First Report of a Toxic Nodularia spumigena (Nostocales/ Cyanobacteria) Bloom in Sub-Tropical Australia. II. Bioaccumulation of Nodularin in Isolated Populations of Mullet (Mugilidae)
Abstract
:1. Introduction
| Species | Common name | Date collected | Matrix | Nodularin concentration # μg/kg | Method | LOD/LOR | References |
|---|---|---|---|---|---|---|---|
| Platichthys flesus | European flounder | 1997–2000 | muscle | ND | ELISA | 0.2 ppb LOD | [7,8,9] |
| Platichthys flesus | European flounder | 2007 | muscle | ND | HPLC-UV | NR | [10] |
| Mytilus edulis | blue mussel | 2007 | whole soft tissues | ND | HPLC-UV | NR | [10] |
| NR | mussel | NR | whole soft tissues | 0.2 ww | LC + PPIA | NR | [11] |
| Platichthys flesus | European flounder | 2005 | muscle | 1.0 dw | ELISA + confirmatory LC-MS | 0.1 ppb LOD | [12] |
| NR | finfish | 2001 | muscle | 2.5 ww | LC-MS | NR | [13] |
| Salmo salar | Atlantic salmon | 1997 | liver | 5 dw | ELISA | 0.31 ppb IC50 | [14] |
| Clupea harengus membras | Baltic herring | 2002 | liver | 5 dw | LC-MS | NR | [15] |
| Clupea harengus membras | Baltic herring | 1997 | muscle | 6.5 dw | ELISA | 0.31 ppb IC50 | [14] |
| Salmo salar | Atlantic salmon | 2002 | liver | 10 dw | LC-MS | NR | [15] |
| Somateria mollissima | eider | 2002 | muscle | 21 dw | LC-MS | NR | [16] |
| NR | prawn | 2001 | muscle | 22 ww | LC-MS | NR | [13] |
| Platichthys flesus | European flounder | 2001 | liver | 28 dw | LC-MS | 5 pg | [17] |
| Somateria mollissima | eider | 2002 | liver | 48 dw | LC-MS | 10 pg on column | [18] |
| Macoma balthica | Baltic clam | 2006 + | whole soft tissues (gut rinsed) | 52 dw (mean of n = 3 specimens) | LC-MS | NR | [19] |
| Gadus morhua | Atlantic cod | 1998 | liver | 53 dw | ELISA | 0.31 ppb IC50 | [9] |
| Mytilus edulis | blue mussel | 2005 | whole soft tissues | 80 dw | LC-MS | 10 pg on column | [18] |
| Sprattus sprattus | sprat | 2004 | whole fish | 100 dw | ELISA | NR | [20] |
| Platichthys flesus | European flounder | 2004 | muscle | 100 dw | ELISA + LC-MS | NR | [16] |
| Salmo trutta | sea trout | NR + | muscle | 125 dw | ELISA | 0.5 ppb | [21] |
| Platichthys flesus | European flounder | 1997 | liver | 140 dw | ELISA | 0.31 ppb IC50 | [9] |
| Mytilus edulis | blue mussel | 2004 | whole soft tissues | 139 dw | ELISA + confirmatory LC-MS | 0.1 ppb LOD | [12] |
| NR | finfish | 2001 | viscera | 152 ww | LC-MS | NR | [13] |
| Somateria mollissima | eider | 2002 | liver | 180 dw (LC-MS SIR) | ELISA + LC-MS | NR | [22] |
| Somateria mollissima | eider | 2002 | liver | 199 dw | LC-MS | NR | [16] |
| Rutilus rutilus | roach | 2004 | muscle | 200 dw | ELISA + LC-MS | NR | [16] |
| Mytilus edulis | blue mussel | 2004 | soft tissues minus hepatopancreas | 200 dw | LC-MS | 50 pg on-column | [23] |
| Clupea harengus membras | Baltic herring | 2004 | whole fish | 220 dw | ELISA | NR | [20] |
| Platichthys flesus | European flounder | NR + | liver | 220 dw | LC-MS | [24] | |
| Mytilus edulis | blue mussel | 2001 | whole soft tissue | 303 dw | LC-MS | 5 pg | [17] |
| Macoma balthica | Baltic clam | NR | whole soft tissues | 320 dw | HPLC + diode array | 120 ppb LOQ | [25] |
| Platichthys flesus | European flounder | 1999 | liver | 399 dw | ELISA + confirmatory LC-MS | 0.2 ppb LOD | [7,8] |
| Platichthys flesus | European flounder | 2000 | liver | 410 dw | ELISA | 0.2 ppb LOD | [8] |
| Platichthys flesus | European flounder | 2005 | liver | 473 dw | ELISA + confirmatory LC-MS | 0.1 ppb LOD | [12] |
| Platichthys flesus | European flounder | 2007 | liver | 557 dw | HPLC-UV | NR | [10] |
| Platichthys flesus | European flounder | 1995 | liver | 637 ww | LC-MS (multiple reactant monitoring) | 1–2 ppb (selected ion recording) | [26] |
| Penaeus monodon | black tiger prawn | 2001/02 | hepatopancreas | 830 dw | ELISA + confirmatory HPLC + diode array | 0.2 ppb (ELISA) 0.3–0.5 ng per injection | [27] |
| Rutilus rutilus | roach | 2004 | liver | 900 dw | ELISA + LC-MS | NR | [16] |
| Platichthys flesus | European flounder | 2004 | liver | 1,100 dw | ELISA + LC-MS | NR | [16] |
