The Public Health Impact of Coccidioidomycosis in Arizona and California
Abstract
:1. Introduction
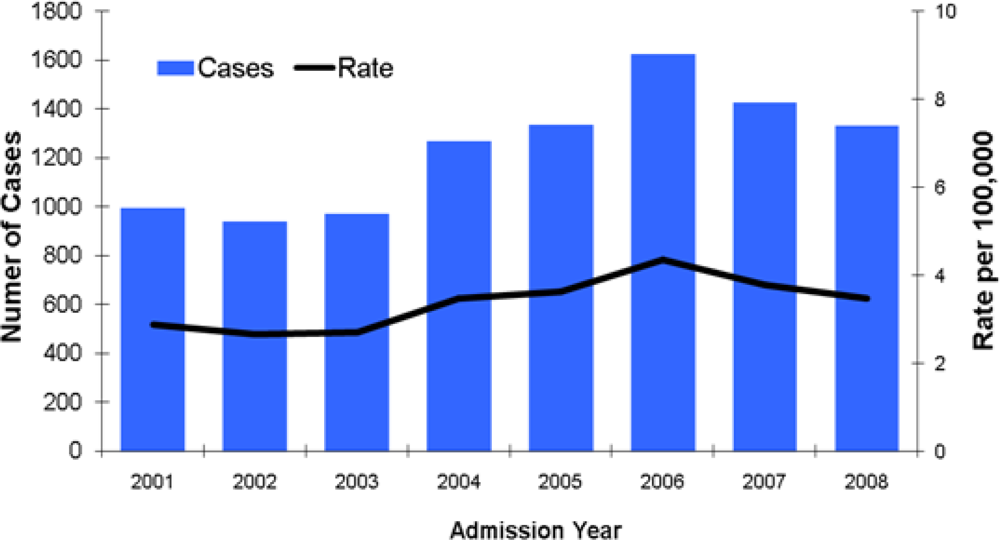
2. The Public Health Impact of Coccidioidomycosis in Arizona
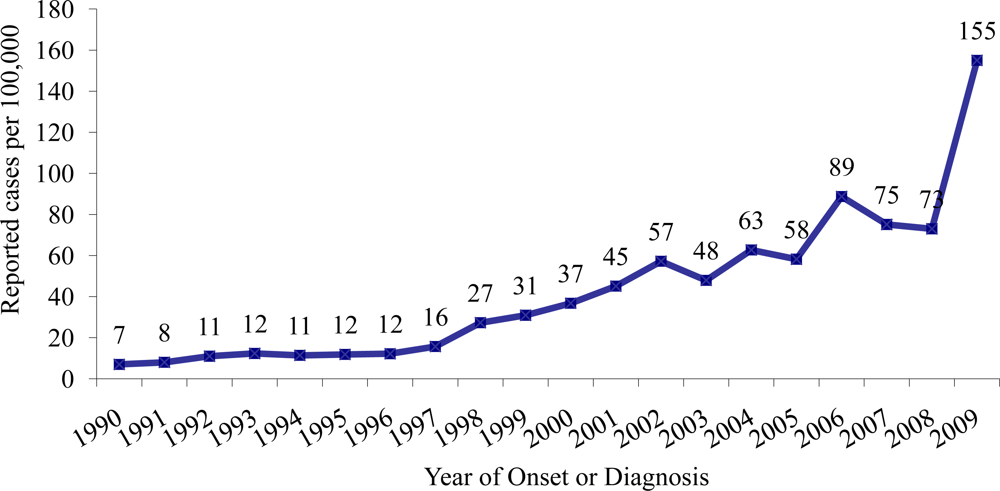
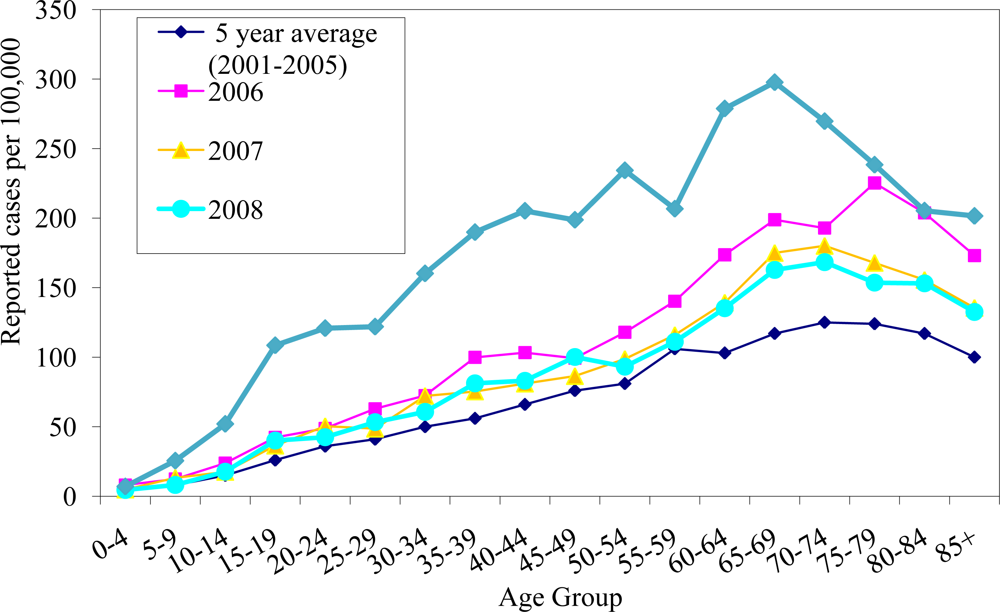
2.1. Epidemiology of Coccidioidomycosis
2.2. Monitoring Laboratory Reporting of Coccidioidomycosis
2.3. Coccidioidomycosis as a Cause of Community-Acquired Pneumonia
2.4. Enhanced Surveillance of Coccidioidomycosis
2.5. Conclusions
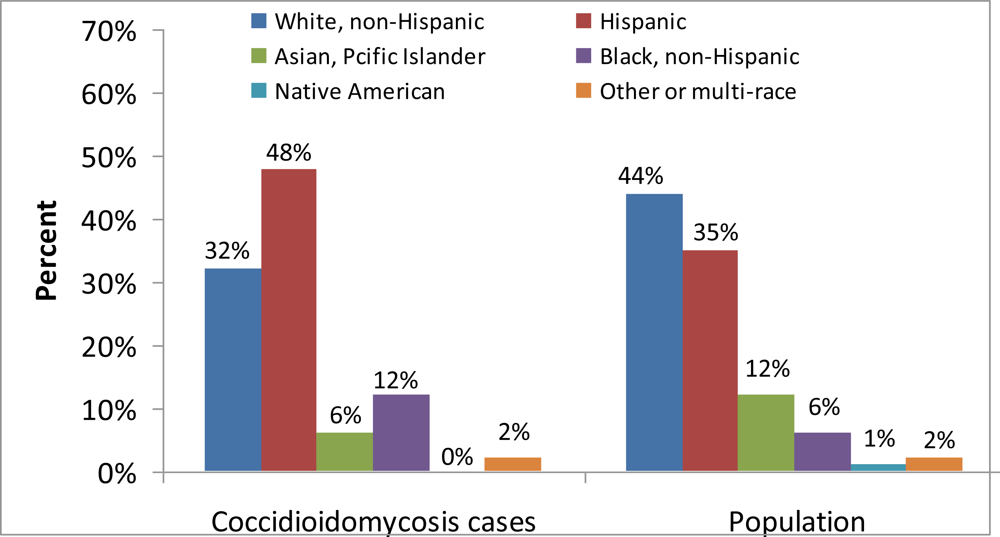
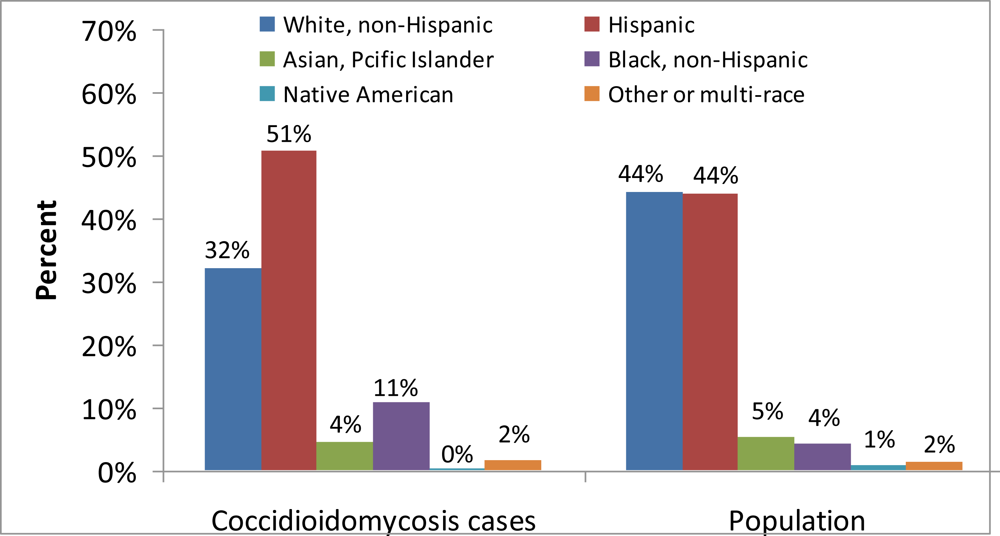
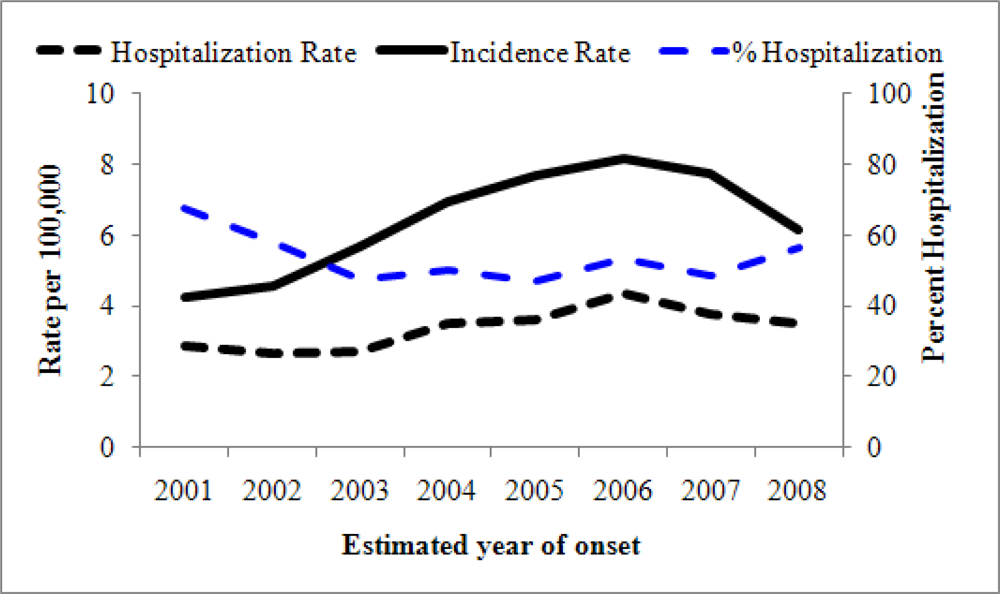
3. The Public Health Impact of Coccidioidomycosis in California, 2001–2009
3.1. Materials and Methods
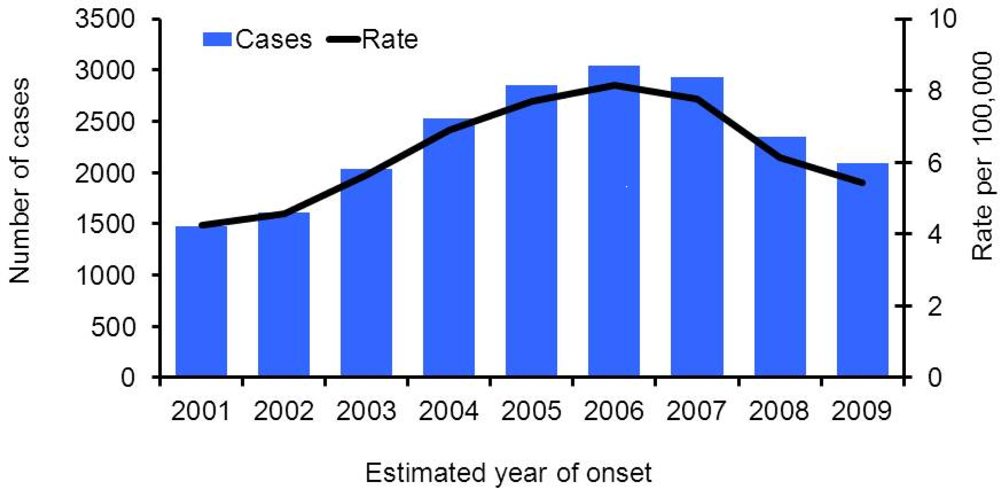
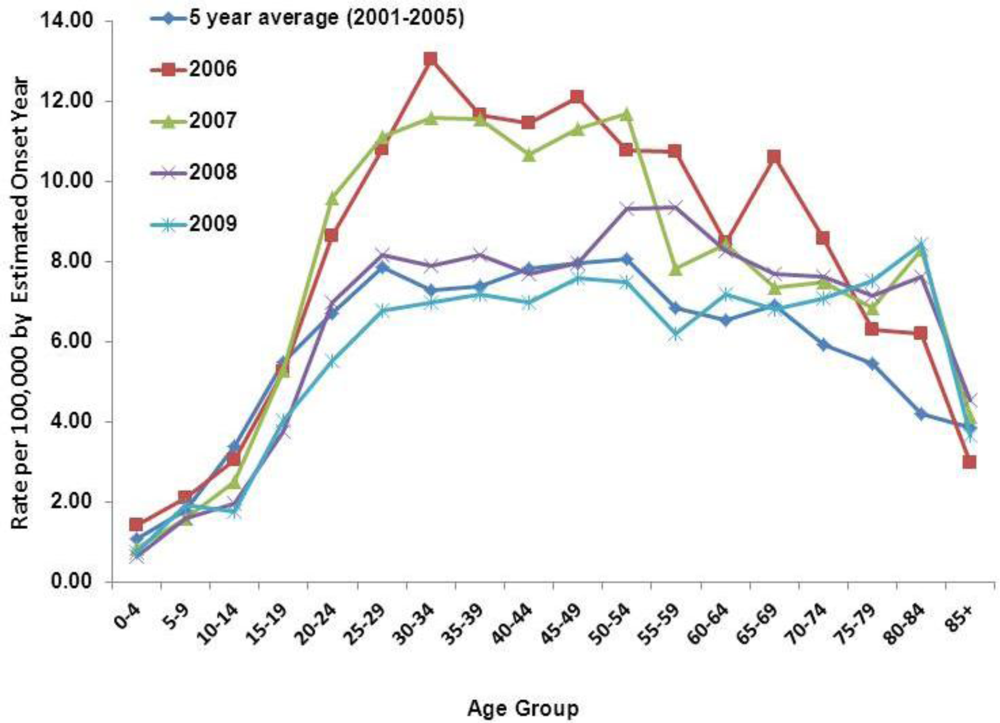
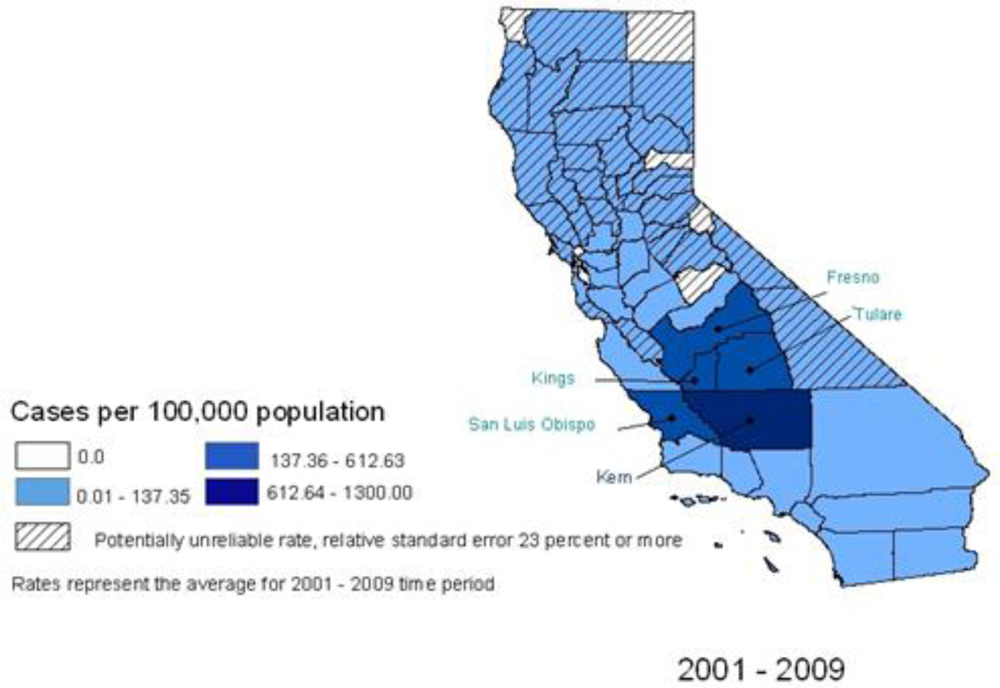
3.2. Results
3.3. Discussion, Limitations, and Comments
4. Diagnosis and Treatment of Coccidioidomycosis
4.1. Improving Diagnostics for Early Coccidioidal Infections
4.2. Current and Future Therapies for Coccidioidomycosis
5. A Preventive Vaccine for Coccidioidomycosis
6. Discussion
Acknowledgments
References
- Fisher, F; Bultman, MW; Pappagianis, D. Operational Guidelines for Geological Fieldwork in Areas Endemic for Coccidioidomycosis (Valley Fever); U.S. Geological Survey Open-File Report; U.S. Department of the Interior: Washington, DC, USA, 2000; pp. 1–16. [Google Scholar]
- Stevens, DA. Coccidioidomycosis. N. Engl. J. Med 1995, 332, 1077–1082. [Google Scholar]
- Chiller, TM; Galgiani, JN; Stevens, DA. Coccidioidomycosis. Infect. Dis. Clin. North Am 2003, 17, 41–57. [Google Scholar]
