Pollination Potential of Riparian Hardwood Forests—A Multifaceted Field-Based Assessment in the Vistula Valley, Poland
Abstract
:1. Introduction
- Develop a comprehensive set of indicators to measure pollination potential;
- Calculate indicator values and propose the level of an indicator (based on the distribution of values) reflecting the potential of an ecosystem type;
- Determine mutual relations among indicators.
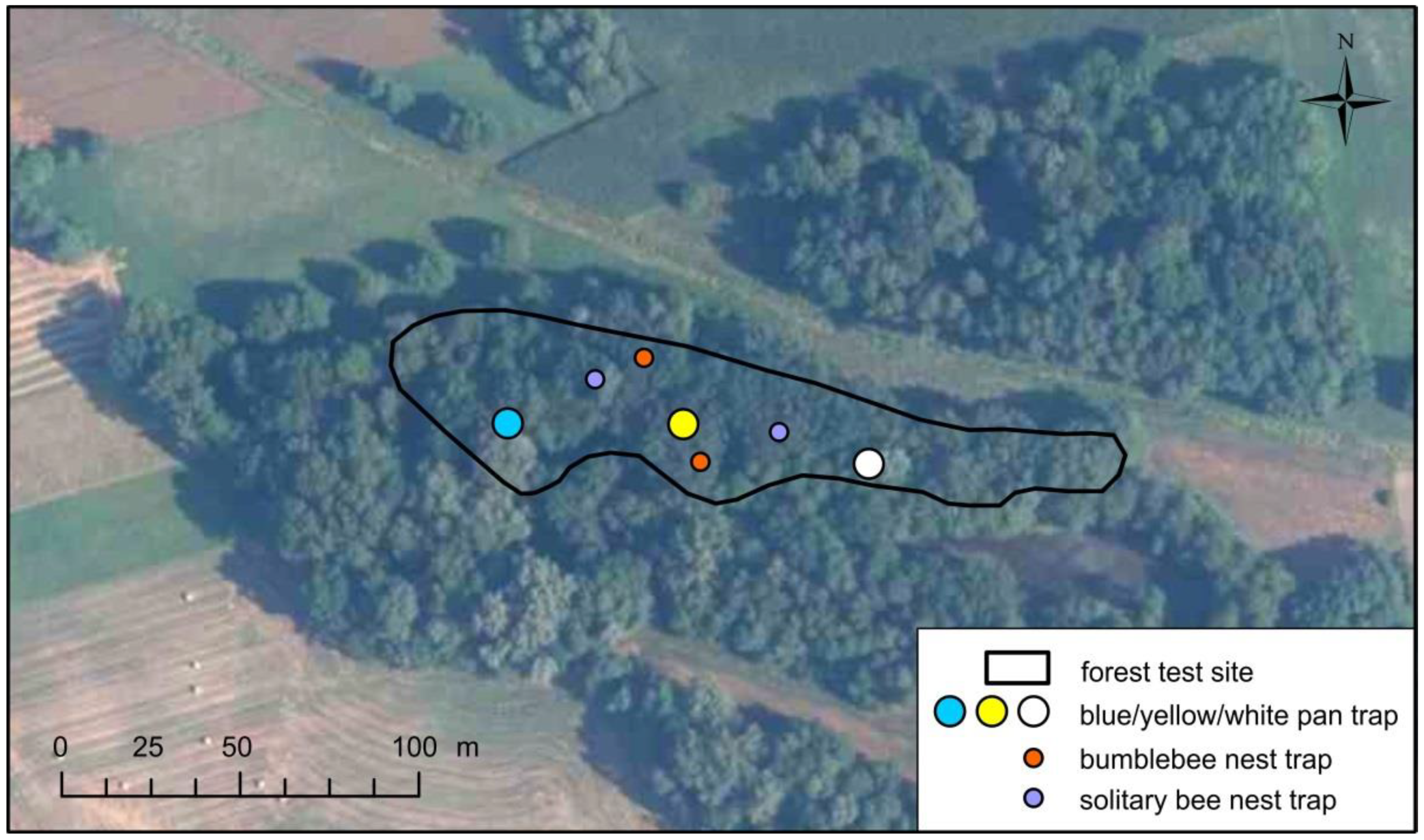
2. Study Area
- -
- Plant community should be unambiguously recognized in the field as Ficario-Ulmetum minoris association;
- -
- Sites should not be subject to strong human pressure, without any recent visible human impact (timber extraction etc.);
- -
- Tree stand should be older than 60 years.
3. Materials and Methods
3.1. Indicators of Pollination Potential
- (I)
- The potential of the habitat for the occurrence and reproduction of pollinating insects;
- (II)
- The potential of pollinating insects that are present in riparian hardwood forests for pollination of nearby crops.
