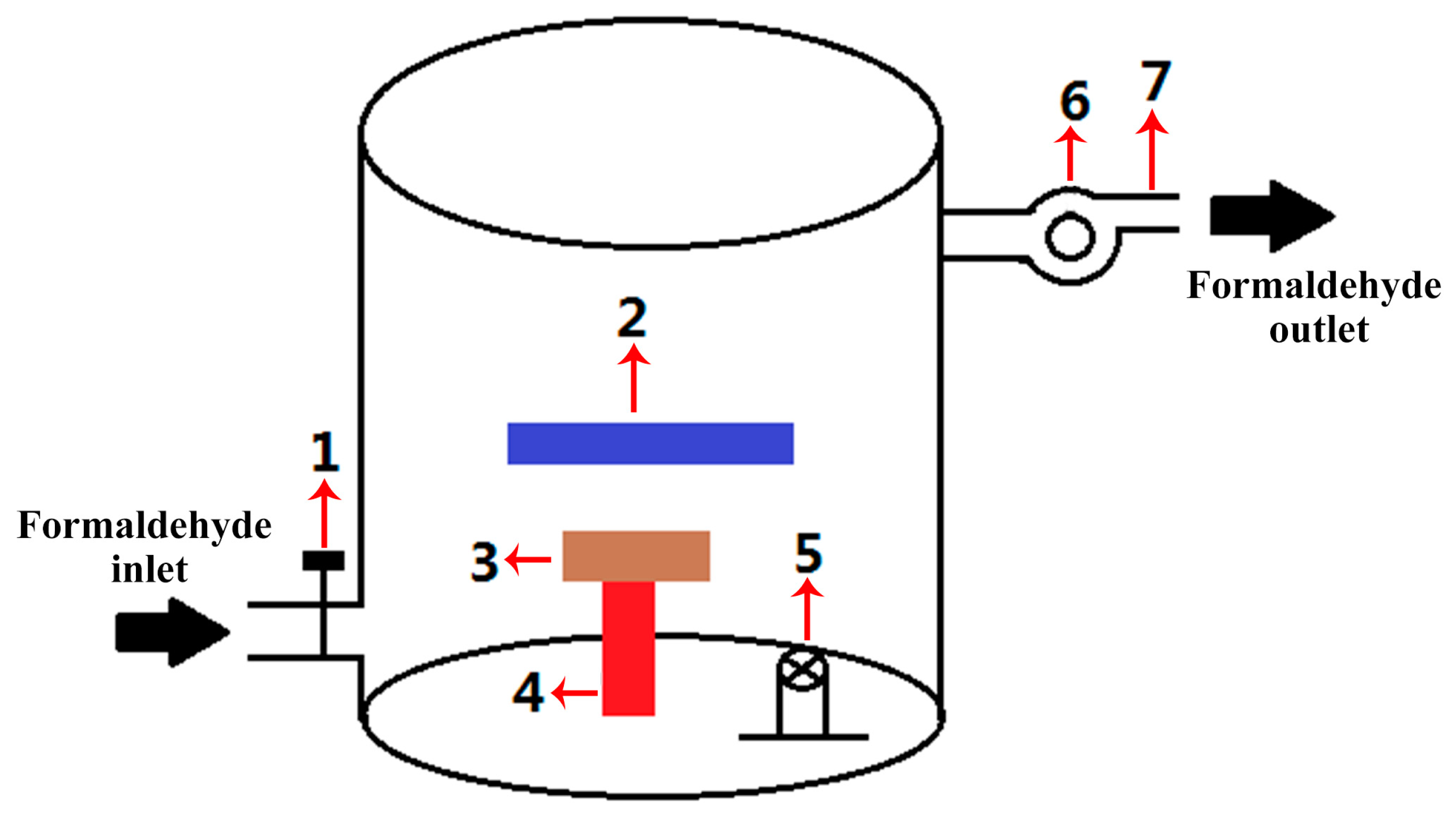
2.5. Formaldehyde Degradation Performance Test
3. Results and Discussion
3.1. Morphological Characterizations
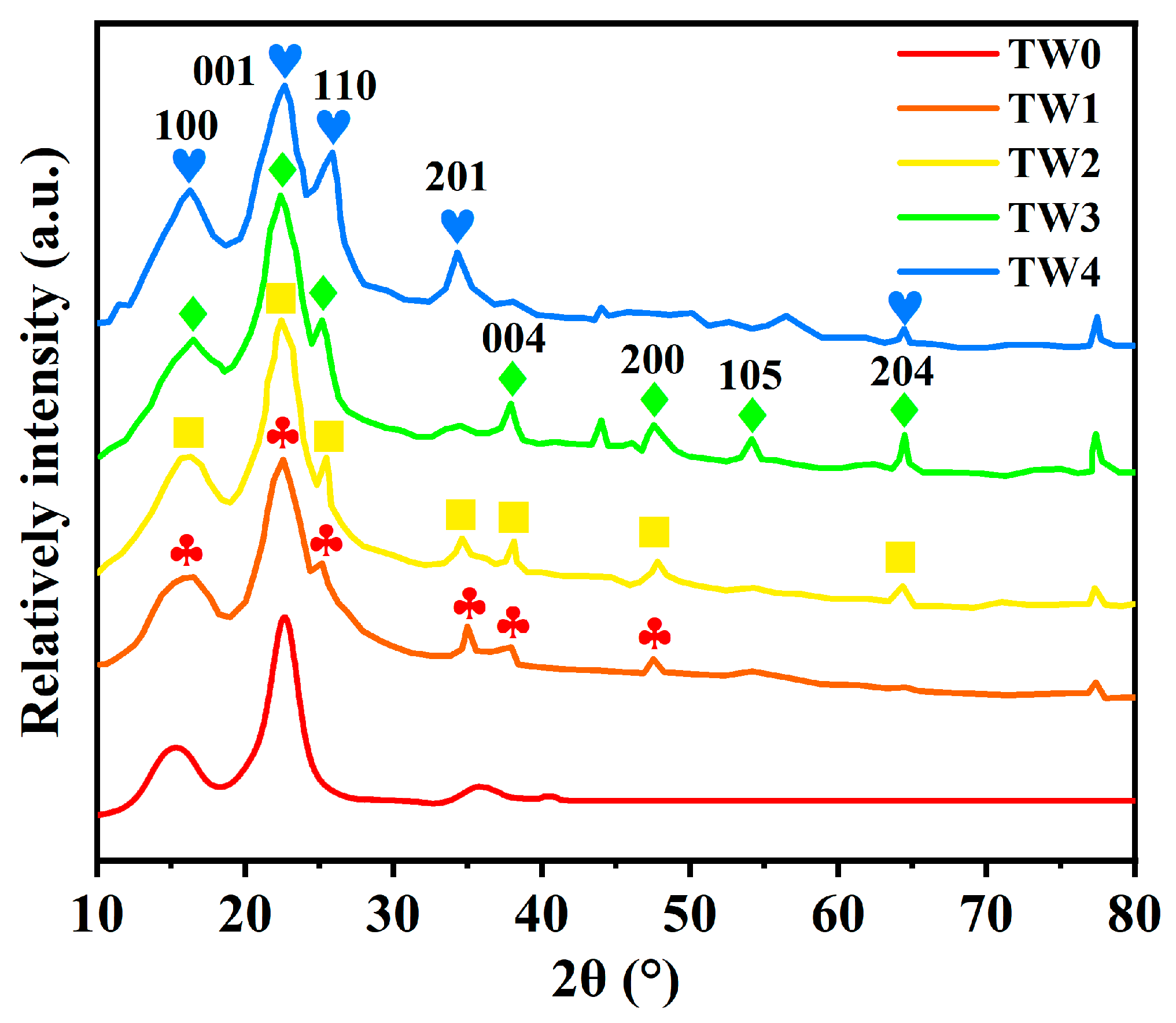
3.2. XRD Analysis
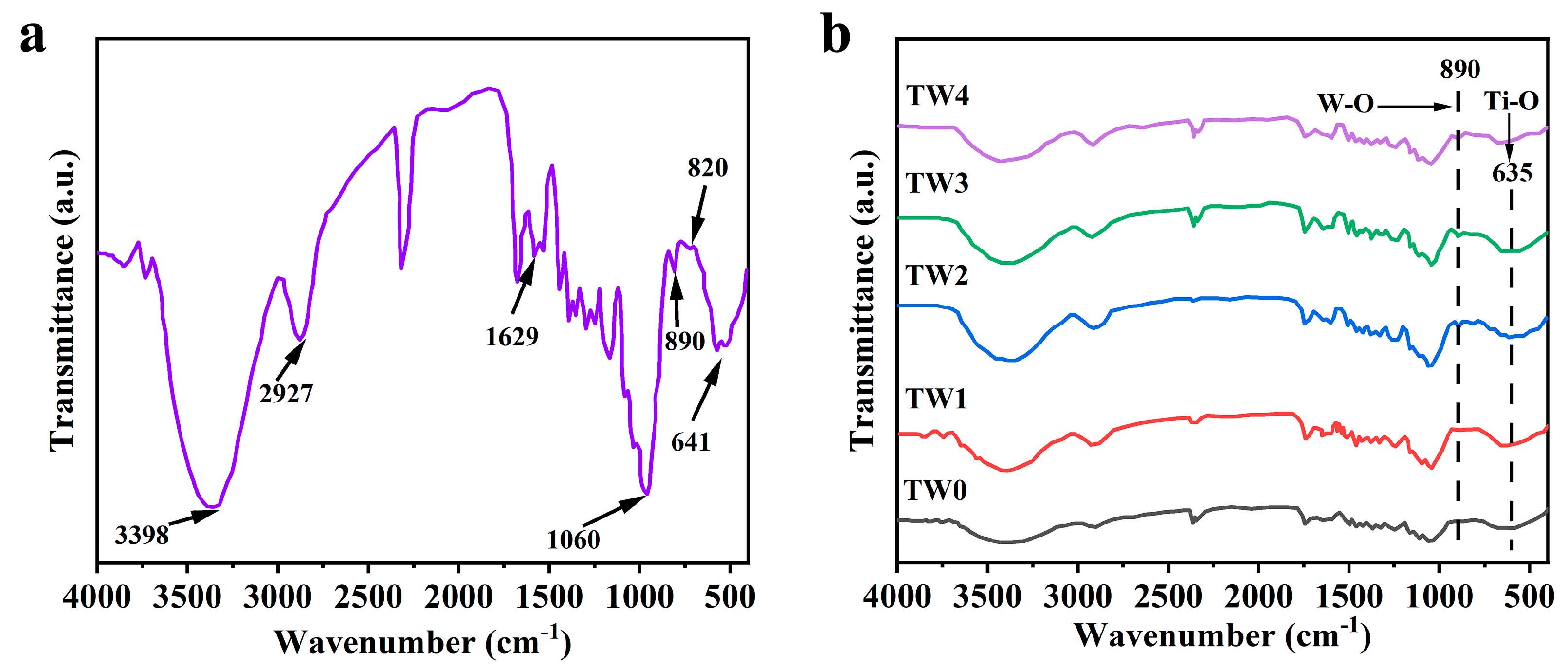
3.3. ATR-FTIR Analysis
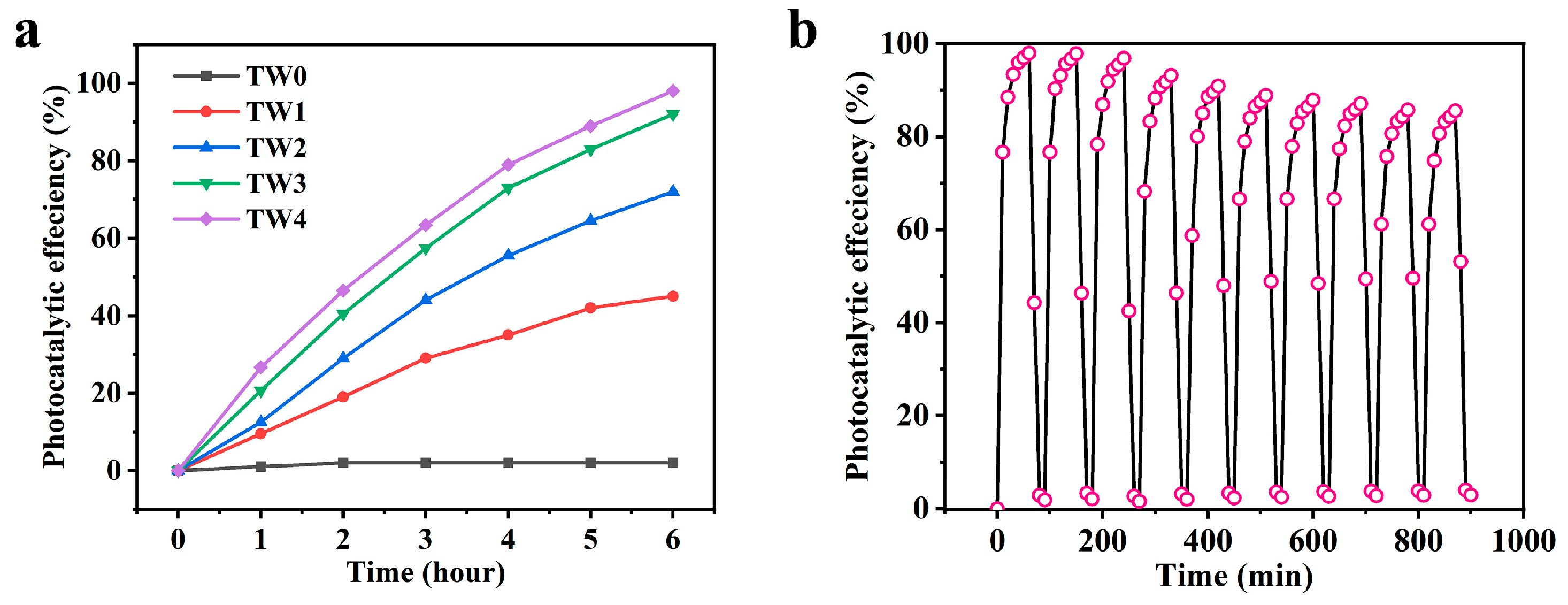
3.4. Photocatalytic Degradation Performance
3.5. Mechanism of Photocatalytic Degradation of Formaldehyde
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malayeri, M.; Bahri, M.; Haghighat, F.; Shah, A. Impact of air distribution on indoor formaldehyde abatement with/without passive removal material: A CFD modeling. Build. Environ. 2022, 212, 108792. [Google Scholar] [CrossRef]
- Li, X.; Shi, R.; Li, J. Effect of clay on properties of melamine-urea-formaldehyde resin. J. For. Eng. 2022, 7, 78–83. [Google Scholar]
- Wang, X.; Hong, S.; Lian, H.; Zhan, X.; Cheng, M.; Huang, Z.; Manzo, M.; Cai, L.; Nadda, A.; Van Le, Q. Photocatalytic degradation of surface-coated tourmaline-titanium dioxide for self-cleaning of formaldehyde emitted from furniture. J. Hazard. Mater. 2021, 420, 126565. [Google Scholar] [CrossRef]
- Soni, V.; Goel, V.; Singh, P.; Garg, A. Abatement of formaldehyde with photocatalytic and catalytic oxidation: A review. Int. J. Chem. React. Eng. 2021, 19, 1–29. [Google Scholar] [CrossRef]
- Sahrin, N.T.; Nawaz, R.; Chong, F.K.; Lee, S.L.; Wirzal, M.D.H. Current perspectives of anodized TiO2 nanotubes towards photodegradation of formaldehyde: A short review. Environ. Technol. Innov. 2021, 22, 101418. [Google Scholar] [CrossRef]
