The Changing Disease-Scape in the Third Epidemiological Transition
Abstract
:1. Introduction
2. Background
3. Improvements upon Earlier Models
4. Profiles of the Major Transitions
4.1. The Paleolithic Baseline
4.2. The First Epidemiological Transition
4.3. The Second Epidemiological Transition
4.4. The Third Epidemiological Transition
5. Challenges to the Epidemiological Transitions Model
6. Conclusion
Acknowledgments
References
- Armelagos, GJ; Barnes, K. The evolution of human disease and the rise of allergy: Epidemiological transitions. Med. Anthropol 1999, 18, 187–213. [Google Scholar]
- Armelagos, GJ; Barnes, KC; Lin, J. Disease in human evolution: The re-emergence of infectious disease in the third epidemiological transition. AnthroNotes 1996, 18, 1–7. [Google Scholar]
- Barrett, R; Kuzawa, CW; McDade, T; Armelagos, GJ. Emerging infectious disease and the third epidemiological transition. In Annual Review of Anthropology; Durham, W, Ed.; Annual Reviews Inc: Palo Alto, CA, USA, 1998; Volume 27, pp. 247–271. [Google Scholar]
- Omran, AR. The epidemiologic transition: A theory of the epidemiology of population change. Milbank Q 1971, 49, 509–538. [Google Scholar]
- Omran, AR. A century of epidemiologic transition in the United States. Prev. Med 1977, 6, 30–51. [Google Scholar]
- Omran, AR. The epidemiologic transition theory revisited thirty years later. World Health Stat. Q 1998, 51, 99–119. [Google Scholar]
- Kirk, D. Demographic Transition theory. Population Stud 1996, 50, 361–387. [Google Scholar]
- McKeown, T; Record, RG. Reasons for the decline in mortality in England and Wales during the nineteenth century. Population Stud 1962, 16, 94–122. [Google Scholar]
- Mosher, WD. Demographic responses and demographic transitions: a case study of Sweden. Demography 1980, 17, 395–412. [Google Scholar]
- Carolina, MS; Gustavo, LF. Epidemiological transition: model or illusion? A look at the problem of health in Mexico. Soc. Sci. Med 2003, 57, 539–550. [Google Scholar]
- Munoz-Tuduri, M; Garcia-Moro, C; Walker, PL. Time series analysis of the epidemiological transition in Minorca, 1634–1997. Hum. Biol 2006, 78, 619–634. [Google Scholar]
- Morand, OF. Economic growth, longevity and the epidemiological transition. Eur. J. Health Econ 2004, 5, 166–174. [Google Scholar]
- Caldwell, JC. Population health in transition. Bull. World Health Organ 2001, 79, 159–160. [Google Scholar]
- Girard, DZ. The cost of epidemiological transition: a study of a decrease in pertussis vaccination coverage. Health Policy 2005, 74, 287–303. [Google Scholar]
- Huicho, L; Trelles, M; Gonzales, F; Mendoza, W; Miranda, J. Mortality profiles in a country facing epidemiological transition: an analysis of registered data. BMC Public Health 2009, 9, 47. [Google Scholar]
- Mackenbach, JP. The epidemiological transition theory. J. Epidemiol. Community Health 1994, 48, 329–331. [Google Scholar]
- Cappuccio, FP. Commentary: epidemiological transition, migration, and cardiovascular disease. Int. J. Epidemiol 2004, 33, 387–388. [Google Scholar]
- Gulliford, MC. Epidemiological transition in Trinidad and Tobago, West Indies 1953−1992. Int. J. Epidemiol 1996, 25, 357–365. [Google Scholar]
- Nakatani, H; Fujii, N; Mori, T; Hoshinot, H. Epidemiological transition of tuberculosis and future agenda of control in Japan: results of the Ad-Hoc National Survey of Tuberculosis 2000. Int. J. Tuberc. Lung Dis 2002, 6, 198–207. [Google Scholar]
- Quigley, MA. Commentary: shifting burden of disease—epidemiological transition in India. Int. J. Epidemiol 2006, 35, 1530–1531. [Google Scholar]
- Schooneveldt, M; Songer, T; Zimmet, P; Thoma, K. Changing mortality patterns in Nauruans: an example of epidemiological transition. J. Epidemiol. Community Health 1988, 42, 89–95. [Google Scholar]
- Thompson, WS. Population. Am. J. Sociol 1929, 34, 959–975. [Google Scholar]
