Post-Fire Demography, Growth, and Control of Eucalyptus globulus Wildlings
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Data Collection
2.3. Data Analysis
3. Results
3.1. Characterisation of Forest Stands
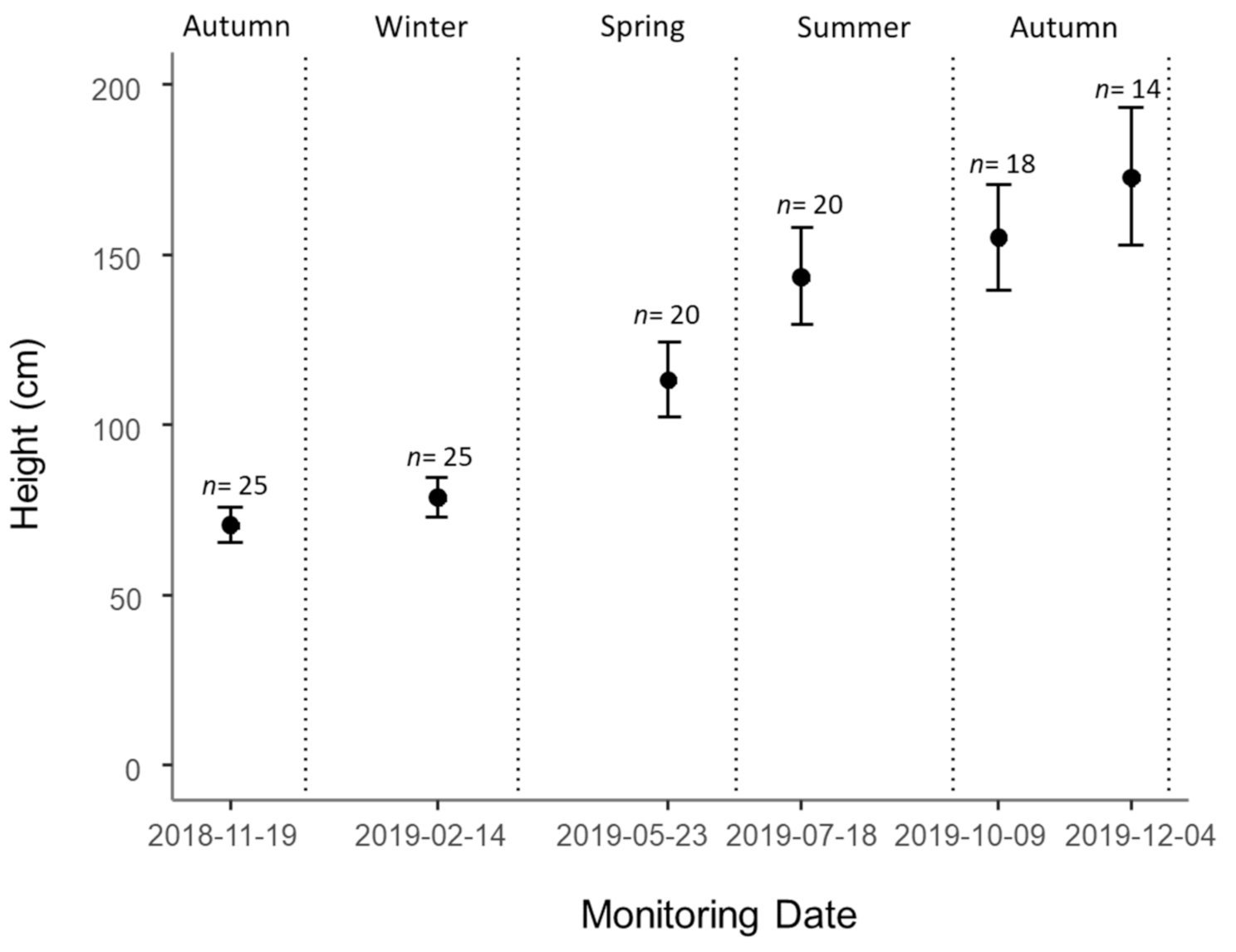
3.2. Demography
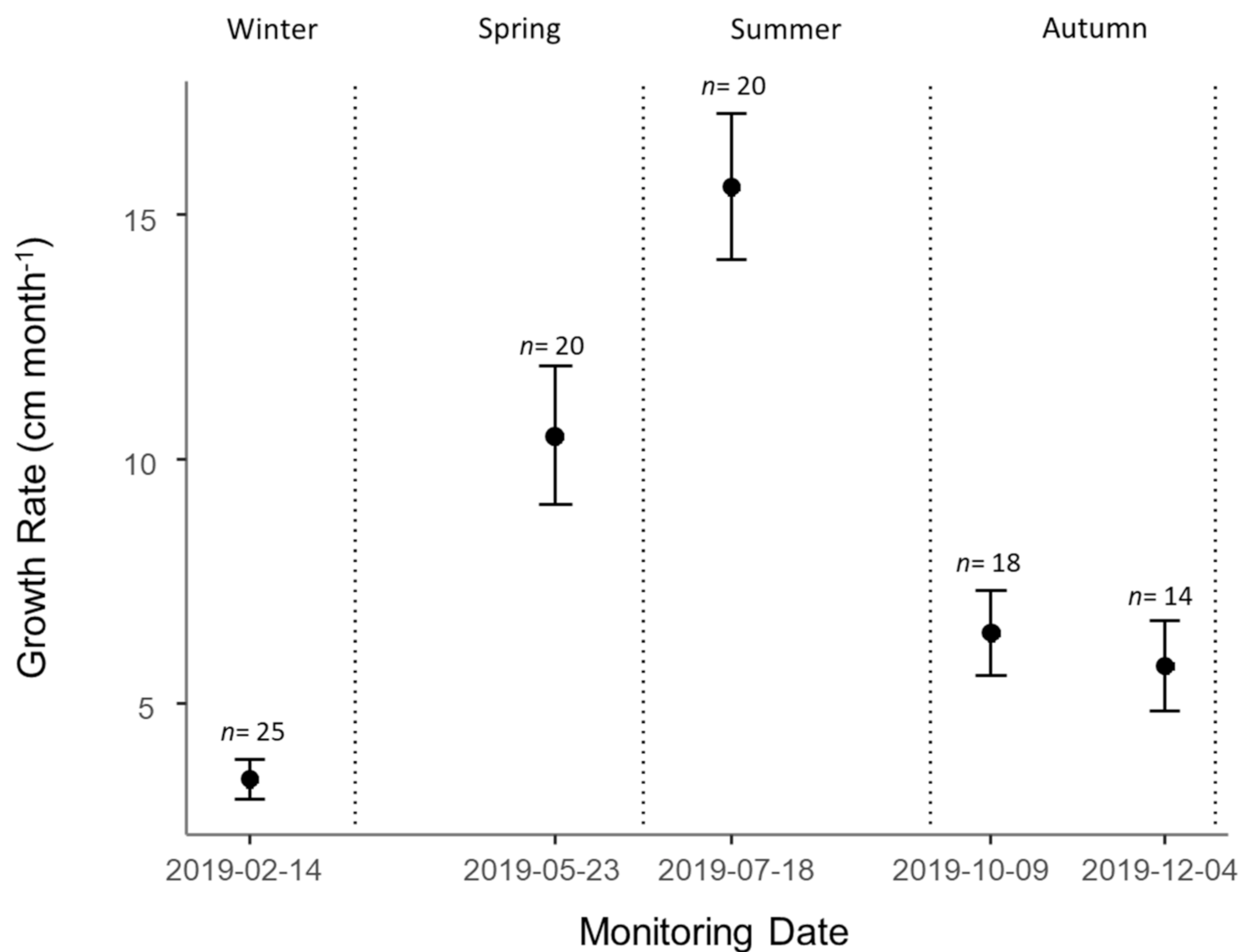
3.3. Growth
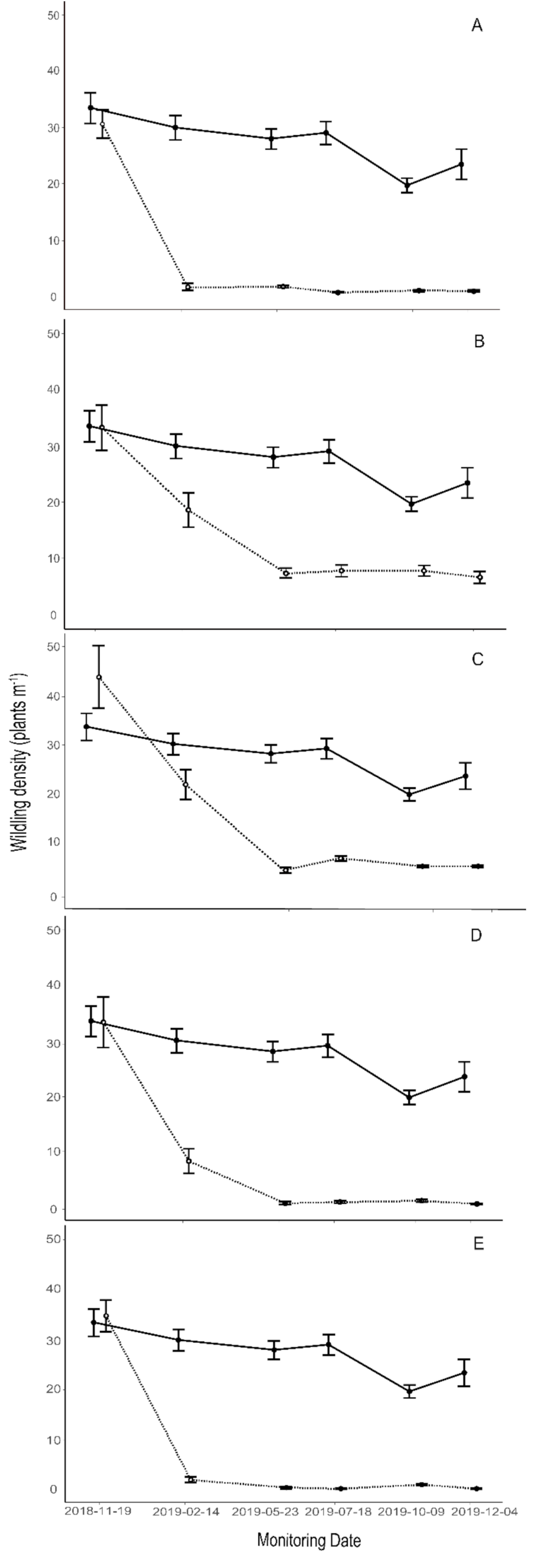
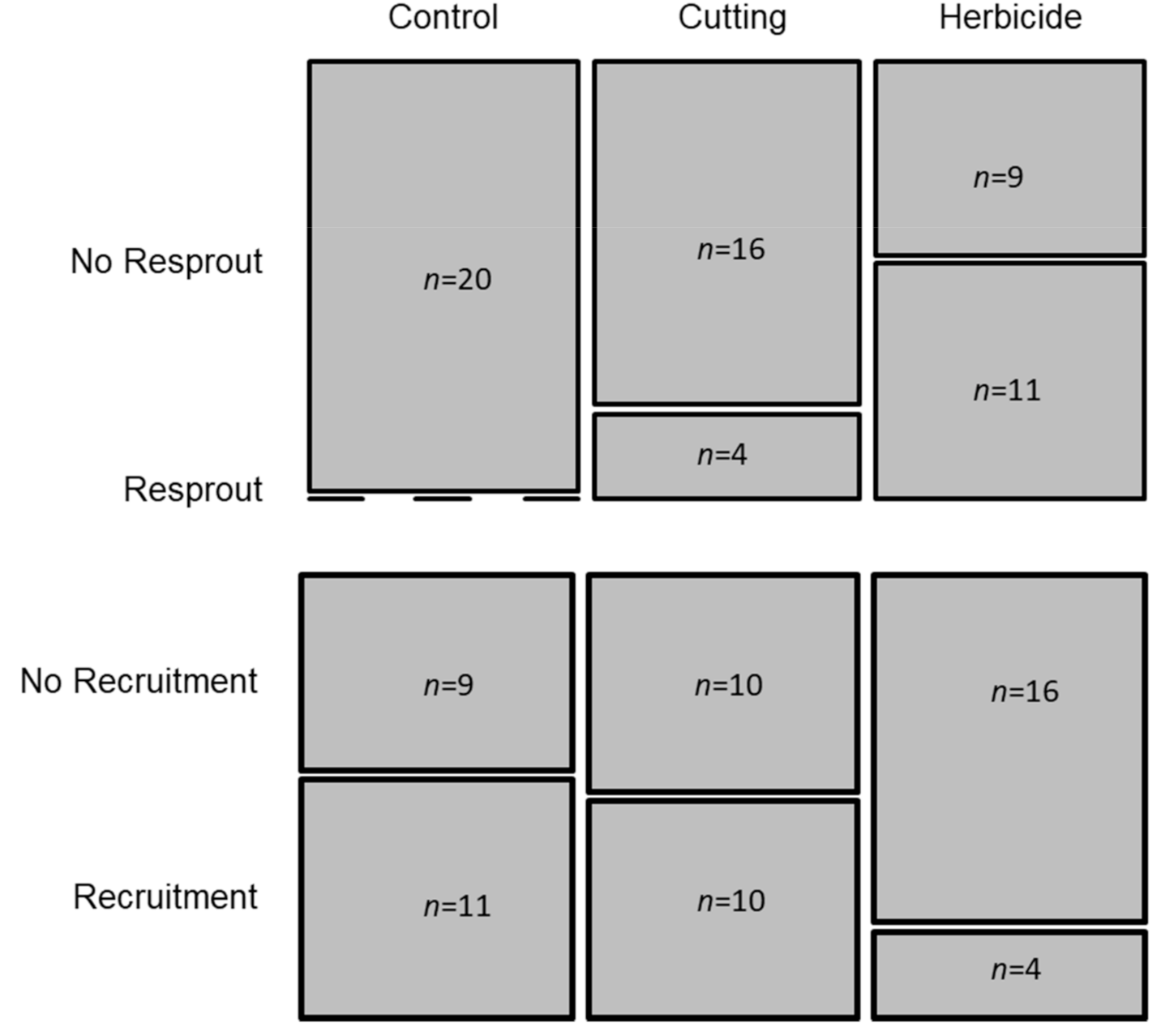
3.4. Treatments
4. Discussion
4.1. Wildling Establishment
4.2. Wildling Demography
4.3. Wildling Growth
4.4. Effectiveness of Control Methods
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jacobs, M.R. Eucalypts for Planting; Food and Agriculture Organization of the United Nations: Rome, Italy, 1981. [Google Scholar]
- Rejmánek, M.; Richardson, D. Eucalypts. In Encyclopedia of Biological Invasions; Simberloff, D., Rejmánek, M., Eds.; University of California Press: Los Angeles, CA, USA, 2011; pp. 203–209. [Google Scholar]
- Potts, B.M.; Vaillancourt, R.E.; Jordan, G.J.; Dutkowski, G.W.; Costa e Silva, J.; McKinnon, G.E.; Steane, D.A.; Volker, P.W.; Lopez, G.A.; Apiolaza, L.A.; et al. Exploration of the Eucalyptus globulus gene pool. In Proceedings of the Eucalyptus in a Changing World, Aveiro, Portugal, 11–15 October; pp. 46–61.
- Rejmánek, M.; Richardson, D.M. Trees and shrubs as invasive alien species–2013 update of the global database. Divers. Distrib. 2013, 19, 1093–1094. [Google Scholar] [CrossRef]
- ICNF. 6º Inventário Florestal Nacional-Relatório Final; Instituto da Conservação da Natureza e das Florestas: Lisboa, Portugal, 2019; p. 58. [Google Scholar]
- Águas, A.; Ferreira, A.; Maia, P.; Fernandes, P.M.; Roxo, L.; Keizer, J.; Silva, J.S.; Rego, F.C.; Moreira, F. Natural establishment of Eucalyptus globulus Labill. in burnt stands in Portugal. For. Ecol. Manag. 2014, 323, 47–56. [Google Scholar]
- Vaz, A.S.; Honrado, J.P.; Lomba, A. Replacement of pine by eucalypt plantations: Effects on the diversity and structure of tree assemblages under land abandonment and implications for landscape management. Landsc. Urban Plan. 2019, 185, 61–67. [Google Scholar]