- Saubolle, MA; McKellar, PP; Sussland, D. Epidemiologic, clinical, and diagnostic aspects of coccidioidomycosis. J. Clin. Microbiol 2007, 45, 26–30. [Google Scholar]
- Blair, JE. Coccidioidomycosis in patients who have undergone transplantation. Ann. NY Acad. Sci 2007, 1111, 365–376. [Google Scholar]
- Kirkland, TN; Fierer, J. Coccidioidomycosis: A reemerging infectious disease. Emerg. Infect. Dis 1996, 2, 192–199. [Google Scholar]
- Ampel, NM. The complex immunology of human coccidioidomycosis. Ann. NY Acad. Sci 2007, 1111, 245–258. [Google Scholar]
- Masannat, FY; Ampel, NM. Coccidioidomycosis in patients with HIV-1 infection in the era of potent antiretroviral therapy. Clin. Infect. Dis 2010, 50, 1–7. [Google Scholar]
- Santelli, AC; Blair, JE; Roust, LR. Coccidioidomycosis in patients with diabetes mellitus. Am. J. Med 2006, 119, 964–969. [Google Scholar]
- Pappagianis, D; Lindsay, S; Beall, S; Williams, P. Ethnic background and the clinical course of coccidioidomycosis. Am. Rev. Resp. Dis 1979, 120, 959–961. [Google Scholar]
- Ruddy, BE; Mayer, AP; Ko, MG; Labonte, HR; Borovansky, JA; Boroff, ES; Blair, JE. Coccidioidomycosis in African Americans. Mayo Clin. Proc 2011, 86, 63–69. [Google Scholar]
- Galgiani, JN; Ampel, NM; Blair, JE; Catanzaro, A; Johnson, RH; Stevens, DA; Williams, PL. Coccidioidomycosis. Clin. Infect. Dis 2005, 41, 1217–1223. [Google Scholar]
- Centers for Disease Control and Prevention. Summary of notifiable diseases—United States, 2006. Morb. Mortal. Wkly. Rep 2008, 55, 1–94. [Google Scholar]
- Centers for Disease Control and Prevention. Coccidioidomycosis—Arizona, 1990–1995. Morb. Mortal. Wkly. Rep 1996, 45, 1069–1073. [Google Scholar]
