The Composition and Diversity of Soil Bacterial and Fungal Communities Along an Urban-To-Rural Gradient in South China
Abstract
1. Introduction
2. Materials and Methods
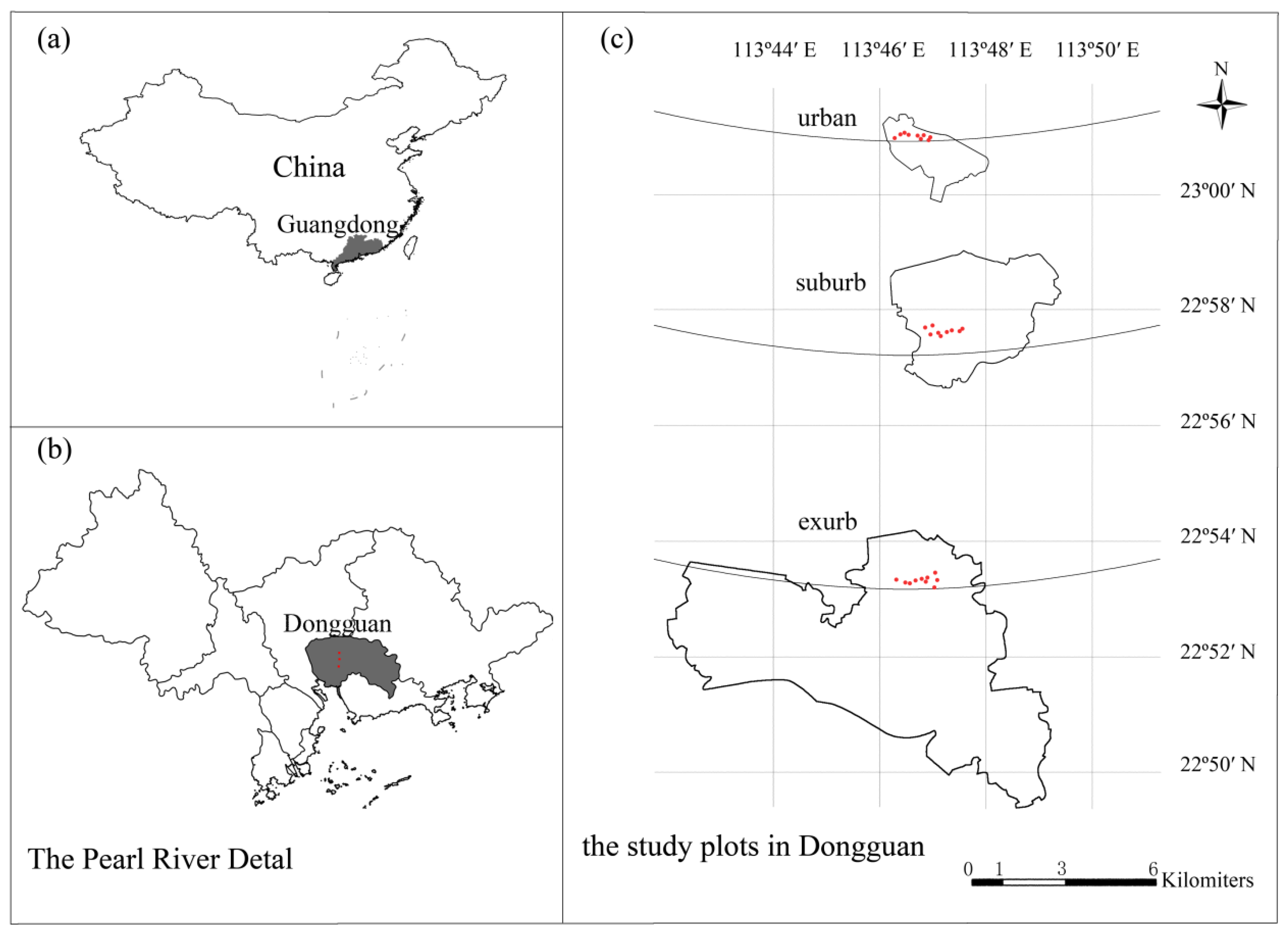
2.1. Study Site
2.2. Experimental Design, Plant Census, and Soil Sampling
2.3. Soil Physicochemical Properties
2.4. Soil Microbial Communities
2.5. Statistical Analyses
3. Results
3.1. Changes in Plant and Soil Variables
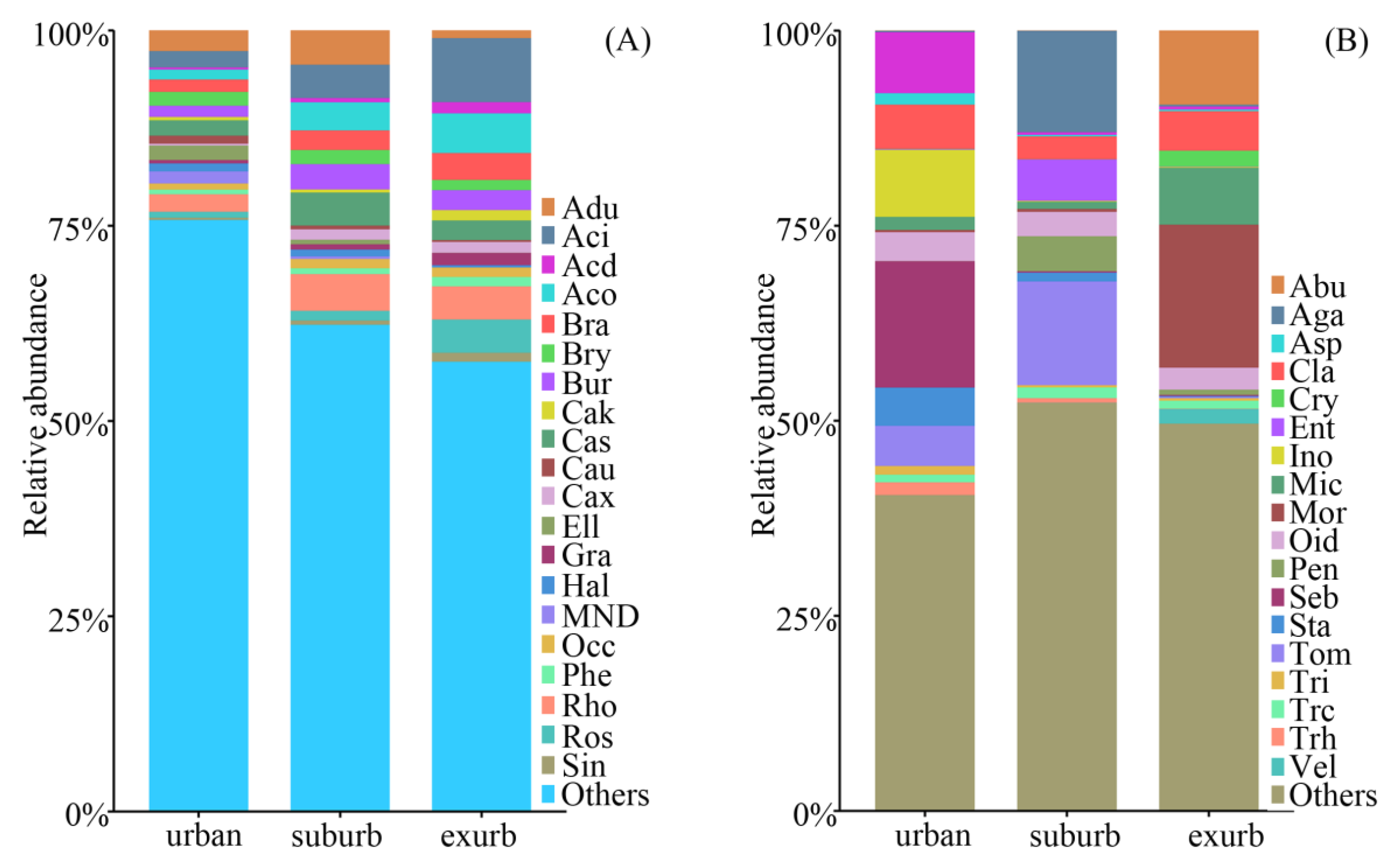
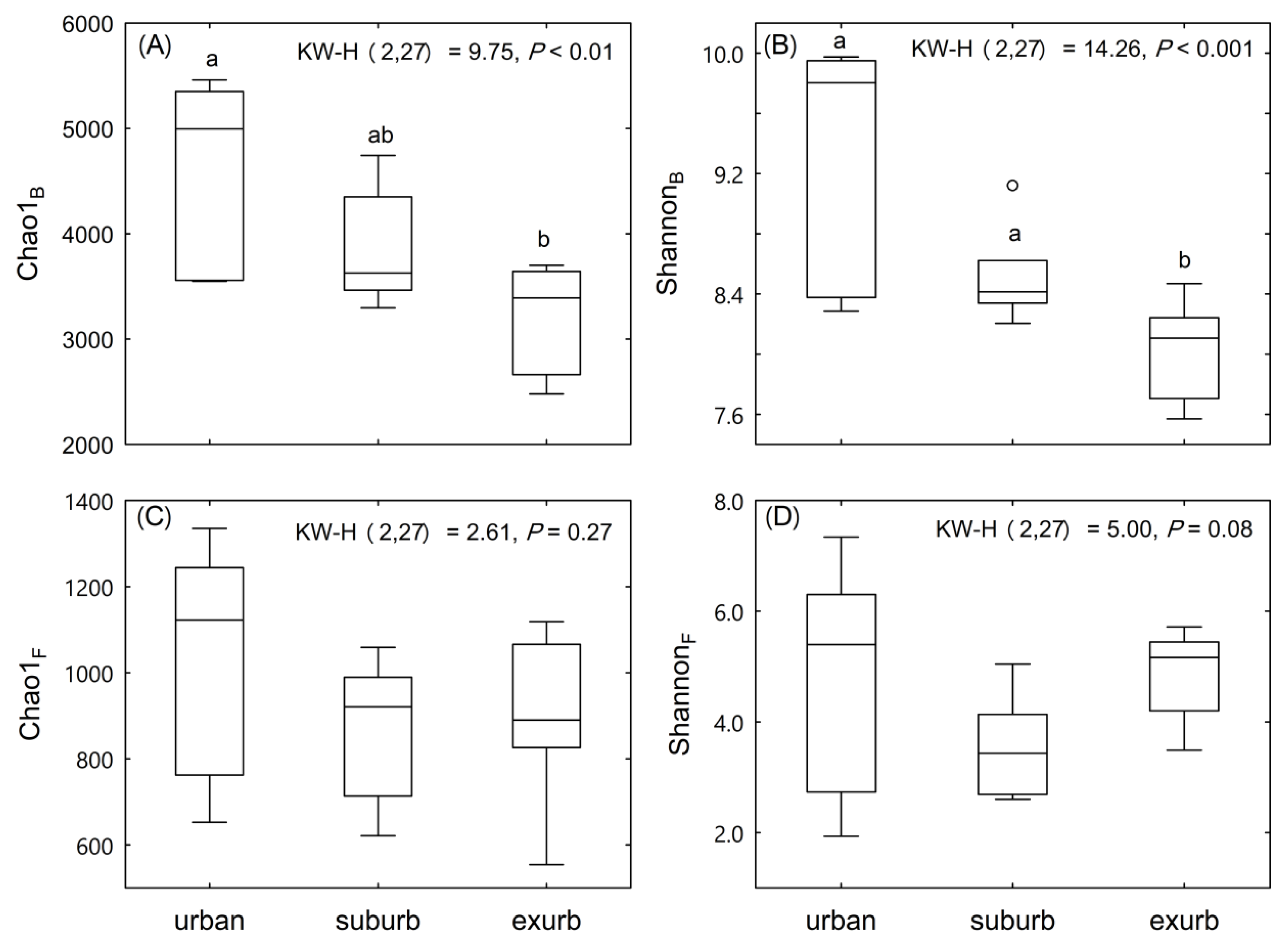
3.2. Communities of Soil Bacteria and Fungi
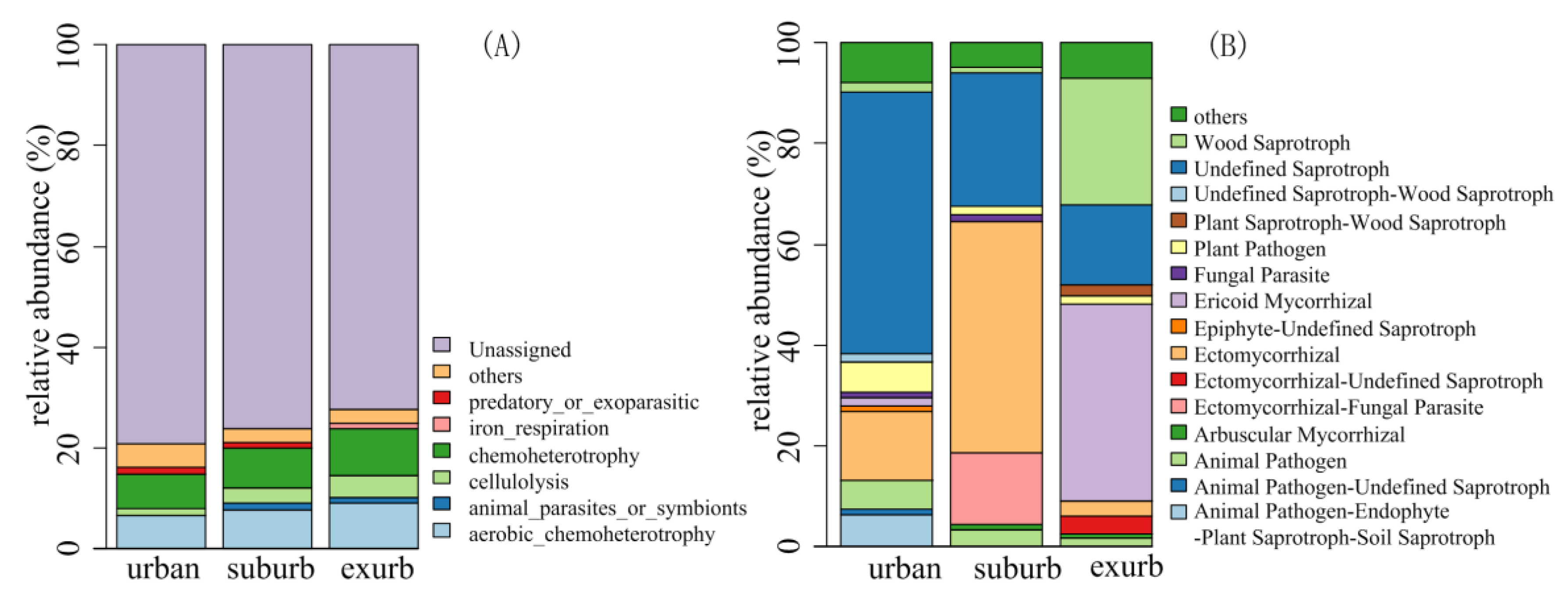
3.3. Bacterial and Fungal Functional Groups
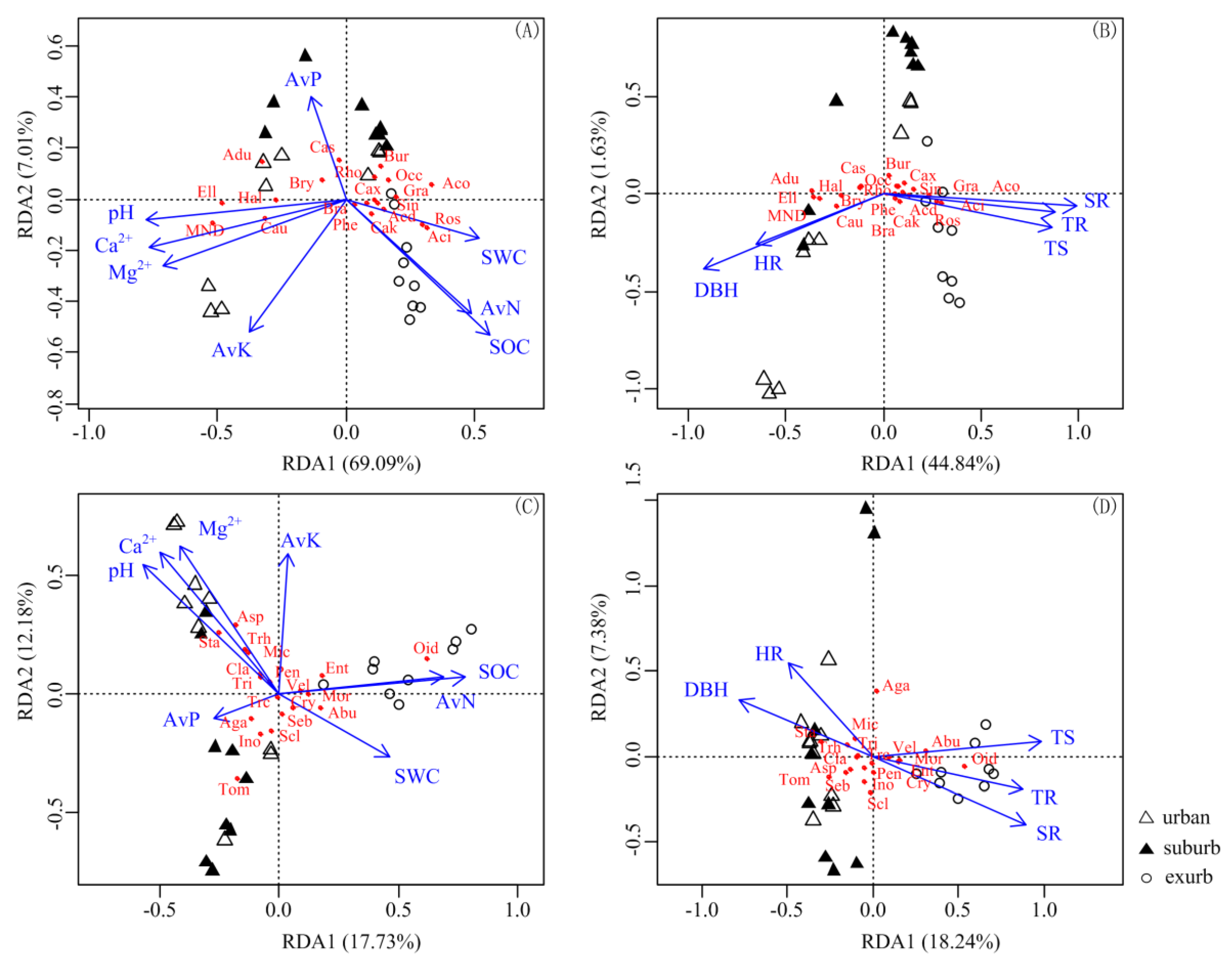
3.4. Correlation of Characteristics of Soil Bacterial and Fungal Communities with Plant and Soil Variables
4. Discussion
4.1. The Composition and Diversity of Soil Microbial Community Varied Along the Urban-To-Rural Gradient
4.2. Plant and Soil Variables Significantly Affect Soil Bacterial Diversity, but Not Fungal Diversity
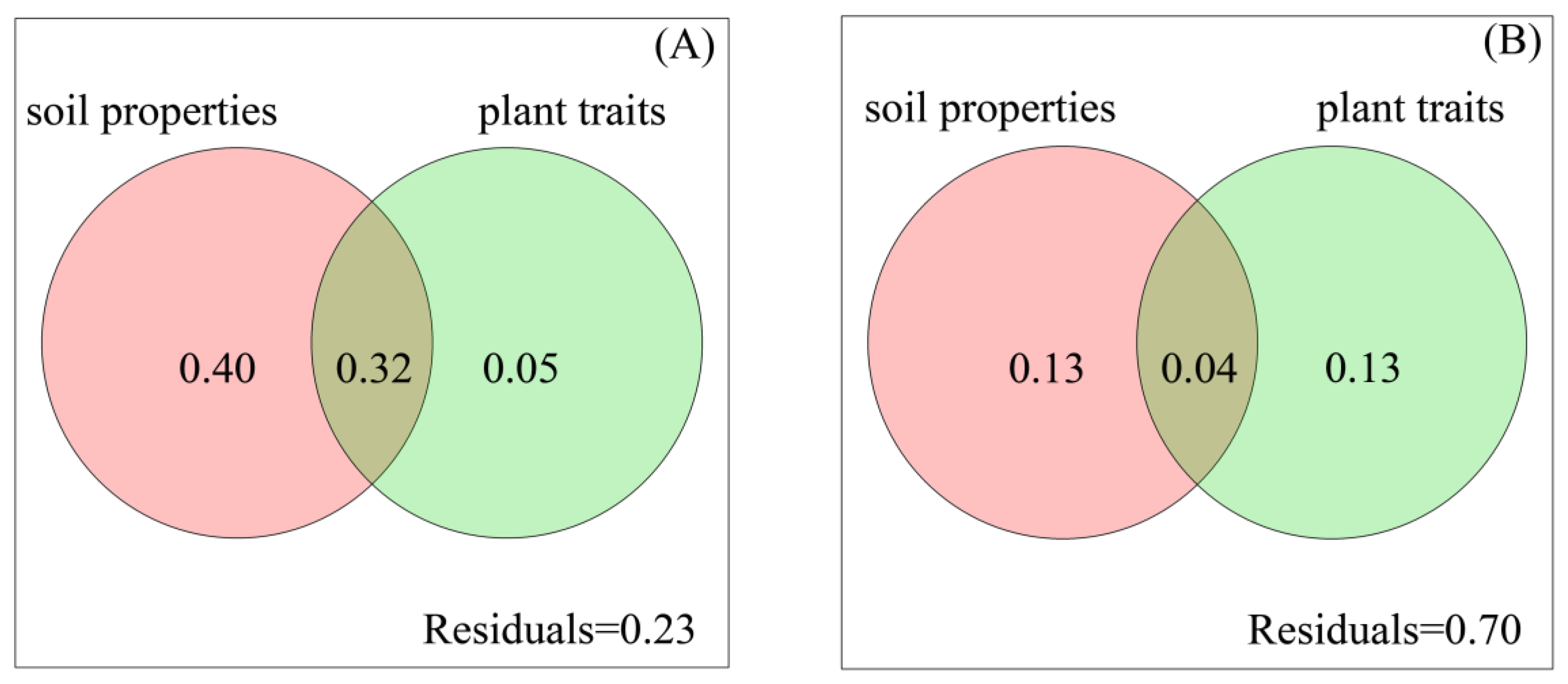
4.3. Soil Properties Affect Bacterial Composition More Than Fungal Composition, While Plant Traits Affect Fungal Composition More Than Bacterial Composition
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Department of Economic and Social Affairs. 2018 Revision of World Urbanization Prospects; United Nations: New York, NY, USA, 2018. [Google Scholar]
- Li, X.M.; Zhou, W.Q. Optimizing urban greenspace spatial pattern to mitigate urban heat island effects: Extending understanding from local to the city scale. Urban For. Urban Green. 2019, 41, 255–263. [Google Scholar] [CrossRef]
- Yazdi, S.K.; Dariani, A.G. CO2 emissions, urbanisation and economic growth: Evidence from Asian countries. Econ. Res.-Ekon. Istraz. 2019, 32, 510–530. [Google Scholar] [CrossRef]
- Cui, Y.Z.; Zhang, W.S.; Bao, H.J.; Wang, C.; Cai, W.J.; Yu, J.; Streets, D.G. Spatiotemporal dynamics of nitrogen dioxide pollution and urban development: Satellite observations over China, 2005–2016. Resour. Conserv. Recy. 2019, 142, 59–68. [Google Scholar] [CrossRef]
