Biodiversity of the Cocoa Agroforests of the Bengamisa-Yangambi Forest Landscape in the Democratic Republic of the Congo (DRC)
Abstract
1. Introduction
2. Materials and Methods
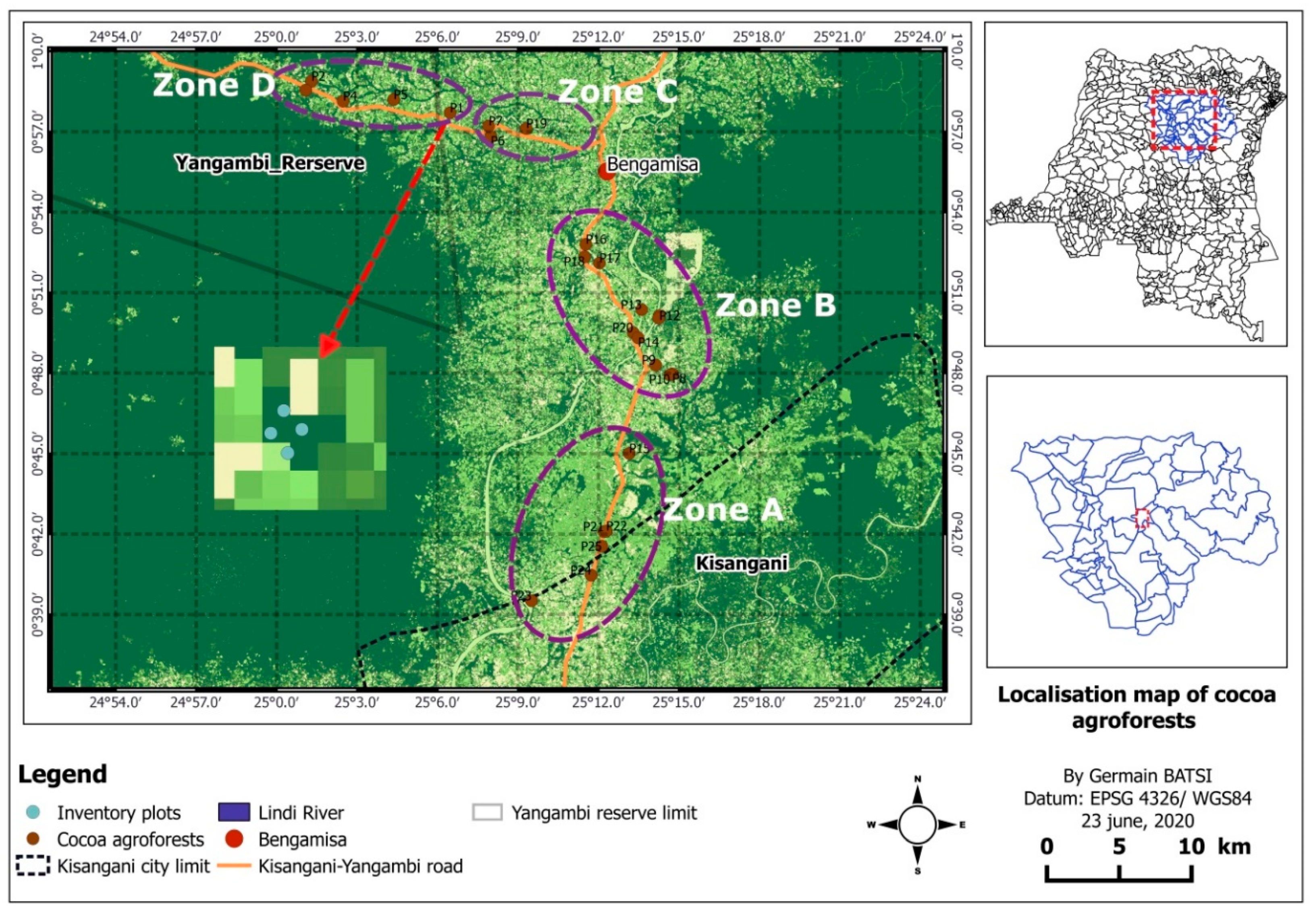
2.1. Study Area
2.2. Methods
2.2.1. Study Design
2.2.2. Collection of Floristic Data
2.2.3. Data Analysis
3. Results
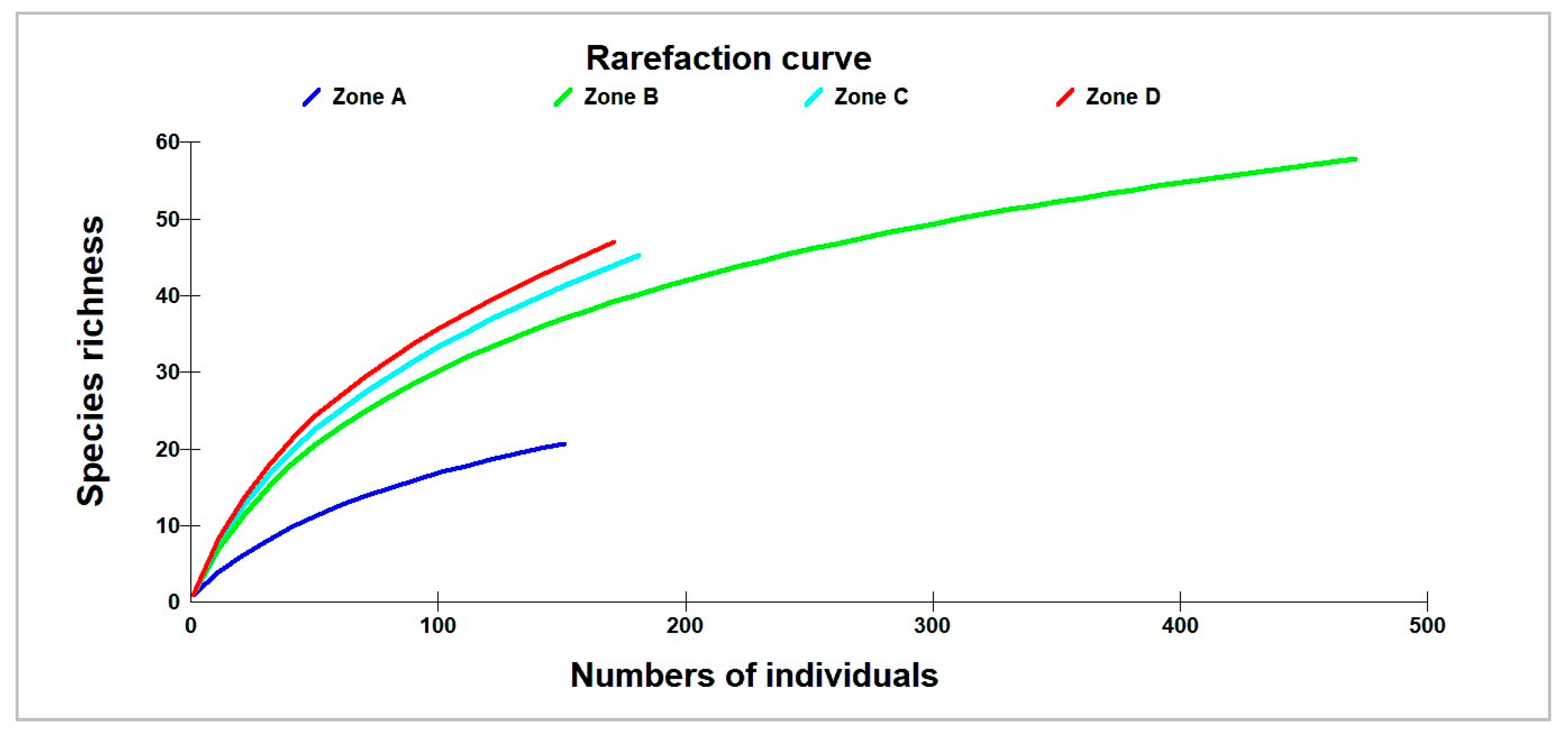
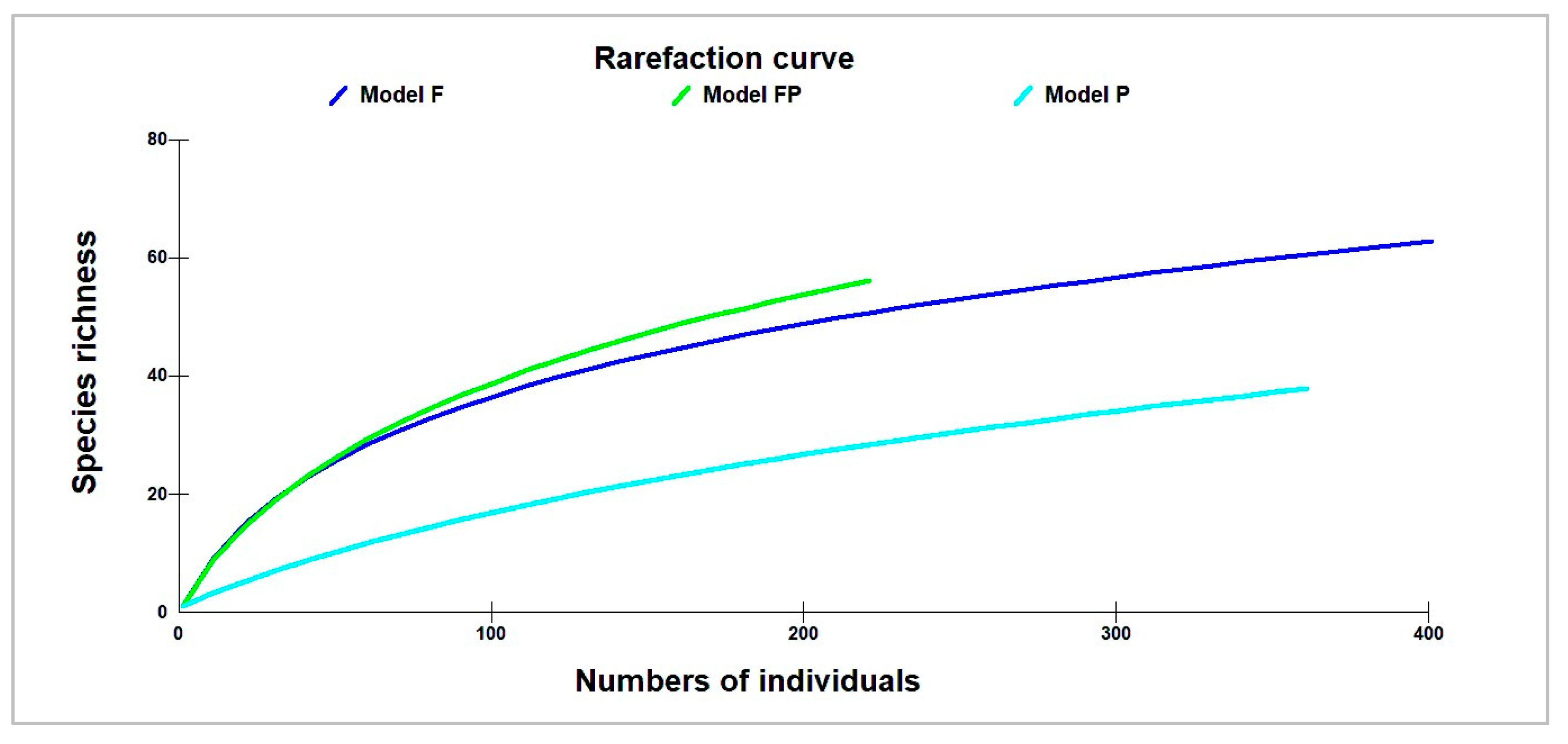
3.1. Species Richness and Diversity Index
3.2. Abundance of Plants Associated with Cocoa
3.3. Suitable and Unsuitable Species for Cocoa
3.3.1. Suitable Species to Cocoa Agronomy
3.3.2. Unsuitable Species for Cocoa Agronomy
3.4. Main Uses of Plants Associated with Cocoa
3.4.1. Main Uses of Plants Associated with Cocoa by Zone (i.e., Main Distance from Kisangani)
3.4.2. Main Uses of Plants Associated with Cocoa by Cocoa Agroforest Models
3.5. Structure of Cocoa Agroforests
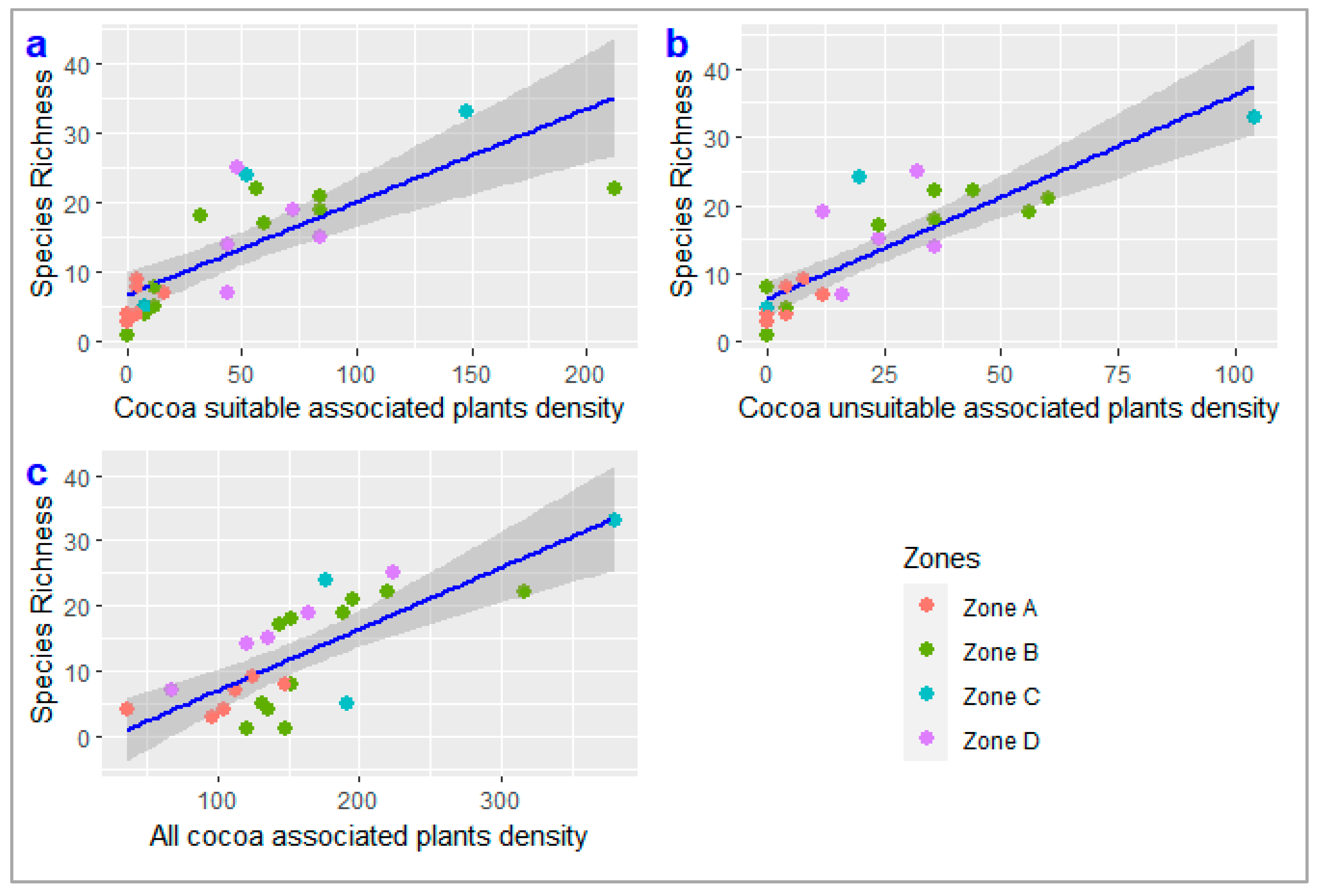
3.6. Relationship between Density and Species Richness of Cocoa Agroforest
4. Discussion
4.1. Floristic Composition of Cocoa Agroforests
4.2. Specific Diversity in the Cocoa Agroforests of the Study Area
4.3. Structure of Cocoa Agroforests
4.4. Main Local Uses of Cocoa-Associated Plants
4.5. Landscape Management Implication
4.6. Limitation of this Paper and Perspectives
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Foley, J.A.; DeFries, R.; Asner, G.P.; Barford, C.; Bonan, G.; Carpenter, S.R.; Chapin, F.S.; Coe, M.T.; Daily, G.C.; Gibbs, H.K.; et al. Global consequences of land-use. Science 2005, 309, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Clough, Y.; Barkmann, J.; Juhrbandt, J.; Kessler, M.; Wanger, T.C.; Anshary, A.; Buchori, D.; Cicuzza, D.; Darras, K.; Dwi Putra, D.; et al. Combining high biodiversity with high yields in tropical agroforests. Proc. Natl. Acad. Sci. USA 2011, 108, 8311–8316. [Google Scholar] [CrossRef] [PubMed]
- Rice, R.A.; Greenberg, R. Cacao cultivation and the conservation of biological diversity. Ambio 2000, 29, 167–173. [Google Scholar] [CrossRef]
- Götz, S.; Harvey, C.A.; Grégoire, V. Agroforestry and biodiversity conservation in tropical landscapes. In Agroforestry Systems; Götz, S., da Fonseca, G., Harvey, C.A., Gascon, C., Vasconcelos, H., Izac, A.-M., Eds.; Island Press: Washington, DC, USA, 2004; Volume 68, pp. 247–249. [Google Scholar]
- Bhagwat, S.A.; Willis, K.J.; Birks, H.J.B.; Whittaker, R.J. Agroforestry: A refuge for tropical biodiversity? Trends Ecol. Evol. 2008, 23, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Cicuzza, D.; Kessler, M.; Clough, Y.; Pitopang, R.; Leitner, D.; Tjitrosoedirdjo, S.S. Conservation value of cacao agroforestry systems for terrestrial herbaceous species in Central Sulawesi, Indonesia. Biotropica 2011, 43, 755–762. [Google Scholar] [CrossRef]
- Sonwa, D.J.; Weise, S.F.; Schroth, G.; Janssens, M.J.J. Howard-Yana Shapiro Plant diversity management in cocoa agroforestry systems in West and Central Africa—Effects of markets and household needs. Agrofor. Syst. 2014, 88, 1021–1034. [Google Scholar] [CrossRef]
- Blaser, W.J.; Oppong, J.; Yeboah, E.; Six, J. Shade trees have limited benefits for soil fertility in cocoa agroforests. Agric. Ecosyst. Environ. 2017, 243, 83–91. [Google Scholar] [CrossRef]
- Schroth, G.; Harvey, C.A. Biodiversity conservation in cocoa production landscapes: An overview. Biodivers. Conserv. 2007, 16, 2237–2244. [Google Scholar] [CrossRef]
- Toppo, P.; Raj, A. Role of agroforestry in climate change mitigation. J. Pharmacogn. Phytochem. 2018, 7, 241–243. [Google Scholar]
- Vebrova, H.; Lojka, B.; Husband, T.P.; Zans, M.E.C.; Van Damme, P.; Rollo, A.; Kalousova, M. Tree diversity in cacao agroforests in San Alejandro, Peruvian Amazon. Agrofor. Syst. 2014, 88, 1101–1115. [Google Scholar] [CrossRef]
