Dynamics of Territorial Occupation by North American Beavers in Canadian Boreal Forests: A Novel Dendroecological Approach
Abstract
1. Introduction
2. Materials and Methods
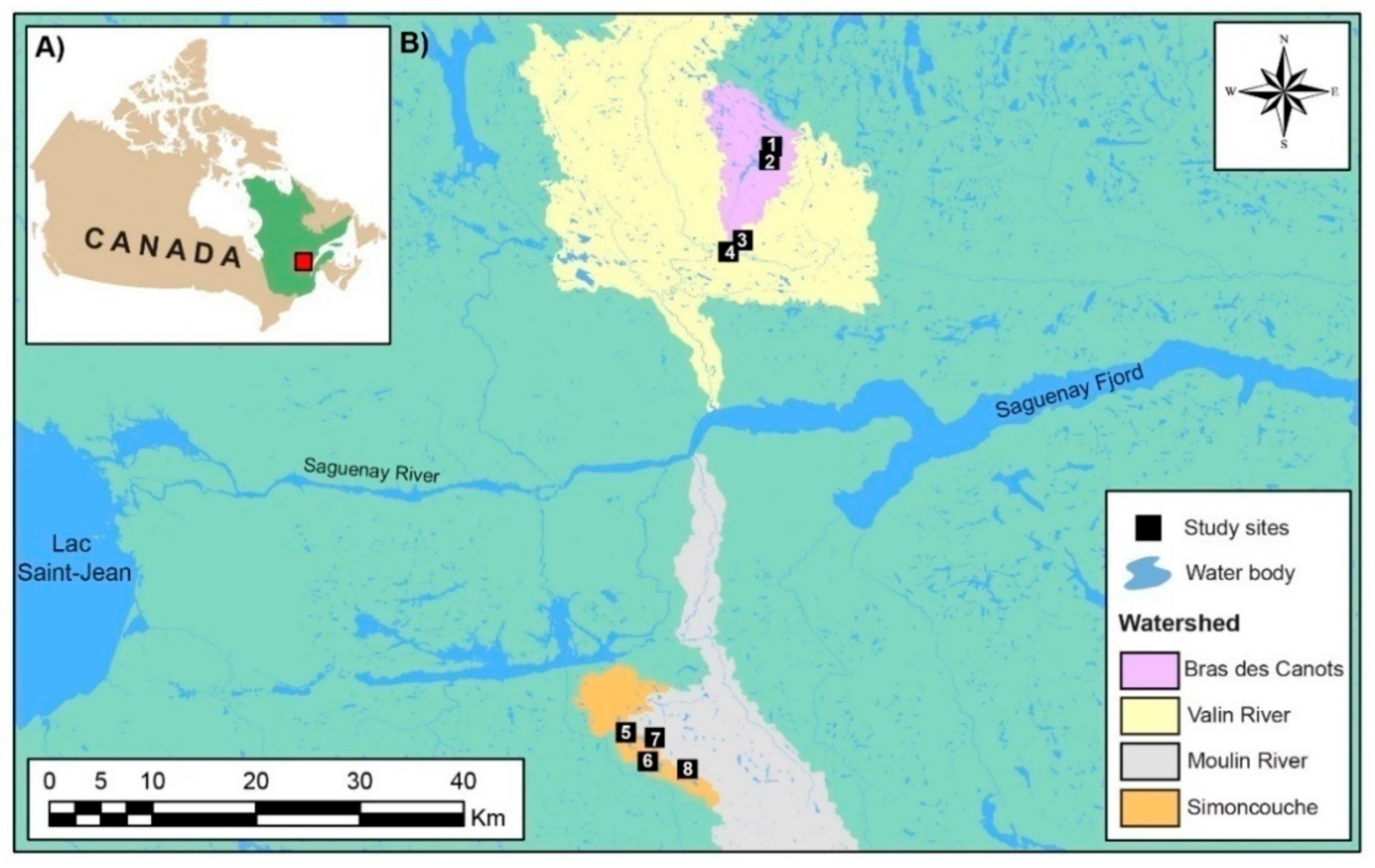
2.1. Study Area
2.2. Experimental Design and Site Selection
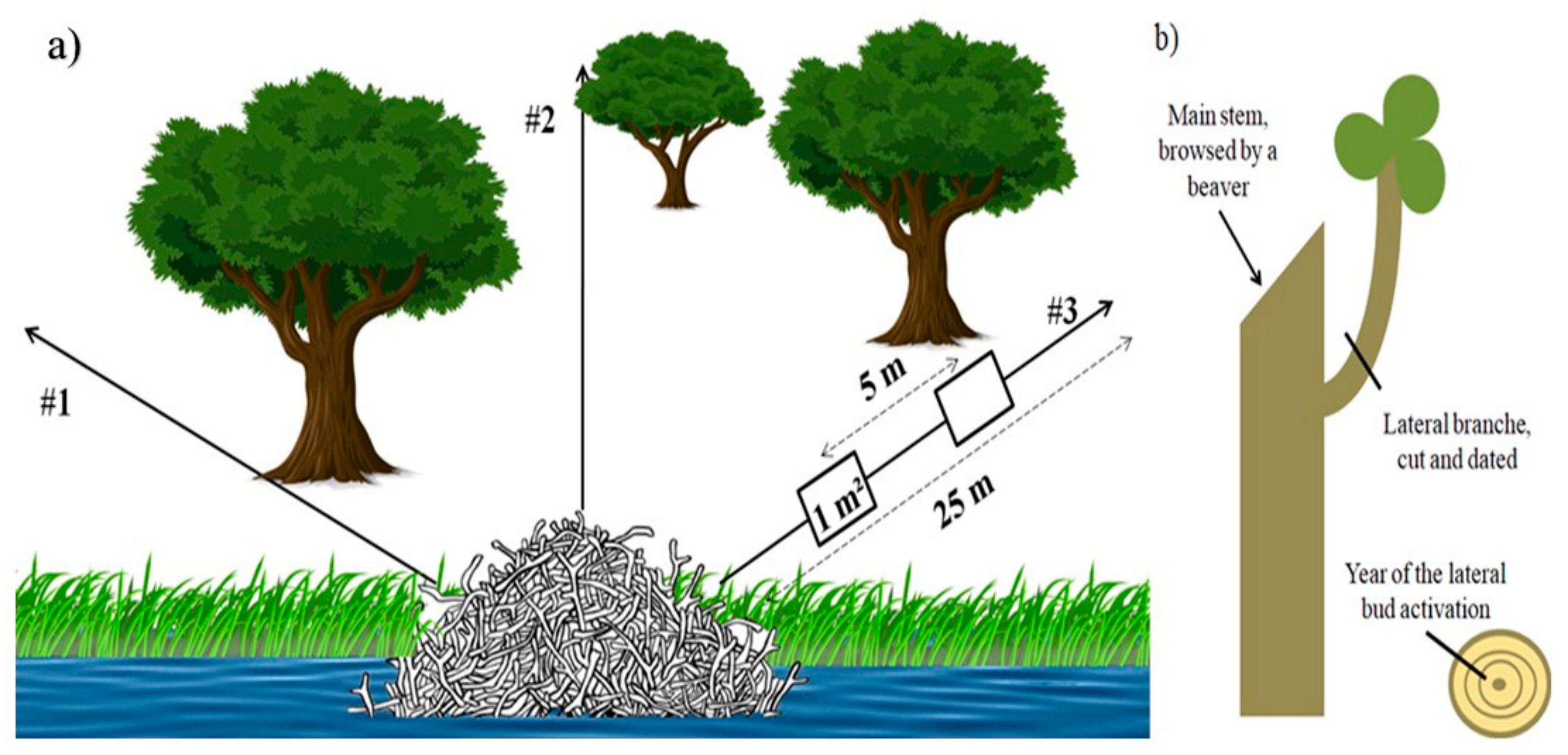
2.3. Dendroecological Approach
2.4. Data Analysis
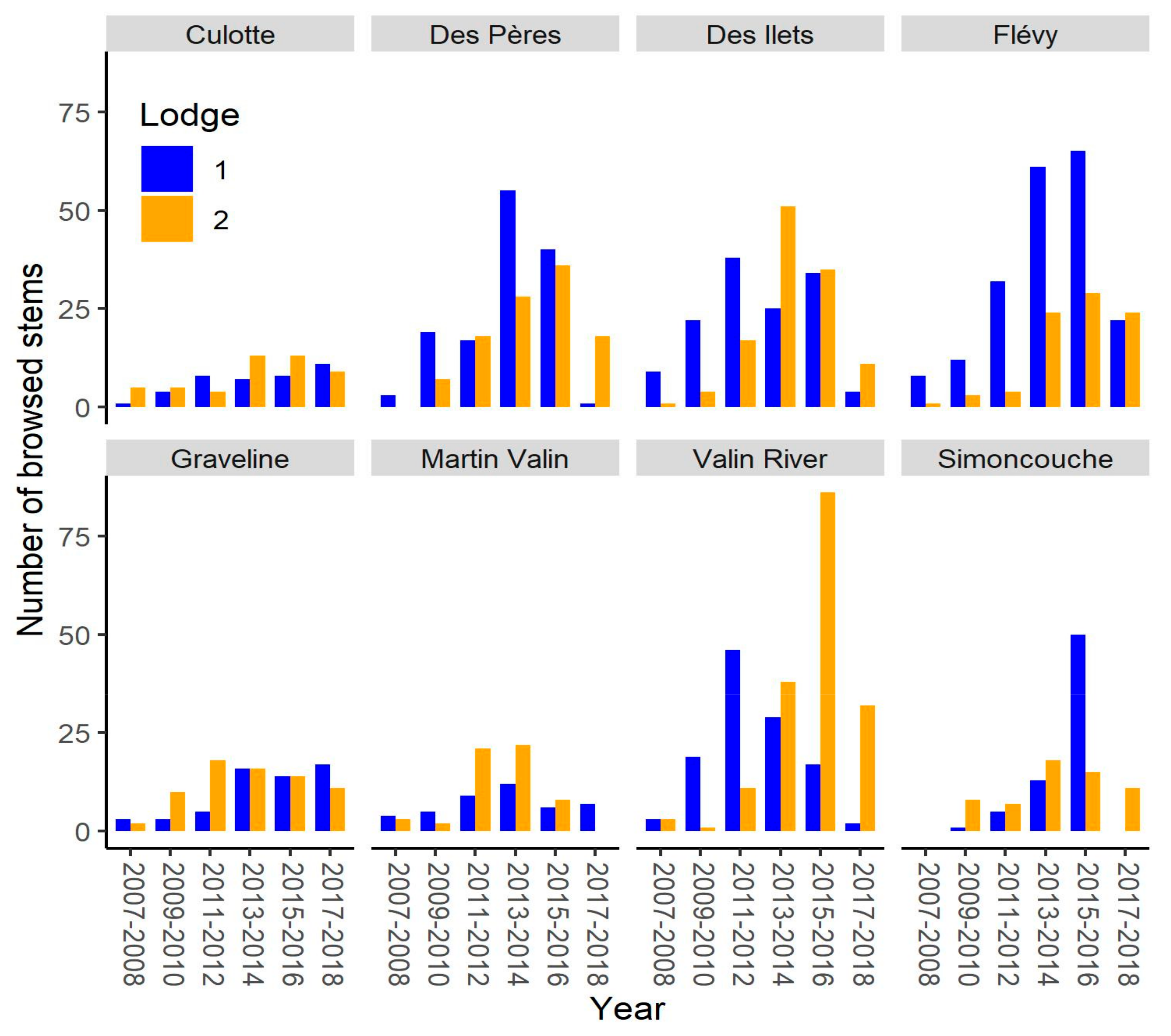
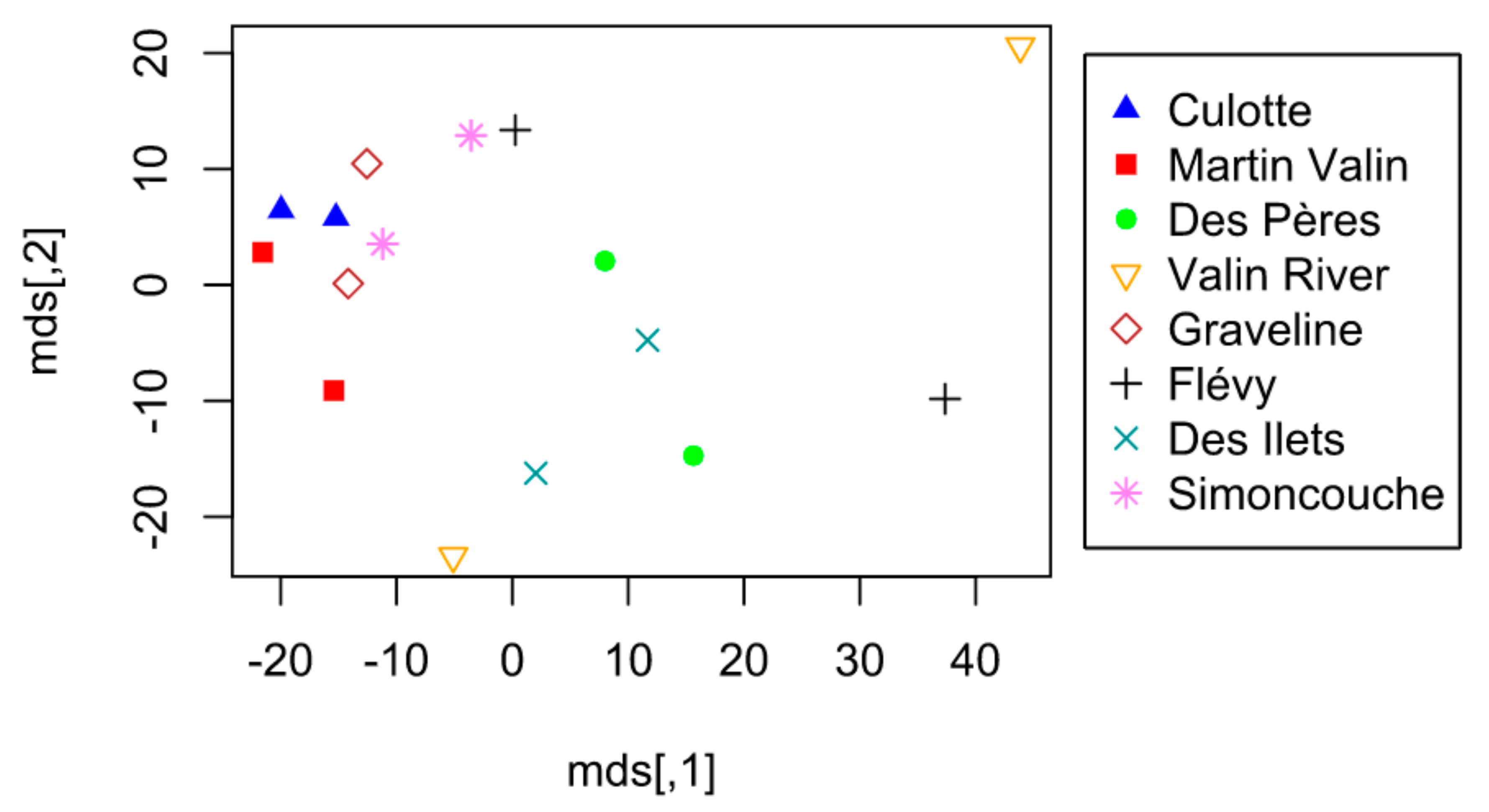
3. Results
4. Discussion
4.1. Alternation of Lodge Use
4.2. Other Observed Patterns
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brassard, B.W.; Han, Y.H.C. Dynamique de Structure et de Composition des Peuplements en Forêt Boréale Mixte: Implications Pour L’aménagement Forestier, 3rd ed.; Réseau de Gestion Durable des Forêts: Alberta, AB, Canada, 2010; p. 21. [Google Scholar]
- Lavoie, J.; Montoro Girona, M.; Morin, H. Vulnerability of Conifer Regeneration to Spruce Budworm Outbreaks in the Eastern Canadian Boreal Forest. Forests 2019, 10, 850. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Navarro, L.; Morin, H. A Secret Hidden in the Sediments: Lepidoptera Scales. Front. Ecol. Evol. 2018, 6. [Google Scholar] [CrossRef]
- Montoro Girona, M. À la Recherche de l’Aménagement Durable en Forêt Boréale: Croissance, Mortalité et Régénération des Pessières Noires Soumises à Différents Systèmes Sylvicoles; Université du Québec à Chicoutimi: Chicoutimi, QC, Canada, 2017; p. 287. [Google Scholar]
