Treatment of Clinical Solid Waste Using a Steam Autoclave as a Possible Alternative Technology to Incineration
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation of Bacteria
| Bacteria | Initial concentration, N0 | Log reduction Log N0/N |
|---|---|---|
| Escherichia coli | 2.90 × 108 | 8.5 |
| Psudomonas aeroginosa | 2.92 × 108 | 8.5 |
| Acinetobacter baumannii | 2.95 × 108 | 8.5 |
| Staphylococcus aurous | 2.55 × 108 | 8.4 |
| Streptococcus pyogenes | 1.25 × 108 | 8.0 |
| Bacillus subtilis (Vegetative form) | 3.4 × 108 | 7.5 |
2.2. Sample Preparation
| Material | % in mass (dry basis) |
|---|---|
| Hard plastic | 40 |
| Broken glass | 25 |
| Fabric | 15 |
| metal | 20 |
| Rubber | 10 |
| Total = 100 | |
2.3. Inactivation of Bacteria in CSW
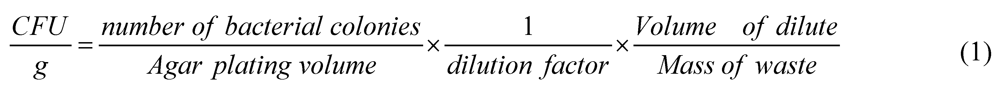

2.4. Bacterial Re-Growth
3. Results and Discussion
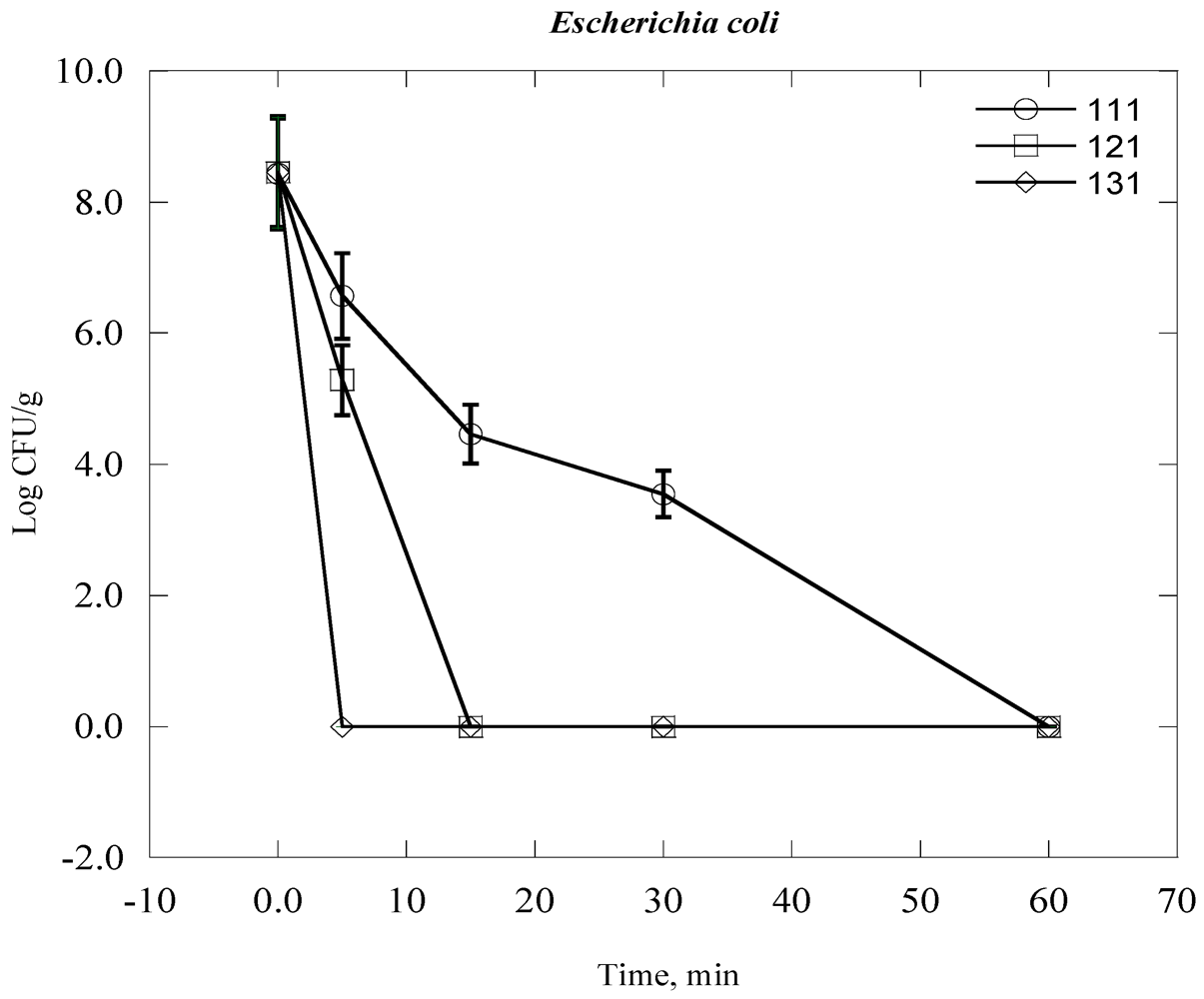
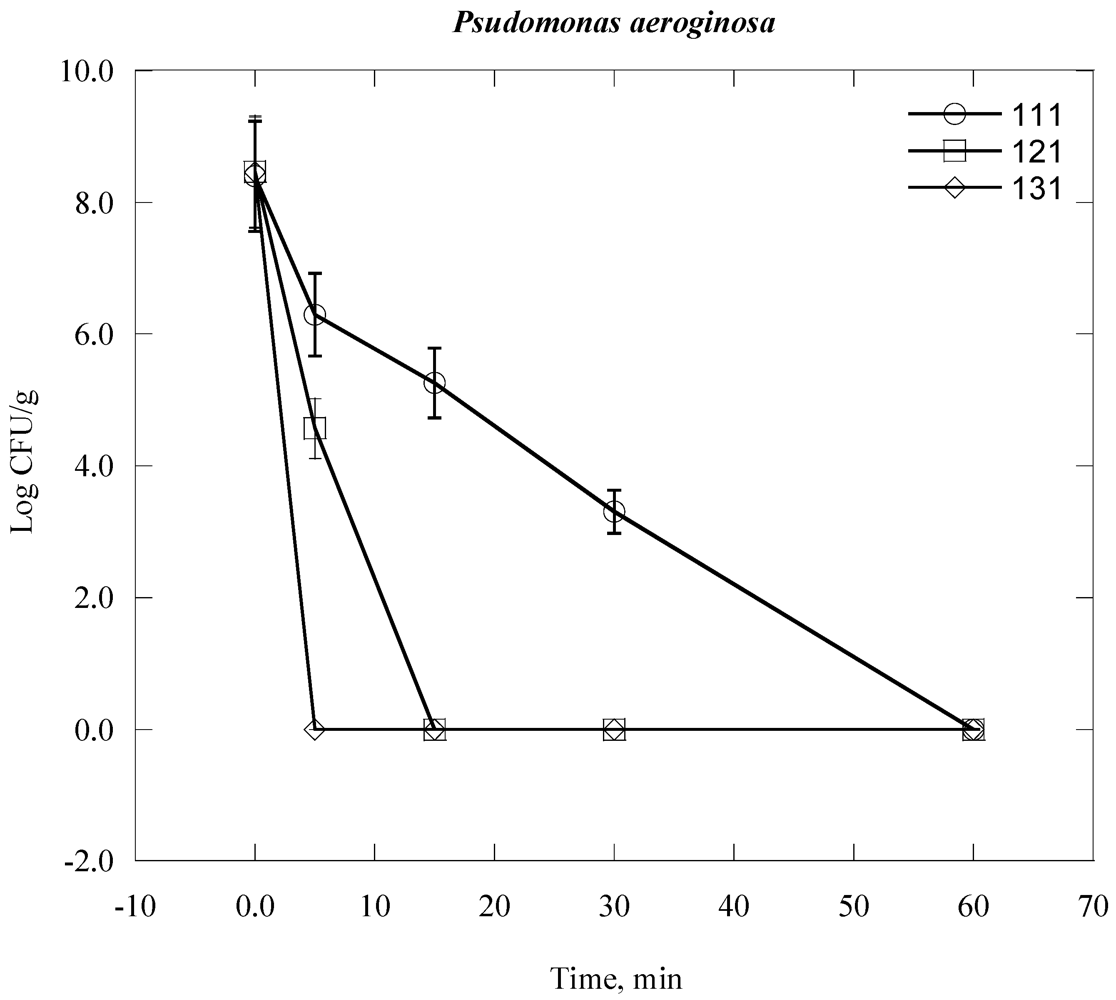
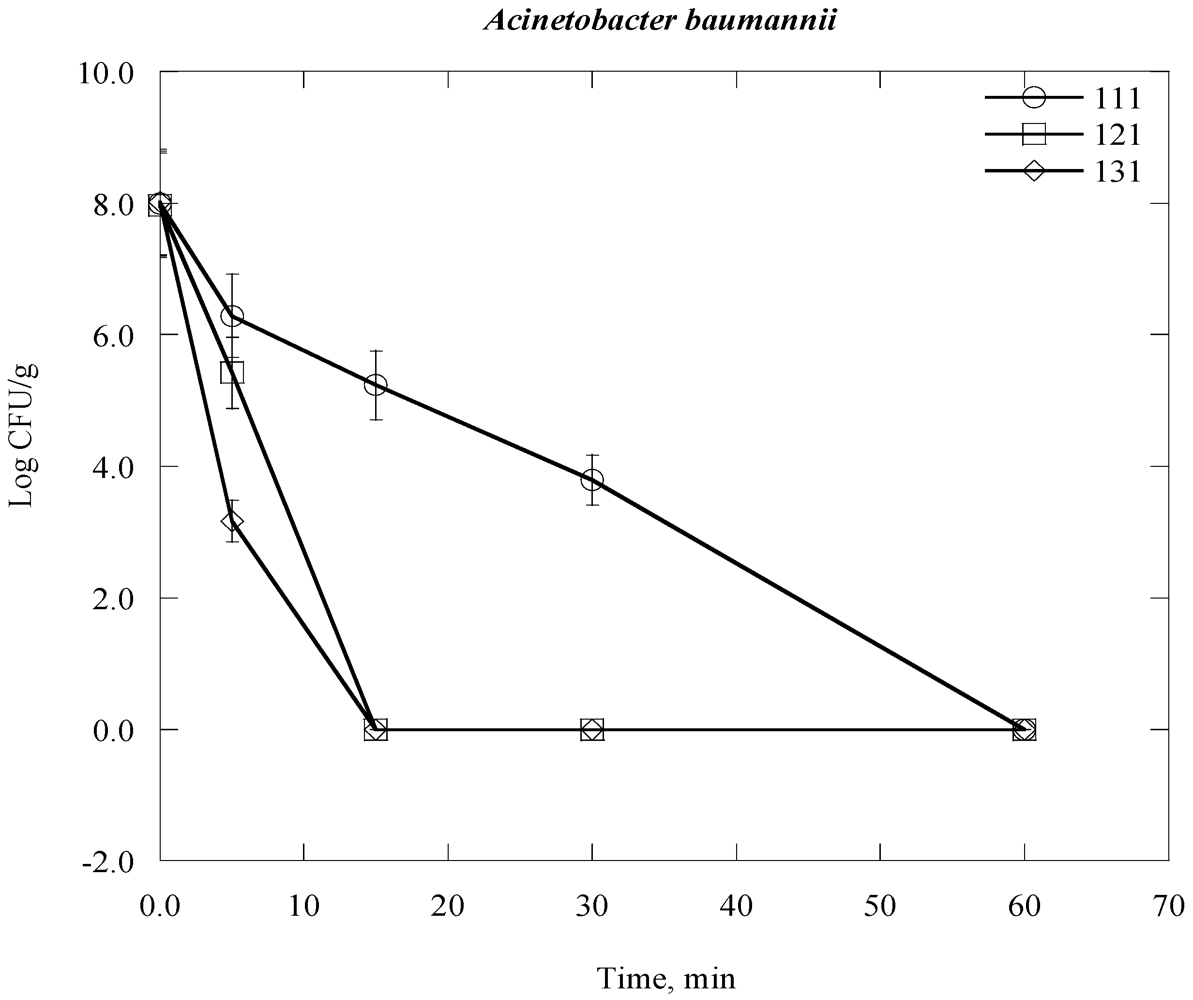
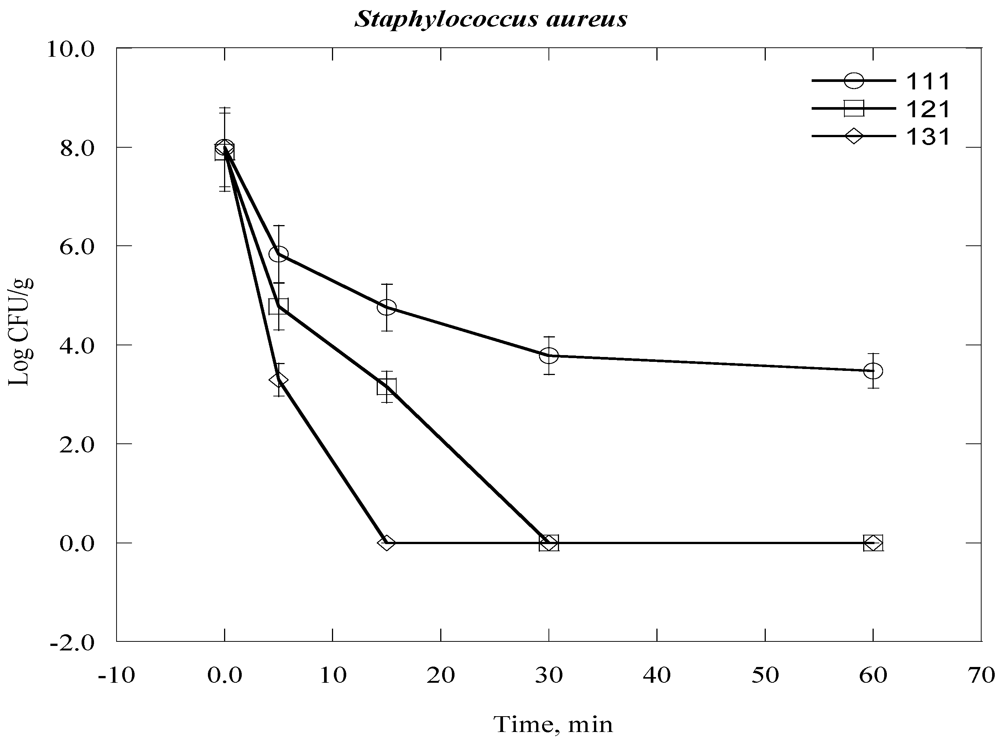
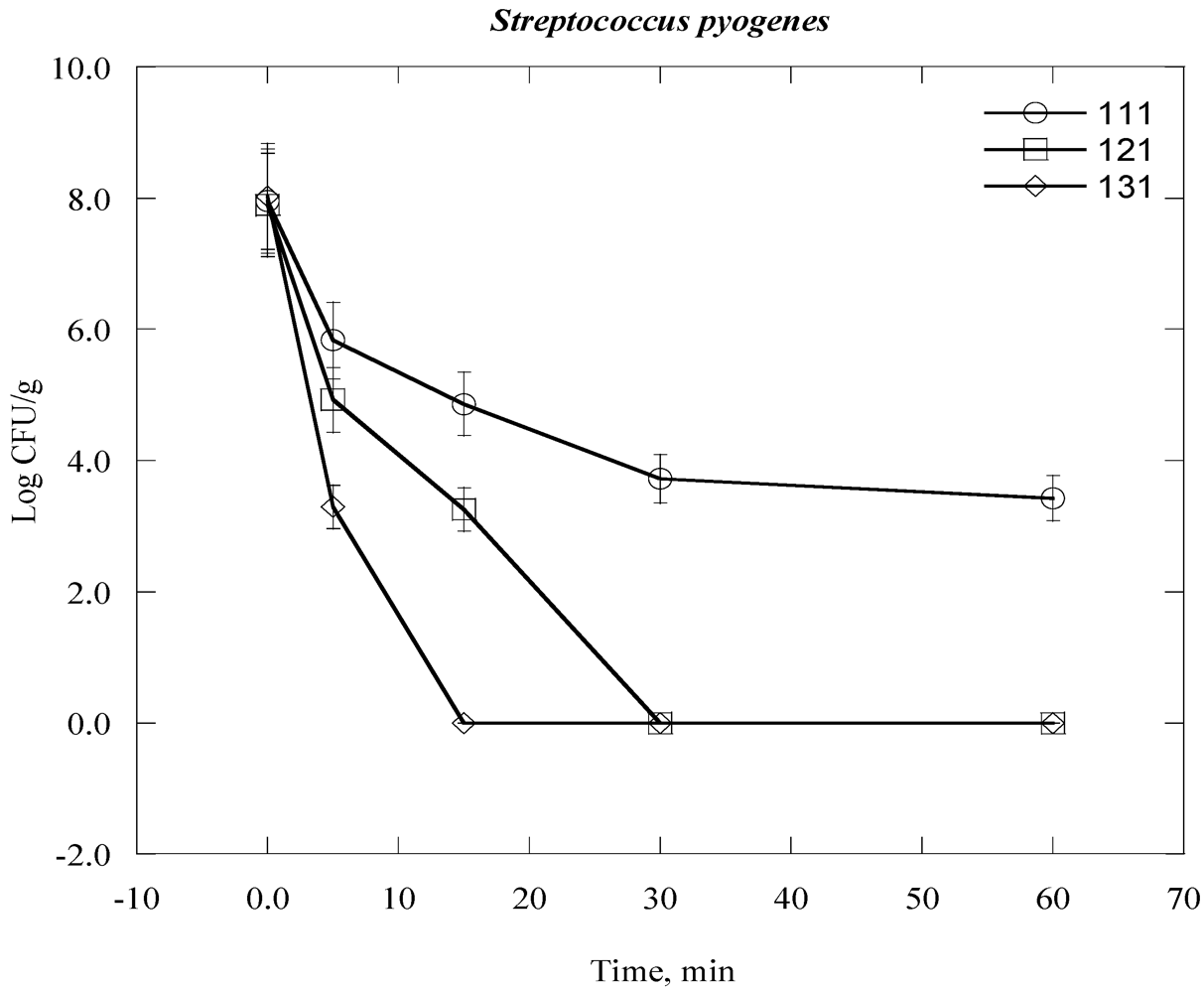
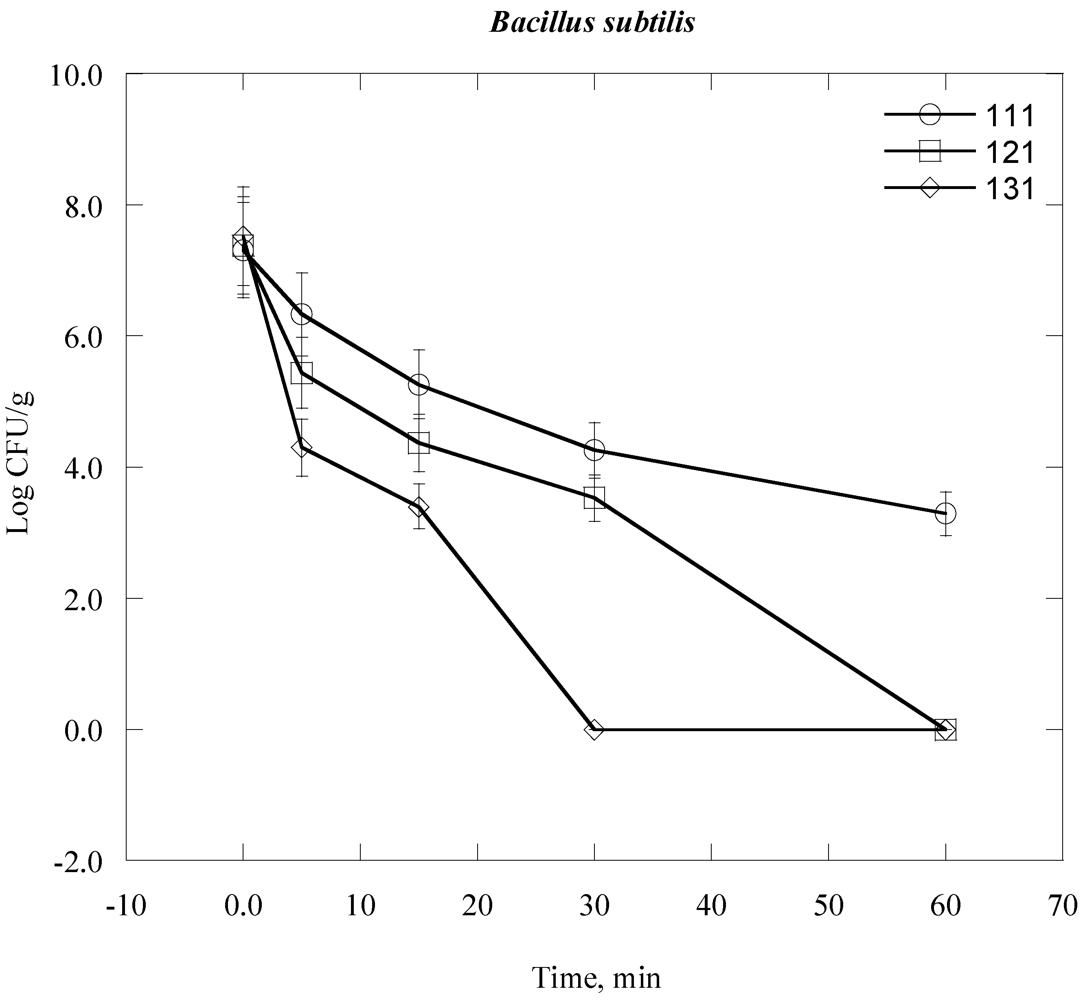