| Mytilus edulis | blue mussel | 2004 | hepatopancreas | 1,100 dw | LC-MS | 50 pg on-column | [23] |
| Salmo trutta | sea trout | NR+ | liver | 1,200 dw | ELISA | 0.5 ppb | [21] |
| Mytilus edulis | blue mussel | 1999 | whole soft tissues | 2,150 dw | ELISA + confirmatory LC-MS | 0.2 ppb LOD | [7,8] |
| Platichthys flesus | European flounder | 2002 | liver | 2,230 dw | ELISA + confirmatory LC-MS | 0.2 ppb * LOD | [28] |
| NR | mussel | 2001 | whole soft tissue | 2,500 ww | LC-MS | NR | [13] |
| NR | prawn | 2001 | viscera | 6,400 ww | LC-MS | NR | [13] |
| Mytilus edulis | blue mussel | 2002 + | whole soft tissue | 13,750 dw | LC-MS | NR | [29] |
| Macoma balthica | Baltic clam | 2000 + | whole soft tissue | 30,300 dw 1,400 dw | ELISA HPLC + diode array | 0.16 ppb 200 ppb LOQ | [30] |
| Mytilus edulis | blue mussel | NR + | whole soft tissue | 80,400 dw | LC-MS | 0.5 ng (abs) | [31,32] |
| Mytilus edulis | blue mussel | NR + | digestive gland | 245,000 dw | LC-MS | 0.5 ng (abs) | [31,32] |
| Common name | Species | Location | Date specimen obtained | Tissue | Nodularin concentration, μg/kg wet weight. Median (range) | C.V. (%) | Nodularin concentration, μg/kg dry weight equivalents. Median (range) |
|---|---|---|---|---|---|---|---|
| sea mullet; n = 3 | Mugil cephalus | recreational lake | November 2008 | Muscle T | 13.6 (10.0, 17.6) | 6.1 | 44.0 (32.4, 57.0) |
| Liver T (n = 2) D (n = 1) | 12,000 (11,200, 13,200) | 8.3 | 43,500 (40,600, 47,800) | ||||
| peritoneal contents T | 195 (132, 277) | 8.3 | |||||
| greasy rockcod; n = 1 | Epinephelus tauvina | recreational lake | November 2008 | Muscle T | ND | ||
| Liver T | 25.2 | 10.7 | 91.3 | ||||
| longfin eel; n = 1 | Anguilla reinhardtii | recreational lake | November 2008 | Muscle T | ND | ||
| liver | 58.6 | 212 | |||||
| yellowfin bream; n = 2 | Acanthopagrus australis | recreational lake | November 2008 | Muscle T | ND | ||
| longfin eel; n = 1 | Anguilla reinhardtii | recreational lake | April 2009 | Liver T | 7.64 | 1.0 | 27.7 |
| sea mullet; n = 3 | Mugil cephalus | recreational lake | 7 September 2009 | Muscle D | 3.07 (2.81, 3.83) | 6.1 | 12.7 (11.6, 15.8) |
| Liver D | 4,740 (4,150, 5,180) | 5.4 | 17,200 (15,000, 18,800) | ||||
| stomach contents D | ND | ||||||
| unidentified mullet; n = 2 | Mugilidae—undifferentiated | recreational lake | 7 September 2009 | Muscle D | ND | ||
| Liver D | 963, 1,150 | 3.9 | 3,490, 4,170 | ||||
| freshwater shrimp; n = 3 | unidentified | recreational lake | 7 September 2009 | whole animal; n = 3 specimens combined and extracted as a single sample | ND | ||
| unidentified mullet; n = 1 | Mugilidae—undifferentiated | reference lake | 7 September 2009 | muscle | ND | ||
| liver | ND | ||||||
| stomach contents | ND | ||||||
| sea mullet (n = 2) | Mugil cephalus | recreational lake | 29 September 2009 | muscle D | 1.94 (n = 1; n = 1: ND) | 12.6 | 8.02 |
| liver D | 5,230, 7,230 | 3.7 | 18,900, 26,200 | ||||
| stomach contents | 4.50, 13.4 | ||||||
| unidentified mullet (n = 2) | Mugilidae—undifferentiated | recreational lake | 29 September 2009 | Muscle D | ND | ||
| Liver D | 1,320, 1,370 | 4.1 | 4,780, 4,960 | ||||
| stomach contents | 3.20, 3.49 | ||||||
| sea mullet (n = 1)+ goldspot mullet (n = 3)+ unidentified mullet (n = 1) | Mugil cephalus + Liza argentia + Mugilidae—undifferentiated | reference lakes | 29 September 2009 | muscle | ND | ||
| Liver D | ND | ||||||
| stomach contents | ND |
2. Results
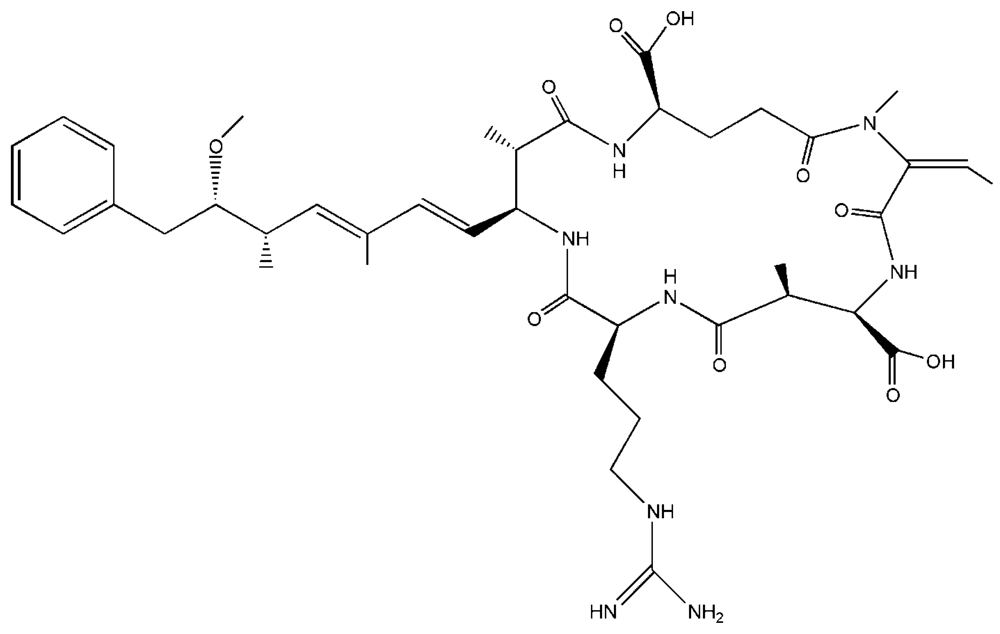
2.1. Systematic Review of Nodularin in Edible Fish, Shellfish and Crustaceans
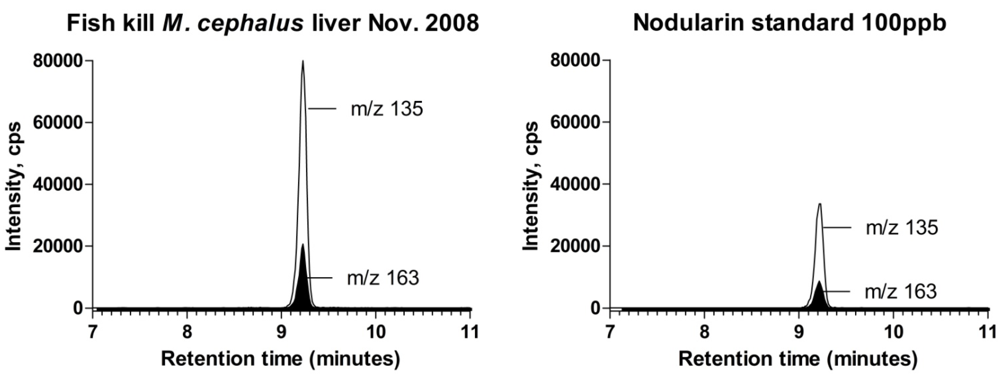
2.2. Fish Species and Measured Nodularin Concentrations in Various Tissues
2.3. Spike Recoveries
| Matrix | Analyte | Recovery (%) median (range) | C.V. (%) |
|---|---|---|---|
| Liver | Nodularin | 83.9 (81.7, 84.0) | 1.53 |
| Liver | MC-LR | 73.8 (73.1, 75.2) | 1.42 |
| Muscle | Nodularin | 80.9 (73.7, 84.9) | 7.12 |
| Muscle | MC-LR | 73.8 (72.4, 76.6) | 2.84 |
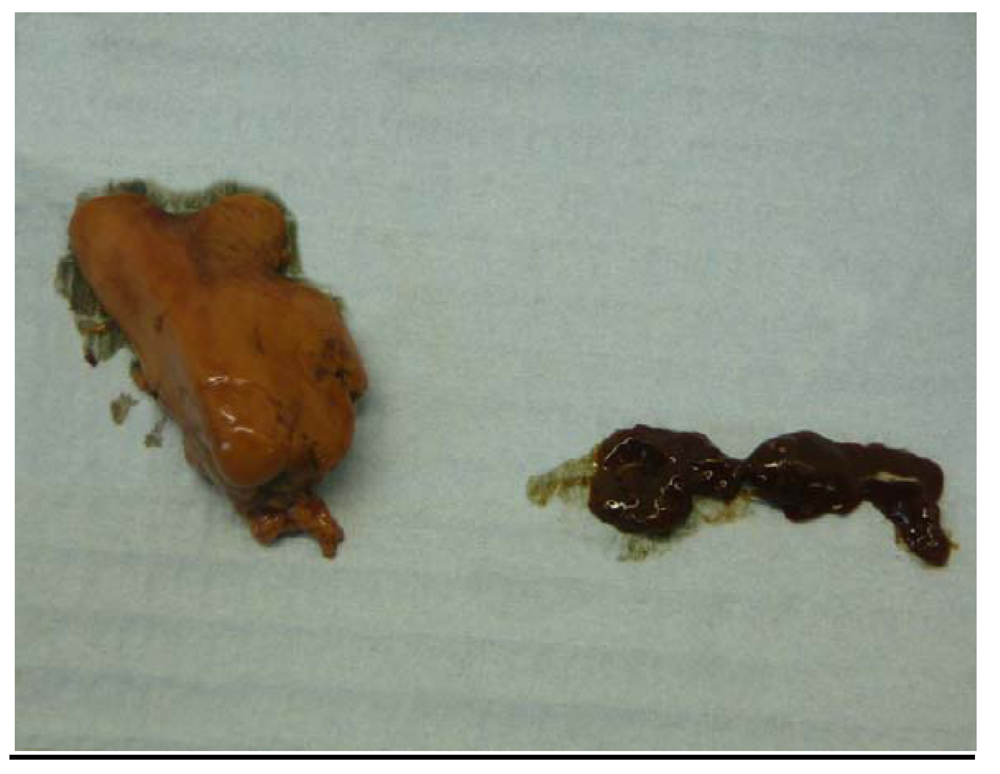
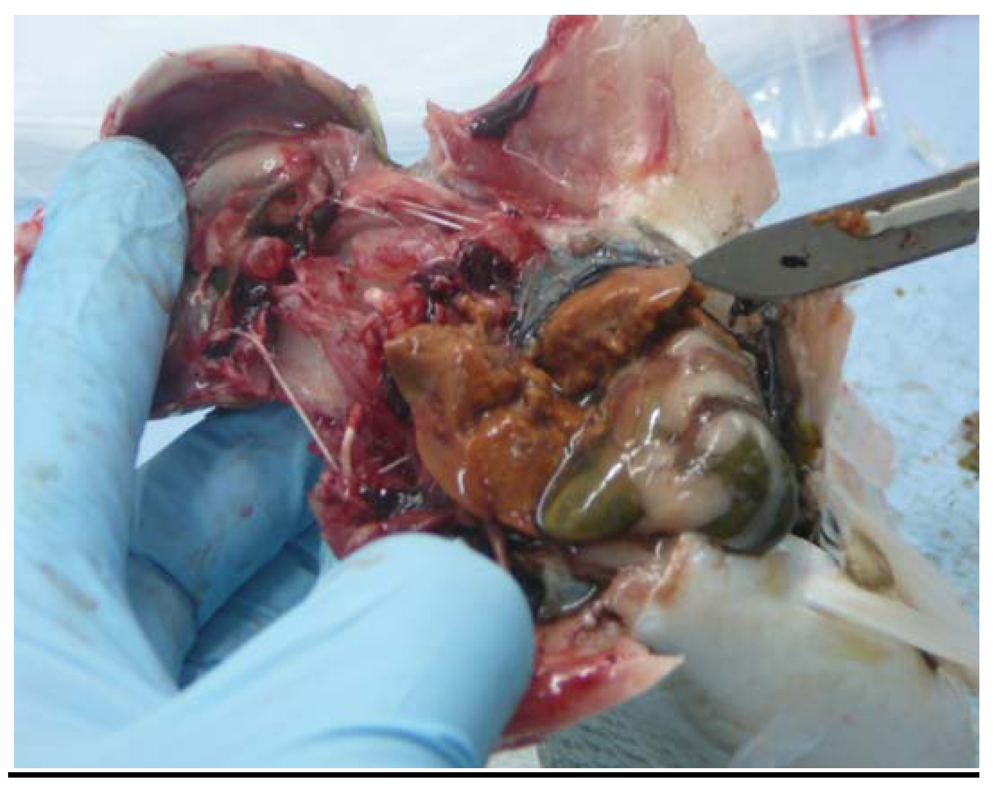
2.4. Gross Appearance of Livers; Liver Histology
