Sources of Propylene Glycol and Glycol Ethers in Air at Home
Abstract
:1. Introduction
2. Experimental Section
2.1. Exposure Assessment
2.1.1. Air Sampling
2.1.2. Laboratory Analysis
2.2. Statistical Analysis
2.2.1. Descriptive analysis
2.2.2. Predictive model of indoor PGE concentration
3. Results
3.1. Pattern of Mean PGE Concentration, Compared to the Patterns of Terpenes, Texanols, BBzP and DEHP
3.2. Comparisons of Trends in Compound Groups
3.3. Inspector Rating of Indoor Air Quality
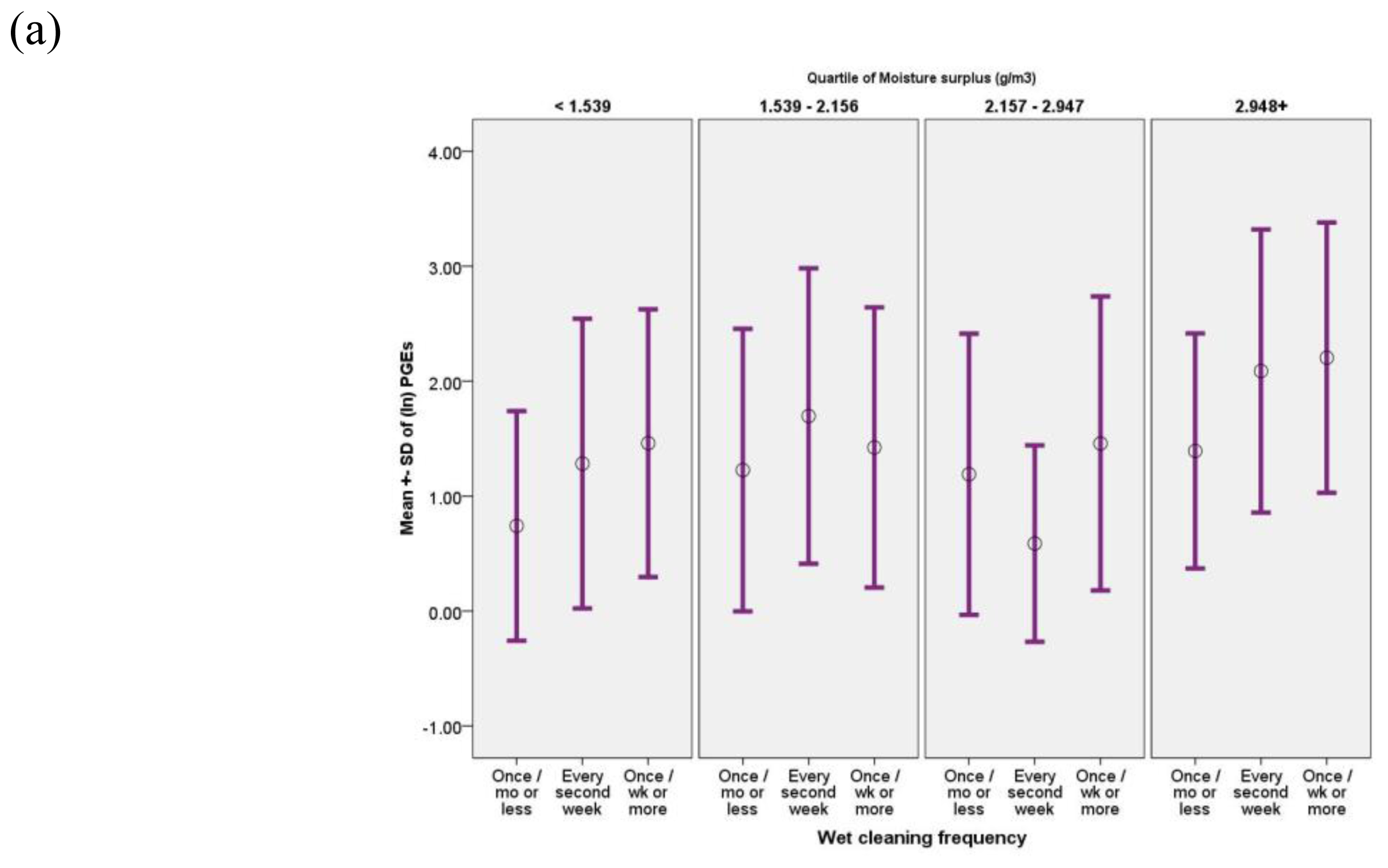
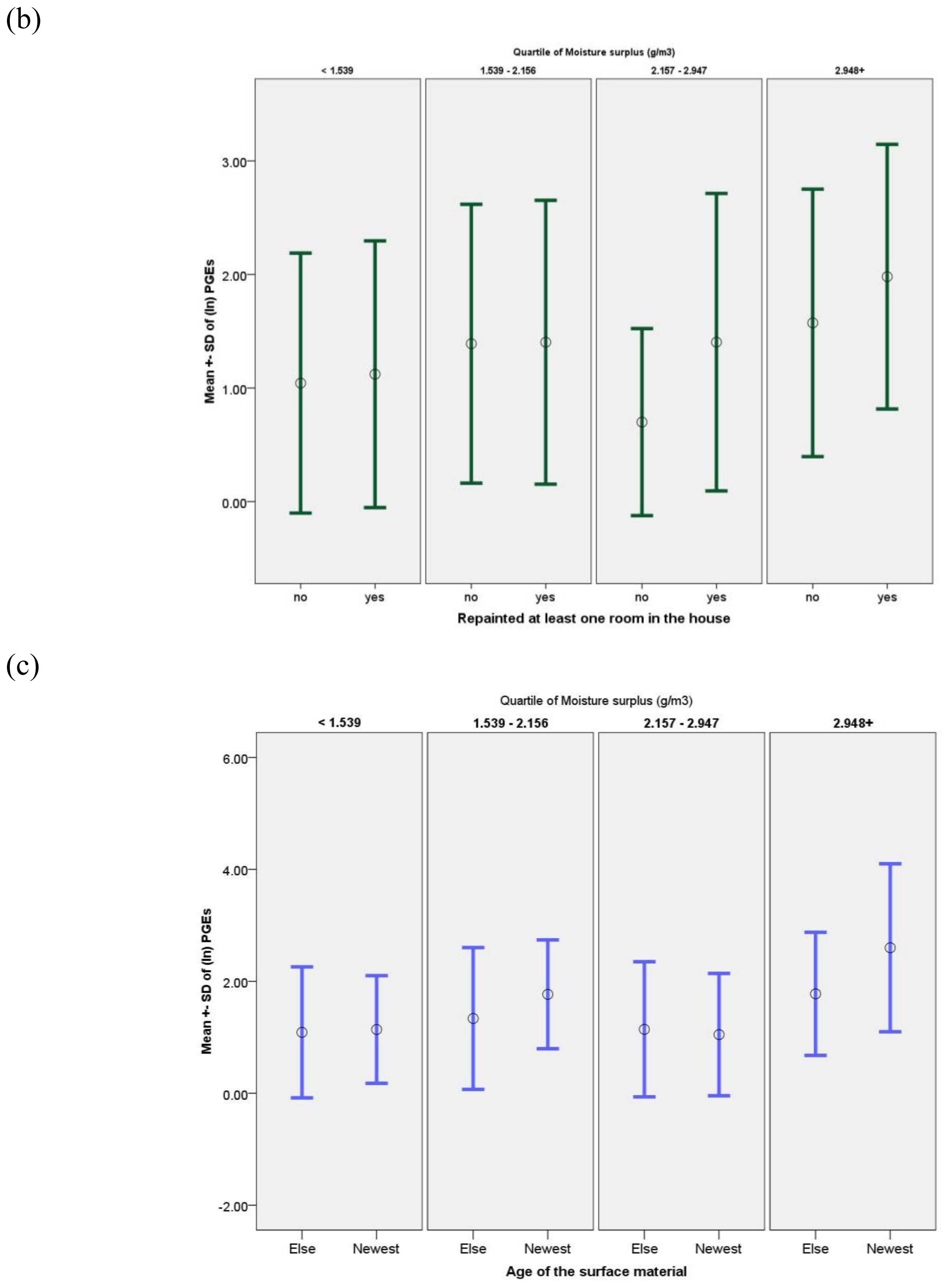
3.4. Modifiers of Indoor PGE Concentrations
3.5. Final Predictive Model
4. Discussion
5. Conclusions
Acknowledgements
Appendix: Online Supporting Document
Detection and Identification
Calibration
Quality Assurance
| Excess Indoor Humidity (g/m3) | Cleaning frequency | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| N | GM | 95% | CI | N | GM | 95% | CI | ||
| 1,2-propanediol | |||||||||
| <1.539 | 29 | 5.26 | 3.5 | 7.92 | ≥once/wk | 64 | 6.12 | 4.85 | 7.73 |
| 1.539–2.156 | 35 | 4.77 | 3.56 | 6.39 | Bi-wkly | 34 | 5.94 | 4.45 | 7.92 |
| 2.157–2.947 | 30 | 5.96 | 3.93 | 9.03 | ≤once/mo | 63 | 4.98 | 3.97 | 6.24 |
| ≥2.948 | 52 | 6.32 | 5.2 | 7.67 | 161 | ||||
| 146 | |||||||||
| 1-methoxy-2-propanol | |||||||||
| <1.539 | 19 | 2.66 | 1.89 | 3.74 | ≥once/wk | 29 | 4.27 | 3.45 | 5.3 |
| 1.539–2.156 | 18 | 4.21 | 2.8 | 6.33 | Bi-wkly | 26 | 3.85 | 2.91 | 5.09 |
| 2.157–2.947 | 15 | 3.54 | 2.58 | 4.85 | ≤once/mo | 31 | 3.01 | 2.34 | 3.89 |
| ≥2.948 | 24 | 4.51 | 3.5 | 5.8 | 86 | ||||
| 76 | |||||||||
| 2-(2-butoxyethoxy)ethanol | |||||||||
| <1.539 | 11 | 1.51 | 0.8 | 2.83 | ≥once/wk | 34 | 3.2 | 2.44 | 4.2 |
| 1.539–2.156 | 10 | 4.51 | 2.28 | 8.92 | Bi-wkly | 14 | 4.42 | 2.67 | 7.33 |
| 2.157–2.947 | 13 | 3.04 | 1.95 | 4.74 | ≤once/mo | 21 | 2.74 | 1.78 | 4.2 |
| ≥2.948 | 21 | 4.30 | 3.16 | 5.83 | 69 | ||||
| 55 | |||||||||
| 1-butoxy-2-propanol | |||||||||
| <1.539 | 2 | 5.11 | 0 | ∞ | ≥once/wk | 4 | 7.44 | 1.12 | 49.34 |
| 1.539–2.156 | 5 | 3.08 | 0.97 | 9.74 | Bi-wkly | 7 | 6.24 | 2.57 | 15.11 |
| 2.157–2.947 | 2 | 2.84 | 0 | ∞ | ≤once/mo | 10 | 3.38 | 1.55 | 7.36 |
| ≥2.948 | 8 | 6.92 | 2.94 | 16.25 | 21 | ||||
| 17 | |||||||||
| 2-(2-butoxyethoxy)ethanol acetate | |||||||||
| <1.539 | 7 | 1.43 | 0.8 | 2.58 | ≥once/wk | 10 | 2.67 | 1.44 | 4.94 |
| 1.539–2.156 | 9 | 2.64 | 1.34 | 5.21 | Bi-wkly | 13 | 2.99 | 1.96 | 4.55 |
| 2.157–2.947 | 5 | 1.89 | 0.67 | 5.29 | ≤once/mo | 10 | 1.67 | 0.98 | 2.85 |
| ≥2.948 | 9 | 3.63 | 2.08 | 6.33 | 33 | ||||
| 30 | |||||||||
| 2-butoxy ethanol | |||||||||
| <1.539 | 2 | 1.7 | 0.03 | 109.77 | ≥once/wk | 14 | 3.25 | 1.95 | 5.44 |
| 1.539–2.156 | 6 | 3.38 | 0.9 | 12.73 | Bi-wkly | 5 | 4.19 | 1.21 | 14.49 |
| 2.157–2.947 | 3 | 1.83 | 0.26 | 13.07 | ≤once/mo | 8 | 2.29 | 1.21 | 4.36 |
| ≥2.948 | 13 | 3.67 | 2.31 | 5.82 | 27 | ||||
| 24 | |||||||||
| 2-(2-(2-butoxyethoxy)ethoxy) ethanol | |||||||||
| <1.539 | 4 | 1.62 | 1.12 | 2.34 | ≥once/wk | 10 | 1.98 | 1.15 | 3.4 |
| 1.539–2.156 | 3 | 0.96 | 0.34 | 2.73 | Bi-wkly | 2 | 2.2 | 0.05 | 102.27 |
| 2.157–2.947 | 3 | 1.39 | 0.33 | 5.78 | ≤once/mo | 8 | 1.28 | 0.87 | 1.89 |
| ≥2.948 | 4 | 3.64 | 1.21 | 10.98 | 20 | ||||
| 14 | |||||||||
| 2-(2-ethoxyethoxy) ethanol | |||||||||
| <1.539 | 3 | 8.07 | 2.43 | 26.79 | ≥once/wk | 7 | 6.4 | 3.63 | 11.31 |
| 1.539–2.156 | 3 | 11.03 | 1.19 | 102.15 | Bi-wkly | 4 | 4.97 | 2.89 | 8.55 |
