Effects of Canopy Microclimate on Chinese Chestnut (Castanea mollissima Blume) Nut Yield and Quality
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site Description and Plant Materials
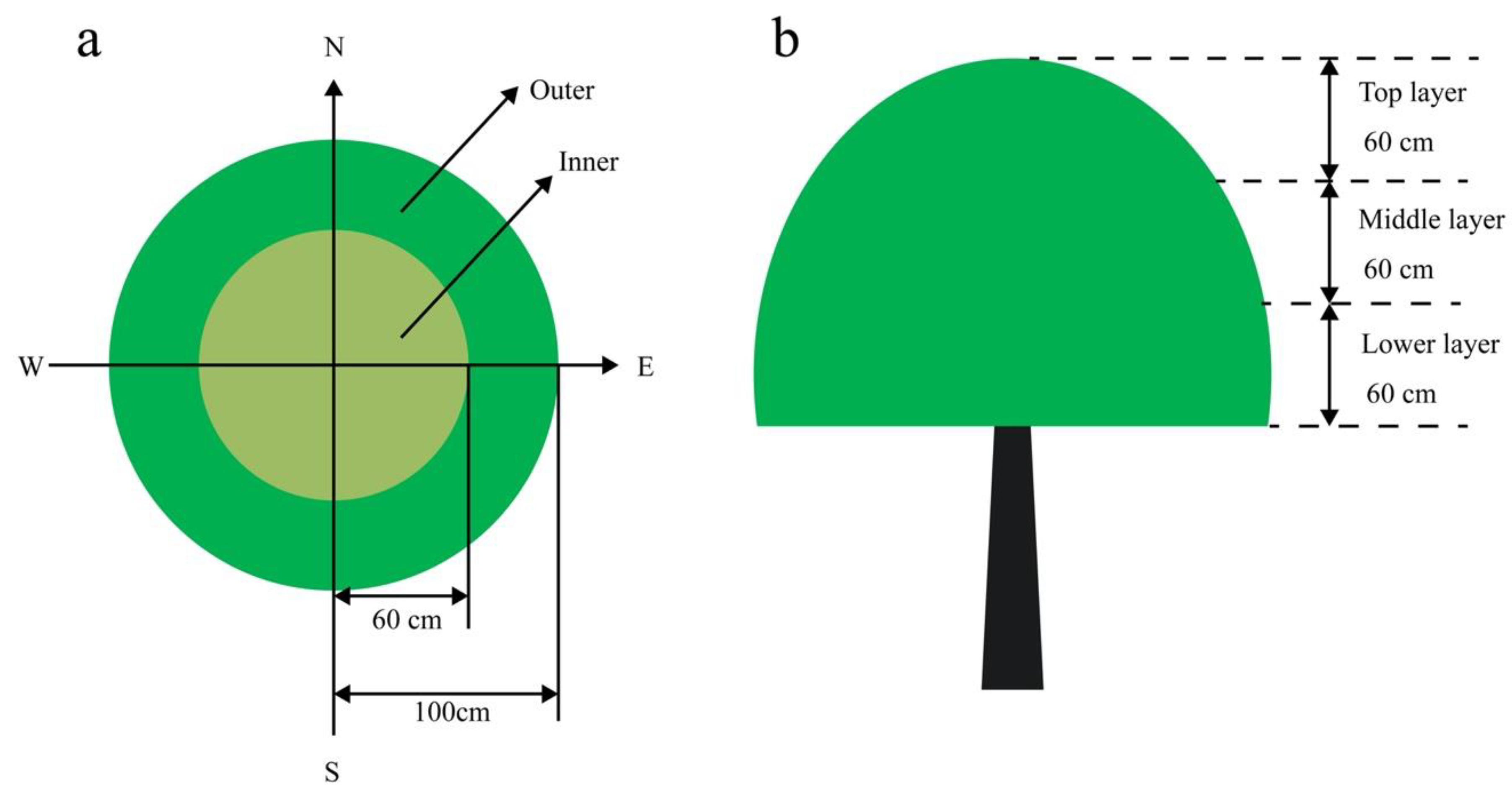
2.2. Canopy Microclimate
2.3. Distribution of Different Branch Types
2.4. Flesh Nut Yield and Quality
2.5. Statistical Analysis
3. Results
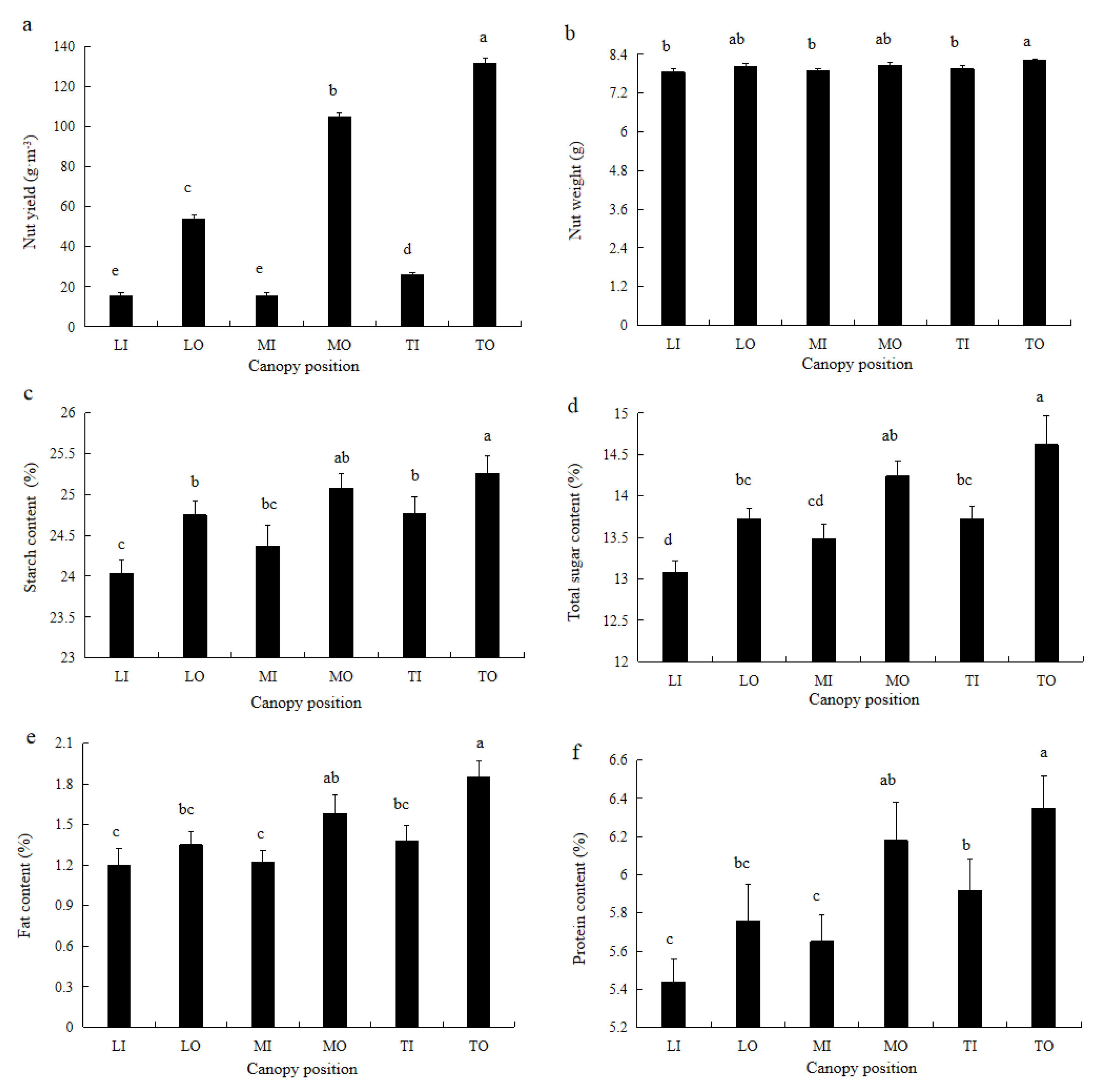
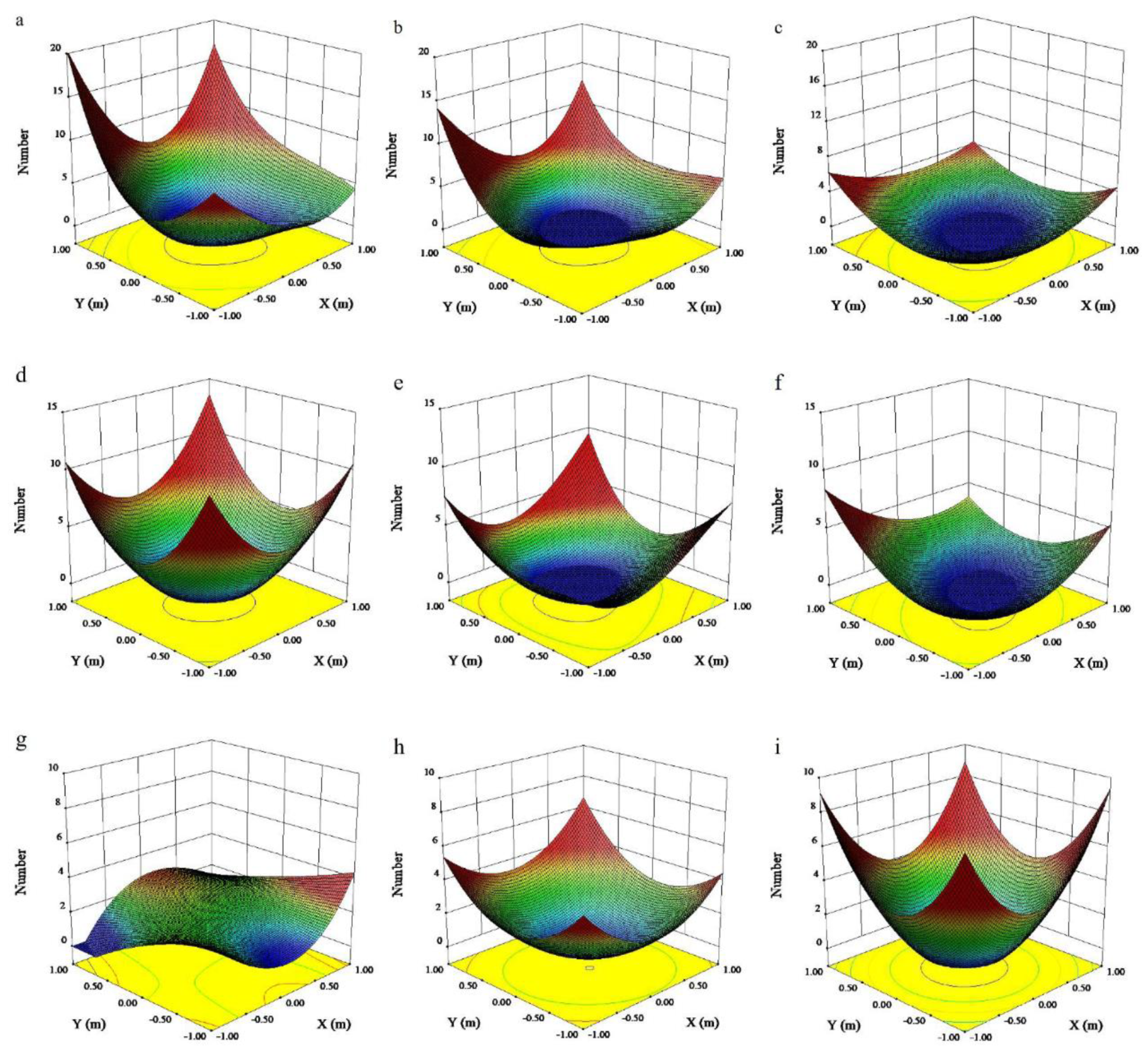
3.1. Nut Yield and Quality at Different Positions within the Canopy
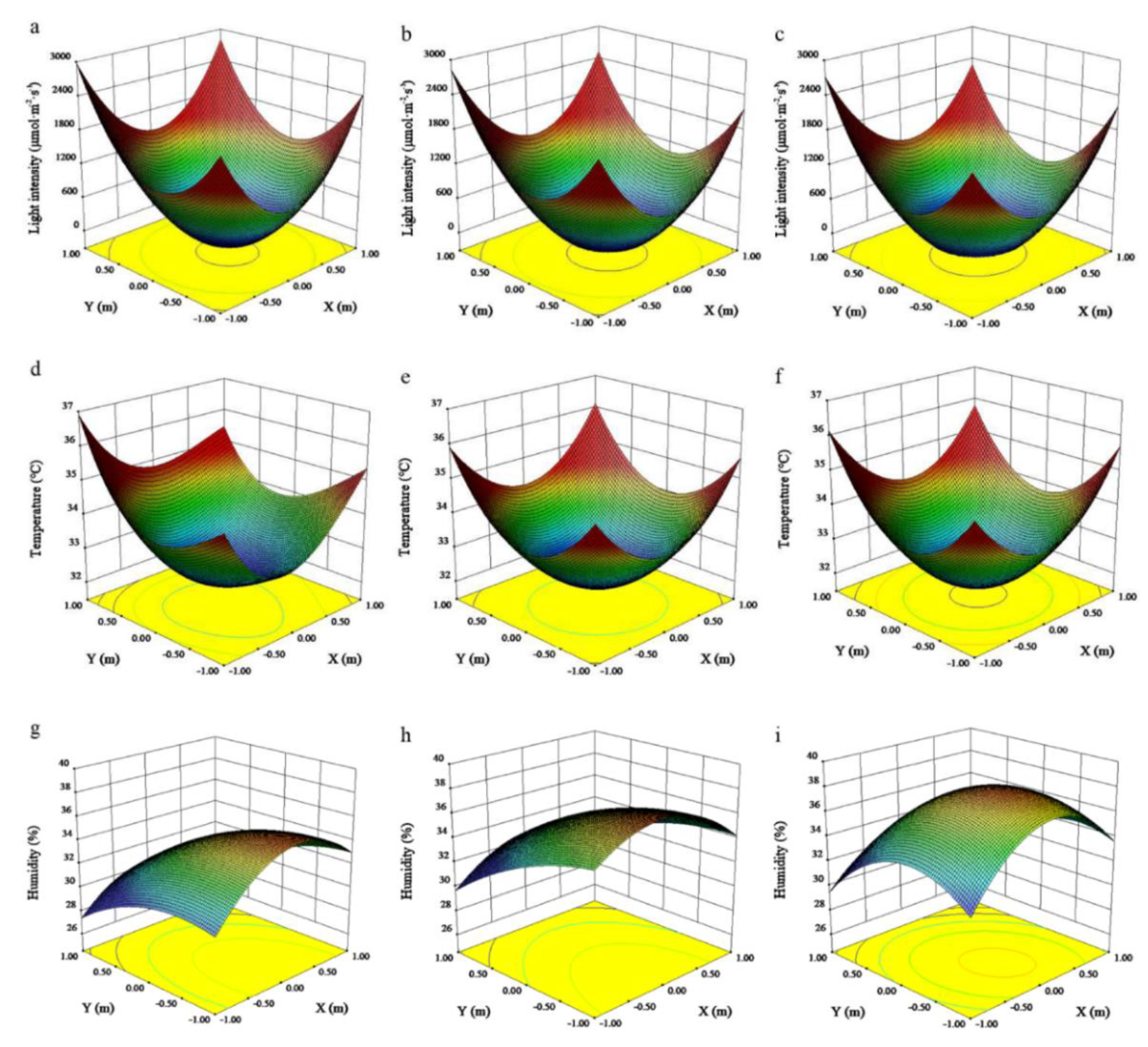
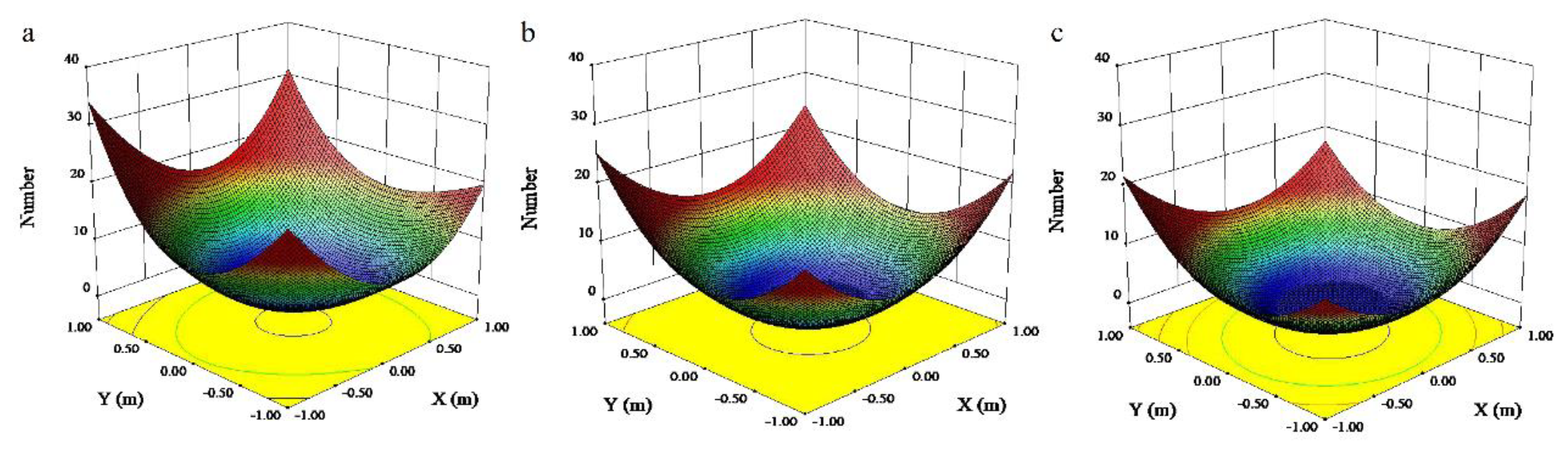
3.2. Microclimate Distribution within the Canopy
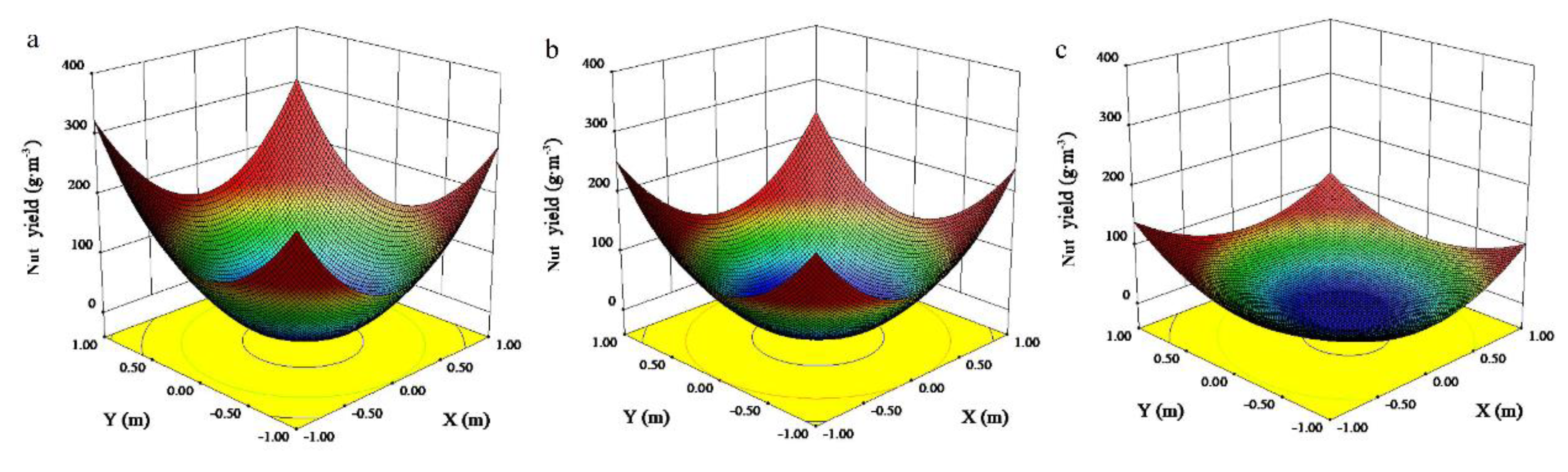
3.3. Canopy Microclimate Effects on Nut Yield
3.4. Distribution of Different Branch Types within the Canopy
3.5. Relationships between the Canopy Microclimate and Nut Yield and Quality
4. Discussion
4.1. Yield and Quality of Nuts from Different Positions within the Canopy
4.2. Microclimate Distribution within the Canopy
4.3. Distribution of Different Branch Types within the Canopy
4.4. Relationships between the Canopy Microclimate and Nut Yield and Quality
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Yang, Y.; Pan, X.; Wang, G. China Food Composition, 2nd ed.; Peking University Medical Press: Beijing, China, 2015. [Google Scholar]
- Xia, R.; Xu, J.; Li, G.; Ma, M. Flowering and fruiting habits and sexual expression in chestnut (Castanea mollissima BL.). J. Wuhan Bot. Res. 1998, 16, 154–158. [Google Scholar]
- Miller, A.; Woeste, K.; Anagnostakis, S.; Jacobs, D. Exploration of a rare population of Chinese chestnut in North America: Stand dynamics, health and genetic relationships. AoB Plants 2014, 6, 11–15. [Google Scholar] [CrossRef]
- Cao, X.; Li, Z.; Zhang, Q.; Qin, L.; Xing, Y. Comprehensive evaluation of resistant starch in different Chinese chestnut varieties (lines). J. Chin. Cereals Oils Assoc. 2019, 34, 39–46. [Google Scholar]
