Fish, Mercury, Selenium and Cardiovascular Risk: Current Evidence and Unanswered Questions
Abstract
:1. Introduction
2. Fish and Cardiovascular Risk
3. Mercury and Cardiovascular Risk
4. Selenium and Cardiovascular Risk
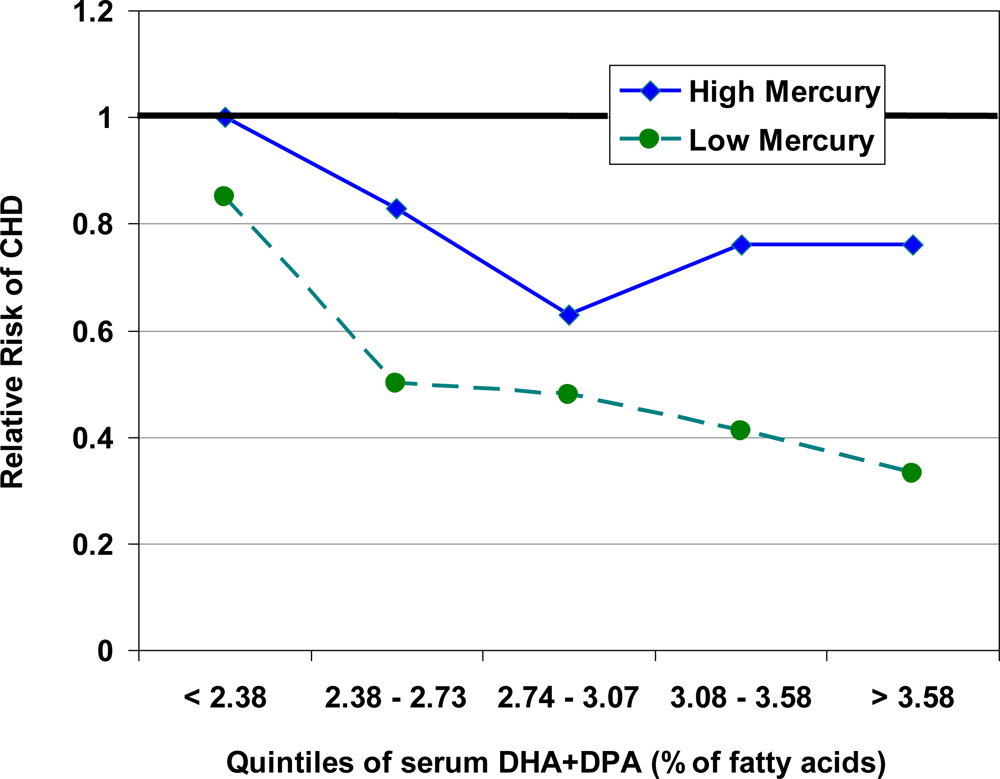
5. Interaction between Mercury, Selenium and Cardiovascular Risk
6. Polychlorinated Biphenyls (PCBs) and Dioxins
7. Conclusions
Acknowledgments
- Disclosures: Research grants (significant) from NHLBI and NIEHS (K08 HL 075628-01, R01 HL 085710-01, R01 ES 014433-01A2); the Searle Scholar Award from the Searle Funds at The Chicago Community Trust; the Genes and Environment Initiative at the Harvard School of Public Health; the Gates Foundation/World Health Organization Global Burden of Diseases, Injuries, and Risk Factors Study; and GSK, Sigma Tau, and Pronova for an investigator-initiated trial of fish oil to prevent post-surgical arrhythmia. Honoraria (modest) for speaking at scientific conferences and reviewing on topics related to diet and cardiovascular disease, including from the U.S. Food and Drug Administration, United Nations, World Health Organization, UpToDate, International Life Sciences Institute, Aramark, and several universities and scientific organizations. No ownership, patents, stocks, advisory board membership, nor speaking board membership.