- Notestein, FW. Population—the long view. In Food for the World; Schultz, TW, Ed.; University of Chicago Press: Chicago, IL, USA, 1945. [Google Scholar]
- Davis, K. The world demographic transition. Ann. Amer. Acad. Polit. Soc. Sci 1945, 237, 1–11. [Google Scholar]
- Angel, J. Health as a crucial factor in the changes from hunting to developed farming in the eastern mediterranean. In Paleopathology at the Origins of Agriculture; Cohen, MN, Armelagos, G, Eds.; Academic Press: Orlando, FL, USA, 1984; pp. 51–73. [Google Scholar]
- Cook, DC. Subsistence and health in the lower Illinois valley: osteological evidence. In Paleopathology at the Origins of Agriculture; Cohen, MN, Armelagos, G, Eds.; Academic Press: Orlando, FL, USA, 1984; pp. 235–269. [Google Scholar]
- Goodman, A; Lallo, J; Armelagos, G; Rose, J. Health changes at Dickson Mounds, Illinois (AD 950–1300). In Paleopathology at the Origins of Agriculture; Cohen, MN, Armelagos, G, Eds.; Academic Press: Orlando, FL, USA, 1984; pp. 271–305. [Google Scholar]
- Armelagos, GJ; Goodman, AH; Jacobs, KH. The origins of agriculture: population growth during a period of declining health. Popul. Environ 1991, 13, 9–22. [Google Scholar]
- Armelagos, GJ; Harper, KN. Disease globalization in the third epidemiological transition. In Globalization, Health and the Environment: An Integrated Perspective; Guest, G, Ed.; AltaMira Press: Walnut Creek, CA, USA, 2005; pp. 27–33. [Google Scholar]
- Lambrecht, FL. Paleoecology of tsetse flies and sleeping sickness in Africa. Proc. Am. Philos. Soc 1980, 124, 367–385. [Google Scholar]
- Lambrecht, FL. Trypanosomiasis in prehistoric and later human populations: A tentative reconstruction. In Diseases in Antiquity; Brothwell, D, Sandison, AT, Eds.; Thomas: Springfield, IL, USA, 1967; pp. 132–151. [Google Scholar]
- Lambrecht, FL. Trypanosomes and hominid evolution. Bioscience 1985, 35, 640–646. [Google Scholar]
- Desowitz, RS. Epidemiological-ecological interactions in savanna environments. In Human Ecology in Savanna Environments; Harris, DR, Ed.; Academic Press: London, UK, 1980; pp. 457–477. [Google Scholar]
- Cockburn, TA. The evolution of human infectious diseases. In Infectious Diseases: Their Evolution and Eradication; Cockburn, TA, Ed.; Charles C. Thomas: Springfield, IL, USA, 1967; pp. 84–107. [Google Scholar]
- Cockburn, TA. Infectious disease in ancient populations. Cur. Anthropol 1971, 12, 45–62. [Google Scholar]
- Polgar, S. Evolution and the ills of mankind. In Horizons of Anthropology; Tax, S, Ed.; Aldine Pub Comp: Chicago, IL, USA, 1964; pp. 200–211. [Google Scholar]
- Armelagos, G. Health and disease in prehistoric populations in transition. In Disease in Populations in Transition; Swedlund, AC, Armelagos, GA, Eds.; Bergin and Garvey: New York, NY, USA, 1990; pp. 127–144. [Google Scholar]
- Armelagos, GJ. Human evolution and the evolution of human disease. Ethn. Dis 1991, 1, 21–26. [Google Scholar]
- Steckels, R; Rose, J. The Body as Evidence; the Body of Evidence; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Armelagos, GJ; Brown, PJ. The body as evidence; the body of evidence. In The Backbone of History; Steckels, R, Rose, J, Eds.; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- May, JM. The ecology of human disease. Ann. N.Y. Acad. Sci 1960, 84, 789–794. [Google Scholar]
- Turshen, M. The political ecology of disease. Rev. Radical Polit. Econ 1977, 9, 45–60. [Google Scholar]
- Link, BG; Phelan, J. Social conditions as fundamental causes of disease. J. Health Soc. Behav 1995, 35, 80–94. [Google Scholar]
- Farmer, P. Social inequalities and emerging infectious diseases. Emerg. Infect. Dis 1996, 2, 259–269. [Google Scholar]
- Working Together for a Healthy World The World Health Report 2006; World Health Organization: Geneva, Switzerland, 2006.
