Branching Processes: Their Role in Epidemiology
Abstract
:1. Introduction
2. Single-type Bienaymé-Galton-Watson Branching Processes
3. Single-type Branching Processes with Population-Dependent Offsprings
4. Set of Single-type Branching Processes with Population-Dependent Offsprings
5. Multitype Branching Processes with Age and Population-Dependent Offsprings
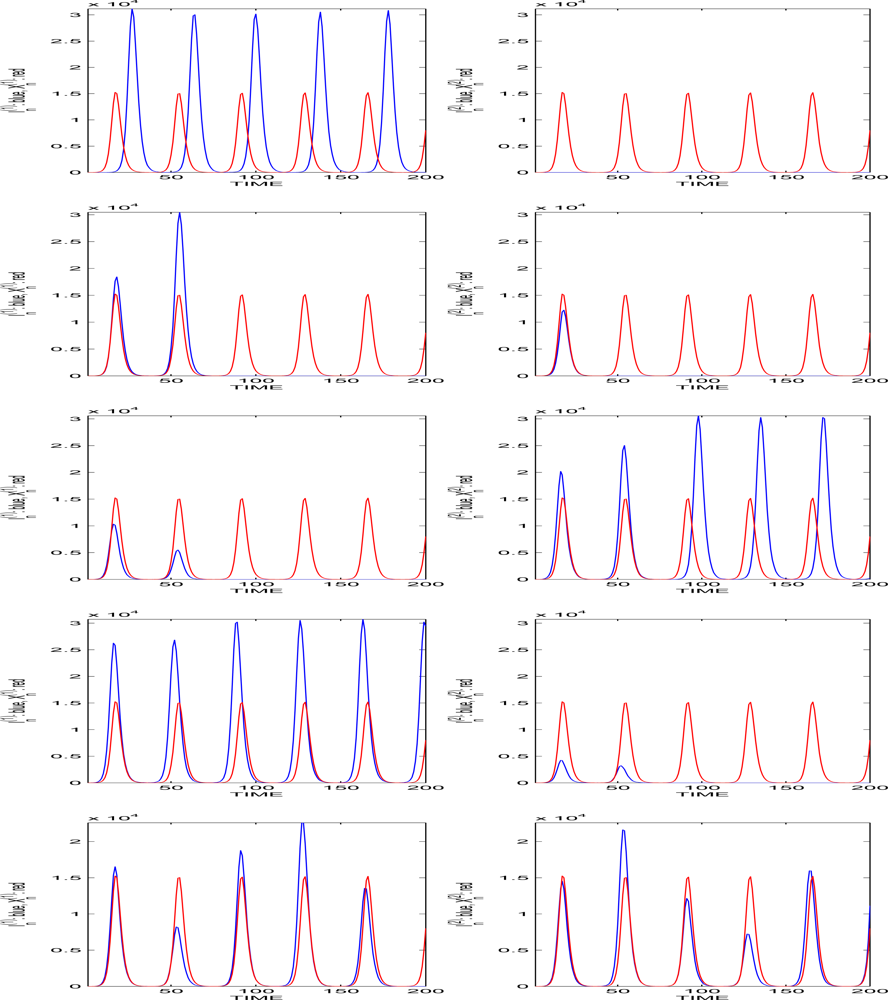
5.1. The Population Size Remains Bounded
5.2. The Initial Population Size N0 is Large and the Disease is not Rare
- {Nn} is positively regular, nonsingular, and checks the xlogx property;
- P(limn Nn = 0 ∪ limn Nn = ∞) = 1
- Let ρ be the first eigenvalue of M͂ defined by E(Ñn|Ñn−1) =: Ñn−1M͂, that is,Then E(Ñnξt) = Ñ0ρnξt, where M͂ξt = ρξt. Moreover ρ ≤ 1 (subcritical and critical cases) implies that P(limn Nn = 0) = 1 (a.s. extinction), and ρ > 1 (supercritical case) implies the existence of an integrable random variable W such that , where Wn := Nn[N0ρn]−1, and P(limn Nn = ∞) = P(W > 0). Finally let us assume that N−l = ρ−lN0, l = 1, ..., d − 1. Then if ρ ∈ , .
- ρ is solution of, and ρ ≤ 1 ⇔R∞ ≤ 1, where (total mean number of offspring generated by an individual).
5.3. The Initial Population Size is Large and the Disease is Rare
- The S and E individuals have the same time-homogeneous survival law {Sa}a;
- There is no over-contamination during the incubation period or the clinical state;
- The number of newborn animals at each birth per individual at time l, is independent of l, i, and of the health state h of i (but the health state of each newborn and his survival during the first time unit may depend on h);
- The population is roughly stable: ;
- The disease is rare at the initial time: ;
- The probability for a given S to be infected at time k + 1 via the horizontal route of excretion, follows a Reed-Frost’s type model, that iswhere is the number of infectious animals at time k (including those in the latest stage of their incubation period). We assume a similar expression for the infection via the horizontal route concerning contaminated meat and bone meal produced from dead infectious animals.