- Lopez, B.E.; Urban, D.; White, P.S. Testing the effects of four urbanization filters on forest plant taxonomic, functional, and phylogenetic diversity. Ecol. Appl. 2018, 28, 2197–2205. [Google Scholar] [CrossRef] [PubMed]
- Ning, Z.H.; Chambers, R.; Abdollahi, K. Modeling air pollutant removal, carbon storage, and CO2 sequestration potential of urban forests in Scotlandville, Louisiana, USA. iForest 2016, 9, 860–867. [Google Scholar] [CrossRef]
- Veresoglou, S.D.; Halley, J.M.; Rillig, M.C. Extinction risk of soil biota. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef]
- Klimek, B.; Chodak, M.; Jazwa, M.; Niklinska, M. Functional diversity of soil microbial communities in boreal and temperate Scots pine forests. Eur. J. For. Res. 2016, 135, 731–742. [Google Scholar] [CrossRef]
- Wardle, D.A.; Bardgett, R.D.; Klironomos, J.N.; Setala, H.; van der Putten, W.H.; Wall, D.H. Ecological linkages between aboveground and belowground biota. Science 2004, 304, 1629–1633. [Google Scholar] [CrossRef]
- Li, J.; Li, Z.A.; Wang, F.M.; Zou, B.; Chen, Y.; Zhao, J.; Mo, Q.F.; Li, Y.W.; Li, X.B.; Xia, H.P. Effects of nitrogen and phosphorus addition on soil microbial community in a secondary tropical forest of China. Biol. Fert. Soils 2015, 51, 207–215. [Google Scholar] [CrossRef]
- Deng, Q.; Cheng, X.L.; Hui, D.F.; Zhang, Q.; Li, M.; Zhang, Q.F. Soil microbial community and its interaction with soil carbon and nitrogen dynamics following afforestation in central China. Sci. Total Environ. 2016, 541, 230–237. [Google Scholar] [CrossRef]
- Strecker, T.; Mace, O.G.; Scheu, S.; Eisenhauer, N. Functional composition of plant communities determines the spatial and temporal stability of soil microbial properties in a long-term plant diversity experiment. Oikos 2016, 125, 1743–1754. [Google Scholar] [CrossRef]