- Armelagos, GJ; Ryan, M; Leatherman, T. The evolution of disease: A biocultural analysis of AIDS. Am. J. Human Biol 1990, 2, 353–363. [Google Scholar]
- Audy, JR; Dunn, FL. Health and disease. In Human Ecology; Sargent, I, Ed.; North Holland Publishing Co.: Amsterdam, The Netherlands, 1974. [Google Scholar]
- Webby, RJ; Webster, RG. Are we ready for pandemic influenza? Science 2003, 302, 1519–1522. [Google Scholar]
- Sabroza, PC; Waltner-Toews, D. Emerging diseases, local systems, and globalization. Cad. Saude Publica 2001, 17, 5. [Google Scholar]
- Thorpe, CM. Shiga Toxin–Producing Escherichia coli Infection. Clin. Infect. Dis 2004, 38, 1298–1303. [Google Scholar]
- Henniga, B; Ormsbeeb, L; Bachasc, L; Silverstoned, A; Milnere, J; Carpenterf, D; Thompsong, C; Sukg, WA. Introductory comments: nutrition, environmental toxins and implications in prevention and intervention of human diseases. J. Nutr. Biochem 2007, 18, 161–162. [Google Scholar]
- Agaibi, CE; Wilson, JP. Trauma, PTSD, and resilience: a review of the literature. Trauma Violence Abuse 2005, 6, 195–216. [Google Scholar]
- Kampa, M; Castanas, E. Human health effects of air pollution. Environ. Pollut 2008, 151, 362–367. [Google Scholar]
- Kahn, RS; Wise, PH; Kennedy, BP; Kawachi, I. State income inequality, household income, and maternal mental and physical health: cross sectional national survey. BMJ 2000, 321, 1311–1315. [Google Scholar]
- Smocovitis, VB. Unifying biology: the evolutionary synthesis and evolutionary biology. J. Hist. Biol 1992, 25, 1–65. [Google Scholar]
- Smocovitis, VB. Unifying Biology: The Evolutionary Synthesis and Evolutionary Biology; Princeton University Press: Princeton, NJ, USA, 1996. [Google Scholar]
- Armelagos, GJ; Goodman, AH; Jacobs, KH. The ecological perspective in disease. In Health and the Human Condition; Logan, M, Hunt, E, Eds.; Duxbury Press: North Scituate, MA, USA, 1978; pp. 71–83. [Google Scholar]
- Rambo, AT. Searching for Vietnam: Selected Writings on Vietnamese Culture and Society; Kyoto University Press: Kyoto, Japan, 2005. [Google Scholar]
- Sponheimer, M; Lee-Thorp, JA. Isotopic evidence for the diet of an early hominid, Australopithecus africanus. Science 1999, 283, 368–370. [Google Scholar]
- Shipman, P. What can you do with a bone fragment? Proc. Natl. Acad. Sci. USA 2001, 98, 1335–1337. [Google Scholar]
- Strait, DS; Weber, GW; Neubauer, S; Chalk, J; Richmond, BG; Lucas, PW; Spencer, MA; Schrein, C; Dechow, PC; Ross, CF; Grosse, IR; Wright, BW; Constantino, P; Wood, BA; Lawn, B; Hylander, WL; Wang, Q; Byron, C; Slice, DE; Smith, AL. The feeding biomechanics and dietary ecology of Australopithecus africanus. Proc. Natl. Acad. Sci. USA 2009, 106, 2124–2129. [Google Scholar]
- Krebs, JR. The gourmet ape: evolution and human food preferences. Am. J. Clin. Nutr 2009, 90, 707–711. [Google Scholar]
- Hockett, B; Haws, J. Nutritional ecology and diachronic trends in Paleolithic diet and health. Evol. Anthropol 2003, 12, 211–216. [Google Scholar]
- Dart, RA. Australopithecus africanus: The man-ape of South Africa. Nature 1925, 115, 195. [Google Scholar]
- Washburn, SL; Devore, I. Social behavior of baboons and early man. In The Social Life of Early Man; Washburn, SL, Ed.; Viking Foundation Publication in Anthropology; Aldine: Chicago, IL, USA, 1961; Volume 31, pp. 91–95. [Google Scholar]
- Kingston, JD; Hill, A; Marino, BD. Isotopic evidence for neogene hominid paleoenvironments in the Kenya Rift Valley. Science 1994, 264, 955–959. [Google Scholar]
