1. Introduction
Cardiovascular disease (CVD) and diabetes are the leading causes of death worldwide. An estimated 17.1 million people died from CVD in 2004, representing 29% of all global deaths. Diabetes causes approximately 5% of all deaths globally each year and its incidence is predicted to increase by over 50% in the next 10 years, according to the World Health Organization (WHO) [
1]. People with diabetes develop CVD at an earlier age and are two to four times more likely to suffer strokes than healthy subjects, and approximately 73% of adults with diabetes are considered pre-hypertensive. These diseases also impose a substantial economic burden on individuals, families, and nations. Healthcare expenditures for diabetes are expected to account for 11.6% of total healthcare spending in the world in 2010 [
2]. Besides excess healthcare expenditures, diabetes and CVD also impose costs in terms of lost productivity and foregone economic growth due to lost work days, lower work productivity, mortality, and permanent disability [
3].
Lifestyle interventions,
i.e., changed dietary habits, increased physical activity, maintaining or reducing body weight, and smoking cessation, are effective in preventing CVD and diabetes. In recent decades, numerous studies have focused on preventing type 2 diabetes (T2DM) via lifestyle intervention. The Malmö feasibility study was the first [
4], followed by other controlled trials, such as the Da Qing study in China [
5], the Diabetes Prevention Program (DPP) in the USA [
6], and the Diabetes Prevention Study (DPS) in Finland [
7]. These trials have had a significant impact on public health policy, providing evidence of lifestyle interventions as preventive factors, and have been followed by similar studies in other countries, for example, India [
8], Japan [
9], and The Netherlands [
10]. Reviews of lifestyle interventions have also indicated that diet and/or physical activity are effective in reducing CVD risk in primary care [
11–
14].
As evidence supports the role of diet and/or physical activity in preventing T2DM and CVD, preventive strategies should aim to reduce population-wide risk. Such risk reduction interventions, even if modest, could cumulatively yield substantial benefits. Given the considerable cost of such interventions, public health interventions are increasingly subject to economic evaluation [
15–
17]. Economic evaluations comprise the comparative analysis of two or more healthcare interventions in terms of their costs and consequences. The results of such evaluations help public health policymakers make informed decisions, ensuring that limited resources are allocated as efficiently as possible to improve overall population health while avoiding allocating resources to interventions with comparatively low cost-effectiveness [
18,
19]. The number of economic evaluations of diet and/or physical activity interventions focusing on T2DM and CVD is also increasing. There is one review of the cost-effectiveness of physical activity interventions, but not specifically regarding CVD or T2DM [
20], one of dietary intervention to prevent CVD [
21], and another of the cost-effectiveness of physical activity in treating disease [
22]. There is also a review of economic evaluations of T2DM prevention [
23], which updated three previous reviews. However, to our knowledge, no review has considered the health economic evidence regarding lifestyle interventions to prevent CVD and T2DM simultaneously, although these diseases share lifestyle risk factors (sometimes referred to as the metabolic syndrome [
24,
25]).
Modeling has become a crucial component of economic evaluations. Computer simulation models are usually a series of mathematical equations combined in a structural framework to allow the projection of short-term data from clinical trials to long-term health outcomes and costs [
18]. Modeling is particularly relevant in the case of T2DM and CVD, since morbidity and mortality stem from chronic complications. Several models of diabetes and related complications and of CVDs have recently been developed [
26,
27]; for example, in the fourth Mount Hood Challenge, seven models of diabetes were analyzed [
28]. There have been previous reviews of diabetes models [
27,
29], one of which concluded that models vary significantly in whether diabetes complications (micro
vs. macro complications) are covered, and less in the detail of such coverage [
29]. Several recent studies have incorporated the latest epidemiological data, enabling advanced modeling of diabetes and related complications.
Models incorporate the short-term outcome of an intervention and project its lifetime effects. Since lifestyle interventions aim to change subject behavior, the beneficial habits are supposed to continue after the interventions have ended. For example, in a followup study of DPS, the intervention group maintained the beneficial lifestyle changes with the relative risk reduction of 36% after the three-year followup of a 4-year intervention period [
30]. Unlike DPS, the 10-year followup of DPP demonstrated that diabetes incidence was the same in the lifestyle and control groups (5.9
vs. 5.6), but that the cumulative incidence was lower in the lifestyle group, leading the authors to conclude that diabetes can be prevented or delayed for at least for 10 years by means of lifestyle intervention [
31]. The 20-year followup study of Da Qing demonstrated that lifestyle intervention still had positive effects on the incidence of T2DM in the intervention group [
32], but had no significant effect on CVD events, CVD mortality, or all-cause mortality relative to the control group. The DPS followup study also demonstrated that the effect of lifestyle intervention on 10-year CVD mortality was same in both control and intervention groups, unlike the Malmö preventive trial in which, after 12 years of followup, total mortality was lower for lifestyle participants [
33]. Although there are unresolved issues concerning the long-term effectiveness of lifestyle interventions, economic evaluation of lifestyle interventions requires empirical evidence or logical assumptions to model probable future health outcomes.
