Development of a Potential Functional Yogurt Using Bioactive Compounds Obtained from the By-Product of the Production of Tannat Red Wine †
Abstract
1. Introduction
2. Methods
2.1. Tannat Grape Skin Treatment and Extract Preparation
2.2. Systems Preparation
- System 1—whey protein isolate and inulin (WPI + I) (control system)
- System 2—whey protein isolate hydrolysate and inulin (WPIH + I) (control system)
- System 3—encapsulant: extract (WPI + I + E) (1:1)
- System 4—encapsulant: extract (WPIH + I + E) (1:1)
- System 5—extract subjected to the spray drying temperature (E c/t) (control system)
- System 6—extract without temperature treatment (E s/t) (control system)
2.3. Evaluation of Encapsulation Efficiency
2.4. Yogurt Preparation
- Yogurt 1—Encapsulant WPI + I (Y WPI + I)
- Yogurt 2—Encapsulant WPIH + I (Y WPIH + I)
- Yogurt 3—Extract with encapsulant WPI + I (Y WPI + I + E)
- Yogurt 4—Extract with encapsulant WPIH + I (Y WPIH + I + E)
- Yogurt 5—Extract at spray drying temperature (Y E w/t)
- Yogurt 6—Extract without drying temperature (Y E wo/t)
- Yogurt 7—Base formulation (Y B)
2.5. Bioactive Compounds’ Release after In Vitro Digestive Simulation
2.6. Statistical Analysis
3. Results and Discussion
3.1. Evaluation of Encapsulation Efficiency
3.2. Bioactive Compounds’ Release
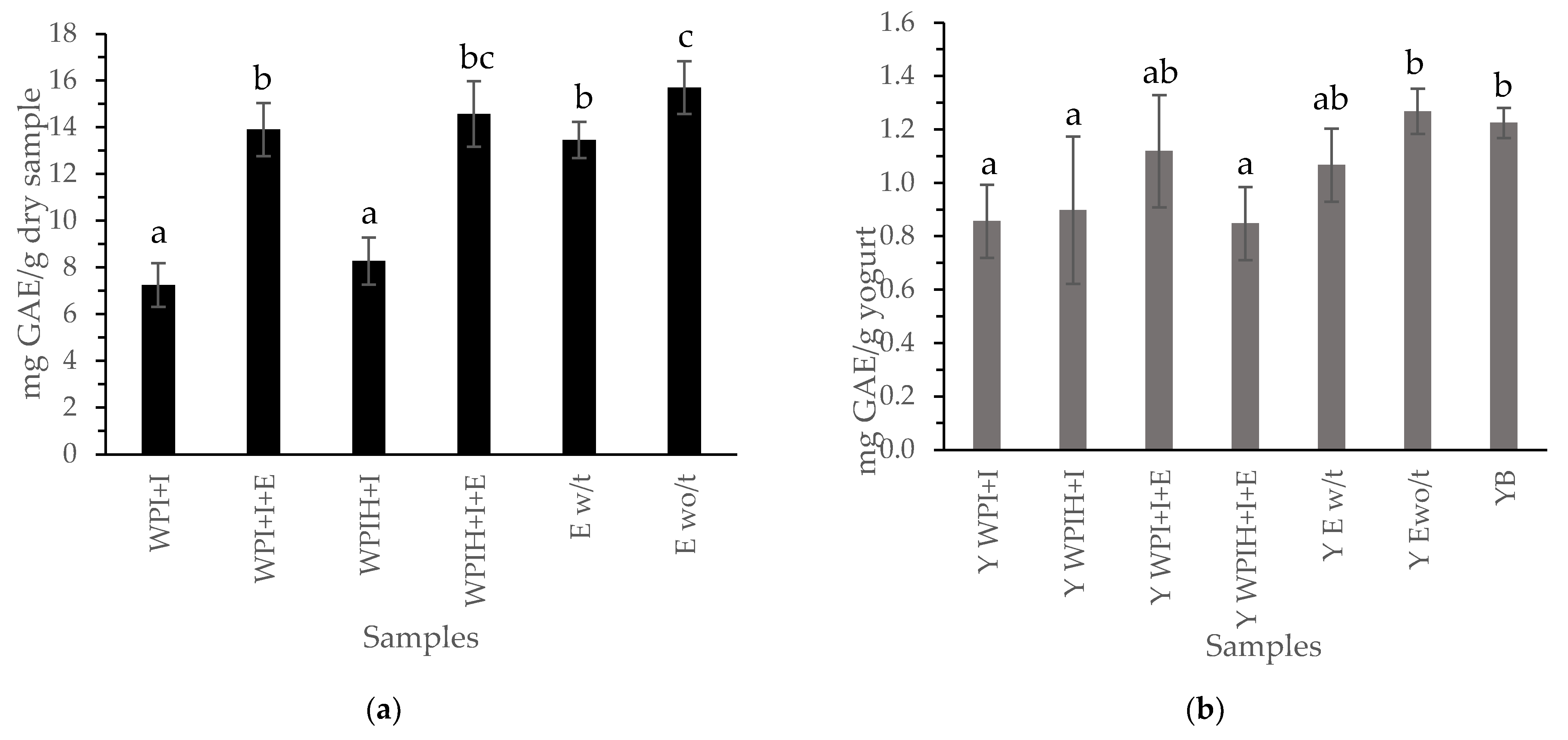
3.2.1. Phenolic Compounds
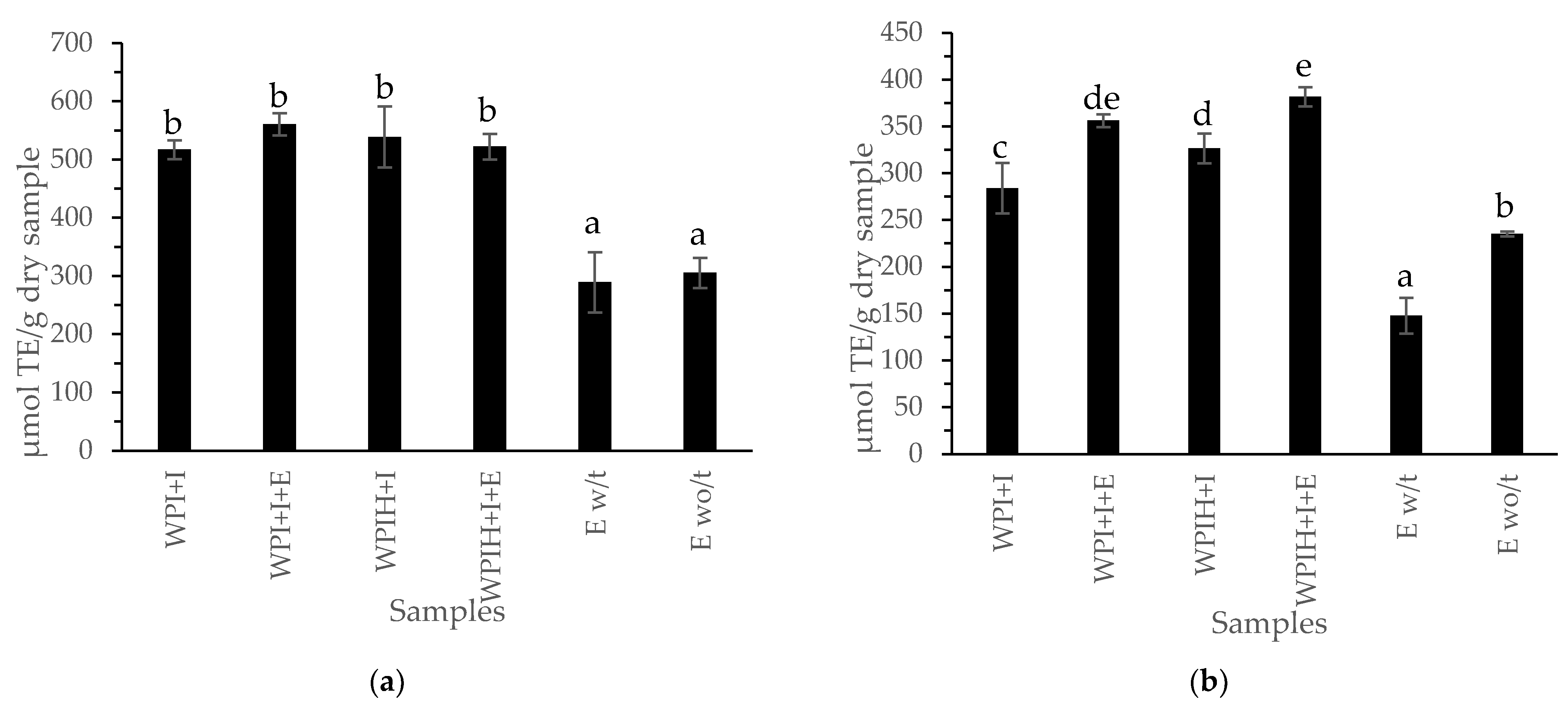
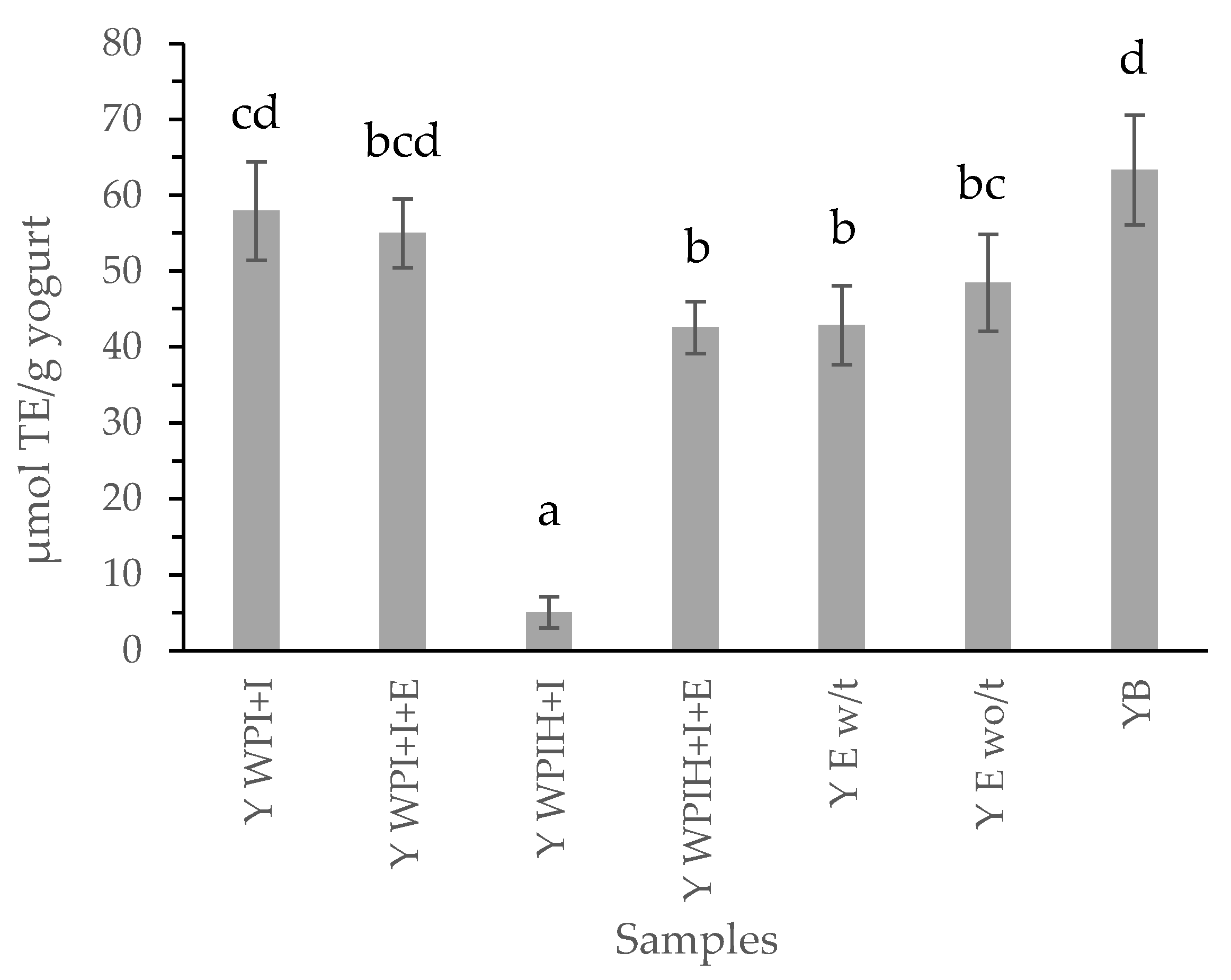