- Fernandes, P.; Antunes, C.; Pinho, P.; Máguas, C.; Correia, O. Natural regeneration of Pinus pinaster and Eucalyptus globulus from plantation into adjacent natural habitats. For. Ecol. Manag. 2016, 378, 91–102. [Google Scholar] [CrossRef]
- Deus, E.; Silva, J.S.; Larcombe, M.J.; Catry, F.X.; Queirós, L.; Santos, P.D.; Matias, H.; Águas, A.; Rego, F.C. Investigating the invasiveness of Eucalyptus globulus in Portugal: Site-scale drivers, reproductive capacity and dispersal potential. Biol. Invasions 2019, 21, 2027–2044. [Google Scholar]
- Águas, A.; Larcombe, M.J.; Matias, H.; Deus, E.; Potts, B.M.; Rego, F.C.; Silva, J.S. Understanding the naturalization of Eucalyptus globulus in Portugal: A comparison with Australian plantations. Eur. J. For. Res. 2017, 3, 433–446. [Google Scholar] [CrossRef]
- Larcombe, M.J.; Silva, J.S.; Vaillancourt, R.E.; Potts, B.M. Assessing the invasive potential of Eucalyptus globulus in Australia: Quantification of wildling establishment from plantations. Biol. Invasions 2013, 15, 2763–2781. [Google Scholar] [CrossRef]
- Deus, E.d.; Silva, J.S.; Castro-Díez, P.; Lomba, A.; Ortiz, M.L.; Vicente, J. Current and future conflicts between eucalypt plantations and high biodiversity areas in the Iberian Peninsula. J. Nat. Conserv. 2018, 45, 107–117. [Google Scholar] [CrossRef]
- Silva, J.S.; Vaz, P.; Moreira, F.; Catry, F.; Rego, F.C. Wildfires as a major driver of landscape dynamics in three fire-prone areas of Portugal. Landscape Urban Plann. 2011, 101, 349–358. [Google Scholar] [CrossRef]
- Queirós, L.; Deus, E.; Silva, J.S.; Vicente, J.; Ortiz, L.; Fernandes, P.M.; Castro-Díez, P. Assessing the drivers and the recruitment potential of Eucalyptus globulus in the Iberian Peninsula. For. Ecol. Manage. 2020, 466, 118147. [Google Scholar] [CrossRef]
- Gill, A.M. Eucalypts and fires: Interdependent or independent? In Eucalypt Ecology: Individuals to Ecosystems; Williams, J.E., Woinarsk, J.C.Z., Eds.; Cambridge University Press: Cambridge, UK, 1997; pp. 151–167. [Google Scholar]
- Nicolle, D. A classification and census of regenerative strategies in the eucalypts (Angophora, Corymbia and Eucalyptus-Myrtaceae), with special reference to the obligate seeders. Aust. J. Bot. 2006, 54, 391–407. [Google Scholar] [CrossRef]