- Bergeron, Y. Species and Stand Dynamics in the Mixed Woods of Quebec’s Southern Boreal Forest. Ecology 2000, 81, 1500–1516. [Google Scholar] [CrossRef]
- Navarro, L.; Morin, H.; Bergeron, Y.; Girona, M.M. Changes in Spatiotemporal Patterns of 20th Century Spruce Budworm Outbreaks in Eastern Canadian Boreal Forests. Front. Plant Sci. 2018, 9, 1905. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Morin, H.; Lussier, J.-M.; Ruel, J.-C. Post-cutting Mortality Following Experimental Silvicultural Treatments in Unmanaged Boreal Forest Stands. Front. For. Glob. Chang. 2019, 2. [Google Scholar] [CrossRef]
- Touihri, M.; Labbe, J.; Imbeau, L.; Darveau, M. North American Beaver (Castor canadensis Kuhl) key habitat characteristics: Review of the relative effects of geomorphology, food availability and anthropogenic infrastructure. Ecoscience 2018, 25, 9–23. [Google Scholar] [CrossRef]
- Collen, P.; Gibson, R. The general ecology of beavers (Castor spp.), as related to their influence on stream ecosystems and riparian habitats, and the subsequent effects on fish—A review. Rev. Fish Biol. Fisher. 2001, 10, 439–461. [Google Scholar] [CrossRef]
- Léger, L. Utilisation de L’habitat par le Castor (Castor Canadensis) au Parc National du Canada Kouchibouguac; Université de Moncton: Moncton, NB, Canada, 2004. [Google Scholar]