- Thakur, M.P.; Milcu, A.; Manning, P.; Niklaus, P.A.; Roscher, C.; Power, S.; Reich, P.B.; Scheu, S.; Tilman, D.; Ai, F.X.; et al. Plant diversity drives soil microbial biomass carbon in grasslands irrespective of global environmental change factors. Glob. Chang. Biol. 2015, 21, 4076–4085. [Google Scholar] [CrossRef]
- Thompson, G.L.; Kao-Kniffin, J. Diversity Enhances NPP, N Retention, and Soil Microbial Diversity in Experimental Urban Grassland Assemblages. PLoS ONE 2016, 11, e0155986. [Google Scholar] [CrossRef]
- Lange, M.; Eisenhauer, N.; Sierra, C.A.; Bessler, H.; Engels, C.; Griffiths, R.I.; Mellado-Vazquez, P.G.; Malik, A.A.; Roy, J.; Scheu, S.; et al. Plant diversity increases soil microbial activity and soil carbon storage. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef]
- Mellado-Vazquez, P.G.; Lange, M.; Bachmann, D.; Gockele, A.; Karlowsky, S.; Milcu, A.; Piel, C.; Roscher, C.; Roy, J.; Gleixner, G. Plant diversity generates enhanced soil microbial access to recently photosynthesized carbon in the rhizosphere. Soil Biol. Biochem. 2016, 94, 122–132. [Google Scholar] [CrossRef]
- Prober, S.M.; Leff, J.W.; Bates, S.T.; Borer, E.T.; Firn, J.; Harpole, W.S.; Lind, E.M.; Seabloom, E.W.; Adler, P.B.; Bakker, J.D.; et al. Plant diversity predicts beta but not alpha diversity of soil microbes across grasslands worldwide. Ecol. Lett. 2015, 18, 85–95. [Google Scholar] [CrossRef]
- Ren, C.J.; Zhang, W.; Zhong, Z.K.; Han, X.H.; Yang, G.H.; Feng, Y.Z.; Ren, G.X. Differential responses of soil microbial biomass, diversity, and compositions to altitudinal gradients depend on plant and soil characteristics. Sci. Total Environ. 2018, 610, 750–758. [Google Scholar] [CrossRef]
- Zhang, Y.G.; Cong, J.; Lu, H.; Li, G.L.; Xue, Y.D.; Deng, Y.; Li, H.; Zhou, J.Z.; Li, D.Q. Soil bacterial diversity patterns and drivers along an elevational gradient on Shennongjia Mountain, China. Microb. Biotechnol. 2015, 8, 739–746. [Google Scholar] [CrossRef]