- P(limn In = 0 ∪ limn In = ∞) = 1;
- ρ > 1 is equivalent to R∞ > 1 (supercritical case) which itself implies the existence of a positive integrable random variable W such that, where P(limn In = ∞) = P(W > 0). Moreover let us assume that I−l = clI0, l = 1, ..., d − 1, where cl is independent of I0. Then .
- ρ ≤ 1 is equivalent to R∞ ≤ 1 (subcritical and critical cases) which implies P(limn In = 0) = 1 (a.s. extinction).
- Let Ĩ0 := (I0, 0, ..., 0). Then, that is:and, .
- Let Ĩ0 := (I0, I−1, , ..., I−(d−1)). Then, where, the {Ni,j} are i.i.d. with and ⊕ means the mutual independence, that isand
6. Discussion
References
- Athreya, KB; Ney, PE. Branching Processes; Springer-Verlag: Berlin, Germany, 1972. [Google Scholar]
- Becker, NG. On parametric estimation for mortal branching processes. Biometrika 1974, 61, 393–399. [Google Scholar]
- Becker, NG. Estimation for an epidemic model. Biometrics 1976, 32, 769–777. [Google Scholar]
- Becker, NG. Estimation for discrete time branching processes with applications to epidemics. Biometrics 1977, 33, 515–522. [Google Scholar]
- Becker, NG. A general chain binomial model for infectious diseases. Biometrics 1981, 37, 251–258. [Google Scholar]
- Becker, NG. Analysis of Infectious Disease Data; Chapman and Hall/CRC Press: Boca Raton, FL, USA, 1989. [Google Scholar]
- Devroye, L. The branching process method in Lagrange random variate generation. Commun. Statistics-Simulat. Comput 1992, 21, 1–14. [Google Scholar]
- Diekmann, O; Heesterbeek, JAP. Mathematical Epidemiology of Infectious Diseases Model Building, Analysis and Interpretation; Wiley Series in Mathematical and Computational Biology. John Wiley Ltd: Chichester, UK, 2000. [Google Scholar]
- Elaydi, S. An Introduction to Difference Equations, 3rd ed; Undergraduate Texts in Mathematics. Springer: New York, NY, USA, 2005. [Google Scholar]
- Farrington, CP; Grant, AD. The distribution of time to extinction in subcritical branching processes: Applications to outbreaks of infectious disease. J. Appl. Probab 1999, 36, 771–779. [Google Scholar]
- Farrington, CP; Kanaan, MN; Gay, NJ. Branching process models for surveillance of infectious diseases controlled by mass vaccination. Biostatistics 2003, 4, 279–295. [Google Scholar]
- Good, IJ. The Lagrange distributions and branching processes. SIAM J. Appl. Math 1975, 28, 270–275. [Google Scholar]
- Haccou, P; Jagers, P; Vatutin, VA. Branching Processes Variation, Growth, and Extinction of Populations; Cambridge Studies in Adaptative Dynamics: Cambridge, UK, 2005. [Google Scholar]
- Haight, FA; Breuer, AB. The Borel-Tanner distribution. Biometrika 1960, 47, 143–150. [Google Scholar]
- Hall, P; Heyde, CC. Martingale Limit Theory and its Application; Probability and Mathematical Statistics: New York, NY, USA, 1980. [Google Scholar]
- Harris, TE. The Theory of Branching Processes; Die Grundlehren der Mathematischen Wissenschaften, Bd 119 Springer-Verlag: Berlin, Germany; Prentice-Hall, Inc: Englewood Cliffs, N.J., USA, 1963. [Google Scholar]
- Heesterbeek, JAP; Dietz, K. The concept of Ro in epidemic theory. Stat. Neerl 1996, 50, 89–110. [Google Scholar]
- Hefferman, JM; Smith, RJ; Wahl, LM. Perspectives on the basic reproductive ratio. J. R. Soc. Interface 2005, 2, 281–293. [Google Scholar]
- Hethcote, HW. The Mathematics of Infectious Diseases. SIAM Rev 2000, 42, 599–653. [Google Scholar]
- Heyde, CC. On assessing the potential severity of an outbreak of a rare infectious disease: a Bayesian approach. Austral. J. Statist 1979, 21, 282–292. [Google Scholar]
- Jacob, C; Viet, AF. Epidemiological modeling in a branching population. Particular case of a general SIS model with two age classes. Math. Biosci 2003, 182, 93–111. [Google Scholar]
- Jacob, C; Magal, P. Influence of the routine slaughtering on the evolution of BSE. Example of the British and French slaughterings. Risk Anal 2007, 27, 1151–1167. [Google Scholar]
- Jacob, C. Branching Processes: Their rôle in Epidemiology; Technical report, UR341, MIA department, INRA: Jouy-en-Josas, France, 2009. [Google Scholar]