- Gallant, D.; Leger, L.; Tremblay, E.; Berteaux, D.; Lecomte, N.; Vasseur, L. Linking time budgets to habitat quality suggests that beavers (Castor canadensis) are energy maximizers. Can. J. Zool. 2016, 94, 671–676. [Google Scholar] [CrossRef]
- Labbe, J. Modélisation de L’utilisation de L’habitat par le Castor Dans le Québec Forestier; Université du Québec en Abitibi-Témiscamingue, Abitibi-Témiscamingue: Rouyn-Noranda, QC, Canada, 2009. [Google Scholar]
- Fortin, C.; Laliberté, M.; Ouzilleau, J. Guide d’aménagement et de gestion du territoire utilisé par le castor au Québec; Fondation de la Faune du Québec: Sainte-Foy, QC, Canada, 2001; p. 112. [Google Scholar]
- Wright, J.P.; Jones, C.G.; Flecker, A.S. An ecosystem engineer, the beaver, increases species richness at the landscape scale. In Oecologia; Springer: Berlin/Heidelberg, Germany, 2002; Volume 132, pp. 96–101. [Google Scholar]
- Milligan, H.E.; Humphries, M.M. The importance of aquatic vegetation in beaver diets and the seasonal and habitat specificity of aquatic-terrestrial linkages in a subartic environment. Oikos 2010, 119, 1877–1886. [Google Scholar] [CrossRef]
- Metts, B.S.; Lanham, J.D.; Russell, K.R. Evaluation of Herpetofaunal Communities on Upland Streams and Beaver-Impounded Streams in the Upper Piedmont of South Carolina. Am. Midl. Nat. 2001, 145, 54–65. [Google Scholar] [CrossRef]
- Nummi, P.; Holopainen, S. Whole-community facilitation by beaver: Ecosystem engineer increases waterbird diversity. Aquat. Conserv. 2014, 24, 623–633. [Google Scholar] [CrossRef]