- Hu, Y.J.; Veresoglou, S.D.; Tedersoo, L.; Xu, T.L.; Ge, T.D.; Liu, L.; Chen, Y.L.; Hao, Z.P.; Su, Y.R.; Rillig, M.C.; et al. Contrasting latitudinal diversity and co-occurrence patterns of soil fungi and plants in forest ecosystems. Soil Biol. Biochem. 2019, 131, 100–110. [Google Scholar] [CrossRef]
- Brant, J.B.; Myrold, D.D.; Sulzman, E.W. Root controls on soil microbial community structure in forest soils. Oecologia 2006, 148, 650–659. [Google Scholar] [CrossRef]
- Shen, C.; Xiong, J.; Zhang, H.; Feng, Y.; Lin, X.; Li, X.; Liang, W.; Chu, H. Soil pH drives the spatial distribution of bacterial communities along elevation on Changbai Mountain. Soil Biol. Biochem. 2013, 57, 204–211. [Google Scholar] [CrossRef]
- Zhou, X.Q.; Guo, Z.Y.; Chen, C.R.; Jia, Z.J. Soil microbial community structure and diversity are largely influenced by soil pH and nutrient quality in 78-year-old tree plantations. Biogeosciences 2017, 14, 2101–2111. [Google Scholar] [CrossRef]
- Wang, C.; Liu, D.W.; Bai, E. Decreasing soil microbial diversity is associated with decreasing microbial biomass under nitrogen addition. Soil Biol. Biochem. 2018, 120, 126–133. [Google Scholar] [CrossRef]
- Apeagyei, E.; Bank, M.S.; Spengler, J.D. Distribution of heavy metals in road dust along an urban-rural gradient in Massachusetts. Atmos. Environ. 2011, 45, 2310–2323. [Google Scholar] [CrossRef]
- Fang, S.B.; Qiao, Y.J.; Yin, C.S.; Yang, X.Y.; Li, N. Characterizing the physical and demographic variables associated with heavy metal distribution along urban-rural gradient. Environ. Monit. Assess. 2015, 187, 570. [Google Scholar] [CrossRef]
- Vakhlamova, T.; Rusterholz, H.P.; Kanibolotskaya, Y.; Baur, B. Changes in plant diversity along an urban-rural gradient in an expanding city in Kazakhstan, Western Siberia. Landsc. Urban Plan. 2014, 132, 111–120. [Google Scholar] [CrossRef]