| Bacteria Time, day | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|---|
| Escherichia coli | × | × | × | × | × | √ | √ | √ |
| Psudomonas aeroginosa | × | × | × | × | × | × | √ | √ |
| Acinetobacter baumannii | × | × | × | × | √ | √ | √ | √ |
| Staphylococcus aurous | × | × | × | √ | √ | √ | √ | √ |
| Streptococcus pyogenes | × | × | × | × | √ | √ | √ | √ |
| Bacillus subtilis | × | × | √ | √ | √ | √ | √ | √ |
| Gram staining reaction | Oxidase Test | Fermentation Test | TSI Test | API Kit | Name of Bacteria |
|---|---|---|---|---|---|
| Gram negative-rod shaped | -ve | NLF | -ve | API 20NE | Acinetobacter baumannii |
| Gram negative-rod shaped | +ve | NLF | -ve | API 20NE | Pseudomonas aeruginosa |
| Gram negative-rod shaped | -ve | LF | +ve | API 20 E | Escherichia coli |
| Gram staining reaction | catalase Test | API Kit | Name of Bacteria |
|---|---|---|---|
| Gram positive cocci | +ve | API 20 staph | Staphylococcus aureous |
| Gram positive cocci | -ve | API 20 strep | Streptococcus pyogene |
| Gram positive bacilli | +ve | API 50 CHB | Bacillus subtilis |
4. Conclusions
Acknowledgments
References