- Remillard, M.M.; Gruendling, G.K.; Bogucki, D.J. Disturbance by Beaver (Castor canadensis Kuhl) and Increased Landscape Heterogeneity. In Landscape Heterogeneity and Disturbance; Turner, M.G., Ed.; Springer: New York, NY, USA, 1987; pp. 103–122. [Google Scholar] [CrossRef]
- Naiman, R.J.; Melillo, J.M.; Hobbie, J.E. Ecosystem Alteation of Boreal Forest Streams by Beaver (Castor Canadensis). Ecology 1986, 67, 1254–1269. [Google Scholar] [CrossRef]
- Johnston, C.A.; Naiman, R.J. Browse selection by beaver: Effects on riparian forest composition. Can. J. For. Res. 1990, 20, 1036–1043. [Google Scholar] [CrossRef]
- Barnes, W.J.; Dibble, E. The effects of beaver in riverbank forest succession. Can. J. Botany. 1988, 66, 40–44. [Google Scholar] [CrossRef]
- St-Pierre, M.L.; Labbé, J.; Darveau, M.; Imbeau, L.; Mazerolle, M.J. Factors Affecting Abundance of Beaver Dams in Forested Landscapes. Wetlands 2017, 37, 941–949. [Google Scholar] [CrossRef]
- Jenkins, S.H. A Size-Distance Relation in Food Selection by Beavers. Ecology 1980, 61, 740–746. [Google Scholar] [CrossRef]
- Ribic, C.A.; Donner, D.M.; Beck, A.J.; Rugg, D.J.; Reinecke, S.; Eklund, D. Beaver Colony Density Trends on the Chequamegon-Nicolet National Forest, 1987–2013. PLoS ONE 2017, 12, e0170099. [Google Scholar] [CrossRef]
- Jenkins, S.H. Food selection by beavers. Oecologia 1975, 21, 157–173. [Google Scholar] [CrossRef]