- Huang, J.; Liu, J.X.; Zhang, W.; Cai, X.A.; Liu, L.; Zheng, M.H.; Mo, J.M. Effects of urbanization on plant phosphorus availability in broadleaf and needleleaf subtropical forests. Sci. Total Environ. 2019, 684, 50–57. [Google Scholar] [CrossRef]
- Barrico, L.; Castro, H.; Coutinho, A.P.; Goncalves, M.T.; Freitas, H.; Castro, P. Plant and microbial biodiversity in urban forests and public gardens: Insights for cities’ sustainable development. Urban For. Urban Green. 2018, 29, 19–27. [Google Scholar] [CrossRef]
- Kivlin, S.N.; Hawkes, C.V. Tree species, spatial heterogeneity, and seasonality drive soil fungal abundance, richness, and composition in Neotropical rainforests. Environ. Microbiol. 2016, 18, 4662–4673. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Williams, L.J.; Vincent, J.B.; Stefanski, A.; Cavender-Bares, J.; Messier, C.; Paquette, A.; Gravel, D.; Reich, P.B.; Kennedy, P.G. Ectomycorrhizal fungal diversity and saprotrophic fungal diversity are linked to different tree community attributes in a field-based tree experiment. Mol. Ecol. 2016, 25, 4032–4046. [Google Scholar] [CrossRef]
- Ito, N.; Iwanaga, H.; Charles, S.; Diway, B.; Sabang, J.; Chong, L.; Nanami, S.; Kamiya, K.; Lum, S.; Siregar, U.J.; et al. Geographical variation in soil bacterial community structure in tropical forests in Southeast Asia and temperate forests in Japan based on pyrosequencing analysis of 16S rRNA. Genes Genet. Syst. 2017, 92, 1–20. [Google Scholar] [CrossRef][Green Version]
- Sun, J.; Li, Y.P.; Gao, P.P.; Suo, C.; Xia, B.C. Analyzing urban ecosystem variation in the City of Dongguan: A stepwise cluster modeling approach. Environ. Res. 2018, 166, 276–289. [Google Scholar] [CrossRef]
- Lu, R.Q. Methods of Soil Agricultural Chemical Analysis; China Agricultural Science and Technology Press: Beijing, China, 1999. [Google Scholar]
- Nguyen, N.H.; Song, Z.; Bates, S.T.; Branco, S.; Tedersoo, L.; Menke, J.; Schilling, J.S.; Kennedy, P.G. FUNGuild: An open annotation tool for parsing fungal community datasets by ecological guild. Fungal. Ecol. 2016, 20, 241–248. [Google Scholar] [CrossRef]