This review critically appraises the literature, particularly seeking to answer the following questions:
How have economic evaluation of lifestyle interventions (i.e., diet and/or physical activity) been implemented in preventing T2DM and CVD?
What models have been used in conducting these evaluations?
What assumptions have been made regarding the long-term effectiveness of interventions when modeling beyond the intervention period?
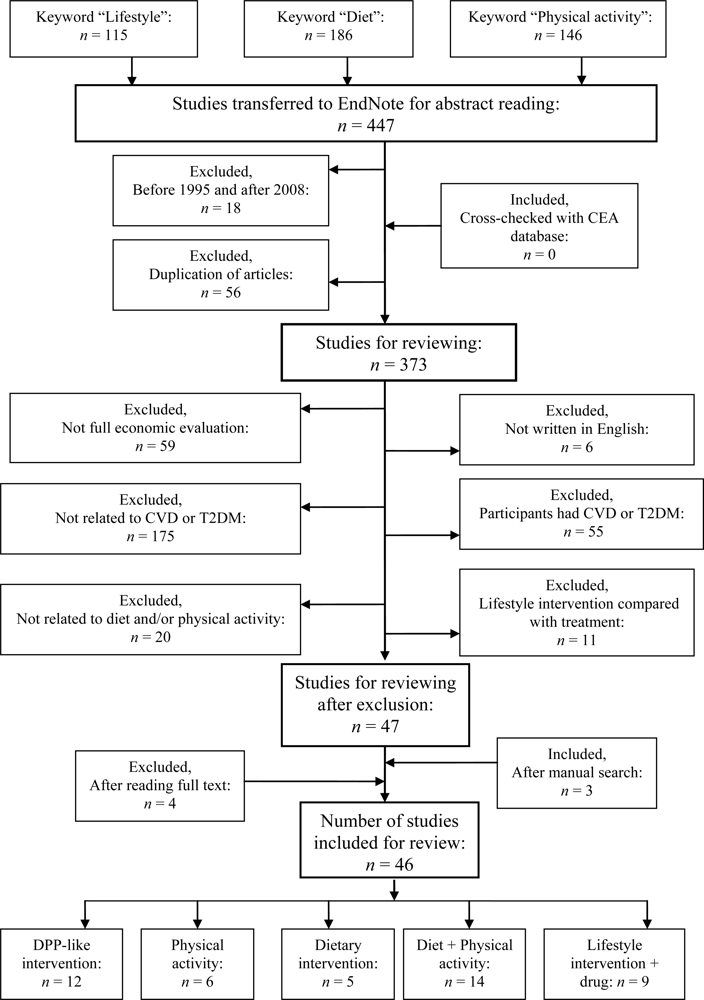
3. Results
The reviewed interventions vary from simple provision of information about behavioral changes to active participation and screening for diabetes or CVD, which might involve universal screening or targeted screening of high-risk groups. The comparator of the studies also varies, being placebo care, standard care, or lifestyle intervention alone, especially when pharmacological interventions are evaluated. The target groups range from school-aged children to subjects over 65 years old. The risk factors vary, the populations ranging from generally healthy to high-risk (
i.e., overweight, obese, and IGT/IFG positive), sometimes being gender specific (
i.e., five articles examined female participants and one examined males). The intervention settings are also diverse: some articles analyze new hypertension guidelines or national policies for countering overweight, others evaluate community or primary care settings, and three studies are school based. The evaluation countries are mainly developed ones, primarily the USA, followed by the UK, with only one study (the Indian Diabetes Prevention Programme—IDPP) from India. Only one study is a CBA; three are CCAs and the rest are either CEAs or CUAs. In the CUA, effectiveness is measured as quality-adjusted life years (QALYs), whereas in the CEAs the measure of effectiveness varies considerably, for example, being life years gained (LYG), incidence of T2DM prevented or delayed, percentage point decrease in 10-year CVD risk, or number needed to treat to prevent one case of diabetes. The effectiveness data are derived from single randomized controlled trials or from literature reviews of trials from the country of a particular intervention study, if available, and otherwise from other countries. Results are sometimes presented as the incremental cost-effectiveness ratio (ICER),
i.e., the ratio of the differences in costs between two alternatives to the differences in effectiveness between the same two alternatives (
Tables 1–
5 present only the results of lifestyle interventions). The discount rate ranges from 3% to 6%. Most studies use the same discount rate for costs and effects, although different rates are used, for example, in all studies from the Netherlands, where 4% is used for costs and 1.5% for effects. Sensitivity analyses are univariate, multivariate, or probabilistic. Of the 46 studies, 31 include decision analytic models (DAMs), such as decision trees, Markov models, and an Archimedes model.
Methodological variation affecting how results are derived makes intra- or inter-group comparison between the studies difficult. However, concentrating only on the monetary figures in the results and agreeing on what is considered cost-effective (50,000 US$/QALY, 20,000–30,000 £/QALY or 50,000 AU$/QALY), most lifestyle interventions are deemed cost-effective.