3.1.1. Habitat Potential
3.1.2. Pollinators Potential
3.2. Threshold Potential Values
3.3. Joint Analysis
4. Results
4.1. Habitat Potential
4.1.1. Suitability for Nesting
4.1.2. Availability of Food Resources
4.2. Pollinators Potential
4.2.1. The Density of Bumblebees
4.2.2. Number of Bees Trapped Daily
4.3. Relationships among Indicators
5. Discussion
5.1. Pollination Potential of Riparian Hardwood Forests
5.2. Methodological Advances and Their Implications
5.3. Linkages among Indicators
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Affek, A.N.; Degórski, M.; Wolski, J.; Solon, J.; Kowalska, A.; Roo-Zielińska, E.; Grabińska, B.; Kruczkowska, B. Ecosystem Service Potentials and Their Indicators in Postglacial Landscapes: Assessment and Mapping; Elsevier: Amsterdam, The Netherlands; Oxford, UK; Cambridge, UK, 2020; ISBN 9780128161340. [Google Scholar]
- TEEB. The Economics of Ecosystems and Biodiversity: Ecological and Economic Foundation; Kumar, P., Ed.; Earthscan: Cambridge, UK, 2010. [Google Scholar]
- Burkhard, B.; Kandziora, M.; Hou, Y.; Müller, F. Ecosystem service potentials, flows and demand—Concepts for spatial localisation, indication and quantification. Landsc. Online 2014, 34, 1–32. [Google Scholar] [CrossRef]
- Bastian, O.; Syrbe, R.U.; Rosenberg, M.; Rahe, D.; Grunewald, K. The five pillar EPPS framework for quantifying, mapping and managing ecosystem services. Ecosyst. Serv. 2013, 4, 15–24. [Google Scholar] [CrossRef]
- Spangenberg, J.H.; von Haaren, C.; Settele, J. The ecosystem service cascade: Further developing the metaphor. Integrating societal processes to accommodate social processes and planning, and the case of bioenergy. Ecol. Econ. 2014, 104, 22–32. [Google Scholar] [CrossRef]
- Bastian, O.; Haase, D.; Grunewald, K. Ecosystem properties, potentials and services—The EPPS conceptual framework and an urban application example. Ecol. Indic. 2012, 21, 7–16. [Google Scholar] [CrossRef]
- Affek, A.N.; Kowalska, A. Ecosystem potentials to provide services in the view of direct users. Ecosyst. Serv. 2017, 26, 183–196. [Google Scholar] [CrossRef]
- Tockner, K.; Stanford, J.A. Riverine flood plains: Present state and future trends. Environ. Conserv. 2002, 29, 308–330. [Google Scholar] [CrossRef] [Green Version]
- European Environment Agency Conservation Status and Trends of Habitats and Species. Available online: https://www.eea.europa.eu/themes/biodiversity/state-of-nature-in-the-eu/article-17-national-summary-dashboards/conservation-status-and-trends (accessed on 14 June 2021).
- Danielewicz, W.; Pawlaczyk, P. Łęgowe lasy dębowo-wiązowo-jesionowe (Ficario-Ulmetum). In Lasy i Bory. Poradniki Ochrony Siedlisk i Gatunków Natura 2000—Podręcznik Metodyczny. Tom 5; Herbich, J., Ed.; Ministerstwo Środowiska: Warszawa, Poland, 2004; pp. 242–258. [Google Scholar]
- Ochrona Przyrody i Środowiska w Dolinach Nizinnych rzek Polski [Nature and Environment Conservation in the Lowland River Valleys of Poland]; Tomiałojć, L. (Ed.) Instytut Ochrony Przyrody PAN: Kraków, Poland, 1993. [Google Scholar]
- Riis, T.; Kelly-Quinn, M.; Aguiar, F.C.; Manolaki, P.; Bruno, D.; Bejarano, M.D.; Clerici, N.; Fernandes, M.R.; Franco, J.C.; Pettit, N.; et al. Global overview of ecosystem services provided by riparian vegetation. BioScience 2020. [Google Scholar] [CrossRef]
- Kowalska, A.; Affek, A.; Wolski, J.; Regulska, E.; Kruczkowska, B.; Zawiska, I.; Kołaczkowska, E.; Baranowski, J. Assessment of regulating ES potential of lowland riparian hardwood forests in Poland. Ecol. Indic. 2021, 120, 106834. [Google Scholar] [CrossRef]