| Site | Date | n | Relative liver weight (%); median (range) | Mann-Whitney U statistic (two-tailed p) |
|---|---|---|---|---|
| Study lake | 7 Sept 09 | 4 | 1.66 (1.17, 2.15) | 0.0 (0.016) |
| Control lake | 7 Sept 09 | 1 | 0.45 | |
| Study lake | 29 Sept 09 | 3 | 0.93 (0.93, 1.08) | 2.0 (0.23) |
| Control lake | 29 Sept 09 | 4 | 0.84 (0.67, 0.95) |
2.5. Histopathology Examination
| Slide number | Nodularin (+/-) | Gross appearance of liver | Histopathology | Histopathology Relative Score (HRS) |
|---|---|---|---|---|
| P19 | ND (“control” lake–west pond) | normal | Normal hepatocytes | 7–8 |
| P20 | ND (“control” lake–model yacht pond) | normal | Melanomacrophages (MMCs) which are darker staining, some pyknotic hepatocytes | 5 |
| P21 | ND (“control” lake–model yacht pond) | normal | Dark, pyknotic hepatocytes with some enlarged nuclei, increased hepatocellular vacuolation. Liver section appears to dehisce, a possible artefact due to suboptimal fixation but not autolytic. | 4–5 |
| P14 | ND (“control” lake–west pond) | normal | Mostly normal hepatocytes, with a small number having pyknotic nuclei. MMCs with darkly staining cells. Single granuloma lesion. | 7 |
| P16 | 1.3 mg/kg | normal | Poor fixation resulting in severe autolysis. Precludes useful interpretation. | No score |
| P17 | 7.2 mg/kg | normal | Thick fibrous capsule of liver section. Dark staining MMCs. Free macrophages evident. Normal hepatocytes. Eosinophilic granulocytes present. Hyperaemia. The liver appears reactive in the absence of any obvious pathogens. | 8.5 |
| P18 | 1.4 mg/kg | normal | Significant autolysis in areas with dispersal of MMCs, and free macrophages. Precludes useful interpretation. | No score. |
| P10A | 4.7 mg/kg | enlarged, orange/brown colour, friable | Liver section appears to dehisce, a possible artefact due to suboptimal fixation but not autolytic. Pyknotic hepatocytes, with advanced degeneration & necrosis. Absence of MMCs. | 3 |
| P10B | 5.2 mg/kg | enlarged, orange/brown colour, friable | Pyknotic hepatocytes with occasional normal hepatocytes containing glycogen. Absence of MMCs. | 6 |
| P10C | 4.1 mg/kg | enlarged, orange/brown colour, friable | Normal hepatocytes generally, although there was some loss of hepatocytes in section. Hepatocytes with glycogen storage and fat vacuolation in some areas. Macrophages present. Absence of MMCs. | 6–7 |
| P10D | ND (“control” lake–west pond) | normal | Autolytic changes. MMCs with increased reaction and darker staining cells. Precludes useful interpretation. | No score |
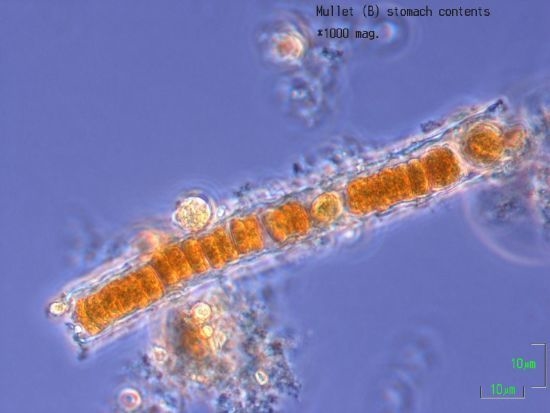
2.6. Stomach Contents

2.7. Comparison of Wet Weight vs. Dry Weight Yields for Nodularin
3. Discussion
3.1. Evidence for Different Mullet Species in the Study Lake
3.2. Mullet Livers: Macroscopic and Microscopic Findings
3.3. No Evidence for Post-Mortem Redistribution of Nodularin
3.4. No Evidence for Biomagnification of Nodularin
3.5. Sources of Nodularin Found in Mullet Tissues
3.6. Public Health Implications
4. Methods
4.1. Systematic Review of the Literature: Nodularin in Seafood and Shellfish
4.2. Fish Specimen Collection
4.3. Detection and Measurement of Nodularin in Fish Tissues
4.4. Spiking Experiments
4.5. Identification of Fish Species
4.6. Liver Histology
4.7. Comparison of Wet Weight vs. Dry Weight Yields for Nodularin
4.8. Nodularia and Nodularin in Stomach Contents
4.9. Statistical Analysis
- Relative liver weights of fish caught in the study lake with those of fish captured from nearby control lakes. These are comparisons between study lake HSI on each capture day, i.e., n = 4 on 7 September 2009 and n = 3 on 29 September 2009, and pooled control lake fish HSI caught on both days (n = 5).
- Wet weight liver nodularin concentrations in M. cephalus and the unknown mugilid species captured from the study lake in September 2009.
5. Conclusions
Acknowledgements
References
- Stewart, I.; Seawright, A.A.; Shaw, G.R. Cyanobacterial poisoning in livestock, wild mammals and birds—An overview. Adv. Exp. Med. Biol. 2008, 619, 613–637. [Google Scholar] [CrossRef]
- Francis, G. Poisonous Australian lake. Nature 1878, 18, 11–12. [Google Scholar] [CrossRef]