- Sari, R.R.; Saputra, D.D.; Hairiah, K.; Rozendaal, D.M.A.; Roshetko, J.M.; van Noordwijk, M. Gendered species preferences link tree diversity and carbon stocks in Cacao agroforest in Southeast Sulawesi, Indonesia. Land 2020, 9, 108. [Google Scholar] [CrossRef]
- Mbow, C.; Smith, P.; Skole, D.; Duguma, L.; Bustamante, M. Achieving mitigation and adaptation to climate change through sustainable agroforestry practices in africa. Curr. Opin. Environ. Sustain. 2014, 6, 8–14. [Google Scholar] [CrossRef]
- Merijn, B.; Simone, S. Biodiversity conservation in cacao agroforestry systems. In Biodiversity Conservation in Agroforestry Landscapes: Challenges and Opportunities; Simonetti, J.A., Ed.; Editorial Universitaria: Santiago, Chile, 2013; pp. 61–76. [Google Scholar]
- Daghela Bisseleua, H.B.; Fotio, D.; Yede; Missoup, A.D.; Vidal, S. Shade tree diversity, cocoa pest damage, yield compensating inputs and farmers’ net returns in West Africa. PLoS ONE 2013, 8, e56115. [Google Scholar] [CrossRef]
- Rajab, Y.; Leuschner, C.; Barus, H.; Tjoa, A.; Hertel, D. Cacao cultivation under diverse shade tree cover allows high carbon storage and sequestration without yield losses. PLoS ONE 2016, 11, e0149949. [Google Scholar] [CrossRef] [PubMed]
- Ruf, F.O. The myth of complex cocoa agroforests: The case of Ghana. Hum. Ecol. 2011, 39, 373–388. [Google Scholar] [CrossRef]
- Wessel, M.; Quist-Wessel, P.M.F. Cocoa production in West Africa, a review and analysis of recent developments. NJAS Wagening. J. Life Sci. 2015, 74–75, 1–7. [Google Scholar] [CrossRef]
- Anderson Bitty, E.; Bi, S.G.; Bene, J.C.K.; Kouassi, P.K.; Scott McGraw, W. Cocoa farming and primate extirpation inside Cote d’Ivoire’s protected areas. Trop. Conserv. Sci. 2015, 8, 95–113. [Google Scholar] [CrossRef]
- Kroeger, A.; Bakhtary, H.; Haupt, F.; Streck, C. Eliminating Deforestation from the Cocoa Supply Chain; World Bank: Washington, DC, USA, 2017; pp. 1–61. [Google Scholar]
- Bernard, F.; Minang, P.A. Community forestry and REDD+ in Cameroon: What future? Ecol. Soc. 2019, 24, 14. [Google Scholar] [CrossRef]
- Downie, R. Assessing the Growth Potential of Eastern Congo’s Coffee and Cocoa Sectors; Center for Strategic and International Studies: Washington, DC, USA, 2018; pp. 1–24. [Google Scholar]
- Jassogne, L.; van Asten, P.; De Beule, H. Cocoa: Driver of deforestation in the Democratic Republic of Congo? CGIAR: Copenhagen, Denmark, 2014; pp. 1–31. [Google Scholar]
- Schroth, G.; Jeusset, A.; da Silva Gomes, A.; Florence, C.T.; Coelho, N.A.P.; Faria, D.; Läderach, P. Climate friendliness of cocoa agroforests is compatible with productivity increase. Mitig. Adapt. Strateg. Glob. Chang. 2016, 21, 67–80. [Google Scholar] [CrossRef]
- Nowak, A.; Rosenstock, T.S.; Hammond, J.; Degrande, A.; Smith, E. Livelihoods of Households Living Near Yangambi Biosphere Reserve, Democratic Republic of Congo; Center for International Forestry Research: Bogor, Indonesia, 2019; pp. 1–10. [Google Scholar]
- Ngo Bieng, M.A.; Gidoin, C.; Avelino, J.; Cilas, C.; Deheuvels, O.; Wery, J. Diversity and spatial clustering of shade trees affect cacao yield and pathogen pressure in Costa Rican agroforests. Basic Appl. Ecol. 2013, 14, 329–336. [Google Scholar] [CrossRef]
- Kyale Koy, J.; Wardell, D.A.; Mikwa, J.; Kabuanga, J.M.; Monga Ngonga, A.M.; Oszwald, J.; Doumenge, C. Dynamics of déforestation dans la Réserve de biosphère de Yangambi (République Démocratique du Congo): Variabilité spatiale et temporelle au cours des 30 dernières années. Bois Forets Trop. 2019, 341, 15. [Google Scholar] [CrossRef]