- Graf, P.M.; Mayer, M.; Zedrosser, A.; Hackländer, K.; Rosell, F. Territory size and age explain movement patterns in the Eurasian beaver. Mamm. Biol. 2016, 81, 587–594. [Google Scholar] [CrossRef]
- Campbell, R.D.; Rosell, F.; Nolet, B.A.; Dijkstra, V.A.A. Territory and group sizes in Eurasian beavers (Castor fiber): Echoes of settlement and reproduction? Behav. Ecol. Sociobiol. 2005, 58, 597–607. [Google Scholar] [CrossRef]
- Gallant, D.; Berube, C.H.; Tremblay, E.; Vasseur, L. An extensive study of the foraging ecology of beavers (Castor canadensis) in relation to habitat quality. Can. J. Zool. 2004, 82, 922–933. [Google Scholar] [CrossRef]
- Bluzma, P. Beaver Abundance and Beaver Site Use in a Hilly Landscape (Eastern Lithuania). Acta Zool. Litu. 2012, 13, 8–14. [Google Scholar] [CrossRef]
- Bloomquist, K.; Nielsen, K.; Shew, J. Spatial Organization of Unexploited Beavers (Castor canadensis) in Southern Illinois. Am. Midl. Nat. 2012, 167, 188–197. [Google Scholar] [CrossRef]
- Hyvönen, T.; Nummi, P. Habitat dynamics of beaver Castor canadensis at two spatial scales. Wildlife Biol 2008, 14, 302–308. [Google Scholar] [CrossRef]
- Havens, R.; Crawford, J.; Nelson, T. Survival, Home Range, and Colony Reproduction of Beavers in East-Central Illinois, an Agricultural Landscape. Am. Midl. Nat. 2013, 169, 17–29. [Google Scholar] [CrossRef]
