Continent-Wide Tree Species Distribution Models May Mislead Regional Management Decisions: A Case Study in the Transboundary Biosphere Reserve Mura-Drava-Danube
Abstract
1. Introduction
2. Materials and Methods
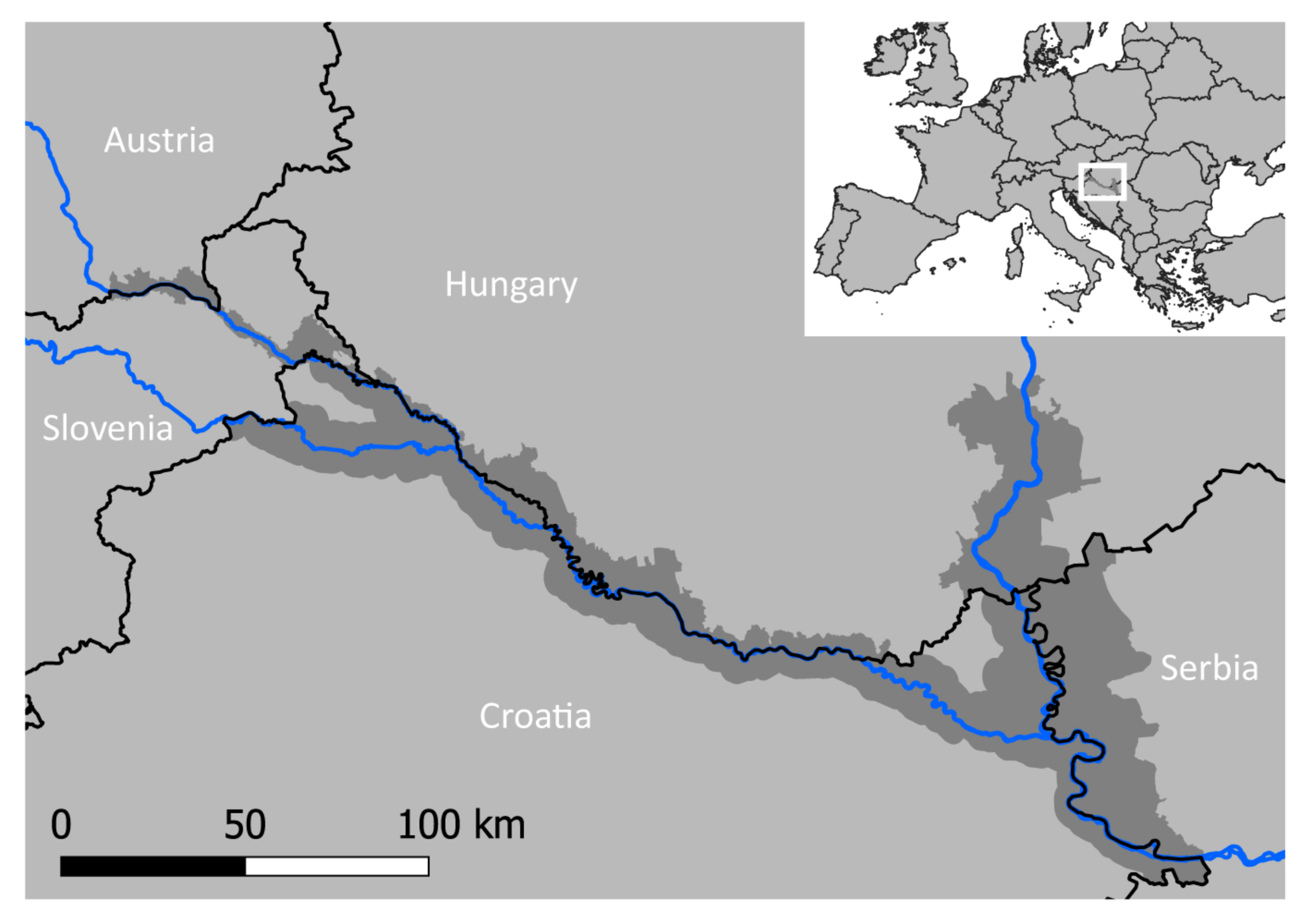
2.1. Study Area
2.2. Tree Species
2.3. Species Occurrence Data
2.4. Environmental Data
2.5. Data Analysis
2.5.1. Variable Selection
2.5.2. Species Distribution Models
3. Results
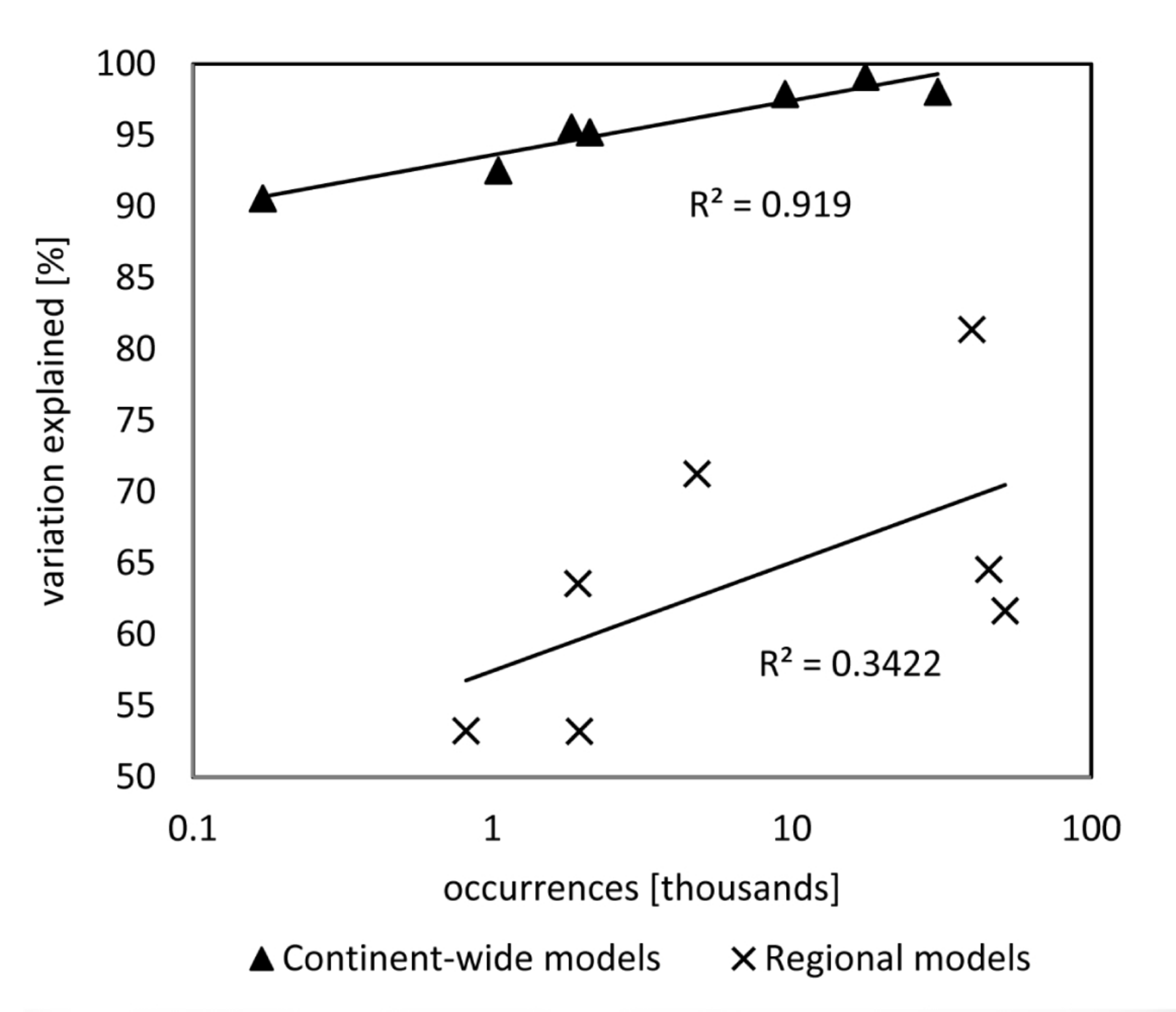
3.1. Model Training Data, Model Performance, and Accuracy
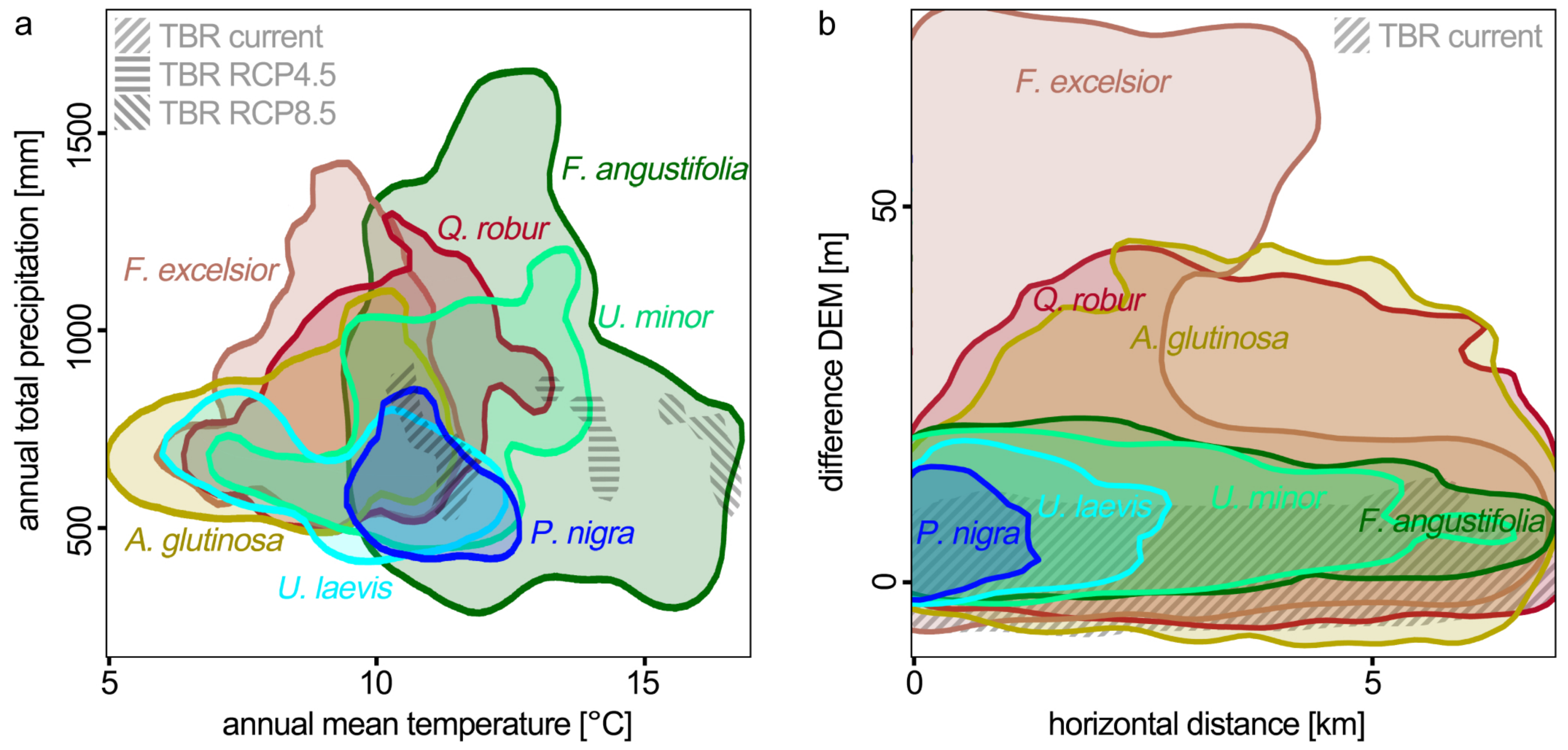
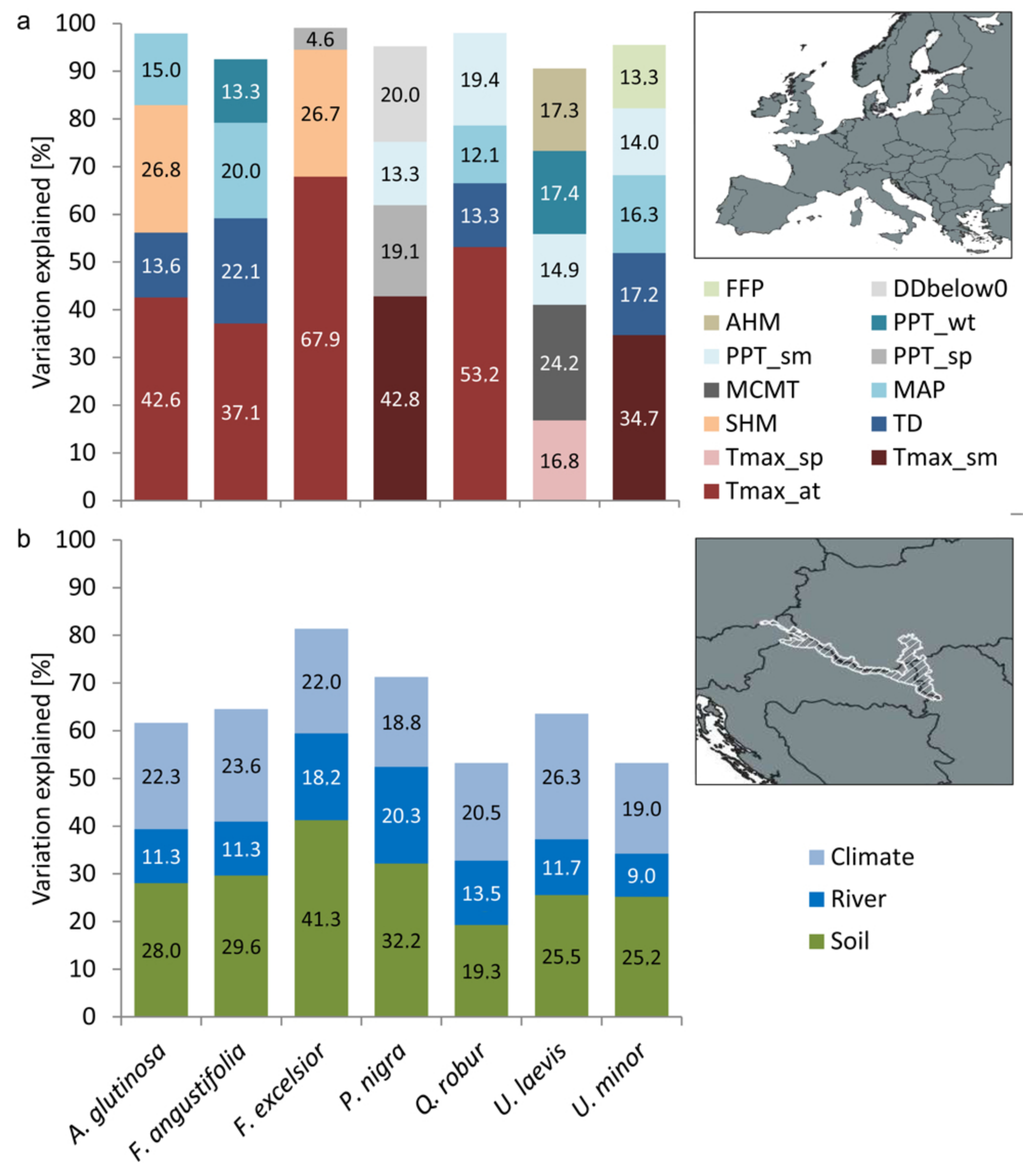
3.2. Drivers for Tree Species Distribution
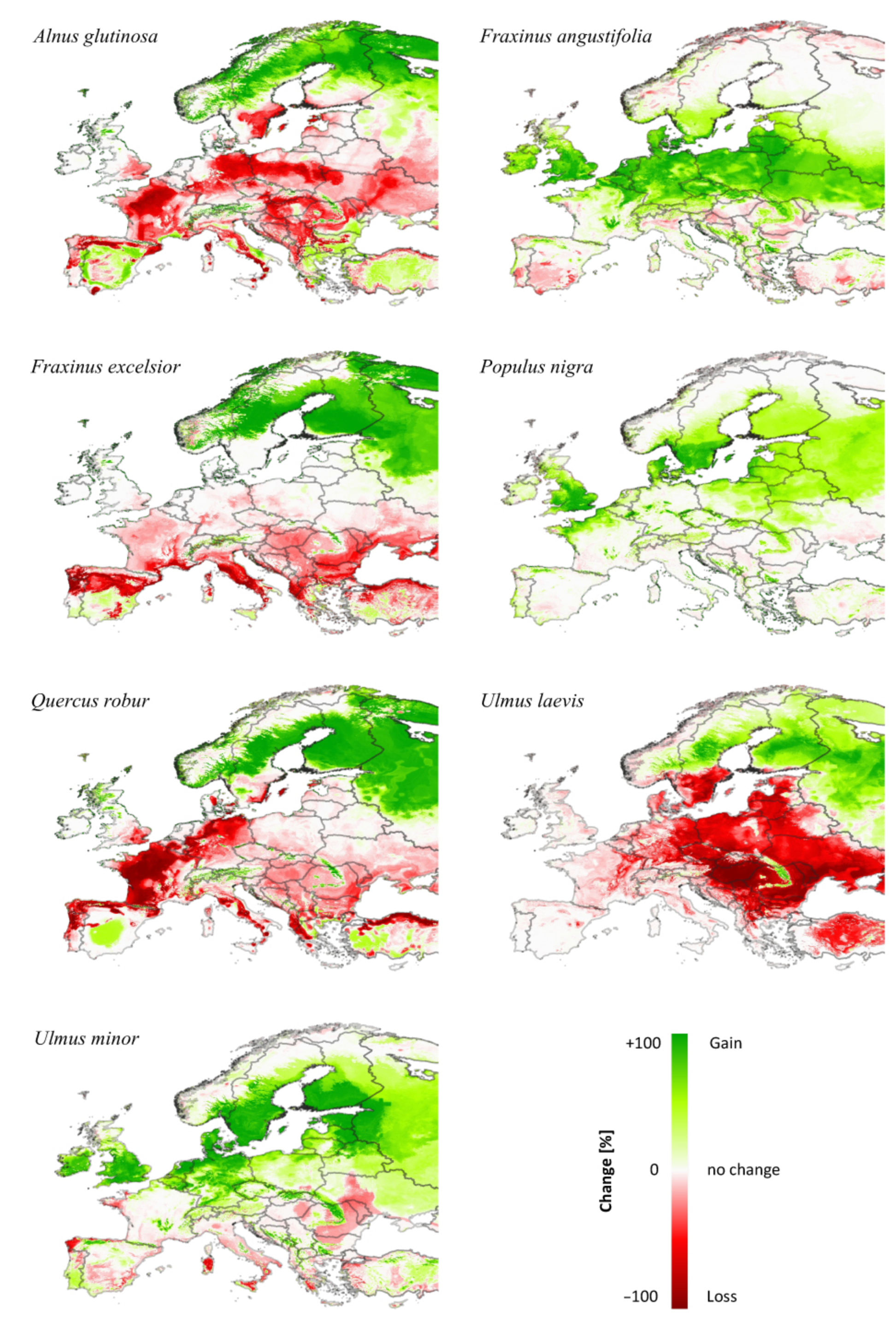
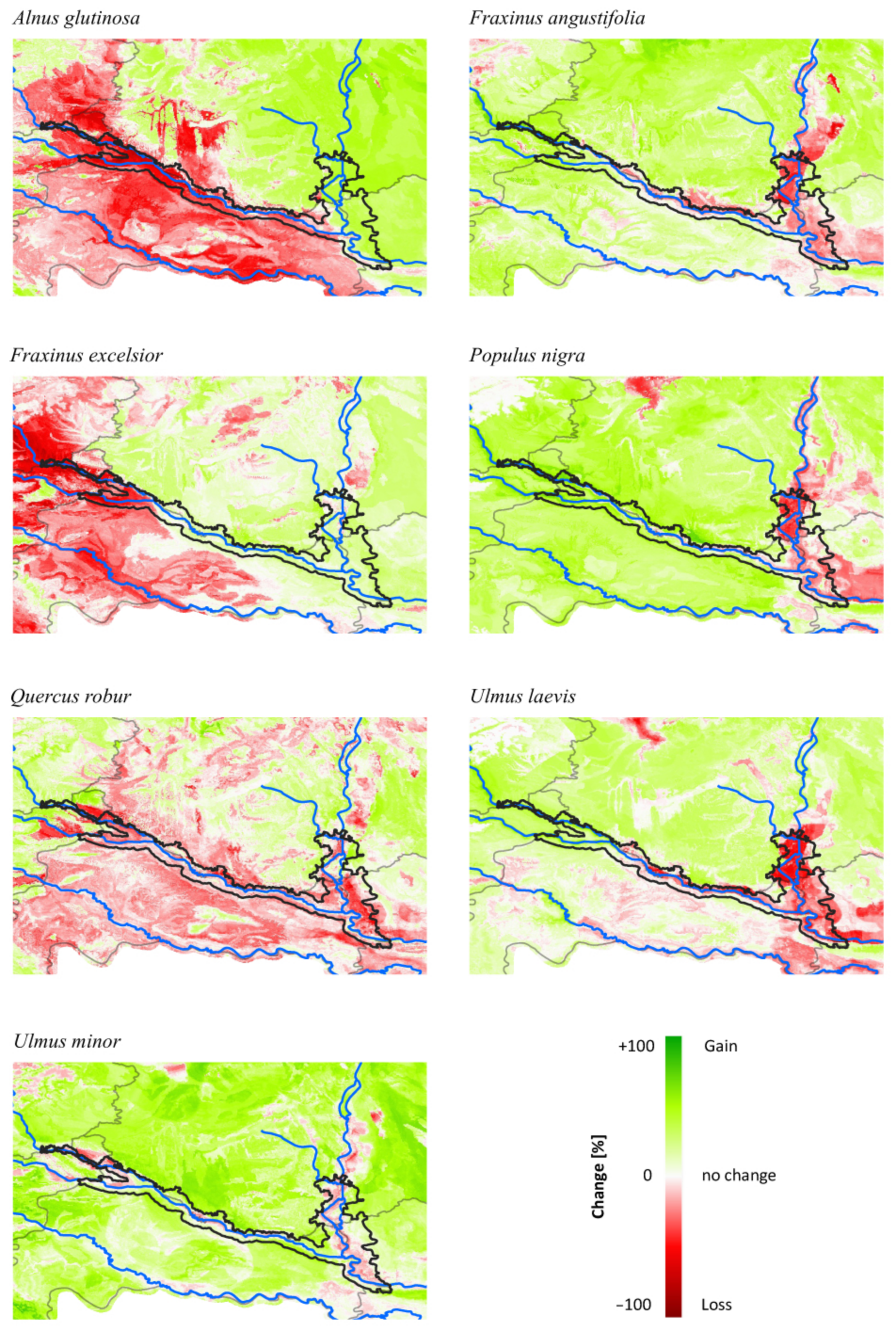
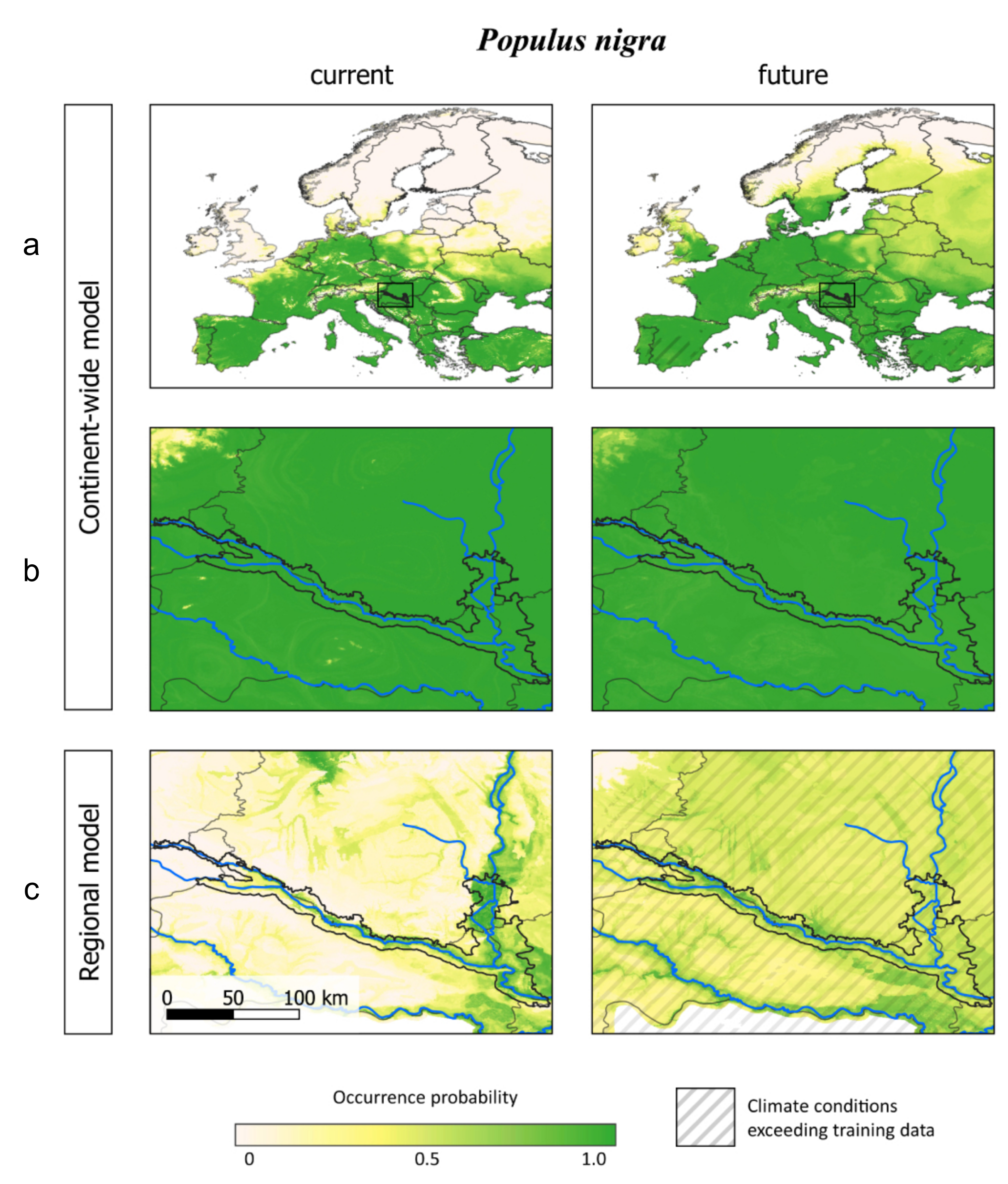
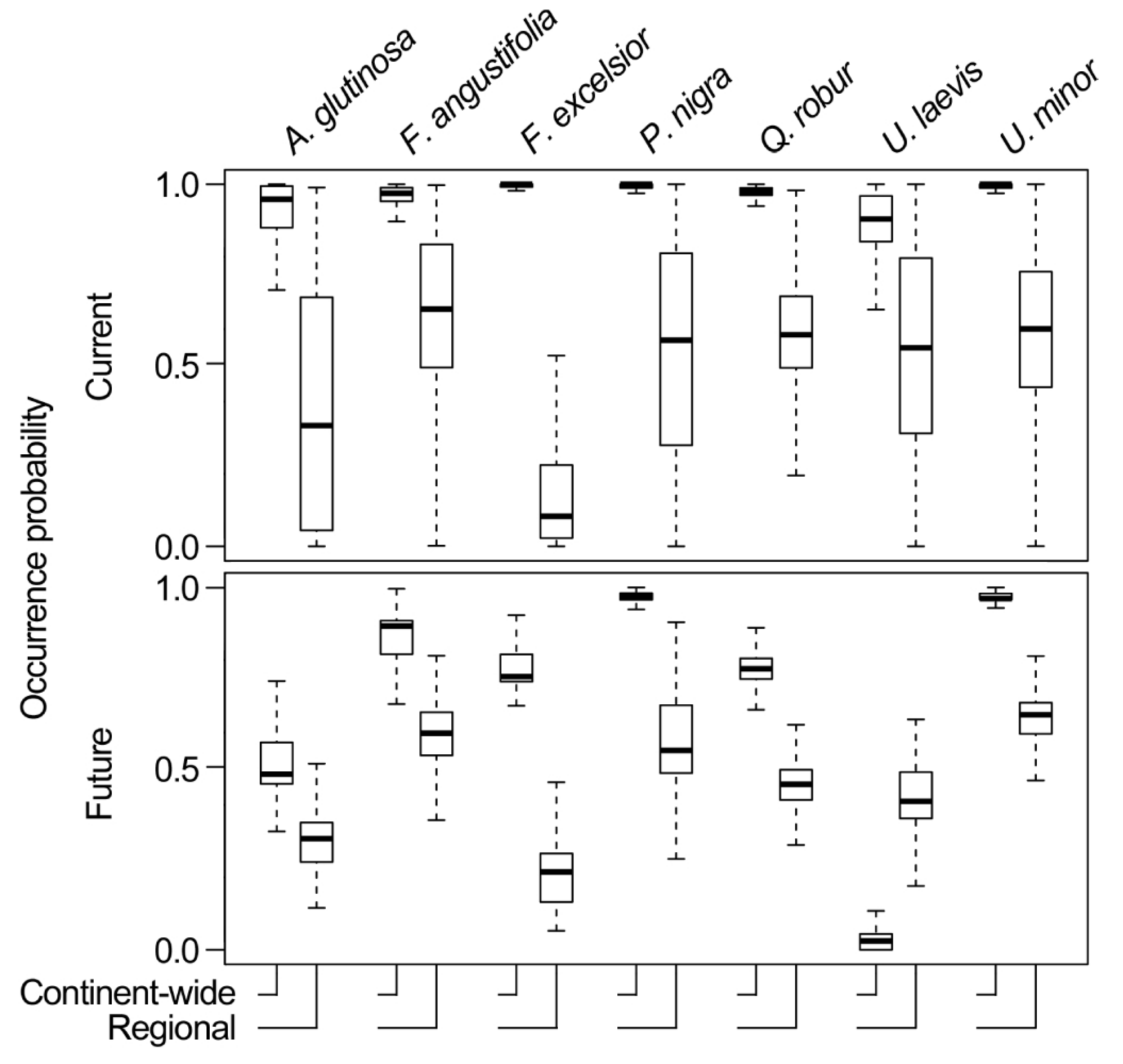
3.3. Comparing Model Type Predictions
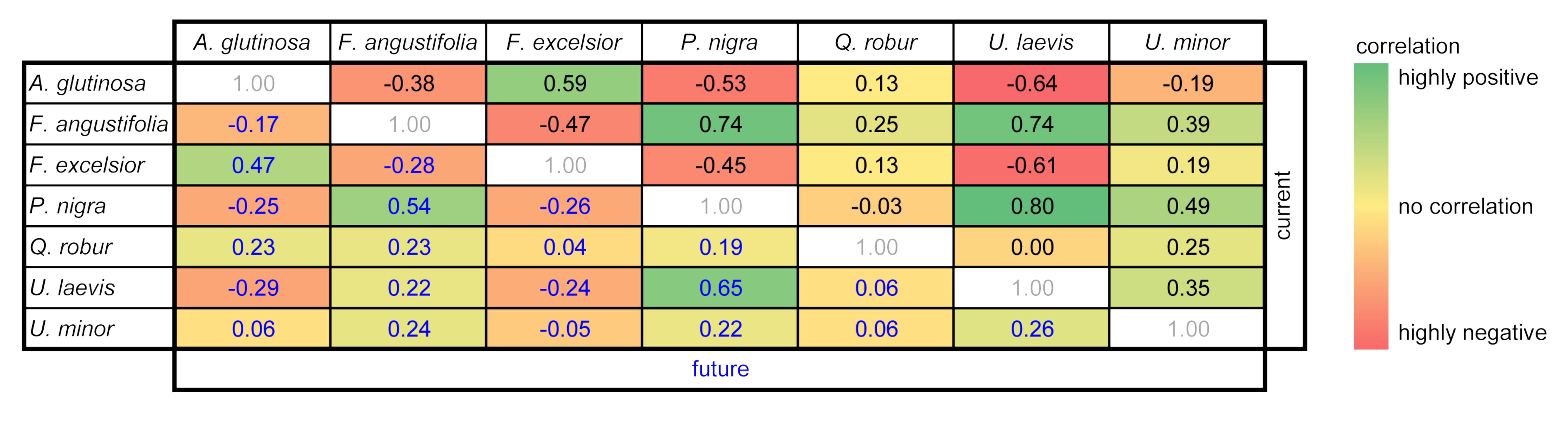
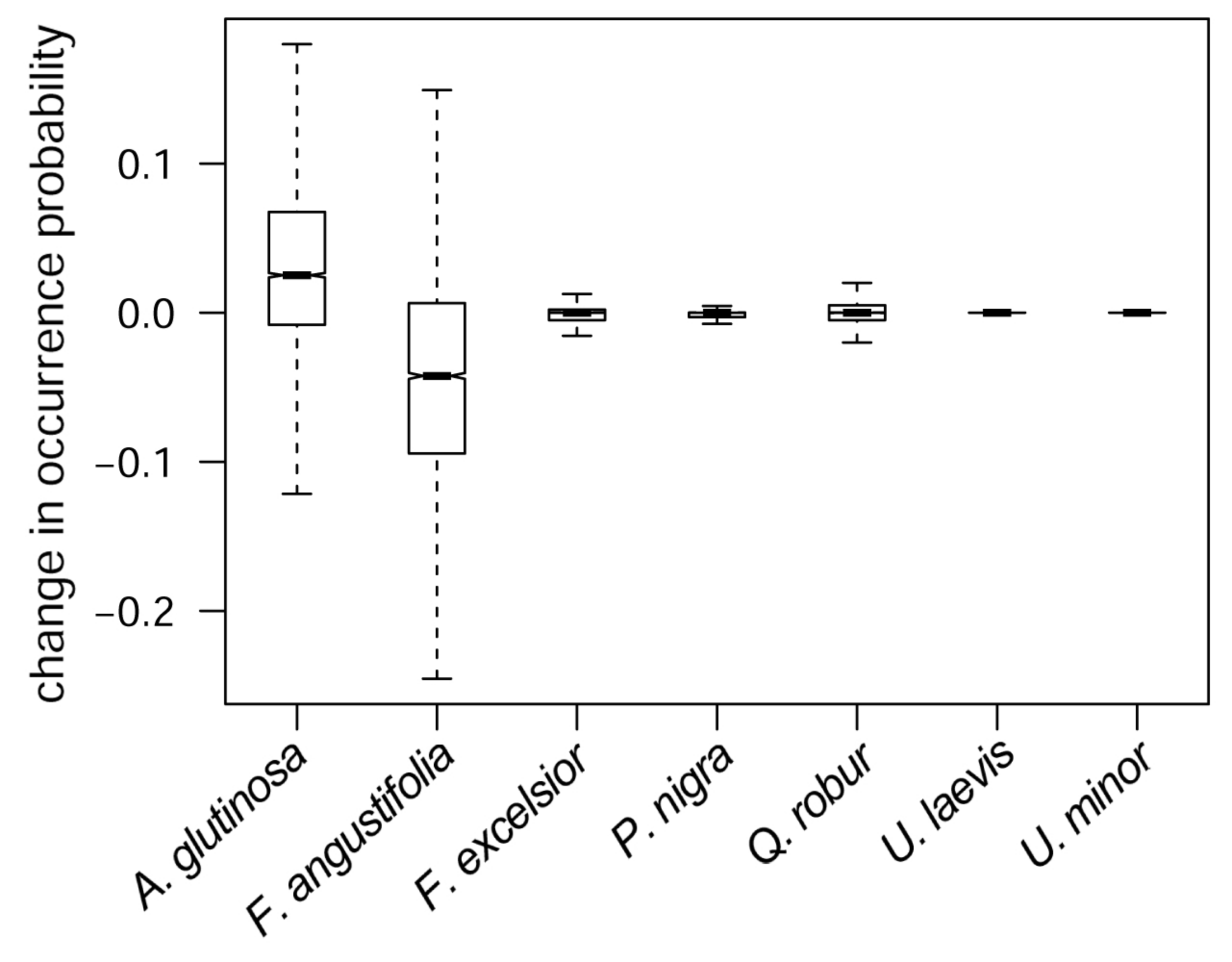
3.4. Predicted Co-Occurrence of Species
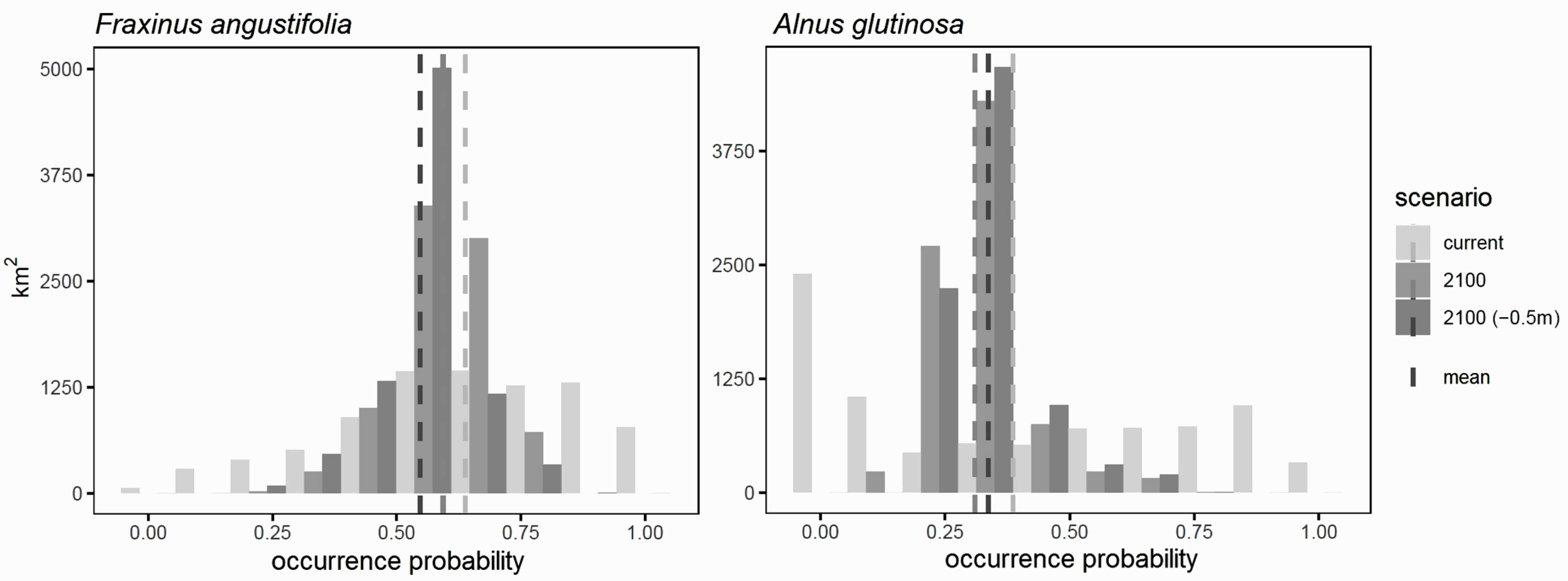
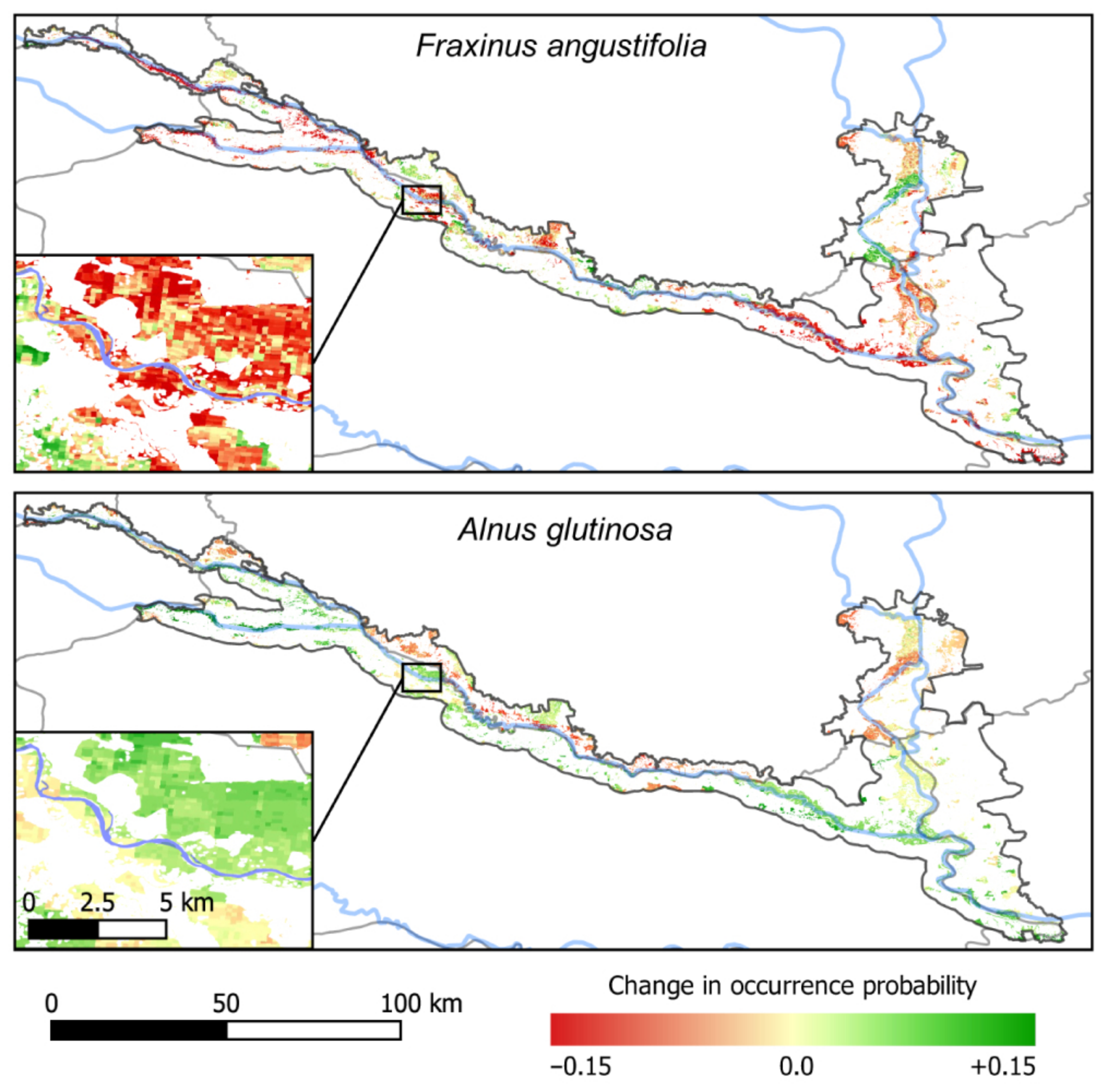
3.5. Consequences of River Deepening for Habitat Availability
4. Discussion
4.1. Consequences for Managing Riparian Forests of the TBR
4.2. Comparison of the Model Results and Predictor Importance with Other Studies
4.3. Data and Model Constraints
4.4. Practical Application of the Models
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Model Type | Alnus glutinosa | Fraxinus angustifolia | Fraxinus excelsior | Populus nigra | Quercus robur | Ulmus laevis | Ulmus minor |
|---|---|---|---|---|---|---|---|
| continent-wide | 9488 | 1046 | 17,596 | 2115 | 30,700 | 171 | 1836 |
| regional | 39,918 | 4821 | 45,445 | 1929 | 51,539 | 816 | 1952 |
| Continent-wide and regional model | AHM | Annual heat:moisture index |
| bFFP | Beginning of FFP | |
| DDabove18 | Degree-days above 18 °C | |
| DDabove5 | Degree-days above 5 °C | |
| DDbelow0 | Degree-days below 0 °C | |
| DDbelow18 | Degree-days below 18 °C | |
| eFFP | Ends of FFP | |
| EMT | Extreme minimum temperature | |
| FFP | Longest frost-free period | |
| MAP | Annual total precipitation | |
| MAT | Annual mean temperature | |
| MCMT | Mean coldest month temperature | |
| MWMT | Mean warmest month temperature | |
| NFFD | Number of frost-free days | |
| PPT_at | Mean autumn precipitation | |