- Moloba, Y.; Mobula, V.; Ntoto, R.; Mahungu, M. Dynamique socio-économique de l’adoption des variétés améliorées du manioc en République Démocratique du Congo (RDC): Cas des provinces de Kongo Central et la Tshopo. Eur. Sci. J. ESJ 2019, 15, 346–362. [Google Scholar]
- Kearsley, E.; Verbeeck, H.; Hufkens, K.; Van de Perre, F.; Doetterl, S.; Baert, G.; Beeckman, H.; Boeckx, P.; Huygens, D. Functional community structure of African monodominant Gilbertiodendron dewevrei forest influenced by local environmental filtering. Ecol. Evol. 2017, 7, 295–304. [Google Scholar] [CrossRef]
- Termote, C.; Van Damme, P.; Djailo, B.D. Eating from the wild: Turumbu, Mbole and Bali traditional knowledge on non-cultivated edible plants, District Tshopo, DR Congo. Genet. Resour. Crop Evol. 2011, 58, 585–618. [Google Scholar] [CrossRef]
- Moonen, P.C.J.; Verbist, B.; Schaepherders, J.; Bwama Meyi, M.; Van Rompaey, A.; Muys, B. Actor-based identification of deforestation drivers paves the road to effective REDD+ in DR Congo. Landuse Policy 2016, 58, 123–132. [Google Scholar] [CrossRef]
- Moonen, P.C.J.; Verbist, B.; Boyemba Bosela, F.; Norgrove, L.; Dondeyne, S.; Van Meerbeek, K.; Kearsley, E.; Verbeeck, H.; Vermeir, P.; Boeckx, P.; et al. Disentangling how management affects biomass stock and productivity of tropical secondary forests fallows. Sci. Total Environ. 2019, 659, 101–114. [Google Scholar] [CrossRef]
- Akkermans, T.; Van Rompaey, A.; Van Lipzig, N.; Moonen, P.; Verbist, B. Quantifying successional land cover after clearing of tropical rainforest along forest frontiers in the Congo Basin. Phys. Geogr. 2013, 34, 417–440. [Google Scholar] [CrossRef]
- Bamba, I.; Barima, Y.S.; Bogaert, J. Influence de la densité de la population sur la structure spatiale d’un paysage forestier dans le Bassin du congo en R. D. Congo. Trop. Conserv. Sci. 2010, 3, 31–44. [Google Scholar] [CrossRef]
- Béguin, H. Densité de population, productivité et développement agricole. Espace Géogr. 1974, 4, 267–272. [Google Scholar] [CrossRef]
- Ministère de l’Environnement Conservation de la Nature et Tourisme (MECNT). Potentiel REDD + de la RDC; MECNT: Kinshasa, Democratic Republic of the Congo, 2009; p. 66. [Google Scholar]
- Torres, B.; Maza, O.J.; Aguirred, P.; Und, L.H.; Günter, S. Contribution of traditional agroforestry to climate change adaptation in the Ecuadorian Amazon: The Chakra System. In Handbook of Climate Change Adaptation; Filho, W.L., Ed.; Springer: Berlin/Heidleberg, Germany, 2014; Volume 1, pp. 1–19. [Google Scholar]
- Zapfack, L.; Engwald, S.; Sonke, B.; Achoundong, G.; Madong, B.A. The impact of land conversion on plant biodiversity in the forest zone of Cameroon. Biodivers. Conserv. 2002, 11, 2047–2061. [Google Scholar] [CrossRef]
- Lejoly, J.L.; Djele, M.N.; Eerinck, D.G. Catalogue-Flore des Plantes Vasculaires des Districts de Kisangani et de la Tshopo (RD Congo); 4ème.; Taxonomania: Bruxelles, Belgium, 2012; pp. 1–313. [Google Scholar]
- Oke, D.O.; Odebiyi, K.A. Traditional cocoa-based agroforestry and forest species conservation in Ondo State, Nigeria. Agric. Ecosyst. Environ. 2007, 122, 305–311. [Google Scholar] [CrossRef]
- Sonwa, D.J.; Nkongmeneck, B.A.; Weise, S.F.; Tchatat, M.; Adesina, A.A.; Janssens, M.J.J. Diversity of plants in cocoa agroforests in the humid forest zone of Southern Cameroon. Biodivers. Conserv. 2007, 16, 2385–2400. [Google Scholar] [CrossRef]
- Abada Mbolo, M.M.; Zekeng, J.C.; Mala, W.A.; Fobane, J.L.; Djomo Chimi, C.; Ngavounsia, T.; Nyako, C.M.; Menyene, L.F.E.; Tamanjong, Y.V. The role of cocoa agroforestry systems in conserving forest tree diversity in the Central region of Cameroon. Agrofor. Syst. 2016, 90, 577–590. [Google Scholar] [CrossRef]