| 2.157–2.947 | 2 | 3.53 | 0.01 | ∞ | ≤once/mo | 5 | 10.71 | 2.6 | 44.08 |
| ≥2.948 | 6 | 5.76 | 3.2 | 10.36 | 16 | ||||
| 14 | |||||||||
| 1-(2-methoxypropoxy)-2-propanol | |||||||||
| <1.539 | 2 | 5.12 | 0.21 | 122.11 | ≥once/wk | 4 | 4.43 | 0.99 | 19.81 |
| 1.539–2.156 | 3 | 4.94 | 0.31 | 79.13 | Bi-wkly | 3 | 7.76 | 1.58 | 38.18 |
| 2.157–2.947 | 1 | 3.52 | ≤once/mo | 4 | 3.48 | 0.73 | 16.48 | ||
| ≥2.948 | 3 | 3.94 | 0.17 | 91.56 | 11 | ||||
| 9 | |||||||||
| Dipropylene glycol methyl ether | |||||||||
| <1.539 | 1 | 9.16 | ≥once/wk | 2 | 3.67 | 2.95 | 4.58 | ||
| 1.539–2.156 | 2 | 4.02 | 0.01 | ∞ | Bi-wkly | 4 | 4.86 | 1.98 | 11.93 |
| 2.157–2.947 | 1 | 3.74 | ≤once/mo | 1 | 1.83 | ||||
| ≥2.948 | 2 | 2.57 | 0.03 | 193.76 | 7 | ||||
| 6 | |||||||||
| 2-(2-methoxyethoxy) ethanol | |||||||||
| <1.539 | 1 | 3.18 | ≥once/wk | 1 | 15.08 | ||||
| 1.539–2.156 | 0 | Bi-wkly | 1 | 4.84 | |||||
| 2.157–2.947 | 1 | 3.88 | ≤once/mo | 4 | 4.27 | 2.61 | 6.99 | ||
| ≥2.948 | 4 | 6.66 | 2.65 | 16.75 | 6 | ||||
| 6 | |||||||||
| 2-(2-hydroxypropoxy)-1-propanol | |||||||||
| <1.539 | 1 | 0.62 | ≥once/wk | 2 | 1.6 | 0.01 | 206.67 | ||
| 1.539–2.156 | 1 | 2.34 | Bi-wkly | 1 | 0.62 | ||||
| 2.157– 2.947 | 2 | 1.34 | 0.1 | 18.48 | ≤once/mo | 1 | 1.65 | ||
| ≥2.948 | 0 | 4 | |||||||
| 4 | |||||||||
| 1-(2-methoxy-1-methylethoxy)-2-propanol | |||||||||
| <1.539 | 1 | 9.08 | ≥once/wk | 1 | 4.13 | ||||
| 1.539–2.156 | 1 | 6.76 | Bi-wkly | 2 | 7.84 | 1.21 | 50.92 | ||
| 2.157–2.947 | 1 | 4.13 | ≤once/mo | 0 | |||||
| ≥2.948 | 0 | 3 | |||||||
| 3 | |||||||||
| 1-propoxy-2-propanol | |||||||||
| <1.539 | ≥once/wk | 0 | |||||||
| 1.539–2.156 | Bi-wkly | 1 | 1.55 | ||||||
| 2.157–2.947 | ≤once/mo | 1 | 8.91 | ||||||
| ≥2.948 | 2 | ||||||||
| 2-(2-ethoxyethoxy) ethanol acetate | |||||||||
| <1.539 | 0 | ≥once/wk | 0 | ||||||
| 1.539–2.156 | 1 | 6.24 | Bi-wkly | 1 | 10.34 | ||||
| 2.157–2.947 | 0 | ≤once/mo | 1 | 6.24 | |||||
| ≥2.948 | 1 | 10.34 | 2 | ||||||
| 2 | |||||||||
| Texanol A | |||||||||
| <1.539 | 4 | 9.06 | 3.26 | 25.15 | ≥once/wk | 12 | 9.56 | 5.9 | 15.49 |
| 1.539–2.156 | 9 | 7.27 | 4.49 | 11.77 | Bi-wkly | 9 | 6.07 | 3.16 | 11.66 |
| 2.157–2.947 | 6 | 7.12 | 3.49 | 14.52 | ≤once/mo | 16 | 8.37 | 5.9 | 11.88 |
| ≥2.948 | 13 | 10.32 | 6.52 | 16.33 | 37 | ||||
| 32 | |||||||||
| Texanol B | |||||||||
| <1.539 | 19 | 1.99 | 1.43 | 2.77 | ≥once/wk | 30 | 3.31 | 2.19 | 4.99 |
| 1.539–2.156 | 18 | 3.86 | 2.48 | 5.99 | Bi-wkly | 21 | 2.94 | 2.12 | 4.07 |
| 2.157–2.947 | 14 | 3.27 | 1.67 | 6.39 | ≤once/mo | 35 | 3.7 | 2.64 | 5.19 |
| ≥2.948 | 28 | 4.46 | 3.11 | 6.39 | 86 | ||||
| 79 | |||||||||
References
- Jaakkola, JJ; Jaakkola, MS. Professional cleaning and asthma. Curr. Opin. Allergy Clin. Immunol 2006, 6, 85–90. [Google Scholar]
- Bornehag, CG; Nanberg, E. Phthalate exposure and asthma in children. Int. J. Androl 2010, 33, 333–345. [Google Scholar]
- Mendell, MJ. Indoor residential chemical emissions as risk factors for respiratory and allergic effects in children: A review. Indoor Air 2007, 17, 259–277. [Google Scholar]
- Medina-Ramon, M; Zock, JP; Kogevinas, M; Sunyer, J; Torralba, Y; Borrell, A; Burgos, F; Anto, JM. Asthma, chronic bronchitis, and exposure to irritant agents in occupational domestic cleaning: a nested case-control study. Occup. Environ. Med 2005, 62, 598–606. [Google Scholar]