- Ampel, NM; Mosley, DG; England, B; Vertz, PD; Komatsu, K; Hajjeh, RA. Coccidioidomycosis in Arizona: Increase in incidence from 1990 to 1995. Clin. Infect. Dis 1998, 27, 1528–1530. [Google Scholar]
- Sunenshine, RH; Anderson, S; Erhard, L; Vossbrink, A; Kelly, PC; Engelthaler, D; Komatsu, K. Public health surveillance for coccidioidomycosis in Arizona. Ann. NY Acad. Sci 2007, 1111, 96–102. [Google Scholar]
- Kaufman, L; Sekhon, AS; Moledina, N; Jalbert, M; Pappagianis, D. Comparative evaluation of commercial Premier EIA and microimmunodiffusion and complement fixation tests for Coccidioides immitis antibodies. J. Clin. Microbiol 2007, 33, 618–619. [Google Scholar]
- Wieden, MA; Lundergan, LL; Blum, J; Delgado, KL; Coolbaugh, R; Howard, R; Peng, T; Pugh, E; Reis, N; Theis, J; Galgianim, JN. Detection of coccidioidal antibodies by 33-kDa spherule antigen, Coccidioides EIA, and standard serologic tests in sera from patients evaluated for coccidioidomycosis. J. Infect. Dis 1996, 173, 1273–1277. [Google Scholar]
- Valdivia, L; Nix, D; Wright, M; Lindberg, E; Fagan, T; Lieberman, D; Stoffer, T; Ampel, NM; Galgiani, JN. Coccidioidomycosis as a common cause of community-acquired pneumonia. Emerg. Infect. Dis 2006, 12, 958–962. [Google Scholar]
- Kim, MM; Blair, JE; Carey, EJ; Wu, Q; Smilack, JD. Coccidioidal pneumonia, Phoenix, Arizona, USA, 2000–2004. Emerg. Infect. Dis 2009, 15, 397–401. [Google Scholar]
- Chang, DC; Anderson, S; Wannemuehler, K; Engelthaler, DM; Erhart, L; Sunenshine, RH; Burwell, LA; Park, BJ. Testing for coccidioidomycosis among patients with community-acquired pneumonia. Emerg. Infect. Dis 2008, 14, 1053–1059. [Google Scholar]