- Dawoe, K.; Asante, W.; Acheampong, E.; Bosu, P. Shade tree diversity and aboveground carbon stocks in Theobroma cacao agroforestry systems: Implications for REDD+ implementation in a West African cacao landscape. Carbon Balance Manag. 2016, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Jadán, O.; Cifuentes, M.; Torres, B.; Selesi, D.; Veintimilla, D.; Günter, S. Influence of tree cover on diversity, carbon sequestration and productivity of cocoa systems in the Ecuadorian Amazon. Bois Forets Trop. 2015, 325, 35–47. [Google Scholar] [CrossRef]
- Noiha, V.N.; Zapfack, L.; Mbade, L.F. Biodiversity management and plant dynamic in a Cocoa Agroforest (Cameroon). Int. J. Plant Soil Sci. 2015, 6, 101–108. [Google Scholar] [CrossRef]
- Somarriba, E.; Cerda, R.; Orozco, L.; Cifuentes, M.; Dávila, H.; Espin, T.; Mavisoy, H.; Ávila, G.; Alvarado, E.; Poveda, V.; et al. Carbon stocks and cocoa yields in agroforestry systems of Central America. Agric. Ecosyst. Environ. 2013, 173, 46–57. [Google Scholar] [CrossRef]
- Hervé, B.; Vidal, S. Plant biodiversity and vegetation structure in traditional cocoa forest gardens in southern Cameroon under different management. Biodivers. Conserv. 2008, 17, 1821–1835. [Google Scholar]
- Sullivan, M.J.P.; Talbot, J.; Lewis, S.L.; Phillips, O.L.; Qie, L.; Begne, S.K.; Chave, J.; Cuni-sanchez, A.; Hubau, W.; Lopez, G.; et al. Diversity and carbon storage across the tropical forest biome. Sci. Rep. 2017, 7–39102, 1–12. [Google Scholar] [CrossRef]
- Shahid, M.; Joshi, S.P. Relationship between tree species diversity and carbon stock density in moist deciduous forest of Western Himalayas, India. J. For. Environ. Sci. 2017, 33, 39–48. [Google Scholar] [CrossRef]
- Grall, J.; Coic, N. Synthèse des Méthodes D’évaluation de la Qualité du Benthos en Milieu Côtier Préliminaire; Institut Universitaire Européen de la Mer–Université de Bretagne Occidentale Laboratoire des Sciences de l’Environnement MARin: Bretagne, France, 2006; pp. 1–90. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; pp. 1–3636. [Google Scholar]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Kindt, R.; Coe, R. Tree Diversity Analysis. A Manual and Software for Common Statistical Methods for Ecological and Biodiversity Studies; World Agroforestry Centre (ICRAF): Nairobi, Kenya, 2005; ISBN 92-9059-179-X. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R package version 2.5-6. Available online: https://CRAN.R-project.org/package=vegan (accessed on 18 July 2020).
- Temgoua, L.F.; Dongmo, W.; Nguimdo, V.; Nguena, C. Diversité ligneuse et stock de carbone des systèmes agroforestiers à base de cacaoyers à l’Est Cameroun: Cas de la forêt d’enseignement et de recherche de l’Université de Dschang. J. Appl. Biosci. 2018, 122, 12274–12286. [Google Scholar] [CrossRef]
- Zapfack, L.; Chimi Djomo, C.; Noiha Noumi, V.; Zekeng, J.C.; Meyan-ya Daghela, G.R.; Tabue Mbobda, R. Correlation between associated trees, cocoa trees and carbon stocks potential in cocoa agroforests of Southern Cameroon. Sustain. Environ. 2016, 1, 71–84. [Google Scholar]
- Salazar, J.C.S.; Bieng, M.A.N.; Melgarejo, L.M.; Di Rienzo, J.A.; Casanoves, F. First typology of cacao (Theobroma cacao L.) systems in Colombian Amazonia, based on tree species richness, canopy structure and light availability. PLoS ONE 2018, 13, e0191003. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.Y.A.; Kpangui, K.B.; Vroh, B.T.A.; Ouattara, D. Pratiques culturales, valeurs d’usage et perception des paysans des espèces compagnes du cacaoyer dans des agroforêts traditionnelles au centre de la Côte d’Ivoire. Rev. d’ethnoécologie 2016, 9, 1–19. [Google Scholar] [CrossRef]