- Ropars, P.; Boudreau, S. Shrub expansion at the forest–tundra ecotone: Spatial heterogeneity linked to local topography. Environ. Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Tape, K.D.; Jones, B.M.; Arp, C.D.; Nitze, I.; Grosse, G. Tundra be dammed: Beaver colonization of the Arctic. Glob Chang. Biol 2018, 24, 4478–4488. [Google Scholar] [CrossRef]
- Czerepko, J.; Wróbel, M.; Boczoń, A.; Sokołowski, K. The response of ash-alder swamp forest to increasing stream water level caused by damming by the European beaver (Castor fiber L.). J. Water Land Dev. 2009, 13. [Google Scholar] [CrossRef]
- Dieter, C.; McCabe, T. Factors Influencing Beaver Lodge-site Selection on a Prairie River. Am. Midl. Nat. 1989, 122, 408–411. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Morin, H.; Lussier, J.-M.; Walsh, D. Radial Growth Response of Black Spruce Stands Ten Years after Experimental Shelterwoods and Seed-Tree Cuttings in Boreal Forest. Forests 2016, 7, 240. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Lussier, J.-M.; Morin, H.; Thiffault, N. Conifer Regeneration after Experimental Shelterwood and Seed-Tree Treatments in Boreal Forests: Finding Silvicultural Alternatives. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Grosbois, G.; del Giorgio, P.A.; Rautio, M. Zooplankton allochthony is spatially heterogeneous in a boreal lake. Freshwater Biol. 2017, 62, 474–490. [Google Scholar] [CrossRef]
- MFFP. Ecoforest Data. Available online: https://geoegl.msp.gouv.qc.ca/igo/mffpecofor/ (accessed on 13 May 2019).