- Haines-Young, R.H.; Potschin, M.B. Common International Classification of Ecosystem Services (CICES) V5.1. and Guidance on the Application of the Revised Structure; Fabis Consulting Ltd: Nottingham, UK, 2018. [Google Scholar]
- Potts, S.G.; Imperatriz-Fonseca, V.; Ngo, H.T.; Aizen, M.A.; Biesmeijer, J.C.; Breeze, T.D.; Dicks, L.V.; Garibaldi, L.A.; Hill, R.; Settele, J.; et al. Safeguarding pollinators and their values to human well-being. Nature 2016, 540, 220–229. [Google Scholar] [CrossRef]
- Ollerton, J.; Winfree, R.; Tarrant, S. How many flowering plants are pollinated by animals? Oikos 2011, 120, 321–326. [Google Scholar] [CrossRef]
- Klein, A.-M.; Vaissiere, B.E.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B 2007, 274, 303–313. [Google Scholar] [CrossRef] [Green Version]
- Lautenbach, S.; Seppelt, R.; Liebscher, J.; Dormann, C.F. Spatial and temporal trends of global pollination benefit. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [Green Version]
- Willmer, P.G.; Cunnold, H.; Ballantyne, G. Insights from measuring pollen deposition: Quantifying the pre-eminence of bees as flower visitors and effective pollinators. Arthropod-Plant Interact. 2017, 11, 411–425. [Google Scholar] [CrossRef] [Green Version]
- Michener, C.D. The Bees of the World, 2nd ed.; The Johns Hopkins University Press: Baltimore, MD, USA, 2007; Volume 85, ISBN 978-0-8018-8573-0. [Google Scholar]
- Nogué, S.; Long, P.R.; Eycott, A.E.; De Nascimento, L.; Fernández-Palacios, J.M.; Petrokofsky, G.; Vandvik, V.; Willis, K.J. Pollination service delivery for European crops: Challenges and opportunities. Ecol. Econ. 2016, 128, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Egoh, B.; Drakou, E.G.; Dunbar, M.B.; Maes, J.; Willemen, L. Indicators for Mapping Ecosystem Services: A Review; Publications Office of the European Union: Luxembourg, 2012. [Google Scholar]
- Kleijn, D.; Winfree, R.; Bartomeus, I.; Carvalheiro, L.G.; Henry, M.; Isaacs, R.; Klein, A.M.; Kremen, C.; M’Gonigle, L.K.; Rader, R.; et al. Delivery of crop pollination services is an insufficient argument for wild pollinator conservation. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [Green Version]
- Bartholomée, O.; Lavorel, S. Disentangling the diversity of definitions for the pollination ecosystem service and associated estimation methods. Ecol. Indic. 2019, 7, 105576. [Google Scholar] [CrossRef]
- Burkhard, B.; Kroll, F.; Nedkov, S.; Müller, F. Mapping ecosystem service supply, demand and budgets. Ecol. Indic. 2012, 21, 17–29. [Google Scholar] [CrossRef]
- Affek, A.N. Indicators of ecosystem potential for pollination and honey production. Ecol. Indic. 2018, 94, 33–45. [Google Scholar] [CrossRef]
- Matuszkiewicz, J.M. Kompleksowe Mapy Roślinności Doliny Wisły Środkowej i Dolnej IGiPZ PAN. Available online: https://www.igipz.pan.pl/roslinnosc-dolina-wisly.html (accessed on 23 December 2020).
- Kowalska, A.; Affek, A.; Regulska, E.; Wolski, J.; Kruczkowska, B.; Kołaczkowska, E.; Zawiska, I.; Baranowski, J. Łęgi jesionowo-wiązowe w dolinie środkowej Wisły—stan ekosystemów pozbawionych zalewów i wytyczne do działań ochronnych [Riparian hardwood forests in the valley of the middle Vistula—ecosystem condition in the absence of flooding, and guidelines for protection]. Prz. Geogr. 2019, 91, 295–323. [Google Scholar] [CrossRef]
- Maes, J.; Teller, A.; Erhard, M.; Grizzetti, B.; Barredo, J.; Paracchini, M.; Condé, S.; Somma, F.; Orgiazzi, A.; Jones, A.; et al. Mapping and Assessment of Ecosystems and Their Services: An Analytical Framework for Ecosystem Condition; Publications Office of the European Union: Luxembourg, 2018. [Google Scholar]