- Sonwa, D.; Weise, L.; Chimi Djomo, C.; Kabelong Banoho, L.-P.; Forbi Preasious, F.; Tsopmejio Temfack, I.; Tajeukem Vice, C.; Ntonmen Yonkeu, A.; Tabue Mboba, R.; Nasang, J. Carbon storage potential of cacao agroforestry systems of different age and management intensity. Clim. Dev. 2018, 1–12. [Google Scholar] [CrossRef]
- Santhyami, S.; Basukriadi, A.; Patria, M.P.; Abdulhadi, R. The comparison of aboveground C-stock between cocoa-based agroforestry system and cocoa monoculture practice in West Sumatra, Indonesia. Biodiversitas 2018, 19, 472–479. [Google Scholar] [CrossRef]
- Häger, A.; Fernández Otárola, M.; Stuhlmacher, M.F.; Acuña Castillo, R.; Contreras Arias, A. Effects of management and landscape composition on the diversity and structure of tree species assemblages in coffee agroforests. Agric. Ecosyst. Environ. 2014, 199, 43–51. [Google Scholar] [CrossRef]
- Bisseleua, D.H.B.; Missoup, A.D.; Vidal, S. Biodiversity conservation, ecosystem functioning, and economic incentives under cocoa agroforestry intensification. Conserv. Biol. 2009, 23, 1176–1184. [Google Scholar] [CrossRef]
- Omatoko, J.; Nshimba, H.; Bogaert, J.; Lejoly, J.; Shutsha, R.; Shaumba, J.P.; Asimonyio, J.; Ngbolua, K.N. Etudes floristique et structurale des peuplements sur sols argileux à Pericopsis elata et sableux à Julbernardia seretii dans la forêt de plaine d’UMA en République Démocratique du Congo. Int. J. Innov. Appl. Stud. 2015, 13, 452–463. [Google Scholar]
- Kambale, J.K.; Asimonyio, J.A.; Shutsha, R.E.; Katembo, E.W.; Tsongo, J.M.; Kavira, P.K. Etudes floristique et structurale des forêts dans le domaine de chasse de Rubi-Télé (Province de Bas-Uélé, République Démocratique du Congo). Int. J. Innov. Sci. Res. 2016, 24, 309–321. [Google Scholar]
- Koubouana, F.; Ifo, S.A.; Ndzai, S.F.; Stoffenne, B. Étude comparative d ’ une forêt primaire et d ’ une forêt dégradée au Nord de la République du Congo par référence à la structure des forêts tropicales humides. Rev. Sci. Tech. Forêt Environ. du Bassin du Congo 2018, 11, 11–25. [Google Scholar]
- Sonwa, D.; Weise, S.F.; Tchatat, M.; Nkongmeneck, B.; Adesina, A.A.; Ndoye, O.; Gockowski, J. Rôle des agroforêts cacao dans la foresterie paysanne et communautaire au Sud-Cameroun. Réseau For. Pour Développement Rural, document du réseau 2001, 25g, 1–11. [Google Scholar]
- Asare, R.; Asare, R.A.; Asante, W.A.; Markussen, B.; RÆbild, A. Influences of shading and fertilization on on-farm yields of cocoa in Ghana. Exp. Agric. 2017, 53, 416–431. [Google Scholar] [CrossRef]
- Bohlman, S.A.; Bourg, N.A.; Brinks, J.; Bunyavejchewin, S.; Butt, N.; Chisholm, R.A.; Muller-Landau, H.C.; Rahman, K.A.; Bebber, D.P.; Bin, Y.; et al. Scale-dependent relationships between tree species richness and ecosystem function in forests. J. Ecol. 2013, 101, 1214–1224. [Google Scholar]
- Sagar, R.; Singh, J.S. Tree density, basal area and species diversity in a disturbed dry tropical forest of northern India: Implications for conservation. Environ. Conserv. 2006, 33, 256–262. [Google Scholar] [CrossRef]
- Blanc, L.; Florès, O.; Molino, J.F.; Gourlet-Fleury, S.; Sabatier, D. Diversité spécifique et regroupement d’espéces arborescentes en Forêt Guyanaise. Rev. For. Fr. 2003, 55, 131–146. [Google Scholar] [CrossRef]
- Asase, A.; Ofori-frimpong, K.; Ekpe, P. Impact of cocoa farming on vegetation in an agricultural landscape in Ghana. Afr. J. Ecol. 2009, 48, 338–346. [Google Scholar] [CrossRef]
- Middendorp, R.; Vanacker, V.; Lambin, E. Impacts of shaded agroforestry management on carbon sequestration, biodiversity and farmers income in cocoa production landscapes. Landsc. Ecol. 2018, 33, 1953–1974. [Google Scholar] [CrossRef]
- Asigbaase, M.; Sjogersten, S.; Lomax, B.; Dawoe, E. Tree diversity and its ecological importance value in organic and conventional cocoa agroforests in Ghana. PLoS ONE 2019, 14, e0210557. [Google Scholar] [CrossRef]