| PPT_sm | Mean summer precipitation | |
| PPT_sp | Mean spring precipitation | |
| PPT_wt | Mean winter precipitation | |
| SHM | Summer heat:moisture index | |
| Tave_at | Mean autumn temperature | |
| Tave_sm | Mean summer temperature | |
| Tave_sp | Mean spring temperature | |
| Tave_wt | Mean winter temperature | |
| TD | Continentality | |
| Tmax_at | Maximum autumn temperature | |
| Tmax_sm | Maximum summer temperature | |
| Tmax_sp | Maximum spring temperature | |
| Tmax_wt | Maximum winter temperature | |
| Tmin_at | Minimum autumn temperature | |
| Tmin_sm | Minimum summer temperature | |
| Tmin_sp | Minimum spring temperature | |
| Tmin_wt | Minimum winter temperature | |
| Regional model | min_direct_dist | Horizontal distance to the next water body (major rivers and inland water bodies) |
| diff_dem | Elevation difference between site and next river. Calculated from a 25 × 25 m Digital Elevation Model | |
| awc_sub | Subsoil available water capacity | |
| awc_top | Topsoil available water capacity | |
| bs_sub | Base saturation of the subsoil | |
| bs_top | Base saturation of the topsoil | |
| cec_sub | Subsoil cation exchange capacity | |
| cec_top | Topsoil cation exchange capacity | |
| dgh | Depth to a gleyed horizon | |
| dimp | Depth to an impermeable layer | |
| dr | Depth to rock | |
| eawc_sub | Subsoil easily available water capacity | |
| eawc_top | Topsoil easily available water capacity | |
| hg | Hydrogeological class | |
| oc_top | Topsoil organic carbon content | |
| pd_sub | Subsoil packing density | |
| pd_top | Topsoil packing density | |
| peat | Peat | |
| pmh | Parent material hydrogeological type | |
| str_sub | Subsoil structure | |
| str_top | Topsoil structure | |
| text | Dominant surface textural class (completed from dominant STU) |
| Species | Occurrences | Variation Explained (%) | Explanatory Variables: InMSE | ||||
|---|---|---|---|---|---|---|---|
| Alnus glutinosa | 9488 | 97.89 | Tmax_at: 76.9 | SHM: 48.4 | MAP: 27.1 | TD: 24.1 | - |
| Fraxinus angustifolia | 1046 | 92.53 | Tmax_at: 42.5 | TD: 25.3 | MAP: 23 | PPT_wt: 15.2 | - |
| Fraxinus excelsior | 17,596 | 99.10 | Tmax_at: 144.5 | SHM: 56.8 | PPT_sp: 9.8 | - | - |
| Populus nigra | 2115 | 95.21 | Tmax_sm: 44.9 | DDbelow0: 21 | PPT_sp: 20.1 | PPT_sm: 14.0 | - |