- Winfree, R. The conservation and restoration of wild bees. Ann. N. Y. Acad. Sci. 2010, 1195, 169–197. [Google Scholar] [CrossRef]
- Greenleaf, S.S.; Williams, N.M.; Winfree, R.; Kremen, C. Bee foraging ranges and their relationship to body size. Oecologia 2007, 153, 589–596. [Google Scholar] [CrossRef]
- Taki, H.; Okochi, I.; Okabe, K.; Inoue, T.; Goto, H.; Matsumura, T.; Makino, S. Succession Influences Wild Bees in a Temperate Forest Landscape: The Value of Early Successional Stages in Naturally Regenerated and Planted Forests. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [Green Version]
- Kells, A.R.; Goulson, D. Preferred nesting sites of bumblebee queens (Hymenoptera: Apidae) in agroecosystems in the UK. Biol. Conserv. 2003. [Google Scholar] [CrossRef]
- Westrich, P. Habitat requirements of central European bees and the problems of partial habitats. In The Conservation of Bees; Matheson, A., Buchmann, S.L., O’Toole, C., Westrich, P., Williams, I.H., Eds.; Academic Press for the Linnean Society of London and IBRA: London, UK, 1996; pp. 1–16. [Google Scholar]
- Braun-Blanquet, J. Pflanzensoziologie. Grundzüge der Vegetationskunde; Springer: Wien, Austria, 1964. [Google Scholar]
- Szklanowska, K. Bory jako baza pożytkowa pszczół [Pine forests as a bee pasture]. Pszczel. Zesz. Nauk. 1973, 17, 51–85. [Google Scholar]
- Demianowicz, Z.; Hłyń, M.; Jabłoński, B.; Maksymiuk, I.; Podgórska, J.; Ruszkowska, B.; Szklanowska, K.; Zimna, J. Wydajność miodowa ważniejszych roślin miododajnych w warunkach Polski. Część I [Honey potential of the major honey-producing plants in Poland. Part I]. Pszczel. Zesz. Nauk. 1960, 4, 87–104. [Google Scholar]
- Szklanowska, K. Nektarowanie i wydajność miodowa ważniejszych roślin runa lasu liściastego [Nectar secretion and honey potential of some more important undergrowth plants in deciduous forest]. Pszczel. Zesz. Nauk. 1979, 23, 123–130. [Google Scholar]
- Maksymiuk, I. Nektarowanie lipy drobnolistnej Tilia Cordata Mill. w Rezerwcie Obrożyska koło Muszyny [The nectar secretion of linden Tilia Cordata Mill. at the Reserve Obrożyska near Muszyna (Carpathians)]. Pszczel. Zesz. Nauk. 1960, 4, 105–125. [Google Scholar]
- Kołtowski, Z. Wielki Atlas Roślin Miododajnych [The Great Atlas of Melliferous Plants]; Przedsiębiorstwo Wydawnicze Rzeczpospolita SA: Warszawa, Poland, 2006. [Google Scholar]
- Ruszkowski, A.; Zadura, M.; Biliński, M.; Gosek, J.; Kaczmarska, K. Wiosenny wzorzec składu gatunkowego trzmieli (Bombus Latr.) na różnych roślinach. Pszczel. Zesz. Nauk. 1997, 41, 43–51. [Google Scholar]
- Jabłoński, B. Notes on the method to investigate nectar secretion rate in flowers. J. Apic. Sci. 2002, 46, 117–125. [Google Scholar]
- Szklanowska, K. Nektarowanie i wydajność miodowa niektórych drzew i krzewów w warunkach Polski. Pszczel. Zesz. Nauk. 1978, 22, 117–128. [Google Scholar]
- Bumblebee Conservation Trust Help Us to Count the UK’s Bumblebees. Available online: https://www.bumblebeeconservation.org/surveys/ (accessed on 23 December 2020).
- Banaszak, J. Studies on methods of censusing the numbers of bees (Hymenoptera, Apoidea). Pol. Ecol. Stud. 1980, 6, 355–366. [Google Scholar]
- Droege, S.; Engler, J.D.; Sellers, E.A.; O’Brien, L.U.S. National Protocol Framework for the Inventory and Monitoring of Bees; Inventory and Monitoring, National Wildlife Refuge System, U.S. Fish and Wildlife Service: Fort Collins, CO, USA, 2016.