- Bartlett, JG; Dowell, SF; Mandell, LA; File, TM, Jr; Musher, DM; Fine, MJ. Practice guidelines for the management of community-acquired pneumonia in adults. Clin. Infect. Dis 2000, 31, 347–382. [Google Scholar]
- Tsang, CA; Anderson, SM; Imholte, SB; Erhart, LM; Chen, S; Park, BJ; Christ, C; Komatsu, KK; Chiller, T; Sunenshine, RH. Enhanced surveillance of coccidioidomycosis, Arizona, USA, 2007–2008. Emerg. Infect. Dis 2010, 16, 1738–1744. [Google Scholar]
- California Code of Regulations, Title 17, Sections 2500, 2502, & 2505.
- Centers for Disease Control and Prevention. Case Definitions for Infectious Conditions Under Public Health Surveillance. Available online: http://www.cdc.gov/ncphi/disss/nndss/casedef/coccidioid2008.htm (accessed on 11 February 2011).
- Race/Ethnic Population with Age and Sex Detail, 2000–2050; State of California, Department of Finance: Sacramento, CA, USA, July 2007. Available online: http://www.dof.ca.gov/research/demographic/data/race-ethnic/2000-50/ (accessed on 14 April 2011).
- Crum, N; Lamb, C; Utz, G; Amundson, D; Wallace, M. Coccidioidomycosis outbreak among United States Navy SEALs training in a Coccidioides immitis-endemic area—Coalinga, California. J. Infect. Dis 2002, 186, 865–868. [Google Scholar]
- Cummings, KC; McDowell, A; Wheeler, C; McNary, J; Das, R; Vugia, DJ; Mohle-Boetani, JC. Point-source outbreak of coccidioidomycosis in construction workers. Epidemiol. Infect 2010, 138, 507–511. [Google Scholar]