3.1. General Characteristics of the Studies
The papers are divided into five groups: (1) DPP-like lifestyle interventions, (2) physical activity interventions, (3) dietary interventions, (4) diet + physical activity interventions, and (5) drug treatment combined with any of the preceding interventions (Sections 3.1.1.–3.1.5.;
Tables 1–
5).
3.1.1. DPP-Like Lifestyle Interventions
Economic evaluations of DPP-like lifestyle interventions have used different methodologies and produced inconsistent results. The first economic evaluation of DPP, which compared lifestyle interventions and pharmacotherapy (metformin) with placebo care, was performed by the DPP Research Group in 2003 [
37]; it covered only the three-year intervention period without any DAM and from a societal perspective. As DPP intervention costs were very high, it was proposed that the intervention be offered on a group basis (10 people per group) instead of face to face; it was assumed that the effectiveness would be similar, so that the costs of lifestyle intervention would be reduced. As a three-year time horizon overestimates the treatment costs and underestimates the benefits of lifestyle intervention and metformin, the same research group later extrapolated the trial data into lifetime costs and benefits using a Markov model [
38]. The progression of IGT to clinical onset of diabetes and from diabetes-related complications to death was assessed.
A later study [
39] used the DPP data for five countries,
i.e., Australia, France, Germany, Switzerland, and the UK, applying a simple three-state Markov model (
i.e., alive with IGT, alive with T2DM, and deceased) over a lifetime horizon. Another study analyzed the DPP results from a different perspective [
40],
i.e., whether investment in a DPP intervention program by a health insurer would cut costs. The model was same as that of the DPP Research Group but used for two time periods. The conclusion was that, if the insurer and Medicare shared the DPP intervention costs (24% borne by the insurer), the insurer would recover the investment via avoided future medical care costs.
Two studies examined [
41,
42] screening for diabetes among overweight and obese people followed by DPP intervention, while Icks
et al. [
42] studied the cost-effectiveness of DPP in a real-world setting,
i.e., when acceptance of and adherence to the intervention is low and the dropout rate is high. In addition to metformin, another drug, acarbose, was examined in Caro
et al. [
43] using a four-state Markov model of a Canadian population over ten years; this study estimated that lifestyle modification would prevent 117 cases of diabetes, while metformin and acarbose would prevent 52 and 74 new cases of diabetes, respectively. The lifetime cost and effectiveness of DPP was estimated by Eddy
et al. [
44] using an Archimedes model, which contains infinite health states. This is the only study estimating that DPP-like lifestyle intervention has a mere 0.1% chance of costing under 50,000 US$/QALY.
DPP or DPP-like interventions have been studied in other countries as well. IDPP was performed by Ramachandran
et al. [
45]; although the analysis examined only the trial period (three years), the cost-effectiveness ratio was much lower than for DPP. Galani
et al. reported two studies [
46,
47] on lifestyle interventions for overweight and obese Swiss population groups, with assumed effectiveness taken from DPS. A seven-state Markov model over a lifetime horizon estimated that lifestyle intervention could be cost-effective depending on sex, age group, and threshold values. DPS effectiveness was applied to a Swedish population [
48] using a Markov model, which included the cost of added life years.
Methodological disagreement is the main issue in DPP-like studies. The results of DPP interventions are reported as 8,800 US$/QALY or 62,600 US$/QALY depending on whether a Markov [
38] or Archimedes model [
44] is applied. If 50,000 US$/QALY is considered a cutoff value for cost-effectiveness, the same trial is cost-effective with one method but not the other. The disagreement stems from different model assumptions on the rates of progression to diabetes and complications [
49,
50]. Both authors provide arguments and counterarguments defending their assumptions [
51,
52].
Despite the disagreement, it was predicted that DPP-like lifestyle intervention would delay the onset of diabetes and lead to fewer complications, longer lives, and improved quality of life [
50]. All the DPP/DPS/IDPP-like lifestyle interventions were cost-effective, except that reported by Eddy
et al. [
44], even if the intervention started with costly screening [
41,
42]. IDPP was much more cost-effective than similar programs in high-income countries, because the intervention cost was much lower even though staff went to participant homes to provide the intervention [
45]. The cost-effectiveness ratio was in the cost-effective range when DPS was applied to a Swedish population [
48], even though unrelated costs for added years of life were included, which is debatable [
53].
3.1.2. Physical Activity Interventions
Several methods have been used to determine the physical activity level of participants in the economic evaluation of physical activity studies. One study used metabolic equivalent (MET) minutes per week to classify participants as inactive, irregularly active, meets guidelines, and highly active [
54]. Some studies used duration and intensity of physical activity,
i.e., ≥2.5 hours of moderate intensity or vigorous exercise per week as marking a physically active person [
55], while other studies used the self-administered physical activity questionnaire (PAQ) [
56,
57]. There were only two Markov models [
54,
55] of physical activity: the CDC MOVE Markov model was based on five diseases (
i.e., coronary heart disease, ischemic stroke, T2DM, breast cancer, and colon cancer), which led to underestimated cost-effectiveness, as physical activity also affects other diseases such as depression or anxiety [
58].