- Hampel, F.R.; Ronchetti, E.M.; Rousseeuw, P.J.; Stahel, W.A. Robust Statistics: The Approach Based on Influence Functions; Wiley: New York, NY, USA, 1986; ISBN 978-0471829218. [Google Scholar]
- Farazi, M.M.; Imon, A. Detection of Outliers in Gene Expression Data Using Expressed Robust-t Test. Malays. J. Math. Sci. 2016, 10, 117–135. [Google Scholar]
- Krishnan, S.; Wiederkehr Guerra, G.; Bertrand, D.; Wertz-Kanounnikoff, S.; Kettle, C.J. The Pollination Services of Forests: A Review of Forest and Landscape Interventions to Enhance Their Cross-Sectoral Benefits; FAO and Biodiversity International: Rome, Italy, 2020; ISBN 978-92-5-132813-2. [Google Scholar]
- Banaszak, J.; Jaroszewicz, B. Bees of the Białowieża National Park and adjacent areas, NE Poland (Hymenoptera: Apoidea, Apiformes). Pol. J. Entomol. 2009, 78, 281–313. [Google Scholar]
- Banaszak, J.; Szefer, P. Pszczoły (Hymenoptera: Apoidea) Równiny Sępopolskiej. Cz. I. Różnorodność gatunkowa. Wiad. Entomol. 2013, 32, 185–201. [Google Scholar]
- Droege, S.; Tepedino, V.J.; Lebuhn, G.; Link, W.; Minckley, R.L.; Chen, Q.; Conrad, C. Spatial patterns of bee captures in North American bowl trapping surveys. Insect Conserv. Divers. 2010. [Google Scholar] [CrossRef]
- Banaszak, J. Pszczoły (Apoidea) grądów i dąbrów świetlistych Niziny Mazowieckiej. Zesz. Nauk. Akad. Bydgoskiej Im. Kazimierza Wielkiego W Bydgoszczy. Studia Przyr. 1990, 8, 23–36. [Google Scholar]
- Sobieraj-Betlińska, A.; Banaszak, J. Zadrzewienia śródpolne jako ostoje pszczół [Mid-field woodlots as refuges for bees]. Wiad. Entomol. 2017, 36, 111–123. [Google Scholar]
- Banaszak, J.; Cierzniak, T. Ocena stopnia zagrożeń i możliwości ochrony owadów w agroekosystemach. Wiad. Entomol. 2000, 18, 73–94. [Google Scholar]
- Banaszak, J.; Krzysztofiak, A. Communities of bees in the forests of Poland. In Natural Resources of Wild Bees in Poland; Banaszak, J., Ed.; Pedagogical University: Bydgoszcz, Poland, 1992; pp. 33–40. [Google Scholar]
- Banaszak, J. Strategy for conservation of wild bees in an agricultural landscape. Agric. Ecosyst. Environ. 1992, 40, 179–192. [Google Scholar] [CrossRef]
- Cunningham-Minnick, M.J.; Crist, T.O. Floral resources of an invasive shrub alter native bee communities at different vertical strata in forest-edge habitat. Biol. Invasions 2020, 22, 2283–2298. [Google Scholar] [CrossRef]
- Smith, C.; Weinman, L.; Gibbs, J.; Winfree, R. Specialist foragers in forest bee communities are small, social or emerge early. J. Anim. Ecol. 2019, 88, 1158–1167. [Google Scholar] [CrossRef]
- Roulston, T.H.; Goodell, K. The role of resources and risks in regulating wild bee populations. Annu. Rev. Entomol. 2011, 56, 293–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winfree, R.; Kremen, C. Are ecosystem services stabilized by differences among species? A test using crop pollination. Proc. R. Soc. B Biol. Sci. 2009, 276, 229–237. [Google Scholar] [CrossRef] [Green Version]
- Torné-Noguera, A.; Rodrigo, A.; Arnan, X.; Osorio, S.; Barril-Graells, H.; Da Rocha-Filho, L.C.; Bosch, J. Determinants of spatial distribution in a bee community: Nesting resources, flower resources, and body size. PLoS ONE 2014, 9, e97255. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez, A.; Kouki, J. Emulating natural disturbance in forest management enhances pollination services for dominant Vaccinium shrubs in boreal pine-dominated forests. For. Ecol. Manag. 2015, 350, 1–12. [Google Scholar] [CrossRef]
- Bystriakova, N.; Griswold, T.; Ascher, J.S.; Kuhlmann, M. Key environmental determinants of global and regional richness and endemism patterns for a wild bee subfamily. Biodivers. Conserv. 2018, 27, 287–309. [Google Scholar] [CrossRef]