- Lee, R; Crum-Cianflone, N. Increasing incidence and severity of coccidioidomycosis at a naval air station. Mil. Med 2008, 173, 769–775. [Google Scholar]
- Burwell, LA; Park, BJ; Wannemuehler, KA; Kendig, N; Pelton, J; Chaput, E; Jinadu, BA; Emery, K; Chavez, G; Fridkin, SK. Outcomes among inmates treated for coccidioidomycosis at a correctional institution during a community outbreak, Kern County, California, 2004. Clin. Infect. Dis 2009, 49, e113–e119. [Google Scholar]
- Pappagianis, D. Coccidioidomycosis in California state correctional institutions. Ann. NY Acad. Sci 2007, 1111, 103–111. [Google Scholar]
- Fiese, MJ. Coccidioidomycosis; Charles C Thomas: Springfield, IL, USA, 1958. [Google Scholar]
- Fiese, MJ. Treatment of disseminated coccidioidomycosis with amphotericin B. Report of a case. Calif. Med 1957, 86, 119–120. [Google Scholar]
- Smith, CE. Epidemiology of acute coccidioidomycosis with erythema nodosum. Am. J. Public Health 1940, 30, 600–611. [Google Scholar]
- Shelton, RM. A survey of coccidioidomycosis at Camp Roberts California. JAMA 1942, 118, 1186–1190. [Google Scholar]
- DiTomasso, JP; Ampel, NM; Sobonya, RE; Bloom, JW. Bronchoscopic diagnosis of pulmonary coccidioidomycosis. Comparison of cytology, culture, and transbronchial biopsy. Diag. Microbiol. Infect. Dis 1994, 18, 83–87. [Google Scholar]
- Pappagianis, D; Zimmer, BL. Serology of coccidioidomycosis. Clin. Microbiol. Rev 1990, 3, 247–268. [Google Scholar]
- Polage, CR; Billetdeaux, E; Litwin, CM; Petti, CA. Revisiting the Sensitivity of Serologic Testing in Culture Positive Coccidioidomycosis. Proceedings of the Annual Meeting of the American Society for Microbiology, Orlando, FL, USA, 21– 25 May 2006.