- Gibbons, A. A new kind of ancestor: Ardipithecus unveiled. Science 2009, 326, 36–40. [Google Scholar]
- Gibbons, A. Habitat for Humanity. Science 2009, 326, 40. [Google Scholar]
- White, TD; Ambrose, SH; Suwa, G; Su, DF; DeGusta, D; Bernor, RL; Boisserie, J-R; Brunet, M; Delson, E; Frost, S; Garcia, N; Giaourtsakis, IX; Haile-Selassie, Y; Howell, FC; Lehmann, T; Likius, A; Pehlevan, C; Saegusa, H; Semprebon, G; Teaford, M; Vrba, E. Macrovertebrate paleontology and the pliocene habitat of Ardipithecus ramidus. Science 2009, 326, 67–93. [Google Scholar]
- White, TD; Asfaw, B; Beyene, Y; Haile-Selassie, Y; Lovejoy, CO; Suwa, G; WoldeGabriel, G. Ardipithecus ramidus and the paleobiology of early hominids. Science 2009, 326, 64–86. [Google Scholar]
- WoldeGabriel, G; Ambrose, SH; Barboni, D; Bonnefille, R; Bremond, L; Currie, B; DeGusta, D; Hart, WK; Murray, AM; Renne, PR; Jolly-Saad, MC; Stewart, KM; White, TD. The geological, isotopic, botanical, invertebrate, and lower vertebrate surroundings of Ardipithecus ramidus. Science 2009, 326, 65–655. [Google Scholar]
- Burnet, FM. Natural History of Infectious Disease; Cambridge University Press: Cambridge, UK, 1962. [Google Scholar]
- Bengmark, S. Bacteria for optimal health. Nutrition 2000, 16, 611–615. [Google Scholar]
- Armelagos, GJ; Harper, KN. Genomics at the origins of agriculture, Part One. Evol. Anthropol 2005, 14, 68–77. [Google Scholar]
- Armelagos, GJ; Harper, KN. Genomics at the origins of agriculture. Part Two. Evol. Anthropol 2005, 14, 100–121. [Google Scholar]
- Hoberg, EP; Jones, A; Rausch, RL; Eom, KS; Gardner, SL. A phylogenetic hypothesis for species of the genus Taenia (Eucestoda: Taeniidae). J. Parasitol 2000, 86, 89–98. [Google Scholar]
- Hoberg, EP; Alkire, NL; de Queiroz, A; Jones, A. Out of Africa: origins of the Taenia tapeworms in humans. Proc. R. Soc. Lond. B Biol. Sci 2001, 268, 781–787. [Google Scholar]
- Laird, M. Vector-borne disease introduced into new areas due to human movements: A historical perspective. In Demography and Vector-Borne Diseases; Service, MW, Ed.; CRC Press Inc.: Boca Raton, FL, USA, 1989; pp. 17–33. [Google Scholar]
- Yong, Z; Fournier, P; Rydkina, E; Raoult, D. The geographical segregation of human lice preceded that of Pediculus humanus capitis and Pediculus humanus humanus. C. R. Biol 2003, 326, 565–574. [Google Scholar]
- Cockburn, TA. Infections of the order primates. In Infectious Diseases: Their Evolution and Eradication; Cockburn, TA, Ed.; C.C. Thomas: Springfield, IL, USA, 1967. [Google Scholar]
- Lovell, N; Jurmain, R; Kilgore, L. Skeletal evidence of probable treponemal infection in free-ranging African apes. Primates 2000, 41, 275–290. [Google Scholar]
- Woodford, M; Butynski, T; Karesh, W. Habituating the great apes: the disease risks. Oryx 2002, 36, 153–160. [Google Scholar]
- Harper, KN; Liu, H; Ocampo, P; Steiner, B; Martin, A; Levert, K; Wang, D; Sutton, M; Armelagos, G. The sequence of the acidic repeat protein (arp) gene differentiates venereal from nonvenereal Treponema pallidum subspecies, and the gene has evolved under strong positive selection in the subspecies that causes syphilis. FEMS Immunol. Medical Microbiol 2008, 53, 322–332. [Google Scholar]
- Harper, KN; Ocampo, P; Steiner, B; George, R; Silverman, M; Bolotin, S; Pillay, A; Saunders, N; Armelagos, G. On the origin of the treponematoses: a phylogenetic approach. PLoS Negl. Trop. Dis 2008, 2, e148. [Google Scholar]