| Quercus robur | 30,700 | 98.04 | Tmax_at: 122.1 | PPT_sm: 44.6 | TD: 30.6 | MAP: 27.9 | - |
| Ulmus laevis | 171 | 90.57 | MCMT: 17.7 | PPT_wt: 12.7 | AHM: 12.7 | Tmax_sp: 12.3 | PPT_sm: 10.9 |
| Ulmus minor | 1836 | 95.54 | Tmax_sm: 41.5 | TD: 20.6 | MAP: 19.5 | PPT_sm: 16.7 | FFP: 16.0 |
| Species | Occurrences | Variation Explained (%) | Explanatory Variables: InMSE |
|---|---|---|---|
| Alnus glutinosa | 39,918 | 61.66 | diff_dem: 78; AHM_199110_1km: 61.8; DDbelow0_199110_1km: 49.4; DDabove5_199110_1km: 42.2; dr: 18.2; hg: 17.7; pmh: 17.4; awc_sub: 15.5; cec_sub: 14; bs_top: 13.5; awc_top: 12.1; oc_top: 11.5; pd_top: 10.6; bs_sub: 10.5; str_top: 10.5; text: 10.1; cec_top: 9.3; pd_sub: 8.9; dgh: 6.9; peat: 6.2; |
| Fraxinus angustifolia | 4821 | 64.56 | DDbelow0_199110_1km: 39.5; AHM_199110_1km: 37.6; min_direct_dist: 25.8; DDabove5_199110_1km: 25.6; diff_dem: 23.4; hg: 15.9; awc_sub: 11.6; cec_sub: 10.5; cec_top: 9.8; awc_top: 9.7; text: 9.7; dr: 9.4; dgh: 8.7; str_top: 8.3; bs_top: 7.4; pd_sub: 7.2; pd_top: 7.1; dimp: 5.3; str_top: 4.2; peat: 4.1; |
| Fraxinus excelsior | 45,445 | 81.38 | AHM_199110_1km: 57.0; min_direct_dist: 50.1; DDbelow18_199110_1km: 42.8; diff_dem: 32.5; awc_sub: 16.9; cec_sub: 16.4; cec_top: 16; hg: 13.4; text: 12.2; pd_sub: 11.9; pmh: 11.4; dr: 11.2; bs_top: 11.2; awc_top: 10.9; bs_sub: 10.9; dgh: 10; oc_top: 8.9; str_top: 8.7; pd_top: 7; str_top: 6.7; peat: 3.7; |
| Populus nigra | 1929 | 71.26 | min_direct_dist: 50.5; diff_dem: 26.5; DDabove18_199110_1km: 25.2; DDbelow0_199110_1km: 24.7; AHM_199110_1km: 21.8; pmh: 10.9; hg: 10.4; cec_top: 10.4; text: 9.7; str_top: 8.9; pd_sub: 7.6; bs_sub: 7.1; awc_top: 7; dr: 6.8; bs_top: 6.8; pd_top: 6.7; cec_sub: 6.6; oc_top: 5.9; dgh: 5.7; dimp: 5.6; str_top: 3.4; peat: 3.1; |
| Quercus robur | 51,539 | 53.21 | DDabove5_199110_1km: 89.2; EMT_199110_1km: 88.9; min_direct_dist: 87.1; diff_dem: 82.4; AHM_199110_1km: 79.5; awc_sub: 20.5; dr: 19.4; str_top: 17.3; cec_sub: 17.2; text: 17; hg: 16.2; bs_top: 15.9; awc_top: 15.6; bs_sub: 15.5; pmh: 15.4; oc_top: 15.2; cec_top: 13.3; dgh: 12.3; pd_top: 11.3; str_top: 9; pd_sub: 6.8; peat: 4.4; |
| Ulmus laevis | 816 | 63.57 | min_direct_dist: 20.7; DDbelow0_199110_1km: 20.2; DDabove18_199110_1km: 19.4; AHM_199110_1km: 13; FFP_199110_1km: 10.2; diff_dem: 7.3; hg: 5.8; text: 5.4; dr: 5.2; awc_sub: 4.7; oc_top: 4.5; awc_top: 4.4; cec_sub: 3.8; pmh: 3.4; cec_top: 3.4; bs_top: 3.1; str_top: 3.1; dimp: 2.9; pd_top: 2.8; dgh: 2.8; pd_sub: 2.2; bs_sub: 2.1; str_top: 1.2; |