- Niether, W.; Armengot, L.; Andres, C.; Schneider, M.; Gerold, G. Shade trees and tree pruning alter throughfall and microclimate in cocoa (Theobroma cacao L.) production systems. Ann. For. Sci. 2018, 75, 38. [Google Scholar] [CrossRef]
- Redd-RDC. Mambasa Pays: République Démocratique Du Congo, Rapport D’évaluation du Projet; Banque Africaine de Développement: Kinshasa, Democratic Republic of the Congo, 2010; pp. 1–18. [Google Scholar]
- Mason, J.J.; Asare, R.; Cenamo, M.; Soares, P.; Carrero, G.; Murphy, A.J.; Bandari, C. Ghana Cocoa REDD+ Programme: Draft Implementation Report; Ghana cocoa Reed+ programme: Accra, Ghana, 2016; pp. 1–77. [Google Scholar]
- Carodenuto, S.; Gromko, D.; Chia, E.L. Zero deforestation cocoa in Cameroon: Private sector engagement to support Reducing Emissions from Deforestation and forest Degradation (REDD+). GIZ-ProPFE Policy Br. 2017, 4, 1–3. [Google Scholar]
- Fernandes, C.D.A.F.; Matsumoto, S.N.; Fernandes, V.S. Carbon stock in the development of different designs of biodiverse agroforestry systems. Rev. Bras. Eng. Agrícola Ambient. 2018, 22, 720–725. [Google Scholar] [CrossRef]
- Sonwa, D.J.; Weise, S.F.; Bernard, A.; Nkongmeneck, M.T.; Janssens, M.J.J. Profiling carbon storage/stocks of cocoa agroforests in the forest landscape of southern Cameroon. In Agroforestry: Anecdotal to Modern Science; Dagar, J., Tewari, V., Eds.; Springer: Singapore, 2017; pp. 739–752. [Google Scholar]
| Indices/Index | Zone A (n = 6 Agroforests) | Zone B (n = 11 Agroforests) | Zone C (n = 3 Agroforests) | Zone D (n = 5 Agroforests) | Whole Region (n = 25 Agroforests) | p-Value |
|---|---|---|---|---|---|---|
| Species Richness | 5.83 (±2.48) | 12.55 (±8.71) | 20.67 (±14.29) | 16 (±6.63) | 12.6 (±8.9) | 0.0725 |
| Shannon-Wiener index | 0.99 (±0.5) | 1.65 (±1.2) | 2.14 (±1.51) | 2.3 (±0.54) | 1.68 (±1.05) | 0.1836 |
| Piélou’s equitability | 0.56 (±0.19) | 0.6 (±0.37) | 0.69 (±0.38) | 0.85 (±0.09) | 0.65 (±0.3) | 0.2828 |
| Simpson’s index | 0.45 (±0.22) | 0.58 (±0.39) | 0.68 (±0.45) | 0.83 (±0.12) | 0.61 (±0.33) | 0.2142 |
| Indices | Model F (n = 10 Agroforests) | Model FP (n = 5 Agroforests) | Model P (n = 10 Agroforests) | Whole region (n = 25 Agroforests) | p-Value |
|---|---|---|---|---|---|
| Species Richness | 16.4 (±6.19) b | 17.4 (±10.92) b | 6.4 (±6.96) a | 12.6 (±8.9) | 0.011 |
| Shannon-Wiener index | 2.33 (±0.63) b | 2.29 (±0.78) b | 0.73 (±0.79) a | 1.68 (±1.05) | 0.00011 |
| Piélou’s equitability | 0.85 (±0.08) b | 0.83 (±0.11) b | 0.36 (±0.25) a | 0.65 (±0.3) | 0.002 |
| Simpson’s index | 0.83 (±0.14) b | 0.81 (±0.16) b | 0.29 (±0.27) a | 0.61 (±0.33) | 8.50 × 10−6 |
| Species | Local Names | Main Uses | Zone A (n = 6 Agroforests) | Zone B (n= 11 Agroforests) | Zone C (n = 3 Agroforests) | Zone D (n = 5 Agroforests) | Whole Region (25 Agroforests) |
|---|---|---|---|---|---|---|---|
| Elaeis guineensis | Adjagale | Edible | 110 | 179 | 51 | 26 | 366 |
| Musanga cecropioides | Kombo | Timber | - | - | 10 | 35 | 45 |
| Pycnanthus angolensis | Gbotugbu | Timber | - | 35 | - | - | 35 |
| Ficus exasperata | Kasage | Medicinal | 3 | 24 | - | - | 27 |
| Pseudospondias microcarpa | Bume | Medicinal | - | 25 | - | - | 25 |
| Maesopsis eminii | Ngana | Medicinal | - | - | 12 | 6 | 18 |
| Petersianthus macrocarpus | Angbeche | Caterpillar | - | 17 | - | - | 17 |
| Carapa procera | Mbindo | Medicinal | - | - | 14 | - | 14 |
| Macaranga monandra | Abou chumbuge | Timber | - | - | 13 | - | 13 |
| Tetrorchidium didymostemon | Aboligi | Timber | - | - | - | 9 | 9 |
| Persea americana | Savoka | Edible | 8 | - | - | - | 8 |
| Bridelia atroviridis | Bubu | Caterpillar | - | - | - | 7 | 7 |
| Dacryodes edulis | Angboka | Edible | 5 | - | - | - | 5 |