- Starting Health Care Waste Management in Medical Institutions, Health Care Waste Practical Information Series 1; World Health Organization: Geneva, Switzerland, 2007.
- Nemathaga, F.; Maringa, S.; Chimuka, L. Hospital solid waste management practices in Limpopo Province, South Africa: A case study of two hospitals. Waste Manag. 2008, 28, 1236–1245. [Google Scholar]
- Jang, Y.-C.; Lee, C.; Yoon, O.-S.; Kim, H. Medical waste management in Korea. J. Environ. Manag. 2006, 80, 107–115. [Google Scholar]
- Oweis, R.; Al-Widyan, M.; Al-Limoon, O. Medical waste management in Jordan: A study at the King Hussein Medical Center. Waste Manag. 2005, 25, 622–625. [Google Scholar]
- Alagöz, A.Z.; Kocasoy, G. Determination of the best appropriate management methods for the health-care wastes in İstanbul. Waste Manag. 2008, 28, 1227–1235. [Google Scholar]
- Blenkharn, J.I. Safe disposal and effective destruction of clinical wastes. J. Hosp. Infect. 2005, 60, 295–297. [Google Scholar]
- Waste Management Licensing Technical Guidance on Clinical Waste Management Facilities; Environment Agency: Bristol, UK, 2003; pp. 17–102.