- Catry, F.X.; Moreira, F.; Tujeira, R.; Silva, J.S. Post-fire survival and regeneration of Eucalyptus globulus in forest plantations in Portugal. For. Ecol. Manag. 2013, 310, 194–203. [Google Scholar] [CrossRef]
- Silva, J.S.; dos Santos, P.; Sério, A.; Gomes, F. Effects of heat on dehiscence and germination in Eucalyptus globulus Labill. Int. J. Wildland Fire 2016, 25, 478–483. [Google Scholar] [CrossRef]
- Stoneman, G.L. Ecology and physiology of establishment of eucalypt seedlings from seed: A review. Aust. For. 1994, 57, 11–29. [Google Scholar]
- Cremer, K. How eucalypt fruits release their seed. Aust. J. Bot. 1965, 13, 11–16. [Google Scholar]
- Vivian, L.M.; Cary, G.J.; Bradstock, R.A.; Gill, A.M. Influence of fire severity on the regeneration, recruitment and distribution of eucalypts in the Cotter River Catchment, Australian Capital Territory. Austral Ecol. 2008, 33, 55–67. [Google Scholar] [CrossRef]
- Calviño-Cancela, M.; Lorenzo, P.; González, L. Fire increases Eucalyptus globulus seedling recruitment in forested habitats: Effects of litter, shade and burnt soil on seedling emergence and survival. For. Ecol. Manag. 2018, 409, 826–834. [Google Scholar] [CrossRef]
- Santos, P.; Matias, H.; Deus, E.; Águas, A.; Silva, J.S. Fire effects on capsules and encapsulated seeds from Eucalyptus globulus in Portugal. Plant Ecol. 2015, 216, 1611–1621. [Google Scholar] [CrossRef]
- Fernandes, P.; Maguas, C.; Correia, O. Combined effects of climate, habitat, and disturbance on seedling establishment of Pinus pinaster and Eucalyptus globulus. Plant Ecol. 2017, 218, 501–515. [Google Scholar] [CrossRef]
- Nereu, M.; Silva, J.S.; Deus, E.; Nunes, M.; Potts, B. The effect of management operations on the demography of Eucalyptus globulus seedlings. For. Ecol. Manag. 2019, 453, 117630. [Google Scholar] [CrossRef]
- Becerra, P.I.; Bustamante, R.O. The effect of herbivory on seedling survival of the invasive exotic species Pinus radiata and Eucalyptus globulus in a Mediterranean ecosystem of Central Chile. For. Ecol. Manag. 2008, 256, 1573–1578. [Google Scholar]