Indirectly measuring the level of physical activity (e.g., via PAQs) raises validity concerns [
59]. The selection of participants for physical activity interventions is also a major concern,
i.e., whether the control group also participates in the program or only includes motivated people; for example, one study [
56] excluded highly active participants. Participation rate and adherence to physical activity are other important issues meriting attention when modeling the long-term benefits of physical activity.
Surprisingly, the ICER of physical activity interventions is much lower in Australia [
55] and New Zealand than in the USA [
54] or UK [
56]. This could be because Roux
et al. [
54] analyzed the physical activity promotional intervention using a model containing considerably more health states and because the participants were older in the Munro
et al. [
56] study; however, all ICERs are in the cost-effective range.
3.1.3. Dietary Interventions
An economic evaluation of ten different nutritional interventions for the Australian population was performed by Dalziel and Segal [
60]. Four Markov models were developed to analyze nutritional interventions. Reduction of dietary sodium intake to reduce hypertension, eating five servings of fruit and vegetables per day, and adhering to a “Mediterranean-style” diet seemed to be cost-effective. The effect of the Mediterranean diet was estimated to reduce the 10-year CVD risk based on the Framingham risk equation. The cost-effectiveness of grains fortified with folic acid as well as vitamin supplementation with folic acid and cyanocobalamin (vitamin B12) to prevent coronary heart disease (CHD) was analyzed in one study [
61] of the entire US population via the CHD Policy Model. Nutritional education was compared in Cox
et al. [
62], one group receiving traditional face-to-face advice, while another received self-administered video lessons. The effectiveness of the intervention was analyzed using a behavior questionnaire on changes in 10 dietary factors.
There are few economic evaluations of dietary interventions and the quality of studies is often limited, since some important economic evaluation parameters (i.e., age, risk factors, sensitivity analysis, and price year) are not clearly specified. The use of self-administered questionnaires to measure food intake raises validity issues.
Dietary interventions are very diverse, and probably not very comparable. Moreover, varying study quality makes it difficult to compare the results of the dietary interventions, but fruit and vegetable intake has the lowest cost-effectiveness ratio (46 AU$/QALY) [
60] and similar results are obtained from universal fortification of grains with folic acid.
3.1.4. Diet + Physical Activity Interventions
Three articles presented economic evaluations of a national policy or action plan [
63–
65]. The new Finnish hypertension care guidelines were compared with previous ones [
64], which did not incorporate lifestyle intervention; the new guidelines were estimated to save an additional 49,000 life years. The Dutch national action plan for counteracting obesity included a community-based intervention in which 90% of the population was screened and received lifestyle advice; an intensive lifestyle program was applied to 10% of the overweight adults in a healthcare setting. The combined program was evaluated using the RIVM chronic disease model, finding that it would save 110,000 life years over 20 years [
63]. Using a diabetes model [
65], it was estimated that diabetes care and prevention strategies applied to the entire Australian population aged 45–74 years would prevent 53,000 new cases of diabetes over 10 years.
Three articles studied childhood obesity prevention programs [
66–
68]. The “traffic light diet,” physical activity reinforcement, self monitoring, and stimulus control were used as interventions in one study; their effectiveness was measured as a reduction in standardized body mass index (Z-BMI) and proportion of overweight children [
67]. Another study [
68] examined Planet Health, a school-based obesity prevention program focused on reducing television viewing, reducing consumption of high-fat foods, increasing fruit and vegetable consumption, and increasing moderate and vigorous physical activity. The randomized controlled trial included male and female subjects, but there were significant reductions in obesity only among female students after two school years. This intervention effect was then extrapolated up to age 64 years using a decision model measuring health benefits as QALYs. The same method was used in another study [
66] of a different school-based obesity prevention program (CATCH) including male and female participants from grades three (age 8) to five (age 11). CATCH appears to be more cost-effective than the Planet Health program.
Two studies have focused solely on reduction of CVD incidence using the Framingham risk equation [
69,
70]. One study compared lifestyle interventions, including video and video + self-help guidelines, with routine care for Australian participants at risk of CVD [
70]. In another study, community-based education to change dietary habits to reduce total cholesterol level was estimated to prevent approximately 4.5 cases of CVD every year, with the highest benefit in the 55–64-year age group, in which it would prevent 8–9 cases [
69] in a population of 5,500. The WISEWOMEN project, which aimed to reduce CVD risk in older uninsured and undernourished women by means of either CVD screening + enhanced lifestyle intervention or CVD screening + minimal intervention, was evaluated in two studies [
71,
72]. The earlier study [
72] measured effectiveness as the percentage reduction of the 10-year probability of having CVD, while the later study [
71] measured it as LYG as well, resulting in a cost per LYG of US$ 4400. The effect of a single randomized controlled trial intervention of diet, exercise, and diet + exercise extrapolated on a cohort of 60-year-old healthy subjects without CVD was evaluated in one study [
73]. The Markov model included 10 health states but not diabetes. The cost-effectiveness of interactive group sessions advising on nutrition and physical activity for cohabiting Australian couples was studied [
74] on an intention-to-treat basis with outcomes measured on 14 variables (e.g., energy intake, fat intake, fiber intake, sodium, fruit and vegetable intakes, BMI, physical activity level, cholesterol profiles, and blood pressure). Two types of lifestyle intervention were evaluated by Jacobs
et al. [
75]—a community-based intervention for the general population covering many people at a relatively low cost, and a healthcare-based intervention for high-risk people covering fewer patients at a higher cost—using the RIVM chronic disease model.