- Pruss, A.; Giroult, E.; Rushbrook, P.E. Safe Management of Wastes from Healthcare Activities; World Health Organization: Geneva, Switzerland, 1999. [Google Scholar]
- Saini, S.; Das, K.B.; Kapil, A.; Nagargan, S.; Sarma, R.K. The study of bacterial flora of different types in hospital waste: Evaluation of waste treatment at aims hospital, New Delhi. Southeast Asian J. Trop. Med. Public Health 2004, 35, 986–989. [Google Scholar]
- Coker, A.; Sangodoyin, A.; Sridhar, M.; Booth, C.; Olomolaiye, P.; Hammond, F. Medical waste management in Ibadan, Nigeria: Obstacles and prospects. Waste Manag. 2009, 29, 804–811. [Google Scholar]
- Park, H.; Lee, H.; Kim, M.; Lee, J.; Seong, S.-Y.; Ko, G. Detection and hazard assessment of pathogenic microorganisms in medical waste. J. Environ. Sci. Health A 2009, 44, 995–1003. [Google Scholar]
- Ananth, A.P.; Prashanthini, V.; Visvanathan, C. Healthcare waste management in Asia. Waste Manag. 2010, 30, 154–161. [Google Scholar]
- Gai, R.; Xu, L.; Li, H.; Zhou, C.; He, J.; Shirayama, Y.; Tang, W.; Kuroiwa, C. Investigation of health care waste management in Binzhou District, China. Waste Manag. 2010, 30, 246–250. [Google Scholar]
- Lee, B.-K.; Ellenbecker, M.J.; Moure-Ersaso, R. Alternatives for treatment and disposal cost reduction of regulated medical wastes. Waste Manag. 2004, 24, 143–151. [Google Scholar]
- Gautam, V.; Thapar, R.; Sharma, M. Biomedical waste management: Incineration vs. environmental safety. Indian J. Med. Microbiol. 2010, 28, 191–192. [Google Scholar] [CrossRef]
- Lindner, G.; Jenkins, A.C.; McCormack, J.; Adrian, R.C. Dioxins and furans in emissions from medical waste incinerators. Chemosphere 1990, 20, 1793–1800. [Google Scholar]
- Matsui, M.; Kashima, Y.; Kawano, M.; Matsuda, M.; Ambe, K.; Wakimoto, T.; Doi, R. Dioxin-like potencies and extractable organohalogens (EOX) in medical, municipal and domestic waste incinerator ashes in Japan. Chemosphere 2003, 53, 971–980. [Google Scholar]
- Nouwen, J.; Cornelis, C.; de Fré, R.; Wevers, M.; Viaene, P.; Mensink, C.; Patyn, J.; Verschaeve, L.; Hooghe, R.; Maes, A.; et al. Health risk assessment of dioxin emissions from municipal waste incinerators: The Neerlandquarter (Wilrijk, Belgium). Chemosphere 2001, 43, 909–923. [Google Scholar] [CrossRef]
- Blenkharn, J.I. The disposal of clinical wastes. J. Hosp. Infect. 1995, 30, 514–520. [Google Scholar]
- Sawalem, M.; Selic, E.; Herbell, J.D. Hospital waste management in Libya: A case study. Waste Manag. 2009, 29, 1370–1375. [Google Scholar]
- Hossain, M.S.; Santhanam, A.; Nik Norulaini, N.A.; Mohd Omar, A.K. Clinical solid waste management practices and its impact on human health and environment—A review. Waste Manag. 2011, 31, 754–766. [Google Scholar]
- Tsakona, M.; Anagnostopoulou, E.; Gidarakos, E. Hospital waste management and toxicity evaluation: A case study. Waste Manag. 2007, 27, 912–920. [Google Scholar]
- State and Territorial Association on Alternative Treatment Technologies (STAATT) III, Executive Summary and Daily Discussions; STAATT: Orlando, FL, USA, 2005.