- Zhu, C.; Ji, F.; Geng, G. Fuzzy comprehensive evaluation on important agronomic characterisitcs of Chinese chestnut cultivars. Nonwood For. Res. 2017, 35, 13–21. [Google Scholar] [CrossRef]
- Xiong, H.; Guo, S.; Peng, J.; Zhou, F.; Li, G.; Lv, W. Effect of tree structure on light distribution and yield and quality of Castanea mollissima BL. J. Nanjing For. Univ. 2014, 38, 68–74. [Google Scholar] [CrossRef]
- Huang, S.; Shen, C.; Yu, Y.; Qu, S. Discussion on climatic and ecological regionalization of Chinese chestnut. Meteorol. J. Hubei 1998, 23–25. [Google Scholar]
- Jiang, X.; Gong, B.; Liu, Q.; Chen, X.; Wu, K.; Deng, Q.; Tang, D. Phenotypic diversity of important agronomic traits of local cultivars of Chinese chestnut. Acta Hort. Sin. 2014, 41, 641–652. [Google Scholar] [CrossRef]
- Li, B.; Guo, S. Advances in research on cultivation techniques for improving yield of Chinese chestnut. J. Hebei For. Coll. 1992, 7, 339–345. [Google Scholar]
- Jiang, G. Cultivation Techniques for Early Fruit Yield of Chinese Chestnut; China Forestry Publishing House: Beijing, China, 1995. [Google Scholar]
- Kumar, M.; Rawat, V.; Rawat, J.; Tomar, Y. Effect of pruning intensity on peach yield and fruit quality. Sci. Hortic. 2010, 125, 218–221. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, R.; Patel, V. Influence of pruning intensity on flowering, fruit yields and floral malformation in three mango cultivars planted under high density. Indian J. Hortic. 2010, 67, 84–89. [Google Scholar]
- Sharma, R.; Singh, R. Pruning intensity modifies canopy microclimate, and influences sex ratio, malformation incidence and development of fruited panicles in ‘Amrapali’ mango (Mangifera indica, L.). Sci. Hortic. 2016, 109, 118–122. [Google Scholar] [CrossRef]
- Sharma, R.; Singh, R. Effect of pruning intensity on light penetration and leaf physiology in Amrapali mango trees under high-density planting. Trop. Sci. 2006, 46, 16–19. [Google Scholar] [CrossRef]