- Calviño-Cancela, M.; Rubido-Bará, M. Invasive potential of Eucalyptus globulus: Seed dispersal, seedling recruitment and survival in habitats surrounding plantations. For. Ecol. Manag. 2013, 305, 129–137. [Google Scholar]
- Silva, J.S.; Moreira, F.; Vaz, P.; Catry, F.; Godinho-Ferreira, P. Assessing the relative fire proneness of different fores types in Portugal. Plant Biosyst. 2009, 143, 597–608. [Google Scholar]
- Souto, X.C.; Bolaño, J.C.; González, L.; Reigosa, M.J. Allelopathic effects of tree species on some soil microbial populations and herbaceous plants. Biol. Plant. 2001, 44, 269–275. [Google Scholar] [CrossRef]
- Babu, R.C.; Kandasamy, O. Allelopathic effect of Eucalyptus globulus Labill. on Cyperus rotundus L. and Cynodon dactylon L. Pers. J. Agron. Crop Sci. 1997, 179, 123–126. [Google Scholar]
- Jayakumar, M.; Eyini, M. Allelopathic effect of Eucalyptus globulus Labil in groundnut and corn. Comp. Physiol. Ecol. 1990, 15, 109–113. [Google Scholar]
- Brundu, G.; Pauchard, A.; Pyšek, P.; Pergl, J.; Bindewald, A.M.; Brunori, A.; Canavan, S.; Campagnaro, T.; Celesti-Grapow, L.; Dechoum, M.D.S.; et al. Global guidelines for the sustainable use of non-native trees to prevent tree invasions and mitigate their negative impacts. NeoBiota 2020, 61, 65–116. [Google Scholar] [CrossRef]
- Andreu, J.; Vila, M.; Hulme, P.E. An assessment of stakeholder perceptions and management of noxious alien plants in Spain. Environ. Manag. 2009, 43, 1244–1255. [Google Scholar]
- Boland, D.J.; Brooker, M.I.H.; Turnbull, J.W. Eucalyptus Seed; CSIRO publishing: Canberra, Australia, 1980; p. 204. [Google Scholar]
- Souza-Alonso, P.; Lorenzo, P.; Rubido-Bará, M.; González, L. Effectiveness of management strategies in Acacia dealbata Link invasion, native vegetation and soil microbial community responses. For. Ecol. Manag. 2013, 304, 464–472. [Google Scholar]
- Constán-Nava, S.; Bonet, A.; Pastor, E.; Lledó, M.J. Long-term control of the invasive tree Ailanthus altissima: Insights from Mediterranean protected forests. For. Ecol. Manag. 2010, 260, 1058–1064. [Google Scholar]