- Autoclave Processing of Medical Waste; AFEC Solutions LLC: Hackettstown, NJ, USA, 2007.
- Oliveira, E.A.; Nogueira, N.G.P.; Innocentini, M.D.M.; Pisani, R., Jr. Microwave inactivation of Bacillus atrophaeus spores in healthcare waste. Waste Manag. 2010, 30, 2327–2335. [Google Scholar]
- Dehghani, F. Biochemical Engineering Notes; Department of Chemical Engineering, University of Sydney: Sydney, Australia, 2006. [Google Scholar]
- Zhang, J.; Dalal, N.; Matthews, M.A.; Waller, L.N.; Saunders, C.; Fox, K.F.; Fox, A. Supercritical carbon dioxide and hydrogen peroxide cause mild changes in spore structures associated with high killing rate of Bacillus anthracis. J. Microbiol. Methods 2007, 70, 442–451. [Google Scholar]
- Rutala, W.A. Disinfection and sterilization of patient-care items. Infect. Control Hosp. Epidemiol. 1996, 17, 377–384. [Google Scholar]
- Navarre, W.W.; Schneewind, O. Surface proteins of Gram-positive bacteria and mechanisms of their targeting to the cell wall envelope. Microbiol. Mol. Biol. Rev. 1999, 63, 174–229. [Google Scholar]
- Moesby, L.; Hansen, E.W.; Christensen, J.D.; Høyer, C.H.; Juhl, G.L.; Olsen, H.B. Dry and moist heat sterilization cannot inactivate pyrogenicity of Gram positive microorganisms. Eur. J. Pharm. Sci. 2005, 26, 318–323. [Google Scholar]
- Grover-Lakomia, L.I.; Fong, E. Microbiology for Health Careers: The Cellular Structure and Characteristics of Prokarvotic Cells, 6th ed; Susan Simpfenderfer: Albany, NY, USA, 1998. [Google Scholar]
- Bähr, C.; Stammen, E.; Thiele, R.; Böhm, S.; Dilger, K.; Büchs, J. Withstanding frequent steam sterilisation: Innovative technique to bond glass and stainless steel composites in Biotechnology and endoscopic medicine. Int. J. Adhes. Adhes. 2012, 33, 15–25. [Google Scholar]
- Zhang, J.; Davis, T.A.; Matthews, M.A.; Drews, M.J.; LaBerge, M.; An, Y.H. Sterilization using high-pressure carbon dioxide. J. Supercrit. Fluids 2006, 38, 354–372. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hossain, M.S.; Balakrishnan, V.; Rahman, N.N.N.A.; Sarker, M.Z.I.; Kadir, M.O.A. Treatment of Clinical Solid Waste Using a Steam Autoclave as a Possible Alternative Technology to Incineration. Int. J. Environ. Res. Public Health 2012, 9, 855-867. https://doi.org/10.3390/ijerph9030855
Hossain MS, Balakrishnan V, Rahman NNNA, Sarker MZI, Kadir MOA. Treatment of Clinical Solid Waste Using a Steam Autoclave as a Possible Alternative Technology to Incineration. International Journal of Environmental Research and Public Health. 2012; 9(3):855-867. https://doi.org/10.3390/ijerph9030855
Chicago/Turabian StyleHossain, Md. Sohrab, Venugopal Balakrishnan, Nik Norulaini Nik Ab Rahman, Md. Zaidul Islam Sarker, and Mohd Omar Ab Kadir. 2012. "Treatment of Clinical Solid Waste Using a Steam Autoclave as a Possible Alternative Technology to Incineration" International Journal of Environmental Research and Public Health 9, no. 3: 855-867. https://doi.org/10.3390/ijerph9030855
APA StyleHossain, M. S., Balakrishnan, V., Rahman, N. N. N. A., Sarker, M. Z. I., & Kadir, M. O. A. (2012). Treatment of Clinical Solid Waste Using a Steam Autoclave as a Possible Alternative Technology to Incineration. International Journal of Environmental Research and Public Health, 9(3), 855-867. https://doi.org/10.3390/ijerph9030855





