Root-Soil Plate Characteristics of Silver Birch on Wet and Dry Mineral Soils in Latvia
Abstract
1. Introduction
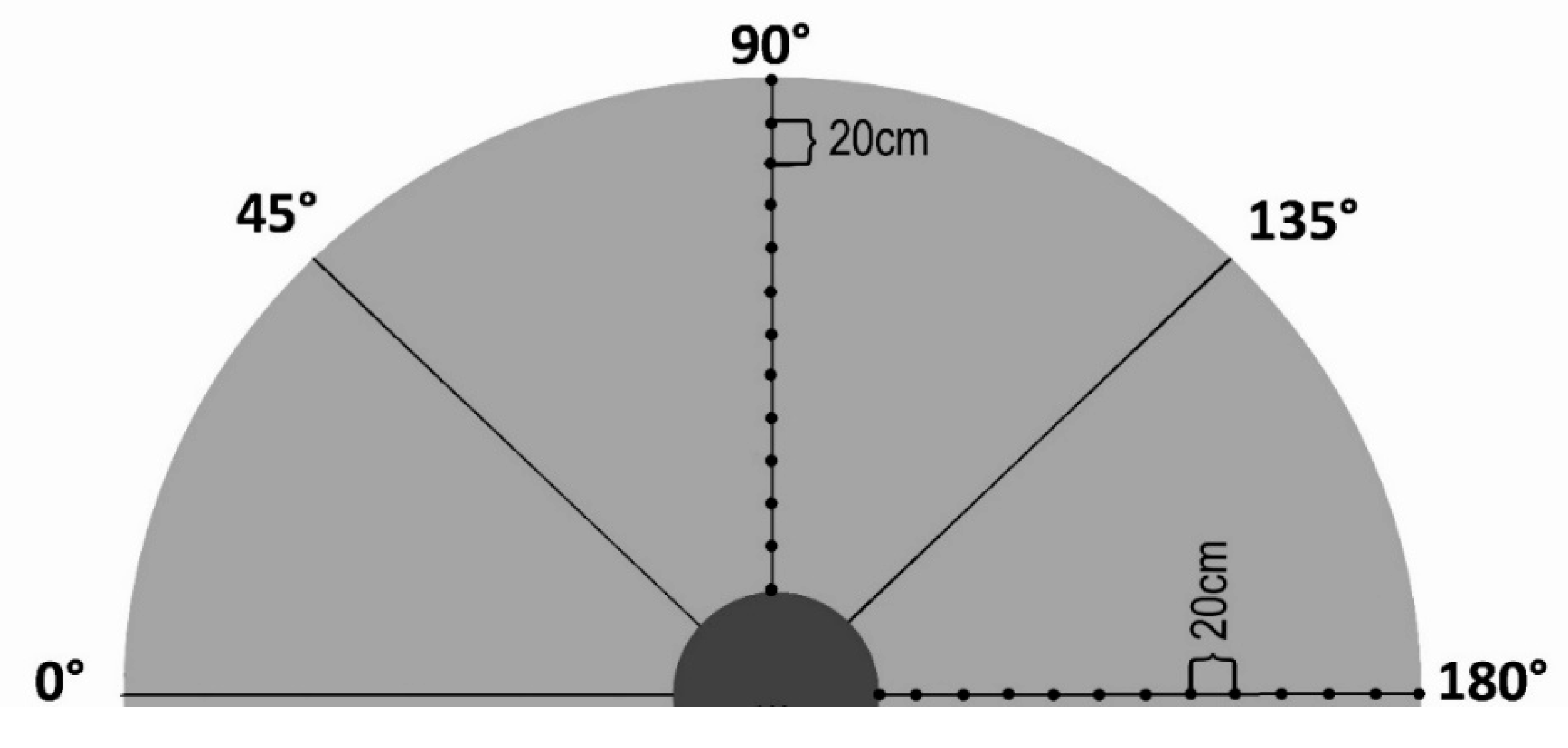
2. Materials and Methods
3. Results
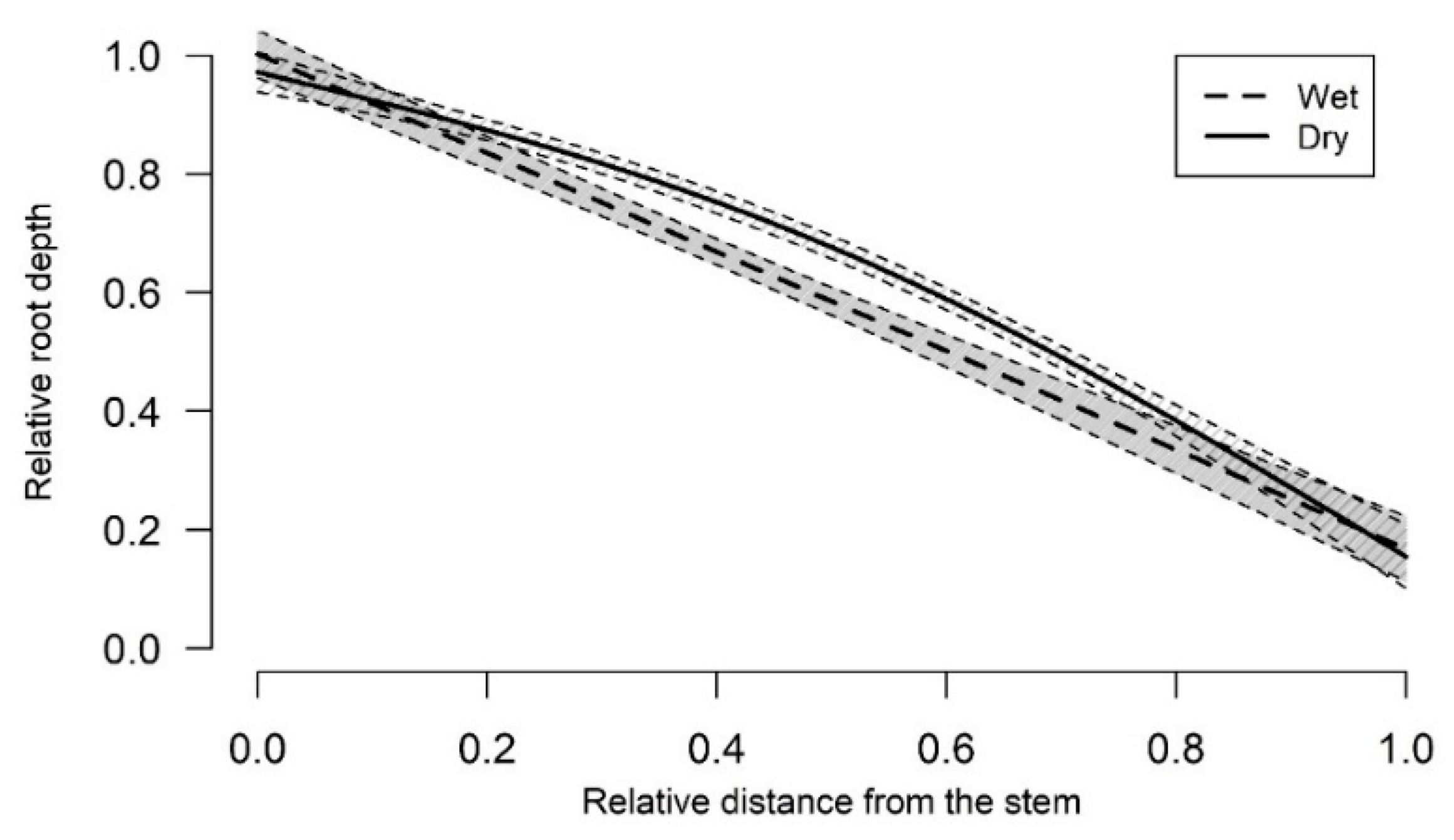
3.1. Root-Soil Plate Depth Distribution
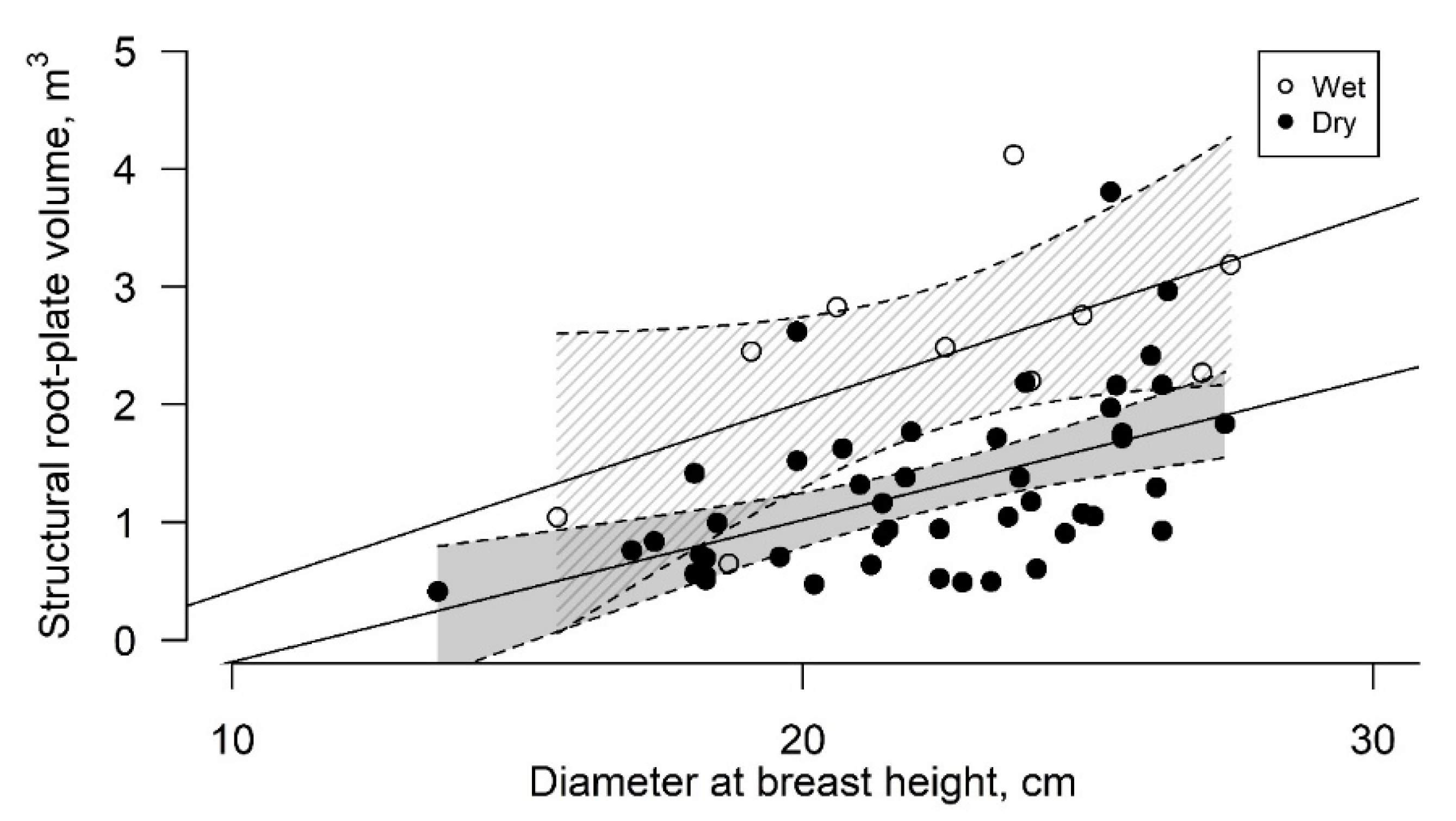
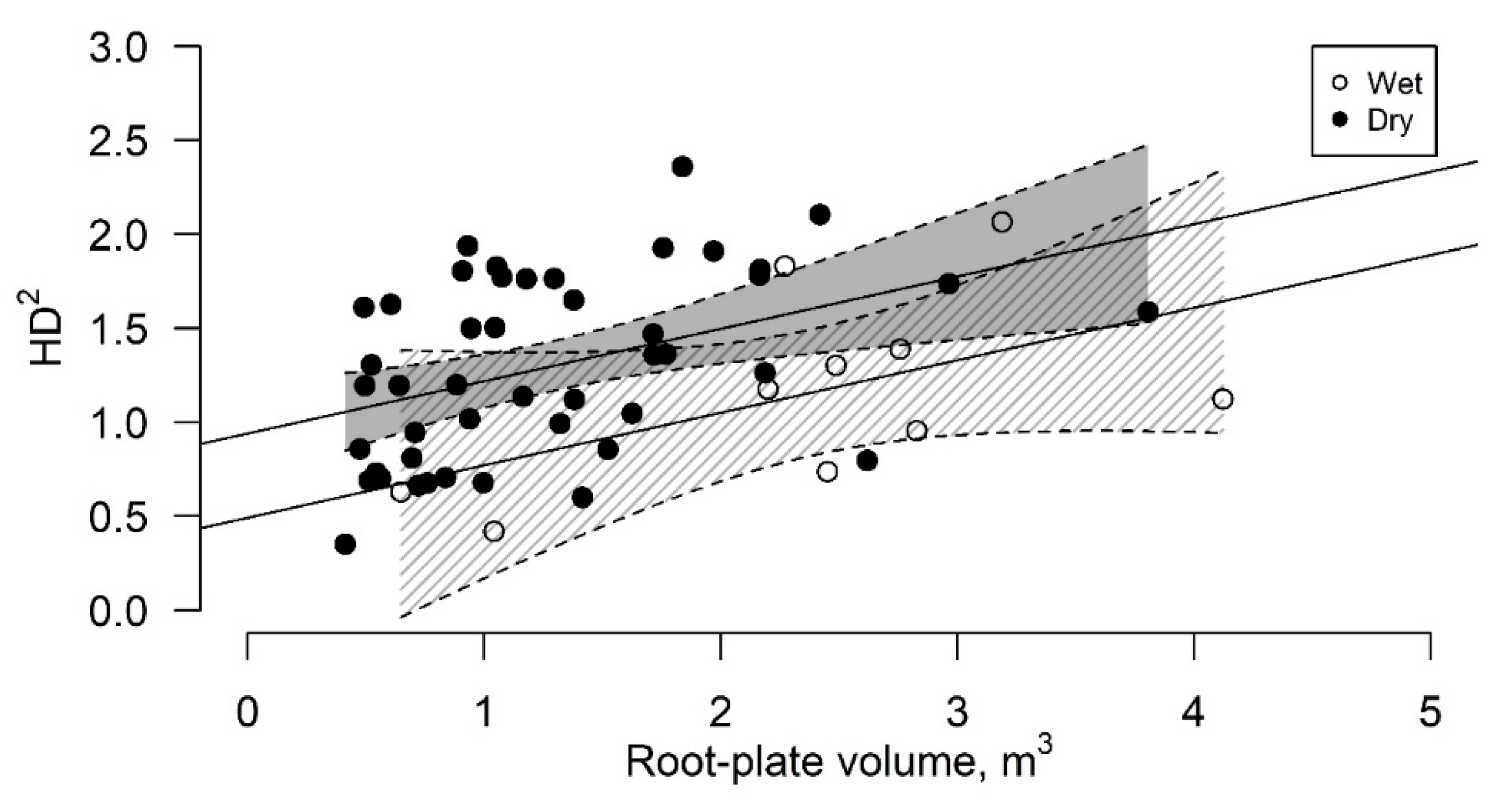
3.2. Root-Plate Volume
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gregow, H.; Laaksonen, A.; Alper, M.E. Increasing large scale windstorm damage in Western, Central and Northern European forests, 1951–2010. Sci. Rep. 2017, 7, 46397. [Google Scholar] [CrossRef] [PubMed]
- IPCC. Summary for Policymakers. In Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; Shukla, P.R., Skea, J., Calvo Buendia, E., Masson-Delmotte, V., Pörtner, H.-O., Roberts, D.C., Zhai, P., Slade, R., Connor, S., van Diemen, R., et al., Eds.; 2019; in press. [Google Scholar]