- Zhu, W.; Kim, D.; Han, M.; Jang, J.; Choi, H.; Kwon, G.; Jeon, Y.; Ryu, D.Y.; Lim, S.-H.; You, J.; et al. Fibrous Cellulose Nanoarchitectonics on N-doped Carbon-based Metal-Free Catalytic Nanofilter for Highly Efficient Advanced Oxidation Process. Chem. Eng. J. 2023, 460, 141593. [Google Scholar] [CrossRef]
- Qin, X.; Liu, Y.; Xu, Y.; Wu, L.; Huang, K.; Huang, J.; Wang, W. Effect of microencapsulation of formaldehyde capture agents on formaldehyde emission from veneered panels. J. For. Eng. 2020, 5, 81–87. [Google Scholar]
- Ran, M.; Cui, W.; Li, K.; Chen, L.; Zhang, Y.; Dong, F.; Sun, Y. Light-induced dynamic stability of oxygen vacancies in BiSbO4 for efficient photocatalytic formaldehyde degradation. Energy Environ. Mater. 2022, 5, 305–312. [Google Scholar] [CrossRef]
- Lin, Z.; Shen, W.; Corriou, J.-P.; Chen, X.; Xi, H. Assessment of multiple environmental factors on the adsorptive and photocatalytic removal of gaseous formaldehyde by a nano-TiO2 colloid: Experimental and simulation studies. J. Colloid Interface Sci. 2022, 608, 1769–1781. [Google Scholar] [CrossRef]
- Wang, J.; Wu, X.; Wang, Y.; Zhao, W.; Zhao, Y.; Zhou, M.; Wu, Y.; Ji, G. Green, Sustainable Architectural Bamboo with High Light Transmission and Excellent Electromagnetic Shielding as a Candidate for Energy-Saving Buildings. Nano-Micro Lett. 2023, 15, 11. [Google Scholar] [CrossRef]
- Grönquist, P.; Wood, D.; Hassani, M.M.; Wittel, F.K.; Menges, A.; Rüggeberg, M. Analysis of hygroscopic self-shaping wood at large scale for curved mass timber structures. Sci. Adv. 2019, 5, eaax1311. [Google Scholar] [CrossRef] [PubMed]