- Carré, G.; Roche, P.; Chifflet, R.; Morison, N.; Bommarco, R.; Harrison-Cripps, J.; Krewenka, K.; Potts, S.G.; Roberts, S.P.M.; Rodet, G.; et al. Landscape context and habitat type as drivers of bee diversity in European annual crops. Agric. Ecosyst. Environ. 2009, 133, 40–47. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Steffan-Dewenter, I.; Kremen, C.; Morales, J.M.; Bommarco, R.; Cunningham, S.A.; Carvalheiro, L.G.; Chacoff, N.P.; Dudenhöffer, J.H.; Greenleaf, S.S.; et al. Stability of pollination services decreases with isolation from natural areas despite honey bee visits. Ecol. Lett. 2011, 14, 1062–1072. [Google Scholar] [CrossRef]
- Potts, S.G.; Vulliamy, B.; Roberts, S.; O’Toole, C.; Dafni, A.; Ne’eman, G.; Willmer, P. Role of nesting resources in organising diverse bee communities in a Mediterranean landscape. Ecol. Entomol. 2005, 30, 78–85. [Google Scholar] [CrossRef]
- Ricou, C.; Schneller, C.; Amiaud, B.; Plantureux, S.; Bockstaller, C. A vegetation-based indicator to assess the pollination value of field margin flora. Ecol. Indic. 2014, 45, 320–331. [Google Scholar] [CrossRef]
- Bartholomée, O.; Aullo, A.; Becquet, J.; Vannier, C.; Lavorel, S. Pollinator presence in orchards depends on landscape-scale habitats more than in-field flower resources. Agric. Ecosyst. Environ. 2020, 293, 106806. [Google Scholar] [CrossRef]
- Potts, S.G.; Vulliamy, B.; Dafni, A.; Ne’eman, G.; Willmer, P. Linking bees and flowers: How do floral communities structure pollinator communities? Ecology 2003, 84, 2628–2642. [Google Scholar] [CrossRef] [Green Version]
- Liss, K.N.; Mitchell, M.G.E.; Macdonald, G.K.; Mahajan, S.L.; Méthot, J.; Jacob, A.L.; Maguire, D.Y.; Metson, G.S.; Ziter, C.; Dancose, K.; et al. Variability in ecosystem service measurement: A pollination service case study. Front. Ecol. Environ. 2013, 11, 414–422. [Google Scholar] [CrossRef] [Green Version]
- Lonsdorf, E.; Kremen, C.; Ricketts, T.; Winfree, R.; Williams, N.; Greenleaf, S. Modelling pollination services across agricultural landscapes. Ann. Bot. 2009, 103, 1589–1600. [Google Scholar] [CrossRef] [Green Version]
- Zulian, G.; Maes, J.; Paracchini, M. Linking Land Cover Data and Crop Yields for Mapping and Assessment of Pollination Services in Europe. Land 2013, 2, 472–492. [Google Scholar] [CrossRef] [Green Version]
- Schulp, C.J.E.; Lautenbach, S.; Verburg, P.H. Quantifying and mapping ecosystem services: Demand and supply of pollination in the European Union. Ecol. Indic. 2014, 36, 131–141. [Google Scholar] [CrossRef]
- Polce, C.; Termansen, M.; Aguirre-Gutiérrez, J.; Boatman, N.D.; Budge, G.E.; Crowe, A.; Garratt, M.P.; Pietravalle, S.; Potts, S.G.; Ramirez, J.A.; et al. Species Distribution Models for Crop Pollination: A Modelling Framework Applied to Great Britain. PLoS ONE 2013, 8, e76308. [Google Scholar] [CrossRef]
- Perennes, M.; Diekötter, T.; Groß, J.; Burkhard, B. A hierarchical framework for mapping pollination ecosystem service potential at the local scale. Ecol. Model. 2021, 444, 109484. [Google Scholar] [CrossRef]
- Everaars, J.; Settele, J.; Dormann, C.F. Fragmentation of nest and foraging habitat affects time budgets of solitary bees, their fitness and pollination services, depending on traits: Results from an individual-based model. PLoS ONE 2018, 13, e0188269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Häussler, J.; Sahlin, U.; Baey, C.; Smith, H.G.; Clough, Y. Pollinator population size and pollination ecosystem service responses to enhancing floral and nesting resources. Ecol. Evol. 2017, 7, 1898–1908. [Google Scholar] [CrossRef]