- Jones, H. Plants and Microclimate: A Quantitative Approach to Environmental, 3rd ed.; Cambridge University Press: Cambridge, UK, 2014; p. 407. [Google Scholar]
- Matloobi, M. Light Harvesting and Photosynthesis by the Canopy. In Advances in Photosynthesis—Fundamental Aspects; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- He, F.; Wang, F.; Wei, Q.; Wang, X.; Zhang, Q. Relationships between the distribution of relative canopy light intensity and the peach yield and quality. Agric. Sci. China 2008, 7, 297–302. [Google Scholar] [CrossRef]
- Zhang, J.; Serra, S.; Leisso, R.; Musacchi, S. Effect of light microclimate on the quality of ‘d’Anjou’ pears in mature open-center tree architecture. Biosyst. Eng. 2016, 141, 1–11. [Google Scholar] [CrossRef]
- Niinemets, Ü.; Valladares, F. Photosynthetic acclimation to simultaneous and interacting environmental stresses along natural light gradients: Optimality and constraints. Plant Biol. 2004, 6, 254–268. [Google Scholar] [CrossRef]
- Bailey, B.; Stoll, R.; Pardyjak, E.; Miller, N. A new three-dimensional energy balance model for complex plant canopy geometries: Model development and improved validation strategies. Agric. For. Meteorol. 2016, 218–219, 146–160. [Google Scholar] [CrossRef]
- Hamadziripi, E.; Theron, K.; Muller, M.; Steyn, W. Apple compositional and peel color differences resulting from canopy microclimate affect consumer preference for eating quality and appearance. Hortscience 2014, 49, 384–392. [Google Scholar] [CrossRef]
- Wen, Y.; Su, S.; Ma, L.; Yang, S.; Wang, Y.; Wang, X. Effects of canopy microclimate on fruit yield and quality of Camellia oleifera. Sci. Hortic. 2018, 235, 132–141. [Google Scholar] [CrossRef]
- Zorer, R.; Volschenk, C.; Hunter, J. Integrating geographic information systems and hemispherical photography in the assessment of canopy light profiles in a vineyard. Agric. For. Meteorol. 2017, 232, 672–681. [Google Scholar] [CrossRef]
- Sansavini, S.; Musacchi, S. European pear orchard design and HDP management: A review. VIII Int. Symp. Pear 2002, 596, 589–601. [Google Scholar] [CrossRef]