- Gan, W.; Wang, Y.; Xiao, K.; Zhai, M.; Wang, H.; Xie, Y. Research review of energy storage and conversion materials based on wood cell wall functional modification. J. For. Eng. 2022, 7, 1–12. [Google Scholar]
- Hill, C.; Altgen, M.; Rautkari, L. Thermal modification of wood—A review: Chemical changes and hygroscopicity. J. Mater. Sci. 2021, 56, 6581–6614. [Google Scholar] [CrossRef]
- Ijaz, M.; Zafar, M. Titanium dioxide nanostructures as efficient photocatalyst: Progress, challenges and perspective. Int. J. Energy Res. 2021, 45, 3569–3589. [Google Scholar] [CrossRef]
- Wang, J.; Hu, X.; Li, M.; Shao, L.; Zhan, J. Preparation of wood-based yttrium doped TiO2 film and its photocatalytic properties. J. For. Eng. 2021, 6, 51–57. [Google Scholar]
- Li, Z.; Wang, S.; Wu, J.; Zhou, W. Recent progress in defective TiO2 photocatalysts for energy and environmental applications. Renew. Sustain. Energy Rev. 2022, 156, 111980. [Google Scholar] [CrossRef]
- Yuan, D.; Liu, Y.; Wang, W.; Gao, L.; Cheng, J. Preparation of titanium dioxide based on poplar template and its application on formaldehyde degradation. J. For. Eng. 2020, 5, 34–40. [Google Scholar]
- Basavarajappa, P.S.; Patil, S.B.; Ganganagappa, N.; Reddy, K.R.; Raghu, A.V.; Reddy, C.V. Recent progress in metal-doped TiO2, non-metal doped/codoped TiO2 and TiO2 nanostructured hybrids for enhanced photocatalysis. Int. J. Hydrog. Energy 2020, 45, 7764–7778. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhu, W.; Li, Z.; Feng, Y.; Qi, W.; Li, S.; Wang, X.; Chen, M. Enhancement of Wood Coating Properties by Adding Silica Sol to UV-Curable Waterborne Acrylics. Forests 2023, 14, 335. [Google Scholar] [CrossRef]