- Louca, S.; Parfrey, L.W.; Doebeli, M. Decoupling function and taxonomy in the global ocean microbiome. Science 2016, 353, 1272–1277. [Google Scholar] [CrossRef]
- Peres-Neto, P.R.; Legendre, P.; Dray, S.; Borcard, D. Variation partitioning of species data matrices: Estimation and comparison of fractions. Ecology 2006, 87, 2614–2625. [Google Scholar] [CrossRef]
- Yan, B.; Li, J.S.; Xiao, N.W.; Qi, Y.; Fu, G.; Liu, G.H.; Qiao, M.P. Urban-development-induced Changes in the Diversity and Composition of the Soil Bacterial Community in Beijing. Sci. Rep. UK 2016, 6. [Google Scholar] [CrossRef]
- Comte, J.; del Giorgio, P.A. Linking the patterns of change in composition and function in bacterioplankton successions along environmental gradients. Ecology 2010, 91, 1466–1476. [Google Scholar] [CrossRef]
- Brundrett, M.C.; Tedersoo, L. Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 2018, 220, 1108–1115. [Google Scholar] [CrossRef]
- Peay, K.G. The Mutualistic Niche: Mycorrhizal Symbiosis and Community Dynamics. Annu. Rev. Ecol. Evol. Syst. 2016, 47, 143–164. [Google Scholar] [CrossRef]
- Sinha, S.; Masto, R.E.; Ram, L.C.; Selvi, V.A.; Srivastava, N.K.; Tripathi, R.C.; George, J. Rhizosphere soil microbial index of tree species in a coal mining ecosystem. Soil. Biol. Biochem. 2009, 41, 1824–1832. [Google Scholar] [CrossRef]
- Tedersoo, L.; Bahram, M.; Polme, S.; Koljalg, U.; Yorou, N.S.; Wijesundera, R.; Ruiz, L.V.; Vasco-Palacios, A.M.; Thu, P.Q.; Suija, A.; et al. Global diversity and geography of soil fungi. Science 2014, 346, 125688. [Google Scholar] [CrossRef]
- Hiiesalu, I.; Bahram, M.; Tedersoo, L. Plant species richness and productivity determine the diversity of soil fungal guilds in temperate coniferous forest and bog habitats. Mol. Ecol. 2017, 26, 4846–4858. [Google Scholar] [CrossRef]