- Cao, Q.; Lan, Y.; Zhou, L.; Lan, W. Study on illumination and fruiting characteristics of three species of chestnut. China Fruits 2006, 15–16. [Google Scholar] [CrossRef]
- Castilloruiz, F.; Jiménezjiménez, F.; Blancoroldán, G.; Solaguirado, R.; Agüeravega, J.; Castrogarcia, S. Analysis of fruit and oil quantity and quality distribution in high-density olive trees in order to improve the mechanical harvesting process. Span. J. Agric. Res. 2015, 13. [Google Scholar] [CrossRef]
- Gómezdelcampo, M.; García, J. Canopy fruit location can affect olive oil quality in ‘Arbequina’ hedgerow orchards. J. Am. Oil Chem. Soc. 2012, 89, 123–133. [Google Scholar] [CrossRef]
- Hatfield, J.; Prueger, J. Temperature extremes: Effect on plant growth and development. Weather Clim. Extrem. 2015, 10, 4–10. [Google Scholar] [CrossRef]
- Trad, M.; Gaaliche, B.; Renard, C.; Mars, M. Inter- and intra-tree variability in quality of figs. Influence of altitude, leaf area and fruit position in the canopy. Sci. Hortic. 2013, 162, 49–54. [Google Scholar] [CrossRef]
- Amarante, C.; Steffens, C.; Argenta, L. Yield and fruit quality of ‘Gala’ and ‘Fuji’ apple trees protected by white anti-hail net. Sci. Hortic. 2011, 129, 79–85. [Google Scholar] [CrossRef]
- Lin, L. Studies on Mineral Nutrients and Fertilizer Application of Castaneamollissima BL. Master’s Thesis, Beijing Forestry University, Beijing, China, 2004. [Google Scholar]
- Peng, F.; Chen, M. A preliminary study on the ecophysiological characteristic of photosynthesis of chestnut in high density planting orchards. J. Nanjing For. Univ. 1998, 22, 6–10. [Google Scholar]
- Liu, Q.; Dong, H.; Liu, P.; Zhang, L.; Wang, F. Studies on the photosynthetic characteristics of chestnut trees. J. Fruit Sci. 2005, 22, 335–338. [Google Scholar] [CrossRef]
- Guo, S.; Xiong, H.; Li, G.; Zhou, F.; Peng, J.; Xie, P. Effect of chestnut (Castanea mollissima BL.) tree structure on canopy light radiation, photosynthesis and yield. J. Northeast For. Univ. 2014, 42, 14–18. [Google Scholar]