| Ulmus minor | 1952 | 53.25 | MAP_199110_1km: 38.5; DDabove5_199110_1km: 29.8; DDbelow0_199110_1km: 26.9; diff_dem: 22.6; min_direct_dist: 22.4; hg: 10.7; dr: 10.7; pmh: 10; awc_sub: 9.7; bs_top: 8.9; bs_sub: 8.7; str_top: 8.5; text: 8.2; oc_top: 7.9; pd_top: 7.3; cec_top: 6.9; cec_sub: 6.7; pd_sub: 6.4; awc_top: 5.5; dgh: 4.9; str_top: 4.2; peat: 0.9; |
| Correlation | Alnus glutinosa | Fraxinus angustifolia | Fraxinus excelsior | Populus nigra | Quercus robur | Ulmus laevis | Ulmus minor |
|---|---|---|---|---|---|---|---|
| (a) Current | −0.17 | 0.71 | −0.44 | 0.07 | 0.27 | −0.45 | −0.04 |
| (b) Future | 0.29 | −0.33 | −0.03 | −0.12 | −0.18 | 0.03 | 0.08 |
| (c) Change | −0.02 | 0.64 | 0.54 | 0.10 | 0.14 | 0.39 | −0.15 |
 |
References
- Jones, K.R.; Venter, O.; Fuller, R.A.; Allan, J.R.; Maxwell, S.L.; Negret, P.J.; Watson, J.E.M. One-third of global protected land is under intense human pressure. Science 2018, 360, 788–791. [Google Scholar] [CrossRef] [PubMed]
- Dyderski, M.K.; Paź, S.; Frelich, L.E.; Jagodziński, A.M. How much does climate change threaten European forest tree species distributions? Glob. Chang. Biol. 2018, 24, 1150–1163. [Google Scholar] [CrossRef] [PubMed]
- Bentz, B.J.; Régnière, J.; Fettig, C.J.; Hansen, E.M.; Hayes, J.L.; Hicke, J.A.; Kelsey, R.G.; Negrón, J.F.; Seybold, S.J. Climate change and bark beetles of the Western United States and Canada: Direct and indirect effects. Bioscience 2010, 60, 602–613. [Google Scholar] [CrossRef]
- Jönsson, A.M.; Appelberg, G.; Harding, S.; Bärring, L. Spatio-temporal impact of climate change on the activity and voltinism of the spruce bark beetle, Ips typographus. Glob. Chang. Biol. 2009, 15, 486–499. [Google Scholar] [CrossRef]
- Beniston, M.; Stephenson, D.B.; Christensen, O.B.; Ferro, C.A.T.; Frei, C.; Goyette, S.; Halsnaes, K.; Holt, T.; Jylhä, K.; Koffi, B.; et al. Future extreme events in European climate: An exploration of regional climate model projections. Clim. Chang. 2007. [Google Scholar] [CrossRef]
- Seidl, R.; Thom, D.; Kautz, M.; Martin-Benito, D.; Peltoniemi, M.; Vacchiano, G.; Wild, J.; Ascoli, D.; Petr, M.; Honkaniemi, J.; et al. Forest disturbances under climate change. Nat. Clim. Chang. 2017, 7, 395–402. [Google Scholar] [CrossRef]