- Jagers, P. Branching Processes with Biological Applications; Wiley Series in Probability and Mathematical Statistics, John Wiley: London, UK, 1975. [Google Scholar]
- Jacob, C; Lalam, N; Yanev, N. Statistical inference for processes depending on environments and application in regenerative processes. Pliska Stud. Math. Bulgar 2005, 17, 109–136. [Google Scholar]
- Jacob, C; Viet, AF. A new class of processes for formalizing and generalizing individual-based models: the semi-semi-Markov processes. Pliska Stud Math Bulgar 2007, 121–144. [Google Scholar]
- Jacob, C. Saturation Effects in Population Dynamics: Use Branching Processes or Dynamical Systems? In Mathematical Modeling of Biological Systems; Deutsch, A, Bravo de la Parra, R, de Boer, R, Dieckmann, O, Jagers, P, Kisdi, E, Kretzschmar, M, Lansky, P, Metz, H, Eds.; Birkhäuser: Boston, MA, USA, 2008; pp. 339–352. [Google Scholar]
- Jacob, C; Maillard-Teyssier, L; Denis, JB; Bidot, C. A branching process approach for the propagation of the Bovine Spongiform Encephalopathy in Great-Britain. Accepted in Branching processes and their Applications, Lecture Notes in Statistics, Springer-Verlag: Berlin, Germany, 2010; pp. 227–242. [Google Scholar]
- Jagers, P. A general stochastic model for population development. Skan. Aktuarietidskr 1969, 52, 84–103. [Google Scholar]
- Klebaner, FC. Population-size-dependent branching process with linear rate of growth. J. Appl. Probab 1983, 20, 242–250. [Google Scholar]
- Klebaner, FC. On population-size-dependent branching processes. Adv. Appl. Probab 1984, 16, 30–55. [Google Scholar]
- Klebaner, FC. Linear growth in near-critical population-size-dependent multitype Galton-Watson processes. J. Appl. Probab 1989, 26, 431–445. [Google Scholar]
- Klebaner, FC. Correction: “Linear growth in near-critical population-size-dependent multitype Galton-Watson processes” [J. Appl. Probab. 26 (1989), no. 3, 431–445] and “Asymptotic behavior of near-critical multitype branching processes” [ibid. 28 (1991), no. 3, 512–519]. J. Appl. Probab 1992, 29, 246. [Google Scholar]
- Klebaner, FC. Population-dependent branching processes with a threshold. Stochastic Process. Appl 1993, 46, 115–127. [Google Scholar]
- Klebaner, FC; Nerman, O. Autoregressive approximation in branching processes with a threshold. Stochastic Process. Appl 1994, 51, 1–7. [Google Scholar]
- Klebaner, FC; Zeitouni, O. The exit problem for a class of density-dependent branching systems. Ann. Appl. Probab 1994, 4, 1188–1205. [Google Scholar]
- Klebaner, FC. Population and density dependent branching processes. In Classical and Modern Branching Processes; IMA Vol Math Appl, 84, Springer: New York, NY, USA, 1997; pp. 165–169. [Google Scholar]
- Klebaner, FC; Lazar, J; Zeitouni, O. On the quasi-stationary distribution for some randomly perturbed transformations of an interval. Ann. Appl. Probab 1998, 8, 300–315. [Google Scholar]
- Yanev, NM; Mitov, KV. Regenerative Branching Processes. In Records and Branching processes; Ahsanullah, M, Yanev, GP, Eds.; Nova Science Publishers, Inc: New York, NY, USA, 2008; Chapter 3; pp. 37–62. [Google Scholar]
- Viet, AF; Fourichon, C; Seegers, H; Jacob, C; Guihenneuc-Jouyaux, C. A model of the spread of the bovine viral-diarrhoea virus within a dairy herd. Prev. Vet. Med 2004, 63, 211–236. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jacob, C. Branching Processes: Their Role in Epidemiology. Int. J. Environ. Res. Public Health 2010, 7, 1186-1204. https://doi.org/10.3390/ijerph7031204
Jacob C. Branching Processes: Their Role in Epidemiology. International Journal of Environmental Research and Public Health. 2010; 7(3):1186-1204. https://doi.org/10.3390/ijerph7031204
Chicago/Turabian StyleJacob, Christine. 2010. "Branching Processes: Their Role in Epidemiology" International Journal of Environmental Research and Public Health 7, no. 3: 1186-1204. https://doi.org/10.3390/ijerph7031204
APA StyleJacob, C. (2010). Branching Processes: Their Role in Epidemiology. International Journal of Environmental Research and Public Health, 7(3), 1186-1204. https://doi.org/10.3390/ijerph7031204