- Zhang, W. Fruit Tree Method, 3rd ed.; China Agriculture Press: Beijing, China, 1995. [Google Scholar]
- Mika, A.; Buler, Z.; Treder, W.; Sopyla, C. Relationship between fruit distribution within ‘Jonagold’ apple canopy, fruit quality and illumination. J. Fruit Ornam. Plant. Res. 2002, 10, 75–84. [Google Scholar]
- Inglese, P.; Costanza, P.; Gugliuzza, G. Influence of within-tree and environmental factors on fruit, quality of cactus pear (Opuntiaficus indica) in Italy. Fruits 2010, 65, 179–189. [Google Scholar] [CrossRef]
- Gullo, G.; Motisi, A.; Zappia, R.; Dattola, A.; Diamanti, J.; Mezzetti, B. Rootstock and fruit canopy position affect peach [Prunus persica (L.) Batsch] (cv. Rich May) plant productivity and fruit sensorial and nutritional quality. Food Chem. 2014, 153, 234–242. [Google Scholar] [CrossRef]
- Lewallen, K.S.; Marini, R.P. Relationship between flesh firmness and ground color in peach as influenced by light and canopy position. J. Am. Soc. Hortic. Sci. 2003, 128, 163–170. [Google Scholar] [CrossRef]
- Alcobendas, R.; Mirás-Avalos, J.; Alarcón, J.; Nicolás, E. Effects of irrigation and fruit position on size, colour, firmness and sugar contents of fruits in a mid-late maturing peach cultivar. Sci. Hortic. 2013, 164, 340–347. [Google Scholar] [CrossRef]
- Castro, F.; Fetcher, N. Three dimensional model of the interception of light by a canopy. Agric. For. Meteorol. 1998, 90, 215–233. [Google Scholar] [CrossRef]
- Xue, H.; Han, Y.; Li, Y.; Wang, G.; Feng, L.; Fan, Z.; Mao, S. Spatial distribution of light interception by different plant population densities and its relationship with yield. Field Crop Res. 2015, 184, 17–27. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.; Ahn, T.; Shin, J.; Park, K.; Son, J. Sweet pepper (Capsicum annuum L.) canopy photosynthesis modeling using 3D plant architecture and light ray-tracing. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, P. Effect of light intensity on growth, development and photosynthesis of the chestnut tree. Nonwood For. Res. 1995, 37, 13–15. [Google Scholar] [CrossRef]