- Dubrovský, M.; Hayes, M.; Duce, P.; Trnka, M.; Svoboda, M.; Zara, P. Multi-GCM projections of future drought and climate variability indicators for the Mediterranean region. Reg. Environ. Chang. 2014, 14, 1907–1919. [Google Scholar] [CrossRef]
- Hanel, M.; Rakovec, O.; Markonis, Y.; Máca, P.; Samaniego, L.; Kyselý, J.; Kumar, R. Revisiting the recent European droughts from a long-term perspective. Sci. Rep. 2018, 8, 9499. [Google Scholar] [CrossRef] [PubMed]
- Arnell, N.W.; Gosling, S.N. The impacts of climate change on river flood risk at the global scale. Clim. Chang. 2016, 134, 387–401. [Google Scholar] [CrossRef]
- Dottori, F.; Szewczyk, W.; Ciscar, J.C.; Zhao, F.; Alfieri, L.; Hirabayashi, Y.; Bianchi, A.; Mongelli, I.; Frieler, K.; Betts, R.A.; et al. Increased human and economic losses from river flooding with anthropogenic warming. Nat. Clim. Chang. 2018, 8, 781–786. [Google Scholar] [CrossRef]
- Winsemius, H.C.; Aerts, J.C.J.H.; Van Beek, L.P.H.; Bierkens, M.F.P.; Bouwman, A.; Jongman, B.; Kwadijk, J.C.J.; Ligtvoet, W.; Lucas, P.L.; Van Vuuren, D.P.; et al. Global drivers of future river flood risk. Nat. Clim. Chang. 2016, 6, 381–385. [Google Scholar] [CrossRef]
- Mikulová, K.; Jarolímek, I.; Šibík, J.; Bacigál, T.; Šibíková, M. Long-Term changes of softwood floodplain forests—Did the disappearance of wet vegetation accelerate the invasion process? Forests 2020, 11, 1218. [Google Scholar] [CrossRef]
- Hulme, P.E. Trade, transport and trouble: Managing invasive species pathways in an era of globalization. J. Appl. Ecol. 2009, 46, 10–18. [Google Scholar] [CrossRef]
- Meyerson, L.A.; Mooney, H.A. Invasive alien species in an era of globalization. Front. Ecol. Environ. 2007, 5, 199–208. [Google Scholar] [CrossRef]
- Boyd, I.L.; Freer-Smith, P.H.; Gilligan, C.A.; Godfray, H.C.J. The consequence of tree pests and diseases for ecosystem services. Science 2013, 342. [Google Scholar] [CrossRef] [PubMed]
- Hansen, E.M. Alien forest pathogens: Phytophthora species are changing world forests. Boreal Environ. Res. 2008, 13, 33–41. [Google Scholar]
- Allard, G.; Sigaud, P. Alien Invasive Species: Impacts on Forests and Forestry—A Review. Available online: http://www.fao.org/3/j6854e/J6854E06.htm (accessed on 30 March 2020).