- Guo, Q.; Zhou, C.; Ma, Z.; Yang, X. Fundamentals of TiO2 photocatalysis: Concepts, mechanisms, and challenges. Adv. Mater. 2019, 31, 1901997. [Google Scholar] [CrossRef]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, C.P.M.; Moreira, V.R.; Lebron, Y.A.R.; de Vasconcelos, C.K.B.; Koch, K.; Viana, M.M.; Drewes, J.E.; Amaral, M.C.S. Converting recycled membranes into photocatalytic membranes using greener TiO2-GRAPHENE oxide nanomaterials. Chemosphere 2022, 306, 135591. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.; Lim, C.; Lee, H.; Kim, S.; Lee, Y.-S. Visible light photocatalytic activity of TiO2 with carbon-fluorine heteroatoms simultaneously introduced by CF4 plasma. Korean J. Chem. Eng. 2022, 39, 3334–3342. [Google Scholar] [CrossRef]
- Yu, H.; Zhao, S.; Mao, W.; Faheem, M.; Ding, D.; Chen, L.; Cai, T. Facile synthesis of N-doped TiO2 and its photocatalytic properties for ciprofloxacin degradation. Funct. Mater. Lett. 2022, 15, 2250028. [Google Scholar] [CrossRef]
- Liu, K.; Kong, F.; Zhu, C.; Jiang, G. Photocatalytic activity of phosphorus and nitrogen co-doped carbon quantum dots/TiO2 nanosheets. Nano 2020, 15, 2050151. [Google Scholar] [CrossRef]
- Chen, D.; Cheng, Y.; Zhou, N.; Chen, P.; Wang, Y.; Li, K.; Huo, S.; Cheng, P.; Peng, P.; Zhang, R. Photocatalytic degradation of organic pollutants using TiO2-based photocatalysts: A review. J. Clean. Prod. 2020, 268, 121725. [Google Scholar] [CrossRef]