- Agele, S.; Maraiyesa, I.; Adeniji, I. Effects of variety and row spacing on radiation interception, partitioning of dry matter and seed set efficiency in late season sunflower (Helianthus annuus L.) in a humid zone of Nigeria. Afr. J. Agric. Res. 2007, 2, 80–88. [Google Scholar]
- Liu, T.; Song, F. Maize photosynthesis and microclimate within the canopies at grain-filling stage in response to narrow-wide row planting patterns. Photosynthetica 2012, 50, 215–222. [Google Scholar] [CrossRef]
- Jung, S.; Choi, H. Light penetration, growth, and fruit productivity in ‘Fuji’ apple trees trained to four growing systems. Sci. Hortic. 2010, 125, 672–678. [Google Scholar] [CrossRef]
- Xi, R.; Liu, M. Chinese Dried Fruits; China Forestry Publishing House: Beijing, China, 2005. [Google Scholar]
- Liu, Y.; Wei, Q.; Gao, Z.; Wang, X.; Wei, S. Relationship between Distribution of relative light radiation and yield and quality in different tree shapes for “Fuji” apple. J. Anhui Agric. Univ. 2004, 31, 353–357. [Google Scholar] [CrossRef]
- Main, G.; Morris, J. Leaf-removal effects on Cynthiana yield, juice composition, and wine composition. Am. J. Enol. Viticult. 2004, 55, 147–152. [Google Scholar]
- Kliewer, W.; Dokoozlian, N. Leaf area/crop weight ratios of grapevines: Influence on fruit composition and wine quality. Am. J. Enol. Viticult. 2005, 56, 170–181. [Google Scholar]
| Nut Characteristic | Nut Yield | Nut Weight | Starch Content | Total Sugar Content | Fat Content | Protein Content |
|---|---|---|---|---|---|---|
| Nut yield | 1 | |||||
| Nut weight | 0.615 ** | 1 | ||||
| Starch content | 0.598 ** | 0.884 ** | 1 | |||
| Total sugar content | 0.726 ** | 0.871 ** | 0.893 ** | 1 | ||
| Fat content | 0.628 ** | 0.759 ** | 0.793 ** | 0.790 ** | 1 | |