- Nehring, S. Mortality of dogs associated with a mass development of Nodularia spumigena (Cyanophyceae) in a brackish lake at the German North Sea coast. J. Plankton Res. 1993, 15, 867–872. [Google Scholar] [CrossRef]
- Algermissen, D.; Mischke, R.; Seehusen, F.; Göbel, J.; Beineke, A. Lymphoid depletion in two dogs with nodularin intoxication. Vet. Rec. 2011, 169, 15. [Google Scholar]
- Rinehart, K.L.; Harada, K.; Namikoshi, M.; Chen, C.; Harvis, C.A.; Munro, M.H.G.; Blunt, J.W.; Mulligan, P.E.; Beasley, V.R.; Dahlem, A.M.; et al. Nodularin, microcystin, and the configuration of ADDA. J. Am. Chem. Soc. 1988, 110, 8557–8558. [Google Scholar]
- Falconer, I.R.; Choice, A.; Hosja, W. Toxicity of edible mussels (Mytilus edulis) growing naturally in an estuary during a water bloom of the blue-green alga Nodularia spumigena. Environ. Toxicol. Water Qual. 1992, 7, 119–123. [Google Scholar] [CrossRef]
- Sipiä, V.O.; Kankaanpää, H.T.; Flinkman, J.; Lahti, K.; Meriluoto, J.A.O. Time-dependent accumulation of cyanobacterial hepatotoxins in flounders (Platichthys flesus) and mussels (Mytilus edulis) from the northern Baltic Sea. Environ. Toxicol. 2001, 16, 330–336. [Google Scholar] [CrossRef]
- Sipiä, V.O.; Kankaanpää, H.T.; Pflugmacher, S.; Flinkman, J.; Furey, A.; James, K.J. Bioaccumulation and detoxication of nodularin in tissues of flounder (Platichthys flesus), mussels (Mytilus edulis, Dreissena polymorpha), and clams (Macoma balthica) from the northern Baltic Sea. Ecotoxicol. Environ. Saf. 2002, 53, 305–311. [Google Scholar] [CrossRef]
- Sipiä, V.; Kankaanpää, H.; Lahti, K.; Carmichael, W.W.; Meriluoto, J. Detection of nodularin in flounders and cod from the Baltic Sea. Environ. Toxicol. 2001, 16, 121–126. [Google Scholar] [CrossRef]
- Persson, K.J.; Legrand, C.; Olsson, T. Detection of nodularin in European flounder (Platichthys flesus) in the west coast of Sweden: Evidence of nodularin mediated oxidative stress. Harmful Algae 2009, 8, 832–838. [Google Scholar] [CrossRef]
- Chen, D.Z.X.; Boland, M.P.; Smillie, M.A.; Klix, H.; Ptak, C.; Andersen, R.J.; Holmes, C.F.B. Identification of protein phosphatase inhibitors of the microcystin class in the marine environment. Toxicon 1993, 31, 1407–1414. [Google Scholar] [CrossRef]
- Mazur-Marzec, H.; Tymińska, A.; Szafranek, J.; Pliński, M. Accumulation of nodularin in sediments, mussels, and fish from the Gulf of Gdansk, Southern Baltic Sea. Environ. Toxicol. 2007, 22, 101–111. [Google Scholar] [CrossRef]
- van Buynder, P.G.; Oughtred, T.; Kirkby, B.; Phillips, S.; Eaglesham, G.; Thomas, K.; Burch, M. Nodularin uptake by seafood during a cyanobacterial bloom. Environ. Toxicol. 2001, 16, 468–471. [Google Scholar] [CrossRef]
- Sipiä, V.O.; Lahti, K.; Kankaanpää, H.T.; Vuorinen, P.J.; Meriluoto, J.A.O. Screening for cyanobacterial hepatotoxins in herring and salmon from the Baltic Sea. Aquat. Ecosyst. Health Manag. 2002, 5, 451–456. [Google Scholar] [CrossRef]
- Sipiä, V.; Kankaanpää, H.; Peltonen, H.; Vinni, M.; Meriluoto, J. Transfer of nodularin to three-spined stickleback (Gasterosteus aculeatus L.), herring (Clupea harengus L.), and salmon (Salmo salar L.) in the northern Baltic Sea. Ecotoxicol. Environ. Saf. 2007, 66, 421–425. [Google Scholar] [CrossRef]
- Sipiä, V.O.; Sjövall, O.; Valtonen, T.; Barnaby, D.L.; Codd, G.A.; Metcalf, J.S.; Kilpi, M.; Mustonen, O.; Meriluoto, J.A.O. Analysis of nodularin-R in eider (Somateria mollissima), roach (Rutilus rutilus L.), and flounder (Platichthys flesus L.) liver and muscle samples from the western Gulf of Finland, Northern Baltic Sea. Environ. Toxicol. Chem. 2006, 25, 2834–2839. [Google Scholar]
- Karlsson, K.; Sipiä, V.; Kankaanpää, H.; Meriluoto, J. Mass spectrometric detection of nodularin and desmethylnodularin in mussels and flounders. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2003, 784, 243–253. [Google Scholar] [CrossRef]
- Sipiä, V.O.; Neffling, M.R.; Metcalf, J.S.; Nybom, S.M.K.; Meriluoto, J.A.O.; Codd, G.A. Nodularin in feathers and liver of eiders (Somateria mollissima) caught from the western Gulf of Finland in June–September 2005. Harmful Algae 2008, 7, 99–105. [Google Scholar] [CrossRef]