- Medina-Ramon, M; Zock, JP; Kogevinas, M; Sunyer, J; Basagana, X; Schwartz, J; Burge, PS; Moore, V; Anto, JM. Short-term respiratory effects of cleaning exposures in female domestic cleaners. Eur. Respir. J 2006, 27, 1196–1203. [Google Scholar]
- Wieslander, G; Norback, D; Edling, C. Airway symptoms among house painters in relation to exposure to volatile organic compounds (VOCS)—a longitudinal study. Ann. Occup. Hyg 1997, 41, 155–166. [Google Scholar]
- Wieslander, G; Norback, D; Lindgren, T. Experimental exposure to propylene glycol mist in aviation emergency training: acute ocular and respiratory effects. Occup. Environ. Med 2001, 58, 649–655. [Google Scholar]
- Wieslander, G; Norback, D; Nordstrom, K; Walinder, R; Venge, P. Nasal and ocular symptoms, tear film stability and biomarkers in nasal lavage, in relation to building-dampness and building design in hospitals. Int. Arch. Occup. Environ. Health 1999, 72, 451–461. [Google Scholar]
- Kogevinas, M; Zock, JP; Jarvis, D; Kromhout, H; Lillienberg, L; Plana, E; Radon, K; Toren, K; Alliksoo, A; Benke, G; Blanc, PD; Dahlman-Hoglund, A; D’Errico, A; Hery, M; Kennedy, S; Kunzli, N; Leynaert, B; Mirabelli, MC; Muniozguren, N; Norback, D; Olivieri, M; Payo, F; Villani, S; Van Sprundel, M; Urrutia, I; Wieslander, G; Sunyer, J; Anto, JM. Exposure to substances in the workplace and new-onset asthma: An international prospective population-based study (ECRHS-II). Lancet 2007, 370, 336–341. [Google Scholar]
- Mazurek, JM; Filios, M; Willis, R; Rosenman, KD; Reilly, MJ; McGreevy, K; Schill, DP; Valiante, D; Pechter, E; Davis, L; Flattery, J; Harrison, R. Work-related asthma in the educational services industry: California, Massachusetts, Michigan, and New Jersey, 1993–2000. Am. J. Ind. Med 2008, 51, 47–59. [Google Scholar]
- Obadia, M; Liss, GM; Lou, W; Purdham, J; Tarlo, SM. Relationships between asthma and work exposures among non-domestic cleaners in Ontario. Am. J. Ind. Med 2009, 52, 716–723. [Google Scholar]
- Zock, J-P; Plana, E; Jarvis, D; Anto, JM; Kromhout, H; Kennedy, SM; Kunzli, N; Villani, S; Olivieri, M; Toren, K; Radon, K; Sunyer, J; Dahlman-Hoglund, A; Norback, D; Kogevinas, M. The use of household cleaning sprays and adult asthma: An international longitudinal study. Am. J. Respir. Crit. Care Med 2007, 176, 735–741. [Google Scholar]
- Nielsen, GD; Larsen, ST; Olsen, O; Lovik, M; Poulsen, LK; Glue, C; Wolkoff, P. Do indoor chemicals promote development of airway allergy? Indoor Air 2007, 17, 236–255. [Google Scholar]
- Emmen, HH; Muijser, H; Arts, JHE; Prinsen, MK. Human volunteer study with PGME: Eye irritation during vapour exposure. Toxicol Lett 2003. [Google Scholar]
- Choi, H; Schmidbauer, N; Sundell, J; Hasselgren, M; Spengler, J; Bornehag, CG. Common household chemicals and the allergy risks in pre-school age children. PLoS One 2010, 5, e13423. [Google Scholar]
- Singer, BC; Destaillats, H; Hodgson, AT; Nazaroff, WW. Cleaning products and air fresheners: Emissions and resulting concentrations of glycol ethers and terpenoids. Indoor Air 2006, 16, 179–191. [Google Scholar]
- Wieslander, G; Norback, D. Ocular symptoms, tear film stability, nasal patency, and biomarkers in nasal lavage in indoor painters in relation to emissions from water-based paint. Int. Arch. Occup. Environ. Health 2010, 83, 733–741. [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR), Toxicological Profile for 2-butoxyethanol and 2-butoxyethanol Acetate; Department of Health and Human Services, Public Health Service: Atlanta, GA, USA, 1998.