- Clark, KA; McAllister, D. Direct detection of Coccidioides immitis in clinical specimens using target amplification. In Coccidioidomycosis. Proceedings of the 5th International Conference; Einstein, HE, Catanzaro, A, Eds.; National Foundation for Infectious Diseases: Washington, DC, USA, 1996; pp. 129–136. [Google Scholar]
- Binnicker, MJ; Buckwalter, SP; Eisberner, JJ; Stewart, RA; McCullough, AE; Wohlfiel, SL; Wengenack, NL. Detection of Coccidioides species in clinical specimens by real-time PCR. J. Clin. Microbiol 2007, 45, 173–178. [Google Scholar]
- Johnson, SM; Simmons, KA; Pappagianis, D. Amplification of coccidioidal DNA in clinical specimens by PCR. J. Clin. Microbiol 2004, 42, 1982–1985. [Google Scholar]
- Vucicevic, D; Blair, JE; Binnicker, MJ; McCullough, AE; Kusne, S; Vikram, HR; Parish, JM; Wengenack, NL. The utility of Coccidioides polymerase chain reaction testing in the clinical setting. Mycopathologia 2010, 170, 345–351. [Google Scholar]
- Yoshinoya, S; Cox, RA; Pope, RM. Circulating immune complexes in coccidioidomycosis. Detection and characterization. J. Clin. Invest 1980, 66, 655–663. [Google Scholar]
- Weiner, MH. Antigenemia detected in human coccidioidomycosis. J. Clin. Microbiol 1983, 18, 136–142. [Google Scholar]
- Galgiani, JN; Dugger, KO; Ito, JI; Wieden, MA. Antigenemia in primary coccidioidomycosis. Am. J. Trop. Med. Hyg 1984, 33, 645–649. [Google Scholar]
- Galgiani, JN; Grace, GM; Lundergan, LL. New serologic tests for early detection of coccidioidomycosis. J. Infect. Dis 1991, 163, 671–674. [Google Scholar]
- Durkin, M; Connollym, P; Kuberski, T; Myers, R; Kubak, BM; Bruckner, D; Pegues, D; Wheat, LJ. Diagnosis of coccidioidomycosis with use of the Coccidioides antigen enzyme immunoassay. Clin. Infect. Dis 2008, 47, e69–e73. [Google Scholar]
- Johnson, RH; Einstein, HE. Amphotericin B and coccidioidomycosis. Ann. NY Acad. Sci 2007, 1111, 434–441. [Google Scholar]
- Ampel, NM. Combating opportunistic infections: Coccidioidomycosis. Expert. Opin. Pharmacother 2004, 5, 255–261. [Google Scholar]
- Clemons, KV; Capilla, J; Sobel, RA; Martinez, M; Tong, AJ; Stevens, DA. Comparative efficacies of lipid-complexed amphotericin B and liposomal amphotericin B against coccidioidal meningitis in rabbits. Antimicrob. Agents Chemother 2009, 53, 1858–1862. [Google Scholar]
- Clemons, KV; Sobel, RA; Williams, PL; Pappagianis, D; Stevens, DA. Efficacy of intravenous liposomal amphotericin B (AmBisome) against coccidioidal meningitis in rabbits. Antimicrob. Agents Chemother 2002, 46, 2420–2426. [Google Scholar]
- Johnson, RH; Einstein, HE. Coccidioidal meningitis. Clin. Infect. Dis 2006, 42, 103–107. [Google Scholar]
- Stevens, DA; Shatsky, SA. Intrathecal amphotericin in the management of coccidioidal meningitis. Semin. Respir. Infect 2001, 16, 263–269. [Google Scholar]
- Galgiani, JN; Ampel, NM; Blair, JE; Catanzaro, A; Johnson, RH; Stevens, DA; Williams, PL. Coccidioidomycosis. Clin. Infect. Dis 2005, 41, 1217–1223. [Google Scholar]
- Stevens, DA. Miconazole in the treatment of coccidioidomycosis. Drugs 1983, 26, 347–354. [Google Scholar]
- Galgiani, JN; Stevens, DA; Graybill, JR; Dismukes, WE; Cloud, GA. Ketoconazole therapy of progressive coccidioidomycosis. Comparison of 400- and 800-mg doses and observations at higher doses. Am. J. Med 1988, 84, 603–610. [Google Scholar]
- Graybill, JR; Stevens, DA; Galgiani, JN; Sugar, AM; Craven, PC; Gregg, C; Huppert, M; Cloud, G; Dismukes, WE. Ketoconazole treatment of coccidioidal meningitis. Ann. NY Acad. Sci 1988, 544, 488–496. [Google Scholar]
- Galgiani, JN; Catanzaro, A; Cloud, GA; Johnson, RH; Williams, PL; Mirels, LF; Nassar, F; Lutz, JE; Stevens, DA; Sharkey, PK. Comparison of oral fluconazole and itraconazole for progressive, nonmeningeal coccidioidomycosis. A randomized, double-blind trial. Mycoses Study Group. Ann. Intern. Med 2000, 133, 676–686. [Google Scholar]
- Kelly, PC. Coccidioidal Meningitis. In Coccidioidomycosis: A Text; Stevens, DA, Ed.; Plenum Medical Book Company: New York, NY, USA, 1980; pp. 163–193. [Google Scholar]