- Karlson, A.M.L.; Mozuraitis, R. Deposit-feeders accumulate the cyanobacterial toxin nodularin. Harmful Algae 2011, 12, 77–81. [Google Scholar] [CrossRef]
- Karjalainen, M.; Pääkkönen, J.P.; Peltonen, H.; Sipiä, V.; Valtonen, T.; Viitasalo, M. Nodularin concentrations in Baltic Sea zooplankton and fish during a cyanobacterial bloom. Mar. Biol. 2008, 155, 483–491. [Google Scholar] [CrossRef]
- Kankaanpää, H.; Vuorinen, P.J.; Sipiä, V.; Keinänen, M. Acute effects and bioaccumulation of nodularin in sea trout (Salmo trutta m. trutta L.) exposed orally to Nodularia spumigena under laboratory conditions. Aquat. Toxicol. 2002, 61, 155–168. [Google Scholar] [CrossRef]
- Sipiä, V.O.; Karlsson, K.M.; Meriluoto, J.A.O.; Kankaanpää, H.T. Eiders (Somateria mollissima) obtain nodularin, a cyanobacterial hepatotoxin, in Baltic Sea food web. Environ. Toxicol. Chem. 2004, 23, 1256–1260. [Google Scholar] [CrossRef]
- Kankaanpää, H.; Leiniö, S.; Olin, M.; Sjövall, O.; Meriluoto, J.; Lehtonen, K.K. Accumulation and depuration of cyanobacterial toxin nodularin and biomarker responses in the mussel Mytilus edulis. Chemosphere 2007, 68, 1210–1217. [Google Scholar] [CrossRef]
- Vuorinen, P.J.; Sipiä, V.O.; Karlsson, K.; Keinänen, M.; Furey, A.; Allis, O.; James, K.; Perttilä, U.; Rimaila-Pärnänen, E.; Meriluoto, J.A.O. Accumulation and effects of nodularin from a single and repeated oral doses of cyanobacterium Nodularia spumigena on flounder (Platichthys flesus L.). Arch. Environ. Contam. Toxicol. 2009, 57, 164–173. [Google Scholar]
- Kankaanpää, H.T.; Vuorensola, K.M.; Sipiä, V.O.; Meriluoto, J.A.O. Chromatographic and spectral behaviour and detection of hepatotoxic nodularin in fish, clam, mussel and mouse tissues using HPLC analysis. Chromatographia 2002, 55, 157–162. [Google Scholar] [CrossRef]
- Karlsson, K.; Sipiä, V.; Krause, E.; Meriluoto, J.; Pflugmacher, S. Mass spectrometric detection and quantification of nodularin-R in flounder livers. Environ. Toxicol. 2003, 18, 284–288. [Google Scholar] [CrossRef]
- Kankaanpää, H.T.; Holliday, J.; Schröder, H.; Goddard, T.J.; von Fister, R.; Carmichael, W.W. Cyanobacteria and prawn farming in northern New South Wales, Australia—A case study on cyanobacteria diversity and hepatotoxin bioaccumulation. Toxicol. Appl. Pharmacol. 2005, 203, 243–256. [Google Scholar] [CrossRef]
- Kankaanpää, H.; Turunen, A.K.; Karlsson, K.; Bylund, G.; Meriluoto, J.; Sipiä, V. Heterogeneity of nodularin bioaccumulation in northern Baltic Sea flounders in 2002. Chemosphere 2005, 59, 1091–1097. [Google Scholar] [CrossRef]
- Strogyloudi, E.; Giannakourou, A.; Legrand, C.; Ruehl, A.; Granéli, E. Estimating the accumulation and transfer of Nodularia spumigena toxins by the blue mussel Mytilus edulis: An appraisal from culture and mesocosm experiments. Toxicon 2006, 48, 359–372. [Google Scholar] [CrossRef]
- Lehtonen, K.K.; Kankaanpää, H.; Leiniö, S.; Sipiä, V.O.; Pflugmacher, S.; Sandberg-Kilpi, E. Accumulation of nodularin-like compounds from the cyanobacterium Nodularia spumigena and changes in acetylcholinesterase activity in the clam Macoma balthica during short-term laboratory exposure. Aquat. Toxicol. 2003, 64, 461–476. [Google Scholar] [CrossRef]
- Dahlmann, J.; Budakowski, W.R.; Luckas, B. Liquid chromatography-electrospray ionisation-mass spectrometry based method for the simultaneous determination of algal and cyanobacterial toxins in phytoplankton from marine waters and lakes followed by tentative structural elucidation of microcystins. J. Chromatogr. A 2003, 994, 45–57. [Google Scholar] [CrossRef]
- Svensen, C.; Strogyloudi, E.; Wexels Riser, C.; Dahlmann, J.; Legrand, C.; Wassmann, P.; Granéli, E.; Pagou, K. Reduction of cyanobacterial toxins through coprophagy in Mytilus edulis. Harmful Algae 2005, 4, 329–336. [Google Scholar] [CrossRef]
- Seafood Services Australia. Australian Fish Names Standard AS SSA 5300-2007; Seafood Services Australia: Brisbane, Australia, 2007; p. 100. Available online: http://www.seafood.net.au/intro/AFNS_AS_SSA_5300.pdf (accessed on 20 June 2012).