- Rumchev, K; Spickett, J; Bulsara, M; Phillips, M; Stick, S. Association of domestic exposure to volatile organic compounds with asthma in young children. Thorax 2004, 59, 746–751. [Google Scholar]
- Wieslander, G; Norback, D; Venge, P. Changes of symptoms, tear film stability and eosinophilic cationic protein in nasal lavage fluid after re-exposure to a damp office building with a history of flooding. Indoor Air 2007, 17, 19–27. [Google Scholar]
- Stewart, RD; Baretta, ED; Dodd, HC; Torkelson, TR. Experimental human exposure to vapor of propylene glycol monomethyl ether. Experimental human exposure. Arch. Environ. Health 1970, 20, 218–223. [Google Scholar]
- Wieslander, G; Norback, D. A field study on clinical signs and symptoms in cleaners at floor polish removal and application in a Swedish hospital. Int. Arch. Occup. Environ. Health 2010, 83, 585–591. [Google Scholar]
- Miller, RR; Hermann, EA; Young, JT; Calhoun, LL; Kastl, PE. Propylene glycol monomethyl ether acetate (PGMEA) metabolism, disposition, and short-term vapor inhalation toxicity studies. Toxicol. Appl. Pharmacol 1984, 75, 521–530. [Google Scholar]
- Ernstgård, L; Lof, A; Wieslander, G; Norback, D; Johanson, G. Acute effects of some volatile organic compounds emitted from water-based paints. J. Occup. Environ. Med 2007, 49, 880–889. [Google Scholar]
- Wieslander, G; Norback, D; Bjornsson, E; Janson, C; Boman, G. Asthma and the indoor environment: the significance of emission of formaldehyde and volatile organic compounds from newly painted indoor surfaces. Int. Arch. Occup. Environ. Health 1997, 69, 115–124. [Google Scholar]
- Jaakkola, JJ; Oie, L; Nafstad, P; Botten, G; Samuelsen, SO; Magnus, P. Interior surface materials in the home and the development of bronchial obstruction in young children in Oslo, Norway. Am. J. Public Health 1999, 89, 188–192. [Google Scholar]
- Jaakkola, JJK; Parise, H; Kislitsin, V; Lebedeva, NI; Spengler, JD. Asthma, wheezing, and allergies in Russian school children in relation to new surface materials in the Home. Am. J. Public Health 2004, 94, 560–562. [Google Scholar]
- Wolkoff, P; Schneider, T; Kildesø, J; Degerth, R; Jaroszewski, M; Schunk, H. Risk in cleaning: chemical and physical exposure. Sci. Total Environ 1998, 215, 135–156. [Google Scholar]
- Järnström, H; Saarela, K; Kalliokoski, P; Pasanen, AL. Comparison of VOC and ammonia emissions from individual PVC materials, adhesives and from complete structures. Environ. Int 2008, 34, 420–427. [Google Scholar]
- Bornehag, CG; Sundell, J; Weschler, CJ; Sigsgaard, T; Lundgren, B; Hasselgren, M; Hagerhed-Engman, L. The association between asthma and allergic symptoms in children and phthalates in house dust: a nested case-control study. Environ. Health Perspect 2004, 112, 1393–1397. [Google Scholar]
- Klenø, JG; Wolkoff, P; Clausen, PA; Wilkins, CK; Petersen, T. Degradation of the adsorbent Tenax TA by nitrogen oxides, ozone, hydrogen peroxide, OH radical and limonene oxidation products. Environ. Sci. Technol 2002, 36, 4121–4126. [Google Scholar]
- Wolkoff, P; Clausen, PA; Wilkins, CK; Nielsen, GD. Formation of strong airway irritants in terpene/ozone mixtures. Indoor Air 2000, 10, 82–91. [Google Scholar]
- Engman, L; Bornehag, CG; Sundell, J. How valid are parents’ questionnaire responses regarding building characteristics, mouldy odour, and signs of moisture problems in Swedish homes? Scand. J. Public Health 2007, 35, 125–132. [Google Scholar]
- Bornehag, CG; Sundell, J; Sigsgaard, T; Hagerhed-Engman, L. Association between ventilation rates in 390 Swedish homes and allergic symptoms in children: A nested case control study. Indoor Air 2005, 15, 275–280. [Google Scholar]
- Nordtest, Ventilation: Local mean age of air—Homogenous emission techniques. In Nordtest method NT VVS 118; Nordtest, Finland, 1997.