- Tishkoff, SA; Varkonyi, R; Cahinhinan, N; Abbes, S; Argyropoulos, G; Destro-Bisol, G; Drousiotou, A; Dangerfield, B; Lefranc, G; Loiselet, J; Piro, A; Stoneking, M; Tagarelli, A; Tagarelli, G; Touma, EH; Williams, SM; Clark, AG. Haplotype diversity and linkage disequilibrium at human G6PD: recent origin of alleles that confer malarial resistance. Science 2001, 293, 455–462. [Google Scholar]
- Joy, D; Feng, X; Mu, J; Furuya, T; Chotivanich, K; Krettli, A; Ho, M; Wang, A; White, N; Suh, E; Beerli, P; Su, X. Early origin and recent expansion of Plasmodium falciparum. Science 2003, 300, 318–321. [Google Scholar]
- Gordon, S; Brosch, R; Billault, A; Garnier, T; Eiglmeier, K; Cole, S. Identification of variable regions in the genomes of tubercle bacilli using bacterial artificial chromosome arrays. Mol. Microbiol 1999, 32, 643–655. [Google Scholar]
- Mostowy, S; Cousins, D; Brinkman, J; Aranaz, A; Behr, M. Genomic deletions suggest a phylogeny for the Mycobacterium tuberculosis complex. J. Infect. Dis 2002, 186, 74–80. [Google Scholar]
- Audy, JR. The ecology of scrub typhus. In Studies in Disease Ecology; May, JM, Ed.; Hafner Publishing: New York, NY, USA, 1961; pp. 389–432. [Google Scholar]
- Livingstone, FB. Anthropological implications of sickle-cell distribution in West Africa. Am. Anthropol 1958, 60, 533–562. [Google Scholar]
- Stuart-Macadam, P. Anemia in past human populations. In Diet, Demography, and Disease: Changing Perspectives on Anemia; Stuart-Macadam, P, Kent, S, Eds.; Aldine de Gruyter: New York, NY, USA, 1992; pp. 151–172. [Google Scholar]
- Chandra, R. Nutrition and immunology: from the clinic to cellular biology and back again. Proc. Nutr. Soc 1999, 58, 681–683. [Google Scholar]
- Käferstein, F. Food safety: a pressing public-health and economic issue. In Understanding the Global Dimensions of Health; Gunn, S, Mansourian, P, Davies, A, Piel, A, Sayers, B, Eds.; Springer: New York, NY, USA, 2005; pp. 199–212. [Google Scholar]
- Cohen, MN; Armelagos, GJ. Paleopathology at the Origins of Agriculture; Academic Press: New York, NY, USA, 1984. [Google Scholar]
- McKeown, T. The Modern Rise of Population; Academic Press: New York, NY, USA, 1976. [Google Scholar]
- McKeown, T. Determinants of health. Hum. Nature 1978, 1, 60–67. [Google Scholar]
- McKeown, T. The Origins of Human Diseases; Basil Blackwell: Oxford, UK, 1988. [Google Scholar]
- McKeown, T; Brown, RG. Medical evidence related to English population changes in the eighteenth century. Population Stud 1955, 9, 119–141. [Google Scholar]
- Olshansky, S; Ault, A. The fourth stage of the epidemiologic transition: the age of delayed degenerative diseases. Milbank Q 1986, 64, 355–391. [Google Scholar]
- McKeown, T. The Role of Medicine: Dream, Mirage or Nemisis; Princeton University Press: Princeton, NJ, USA, 1979. [Google Scholar]
- Schofield, R; Reher, D. The decline of mortality in Europe. In The Decline of Mortality in Europe; Schofield, R, Reher, D, Bideau, A, Eds.; Claredon Press: Oxford, UK, 1991; pp. 1–17. [Google Scholar]
- Johansson, SR. The health transition: the cultural inflation of morbidity during the decline of mortality. Health Transit. Rev 1991, 1, 39–68. [Google Scholar]
- Johansson, SR. Measuring the cultural inflation of morbidity during the decline in mortality. Health Transit. Rev 1992, 2, 78–89. [Google Scholar]