- Peltola, H.; Ikonen, V.P.; Gregow, H.; Strandman, H.; Kilpeläinen, A.; Venäläinen, A.; Kellomäki, S. Impacts of climate change on timber production and regional risks of win-induced damage to forests in Finland. For. Ecol. Manag. 2010, 260, 833–845. [Google Scholar] [CrossRef]
- Nabuurs, G.J.; Lindner, M.; Verkerk, P.J.; Gunia, K.; Deda, P.; Michalak, R.; Grassi, G. First sign of carbon sink saturation in European forest biomass. Nat. Clim. Chang. 2013, 3, 792–796. [Google Scholar] [CrossRef]
- Seidl, R.; Thom, D.; Kautz, M.; Martin-Benito, D.; Peltoniemi, M.; Vacchiano, G.; Wild, J.; Ascoli, D.; Petr, M.; Honkaniemi, J.; et al. Forest disturbances under climate change. Nat. Clim. Chang. 2017, 7, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Laapas, M.; Lehtonen, I.; Venäläinen, A.; Peltola, H.M. The 10-year return levels of maximum wind speeds under frozen and unfrozen soil forest conditions in Finland. Climate 2019, 7, 62. [Google Scholar] [CrossRef]
- Suvanto, S.; Henttonen, H.M.; Nöjd, P.; Mäkinen, H. Forest susceptibility to storm damage is affected by similar factors regardless of storm type: Comparison of thunder storms and autumn extra-tropical cyclones in Finland. For. Ecol. Manag. 2016, 381, 17–28. [Google Scholar] [CrossRef]
- IPCC. Summary for policymakers. In Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Field, C.B., Barros, V.R., Dokken, D.J., Mach, K.J., Mastrandrea, M.D., Bilir, T.E., Chatterjee, M., Ebi, K.L., Estrada, Y.O., Genova, R.C., et al., Eds.; Cambridge University Press: Cambridge, UK, 2014; pp. 1–32. [Google Scholar]