- Nisbet, D.; Kreutzweiser, D.; Sibley, P.; Scarr, T. Ecological risks posed by emerald ash borer to riparian forest habitats: A review and problem formulation with management implications. For. Ecol. Manag. 2015, 358, 165–173. [Google Scholar] [CrossRef]
- Nadal-Sala, D.; Hartig, F.; Gracia, C.A.; Sabaté, S. Global warming likely to enhance black locust (Robinia pseudoacacia L.) growth in a Mediterranean riparian forest. For. Ecol. Manag. 2019, 449, 117448. [Google Scholar] [CrossRef]
- Tockner, K.; Stanford, J.A. Riverine flood plains: Present state and future trends. Environ. Conserv. 2002, 29, 308–330. [Google Scholar] [CrossRef]
- Nilsson, C.; Berggren, K. Alterations of Riparian Ecosystems Caused by River Regulation. Bioscience 2000, 50, 783. [Google Scholar] [CrossRef]
- Roder, G.; Sofia, G.; Wu, Z.; Tarolli, P. Assessment of Social Vulnerability to floods in the floodplain of northern Italy. Weather Clim. Soc. 2017, 9, 717–737. [Google Scholar] [CrossRef]
- Belletti, B.; Garcia de Leaniz, C.; Jones, J.; Bizzi, S.; Börger, L.; Segura, G.; Castelletti, A.; van de Bund, W.; Aarestrup, K.; Barry, J.; et al. More than one million barriers fragment Europe’s rivers. Nature 2020, 588, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Bonacci, O.; Oskorǔ, D. The changes in the lower Drava River water level, discharge and suspended sediment regime. Environ. Earth Sci. 2010, 59, 1661–1670. [Google Scholar] [CrossRef]
- Bonacci, O.; Oskoruš, D. The influence of three Croatian hydroelectric power plants operation on the river Drava hydrological and sediment regime. Hydrol. Forecast. 2008. [Google Scholar]
- Globevnik, L.; Kaligaric, M. Hydrological changes of the Mura River in Slovenia, accompanied with habitat deterioration in riverine space. RMZ Mater. Geoenviron. 2005, 52, 45–49. [Google Scholar]
- Habersack, H. Wasserbau, Schifffahrt und Ökologie an der Donau–Pilotprojekt Bad Deutsch-Altenburg. Osterr. Wasser Und Abfallwirtschaft 2016, 68, 190–192. [Google Scholar] [CrossRef][Green Version]
- Klimo, E.; Hager, H.; Matič, S.; Anič, I.; Kulhavý, J.; Nilsson, C.; Berggren, K. Floodplain Forests of the Temperate Zone of Europe; Brill Academic Publisher: Leiden, The Netherlands, 2008; ISBN 978-80-87154-16-8. [Google Scholar]
- Netsvetov, M.; Prokopuk, Y.; Puchałka, R.; Koprowski, M. River Regulation Causes Rapid Changes in Relationships Between Floodplain Oak Growth and Environmental Variables. Front. Plant. Sci. 2019, 10, 96. [Google Scholar] [CrossRef]
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Wetlands and Water Synthesis; Max Finlayson, C., Rebecca D’Cruz, N.D., Eds.; World Resources Institute: Washington, DC, USA, 2005. [Google Scholar]
- Leyer, I.; Mosner, E.; Lehmann, B. Managing floodplain-forest restoration in European river landscapes combining ecological and flood-protection issues. Ecol. Appl. 2012, 22, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Sanjou, M.; Okamoto, T.; Nezu, I. Experimental study on fluid energy reduction through a flood protection forest. J. Flood Risk Manag. 2018, 11, e12339. [Google Scholar] [CrossRef]
- Kevey, B. Floodplain forests. In Springer Geography; Lóczy, D., Ed.; Springer: Cham, Switzerland, 2018; Volume PartF5, pp. 299–336. ISBN 978-3-319-92815-9. [Google Scholar]
- Schnitzler, A.; Hale, B.W.; Alsum, E. Biodiversity of floodplain forests in Europe and eastern North America: A comparative study of the Rhine and Mississippi Valleys. Biodivers. Conserv. 2005, 14, 97–117. [Google Scholar] [CrossRef]
- Sikorska, D.; Sikorski, P.; Archiciński, P.; Chormański, J.; Hopkins, R.J. You can’t see the woods for the trees: Invasive Acer negundo L. in Urban riparian forests harms biodiversity and limits recreation activity. Sustainability 2019, 11, 5838. [Google Scholar] [CrossRef]
- Cartisano, R.; Mattioli, W.; Corona, P.; Mugnozza, G.S.; Sabatti, M.; Ferrari, B.; Cimini, D.; Giuliarelli, D. Assessing and mapping biomass potential productivity from poplar-dominated riparian forests: A case study. Biomass Bioenergy 2013, 54, 293–302. [Google Scholar] [CrossRef]
- Seidl, R.; Klonner, G.; Rammer, W.; Essl, F.; Moreno, A.; Neumann, M.; Dullinger, S. Invasive alien pests threaten the carbon stored in Europe’s forests. Nat. Commun. 2018, 9, 1–10. [Google Scholar] [CrossRef]
- Dukes, J.S.; Pontius, J.; Orwig, D.; Garnas, J.R.; Rodgers, V.L.; Brazee, N.; Cooke, B.; Theoharides, K.A.; Stange, E.E.; Harrington, R.; et al. Responses of insect pests, pathogens, and invasive plant species to climate change in the forests of northeastern North America: What can we predict? This article is one of a selection of papers from NE Forests 2100: A Synthesis of Climate Change Impacts on Forests of the Northeastern US and Eastern Canada. Can. J. For. Res. 2009, 39, 231–248. [Google Scholar] [CrossRef]
- Charles, H.; Dukes, J.S. Impacts of Invasive Species on Ecosystem Services. In Biological Invasions; Springer: Berlin, Germany, 2007; pp. 293–310. [Google Scholar] [CrossRef]
- Vilà, M.; Hulme, P.E. Non-native Species, Ecosystem Services, and Human Well-Being. In Impact of Biological Invasions on Ecosystem Services; Springer: Cham, Switzerland, 2017; pp. 1–14. [Google Scholar]
- Ramsfield, T.D.; Bentz, B.J.; Faccoli, M.; Jactel, H.; Brockerhoff, E.G. Forest health in a changing world: Effects of globalization and climate change on forest insect and pathogen impacts. Forestry 2016. [Google Scholar] [CrossRef]
- Buras, A.; Sass-Klaassen, U.; Verbeek, I.; Copini, P. Provenance selection and site conditions determine growth performance of pedunculate oak. Dendrochronologia 2020, 61, 125705. [Google Scholar] [CrossRef]
- Havens, K.; Vitt, P.; Still, S.; Kramer, A.T.; Fant, J.B.; Schatz, K. Seed sourcing for restoration in an era of climate change. Nat. Areas J. 2015, 35, 122–133. [Google Scholar] [CrossRef]
- Konnert, M.; Fady, B.; Gömöry, D.; A’Hara, S.; Wolter, F.; Ducci, F.; Koskela, J.; Bozzano, M.; Maaten, T.; Kowalczyk, J. Use and Transfer of Forest Reproductive Material in Europe in the Context of Climate Change; European Forest Genetic Resources Programme (EUFORGEN); Bioversity International: Rome, Italy, 2015; ISBN 9789292550318. [Google Scholar]
- Falk, W.; Mellert, K.H. Species distribution models as a tool for forest management planning under climate change: Risk evaluation of Abies alba in Bavaria. J. Veg. Sci. 2011, 22, 621–634. [Google Scholar] [CrossRef]
- Bolte, A.; Ammer, C.; Löf, M.; Madsen, P.; Nabuurs, G.J.; Schall, P.; Spathelf, P.; Rock, J. Adaptive forest management in central Europe: Climate change impacts, strategies and integrative concept. Scand. J. For. Res. 2009, 24, 473–482. [Google Scholar] [CrossRef]
- Domisch, S.; Friedrichs, M.; Hein, T.; Borgwardt, F.; Wetzig, A.; Jähnig, S.C.; Langhans, S.D. Spatially explicit species distribution models: A missed opportunity in conservation planning? Divers. Distrib. 2019, 25, 758–769. [Google Scholar] [CrossRef]
- Riordan, E.C.; Montalvo, A.M.; Beyers, J.L. Using Species Distribution Models with Climate Change Scenarios to Aid Ecological Restoration Decisionmaking for Southern California Shrublands. Available online: https://www.fs.fed.us/psw/publications/documents/psw_rp270/psw_rp270.pdf (accessed on 7 November 2020).