- Bornehag, CG; Sundell, J; Lundgren, B; Weschler, CJ; Sigsgaard, T; Hagerhed-Engman, L. Phthalates in indoor dust and their association with building characteristics. Environ. Health Perspect 2005, 113, 1399–1404. [Google Scholar]
- Wu, CH; Feng, CT; Lo, YS; Lin, TY; Lo, JG. Determination of volatile organic compounds in workplace air by multisorbent adsorption/thermal desorption-GC/MS. Chemosphere 2004, 56, 71–80. [Google Scholar]
- Helmig, D. Artifact free preparation, storage and analysis of solid adsorbent sampling cartridges used in the analysis of volatile Organic compounds in air. J. Chromatogr. A 1996, 732, 414–417. [Google Scholar]
- Uhde, E. Application of solid sorbents for the sampling of volatile organic compounds in indoor air. In Organic Indoor Air Pollutants: Occurrence-Measurement-Evaluation; Wilhelm-Klauditz- Institut (WKl), Fraunhofer-Institut für Holzforschung: Braunschweig, Germany, 1999; Volume Chapter 1. [Google Scholar]
- Edwards, RD; Jurvelina, J; Saarela, K; Jantunena, M. VOC concentrations measured in personal samples and residential indoor, outdoor and workplace microenvironments in EXPOLISHelsinki, Finland. Atmos. Environ 2001, 35, 4531–4543. [Google Scholar]
- Chou, MS; Huang, BJ; Chang, HY. Degradation of gas-phase propylene glycol monomethyl ether acetate by ultraviolet/ozone process: A kinetic study. J. Air Waste Manage. Assoc 2006, 56, 767–776. [Google Scholar]
- Zhang, G; Spickett, J; Lee, AH; Rumchev, K; Stick, S. Household hygiene practices in relation to dampness at home and current wheezing and rhino-conjunctivitis among school age children. Pediatr. Allergy Immunol 2005, 16, 587–592. [Google Scholar]
- Bornehag, CG; Sundell, J; Hägerhed-Engman, L; Sigsgaard, T. Association between ventilation rates in 390 Swedish homes and allergic symptoms in children. Indoor Air 2005, 15, 275–280. [Google Scholar]
- Clausen, PA; Lindeberg Bille, RL; Nilsson, T; Hansen, V; Svensmark, B; Bowadt, S. Simultaneous extraction of di(2-ethylhexyl) phthalate and nonionic surfactants from house dust. Concentrations in floor dust from 15 Danish schools. J. Chromatogr A 2003, 986, 179–190. [Google Scholar]
- Bayer, CW; Black, MS; Galloway, LM. Sampling and analysis techniques for trace volatile organic emissions from consumer products. J. Chromatogr. Sci 1988, 26, 168–173. [Google Scholar]
- Kuntasal, OO; Karman, D; Wang, D; Tuncel, SG; Tuncel, G. Determination of volatile organic compounds in different microenvironments by multibed adsorption and short-path thermal desorption followed by gas chromatographic-mass spectrometric analysis. J. Chromatogr. A 2005, 1099, 43–54. [Google Scholar]
- Edwards, RD; Jurvelin, J; Saarela, K; Jantunen, M. VOC concentrations measured in personal samples and residential indoor, outdoor and workplace microenvironments in EXPOLIS-Helsinki, Finland. Atmos. Environ 2001, 35, 4531–4543. [Google Scholar]
| N | Mean | S.D. | Min | 25th | 50th | 75th | Max | |
|---|---|---|---|---|---|---|---|---|
| Percentiles | ||||||||
| Propylene glycol and glycol ethers (CAS #) | ||||||||
| 1,2-propanediol (propylene glycol) (CAS # 57-55-6) | 165 | 8.20 | 8.17 | 0.51 | 2.84 | 5.54 | 10.25 | 48.62 |
| 1-methoxy-2-propanol (α-isomer of Propylene Glycol Monomethyl Ether) (CAS # 107-98-2) | 86 | 4.51 | 3.08 | 0.73 | 2.32 | 3.51 | 6.00 | 15.68 |
| 2-(2-butoxyethoxy)ethanol (CAS # 112-34-5) | 69 | 4.73 | 4.90 | 0.46 | 1.95 | 2.87 | 6.04 | 30.33 |
| 1-butoxy-2-propanol (CAS # 5131-66-8) | 65 | 7.15 | 8.87 | 0.60 | 2.21 | 4.03 | 8.08 | 53.04 |
| 2-(2-butoxyethoxy)ethanol acetate (CAS # 124-17-4) | 33 | 3.30 | 3.05 | 0.53 | 1.62 | 2.32 | 3.57 | 13.26 |
| 2-butoxy ethanol (CAS # 111-76-2) | 27 | 4.74 | 5.67 | 0.78 | 1.73 | 2.36 | 4.84 | 24.39 |
| 2-(2-(2-butoxyethoxy)ethoxy) ethanol (CAS # 143-22-6) | 20 | 2.10 | 1.80 | 0.65 | 0.96 | 1.68 | 2.60 | 8.74 |
| 2-(2-ethoxyethoxy) ethanol (cas # 111-90-0) | 16 | 9.72 | 9.39 | 2.26 | 4.38 | 5.73 | 11.59 | 36.86 |
| 1-(2-methoxypropoxy)-2-propanol (CAS # 13429-07-7) | 11 | 6.63 | 5.74 | 1.39 | 2.73 | 3.99 | 13.91 | 16.15 |
| Dipropylene glycol methyl ether (CAS # 34590-94-8) | 7 | 4.43 | 2.52 | 1.83 | 2.55 | 3.74 | 6.35 | 9.16 |