- Heinonen, T.; Pukkala, T.; Ikonen, V.P.; Peltola, H.; Venäläinen, A.; Dupont, S. Integrating the risk of wind damage into forest planning. For. Ecol. Manag. 2009, 258, 1567–1577. [Google Scholar] [CrossRef]
- Zeng, H.; Peltola, H.; Talkkari, A.; Venäläinen, A.; Strandman, H.; Kellomäki, S.; Wang, K. Influence of clear-cutting on the risk of wind damage at forest edges. For. Ecol. Manag. 2004, 203, 77–88. [Google Scholar] [CrossRef]
- Scott, R.E.; Mitchell, J.S. Empirical modelling of windthrow risk in partially harvested stands using tree, neighbourhood, and stand attributes. For. Ecol. Manag. 2005, 218, 193–209. [Google Scholar] [CrossRef]
- Grime, J.P. Plant Strategies, Vegetation Processes, and Ecosystem Properties; Wiley: Chichester, UK, 2001; p. 456. [Google Scholar]
- Dumroese, K.R.; Terzaghi, M.; Chiatante, D.; Scippa, S.G.; Lasserre, B.; Montagnoli, A. Functional traits of Pinus ponderosa coarse roots in response to slope conditions. Front. Plant Sci. 2019, 10, 947. [Google Scholar] [CrossRef]
- Stubbs, C.J.; Cook, D.D.; Niklas, K.J. A general review of the biomechanics of root anchorage. J. Exp. Bot. 2019, 70, 3439–3451. [Google Scholar] [CrossRef] [PubMed]
- Nicoll, B.C.; Ray, D. Adaptive growth of tree root systems in response to wind action and site conditions. Tree Physiol. 1996, 16, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Hynynen, J.; Niemistö, P.; Viherä-Aarnio, A.; Brunner, A.; Hein, S.; Velling, P. Silviculture of birch (Betula pendula Roth and Betula pubescens Ehrh.) in northern Europe. Int. J. For. Res. 2010, 83, 103–119. [Google Scholar] [CrossRef]
- Ministry of Agriculture. Latvian Forest Sector in Facts and Figures 2019; Zaļās Mājas: Riga, Latvia, 2019; p. 53.