- Robitaille, A.; Saucier, J. Paysages Régionaux du Québec Méridional; Les publications du Québec: Sainte-Foy, France, 1998; p. 213. [Google Scholar]
- Rossi, S.; Morin, H.; Deslauriers, A.; Plourde, P.-Y. Predicting xylem phenology in black spruce under climate warming. Global. Chang. Biol. 2011, 17, 614–625. [Google Scholar] [CrossRef]
- Canada, G.O. Canadian Climate Normals. Available online: http://climat.meteo.gc.ca/climate_normals (accessed on 13 May 2019).
- Jarema, S.I.; Samson, J.; McGill, B.J.; Humphries, M.M. Variation in abundance across a species’ range predicts climate change responses in the range interior will exceed those at the edge: A case study with North American beaver. Global. Chang. Biol. 2009, 15, 508–522. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Rossi, S.; Lussier, J.M.; Walsh, D.; Morin, H. Understanding tree growth responses after partial cuttings: A new approach. PLoS ONE 2017, 12, e0172653. [Google Scholar] [CrossRef] [PubMed]
- Keigley, R.B.; Frisina, M.R.; Fager, C. A Method for Determining the Onset Year of Intense Browsing. J. Range Manag. 2003, 56, 33–38. [Google Scholar] [CrossRef]
- Borg, I.; Groenen, P.J.F. Modern Multidimensional Scaling: Theory and Applications, 2rd ed.; Springer: New York, NY, USA, 2005; p. 613. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. Available online: https://CRAN.R-project.org/package=vegan (accessed on 26 July 2019).