| 2-(2-methoxyethoxy) ethanol (CAS # 111-77-3) | 6 | 6.28 | 4.47 | 3.18 | 3.71 | 4.46 | 8.72 | 15.08 |
| 2-(2-hydroxypropoxy)-1-propanol (CAS # 106-62-7) | 4 | 1.42 | 0.74 | 0.62 | 0.74 | 1.37 | 2.17 | 2.34 |
| 1-(2-methoxy-1-methylethoxy)-2-propanol (CAS # 20324-32-7) | 3 | 6.66 | 2.48 | 4.13 | 4.13 | 6.76 | 9.08 | 9.08 |
| 1-propoxy-2-propanol (CAS # 1569-01-3) | 2 | 5.23 | 5.20 | 1.55 | 1.55 | 5.23 | 8.91 | 8.91 |
| 2-(2-ethoxyethoxy) ethanol acetate (CAS # 112-15-2) | 2 | 8.29 | 2.90 | 6.24 | 6.24 | 8.29 | 10.34 | 10.34 |
| 2,2-oxybis ethanol (Diethylene glycol) (CAS # 111-46-6) | 1 | 7.97 | 7.97 | 7.97 | 7.97 | 7.97 | 7.97 | |
| Ethanediol (Ethylene glycol) (CAS # 107-21-1) | 1 | 1.92 | 1.92 | 1.92 | 1.92 | 1.92 | 1.92 | |
| Terpene hydrocarbons | ||||||||
| γ–Terpinene (CAS # 99-85-4) | 3 | 2.82 | 0.44 | 2.38 | 2.38 | 2.82 | 3.26 | 3.26 |
| iso–Terpinolene (CAS # 586-62-9) | 7 | 3.86 | 1.62 | 2.10 | 2.53 | 3.30 | 5.75 | 6.08 |
| α–Terpinene (CAS # 99-86-5) | 1 | 25.21 | 25.21 | 25.21 | 25.21 | 25.21 | 25.21 | |
| α–Pinene (CAS # 80-56-8) | 239 | 20.76 | 16.40 | 1.93 | 8.47 | 16.27 | 28.58 | 97.53 |
| Limonene (CAS # 5989-27-5) | 383 | 17.78 | 14.50 | 1.36 | 7.84 | 13.61 | 23.19 | 92.99 |
| β–Pinene (CAS # 127-91-3) | 35 | 4.41 | 2.58 | 0.62 | 2.77 | 3.85 | 5.18 | 13.21 |
| Camphene (CAS # 79-92-5) | 155 | 5.62 | 4.24 | 0.71 | 2.82 | 4.59 | 7.07 | 27.16 |
| β–Phellandrene (CAS # 555-10-2) | 14 | 5.81 | 4.34 | 1.39 | 2.37 | 4.97 | 8.08 | 16.35 |
| Texanol® | ||||||||
| Texanol A | 39 | 9.95 | 6.56 | 1.20 | 5.63 | 7.58 | 14.33 | 27.89 |
| Texanol B | 88 | 5.60 | 7.13 | 0.47 | 1.72 | 2.76 | 6.55 | 42.48 |
| Phthalate compounds in dust samplea) | ||||||||
| n-butyl benzyl phthalate (BBzP) | 296 | 0.38 | 2.64 | 0.01 | 0.09 | 0.16 | 0.30 | 45.55 |
| di(2-ethylhexyl)phthalate (DEHP) | 351 | 1.31 | 2.59 | 0.02 | 0.46 | 0.77 | 1.31 | 40.46 |
| PGEs | Terpenes | Texanols | BBzP a) b) | DEHP a) b) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| nb) | GM ± GSD (μg/m3) | nb) | GM ± GSD (μg/m3) | nb) | GM ± GSD (μg/m3) | n | GM ± GSD (μg/m3) | n | GM ± GSD (μg/m3) | |
| Suspected Sources | ||||||||||
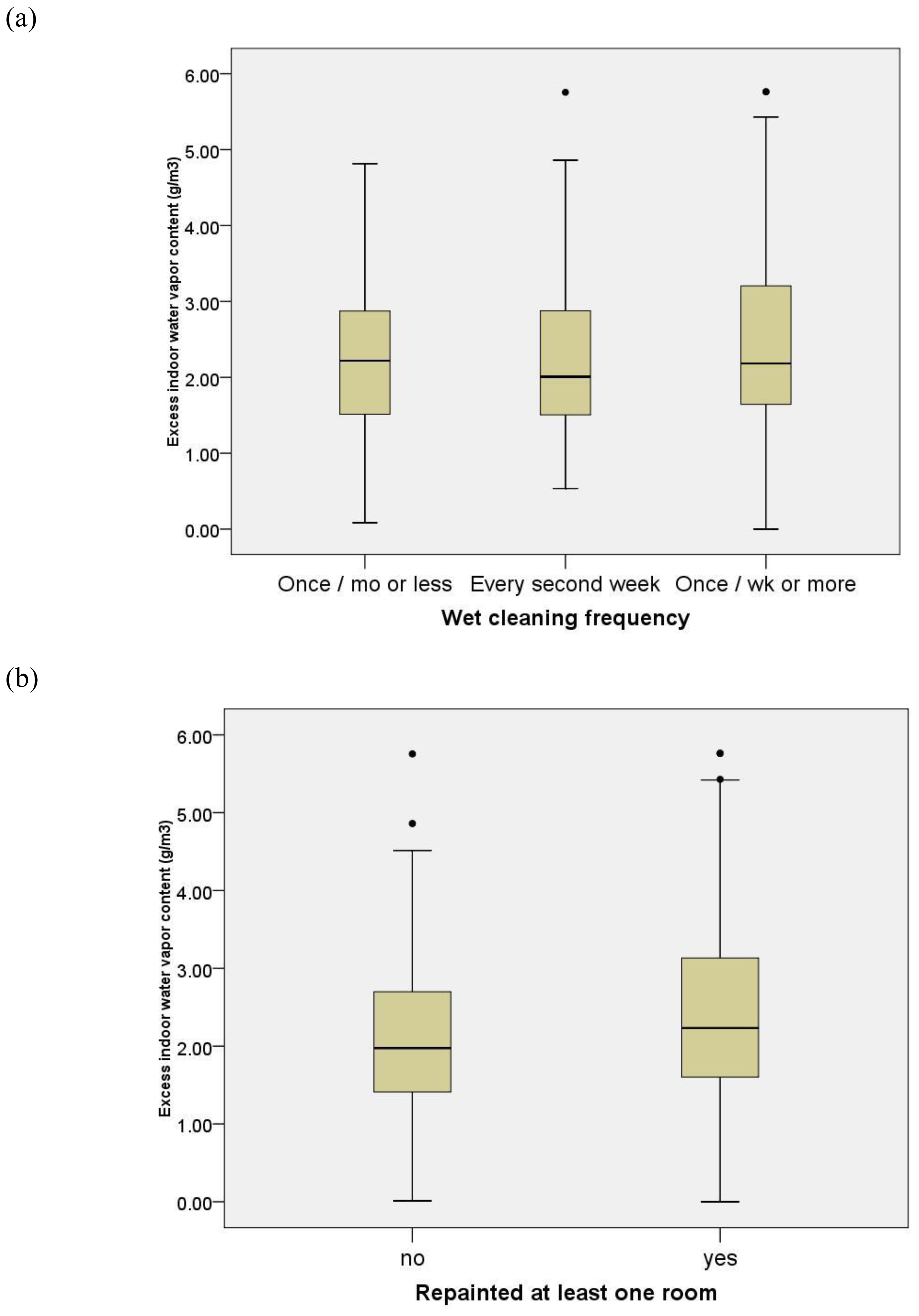
| Water-based cleaning frequency | ||||||||||
| P-for-linear-trend | 0.046 | 0.800 | 0.522 | 0.048 | 0.026 | |||||
| ≥ Once / week | 121 | 5.40 ± 3.42 c) | 121 | 21.14 ± 2.77 | 30 | 4.16 ± 3.79 | 95 | 0.18 ± 2.77 | 109 | 0.86 ± 2.68 |
| Every other week | 99 | 3.94 ± 3.51 | 99 | 24.25 ± 2.87 | 26 | 3.91 ± 2.46 | 72 | 0.17 ± 2.57 | 85 | 0.88 ± 2.72 |
| ≤ Once / month | 165 | 3.13 ± 3.16 | 165 | 23.09 ± 2.34 | 38 | 4.71 ± 3.25 | 125 | 0.14 ± 2.42 | 151 | 0.68 ± 2.48 |
| At least one room was repainted prior to/following the child’s birth | ||||||||||
| P-for-ANOVA | 0.014 | 0.320 | 0.851 | 0.292 | 0.457 | |||||
| Yes | 247 | 4.28 ± 3.47 | 247 | 23.07 ± 2.62 | 66 | 4.64 ± 3.15 | 190 | 0.17 ± 2.67 | 224 | 0.80 ± 2.58 |
| No | 143 | 3.37 ± 3.18 | 143 | 21.49 ± 2.68 | 29 | 3.70 ± 3.18 | 106 | 0.15 ± 2.40 | 127 | 0.73 ± 2.68 |
| Flooring material, child’s bedroom | ||||||||||
| P-for-ANOVA | 0.110 | 0.382 | 0.178 | 0.000 | 0.002 | |||||
| Linoleum | 13 | 4.44 ± 3.89 | 13 | 22.77 ± 1.95 | 13 | 1.61 ± 1.03 | 12 | 0.12 ± 3.39 | 13 | 0.54 ± 2.98 |
| PVC | 211 | 4.42 ± 3.35 | 211 | 22.21 ± 2.64 | 211 | 4.25 ± 3.11 | 171 | 0.20 ± 2.46 | 188 | 0.95 ± 2.59 |
| Wood | 120 | 3.36 ± 3.43 | 120 | 24.44 ± 2.57 | 120 | 5.62 ± 3.40 | 82 | 0.11 ± 2.53 | 106 | 0.60 ± 2.42 |
| Laminate | 39 | 3.81 ± 3.11 | 39 | 21.25 ± 2.87 | 39 | 2.13 ± 1.71 | 25 | 0.16 ± 2.31 | 37 | 0.65 ± 2.81 |
| Other d) | 7 | 1.32 ± 1.89 | 7 | 10.26 ± 3.41 | 7 | 6 | 0.13 ± 1.40 | 7 | 0.79 ± 1.75 | |
| Age of surface materials in the child’s bedroom | ||||||||||
| P-for-linear-trend | 0.049 | 0.556 | 0.073 | 0.727 | 0.027 | |||||
| Very old | 19 | 3.15 ± 3.23 | 19 | 21.82 ± 2.02 | 5 | 1.22 ± 2.16 | 15 | 0.17 ± 2.90 | 18 | 0.99 ± 1.98 |
| Old | 76 | 3.51 ± 3.31 | 76 | 26.04 ± 2.39 | 16 | 3.07 ± 3.22 | 56 | 0.16 ± 2.87 | 70 | 0.88 ± 2.92 |
| Mixture of old/new | 136 | 3.77 ± 3.35 | 136 | 20.69 ± 3.02 | 33 | 6.28 ± 2.77 | 104 | 0.15 ± 2.19 | 122 | 0.74 ± 2.47 |
| Overall new | 112 | 3.65 ± 3.31 | 112 | 21.96 ± 2.52 | 31 | 3.95 ± 3.02 | 90 | 0.17 ± 2.91 | 101 | 0.76 ± 2.72 |