- Ennos, A. The mechanics of root anchorage. Adv. Bot. Res. 2000, 33, 133–157. [Google Scholar] [CrossRef]
- Krause, C.; Lemay, A.; Tremblay, S.; Ruel, J.C.; Plourde, P.Y. How does the root system inhibit windthrow in thinned black spruce sites in the boreal forest? Trees 2014, 28, 1723–1735. [Google Scholar] [CrossRef]
- Peltola, H.; Kellomäki, S.; Hassinen, A.; Granander, M. Mechanical stability of Scots pine, Norway spruce and birch: An analysis of tree-pulling experiments in Finland. For. Ecol. Manag. 2000, 135, 143–153. [Google Scholar] [CrossRef]
- Ancelin, P.; Courbaud, B.; Fourcaud, T. Development of an individual tree-based mechanical model to predict wind damage within forest stands. For. Ecol. Manag. 2004, 203, 101–121. [Google Scholar] [CrossRef]
- Peltola, H.; Kellomäki, S.; Väisänen, H.; Ikonen, V.P. A mechanistic model for assessing the risk of wind and snow damage to single trees and stands of Scots pine, Norway spruce, and birch. Can. J. For. Res. 1999, 29, 647–661. [Google Scholar] [CrossRef]
- Gardiner, B.; Byrne, K.; Hale, S.; Kamimura, K.; Mitchell, S.J.; Peltola, H.; Ruel, J.C. A review of mechanistic modelling of wind damage risk to forests. Forestry 2008, 81, 447–463. [Google Scholar] [CrossRef]
- LEGMC, Latvian Environment, Geology and Meteorology Centre. Climate in Latvia. 2020. Available online: https://klimats.meteo.lv/klimats/latvijas_klimats/ (accessed on 8 December 2020).
- Jaagus, J.; Briede, A.; Rimkus, E.; Remm, K. Precipitation pattern in the Baltic countries under the influence of large-scale atmospheric circulation and local. Int. J. Climatol. 2010, 30, 705–720. [Google Scholar] [CrossRef]
- Bušs, K. Forest ecosystem classification in Latvia. In Proceedings of the Latvian Academy of Sciences B, Rīga, Latvia, 15 January 1997; pp. 204–218. [Google Scholar]
- Krisans, O.; Matisons, R.; Kitenberga, M.; Rust, S.; Elferts, D.; Jansons, Ā. Influence of soil moisture on wind resistance of birch (Betula spp) in hemiboreal forests. Forests 2020. under review. [Google Scholar]
- Krišāns, O.; Samariks, V.; Donis, J.; Jansons, Ā. Structural root-plate characteristics of wind-thrown Norway spruce in hemiboreal forests of Latvia. Forests 2020, 11, 1143. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 12 November 2020).