- R Core Team. R: A language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2013. [Google Scholar]
- Speer, J.H. Fundamentals of Tree-Ring Research; University of Arizona Press: Tucson, AZ, USA, 2012. [Google Scholar]
- Coomes, D.A.; Allen, R.B. Effect of Size, Competition and Altitude on Tree Growth. J. Ecol. 2007, 95, 1084–1097. [Google Scholar] [CrossRef]
- Tremblay, G.; Valeria, O.; Imbeau, L. Characterisation of Beaver Habitat Parameters That Promote the Use of Culverts as Dam Construction Sites: Can We Limit the Damage to Forest Roads? Forests 2017, 8, 494. [Google Scholar] [CrossRef]
| Region | Watershed | Water Body (Name—Type) | Location | Size (ha) | Stand Type | Number of Lodges | Distance between the Studied Lodges (m) |
|---|---|---|---|---|---|---|---|
| Simoncouche | Simoncouche | Simoncouche—Lake | 48°13′50.56″ N 71°15′02.32″ W | 82.9 | Trembling aspen | 11 | 2200 |
| Des Îlets—Lake | 48°11′57.90″ N 71°14′13.81″ W | 167.7 | Trembling aspen, balsam fir | 2 | 1400 | ||
| Rivière du Moulin | Flévy—Lake | 48°13′02.08″ N 71°12′58.86″ W | 4.0 | White birch, trembling aspen | 2 | 200 | |
| Graveline—Lake | 48°10′55.18″ N 71°11′22.48″ W | 4.0 | Trembling aspen, black spruce | 3 | 200 | ||
| Monts-Valin | Bras-des-Canots | Culotte—Lake | 48°38′18.31″ N 70°47′27.55″ W | 11.1 | Balsam fir, black spruce | 4 | 180 |
| Martin-Valin—Lake | 48°38′37.24″ N 70°46′46.14″ W | 94.3 | Balsam fir, black spruce | 8 | 580 | ||
| Rivière Valin | Des Pères—Lake | 48°35′10.17″ N 70°52′00.40″ W | 2.0 | Yellow birch, white birch, trembling aspen, balsam fir | 2 | 240 | |
| Valin River—River | 48°34′42.29″ N 70°52′45.34″ W | -------- | Balsam fir, trembling aspen | 6 | 730 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Labrecque-Foy, J.-P.; Morin, H.; Girona, M.M. Dynamics of Territorial Occupation by North American Beavers in Canadian Boreal Forests: A Novel Dendroecological Approach. Forests 2020, 11, 221. https://doi.org/10.3390/f11020221
Labrecque-Foy J-P, Morin H, Girona MM. Dynamics of Territorial Occupation by North American Beavers in Canadian Boreal Forests: A Novel Dendroecological Approach. Forests. 2020; 11(2):221. https://doi.org/10.3390/f11020221
Chicago/Turabian StyleLabrecque-Foy, Julie-Pascale, Hubert Morin, and Miguel Montoro Girona. 2020. "Dynamics of Territorial Occupation by North American Beavers in Canadian Boreal Forests: A Novel Dendroecological Approach" Forests 11, no. 2: 221. https://doi.org/10.3390/f11020221
APA StyleLabrecque-Foy, J.-P., Morin, H., & Girona, M. M. (2020). Dynamics of Territorial Occupation by North American Beavers in Canadian Boreal Forests: A Novel Dendroecological Approach. Forests, 11(2), 221. https://doi.org/10.3390/f11020221