- Tucker, RM; Denning, DW; Dupont, B; Stevens, DA. Itraconazole therapy for chronic coccidioidal meningitis. Ann. Intern. Med 1990, 112, 108–112. [Google Scholar]
- Galgiani, JN; Catanzaro, A; Cloud, GA; Higgs, J; Friedman, BA; Larsen, RA; Graybill, JR. Fluconazole therapy for coccidioidal meningitis. The NIAID-Mycoses Study Group. Ann. Intern. Med 1993, 119, 28–35. [Google Scholar]
- Catanzaro, A; Cloud, GA; Stevens, DA; Levine, BE; Williams, PL; Johnson, RH; Rendon, A; Mirels, LF; Lutz, JE; Holloway, M; Galgiani, JN. Safety, tolerance, and efficacy of posaconazole therapy in patients with nonmeningeal disseminated or chronic pulmonary coccidioidomycosis. Clin. Infect. Dis 2007, 45, 562–568. [Google Scholar]
- Stevens, DA; Rendon, A; Gaona-Flores, V; Catanzaro, A; Anstead, GM; Pedicone, L; Graybill, JR. Posaconazole therapy for chronic refractory coccidioidomycosis. Chest 2007, 132, 952–958. [Google Scholar]
- Prabhu, RM; Bonnell, M; Currier, BL; Orenstein, R. Successful treatment of disseminated nonmeningeal coccidioidomycosis with voriconazole. Clin. Infect. Dis 2004, 39, e74–e77. [Google Scholar]
- Proia, LA; Tenorio, AR. Successful use of voriconazole for treatment of Coccidioides meningitis. Antimicrob. Agents Chemother 2004, 48, 2341. [Google Scholar]
- Cortez, KJ; Walsh, TJ; Bennett, JE. Successful treatment of coccidioidal meningitis with voriconazole. Clin. Infect. Dis 2003, 36, 1619–1622. [Google Scholar]
- Ampel, NM; Giblin, A; Mourani, JP; Galgiani, JN. Factors and outcomes associated with the decision to treat primary pulmonary coccidioidomycosis. Clin. Infect. Dis 2009, 48, 172–178. [Google Scholar]
- Hector, RF; Zimmer, BL; Pappagianis, D. Evaluation of nikkomycins X and Z in murine models of coccidioidomycosis, histoplasmosis, and blastomycosis. Antimicrob. Agents Chemother 1990, 34, 587–593. [Google Scholar]
- Nix, DE; Hoover, SE; Thompson, C; Hanan, NJ; Chavez, SE; Galgiani, JN. Pharmacokinetics and Safety of Nikkomycin Z Following Multiple Rising Doses in Healthy Subjects. Proceedings of the 50th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC), Boston, MA, USA, 12– 15 September 2010. Abstract no. A1-031..
- Ostrosky-Zeichner, L; Casadevall, A; Galgiani, JN; Odds, FC; Rex, JH. An insight into the antifungal pipeline: selected new molecules and beyond. Nat. Rev. Drug Discov 2010, 9, 719–727. [Google Scholar]
- Pappagianis, D. Seeking a vaccine against Coccidioides immitis and serologic studies: Expectations and realities. Fungal Genet. Biol 2001, 32, 1–9. [Google Scholar]
- Stevens, DA. Coccidioidomycosis: A Text; Plenum Medical: New York, NY, USA, 1980. [Google Scholar]
- Pappagianis, D. Epidemiology of coccidioidomycosis. Curr. Top. Med. Mycol 1988, 2, 199–238. [Google Scholar]
- Levine, HB; Cobb, JM; Smith, CE. Immunity to coccidioidomycosis induced in mice by purified spherule, arthrospore, and mycelial vaccines. Trans. NY Acad. Sci 1960, 22, 436–447. [Google Scholar]
- Pappagianis, D; Levine, HB. The present status of vaccination against coccidioidomycosis in man. Am. J. Epidemiol 1975, 102, 30–41. [Google Scholar]
- Pappagianis, D. Evaluation of the protective efficacy of the killed Coccidioides immitis spherule vaccine in humans. The Valley Fever Vaccine Study Group. Am. Rev. Respir. Dis 1993, 148, 656–660. [Google Scholar]
- Delgado, N; Xue, J; Yu, JJ; Hung, CY; Cole, GT. A recombinant beta-1,3-glucanosyltransferase homolog of Coccidioides posadasii protects mice against coccidioidomycosis. Infect Immun 2003, 71, 3010–3019. [Google Scholar]
- Ivey, FD; Magee, DM; Woitaske, MD; Johnston, SA; Cox, RA. Identification of a protective antigen of Coccidioides immitis by expression library immunization. Vaccine 2003, 21, 4359–4367. [Google Scholar]
- Johnson, SM; Lerche, NW; Pappagianis, D; Yee, JL; Galgiani, JN; Hector, RF. Safety, antigenicity, and efficacy of a recombinant coccidioidomycosis vaccine in cynomolgus macaques (Macaca fascicularis). Ann. NY Acad. Sci 2007, 1111, 290–300. [Google Scholar]
- Shubitz, LF; Yu, JJ; Hung, CY; Kirkland, TN; Peng, T; Perrill, R; Simons, J; Xue, J; Herr, RA; Cole, GT; Galgiani, JN. Improved protection of mice against lethal respiratory infection with Coccidioides posadasii using two recombinant antigens expressed as a single protein. Vaccine 2006, 24, 5904–5911. [Google Scholar]