- Perala, D.A.; Alm, A.A. Reproductive ecology of birch: A review. For. Ecol. Manag. 1990, 32, 1–38. [Google Scholar] [CrossRef]
- Marimoto, J.; Aiba, M.; Furukawa, F.; Mishima, Y.; Yoshimura, N.; Nayak, S.; Takemi, T.; Haga, C.; Matusi, T.; Nakamura, F. Risk assessment of forest disturbance by typhoons with heavy precipitation in northern Japan. For. Ecol. Manag. 2021, 479, 118521. [Google Scholar] [CrossRef]
- Crook, M.J.; Ennos, A.R. The increase in anchorage with tree size of the tropical tap rooted tree Mallotus wrayi, King (Eiphorbiaceae). Ann. Bot. 1998, 82, 291–296. [Google Scholar] [CrossRef]
- Bolte, A.; Tahmann, T.; Kuhr, M.; Pogoda, P.; Murach, D.; Gadow, K.V. Relationship between tree dimension and coarse root biomass in mixed stands of European beech (Fagus sylvatica L.) and Norway spruce (Picea abies [L.] Karst.). Plant Soil. 2004, 264, 1–11. [Google Scholar] [CrossRef]
- James, K.R.; Haritos, N.; Ades, P.K. Mechanical stability of trees under dynamic loads. Am. J. Bot. 2006, 93, 1522–1530. [Google Scholar] [CrossRef]
- Gardiner, B.; Berry, P.; Moulia, B. Review: Wind impacts on plant growth, mechanics and damage. Plant Sci. 2016, 245, 94–118. [Google Scholar] [CrossRef]
- Coutts, M.P. Developmental process in tree root systems. Can. J. For. Res. 1987, 17, 761–767. [Google Scholar] [CrossRef]
- Nicoll, B.C.; Gardiner, B.A.; Rayner, B.; Peace, A.J. Anchorage of coniferous trees in relation to species, soil type, and rooting depth. Can. J. For. Res. 2006, 36, 1871–1883. [Google Scholar] [CrossRef]
- Ray, D.; Nicoll, C.B. The effect of soil water-table depth on root-plate development and stability of Sitka spruce. Forests 1998, 71, 169–182. [Google Scholar] [CrossRef]
- Fourcaud, T.; Zhang, J.-N.; Ji, Z.-Q.; Stokes, A. Understanding the impact of root morphology on overturning mechanisms: A modeling approach. Ann. Bot. 2008, 101, 1267–1280. [Google Scholar] [CrossRef] [PubMed]
| Variable | Dry Mineral Soil | Wet Mineral Soil | ||||
|---|---|---|---|---|---|---|
| Min | Max | Mean (±95% CI) | Min | Max | Mean (±95% CI) | |
| N | 46 | 10 | ||||
| DBH (cm) | 13.6 | 27.4 | 22.19 ± 0.95 | 15.7 | 27.5 | 22.37 ± 2.72 |
| H (m) | 18.3 | 31.4 | 25.13 ± 1.08 | 17 | 27.3 | 21.86 ± 2.41 |
| Root-plate width (m) | 0.45 | 1.9 | 1.05 ± 0.04 | 0.5 | 2.35 | 1.48 ± 0.10 |
| Root-plate depth (m) | 0.05 | 1.0 | 0.52 ± 0.02 | 0.1 | 0.9 | 0.52 ± 0.3 |
| GWCsoil (%) | 6.5 | 26.6 | 16.9 ± 8.4 | 24.6 | 83.7 | 38.9 ± 19.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samariks, V.; Īstenais, N.; Seipulis, A.; Miezīte, O.; Krišāns, O.; Jansons, Ā. Root-Soil Plate Characteristics of Silver Birch on Wet and Dry Mineral Soils in Latvia. Forests 2021, 12, 20. https://doi.org/10.3390/f12010020
Samariks V, Īstenais N, Seipulis A, Miezīte O, Krišāns O, Jansons Ā. Root-Soil Plate Characteristics of Silver Birch on Wet and Dry Mineral Soils in Latvia. Forests. 2021; 12(1):20. https://doi.org/10.3390/f12010020
Chicago/Turabian StyleSamariks, Valters, Nauris Īstenais, Andris Seipulis, Olga Miezīte, Oskars Krišāns, and Āris Jansons. 2021. "Root-Soil Plate Characteristics of Silver Birch on Wet and Dry Mineral Soils in Latvia" Forests 12, no. 1: 20. https://doi.org/10.3390/f12010020
APA StyleSamariks, V., Īstenais, N., Seipulis, A., Miezīte, O., Krišāns, O., & Jansons, Ā. (2021). Root-Soil Plate Characteristics of Silver Birch on Wet and Dry Mineral Soils in Latvia. Forests, 12(1), 20. https://doi.org/10.3390/f12010020