- Zhang, L.; Qin, M.; Yu, W.; Zhang, Q.; Xie, H.; Sun, Z.; Shao, Q.; Guo, X.; Hao, L.; Zheng, Y. Heterostructured TiO2/WO3 nanocomposites for photocatalytic degradation of toluene under visible light. J. Electrochem. Soc. 2017, 164, H1086. [Google Scholar] [CrossRef]
- Samuel, O.; Othman, M.H.D.; Kamaludin, R.; Sinsamphanh, O.; Abdullah, H.; Puteh, M.H.; Kurniawan, T.A. WO3–based photocatalysts: A review on synthesis, performance enhancement and photocatalytic memory for environmental applications. Ceram. Int. 2021, 48, 5845–5875. [Google Scholar] [CrossRef]
- Tahir, M.; Ali, S.; Rizwan, M. A review on remediation of harmful dyes through visible light-driven WO3 photocatalytic nanomaterials. Int. J. Environ. Sci. Technol. 2019, 16, 4975–4988. [Google Scholar] [CrossRef]
- Thirumoorthi, G.; Gnanavel, B.; Kalaivani, M.; Ragunathan, A.; Venkatesan, H. Suitability of Iron (Fe)-Doped Tungsten Oxide (WO3) Nanomaterials for Photocatalytic and Antibacterial Applications. Int. J. Nanosci. 2021, 20, 2150042. [Google Scholar] [CrossRef]
- Karthikeyan, S.; Selvapandiyan, M.; Sasikumar, P.; Parthibavaraman, M.; Nithiyanantham, S.; Srisuvetha, V. Investigation on the properties of vanadium doping WO3 nanostructures by hydrothermal method. Mater. Sci. Energy Technol. 2022, 5, 411–415. [Google Scholar] [CrossRef]
- Jiang, H.; Li, Y.; Wang, D.; Hong, X.; Liang, B. Recent advances in heteroatom doped graphitic carbon nitride (g-C3N4) and g-C3N4/metal oxide composite photocatalysts. Curr. Org. Chem. 2020, 24, 673–693. [Google Scholar] [CrossRef]
- Zhu, W.; Zhang, Y.; Wang, X.; Wu, Y.; Han, M.; You, J.; Jia, C.; Kim, J. Aerogel nanoarchitectonics based on cellulose nanocrystals and nanofibers from eucalyptus pulp: Preparation and comparative study. Cellulose 2022, 29, 817–833. [Google Scholar] [CrossRef]