| Senna siamea | Ngbangaolaya | Medicinal | 4 | - | - | - | 4 |
| Total of top five species | 130 | 280 | 100 | 83 | 593 | ||
| Total of all species in study area | 155 | 476 | 187 | 178 | 996 | ||
| Percentage of top five species | 83.87 | 58.82 | 53.48 | 46.63 | 59.54 |
| Species | Zone A (n = 6 Agroforests) | Zone B (n = 11 Agroforests) | Zone C (n = 3 Agroforests) | Zone D (n = 5 Agroforests) | Whole Region (n = 25 Agroforests) |
|---|---|---|---|---|---|
| Musanga cecropioides | 2 | 10 | 10 | 35 | 57 |
| Pycnanthus angolensis | 0 | 35 | 3 | 0 | 38 |
| Ficus exasperata | 3 | 24 | 1 | 1 | 29 |
| Petersianthus macrocarpus | 0 | 17 | 6 | 4 | 27 |
| Zanthoxylum gilletii | 0 | 12 | 8 | 6 | 26 |
| Macaranga monandra | 0 | 2 | 13 | 4 | 19 |
| Bridelia atroviridis | 0 | 9 | 0 | 7 | 16 |
| Macaranga spinosa | 0 | 5 | 4 | 6 | 15 |
| Albizia gummifera | 0 | 10 | 1 | 1 | 12 |
| Albizia adianthifolia | 0 | 4 | 2 | 2 | 8 |
| Alstonia boonei | 1 | 4 | 0 | 1 | 6 |
| Albizia ferruginea | 0 | 3 | 0 | 2 | 5 |
| Canarium schweinfurthii | 0 | 0 | 3 | 0 | 3 |
| Croton haumanianus | 0 | 2 | 0 | 0 | 2 |
| Ficus elastica | 0 | 1 | 1 | 0 | 2 |
| Ficus mucuso | 1 | 0 | 0 | 1 | 2 |
| Ficus wildemaniana | 0 | 1 | 0 | 1 | 2 |
| Zanthoxylum lemairei | 0 | 1 | 0 | 1 | 2 |
| Harungana madagascariensis | 0 | 0 | 0 | 1 | 1 |
| Total of suitable plants | 7 | 140 | 52 | 73 | 272 |
| Total of all plants in study area | 155 | 476 | 187 | 178 | 996 |
| Percentage of suitable plants | 4.52 | 29.41 | 27.81 | 41.01 | 27.31 |
| Species | Zone A (n = 6 Agroforests) | Zone B (n = 11 Agroforests) | Zone C (n = 3 Agroforests) | Zone D (n = 5 Agroforests) | Whole Region (n = 25 Agroforests) |
|---|---|---|---|---|---|
| Pseudospondias microcarpa | 1 | 25 | 0 | 0 | 26 |
| Myrianthus arboreus | 0 | 16 | 0 | 0 | 16 |
| Carapa procera | 0 | 1 | 14 | 0 | 15 |
| Dacryodes edulis | 5 | 3 | 1 | 3 | 12 |
| Pterocarpus soyauxii | 0 | 1 | 4 | 6 | 11 |
| Rauvolfia vomitoria | 0 | 4 | 1 | 3 | 8 |
| Trichilia gilgiana | 0 | 4 | 2 | 2 | 8 |
| Vernonia conferta | 0 | 0 | 0 | 6 | 6 |
| Desplatsia dewevrei | 0 | 4 | 1 | 0 | 5 |
| Oncoba welwitschii | 0 | 1 | 0 | 4 | 5 |
| Blighia welwitschii | 0 | 2 | 0 | 1 | 3 |
| Synsepalum subcordatum | 0 | 0 | 3 | 0 | 3 |
| Uapaca guineensis | 1 | 0 | 1 | 1 | 3 |
| Barteria fistulosa | 0 | 1 | 0 | 1 | 2 |
| Canthium subcordatum | 0 | 0 | 1 | 1 | 2 |
| Cola lateritia | 0 | 2 | 0 | 0 | 2 |
| Anonidium mannii | 0 | 0 | 1 | 0 | 1 |
| Cola marsupium | 0 | 0 | 1 | 0 | 1 |
| Drypetes gossweileri | 0 | 0 | 1 | 0 | 1 |
| Gilbertiodendron dewevrei | 0 | 0 | 0 | 1 | 1 |
| Homalium longistylum | 0 | 1 | 0 | 0 | 1 |
| Panda oleosa | 0 | 0 | 0 | 1 | 1 |
| Total of unsuitable plants | 7 | 65 | 31 | 30 | 133 |
| Total of all plants in study area | 155 | 476 | 187 | 178 | 996 |
| Percentage of unsuitable plants | 4.52 | 13.66 | 16.58 | 16.85 | 13.35 |
| Main Uses | Zone A (n = 6 Agroforests) | Zone B (n = 11 Agroforests) | Zone C (n = 3 Agroforests) | Zone D (n = 5 Agroforests) | Whole Region (n = 25 Agroforests) | p-Value |
|---|---|---|---|---|---|---|
| Edible | 85.33 (±32.36) | 78.91 (±49.48) | 77.33 (±85.54) | 30.4 (±39.76) | 70.56 (±50.4) | 0.265 |
| Hosts for caterpillars | 0.67 (±1.63) | 12 (±14.86) | 9.33 (±6.11) | 12.8 (±10.35) | 9.12 (±11.75) | 0.07834 |
| Medicinal | 11.33 (±8.55) | 38.91 (±37.23) | 76 (±79.9) | 25.6 (±17.57) | 34.08 (±39.38) | 0.115 |
| Timber | 1.33 (±2.07) a | 13.45 (±12.93) a | 53.33 (±52.2) b | 52 (±18.76) b | 23.04 (±28.38) | 0.002034 |
| Others (minor or no known uses) | 4.67 (±7.34) | 29.82 (±42.61) | 33.33 (±34.02) | 21.6 (±14.59) | 22.56 (±31.91) | 0.2558 |
| Total | 103.33 (±37.64) a | 173.09 (±56.23) b | 249.33 (±13.44) b | 142.4 (±7.45) ab | 159.36 (±71.96) | 0.01405 |