Many studies lack detailed information about certain important economic evaluation parameters and on how data have been derived, for example, via meta analysis, systematic review, selective studies, or expert opinion. These parameters include cost (e.g., unit cost, total cost, intervention cost, disease cost, and productivity cost) [
65,
72,
74], utility [
63], and disability weight [
65]. In childhood obesity studies [
66,
68], the explicit assumption requires evidence, which is missing, from epidemiological studies linking childhood overweight to adult overweight and to weight regain after two-year interventions. The same issue applies in CVD cases: a time lag after termination of lifestyle intervention in improvements of CVD risk factors. Lindholm
et al. [
69] considered a five-year lag, whereas
Dzator
et al. [
74] considered no time lag. The transferability of one country’s clinical trial data to another [
65] also requires adjustment. For community-based interventions [
63,
69,
75], special attention is required to consider the spillover effect of lifestyle interventions and the efficacy of interventions in various socioeconomic groups,
i.e., the equity concern is missing from the studies. However, the main question concerns the applicability of intervention effectiveness data from clinical settings to real-world settings, particularly when national action plans or policies are being evaluated [
63–
65].
Interventions starting from childhood have a low cost-effectiveness ratio (900 US$/QALY) [
66]. The community-based approach is also attractive, as seen in the Dutch [
63,
75] and Swedish studies [
69]. The advantage of community-based lifestyle prevention programs is that the health gains achieved through population-based approaches often exceed those achievable by targeting specific groups in clinical or subclinical settings.
3.1.5. Combined Drug and Lifestyle Interventions
Three articles [
76–
78] concerning the drug sibutramine were studied by the same research group using the same model but in different country settings,
i.e., Finland, Germany, the USA, the UK, and Switzerland. Lifestyle intervention was included along with the drug to treat overweight or obese people. The total number of fatal and non-fatal CHD events avoided in five years of analysis was estimated as 3.49 in Finland, 4.18 in Germany, 4.49 in Switzerland, and 1.96 in the UK per 1,000 people, while the average number of diabetes cases avoided was 3.0. In an Italian obese population, another drug, orlistat, was studied [
79]. The same drug combined with a low-calorie diet for obese patients was also assessed for the whole Dutch population, estimated over a lifetime horizon using the RIVM chronic disease model [
80]; the combined therapy was estimated to produce an additional 34,000 life years.
A weight-reduction drug, rimonabant, was compared with lifestyle intervention [
81] using a decision tree with five arms,
i.e., treatment options. Two years’ treatment with rimonabant combined with lifestyle intervention produced the most cost-effective option. Employing a Markov model, Roux
et al. [
82] compared four weight-loss strategies in overweight and obese women. The strategies were diet only, diet + pharmacotherapy (orlistat), diet + exercise, and diet + exercise combined with behavior therapy. The weight-loss intervention consisted of six months of intervention followed by a six-month maintenance program. Diet + exercise combined with behavior modification was the best strategy.
When statin drugs and cholesterol-lowering diets were compared with no interventions for patients with a high cholesterol level using the CHD Policy Model over a 30-year period, both interventions produced much higher cost/QALY in the primary prevention [
83]. This may be because statins were expensive at the time (1997), before they were available as generics. In another study [
84], four strategies for preventing or delaying diabetes,
i.e., screening for early detection, screening + lifestyle intervention, screening + lifestyle + pharmacological intervention, and no screening, were compared with a hybrid model using a three-arm decision tree and seven-state Markov model. Screening followed by lifestyle intervention was the most cost-effective strategy.
All pharmacological studies included a short-term trial period (six months to one year) extrapolated to a five- or ten-year time horizon, except one that employed a lifetime horizon [
80]. One issue in pharmacological interventions was weight regain after intervention end; the National Institute of Clinical Excellence (NICE) recommendation [
85] on assumed uniform weight regain over a three-year period had not been taken into consideration. Furthermore, though all drugs had some side effects, the related loss of quality of life was not considered. We agree with previous review findings that the main sources of uncertainty in pharmacological interventions are weight-loss sustainability, utility gains associated with weight loss, extrapolation of long-term benefits from short-term trials, dropout rate, side effects, and bias towards the funding authority [
86].
Surprisingly, all effectiveness data for drug interventions are based on literature reviews, except data from the study [
79] reporting the highest ICER (75,300 €/QALY). When lifestyle interventions are compared with pharmacotherapy, lifestyle interventions are more effective,
i.e., in survival years, disease-free time, and quality-adjusted life expectancy [
82], indicating that lifestyle interventions are better options for preventing lifestyle diseases.