- Kremen, C.; Williams, N.M.; Aizen, M.A.; Gemmill-Herren, B.; LeBuhn, G.; Minckley, R.; Packer, L.; Potts, S.G.; Roulston, T.; Steffan-Dewenter, I.; et al. Pollination and other ecosystem services produced by mobile organisms: A conceptual framework for the effects of land-use change. Ecol. Lett. 2007, 10, 299–314. [Google Scholar] [CrossRef]
- Ricketts, T.H.; Williams, N.M.; Mayfield, M.M. Connectivity and ecosystem services: Crop pollination in agricultural landscapes. In Connectivity Conservation; Crooks, K., Sanjayan, M., Eds.; Cambridge University Press: Cambridge, UK, 2006; pp. 255–290. [Google Scholar]
- Steffan-Dewenter, I.; Münzenberg, U.; Bürger, C.; Thies, C.; Tscharntke, T. Scale-dependent effects of landscape context on three pollinator guilds. Ecology 2002, 83, 1421–1432. [Google Scholar] [CrossRef]
- Westphal, C.; Steffan-Dewenter, I.; Tscharntke, T. Bumblebees experience landscapes at different spatial scales: Possible implications for coexistence. Oecologia 2006, 149, 289–300. [Google Scholar] [CrossRef]
- Gathmann, A.; Tscharntke, T. Foraging ranges of solitary bees. J. Anim. Ecol. 2002, 71, 757–764. [Google Scholar] [CrossRef]
- Williams, N.M.; Tepedino, V.J. Consistent mixing of near and distant resources in foraging bouts by the solitary mason bee Osmia lignaria. Behav. Ecol. 2003. [Google Scholar] [CrossRef] [Green Version]
- Winfree, R.; Griswold, T.; Kremen, C. Effect of human disturbance on bee communities in a forested ecosystem. Conserv. Biol. 2007, 21, 213–223. [Google Scholar] [CrossRef]
- Roberts, H.P.; King, D.I.; Milam, J. Factors affecting bee communities in forest openings and adjacent mature forest. For. Ecol. Manag. 2017, 394, 111–122. [Google Scholar] [CrossRef] [Green Version]
- Carvell, C.; Meek, W.R.; Pywell, R.F.; Goulson, D.; Nowakowski, M. Comparing the efficacy of agri-environment schemes to enhance bumble bee abundance and diversity on arable field margins. J. Appl. Ecol. 2007, 44, 29–40. [Google Scholar] [CrossRef]
- Broussard, M.; Rao, S.; Stephen, W.P.; White, L. Native bees, Honeybees, and pollination in Oregon cranberries. HortScience 2011, 46, 885–888. [Google Scholar] [CrossRef] [Green Version]








| No | Indicator | Measure | Unit | Reference | N |
|---|---|---|---|---|---|
| 1 | Soil dryness | 100-gravimetric water content in topsoil | % | soil sample | 22 |
| 2 | Presence of large trees | DBH of the largest tree | cm | relevé | 18 |
| 3 | Habitat suitability for bumblebees | Occupancy degree of bumblebee nest traps | % | trap | 12 |
| 4 | Habitat suitability for solitary bees | Occupancy degree of solitary bee nest traps | % | trap | 12 |
| 5 | Food base | Honey potential of the ecosystem | kg ha−1 year−1 | relevé | 51 |
| 6 | Abundance of bumblebees | Bumblebee density from the route method | individuals ha−1 | observation | 51 |
| 7 | Abundance of bees (Apoidea) | Number of captured Apoidea per day into the pan trap | individuals day−1 | sample | 126 |
| Dimension | Pollination Indicator | Min | Mean | Median | AdjMax | Max | |
|---|---|---|---|---|---|---|---|
| Habitat potential | 1 | Soil dryness [%] | 47.1 | 75.6 | 78.3 | 90.4 | 90.4 |
| 2 | Presence of large trees [max DBH in cm] | 36.5 | 66.2 | 69.0 | 110.0 | 110.0 | |
| 3 | Habitat suitability for bumblebees | 0 | 0 | 0 | 0 | 0 | |
| 4 | Habitat suitability for solitary bees | 0 | 0 | 0 | 0 | 0 | |
| 5 | Nectar food base [kg ha−1] | 7.5 | 112.8 | 57.4 | 200.2 | 575.1 | |
| Pollinators potential | 6 | Abundance of bumblebees [individuals ha−1] | 0 | 163 | 100 | 500 | 850 |
| 7 | Abundance of bees (Apoidea) [individuals day−1 trap−1] | 0 | 0.14 | 0.08 | 0.42 | 1.60 |
| No. | Melliferous Plant | Honey Potential [kg ha−1] | Cover in Tree Layer [%] | Cover in Shrub Layer [%] | Cover in Herb Layer [%] | Honey Potential Weighted by Overall Cover [kg ha−1] |