- Buras, A.; Menzel, A. Projecting tree species composition changes of european forests for 2061–2090 under RCP 4.5 and RCP 8.5 scenarios. Front. Plant Sci. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Marchi, M.; Ducci, F. Some refinements on species distribution models using tree-level national forest inventories for supporting forest management and marginal forest population detection. iForest 2018, 11, 291–299. [Google Scholar] [CrossRef]
- Thurm, E.A.; Hernandez, L.; Baltensweiler, A.; Ayan, S.; Rasztovits, E.; Bielak, K.; Zlatanov, T.M.; Hladnik, D.; Balic, B.; Freudenschuss, A.; et al. Alternative tree species under climate warming in managed European forests. For. Ecol. Manag. 2018, 430, 485–497. [Google Scholar] [CrossRef]
- Roces-Díaz, J.V.; Jiménez-Alfaro, B.; Álvarez-Álvarez, P.; Álvarez-García, M.A. Environmental niche and distribution of six deciduous tree species in the spanish atlantic region. iForest 2014, 8, 214–221. [Google Scholar] [CrossRef]
- Walentowski, H.; Falk, W.; Mette, T.; Kunz, J.; Bräuning, A.; Meinardus, C.; Zang, C.; Sutcliffe, L.M.E.; Leuschner, C. Assessing future suitability of tree species under climate change by multiple methods: A case study in southern Germany. Ann. For. Res. 2017, 60, 101–126. [Google Scholar] [CrossRef]
- Bastin, J.-F.; Finegold, Y.; Garcia, C.; Mollicone, D.; Rezende, M.; Routh, D.; Zohner, C.M.; Crowther, T.W. The global tree restoration potential. Science 2019, 366, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Heikkinen, R.K.; Luoto, M.; Araújo, M.B.; Virkkala, R.; Thuiller, W.; Sykes, M.T. Methods and uncertainties in bioclimatic envelope modelling under climate change. Prog. Phys. Geogr. 2006, 30, 751–777. [Google Scholar] [CrossRef]
- Diekmann, M.; Michaelis, J.; Pannek, A. Know your limits–The need for better data on species responses to soil variables. Basic Appl. Ecol. 2015, 16, 563–572. [Google Scholar] [CrossRef]
- Mellert, K.H.; Lenoir, J.; Winter, S.; Kölling, C.; Čarni, A.; Dorado-Liñán, I.; Gégout, J.C.; Göttlein, A.; Hornstein, D.; Jantsch, M.; et al. Soil water storage appears to compensate for climatic aridity at the xeric margin of European tree species distribution. Eur. J. For. Res. 2018, 137, 79–92. [Google Scholar] [CrossRef]
- Velazco, S.J.E.; Galvão, F.; Villalobos, F.; De Marco, P. Using worldwide edaphic data to model plant species niches: An assessment at a continental extent. PLoS ONE 2017, 12, 0186025. [Google Scholar] [CrossRef]
- Walthert, L.; Meier, E.S. Tree species distribution in temperate forests is more influenced by soil than by climate. Ecol. Evol. 2017, 7, 9473–9484. [Google Scholar] [CrossRef]
- Chauvier, Y.; Thuiller, W.; Brun, P.; Lavergne, S.; Descombes, P.; Karger, D.N.; Renaud, J.; Zimmermann, N.E. Influence of climate, soil, and land cover on plant species distribution in the European Alps. Ecol. Monogr. 2020. [Google Scholar] [CrossRef]
- Thuiller, W.; Araújo, M.B.; Lavorel, S. Do we need land-cover data to model species distributions in Europe? J. Biogeogr. 2004, 31, 353–361. [Google Scholar] [CrossRef]
- Austin, M.P.; Van Niel, K.P. Improving species distribution models for climate change studies: Variable selection and scale. J. Biogeogr. 2011, 38, 1–8. [Google Scholar] [CrossRef]
- Silva, L.D.; de Azevedo, E.B.; Reis, F.V.; Elias, R.B.; Silva, L. Limitations of species distribution models based on available climate change data: A case study in the azorean forest. Forests 2019, 10, 575. [Google Scholar] [CrossRef]
- Piedallu, C.; Gégout, J.C.; Perez, V.; Lebourgeois, F. Soil water balance performs better than climatic water variables in tree species distribution modelling. Glob. Ecol. Biogeogr. 2013, 22, 470–482. [Google Scholar] [CrossRef]
- Mod, H.K.; Scherrer, D.; Luoto, M.; Guisan, A. What we use is not what we know: Environmental predictors in plant distribution models. J. Veg. Sci. 2016, 27, 1308–1322. [Google Scholar] [CrossRef]
- Sinclair, S.J.; White, M.D.; Newell, G.R. How useful are species distribution models for managing biodiversity under future climates? Ecol. Soc. 2010, 15. [Google Scholar] [CrossRef]
- Riis, T.; Kelly-Quinn, M.; Aguiar, F.C.; Manolaki, P.; Bruno, D.; Bejarano, M.D.; Clerici, N.; Fernandes, M.R.; Franco, J.C.; Pettit, N.; et al. Global overview of ecosystem services provided by riparian vegetation. Bioscience 2020, 70, 501–514. [Google Scholar] [CrossRef]
- Van Looy, K.; Tormos, T.; Souchon, Y.; Gilvear, D. Analyzing riparian zone ecosystem services bundles to instruct river management. Int. J. Biodivers. Sci. Ecosyst. Serv. Manag. 2017, 13, 330–341. [Google Scholar] [CrossRef]
- Mauri, A.; Strona, G.; San-Miguel-Ayanz, J. EU-Forest, a high-resolution tree occurrence dataset for Europe. Sci. Data 2017, 4, 1–8. [Google Scholar] [CrossRef]
- Titeux, N.; Maes, D.; Van Daele, T.; Onkelinx, T.; Heikkinen, R.K.; Romo, H.; García-Barros, E.; Munguira, M.L.; Thuiller, W.; van Swaay, C.A.M.; et al. The need for large-scale distribution data to estimate regional changes in species richness under future climate change. Divers. Distrib. 2017, 23, 1393–1407. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2019. [Google Scholar]
- Chakraborty, D.; Schueler, S.; Lexer, M.J.; Wang, T. Genetic trials improve the transfer of Douglas-fir distribution models across continents. Ecography 2019, 42, 88–101. [Google Scholar] [CrossRef]
- Chakraborty, D.; Dobor, L.; Zolles, A.; Hlásny, T.; Schueler, S. High-resolution gridded climate data for Europe based on bias-corrected EURO-CORDEX: The ECLIPS dataset. Geosci. Data J. 2020. [Google Scholar] [CrossRef]
- Barbet-Massin, M.; Jiguet, F.; Albert, C.H.; Thuiller, W. Selecting pseudo-absences for species distribution models: How, where and how many? Methods Ecol. Evol. 2012, 3, 327–338. [Google Scholar] [CrossRef]
- QGIS.org. QGIS Geographic Information System 2019. Available online: https://www.qgis.org/en/site/ (accessed on 10 December 2020).
- Van Vuuren, D.P.; Edmonds, J.; Kainuma, M.; Riahi, K.; Thomson, A.; Hibbard, K.; Hurtt, G.C.; Kram, T.; Krey, V.; Lamarque, J.F.; et al. The representative concentration pathways: An overview. Clim. Chang. 2011, 109, 5–31. [Google Scholar] [CrossRef]
- European Environment Agency. EU-DEM v1.1—Copernicus Land Monitoring Service. Available online: https://land.copernicus.eu/imagery-in-situ/eu-dem/eu-dem-v1.1?tab=metadata (accessed on 7 November 2020).
- European Environment Agency. EU-Hydro–River Network Database, Version 1.2. Available online: https://land.copernicus.eu/imagery-in-situ/eu-hydro/eu-hydro-river-network-database?tab=metadata. (accessed on 7 November 2020).