| Main Uses | Model F (n = 10 Agroforests) | Model FP (n = 5 Agroforests) | Model P (n = 10 Agroforests) | Whole Region (n = 25 Agroforests) | p-Value |
|---|---|---|---|---|---|
| Edible | 29.6 (±25.24) a | 50.4 (±35.05) a | 121.6 (±24.6) b | 70.56 (±50.4) | 0.0001507 |
| Tree-hosting caterpillars | 15.6 (±13.91) a | 11.2 (±11.1) ab | 1.6 (±2.8) b | 9.12 (±11.75) | 0.01258 |
| Medicinal | 48.8 (±31.88) a | 52.8 (±64.96) ab | 10 (±12.82) b | 34.08 (±39.38) | 0.007181 |
| Timber | 30.4 (±25.38) ab | 39.2 (±43.58) a | 7.6 (±13.79) b | 23.04 (±28.38) | 0.04679 |
| Others (minor or no known uses) | 37.6 (±41.06) a | 28.8 (±25.2) ab | 4.4 (±10.41) b | 22.56 (±31.91) | 0.0071 |
| Whole region | 162 (±77.98) | 182.4 (±113.3) | 145.2 (±38.69) | 159.36 (±71.96) | 0.653 |
| Cocoa Agroforest Structure | Zone A (n = 6 Agroforests) | Zone B (n = 11 Agroforests) | Zone C (n =3 Agroforests) | Zone D (n = 5 Agroforests) | Whole Region (n = 25 Agroforests) | p-Value |
|---|---|---|---|---|---|---|
| Density of associated plants (n/ha) | 103.33 (±37.64) a | 173.09 (±56.23) b | 249.33 (±113.44) b | 142.4 (±7.45) ab | 159.36 (±71.96) | 0.01405 |
| Basal area of associated plants (m2/ha) | 5.59 (±5.28) | 10.95 (±9.4) | 15.36 (±3.35) | 8.26 (±4.66) | 9.65 (±7.52) | 0.273 |
| Density of (n/ha) of cocoa | 913.33 (±213.07) | 959.27 (±154.07) | 746.67 (±78.93) | 789.6 (±92.81) | 888.8 (±168.24) | 0.107 |
| Basal area of cocoa | 10.91 (±2.63) | 9.14 (±1.94) | 8.2 (±2.04) | 9.68 (±3.9) | 9.56 (±2.58) | 0.453 |
| Density of whole region | 1016.67 (±77.94) | 1132.36(±187.62) | 996 (±34.64) | 932 (±77.82) | 1048.16 (±70.36) | 0.133 |
| Basal area of whole region | 13.13 (±3.04) | 19.19 (±7.73) | 17.28 (±2) | 18.08 (±3.13) | 17.28 (±5.91) | 0.248 |
| Cocoa Agroforest Structures | Model F (n = 10 Agroforests) | Model FP (n = 5 Agroforests) | Model P (n = 10 Agroforests) | Whole Region (n = 25 Agroforests) | p-Value |
|---|---|---|---|---|---|
| Density of associated plants (n/ha) | 162 (±77.98) | 182.4 (±113.3) | 145.2 (±38.69) | 159.36 (±71.96) | 0.653 |
| Basal area of associated plants (m2/ha) | 12.38 (±9.48) | 9.87 (±4.83) | 6.81 (±5.77) | 9.65 (±7.52) | 0.263 |
| Density (n/ha) cocoa | 984 (±200.55) | 824 (±109.8) | 826 (±114.53) | 888.8 (±168.24) | 0.0629 |
| Basal area of cocoa | 9.6 (±2.97) | 9.81 (±3.45) | 9.4 (±1.91) | 9.56 (±2.58) | 0.961 |
| Density of whole region | 1146 (±217.52) | 1006.4 (±50.72) | 971.2 (±103.67) | 1048.16 (±70.36) | 0.0525 |
| Basal area of whole region | 22.99 (±3.86) a | 16.22 (±2.77) b | 12.1 (±2.85) b | 17.28 (±5.91) | 9.62 × 10−7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batsi, G.; Sonwa, D.J.; Mangaza, L.; Ebuy, J.; Kahindo, J.-M. Biodiversity of the Cocoa Agroforests of the Bengamisa-Yangambi Forest Landscape in the Democratic Republic of the Congo (DRC). Forests 2020, 11, 1096. https://doi.org/10.3390/f11101096
Batsi G, Sonwa DJ, Mangaza L, Ebuy J, Kahindo J-M. Biodiversity of the Cocoa Agroforests of the Bengamisa-Yangambi Forest Landscape in the Democratic Republic of the Congo (DRC). Forests. 2020; 11(10):1096. https://doi.org/10.3390/f11101096
Chicago/Turabian StyleBatsi, Germain, Denis Jean Sonwa, Lisette Mangaza, Jérôme Ebuy, and Jean-Marie Kahindo. 2020. "Biodiversity of the Cocoa Agroforests of the Bengamisa-Yangambi Forest Landscape in the Democratic Republic of the Congo (DRC)" Forests 11, no. 10: 1096. https://doi.org/10.3390/f11101096
APA StyleBatsi, G., Sonwa, D. J., Mangaza, L., Ebuy, J., & Kahindo, J.-M. (2020). Biodiversity of the Cocoa Agroforests of the Bengamisa-Yangambi Forest Landscape in the Democratic Republic of the Congo (DRC). Forests, 11(10), 1096. https://doi.org/10.3390/f11101096