| Newest | 38 | 6.42 ± 3.54 | 38 | 22.65 ± 2.54 | 7 | 5.60 ± 4.31 | 27 | 0.15 ± 2.11 | 33 | 0.57 ± 2.13 |
| Building type | ||||||||||
| P-for-ANOVA | 0.805 | 0.706 | 0.096 | 0.308 | 0.509 | |||||
| Single family house | 321 | 3.85 ± 3.31 | 321 | 23.16 ± 2.64 | 83 | 4.65 ± 3.17 | 243 | 0.16 ± 2.56 | 291 | 0.75 ± 2.57 |
| Two family house | 22 | 4.65 ± 4.27 | 22 | 21.52 ± 2.08 | 3 | 7.80 ± 4.20 | 16 | 0.15 ± 2.14 | 19 | 0.81 ± 2.43 |
| Multifamily house | 44 | 3.94 ± 3.50 | 44 | 18.69 ± 2.96 | 8 | 1.83 ± 2.01 | 34 | 0.15 ± 2.94 | 39 | 0.94 ± 3.09 |
| Another | 3 | 6.35 ± 4.73 | 3 | 18.76 ± 1.53 | 1 | 2.35 ± -- | 3 | 0.43 ± 1.38 | 2 | 0.49 ± 1.34 |
| Secondhand smoke | ||||||||||
| P-for-ANOVA | 0.755 | 0.394 | 0.355 | 0.421 | 0.632 | |||||
| Yes | 67 | 4.07 ± 3.16 | 67 | 20.34 ± 3.19 | 17 | 3.16 ± 3.09 | 55 | 0.17 ± 2.54 | 63 | 0.82 ± 2.58 |
| No | 313 | 3.87 ± 3.40 | 313 | 22.73 ± 2.52 | 75 | 4.59 ± 3.20 | 235 | 0.16 ± 2.60 | 280 | 0.77 ± 2.63 |
| Suspected Modifiers | ||||||||||
| Quartiles of excess indoor water vapor content over outdoor content (g/m3) | ||||||||||
| P-for-linear-trend | <0.001 | 0.137 | 0.031 | 0.007 | 0.165 | |||||
| < 1.539 | 84 | 2.98 ± 3.18 | 84 | 21.28 ± 2.45 | 20 | 2.52 ± 2.77 | 65 | 0.12 ± 2.19 | 80 | 0.68 ± 2.61 |
| 1.539 - 2.156 | 84 | 4.04 ± 3.43 | 84 | 20.60 ± 2.75 | 18 | 5.09 ± 3.08 | 67 | 0.17 ± 2.17 | 82 | 0.78 ± 2.21 |
| 2.157 - 2.947 | 84 | 3.11 ± 3.30 | 84 | 18.92 ± 2.77 | 14 | 4.13 ± 4.06 | 59 | 0.16 ± 2.53 | 69 | 0.72 ± 2.91 |
| ≥ 2.948 | 84 | 6.51 ± 3.24 c) | 84 | 27.58 ± 2.39 | 30 | 5.79 ± 3.05 | 66 | 0.20 ± 3.45 | 75 | 0.88 ± 2.82 |
| Quartiles of Temperature (°C) | ||||||||||
| P-for-linear-trend | 0.003 | 0.279 | 0.323 | 0.064 | 0.589 | |||||
| < 20.18 | 96 | 2.75 ± 2.90 | 96 | 19.95 ± 2.72 | 20 | 5.67 ± 2.53 | 71 | 0.13 ± 2.37 | 87 | 0.72 ± 2.72 |
| 20.18 - 20.96 | 98 | 4.86 ± 3.27 | 98 | 24.26 ± 2.45 | 24 | 4.21 ± 3.77 | 75 | 0.16 ± 2.36 | 87 | 0.84 ± 2.72 |
| 20.97 - 21.67 | 97 | 3.32 ± 3.36 | 97 | 21.10 ± 2.80 | 25 | 4.06 ± 3.46 | 71 | 0.17 ± 3.13 | 84 | 0.70 ± 2.69 |
| ≥ 21.68 | 98 | 5.32 ± 3.64 c) | 98 | 24.50 ± 2.56 | 25 | 3.92 ± 2.95 | 78 | 0.18 ± 2.47 | 92 | 0.83 ± 2.38 |
| Quartiles of ventilation rate, child’s bedroom (air change / hr) | ||||||||||
| P-for-linear-trend | 0.198 | 0.372 | 0.111 | 0.125 | 0.006 | |||||
| < 0.220 | 94 | 4.64 ± 3.52 | 94 | 25.19 ± 2.49 | 24 | 6.28 ± 2.44 | 71 | 0.15 ± 2.25 | 82 | 0.69 ± 2.57 |
| 0.220 - 0.315 | 93 | 4.09 ± 3.48 | 93 | 22.16 ± 2.73 | 27 | 3.90 ± 3.05 | 76 | 0.14 ± 3.10 | 90 | 0.61 ± 2.78 |
| 0.316 - 0.435 | 94 | 3.65 ± 3.16 | 94 | 21.89 ± 2.42 | 22 | 4.23 ± 4.23 | 74 | 0.18 ± 2.53 | 86 | 0.85 ± 2.52 |
| ≥ 0.436 | 97 | 3.78 ± 3.40 | 97 | 22.19 ± 2.73 | 18 | 3.33 ± 3.24 | 69 | 0.17 ± 2.43 | 85 | 0.95 ± 2.44 |
| Type of ventilation system at home | ||||||||||
| P-for-ANOVA | 0.552 | 0.840 | 0.015 | 0.175 | 0.572 | |||||
| Natural, and do not use a kitchen fan | 20 | 2.81 ± 3.55 | 20 | 23.44 ± 1.83 | 8 | 3.43 ± 3.04 | 15 | 0.19 ± 3.02 | 18 | 0.75 ± 4.08 |
| Natural and use a kitchen fan | 229 | 4.29 ± 3.37 | 229 | 22.75 ± 2.85 | 62 | 5.09 ± 3.15 | 171 | 0.16 ± 2.57 | 211 | 0.76 ± 2.63 |
| Exhaust air system | 92 | 3.95 ± 3.62 | 92 | 20.80 ± 2.52 | 19 | 3.79 ± 3.03 | 73 | 0.17 ± 2.53 | 79 | 0.86 ± 2.49 |
| Exhaust & supply air system | 12 | 3.46 ± 3.02 | 12 | 15.91 ± 3.29 | 4 | 1.35 ± 1.61 | 9 | 0.16 ± 2.54 | 10 | 0.88 ± 1.93 |
| Exhaust & supply with heat recovery | 25 | 3.39 ± 2.82 | 25 | 27.41 ± 1.86 | 1 | 0.63 ± -- | 18 | 0.15 ± 2.63 | 21 | 0.74 ± 2.19 |
| Other | 7 | 1.76 ± 2.24 | 7 | 22.65 ± 1.73 | 7 | 0.08 ± 2.43 | 7 | 0.39 ± 2.52 | ||
| Water damage since spring 2000 | ||||||||||
| P-for-ANOVA | 0.915 | 0.813 | 0.110 | 0.764 | 0.625 | |||||
| Yes | 42 | 4.00 ± 3.77 | 42 | 21.94 ± 3.40 | 5 | 1.95 ± 2.65 | 33 | 0.15 ± 2.56 | 40 | 0.82 ± 3.15 |
| No | 344 | 3.91 ± 3.34 | 344 | 22.78 ± 2.55 | 89 | 4.56 ± 3.16 | 259 | 0.16 ± 2.60 | 306 | 0.76 ± 2.56 |
| Building Inspector Rating | ||||||||||
| First impression of stuffy or unventilated air | ||||||||||
| P-for-ANOVA | 0.027 | 0.029 | 0.925 | 0.207 | 0.367 | |||||
| Obvious | 33 | 6.76 ± 3.81 | 33 | 31.77 ± 2.14 | 9 | 8.22 ± 3.29 | 26 | 0.20 ± 2.09 | 29 | 1.02 ± 2.00 |