- Vargas, F.; Rubilar, R.; Gonzalez-Benecke, C.A.; Sanchez-Olate, M.; Aracena, P. Long-term response to area of competition control in Eucalyptus globulus plantations. New For. 2018, 49, 383–398. [Google Scholar] [CrossRef]
- Bean, C.; Russo, M.J. Elemental Stewardship Abstract for Eucalyptus globulus; The Nature Conservancy: Richmond, VA, USA, 1989; Available online: http://www.invasive.org (accessed on 10 August 2020).
- Boyd, D. Eucalyptus removal on Angel Island. In Proceedings of the California Exotic Pest Plant Council 1997 Symposium Proceedings, Concord, CA, USA, 2–4 October; pp. 1–3.
- Burrows, G.E. Buds, bushfires and resprouting in the eucalypts. Aust. J. Bot. 2013, 61, 331–349. [Google Scholar] [CrossRef]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World map of the Köppen-Geiger climate classification updated. Meteorol. Z 2006, 15, 259–263. [Google Scholar]
- CMDFCI. Plano Municipal de Defesa da Floresta Contra Incêndios-Caderno I Diagnóstico; Câmara Municipal de Santa Comba Dão: Santa Comba Dão, Portugal, 2015. [Google Scholar]
- DGRF. Estratégia Nacional Para as Florestas; DGRF: Lisboa, Portugal, 2006; p. 189. [Google Scholar]
- Sprankle, P.; Meggitt, W.F.; Penner, D. Absorption, action, and translocation of glyphosate. Weed Sci. 1975, 23, 235–240. [Google Scholar]
- Cottam, G.; Curtis, J.T.; Hale, B.W. Some sampling characteristics of a population of randomly dispersed individuals. Ecology 1953, 34, 741–757. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop evapotranspiration-Guidelines for computing crop water requirements-FAO Irrigation and drainage paper 56. FAO Rome 1998, 300, D05109. [Google Scholar]
- Pohlert, T. The Pairwise Multiple Comparison of Mean Ranks (PMCMR)R Package. 2014. Available online: https://cran.r-project.org/web/packages/PMCMR/vignettes/PMCMR.pdf (accessed on 15 May 2020).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression; Sage publications: Los Angeles, CA, USA, 2018; p. 608. [Google Scholar]
- Zuur, A.F.; Ieno, E.N.; Walker, N.J.; Saveliev, A.A.; Smith, G.M. Mixed effects models and extensions in ecology with R. Springer: New York, NY, USA, 2009. [Google Scholar]