3.2.2. Antioxidant Compounds of the Systems and Yogurts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dávila, I.; Robles, E.; Egüés, I.; Labidi, J.; Gullón, P. The Biorefinery Concept for the Industrial Valorization of Grape Processing By-Products. In Handbook of Grape Processing By-Products: Sustainable Solutions; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 29–53. ISBN 9780128098714. [Google Scholar]
- INAVI. Estadisticas de Viñedos—Datos Nacionales 2019; INAVI: Canelones, Uruguay, 2019. [Google Scholar]
- Fernández-Fernández, A.M.; Iriondo-DeHond, A.; Dellacassa, E.; Medrano-Fernandez, A.; del Castillo, M.D. Assessment of antioxidant, antidiabetic, antiobesity, and anti-inflammatory properties of a Tannat winemaking by-product. Eur. Food Res. Technol. 2019, 245, 1539–1551. [Google Scholar] [CrossRef]
- Drosou, C.; Kyriakopoulou, K.; Bimpilas, A.; Tsimogiannis, D.; Krokida, M. A comparative study on different extraction techniques to recover red grape pomace polyphenols from vinification byproducts. Ind. Crops Prod. 2015, 75, 141–149. [Google Scholar] [CrossRef]
- Beres, C.; Costa, G.N.S.; Cabezudo, I.; da Silva-James, N.K.; Teles, A.S.C.; Cruz, A.P.G.; Mellinger-Silva, C.; Tonon, R.V.; Cabral, L.M.C.; Freitas, S.P. Towards integral utilization of grape pomace from winemaking process: A review. Waste Manag. 2017, 68, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Helal, A.; Agliazucch, D. Impact of in-vitro gastro-pancreatic digestion on polyphenols and cinnamaldehyde bioaccessibility and antio-annotated.pdf. Food Sci. Technol. 2018, 89, 164–170. [Google Scholar]