- Van Liedekerke, M.; Jones, A.; Panagos, P. ESDBv2 Raster Library–A Set of Rasters Derived from the European Soil Database Distribution v2.0; CD-ROM, EUR 19945; Commission and the European Soil Bureau Network: Brussels, Belgium, 2006. [Google Scholar]
- Panagos, P. The European soil database. GEO Connex. 2006, 5, 32–33. [Google Scholar]
- Liaw, A.; Wiener, M. Classification and Regression by RandomForest. Forest 2002, 2, 18–22. [Google Scholar]
- Naimi, B.; Araújo, M.B. Sdm: A reproducible and extensible R platform for species distribution modelling. Ecography 2016, 39, 368–375. [Google Scholar] [CrossRef]
- Hengl, M.; Huber, B.; Krouzecky, N. The Integrated River Engineering Project on the Danube to the East of Vienna? River Bed Stabilisation by Coarsening of Bed Material. In Proceedings of the 33rd IAHR Congress: Water Engineering for a Sustainable Environment, Vancouver, BC, Canada, 9–14 August 2009; International Association of Hydraulic Engineering & Research (IAHR): Madrid, Spain; Curran Associates, Inc.: Red Hook, NY, USA, 2009. [Google Scholar]
- Venables, W.; Ripley, B. Tree-Based Methods. In Modern Applied Statistics with S-PLUS. Statistics and Computing; Springer: New York, NY, USA, 1997; pp. 413–430. ISBN 9781441930088. [Google Scholar]
- Ruete, A.; Leynaud, G. Goal-oriented evaluation of species distribution models’ accuracy and precision: True Skill Statistic profile and uncertainty maps. PeerJ. Prepr. 2015. [Google Scholar] [CrossRef]
- Mainali, K.P.; Warren, D.L.; Dhileepan, K.; Mcconnachie, A.; Strathie, L.; Hassan, G.; Karki, D.; Shrestha, B.B.; Parmesan, C. Projecting future expansion of invasive species: Comparing and improving methodologies for species distribution modeling. Glob. Chang. Biol. 2015, 21, 4464–4480. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, T. Chalara fraxinea sp. nov. associated with dieback of ash (Fraxinus excelsior) in Poland. For. Pathol. 2006. [Google Scholar] [CrossRef]
- Brasier, C.M. Ophiostoma novo-ulmi sp. nov., causative agent of current Dutch elm disease pandemics. Mycopathologia 1991, 115, 151–161. [Google Scholar] [CrossRef]
- Loarie, S.R.; Duffy, P.B.; Hamilton, H.; Asner, G.P.; Field, C.B.; Ackerly, D.D. The velocity of climate change. Nature 2009, 462, 1052–1055. [Google Scholar] [CrossRef] [PubMed]
- Schueler, S.; Falk, W.; Koskela, J.; Lefèvre, F.; Bozzano, M.; Hubert, J.; Kraigher, H.; Longauer, R.; Olrik, D.C. Vulnerability of dynamic genetic conservation units of forest trees in Europe to climate change. Glob. Chang. Biol. 2014, 20, 1498–1511. [Google Scholar] [CrossRef]
- Araújo, M.B.; Alagador, D.; Cabeza, M.; Nogués-Bravo, D.; Thuiller, W. Climate change threatens European conservation areas. Ecol. Lett. 2011, 14, 484–492. [Google Scholar] [CrossRef]
- Jump, A.S.; Peñuelas, J. Running to stand still: Adaptation and the response of plants to rapid climate change. Ecol. Lett. 2005, 8, 1010–1020. [Google Scholar] [CrossRef]
- Takolander, A.; Hickler, T.; Meller, L.; Cabeza, M. Comparing future shifts in tree species distributions across Europe projected by statistical and dynamic process-based models. Reg. Environ. Chang. 2019, 19, 251–266. [Google Scholar] [CrossRef]
- Thompson, I.; Mackey, B.; McNulty, S.; Mosseler, A. Forest Resilience, Biodiversity, and Climate Change: A Synthesis of the Biodiversity/Resilience/Stability Relationship in Forest Ecosystems; Technical Series No. 43; Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2009; ISBN 9292251376. [Google Scholar]
- Dyakov, N.R. Testing for assembly rules along disturbance gradients in a riparian broadleaved forest. Appl. Ecol. Environ. Res. 2019, 17, 1–13. [Google Scholar] [CrossRef]
- Zedler, J.B.; Kercher, S. Causes and consequences of invasive plants in wetlands: Opportunities, opportunists, and outcomes. CRC Crit. Rev. Plant. Sci. 2004, 23, 431–452. [Google Scholar] [CrossRef]
- Planty-Tabacchi, A.-M.; Tabacchi, E.; Naiman, R.J.; Deferrari, C.; Decamps, H. Invasibility of Species-Rich Communities in Riparian Zones. Conserv. Biol. 1996, 10, 598–607. [Google Scholar] [CrossRef]
- Von Holle, B.; Simberloff, D. Ecological resistance to biological invasion overwhelmed by propagule pressure. Ecology 2005, 86, 3212–3218. [Google Scholar] [CrossRef]
- Richardson, D.M.; Holmes, P.M.; Esler, K.J.; Galatowitsch, S.M.; Stromberg, J.C.; Kirkman, S.P.; Pyšek, P.; Hobbs, R.J. Riparian vegetation: Degradation, alien plant invasions, and restoration prospects. Divers. Distrib. 2007, 13, 126–139. [Google Scholar] [CrossRef]
- Schnitzler, A.; Hale, B.W.; Alsum, E.M. Examining native and exotic species diversity in European riparian forests. Biol. Conserv. 2007, 138, 146–156. [Google Scholar] [CrossRef]
- Aitken, S.N.; Yeaman, S.; Holliday, J.A.; Wang, T.; Curtis-McLane, S. Adaptation, migration or extirpation: Climate change outcomes for tree populations. Evol. Appl. 2008, 1, 95–111. [Google Scholar] [CrossRef]
- Vítková, M.; Müllerová, J.; Sádlo, J.; Pergl, J.; Pyšek, P. Black locust (Robinia pseudoacacia) beloved and despised: A story of an invasive tree in Central Europe. For. Ecol. Manag. 2017, 384, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Von Holle, B.; Neill, C.; Largay, E.F.; Budreski, K.A.; Ozimec, B.; Clark, S.A.; Lee, K. Ecosystem legacy of the introduced N2-fixing tree Robinia pseudoacacia in a coastal forest. Oecologia 2013. [Google Scholar] [CrossRef]
- Guisan, A.; Graham, C.H.; Elith, J.; Huettmann, F.; Dudik, M.; Ferrier, S.; Hijmans, R.; Lehmann, A.; Li, J.; Lohmann, L.G.; et al. Sensitivity of predictive species distribution models to change in grain size. Divers. Distrib. 2007, 13, 332–340. [Google Scholar] [CrossRef]
- Hanberry, B.B. Finer grain size increases effects of error and changes influence of environmental predictors on species distribution models. Ecol. Inform. 2013, 15, 8–13. [Google Scholar] [CrossRef]
- Dubuis, A.; Giovanettina, S.; Pellissier, L.; Pottier, J.; Vittoz, P.; Guisan, A. Improving the prediction of plant species distribution and community composition by adding edaphic to topo-climatic variables. J. Veg. Sci. 2013, 24, 593–606. [Google Scholar] [CrossRef]
- Austin, M.P.; Nicholls, A.O.; Margules, C.R. Measurement of the realized qualitative niche: Environmental niches of five eucalyptus species. Ecol. Monogr. 1990, 60, 161–177. [Google Scholar] [CrossRef]
- Guisan, A.; Thuiller, W. Predicting species distribution: Offering more than simple habitat models. Ecol. Lett. 2005, 8, 993–1009. [Google Scholar] [CrossRef]
- Thuiller, W.; Vayreda, J.; Pino, J.; Sabate, S.; Lavorel, S.; Gracia, C. Large-scale environmental correlates of forest tree distributions in Catalonia (NE Spain). Glob. Ecol. Biogeogr. 2003, 12, 313–325. [Google Scholar] [CrossRef]
- Serra-Diaz, J.M.; Enquist, B.J.; Maitner, B.; Merow, C.; Svenning, J.C. Big data of tree species distributions: How big and how good? For. Ecosyst. 2017, 4, 30. [Google Scholar] [CrossRef]
- Copenhaver-Parry, P.E.; Shuman, B.N.; Tinker, D.B. Toward an improved conceptual understanding of North American tree species distributions. Ecosphere 2017, 8. [Google Scholar] [CrossRef]
- Boivin, N.L.; Zeder, M.A.; Fuller, D.Q.; Crowther, A.; Larson, G.; Erlandson, J.M.; Denhami, T.; Petraglia, M.D. Ecological consequences of human niche construction: Examining long-term anthropogenic shaping of global species distributions. Proc. Natl. Acad. Sci. USA 2016, 113, 6388–6396. [Google Scholar] [CrossRef]
- Yates, K.L.; Bouchet, P.J.; Caley, M.J.; Mengersen, K.; Randin, C.F.; Parnell, S.; Fielding, A.H.; Bamford, A.J.; Ban, S.; Barbosa, A.M.; et al. Outstanding Challenges in the Transferability of Ecological Models. Trends Ecol. Evol. 2018, 33, 790–802. [Google Scholar] [CrossRef] [PubMed]
- Porfirio, L.L.; Harris, R.M.B.; Lefroy, E.C.; Hugh, S.; Gould, S.F.; Lee, G.; Bindoff, N.L.; Mackey, B. Improving the use of species distribution models in conservation planning and management under climate change. PLoS ONE 2014, 9, e113749. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, L.; Graham, E.; Duursma, D.; Wilson, P.; Cabrelli, A.; Baumgartner, J.; Hallgren, W.; Esperón-Rodríguez, M.; Nipperess, D.; Warren, D.; et al. Which species distribution models are more (or less) likely to project broad-scale, climate-induced shifts in species ranges? Ecol. Modell. 2016, 342, 135–146. [Google Scholar] [CrossRef]
- Araújo, M.B.; Cabeza, M.; Thuiller, W.; Hannah, L.; Williams, P.H. Would climate change drive species out of reserves? An assessment of existing reserve-selection methods. Glob. Chang. Biol. 2004, 10, 1618–1626. [Google Scholar] [CrossRef]
- Guisan, A.; Tingley, R.; Baumgartner, J.B.; Naujokaitis-Lewis, I.; Sutcliffe, P.R.; Tulloch, A.I.T.; Regan, T.J.; Brotons, L.; McDonald-Madden, E.; Mantyka-Pringle, C.; et al. Predicting species distributions for conservation decisions. Ecol. Lett. 2013, 16, 1424–1435. [Google Scholar] [CrossRef] [PubMed]
| Model Type | Species | Sensitivity | Specificity | TSS | Kappa |
|---|---|---|---|---|---|
| Continent-wide | A. glutinosa | 1.000 | 1.000 | 1.000 | 1.000 |
| F. angustifolia | 0.998 | 0.998 | 0.996 | 0.996 | |
| F. excelsior | 1.000 | 1.000 | 1.000 | 1.000 | |
| P. nigra | 0.999 | 0.999 | 0.998 | 0.998 | |
| Q. robur | 1.000 | 1.000 | 1.000 | 1.000 | |
| U. laevis | 1.000 | 1.000 | 1.000 | 1.000 | |
| U. minor | 0.999 | 0.999 | 0.999 | 0.999 | |
| Regional | A. glutinosa | 0.895 | 0.895 | 0.790 | 0.790 |
| F. angustifolia | 0.938 | 0.938 | 0.875 | 0.875 | |
| F. excelsior | 0.958 | 0.958 | 0.916 | 0.915 | |
| P. nigra | 0.943 | 0.943 | 0.886 | 0.886 | |
| Q. robur | 0.899 | 0.899 | 0.797 | 0.796 | |
| U. laevis | 0.958 | 0.957 | 0.914 | 0.914 | |
| U. minor | 0.908 | 0.908 | 0.814 | 0.816 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sallmannshofer, M.; Chakraborty, D.; Vacik, H.; Illés, G.; Löw, M.; Rechenmacher, A.; Lapin, K.; Ette, S.; Stojanović, D.; Kobler, A.; et al. Continent-Wide Tree Species Distribution Models May Mislead Regional Management Decisions: A Case Study in the Transboundary Biosphere Reserve Mura-Drava-Danube. Forests 2021, 12, 330. https://doi.org/10.3390/f12030330
Sallmannshofer M, Chakraborty D, Vacik H, Illés G, Löw M, Rechenmacher A, Lapin K, Ette S, Stojanović D, Kobler A, et al. Continent-Wide Tree Species Distribution Models May Mislead Regional Management Decisions: A Case Study in the Transboundary Biosphere Reserve Mura-Drava-Danube. Forests. 2021; 12(3):330. https://doi.org/10.3390/f12030330
Chicago/Turabian StyleSallmannshofer, Markus, Debojyoti Chakraborty, Harald Vacik, Gábor Illés, Markus Löw, Andreas Rechenmacher, Katharina Lapin, Sophie Ette, Dejan Stojanović, Andrej Kobler, and et al. 2021. "Continent-Wide Tree Species Distribution Models May Mislead Regional Management Decisions: A Case Study in the Transboundary Biosphere Reserve Mura-Drava-Danube" Forests 12, no. 3: 330. https://doi.org/10.3390/f12030330
APA StyleSallmannshofer, M., Chakraborty, D., Vacik, H., Illés, G., Löw, M., Rechenmacher, A., Lapin, K., Ette, S., Stojanović, D., Kobler, A., & Schueler, S. (2021). Continent-Wide Tree Species Distribution Models May Mislead Regional Management Decisions: A Case Study in the Transboundary Biosphere Reserve Mura-Drava-Danube. Forests, 12(3), 330. https://doi.org/10.3390/f12030330