| Protein content | 0.577 ** | 0.764 ** | 0.823 ** | 0.849 ** | 0.830 ** | 1 |
| Month | Position | Light Intensity (μmol·m2·s−1) | Temperature (°C) | Humidity (%) |
|---|---|---|---|---|
| 5 | LI | 692.12 ± 5.43 f | 24.01 ± 0.01 e | 28.61 ± 0.02 a |
| LO | 1390.92 ± 13.21 c | 24.82 ± 0.14 c | 25.92 ± 0.02 d | |
| MI | 732.09 ± 7.23 e | 24.31 ± 0.04 d | 28.51 ± 0.04 b | |
| MO | 1432.23 ± 7.71 b | 25.71 ± 0.04 b | 25.80 ± 0.03 e | |
| TI | 764.22 ± 9.18 d | 24.63 ± 0.15 c | 27.17 ± 0.02 c | |
| TO | 1468.89 ± 10.56 a | 25.90 ± 0.10 a | 24.94 ± 0.04 f | |
| 6 | LI | 680.09 ± 5.66 e | 25.13 ± 0.07 e | 43.81 ± 0.09 a |
| LO | 1359.11 ± 16.31 c | 26.16 ± 0.14 b | 40.20 ± 0.05 d | |
| MI | 736.21 ± 9.32 d | 25.41 ± 0.11 d | 41.92 ± 0.07 b | |
| MO | 1409.21 ± 18.42 b | 26.92 ± 0.10 a | 38.93 ± 0.02 e | |
| TI | 743.23 ± 12.85 d | 25.90 ± 0.10 c | 41.17 ± 0.05 c | |
| TO | 1450.21 ± 14.32 a | 27.01 ± 0.16 a | 38.15 ± 0.03 f | |
| 7 | LI | 667.24 ± 6.90 f | 26.71 ± 0.10 f | 48.23 ± 0.11 a |
| LO | 1330.89 ± 15.11 c | 29.62 ± 0.11 c | 46.03 ± 0.07 d | |
| MI | 710.09 ± 9.91 e | 27.34 ± 0.13 e | 47.92 ± 0.09 b | |
| MO | 1389.23 ± 10.39 b | 30.93 ± 0.08 b | 45.15 ± 0.07 e | |
| TI | 740.22 ± 11.56 d | 28.10 ± 0.10 d | 47.27 ± 0.10 c | |
| TO | 1439.30 ± 23.11 a | 31.57 ± 0.12 a | 44.71 ± 0.12 f | |
| 8 | LI | 674.78 ± 10.45 e | 28.71 ± 0.09 f | 44.51 ± 0.07 a |
| LO | 1343.79 ± 23.57 b | 32.32 ± 0.16 c | 41.30 ± 0.10 d | |
| MI | 722.66 ± 13.21 d | 29.00 ± 0.14 e | 43.46 ± 0.08 b | |
| MO | 1413.13 ± 20.34 a | 33.10 ± 0.09 b | 41.22 ± 0.02 d | |
| TI | 757.10 ± 17.21 c | 30.10 ± 0.08 d | 42.29 ± 0.10 c | |
| TO | 1475.22 ± 14.55 a | 33.56 ± 0.16 a | 40.10 ± 0.09 e | |
| 9 | LI | 656.56 ± 9.12 f | 27.11 ± 0.14 e | 37.71 ± 0.05 a |
| LO | 1325.96 ± 21.32 c | 29.42 ± 0.11 c | 35.80 ± 0.07 d | |
| MI | 717.45 ± 10.31 e | 27.60 ± 0.17 d | 36.53 ± 0.04 b | |
| MO | 1395.36 ± 20.81 b | 30.09 ± 0.09 b | 35.22 ± 0.10 e | |
| TI | 750.89 ± 14.81 d | 27.81 ± 0.12 d | 36.01 ± 0.07 c | |