- Zhu, Z.; Long, Y.; Xue, X.; Yin, Y.; Zhu, B.; Xu, B. Phase-transition kinetics of calcium-doped TiO2: A high-temperature XRD study. Ceram. Int. 2022, 48, 25056–25063. [Google Scholar] [CrossRef]
- Fan, M.; Lin, Z.; Zhang, P.; Ma, X.; Wu, K.; Liu, M.; Xiong, X. Synergistic effect of nitrogen and sulfur dual-doping endows TiO2 with exceptional sodium storage performance. Adv. Energy Mater. 2021, 11, 2003037. [Google Scholar] [CrossRef]
- Wang, S.; Xu, H.; Hao, T.; Wang, P.; Zhang, X.; Zhang, H.; Xue, J.; Zhao, J.; Li, Y. In situ XRD and operando spectra-electrochemical investigation of tetragonal WO3-x nanowire networks for electrochromic supercapacitors. NPG Asia Mater. 2021, 13, 51. [Google Scholar] [CrossRef]
- Zhu, W.; Han, M.; Kim, D.; Zhang, Y.; Kwon, G.; You, J.; Jia, C.; Kim, J. Facile preparation of nanocellulose/Zn-MOF-based catalytic filter for water purification by oxidation process. Environ. Res. 2022, 205, 112417. [Google Scholar] [CrossRef]
- Wu, L.; Chen, M.; Xu, J.; Fang, F.; Li, S.; Zhu, W. Nano-SiO2-Modified Waterborne Acrylic Acid Resin Coating for Wood Wallboard. Coatings 2022, 12, 1453. [Google Scholar] [CrossRef]
- Li, W.; Chen, Z.; Yu, H.; Li, J.; Liu, S. Wood-Derived Carbon Materials and Light-Emitting Materials. Adv. Mater. 2021, 33, 2000596. [Google Scholar] [CrossRef]
- Sheng, C.; Wang, C.; Wang, H.; Jin, C.; Sun, Q.; Li, S. Self-photodegradation of formaldehyde under visible-light by solid wood modified via nanostructured Fe-doped WO3 accompanied with superior dimensional stability. J. Hazard. Mater. 2017, 328, 127–139. [Google Scholar] [CrossRef]