- Bardelli, T.; Ascher-Jenull, J.; Stocker, E.B.; Fornasier, F.; Arfaioli, P.; Fravolini, G.; Medeiros, L.R.A.; Egli, M.; Pietramellara, G.; Insam, H.; et al. Impact of slope exposure on chemical and microbiological properties of Norway spruce deadwood and underlying soil during early stages of decomposition in the Italian Alps. Catena 2018, 167, 100–115. [Google Scholar] [CrossRef]
- Rineau, F.; Shah, F.; Smits, M.M.; Persson, P.; Johansson, T.; Carleer, R.; Troein, C.; Tunlid, A. Carbon availability triggers the decomposition of plant litter and assimilation of nitrogen by an ectomycorrhizal fungus. ISME J. 2013, 7, 2010–2022. [Google Scholar] [CrossRef]
- Härdter, R.; Rex, M.; Orlovius, K. Effects of different Mg fertilizer sources on the magnesium availability in soils. Nutr. Cycl. Agroecosyst. 2004, 70, 249–259. [Google Scholar] [CrossRef]
| Site | DBH Mean ± S.E. | H Mean ± S.E. | Representative Species |
|---|---|---|---|
| urban | 14.31 ± 0.41 cm | 8.79 ± 0.19 m | Cinnamomum burmanni, Ficus microcarpa and Albizia falcataria |
| suburb | 11.55 ± 0.27 cm | 8.21 ± 0.14 m | Eucalyptus urophylla, Elaeocarpus sylvestris and Spathodea campanulata |
| exurb | 7.71 ± 0.13 cm | 6.70 ± 0.08 m | Schefflera octophylla, Acronychia pedunculata and Acronychia pedunculata |
| Variables | Urban | Suburb | Exurb | p Value |
|---|---|---|---|---|
| pH | 5.47 ± 0.26 a | 4.66 ± 0.09 a | 4.24 ± 0.04 b | <0.001 |
| SWC (g·kg−1) | 155.65 ± 13.19 b | 187.43 ± 9.15 ab | 208.97 ± 13.33 a | <0.05 |
| SOC (g·kg−1) | 15.95 ± 1.6 b | 17.2 ± 0.9 b | 32.72 ± 2.36 a | <0.001 |
| AvN (mg·kg−1) | 84.29 ± 6.37 b | 84.58 ± 4.85 b | 142.25 ± 10.07 a | <0.001 |
| AvP (mg·kg−1) | 3.98 ± 0.53 b | 12.92 ± 3.76 a | 2.44 ± 0.09 b | <0.01 |
| AvK (mg·kg−1) | 74.05 ± 5.06 | 57.01 ± 4.88 | 68.48 ± 3.57 | 0.074 |
| Ca2+ (mg·kg−1) | 660.53 ± 146.93 a | 214.54 ± 73.14 b | 72.81 ± 6.92 b | <0.01 |
| Mg2+ (mg·kg−1) | 33.13 ± 5.39 a | 11.51 ± 2.17 b | 11.17 ± 0.91 b | <0.01 |
| DBH (cm) | 14.71 ± 0.84 a | 11.49 ± 1.09 b | 7.76 ± 0.37 c | <0.001 |