- Nakagawa, S.; Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- Thomas, R.; Vaughan, I.; Lello, J.; Medeiros, R.; Pollard, A.; Seward, A.; Smith, J.; Vafidis, J. Data Analysis with R Statistical Software: A Guidebook for Scientists; Eco-explore: Cardiff, South Wales, UK, 2015; p. 285. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Ruthrof, K.X.; Loneragan, W.A.; Yates, C.J. Comparative population dynamics of Eucalyptus cladocalyx in its native habitat and as an invasive species in an urban bushland in south-western Australia. Divers. Distrib. 2003, 9, 469–483. [Google Scholar]
- Ruthrof, K.X. Invasion by Eucalyptus megacornuta of an Urban Bushland in Southwestern Australia. Weed Technol. 2004, 18, 1376–1380. [Google Scholar]
- Simberloff, D. The role of propagule pressure in biological invasions. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 81–102. [Google Scholar]
- Catry, F.X.; Moreira, F.; Deus, E.; Silva, J.S.; Águas, A. Assessing the extent and the environmental drivers of Eucalyptus globulus wildling establishment in Portugal: Results from a countrywide survey. Biol. Invasions 2015, 17, 3163–3181. [Google Scholar] [CrossRef]
- Bond, W.J. Fire survival of Cape Proteaceae-influence of fire season and seed predators. Vegetatio 1984, 56, 65–74. [Google Scholar] [CrossRef]
- Deus, E.; Silva, J.S.; Marchante, H.; Marchante, E.; Félix, C. Are post-dispersed seeds of Eucalyptus globulus predated in the introduced range? Evidence from an experiment in Portugal. Web Ecol. 2018, 18, 67–79. [Google Scholar] [CrossRef]
- Ehrlén, J.; Münzbergova, Z.; Diekmann, M.; Eriksson, O. Long-term assessment of seed limitation in plants: Results from an 11-year experiment. J. Ecol. 2006, 94, 1224–1232. [Google Scholar] [CrossRef]
- Bailey, T.G.; Davidson, N.J.; Close, D.C. Understanding the regeneration niche: Microsite attributes and recruitment of eucalypts in dry forests. For. Ecol. Manag. 2012, 269, 229–238. [Google Scholar]
- Sanz-Perez, V.; Castro-Diez, P.; Joffre, R. Seasonal carbon storage and growth in Mediterranean tree seedlings under different water conditions. Tree Physiol. 2009, 29, 1105–1116. [Google Scholar] [CrossRef]