| TO | 1452.11 ± 20.11 a | 30.52 ± 0.11 a | 34.93 ± 0.08 f | |
| 10 | LI | 635.27 ± 8.31 f | 26.13 ± 0.12 f | 33.60 ± 0.07 a |
| LO | 1310.35 ± 20.11 c | 28.14 ± 0.15 c | 30.11 ± 0.08 d | |
| MI | 703.02 ± 9.99 e | 26.80 ± 0.14 e | 32.53 ± 0.09 b | |
| MO | 1372.11 ± 13.01 b | 28.91 ± 0.19 b | 29.55 ± 0.11 e | |
| TI | 732.96 ± 11.87 d | 27.15 ± 0.10 d | 31.20 ± 0.08 c | |
| TO | 1410.33 ± 18.34 a | 29.26 ± 0.17 a | 28.17 ± 0.12 f |
| Nut Yield (g·m−3) | Microclimate Factor | ||
|---|---|---|---|
| Light Intensity (μmol·m2·s−1) | Temperature (°C) | Humidity (%) | |
| 200 | 1500.0 | 34.4 | 27.5 |
| 150 | 1125.0 | 33.8 | 30.0 |
| 100 | 750.0 | 33.2 | 32.5 |
| 50 | 375.0 | 32.6 | 35.0 |
| Quality Factor | Correlation Coefficients | ||
|---|---|---|---|
| Light Intensity | Temperature | Humidity | |
| Nut yield (g·m−3) | 0.796 ** | 0.836 ** | −0.593 ** |
| Nut weight (g) | 0.789 ** | 0.804 ** | −0.756 ** |
| Starch content (%) | 0.742 ** | 0.674 ** | −0.519 ** |
| Fat content (%) | 0.639 ** | 0.583 ** | −0.473 * |
| Total sugar content (%) | 0.657 ** | 0.671 ** | −0.596 ** |
| Protein content (%) | 0.785 ** | 0.684 ** | −0.564 ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, Y.; Su, S.; Zhang, H. Effects of Canopy Microclimate on Chinese Chestnut (Castanea mollissima Blume) Nut Yield and Quality. Forests 2020, 11, 97. https://doi.org/10.3390/f11010097
Wen Y, Su S, Zhang H. Effects of Canopy Microclimate on Chinese Chestnut (Castanea mollissima Blume) Nut Yield and Quality. Forests. 2020; 11(1):97. https://doi.org/10.3390/f11010097
Chicago/Turabian StyleWen, Yue, Shuchai Su, and Haicheng Zhang. 2020. "Effects of Canopy Microclimate on Chinese Chestnut (Castanea mollissima Blume) Nut Yield and Quality" Forests 11, no. 1: 97. https://doi.org/10.3390/f11010097
APA StyleWen, Y., Su, S., & Zhang, H. (2020). Effects of Canopy Microclimate on Chinese Chestnut (Castanea mollissima Blume) Nut Yield and Quality. Forests, 11(1), 97. https://doi.org/10.3390/f11010097