- Li, X.; Li, H.; Huang, Y.; Cao, J.; Huang, T.; Li, R.; Zhang, Q.; Lee, S.-C.; Ho, W. Exploring the photocatalytic conversion mechanism of gaseous formaldehyde degradation on TiO2–x-OV surface. J. Hazard. Mater. 2022, 424, 127217. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Zhao, W.; Li, S.; Wang, S.; Fan, Y.; Wang, F.; Qiu, X.; Zhu, Y.; Zhang, Y.; Long, C. Elevating Photooxidation of Methane to Formaldehyde via TiO2 Crystal Phase Engineering. J. Am. Chem. Soc. 2022, 144, 15977–15987. [Google Scholar] [CrossRef] [PubMed]


| Samples | TW0 | TW1 | TW2 | TW3 | TW4 | |
|---|---|---|---|---|---|---|
| Molar Ratio | ||||||
| WO3:TiO2 | 0:0 | 1:0 | 1:1 | 1:3 | 1:5 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Li, Z.; Li, L.; Dai, X.; Chen, M.; Zhu, W. TiO2-WO3 Loaded onto Wood Surface for Photocatalytic Degradation of Formaldehyde. Forests 2023, 14, 503. https://doi.org/10.3390/f14030503
Li S, Li Z, Li L, Dai X, Chen M, Zhu W. TiO2-WO3 Loaded onto Wood Surface for Photocatalytic Degradation of Formaldehyde. Forests. 2023; 14(3):503. https://doi.org/10.3390/f14030503
Chicago/Turabian StyleLi, Song, Zequn Li, Luming Li, Xiangdong Dai, Meiling Chen, and Wenkai Zhu. 2023. "TiO2-WO3 Loaded onto Wood Surface for Photocatalytic Degradation of Formaldehyde" Forests 14, no. 3: 503. https://doi.org/10.3390/f14030503
APA StyleLi, S., Li, Z., Li, L., Dai, X., Chen, M., & Zhu, W. (2023). TiO2-WO3 Loaded onto Wood Surface for Photocatalytic Degradation of Formaldehyde. Forests, 14(3), 503. https://doi.org/10.3390/f14030503