| TS | 53.11 ± 4.31 c | 82.89 ± 5.72 b | 153.67 ± 5.06 a | <0.001 |
| TR | 7.67 ± 0.91 c | 11.56 ± 1.18 b | 17.67 ± 0.62 a | <0.001 |
| SR | 10.33 ± 1.52 b | 14.67 ± 2.02 b | 29.67 ± 1.09 a | <0.001 |
| HR | 10.11 ± 1.33 a | 7.67 ± 1.26 ab | 5.44 ± 0.38 b | <0.05 |
| Variables | Chao1B | ShannonB | Chao1F | ShannonF |
|---|---|---|---|---|
| DBH (cm) | 0.63 *** | 0.69 *** | 0.20 | −0.13 |
| TS | −0.55 ** | −0.67 *** | −0.29 | −0.01 |
| TR | −0.67 *** | −0.77 *** | −0.23 | 0.00 |
| SR | −0.69 *** | −0.78 *** | −0.08 | 0.22 |
| HR | 0.43 * | 0.48 * | 0.02 | −0.24 |
| pH | 0.78 *** | 0.79 *** | 0.26 | −0.04 |
| SOC (g·kg−1) | −0.71 *** | −0.82 *** | −0.22 | −0.03 |
| AvN (mg·kg−1) | −0.62 ** | −0.68 ** | −0.19 | 0.04 |
| AvP (mg·kg−1) | 0.38 | 0.45 * | 0.29 | −0.03 |
| AcK (mg·kg−1) | 0.34 | 0.24 | 0.29 | 0.14 |
| Ca2+ (mg·kg−1) | 0.74 *** | 0.71 *** | 0.28 | 0.04 |
| Mg2+ (mg·kg−1) | 0.65 *** | 0.59 ** | 0.38 | 0.21 |
| SWC (g·kg−1) | −0.60 ** | −0.69 *** | −0.09 | 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, X.; Kan, L.; Su, Z.; Liu, X.; Zhang, L. The Composition and Diversity of Soil Bacterial and Fungal Communities Along an Urban-To-Rural Gradient in South China. Forests 2019, 10, 797. https://doi.org/10.3390/f10090797
Tan X, Kan L, Su Z, Liu X, Zhang L. The Composition and Diversity of Soil Bacterial and Fungal Communities Along an Urban-To-Rural Gradient in South China. Forests. 2019; 10(9):797. https://doi.org/10.3390/f10090797
Chicago/Turabian StyleTan, Xuelian, Lei Kan, Zhiyao Su, Xiaodong Liu, and Lu Zhang. 2019. "The Composition and Diversity of Soil Bacterial and Fungal Communities Along an Urban-To-Rural Gradient in South China" Forests 10, no. 9: 797. https://doi.org/10.3390/f10090797
APA StyleTan, X., Kan, L., Su, Z., Liu, X., & Zhang, L. (2019). The Composition and Diversity of Soil Bacterial and Fungal Communities Along an Urban-To-Rural Gradient in South China. Forests, 10(9), 797. https://doi.org/10.3390/f10090797