3.2. Decision Analytic Models (DAM)
Of 46 studies, 31 employed DAM: six used decision trees, 20 used Markov models, and one used an Archimedes model (
Table 6). One article used both a Markov model and a decision tree [
84], one used four Markov models [
60], one used an Archimedes model [
44], and two used a life table approach. A decision tree is a simple visual representation of possible options and their consequences. Decision trees start with the options, each of which branches out to explore all potential health outcomes and their respective probabilities and costs. In Markov models, participants move from defined health states (Markov states) in discrete time periods (Markov cycles). Each health state incurs particular costs and health consequences [
18]. It is common to use a previously developed model; all the
Dutch [
63,
75,
80] studies used the RIVM chronic disease model adapted to the study objectives, while the CHD Policy Model [
61,
83] and the CDC diabetes model [
38,
40,
41] have been used in many studies.
Philips
et al. [
92] emphasized several issues for good practice in modeling: model structure; data employed,
i.e., inputs (costs) and outcomes (health benefits); and model consistency or validity. For Markov models, the structure generally concerns the health states included, as inclusion of costly diseases (e.g., stroke due to diabetes in diabetes progression models) might overestimate the long-term results. Another example is that the RIVM chronic disease model [
63] included certain types of cancer absent from another model [
46], although both models were developed for overweight and obesity.
The effectiveness data were taken from a single trial or literature review. For most models, the model population was a hypothetical cohort but, when the effectiveness of a trial was transferred to different country settings, the model population was often matched with the study population; for example, the survey population from the US National Health and Nutrition Survey [
41] and the participants in the German KORA study were matched with the DPP participants [
42], and similarly in three other studies [
39,
44,
60].
Two types of cost data were included in the models: cost of intervention and cost of disease avoided. Some studies estimated the intervention cost from a clinical trial [
91], some calculated the cost retrospectively [
71], and some based the cost on national administrative databases [
65], expert opinion [
73], or even modeler opinion. The avoided disease costs were country specific if available; otherwise, the applicable data were taken from other countries. The model outcomes were QALYs, DALYs, LYG, or other measures of health. Different countries were found to have used different instruments when estimating QALYs: QWB-SA was used for the DPP models, European studies frequently used EQ-5D, while Finnish and Australian studies preferred their own instruments (
i.e., 15D and Aqol). Models are subjected to internal validation (
i.e., comparing model output with the data used in building model), external validation (
i.e., checking whether the model output is consistent with the disease outcome and epidemiological data), and between-model validation (
i.e., comparing the estimated intervention outcome with the outcomes of other models based on similar assumptions and addressing similar diseases) [
92,
93], as model quality depends largely on input data quality [
94]. No studies provided any details on all the three types of validation. However, in modeling, it is recommended that a technical report [
19],
i.e., a detailed description of all assumptions and parameter values used to construct the model, be provided. Not all model studies mentioned technical reports, though some provided supplementary materials.
3.3. Long-Term Effectiveness of Lifestyle Intervention
An important expectation in a lifestyle intervention is long-term adherence to the changed behavior, either the change in dietary habit or the increase in physical activity. A key issue in economic evaluations of such interventions is to link short-term evidence from clinical trials or epidemiological data to the long-term benefits of changed behavior. When considering long-term effectiveness, researchers often analyze different scenarios with optimistic or pessimistic assumptions and then assess the uncertainty in sensitivity analyses. However, there is some indecision concerning the optimistic and pessimistic assumptions or the best- and worst-case scenarios. The base case assumptions used in the lifestyle interventions, alternatives used for sensitivity analyses and the changes in baseline results of the sensitivity analysis are highlighted in
Table 7.
The assumption about whether the intervention effect is maintained after the intervention (or trial) dramatically affects the cost-effectiveness ratio or result. We believe it is pessimistic to assume that the effectiveness will persist only as long as the intervention period, as has been done in several studies [
39,
63,
65,
70,
75], and too optimistic to assume that the effectiveness will persist until death, as is done elsewhere [
71,
73]. For example, one study assuming that the effectiveness would persist only for the intervention period (pessimistic assumption) reported a result of 152,000 AU$/QALY; however, if the effectiveness had been assumed to persist one additional year, the result would be 6,600 AU$/QALY [
70]. On the other hand, if intervention effectiveness is assumed to be one year (pessimistic assumption), instead of lifelong the cost-effectiveness ratio would be 10 times higher than the base case (lifelong effectiveness) result [
71]. In the case of DPP, some researchers assumed that the intervention and its effectiveness would persist until the participants developed diabetes or died— an optimistic assumption [
38,
40,
41,
44]. On the other hand, other researchers assumed that the effectiveness would decline by 20% each year [
41] and 50% over the total period [
38,
40]. Reducing the effectiveness by 20% resulted in 1.5 times [
41] and almost seven times [
38] higher total cost-effectiveness ratios than in the base case analysis. In the DPP trial, the lifestyle intervention was 58% effective, which Caro
et al. [
43] used for the base case analysis with 30% as the worst- and 70% as the best-case value. With 70% effectiveness, the result was dominant,
i.e., the intervention is more effective and less costly than the alternative. Roux
et al. [
82] assumed in the base case analysis that only 20% of participants would maintain the changed behavior in the long term, with optimistic and pessimistic assumptions,
i.e., over 40% and under 10%, respectively, which resulted in estimates four times higher or half the base case cost-effectiveness ratio. Van Baal
et al. [
80] assumed that 23% of the intervention weight loss would persist in the long run as the pessimistic assumption, with optimistic values of 50% or 100% maintained weight loss. With 100% maintained weight loss, the cost-effectiveness ratio was almost three times lower than the base case value.