- Blaber, S.J.M. The food and feeding ecology of Mugilidae in the St Lucia lake system. Biol. J. Linn. Soc. 1976, 8, 267–277. [Google Scholar] [CrossRef]
- McGregor, G.B.; Stewart, I.; Sendall, B.C.; Sadler, R.; Reardon, K.; Carter, S.; Wruck, D.; Wickramasinghe, W. First report of a toxic Nodularia spumigena (Nostocales/Cyanobacteria) bloom from sub-tropical Australia. I. Phycological and public health investigations. Int. J. Environ. Res. Public Health 2012, 9, 2396–2411. [Google Scholar] [CrossRef]
- Hickey, C.W.; Gibbs, M.M. Lake sediment phosphorus release management—Decision support and risk assessment framework. New Zealand J. Mar. Freshwater Res. 2009, 43, 819–854. [Google Scholar] [CrossRef]
- Robb, M.; Greenop, B.; Goss, Z.; Douglas, G.; Adeney, J. Application of Phoslock™, an innovative phosphorus binding clay, to two Western Australian waterways: Preliminary findings. Hydrobiologia 2003, 494, 237–243. [Google Scholar] [CrossRef]
- Namikoshi, M.; Choi, B.W.; Sakai, R.; Sun, F.; Rinehart, K.L.; Carmichael, W.W.; Evans, W.R.; Cruz, P.; Munro, M.H.G.; Blunt, J.W. New nodularins: A general method for structure assignment. J. Org. Chem. 1994, 59, 2349–2357. [Google Scholar]
- Suikkanen, S.; Engström-Öst, J.; Jokela, J.; Sivonen, K.; Viitasalo, M. Allelopathy of Baltic Sea cyanobacteria: No evidence for the role of nodularin. J. Plankton Res. 2006, 28, 543–550. [Google Scholar] [CrossRef]
- Thomson, J.M. The grey mullets. Oceanogr. Mar. Biol. Ann. Rev. 1966, 4, 301–335. [Google Scholar]
- Blaber, S.J.M. Feeding ecology and relative abundance of mullet (Mugilidae) in Natal and Pondoland estuaries. Biol. J. Linn. Soc. 1977, 9, 259–275. [Google Scholar] [CrossRef]
- Malbrouck, C.; Kestemont, P. Effects of microcystins on fish. Environ. Toxicol. Chem. 2006, 25, 72–86. [Google Scholar] [CrossRef]
- Kotak, B.G.; Semalulu, S.; Fritz, D.L.; Prepas, E.E.; Hrudey, S.E.; Coppock, R.W. Hepatic and renal pathology of intraperitoneally administered microcystin-LR in rainbow trout (Oncorhynchus mykiss). Toxicon 1996, 34, 517–525. [Google Scholar] [CrossRef]
- Best, J.H.; Eddy, F.B.; Codd, G.A. Effects of Microcystis cells, cell extracts and lipopolysaccharide on drinking and liver function in rainbow trout Oncorhynchus mykiss Walbaum. Aquat. Toxicol. 2003, 64, 419–426. [Google Scholar] [CrossRef]
- Agius, C.; Roberts, R.J. Melano-macrophage centres and their role in fish pathology. J. Fish Dis. 2003, 26, 499–509. [Google Scholar] [CrossRef]
- Leikin, J.B.; Watson, W.A. Post-mortem toxicology: What the dead can and cannot tell us. J. Toxicol. Clin. Toxicol. 2003, 41, 47–56. [Google Scholar] [CrossRef]
- Yarema, M.C.; Becker, C.E. Key concepts in postmortem drug redistribution. Clin. Toxicol. (Phila) 2005, 43, 235–241. [Google Scholar]
- Fischer, W.J.; Altheimer, S.; Cattori, V.; Meier, P.J.; Dietrich, D.R.; Hagenbuch, B. Organic anion transporting polypeptides expressed in liver and brain mediate uptake of microcystin. Toxicol. Appl. Pharmacol. 2005, 203, 257–263. [Google Scholar] [CrossRef]
- Zhang, Z.; Yu, S.; Chen, C.; Wei, G. Study on the distribution of nodularin in tissues and cell level in mice. Chin. J. Prev. Med. 2002, 36, 100–102. [Google Scholar]
- Ibelings, B.W.; Havens, K.E. Cyanobacterial toxins: A qualitative meta-analysis of concentrations, dosage and effects in freshwater, estuarine and marine biota. Adv. Exp. Med. Biol. 2008, 619, 675–732. [Google Scholar] [CrossRef]
- Berry, J.P.; Lee, E.; Walton, K.; Wilson, A.E.; Bernal-Brooks, F. Bioaccumulation of microcystins by fish associated with a persistent cyanobacterial bloom in Lago de Patzcuaro (Michoacan, Mexico). Environ. Toxicol. Chem. 2011, 30, 1621–1628. [Google Scholar] [CrossRef]
- Eriksson, J.E.; Grönberg, L.; Nygård, S.; Slotte, J.P.; Meriluoto, J.A. Hepatocellular uptake of 3H-dihydromicrocystin-LR, a cyclic peptide toxin. Biochim. Biophys. Acta 1990, 1025, 60–66. [Google Scholar] [CrossRef]
- Tani, Y.; Matsumoto, G.I.; Soma, M.; Soma, Y.; Hashimoto, S.; Kawai, T. Photosynthetic pigments in sediment core HDP-04 from Lake Hovsgol, Mongolia, and their implication for changes in algal productivity and lake environment for the last 1 Ma. Quat. Int. 2009, 205, 74–83. [Google Scholar] [CrossRef]
- Matsumoto, G.I.; Tani, Y.; Seto, K.; Tazawa, T.; Yamamuro, M.; Watanabe, T.; Nakamura, T.; Takemura, T.; Imura, S.; Kanda, H. Holocene palaeolimnological changes in Lake Skallen Oike in the Syowa Station area of Antarctica inferred from organic components in a sediment core (Sk4C-02). J. Paleolimnol. 2010, 44, 677–693. [Google Scholar] [CrossRef]
- Tsuji, K.; Masui, H.; Uemura, H.; Mori, Y.; Harada, K. Analysis of microcystins in sediments using MMPB method. Toxicon 2001, 39, 687–692. [Google Scholar] [CrossRef]
- Toruńska, A.; Bolałek, J,; Pliński, M.; Mazur-Marzek, H. Biodegradation and sorption of nodularin (NOD) in fine-grained sediments. Chemosphere 2008, 2039–2046. [Google Scholar]