- Kunitz, SJ. The personal physician and the decline of mortality. In The Decline of Mortality in Europe; Sclofield, DR, Bideau, A, Eds.; Clarendon Press: Oxford, UK, 1991; pp. 248–262. [Google Scholar]
- Woods, R. The role of Public Health in the nineteenth-century mortality decline. In What We Know About Health Transition: The Cultural, Social, and Behavioral Determinants of Health; Caldwell, J, Findley, S, Caldwell, P, Santow, G, Cosford, W, Braid, J, Broers_Freeman, D, Eds.; Health Transition Centre: Canberra, Australia, 1990; Volume 1, pp. 110–115. [Google Scholar]
- Colgrove, J. The McKeown thesis: a historical controversy and its enduring influence. Am. J. Public Health 2002, 92, 725–729. [Google Scholar]
- Bynum, B. The McKeown thesis. Lancet 2008, 371, 644–645. [Google Scholar]
- Grundy, E. Commentary: The McKeown debate: time for burial. Int. J. Epidemiol 2005, 34, 529–533. [Google Scholar]
- Lawson, J. Rethinking McKeown. Am J Public Health 2003, 93, 1032. [Google Scholar]
- Szreter, S. Rethinking McKeown: The relationship between Public Health and social change. Am. J. Public Health 2002, 92, 722–725. [Google Scholar]
- Aberg, N; Hesselmar, B; Aberg, B; Eriksson, B. Increase of asthma, allergic rhinitis and eczema in Swedish schoolchildren between 1979 and 1991. Clin. Exper. Allergy 2005, 25, 815–819. [Google Scholar]
- Maziak, W; Behrens, T; Brasky, T; Duhme, H; Rzehak, P; Weiland, S; Keil, U. Are asthma and allergies in children and adolescents increasing? Results from ISAAC phase I and phase III surveys in Münster, Germany. Allergy 2003, 58, 572–579. [Google Scholar]
- Armelagos, G. The Paleolithic disease-scape, the hygiene hypothesis, and the second epidemiological transition. In The Hygiene Hypothesis and Darwinian Medicine; Rook, G, Ed.; Birkhäuser: Basel, Switzerland, 2009. [Google Scholar]
- Strachan, D. Hay fever, hygiene, and household size. BMJ 1989, 299, 1259–1260. [Google Scholar]
- Bremner, SA; Carey, IM; DeWilde, S; Richards, N; Maier, WC; Hilton, SR; Strachan, DP; Cook, DG. Infections presenting for clinical care in early life and later risk of hay fever in two UK birth cohorts. Allergy 2008, 63, 274–283. [Google Scholar]
- Rook, GA. The hygiene hypothesis and the increasing prevalence of chronic inflammatory disorders. Trans. R. Soc. Trop. Med. Hyg 2007, 101, 1072–1074. [Google Scholar]
- Rook, GA; Adams, V; Hunt, J; Palmer, R; Martinelli, R; Brunet, LR. Mycobacteria and other environmental organisms as immunomodulators for immunoregulatory disorders. Springer Semin. Immun 2004, 25, 237–255. [Google Scholar]
- Rook, GA; Brunet, LR. Old friends for breakfast. Clin. Exp. Allergy 2005, 35, 841–842. [Google Scholar]
- Rook, GA; Brunet, LR. Microbes, immunoregulation, and the gut. Gut 2005, 54, 317–320. [Google Scholar]
- Rook, GA; Hamelmann, E; Brunet, LR. Mycobacteria and allergies. Immunobiology 2007, 212, 461–473. [Google Scholar]
- Thrall, P; Antonovics, J; Wilson, W. Allocation to sexual versus nonsexual disease transmission. Am. Nat 1998, 151, 29–45. [Google Scholar]
- Lockhart, A; Trhall, P; Antonovics, J. Sexually transmitted diseases in animals: ecological and evolutionary implications. Biol. Rev 1996, 71, 415–471. [Google Scholar]
- Cowan, F; Johnson, A; Ashley, R; Corey, L; Mindel, A. Relationship between antibodies to herpes simplex virus (HSV) and symptoms of HSV infection. J. Infect. Dis 1996, 174, 470–475. [Google Scholar]