- Rubin, J.L.; Gaines, C.G.; Jensen, R.A. Enzymological basis for herbicidal action of glyphosate. Plant Physiol. 1982, 70, 833–839. [Google Scholar] [CrossRef]
- Sammons, R.D.; Gaines, T.A. Glyphosate resistance: State of knowledge. Pest Manag. Sci. 2014, 70, 1367–1377. [Google Scholar] [CrossRef]
- Borzak, C.L.; Potts, B.M.; O’Reilly-Wapstra, J.M. Survival and recovery of Eucalyptus globulus seedlings from severe defoliation. For. Ecol. Manag. 2016, 379, 243–251. [Google Scholar] [CrossRef]
- Rolando, C.; Baillie, B.; Thompson, D.; Little, K. The risks associated with glyphosate-based herbicide use in planted forests. Forests 2017, 8, 208. [Google Scholar] [CrossRef]
- Patten, K. Comparison of chemical and mechanical control efforts for invasive Spartina in Willapa Bay, Washington. In Proceedings of the Conference on Invasive Spartina, San Francisco, CA, USA, 8–9 November 2004. [Google Scholar]
- Reoyo-Prats, B.; Aubert, D.; Menniti, C.; Ludwig, W.; Sola, J.; Pujo-Pay, M.; Conan, P.; Verneau, O.; Palacios, C. Multicontamination phenomena occur more often than expected in Mediterranean coastal watercourses: Study case of the Têt River (France). Sci. Total Environ. 2017, 579, 10–21. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Arico, S.; Aronson, J.; Baron, J.S.; Bridgewater, P.; Cramer, V.A.; Epstein, P.R.; Ewel, J.J.; Klink, C.A.; Lugo, A.E.; et al. Novel ecosystems: Theoretical and management aspects of the new ecological world order. Glob. Ecol. Biogeogr. 2006, 15, 1–7. [Google Scholar] [CrossRef]
| Forest Stand Characteristics | Colmeosa | Barba | Póvoa | Soito | Torroal |
|---|---|---|---|---|---|
| Coordinates | 40°24′19.06″ N 08°08′50.65″ W | 40°25′2.71″ N 08°09′1.55″ W | 40°25′35.33″ N 08°09′01.17″ W | 40°25′00.44″ N 08°08′44.89″ W | 40°25′45.26″ N 08°08′55.17″ W |
| Altitude | 269 | 269 | 284 | 280 | 299 |
| DBH Eg (cm) | 13.9 ± 4.0 | 22.7 ± 3.2 | 13.0 ± 3.2 | 9.1 ± 0.7 | 50.7 ± 7.2 |
| DBH other spp. (cm) | 3.5 ± 0.6 | 4.1 ± 0.7 | 3.3 ± 0.5 | 14.4 ± 5.0 | 20.8 ± 4.2 |