When long-term effectiveness issues are addressed using univariate sensitivity analysis, the changes in results are obvious. However, when probabilistic sensitivity analysis is performed, the changes in results due to effectiveness uncertainty are not clearly distinguished, as seen in two studies by the same author [
46,
47]. The assumptions in these studies were that weight loss (the intervention effect) would persist up to six years and the regain process would take four more years. After 10 years, the weight of the participants would be same as at the intervention start.
For the pharmacological weight reduction, the weight regain process was often assumed to be completed within five years of a one-year intervention [
76–
78], and a confidence interval (CI) was used in the sensitivity analysis. Assuming one year of sustained weight reduction [
81] made the cost-effectiveness ratio unfavorable. Nevertheless, if the weight loss persisted for three years, the value was in the cost-effective range (under US$ 50,000); using the probabilistic sensitivity analysis, the authors demonstrated that at US$ 50,000 willingness to pay, the intervention had 40% chance of being cost-effective.
4. Discussion
It is difficult to compare the results of one cost-effectiveness analysis with another, because of differences in methodology, types of costs included, outcomes, and population groups and related baseline risk. There may also be differences in, for example, healthcare systems, incentives to healthcare professionals and institutions, clinical practices, population values, availability and accessibility of technologies, and currency purchasing power.
Establishing that an intervention is cost-effective is still problematic, since the threshold for cost-effectiveness,
i.e., decision maker willingness to pay, is controversial. NICE in the UK uses a cost-effectiveness threshold range of £20,000 to £30,000 per QALY gained [
95,
96]. In contrast, there are no official guidelines for the USA and Australia, though US researchers frequently employ 50,000 US$/QALY [
97], while Australian researchers use 50,000 AU$/DALY [
98] as thresholds. WHO has recommended that interventions be considered cost-effective if costs per DALY are 1–3 times GDP per capita [
99]. Some argue that cost-effectiveness thresholds may be too high [
100,
101], others argue that they are too low [
102,
103], while still other claim the well-accepted US$ 50,000 threshold is misused [
104]. Moreover, one review of cost-effectiveness analyses suggested that published studies tended to report results below US$ 50,000 per QALY [
105].
Some recent DAM guidelines emphasize that models should be kept as simple as possible, providing they capture all essential parts of the disease processes, including effects of health technologies, to help policymakers make informed decisions [
93,
106–
108]. Certain standard criteria should be considered when developing a model, in what is often referred to as validating [
109,
110] and calibrating [
94,
111] a model. It is not always possible to apply all recommendations in one model, so researchers often make tradeoffs between model accuracy and transparency. Transparency refers to the understandability of the logical arguments of a model, to enable it to be reproduced; accuracy refers to a model’s ability to capture real-life situations [
112]. Balance between accuracy and transparency is difficult to obtain in a model: as a model is made more accurate, its complexity increases, which in turn reduces its understandability to decision makers. Accordingly, some researchers emphasize model transparency [
93], whereas others argue that accuracy should be paramount [
112].
When an intervention leads to significant health benefits in comparison with the comparator, the ICER is supposed to be low. For example, when a physical activity prescription was effective in a target population in New Zealand, the ICER was very low [
55]; however, when interventions had no significant impacts on target groups, as seen in two studies of video-based lessons [
62,
70], the interventions were less cost-effective. Another aspect is the analytical time horizon: if it is short, health benefits are limited, likely resulting in high ICERs. DPP provides a good example; the ICERs for the short-term, three-year trial period [
37] are much higher than those for the long-term lifetime analysis [
38]. The age of the target population might also affect ICERs, as young target groups might achieve greater health benefits than older groups. Interventions for school children are very beneficial [
66], as are those starting at ages around 20 [
54,
75,
76]. In contrast, interventions starting later in life, such as those examined by Lindgren
et al. [
48,
73], who use 60 years as the starting age, result in much higher ICERs. ICERs are also affected by the risk level of the population. When the risk is high, as it is among the overweight or obese, the potential health gains from interventions are higher; this was illustrated nicely in two articles [
46,
47].