- Fleming, D; McQuillan, G; Johnson, R; Nahmias, A; Aral, S; Lee, F; Louis, MS. Herpes simplex virus type 2 in the United States, 1976 to 1994. N. Engl. J. Med 1997, 337, 1105–1111. [Google Scholar]
- Johnson, R; Nahmias, A; Magder, L; Lee, F; Brooks, C; Snowden, C. A seroepidemiologic survey of the prevalence of herpes simplex virus type 2 infection in the United States. N. Engl. J. Med 1989, 321, 7–12. [Google Scholar]
- Becker, T; Blount, J; Guinan, M. Genital herpes infections in private practice in the United States, 1966 to 1981. JAMA 1985, 253, 1601–1603. [Google Scholar]
- Smith, I; Peutherer, J; MacCallum, F. The incidence of herpesvirus hominis antibody in the population. J. Hyg. (London) 1967, 65, 395–408. [Google Scholar]
- Nahmias, A; Lee, F; Beckman-Nahmias, S. Seroepidemiological and sociological patterns of herpes simplex virus infection in the world. Scand. J. Infect. Dis 1990, 69, 19–36. [Google Scholar]
- Van der Sluis, L. The Treponematosis of Tahiti. Its Origin and Evolution. A Study of the Sources; Peco: Amsterdam, The Netherland, 1969. [Google Scholar]
- Willcox, R. Venereal diseases in the Pacific Islands: Papua New Guinea. Brit. J. Ven. Dis 1980, 56, 277–281. [Google Scholar]
- Brandt, A. No Magic Bullet: A Social History of Venereal Disease in the United States Since 1880; Oxford University Press: Oxford, UK, 1987. [Google Scholar]
- Kombe, GC; Darrow, DM. Revisiting emerging infectious diseases: the unfinished agenda. J. Community Health 2001, 26, 113–122. [Google Scholar]
- McMichael, AJ. The urban environment and health in a world of increasing globalization: issues for developing countries. Bull. World Health Organ 2000, 78, 1117–1126. [Google Scholar]
- Woolhouse, ME. Epidemiology: emerging diseases go global. Nature 2008, 451, 898–899. [Google Scholar]
- Raviglione, M; Smith, I. XDR tuberculosis—implications for global public health. N. Engl. J. Med 2007, 356, 656–659. [Google Scholar]
- Quirk, M. First VRSA isolate identified in USA. Lancet 2002, 2, 510. [Google Scholar]
- Waters, WF. Globalization, socioeconomic restructuring, and community health. J. Community Health 2001, 26, 79–92. [Google Scholar]
- Armelagos, GJ. The viral superhighway. The Sciences 1998, 38, 24–30. [Google Scholar]
- Satcher, D. Emerging infections: getting ahead of the curve. Emerg. Infect. Dis 1995, 1, 1–6. [Google Scholar]
- Lederberg, J. Infectious disease as an evolutionary paradigm. Emerg. Infect. Dis 1997, 3, 417–423. [Google Scholar]
- Morse, SS. Factors in the emergence of infectious diseases. Emerg. Infect. Dis 1995, 1, 7–15. [Google Scholar]
- Farmer, P. Pathologies of Power: Health, Human Rights, and the New War on the Poor; University of California Press: Berkeley, CA, USA, 2005. [Google Scholar]
- Olshansky, S; Passaro, D; Hershow, R; Layden, J; Carnes, B; Brody, J; Hayflick, L; Butler, R; Allison, D; Ludwig, D. A potential decline in life expectancy in the United States in the 21st century. N. Engl. J. Med 2005, 352, 1138–1145. [Google Scholar]
- McNeill, W. Plagues and Peoples; Anchor: New York, NY, USA, 1977. [Google Scholar]
- Parkin, D. Global cancer statistics in the year 2000. Lancet Oncol 2001, 2, 533–543. [Google Scholar]
- Bonow, R; Gheorghiade, M. The diabetes epidemic: a national and global crisis. Am. J. Med 2004, 116, 2–10. [Google Scholar]
- Kinsella, K. Changes in life expectancy 1900−1990. Am. J. Clin. Nutr 1992, 55, S1196–1202. [Google Scholar]