| Tree density (trees/ha) | 3199 ± 1448 | 2012 ± 1448 | 1572 ± 520 | 589 ± 162 | 1214 ± 1535 |
| Proportion Eg (%) | 75% | 25% | 58% | 17% | 50% |
| Basal area Eg (m2 ha−1) | 36.4 | 20.4 | 12.1 | 0.01 | 122.5 |
| Basal area other spp. (m2 ha−1) | 0.8 | 2.0 | 0.6 | 8.0 | 20.6 |
| Closest Eg (m) | 2.5 ± 0.3 | 3.9 ± 2.3 | 1.3 ± 0.1 | 7.1 ± 1.5 | 5.2 ± 2.3 |
| Average density (plants m−1) | 80.4 ± 2.4 a | 23.0 ± 1.3 b | 19.5 ± 1.0 b | 15.8 ± 0.9 b | 21.9 ± 1.6 b |
| Average height (cm) | 70.0 ± 2.7 abc | 85.0 ± 3.9 ab | 66.9 ± 4.0 ce | 56.4 ± 4.7 e | 87.2 ± 4.3 a |
| Estimate | Std. Error | t Value | Pr (>|t|) | |
|---|---|---|---|---|
| (Intercept) | 2.221 | 1.527 | 1.455 | ns |
| Wildling height | 0.059 | 0.008 | 7.190 | *** |
| Maximum temperature | −1.339 | 0.220 | −6.091 | *** |
| Evapotranspiration | −0.053 | 0.014 | −3.806 | *** |
| Solar radiation | 1.597 | 0.163 | 9.780 | *** |
| Estimate | Std. Error | z Value | Pr (>|z|) | |
|---|---|---|---|---|
| (Intercept) | 1.392 | 0.562 | 2.478 | * |
| Cutting | 0.793 | 0.110 | 7.219 | *** |
| Herbicide 0.5% | 0.763 | 0.160 | 4.749 | *** |
| Herbicide 1.0% | 0.927 | 0.171 | 5.407 | *** |
| Herbicide 1.5% | 1.165 | 0.186 | 6.260 | *** |
| Herbicide 2.5% | 0.978 | 0.203 | 4.821 | *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, J.S.; Nereu, M.; Pinho, S.; Queirós, L.; Jesús, C.; Deus, E. Post-Fire Demography, Growth, and Control of Eucalyptus globulus Wildlings. Forests 2021, 12, 156. https://doi.org/10.3390/f12020156
Silva JS, Nereu M, Pinho S, Queirós L, Jesús C, Deus E. Post-Fire Demography, Growth, and Control of Eucalyptus globulus Wildlings. Forests. 2021; 12(2):156. https://doi.org/10.3390/f12020156
Chicago/Turabian StyleSilva, Joaquim S., Mauro Nereu, Simão Pinho, Luís Queirós, Cláudio Jesús, and Ernesto Deus. 2021. "Post-Fire Demography, Growth, and Control of Eucalyptus globulus Wildlings" Forests 12, no. 2: 156. https://doi.org/10.3390/f12020156
APA StyleSilva, J. S., Nereu, M., Pinho, S., Queirós, L., Jesús, C., & Deus, E. (2021). Post-Fire Demography, Growth, and Control of Eucalyptus globulus Wildlings. Forests, 12(2), 156. https://doi.org/10.3390/f12020156