- Nyakairu, G.W.A.; Nagawa, C.B.; Mbabazi, J. Assessment of cyanobacteria toxins in freshwater fish: A case study of Murchison Bay (Lake Victoria) and Lake Mburo, Uganda. Toxicon 2010, 55, 939–946. [Google Scholar] [CrossRef]
- Garcia, A.C.; Bargu, S.; Dash, P.; Rabalais, N.N.; Sutor, M.; Morrison, W.; Walker, N.D. Evaluating the potential risk of microcystins to blue crab (Callinectes sapidus) fisheries and human health in a eutrophic estuary. Harmful Algae 2010, 9, 134–143. [Google Scholar] [CrossRef]
- Papadimitriou, T.; Kagalou, I.; Bacopoulos, V.; Leonardos, I.D. Accumulation of microcystins in water and fish tissues: An estimation of risks associated with microcystins in most of the Greek lakes. Environ. Toxicol. 2010, 25, 418–427. [Google Scholar]
- Poste, A.E.; Hecky, R.E.; Guildford, S.J. Evaluating microcystin exposure risk through fish consumption. Environ. Sci. Technol. 2011, 45, 5806–5811. [Google Scholar]
- Saker, M.L.; Eaglesham, G.K. The accumulation of cylindrospermopsin from the cyanobacterium Cylindrospermopsis raciborskii in tissues of the redclaw crayfish Cherax quadricarinatus. Toxicon 1999, 37, 1065–1077. [Google Scholar] [CrossRef]
- Negri, A.P.; Jones, G.J. Bioaccumulation of paralytic shellfish poisoning (PSP) toxins from the cyanobacterium Anabaena circinalis by the freshwater mussel Alathyria condola. Toxicon 1995, 33, 667–678. [Google Scholar] [CrossRef]
- Ibelings, B.W.; Chorus, I. Accumulation of cyanobacterial toxins in freshwater “seafood” and its consequences for public health: A review. Environ. Pollut. 2007, 150, 177–192. [Google Scholar] [CrossRef]
- Mulvenna, V.; Dale, K.; Priestly, B.; Mueller, U.; Humpage, A.; Shaw, G.; Allinson, G.; Falconer, I. Health risk assessment for cyanobacterial toxins in seafood. Int. J. Environ. Res. Public Health 2012, 9, 807–820. [Google Scholar] [CrossRef]
- Chorus, I. Current Approaches to Cyanotoxin Risk Assessment, Risk Management and Regulations in Different Countries; Federal Environmental Agency (Umweltbundesamt): Berlin, Germany, 2005; p. 117. Available online: http://www.umweltdaten.de/publikationen/fpdf-l/2910.pdf (accessed on 20 June 2012).
- van Egmond, H.P.; van Apeldoorn, M.E.; Speijers, G.J.A. Marine Biotoxins; FAO Food and Nutrition Paper No. 80; Food and Agriculture Organization of the United Nations: Rome, Italy, 2004; pp. 1–278. Available online: ftp://ftp.fao.org/docrep/fao/007/y5486e/ (accessed on 20 June 2012).
- Blue-green Algae in the Gippsland Lakes—December 2011. Available online: http://www.health.vic.gov.au/environment/water/bluegreenalgae_gippslandlakes.htm (accessed on 2 March 2012).
- Heresztyn, T.; Nicholson, B.C. Nodularin concentrations in Lakes Alexandrina and Albert, South Australia, during a bloom of the cyanobacterium (blue-green alga) Nodularia spumigena and degradation of the toxin. Environ. Toxicol. Water Qual. 1997, 12, 273–282. [Google Scholar] [CrossRef]
- Stephens, A.; Biggins, N.; Brett, S. Algal Bloom Dynamics in the Estuarine Gippsland Lakes; Publication SR4; Environment Protection Authority Victoria: Melbourne, Australia, 2004; p. 23. Available online: http://epanote2.epa.vic.gov.au/EPA/Publications.nsf/d85500a0d7f5f07b4a2565d 1002268f3/64e0b189afb26cdeca256e3c0002705f/$FILE/SR4.pdf (accessed on 20 June 2012).
- Lenanton, R.C.J.; Loneragan, N.R.; Potter, I.C. Blue-green algal blooms and the commercial fishery of a large Australian estuary. Mar. Pollut. Bull. 1985, 16, 477–482. [Google Scholar] [CrossRef]
- Hallegraeff, G.M. Harmful algal blooms in the Australian region. Mar. Pollut. Bull. 1992, 25, 186–190. [Google Scholar] [CrossRef]
- Ecological Character Description for the Vasse-Wonnerup Wetlands Ramsar Site in South-West Western Australia; WRM: Perth, Australia, 2007; pp. 1–218. Available online: http://www.water.wa.gov.au/PublicationStore/first/83829_Appendices%20D-E.pdf (accessed on 20 June 2012).
- Jones, G.J.; Blackburn, S.I.; Parker, N.S. A toxic bloom of Nodularia spumigena Mertens in Orielton Lagoon, Tasmania. Aust. J. Mar. Freshwater Res. 1994, 45, 787–800. [Google Scholar] [CrossRef]
- Department of Primary Industries. Fisheries Victoria Commercial Fish Production Information Bulletin 2009; Fisheries Victoria: Victoria, Australia, 2009. Available online: http://www.dpi.vic.gov.au/__data/assets/pdf_file/0017/29042/FV-Info-Bulletin-2009.pdf (accessed on 20 June 2012).
- Fletcher, W.J.; Santoro, K. ,State of the Fisheries Report 2008/09; Department of Fisheries, Western Australia: Perth, Australia, 2009; p. 295. Available online: http://www.fish.wa.gov.au/docs/sof/2008/state-of-the-fisheries-2008-09-full.pdf (accessed on 20 June 2012).
- Higham, J.; Ferguson, G.; Ye, Q. Lakes and Coorong Yellow-Eye Mullet (Aldrichetta forsteri) Fishery: Fishery Assessment Report to PIRSA Fisheries. SARDI Aquatic Sciences Publication No. RD04/0162; South Australian Research and Development Institute: Adelaide, Australia, 2005; pp. 1–43. Available online: http://www.pir.sa.gov.au/__data/assets/pdf_file/0018/12744/stock_assmnt_report_yellow_eye_mullet_jan_2005.pdf (accessed on 20 June 2012).