Naturally, intervention cost drastically affects cost-effectiveness, as is obvious in the DPP trial of costly face-to-face
vs. cheaper group-based counseling. Another aspect is whether screening for high-risk individuals is included in the interventions. Icks
et al. [
42] reported that diabetes screening comprises 36% of the total intervention cost, which was one reason for the high ICER. The costs of developing a website [
91] to motivate participants to increase physical activity can also make the cost-effectiveness ratio unattractive.
The cost-effectiveness ratio of our reviewed studies range from 46 AU$/QALY [
55] for fruit and vegetable intake to as high as 143,000 US$/QALY for DPP lifestyle intervention [
44]. Community-based interventions seem to have low cost-effectiveness ratios [
63,
69,
75] ranging from 1,100 to 5,000 Euro/QALY. School-based interventions are also attractive, at 900 US$/QALY [
66] and 4,305 US$/QALY [
68], as are targeted screening followed by lifestyle interventions [
41,
42]. However, any targeted intervention could be made more favorable by dealing with the abovementioned issues (
i.e., risk level in target groups, intervention cost, intervention effectiveness, and starting intervention at young age) and adjusting the assumptions of the model parameters. So, cautious interpretation is required to generalize the results.
Icks
et al. [
42] argued that we lack information on the long-term effects of T2DM prevention interventions, and lack valid data regarding the natural course of T2DM from early onset to death. However, recent studies have examined the long-term effects of the DPP [
31], Da Qing [
32], and DPS trials [
113]. This suggests that it is time to update models, so they are based on recent epidemiological data. Nevertheless, if long-term clinical effectiveness data are unavailable, the only way to explicitly explore the future effect of an intervention after its completion is by modeling; that modeling permits such exploration should be considered one of its major strengths, not a weakness [
114]. The validity of long-term effectiveness assumptions would, however, benefit from some kind of consensus and harmonization, apart from the recommendation to perform relevant sensitivity analyses.
This review is limited in that it is not systematic and in that it omits studies not included in the NHS-EED database, such as monographs, some gray literatures, and book chapters. Smoking cessation is an important lifestyle intervention but, as the topic has been subject to extensive review [
115,
116], it is excluded here. Dieting is a popular lifestyle intervention, but we found no articles focusing specifically on dieting as a weight-loss intervention. However, one objective of DPP-like interventions was to reduce weight by 7% using both diet and physical activity interventions. Roux
et al. have used diet as a sole intervention for weight loss in women [
82], but three other weight-loss strategies were also addressed at the same time. Another study [
73] used a sole dietary intervention, though the objective was not weight loss. The ICERs in the studies have not been converted to a common price year, since there is no fixed cost-effectiveness threshold, and the ICERs reported depend on the comparators, which vary widely in the studies reviewed. The actual incremental costs per health outcome reported are better regarded as indications of cost-effectiveness. Another important limitation is that none of the three available quality checklists [
117–
119] is used for assessing article quality, partly because the checklists were developed only recently [
117,
119], after several of the included studies. Furthermore, a recent study states that the quality appraisals depends on the researchers and not on the checklists [
120], as ICERs are unlikely to be affected by a single factor but rather by a combination of several.
This is the first study, to our knowledge, to include dietary and physical activity lifestyle interventions that affect T2DM and/or CVDs, with a special focus on DPP-type interventions. A recent study by Anderson [
121] questioned the use of systematic reviews of economic evaluations, partly because the interplay of 26 factors makes the results of cost-effectiveness analyses vary depending on setting and location [
122]; as well, 14 factors had to be considered to ensure transferability of results from one country to another [
123]. On the other hand, there are three good reasons to review economic evaluations: (1) to study the development of new decision models; (2) to identify the most relevant studies for a particular decision making context; and (3) to identify the “how and why” causality of interventions that are cost-effective in certain settings but not in others, including the principal economic tradeoffs in particular decision areas. This review identifies several new decision models, such as models of screening for diabetes and ensuing interventions [
84], a model of multiple behavior modification [
82], a model of cardio–metabolic disorders [
46], and a model used for DPS [
48] that differs from older models, such as the CHD Policy Model [
61,
83], the Johannesson model [
70], and the CDC model [
38,
40–
41]. The MOVE model [
54] is a new updated physical activity model, differing from a previous model developed by NICE [
124]. The models developed to study national plans of action/policy are new and advanced, and economic evaluations of national action plans may well constitute a new research area [
63–
65]. The economic tradeoff in intervention options is well demonstrated in DPP-like studies, in which intervention provision (individual
vs. group counseling) and different country settings (USA
vs. India) are key factors [
37,
45]. Tradeoffs have also been identified in Dutch studies in which community-based, high-cost/low-effectiveness intervention is compared with healthcare-based highly effective low-cost intervention [
63,
75]. The explanatory theory-building aspects of how and why an intervention works are hard to establish in public health interventions because of the complex and inherent interplay of several health determinants. Moreover, the reviewed studies were not informative enough in explaining how the intervention, cost, and health effect outcomes are affected by different configurations of input variables (e.g., patient characteristics and context) to build such theory [
121]. However, it was found that using different modeling techniques with different assumptions leads to different results, as in the Archimedes model [
38] and the Markov model [
44] in the DPP trial.