- Fernández-Fernández, A.M.; Iriondo-DeHond, A.; Nardin, T.; Larcher, R.; Dellacassa, E.; Medrano-Fernandez, A.; Castillo, M.D.d. In Vitro Bioaccessibility of Extractable Compounds from Tannat Grape Skin Possessing Health Promoting Properties with Potential to Reduce the Risk of Diabetes. Foods 2020, 9, 1575. [Google Scholar] [CrossRef] [PubMed]
- El-Messery, T.M.; El-Said, M.M.; Demircan, E.; Ozçelik, B. Microencapsulation of Natural Polyphenolic Compounds extracted form apple peel and its application in yoghurt. Acta Sci. Pol. Technol. Aliment. 2019, 18, 25–34. [Google Scholar] [PubMed]
- Petrotos, K.B.; Karkanta, F.K.; Gkoutsidis, P.E.; Giavasis, I.; Papatheodorou, N.; Ntontos, A.C. Production of Novel Bioactive Yogurt Enriched with Olive Fruit Polyphenols. World Acad. Sci. Eng. Technol. 2012, 64, 867–872. [Google Scholar]
- Agnetti, C.; Jorcín, S.; Medrano, A.; López, T. Encapsulation of whey protein isolate hydrolysate by spray drying as delivery systems in functional foods. In Proceedings of the 1st Ibero-American Congress of Bioactive Peptides (CIAPep), Campinas, Brazil, 3 November 2019. [Google Scholar]
- Robert, P.; Torres, V.; García, P.; Vergara, C.; Carmen, S. The encapsulation of purple cactus pear (Opuntia ficus-indica) pulp by using polysaccharide-proteins as encapsulating agents. Food Sci. Technol. 2015, 60, 1039–1045. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Wu, Y.; Chen, Y.; Qie, X.; Zeng, M.; Wang, Z.; Qin, F.; Chen, J.; He, Z. Analysis of the interaction between cyanidin-3-O-glucoside and casein hydrolysates and its effect on the antioxidant ability of the complexes. Food Chem. 2020, 340, 127915. [Google Scholar] [CrossRef] [PubMed]
- Olt, V.; Baéz, J.; Jorcin, S.; López, T.; Fernández-Fernández, A.M.; Medrano Fernandez, A. Encapsulated bioactive compounds from a winemaking byproduct for its application as functional ingredient in yogurt. Agroci. Uruguay 2021, 25, e794. [Google Scholar] [CrossRef]
- Ky, I.; Lorrain, B.; Kolbas, N.; Crozier, A.; Teissedre, P.L. Wine by-Products: Phenolic characterization and antioxidant activity evaluation of grapes and grape pomaces from six different French grape varieties. Molecules 2014, 19, 482–506. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Gumienna, M.; Lasik, M.; Czarnecki, Z. Bioconversion of grape and chokeberry wine polyphenols during simulated gastrointestinal in vitro digestion. Int. J. Food Sci. Nutr. 2011, 62, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Fernández, A.M.; Dellacassa, E.; Nardin, T.; Larcher, R.; Ibañez, C.; Terán, D.; Gámbaro, A.; Medrano-Fernandez, A.; del Castillo, M.D. Tannat Grape Skin: A Feasible Ingredient for the Formulation of Snacks with Potential for Reducing the Risk of Diabetes. Nutrients 2022, 14, 419. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Fernández, A.M.; Dumay, E.; López-Pedemonte, T.; Medrano-Fernandez, A. Bioaccessibility and Cell Metabolic Activity Studies of Antioxidant Low Molecular Weight Peptides Obtained by Ultrafiltration of α Lactalbumin Enzymatic Hydrolysates. Food Nutr. Sci. 2018, 9, 1047–1065. [Google Scholar] [CrossRef][Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olt, V.; Báez, J.; Jorcin, S.; López, T.; Fernández, A.; Medrano, A. Development of a Potential Functional Yogurt Using Bioactive Compounds Obtained from the By-Product of the Production of Tannat Red Wine. Biol. Life Sci. Forum 2021, 6, 93. https://doi.org/10.3390/Foods2021-10998
Olt V, Báez J, Jorcin S, López T, Fernández A, Medrano A. Development of a Potential Functional Yogurt Using Bioactive Compounds Obtained from the By-Product of the Production of Tannat Red Wine. Biology and Life Sciences Forum. 2021; 6(1):93. https://doi.org/10.3390/Foods2021-10998
Chicago/Turabian StyleOlt, Victoria, Jessica Báez, Santiago Jorcin, Tomás López, Adriana Fernández, and Alejandra Medrano. 2021. "Development of a Potential Functional Yogurt Using Bioactive Compounds Obtained from the By-Product of the Production of Tannat Red Wine" Biology and Life Sciences Forum 6, no. 1: 93. https://doi.org/10.3390/Foods2021-10998
APA StyleOlt, V., Báez, J., Jorcin, S., López, T., Fernández, A., & Medrano, A. (2021). Development of a Potential Functional Yogurt Using Bioactive Compounds Obtained from the By-Product of the Production of Tannat Red Wine. Biology and Life Sciences Forum, 6(1), 93. https://doi.org/10.3390/Foods2021-10998