- Australian Bureau of Agricultural and Resource Economics and Sciences. Australian Fisheries Statistics 2010; ABARES: Canberra, Australia, 2011; pp. 1–100. Available online: http://adl.brs.gov.au/data/warehouse/pe_abares20110830.01/AustFishStats_2010_rev20110905.pdf (accessed on 20 June 2012).
- Lee, C.S.; Ostrowski, A.C. Current status of marine finfish larviculture in the United States. Aquaculture 2001, 200, 89–109. [Google Scholar] [CrossRef]
- Lovatelli, A.; Holthus, P.F. (Eds.) Capture-Based Aquaculture: Global Overview; FAO Fisheries Technical Paper No. 508; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008; pp. i–x; 1–298. Available online: ftp://ftp.fao.org/docrep/fao/011/i0254e/i0254e.pdf (accessed on 20 June 2012).
- Saleh, M. Capture-Based Aquaculture of Mullets in Egypt. Capture-Based Aquaculture: Global Overview; FAO Fisheries Technical Paper No. 508; Lovatelli, A., Holthus, P.F., Eds.; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008; pp. 109–126. Available online: ftp://ftp.fao.org/docrep/fao/011/i0254e/i0254e.pdf (accessed on 20 June 2012).
- Lupatsch, I.; Katz, T.; Angel, D.L. Assessment of the removal efficiency of fish farm effluents by grey mullets: A nutritional approach. Aquac. Res. 2003, 34, 1367–1377. [Google Scholar] [CrossRef]
- Porter, C.B.; Krost, P.; Gordin, H.; Angel, D.L. Preliminary assessment of grey mullet (Mugil cephalus) as a forager of organically enriched sediments below marine fish farms. Isr. J. Aquac. Bamid. 1996, 48, 47–55. [Google Scholar]
- Englberger, L.; Aalbersberg, W.; Schierle, J.; Hoffman, P.; Humphries, J.; Huang, A.; Marks, G.C.; Lorens, A.; Alfred, J.; Iuta, T.; et al. Identification of Micronutrient-Rich Locally Grown Foods in the Federated States of Micronesia, Marshall Islands and Kiribati. Proceedings of the Seventh OCEANIAFOODS Conference: Innovations in Nutrient Information; Greenfield, H., Athar, N., Murphy, J., Eds.; New Zealand Institute for Crop & Food Research: Wellington, New Zealand, 2005; pp. 103–110. Available online: http://www.pdfio.com/k-215195.html# (accessed on 20 June 2012).
- Suñer, M.A.; Devesa, V.; Muñoz, O.; López, F.; Montoro, R.; Arias, A.M.; Blasco, J. Total and inorganic arsenic in the fauna of the Guadalquivir estuary: Environmental and human health implications. Sci. Total Environ. 1999, 242, 261–270. [Google Scholar] [CrossRef]
- Bledsoe, G.E.; Bledsoe, C.D.; Rasco, B. Caviars and fish roe products. Crit. Rev. Food Sci. Nutr. 2003, 43, 317–356. [Google Scholar] [CrossRef]
- Barker, T.; Ross, A. Exploring Cultural Constructs: The Case of Sea Mullet Management in Moreton Bay, South East Queensland, Australia. Putting Fishers’ Knowledge to Work: Conference Proceedings; Haggan, N., Brignall, C., Wood, L., Eds.; University of British Columbia: Vancouver, BC, Canada, 2003; pp. 290–305. Available online: http://fisheries.ubc.ca/sites/fisheries.ubc.ca/files/pdfs/fcrrs/11-1b.pdf (accessed on 4 July 2012).
- Katselis, G.; Koutsikopoulos, C.; Rogdakis, Y.; Lachanas, T.; Dimitriou, E.; Vidalis, K. A model to estimate the annual production of roes (avgotaracho) of flathead mullet (Mugil cephalus) based on the spawning migration of species. Fish Res. 2005, 75, 138–148. [Google Scholar] [CrossRef]
- Ojaveer, H. Exploitation of biological resources of the Baltic Sea by Estonia in 1928–1995. Limnologica 1999, 29, 224–226. [Google Scholar] [CrossRef]
- Stewart, I.; Eaglesham, G.K.; Poole, S.; Graham, G.; Paulo, C.; Wickramasinghe, W.; Sadler, R.; Shaw, G.R. Establishing a public health analytical service based on chemical methods for detecting and quantifying pacific ciguatoxin in fish samples. Toxicon 2009, 56, 804–812. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Stewart, I.; Eaglesham, G.K.; McGregor, G.B.; Chong, R.; Seawright, A.A.; Wickramasinghe, W.A.; Sadler, R.; Hunt, L.; Graham, G. First Report of a Toxic Nodularia spumigena (Nostocales/ Cyanobacteria) Bloom in Sub-Tropical Australia. II. Bioaccumulation of Nodularin in Isolated Populations of Mullet (Mugilidae). Int. J. Environ. Res. Public Health 2012, 9, 2412-2443. https://doi.org/10.3390/ijerph9072412
Stewart I, Eaglesham GK, McGregor GB, Chong R, Seawright AA, Wickramasinghe WA, Sadler R, Hunt L, Graham G. First Report of a Toxic Nodularia spumigena (Nostocales/ Cyanobacteria) Bloom in Sub-Tropical Australia. II. Bioaccumulation of Nodularin in Isolated Populations of Mullet (Mugilidae). International Journal of Environmental Research and Public Health. 2012; 9(7):2412-2443. https://doi.org/10.3390/ijerph9072412
Chicago/Turabian StyleStewart, Ian, Geoffrey K. Eaglesham, Glenn B. McGregor, Roger Chong, Alan A. Seawright, Wasantha A. Wickramasinghe, Ross Sadler, Lindsay Hunt, and Glenn Graham. 2012. "First Report of a Toxic Nodularia spumigena (Nostocales/ Cyanobacteria) Bloom in Sub-Tropical Australia. II. Bioaccumulation of Nodularin in Isolated Populations of Mullet (Mugilidae)" International Journal of Environmental Research and Public Health 9, no. 7: 2412-2443. https://doi.org/10.3390/ijerph9072412
APA StyleStewart, I., Eaglesham, G. K., McGregor, G. B., Chong, R., Seawright, A. A., Wickramasinghe, W. A., Sadler, R., Hunt, L., & Graham, G. (2012). First Report of a Toxic Nodularia spumigena (Nostocales/ Cyanobacteria) Bloom in Sub-Tropical Australia. II. Bioaccumulation of Nodularin in Isolated Populations of Mullet (Mugilidae). International Journal of Environmental Research and Public Health, 9(7), 2412-2443. https://doi.org/10.3390/ijerph9072412