- Tarcha, EJ; Basrur, V; Hung, CY; Gardner, MJ; Cole, GT. Multivalent recombinant protein vaccine against coccidioidomycosis. Infect. Immun 2006, 74, 5802–5813. [Google Scholar]
- Tarcha, EJ; Basrur, V; Hung, CY; Gardner, MJ; Cole, GT. A recombinant aspartyl protease of Coccidioides posadasii induces protection against pulmonary coccidioidomycosis in mice. Infect. Immun 2006, 74, 516–527. [Google Scholar]
- Jianmin, X; Chen, X; Selby, D; Hung, CY; Yu, JJ; Cole, GT. A genetically engineered live attenuated vaccine of Coccidioides posadasii protects BALB/c mice against coccidioidomycosis. Infect. Immun 2009, 77, 3196–3208. [Google Scholar]
- Cole, GT; Xue, JM; Okeke, CN; Tarcha, EJ; Basrur, V; Schaller, RA; Herr, RA; Yu, JJ; Hung, CY. A vaccine against coccidioidomycosis is justified and attainable. Med. Mycol 2004, 42, 189–216. [Google Scholar]
- Magee, DM; Cox, RA. Interleukin-12 regulation of host defenses against Coccidioides immitis. Infect. Immun 1996, 64, 3609–3613. [Google Scholar]
- Kirkland, TN; Raz, E; Datta, SK. Molecular and cellular mechanisms of protective immunity to coccidioidomycosis. Vaccine 2006, 24, 495–500. [Google Scholar]
- Jiang, C; Magee, DM; Cox, RA. Construction of a single-chain interleukin-12-expressing retroviral vector and its application in cytokine gene therapy against experimental coccidioidomycosis. Infect. Immun 1999, 67, 2996–3001. [Google Scholar]
- Wüthrich, M; Gern, B; Hung, CY; Ersland, K; Rocco, N; Pick-Jacobs, J; Galles, K; Filutowicz, H; Warner, T; Evans, M; Cole, G; Klein, B. Vaccine-induced protection against 3 systemic mycoses endemic to North America requires Th17 cells in mice. J. Clin. Invest 2011, 121, 554–568. [Google Scholar]
- Magee, DM; Friedberg, RL; Woitaske, MD; Johnston, SA; Cox, RA. Role of B cells in vaccine-induced immunity against coccidioidomycosis. Infect. Immun 2005, 73, 7011–7013. [Google Scholar]
- Johnson, SM; Kerekes, KM; Lunetta, JM; Pappagianis, D. Characteristics of the protective subcellular coccidioidal T27K vaccine. Ann. NY Acad. Sci 2007, 1111, 275–289. [Google Scholar]
- Herr, RA; Hung, CY; Cole, GT. Evaluation of two homologous proline-rich proteins of Coccidioides posadasii as candidate vaccines against coccidioidomycosis. Infect. Immun 2007, 75, 5777–5787. [Google Scholar]
- Barnato, AE; Sanders, GD; Owens, DK. Cost-effectiveness of a potential vaccine for Coccidioides immitis. Emerg Infect. Dis 2001, 7, 797–806. [Google Scholar]
- Wright, JC; Weinstein, MC. Gains in life expectancy from medical interventions—standardizing data on outcomes. N. Engl. J. Med 1998, 339, 380–386. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hector, R.F.; Rutherford, G.W.; Tsang, C.A.; Erhart, L.M.; McCotter, O.; Anderson, S.M.; Komatsu, K.; Tabnak, F.; Vugia, D.J.; Yang, Y.; et al. The Public Health Impact of Coccidioidomycosis in Arizona and California. Int. J. Environ. Res. Public Health 2011, 8, 1150-1173. https://doi.org/10.3390/ijerph8041150
Hector RF, Rutherford GW, Tsang CA, Erhart LM, McCotter O, Anderson SM, Komatsu K, Tabnak F, Vugia DJ, Yang Y, et al. The Public Health Impact of Coccidioidomycosis in Arizona and California. International Journal of Environmental Research and Public Health. 2011; 8(4):1150-1173. https://doi.org/10.3390/ijerph8041150
Chicago/Turabian StyleHector, Richard F., George W. Rutherford, Clarisse A. Tsang, Laura M. Erhart, Orion McCotter, Shoana M. Anderson, Kenneth Komatsu, Farzaneh Tabnak, Duc J. Vugia, Ying Yang, and et al. 2011. "The Public Health Impact of Coccidioidomycosis in Arizona and California" International Journal of Environmental Research and Public Health 8, no. 4: 1150-1173. https://doi.org/10.3390/ijerph8041150
APA StyleHector, R. F., Rutherford, G. W., Tsang, C. A., Erhart, L. M., McCotter, O., Anderson, S. M., Komatsu, K., Tabnak, F., Vugia, D. J., Yang, Y., & Galgiani, J. N. (2011). The Public Health Impact of Coccidioidomycosis in Arizona and California. International Journal of Environmental Research and Public Health, 8(4), 1150-1173. https://doi.org/10.3390/ijerph8041150