- Guyer, B; Freedman, M; Strobino, D; Sondik, E. Annual summary of vital statistics: trends in the health of Americans during the 20th century. Pediatrics 2000, 106, 1307–1317. [Google Scholar]
- Armstrong, G; Conn, L; Pinner, R. Trends in infectious disease mortality in the United States during the 20th century. JAMA 1999, 281, 61–66. [Google Scholar]
- Cooper, R; Cutler, J; Desvigne-Nickens, P; Fortmann, S; Friedman, L; Havlik, R; Hogelin, G; Marler, J; McGovern, P; Morosco, G; Mosca, L; Pearson, T; Stamler, J; Stryer, D; Thom, T. Trends and disparities in coronary heart disease, stroke, and other cardiovascular diseases in the United States: findings of the National Conference on Cardiovascular Disease Prevention. Circulation 2000, 102, 3137–3147. [Google Scholar]
- Wrigley, E; Schofield, R. The Population History of England 1541–1871; Cambridge University Press: Cambridge, UK, 1989; Volume 46. [Google Scholar]
- Flinn, M. The European Demographic System, 1500−1820; Johns Hopkins University Press: Baltimore, MA, USA, 1985. [Google Scholar]
- Hart, C; Kariuki, S. Antimicrobial resistance in developing countries. BMJ 1998, 317, 647–650. [Google Scholar]
- Vlieghe, E; Phoba, M; Tamfun, J; Jacobs, J. Antibiotic resistance among bacterial pathogens in Central Africa: a review of the published literature between 1955 and 2008. Int. J. Antimicrob. Ag 2009, 34, 295–303. [Google Scholar]
- Levy, S. Antibiotic resistance: consequences of inaction. Clin. Infect. Dis 2001, 33, S124–129. [Google Scholar]
- Popkin, B. The nutrition transition and obesity in the developing world. J. Nutr 2001, 131, S871–873. [Google Scholar]
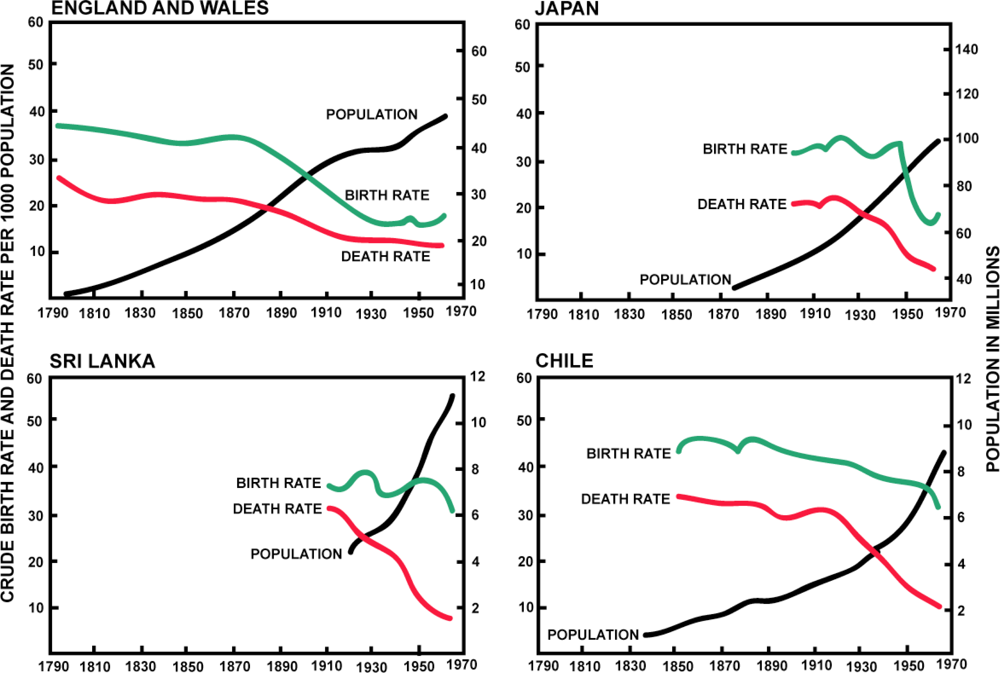
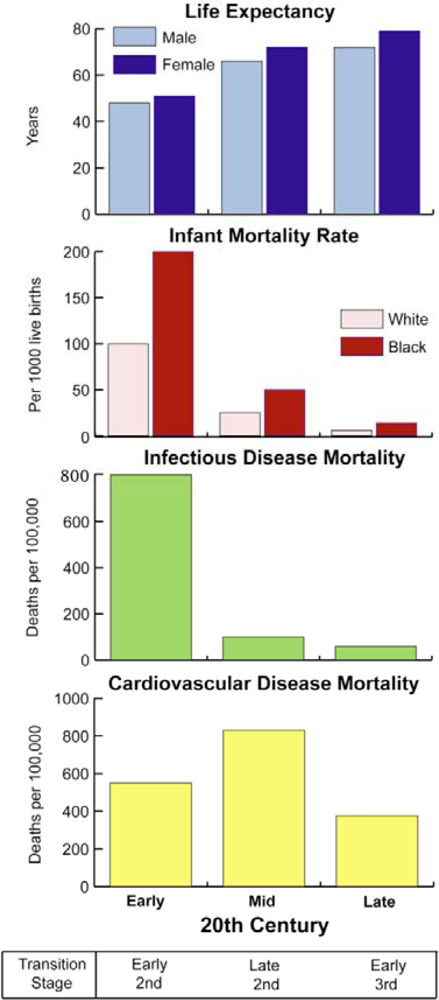
| Transition | Paleolithic Baseline | First Transition | Second Transition | Third Transition |
|---|---|---|---|---|
| Time Period | Pre-Neolithic Cultures; More recent hunter-gatherer cultures with little outside contact | Neolithic cultures-Early Modern Times in Western Europe and United States; Still characterizes many low-income countries | Early Modern times to 20th century in Western Europe, United States; Occurred more recently in some other high-income countries and is in progress in lower income countries | End of the 20th century to the present, global |
| Characteristics |
|
|
|
|
| Common causes of morbidity and mortality | Infections such as tapeworms, body lice, pinworms, typhoid, staph, and possibly yaws |
|
|
|
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Harper, K.; Armelagos, G. The Changing Disease-Scape in the Third Epidemiological Transition. Int. J. Environ. Res. Public Health 2010, 7, 675-697. https://doi.org/10.3390/ijerph7020675
Harper K, Armelagos G. The Changing Disease-Scape in the Third Epidemiological Transition. International Journal of Environmental Research and Public Health. 2010; 7(2):675-697. https://doi.org/10.3390/ijerph7020675
Chicago/Turabian StyleHarper, Kristin, and George Armelagos. 2010. "The Changing Disease-Scape in the Third Epidemiological Transition" International Journal of Environmental Research and Public Health 7, no. 2: 675-697. https://doi.org/10.3390/ijerph7020675
APA StyleHarper, K., & Armelagos, G. (2010). The Changing Disease-Scape in the Third Epidemiological Transition. International Journal of Environmental Research and Public Health, 7(2), 675-697. https://doi.org/10.3390/ijerph7020675