|---|---|---|---|---|---|---|
| 1 | Solidago gigantea Aiton 1 | 700 | 0.00 | 0.00 | 8.05 | 56.33 |
| 2 | Scrophularia nodosa L. | 700 | 0.00 | 0.00 | 0.88 | 6.18 |
| 3 | Angelica sylvestris L. | 200 | 0.00 | 0.00 | 0.01 | 0.01 |
| 4 | Tilia cordataMill. | 200 | 1.03 | 0.29 | 0.20 | 2.65 |
| 5 | Stachys sylvatica L.* | 140 | 0.00 | 0.00 | 1.47 | 2.06 |
| 6 | Lamium maculatum L. | 140 | 0.00 | 0.00 | 8.92 | 12.49 |
| 7 | Ajuga reptans L. | 120 | 0.00 | 0.00 | 0.20 | 0.24 |
| 8 | Rubus idaeus L. | 120 | 0.00 | 6.72 | 1.91 | 10.35 |
| 9 | Acer platanoides L. | 100 | 6.86 | 0.49 | 2.80 | 7.36 |
| 10 | Frangula alnusMill. | 80 | 0.00 | 0.39 | 0.00 | 0.31 |
| 11 | Ribes nigrum L. | 60 | 0.00 | 0.74 | 0.00 | 0.44 |
| 12 | Acer pseudoplatanus L. | 50 | 0.29 | 0.10 | 0.20 | 0.20 |
| 13 | Acer campestre L. | 50 | 0.10 | 0.00 | 0.10 | 0.05 |
| 14 | Pulmonaria obscuraDumort. | 35 | 0.00 | 0.00 | 3.19 | 1.12 |
| 15 | Acer negundo L. *,2 | 30 | 0.44 | 0.30 | 0.00 | 0.22 |
| 16 | Prunus cerasiferaEhrh. | 30 | 0.00 | 0.10 | 0.00 | 0.03 |
| 17 | Salix alba L. | 30 | 0.20 | 0.10 | 0.00 | 0.09 |
| 18 | Galeobdolon luteumHuds. | 20 | 0.00 | 0.00 | 0.44 | 0.09 |
| 19 | Sorbus aucupariaL. Em.Hedl. | 20 | 0.54 | 0.59 | 0.39 | 0.23 |
| 20 | Rubus caesius L. | 20 | 0.00 | 0.34 | 12.01 | 2.47 |
| 21 | Taraxacum officinaleF. H. Wigg. 2 | 20 | 0.00 | 0.00 | 0.01 | 0.00 |
| 22 | Malus sylvestris L. | 15 | 0.10 | 0.00 | 0.00 | 0.01 |
| 23 | Crataegus monogynaJacq. | 15 | 1.62 | 0.93 | 0.39 | 0.38 |
| 24 | Prunus padus L. | 12 | 21.27 | 14.26 | 4.61 | 4.26 |
| 25 | Glechoma hederacea L. * | 10 | 0.00 | 0.00 | 32.70 | 3.27 |
| 26 | Viola reichenbachianaBoreau | 10 | 0.00 | 0.00 | 0.74 | 0.07 |
| 27 | Chelidonium majus L. * | 10 | 0.00 | 0.00 | 4.36 | 0.44 |
| 28 | Polygonatum multiflorum(L.) All. * | 10 | 0.00 | 0.00 | 0.39 | 0.04 |
| 29 | Ribes spicatumE.Robson * | 10 | 0.00 | 5.79 | 0.10 | 0.59 |
| 30 | Galeopsis pubescensBesser *,2 | 10 | 0.00 | 0.00 | 1.08 | 0.11 |
| 31 | Galeopsis speciosaMill. * | 10 | 0.00 | 0.00 | 1.13 | 0.11 |
| 32 | Oxalis acetosella L. * | 10 | 0.00 | 0.00 | 5.93 | 0.59 |
| Abundance of Bees (Apoidea) | Food Base | Abundance of Bumblebees | Soil Dryness | Presence of Large Trees | |
|---|---|---|---|---|---|
| Abundance of bees (Apoidea) | 0.26 | −0.64 | 0.26 | −0.20 | |
| Food base | 0.62 | −0.55 | 0.31 | 0.55 | |
| Abundance of bumblebees | 0.17 | 0.26 | 0.23 | −0.02 | |
| Soil dryness | 0.62 | 0.54 | 0.66 | 0.23 | |
| Presence of large trees | 0.70 | 0.26 | 0.98 | 0.66 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Affek, A.N.; Regulska, E.; Kołaczkowska, E.; Kowalska, A.; Affek, K. Pollination Potential of Riparian Hardwood Forests—A Multifaceted Field-Based Assessment in the Vistula Valley, Poland. Forests 2021, 12, 907. https://doi.org/10.3390/f12070907
Affek AN, Regulska E, Kołaczkowska E, Kowalska A, Affek K. Pollination Potential of Riparian Hardwood Forests—A Multifaceted Field-Based Assessment in the Vistula Valley, Poland. Forests. 2021; 12(7):907. https://doi.org/10.3390/f12070907
Chicago/Turabian StyleAffek, Andrzej N., Edyta Regulska, Ewa Kołaczkowska, Anna Kowalska, and Katarzyna Affek. 2021. "Pollination Potential of Riparian Hardwood Forests—A Multifaceted Field-Based Assessment in the Vistula Valley, Poland" Forests 12, no. 7: 907. https://doi.org/10.3390/f12070907
APA StyleAffek, A. N., Regulska, E., Kołaczkowska, E., Kowalska, A., & Affek, K. (2021). Pollination Potential of Riparian Hardwood Forests—A Multifaceted Field-Based Assessment in the Vistula Valley, Poland. Forests, 12(7), 907. https://doi.org/10.3390/f12070907