| Weak | 85 | 3.79 ± 3.41 | 85 | 19.85 ± 3.08 | 18 | 8.65 ± 3.97 | 64 | 0.15 ± 2.79 | 75 | 0.85 ± 2.74 |
| P-for-ANOVA | 0.027 | 0.029 | 0.925 | 0.207 | 0.367 | |||||
| Obvious | 33 | 6.76 ± 3.81 | 33 | 31.77 ± 2.14 | 9 | 8.22 ± 3.29 | 26 | 0.20 ± 2.09 | 29 | 1.02 ± 2.00 |
| Weak | 85 | 3.79 ± 3.41 | 85 | 19.85 ± 3.08 | 18 | 8.65 ± 3.97 | 64 | 0.15 ± 2.79 | 75 | 0.85 ± 2.74 |
| Stuffy, earthy, or microbial smell | ||||||||||
| P-for-ANOVA | 0.954 | 0.688 | 0.111 | 0.640 | 0.436 | |||||
| Obvious | 39 | 3.36 ± 3.21 | 39 | 25.38 ± 2.67 | 8 | 2.20 ± 2.56 | 30 | 0.15 ± 2.58 | 38 | 0.85 ± 2.85 |
| Weak | 47 | 3.31 ± 3.51 | 47 | 23.20 ± 2.92 | 11 | 5.80 ± 4.10 | 35 | 0.14 ± 2.48 | 42 | 0.70 ± 3.05 |
| Chemical smell | ||||||||||
| P-for-ANOVA | 0.451 | 0.950 | 0.137 | 0.865 | 0.429 | |||||
| Obvious | 13 | 2.47 ± 2.26 | 13 | 26.86 ± 3.53 | 1 | 3.84 ± -- | 7 | 0.14 ± 3.25 | 12 | 0.60 ± 4.13 |
| Weak | 29 | 3.41 ± 4.13 | 29 | 27.57 ± 3.44 | 3 | 22.33 ± 1.88 | 26 | 0.13 ± 2.57 | 27 | 0.84 ± 2.91 |
| PGEs | Terpenes | Texanols | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| β | (95 % CI) | P | β | (95 % CI) | P | β | (95 % CI) | P | |||||
| Excess humidity | |||||||||||||
| ≤2.157 g/m3 | y-intercept | −1.567 | −4.507 | 1.373 | 0.294 | 3.977 | 1.707 | 6.247 | 0.001 | 5.667 | 0.083 | −0.776 | 12.110 |
| Wet-clean once/week | 0.477 | 0.040 | 0.915 | 0.033 | −0.247 | −0.585 | 0.091 | 0.151 | −0.417 | 0.397 | −1.408 | 0.574 | |
| Wet-clean every other week | 0.581 | 0.131 | 1.031 | 0.012 | 0.406 | 0.059 | 0.753 | 0.022 | −0.420 | 0.443 | −1.525 | 0.684 | |
| Repainted ≥ one room | 0.005 | −0.367 | 0.376 | 0.980 | 0.074 | −0.213 | 0.361 | 0.610 | −0.357 | 0.429 | −1.268 | 0.554 | |
| “Newest” surface material | 0.423 | −0.195 | 1.042 | 0.178 | 0.148 | −0.330 | 0.625 | 0.543 | 0.155 | 0.791 | −1.029 | 1.338 | |
| Temperature (quartile unit) | 0.125 | −0.017 | 0.267 | 0.084 | −0.049 | −0.159 | 0.061 | 0.380 | −0.170 | 0.255 | −0.470 | 0.129 | |
| Ventilation rate in the child‘s bedroom (quartile unit) | −0.054 | −0.222 | 0.113 | 0.523 | 0.004 | −0.126 | 0.133 | 0.955 | −0.149 | 0.432 | −0.532 | 0.234 | |
| ≥2.158 g/m3 | y-intercept | −2.824 | −6.290 | 0.642 | 0.110 | 0.398 | −2.421 | 3.217 | 0.781 | 3.496 | 0.362 | −4.189 | 11.181 |
| Wet-clean once/week | 0.519 | 0.100 | 0.938 | 0.015 | 0.104 | −0.236 | 0.444 | 0.547 | 0.246 | 0.594 | −0.682 | 1.174 | |
| Wet-clean every other week | 0.217 | −0.260 | 0.694 | 0.370 | −0.138 | −0.526 | 0.249 | 0.482 | −0.285 | 0.547 | −1.235 | 0.665 | |
| Repainted ≥ one room | 0.630 | 0.243 | 1.017 | 0.002 | 0.100 | −0.215 | 0.415 | 0.532 | 0.016 | 0.971 | −0.847 | 0.878 | |
| “Newest” surface material | 0.409 | −0.195 | 1.013 | 0.183 | −0.098 | −0.589 | 0.393 | 0.695 | 2.386 | 0.089 | −0.385 | 5.157 | |
| Temperature (quartile unit) | 0.185 | 0.020 | 0.350 | 0.028 | 0.127 | −0.008 | 0.261 | 0.065 | −0.064 | 0.725 | −0.431 | 0.302 | |
| Ventilation rate in the child‘s bedroom (quartile unit) | −0.081 | −0.255 | 0.093 | 0.358 | 0.026 | −0.116 | 0.167 | 0.718 | −0.257 | 0.201 | −0.658 | 0.143 | |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Choi, H.; Schmidbauer, N.; Spengler, J.; Bornehag, C.-G. Sources of Propylene Glycol and Glycol Ethers in Air at Home. Int. J. Environ. Res. Public Health 2010, 7, 4213-4237. https://doi.org/10.3390/ijerph7124213
Choi H, Schmidbauer N, Spengler J, Bornehag C-G. Sources of Propylene Glycol and Glycol Ethers in Air at Home. International Journal of Environmental Research and Public Health. 2010; 7(12):4213-4237. https://doi.org/10.3390/ijerph7124213
Chicago/Turabian StyleChoi, Hyunok, Norbert Schmidbauer, John Spengler, and Carl-Gustaf Bornehag. 2010. "Sources of Propylene Glycol and Glycol Ethers in Air at Home" International Journal of Environmental Research and Public Health 7, no. 12: 4213-4237. https://doi.org/10.3390/ijerph7124213
APA StyleChoi, H., Schmidbauer, N., Spengler, J., & Bornehag, C.-G. (2010). Sources of Propylene Glycol and Glycol Ethers in Air at Home. International Journal of Environmental Research and Public Health, 7(12), 4213-4237. https://doi.org/10.3390/ijerph7124213