References
- Mozaffarian, D; Rimm, EB. Fish intake, contaminants, and human health: evaluating the risks and the benefits. JAMA 2006, 296, 1885–1899. [Google Scholar]
- Mahaffey, KR. Methylmercury: a new look at the risks. Public Health Rep 1999, 114, 396–399. [Google Scholar]
- Committee on the Toxicological Effects of Methylmercury; Board on Environmental Studies and Toxicology; Commission on Life Sciences; National Research Council. Toxicological Effects of Methylmercury; National Academy Press: Washington, DC, USA, 2000. [Google Scholar]
- Rice, DC; Schoeny, R; Mahaffey, K. Methods and rationale for derivation of a reference dose for methylmercury by the U.S. EPA. Risk Anal 2003, 23, 107–115. [Google Scholar]
- Rice, DC. The US EPA reference dose for methylmercury: sources of uncertainty. Environ. Res 2004, 95, 406–413. [Google Scholar]
- Chan, HM; Egeland, GM. Fish consumption, mercury exposure, and heart diseases. Nutr. Rev 2004, 62, 68–72. [Google Scholar]
- U.S. Environmental Protection Agency. Mercury Study Report to Congress.
- Gochfeld, M. Cases of mercury exposure, bioavailability, and absorption. Ecotoxicol. Environ. Saf 2003, 56, 174–179. [Google Scholar]
- Risher, JF. Too much of a good thing (fish): methylmercury case study. J Environ Health 2004, 67. [Google Scholar]
- Mozaffarian, D. Does alpha-linolenic acid intake reduce the risk of coronary heart disease? A review of the evidence. Altern. Ther. Health Med 2005, 11, 24–30. [Google Scholar]
- Institute of Medicine of the National Acadamies. Seafood Choices: Balancing Benefits and Risks. 2006.
- U.S. Department of Health and Human Services; U.S. Food and Drug Administration. What You Need to Know About Mercury in Fish and Shellfish: 2004 EPA and FDA Advice for women who might become pregnant, women who are pregnant, nursing mothers, young children.
- Leaf, A; Kang, JX; Xiao, YF; Billman, GE. Clinical prevention of sudden cardiac death by n-3 polyunsaturated fatty acids and mechanism of prevention of arrhythmias by n-3 fish oils. Circulation 2003, 107, 2646–2652. [Google Scholar]
- McLennan, PL. Myocardial membrane fatty acids and the antiarrhythmic actions of dietary fish oil in animal models. Lipids 2001, 36, S111–114. [Google Scholar]
- Charnock, JS; McLennan, PL; Abeywardena, MY. Dietary modulation of lipid metabolism and mechanical performance of the heart. Mol. Cell Biochem 1992, 116, 19–25. [Google Scholar]
- Kenny, D; Warltier, DC; Pleuss, JA; Hoffmann, RG; Goodfriend, TL; Egan, BM. Effect of omega-3 fatty acids on the vascular response to angiotensin in normotensive men. Am. J. Cardiol 1992, 70, 1347–1352. [Google Scholar]
- Chin, JP; Gust, AP; Nestel, PJ; Dart, AM. Marine oils dose-dependently inhibit vasoconstriction of forearm resistance vessels in humans. Hypertension 1993, 21, 22–28. [Google Scholar]
- Geleijnse, JM; Giltay, EJ; Grobbee, DE; Donders, AR; Kok, FJ. Blood pressure response to fish oil supplementation: metaregression analysis of randomized trials. J. Hypertens 2002, 20, 1493–1499. [Google Scholar]
- Mozaffarian, D; Geelen, A; Brouwer, IA; Geleijnse, JM; Zock, PL; Katan, MB. Effect of fish oil on heart rate in humans: A meta-analysis of randomized controlled trials. Circulation 2005, 112, 1945–1952. [Google Scholar]
- Mozaffarian, D; Gottdiener, JS; Siscovick, DS. Intake of tuna or other broiled or baked fish vs. fried fish and cardiac structure, function, and hemodynamics. Am. J. Cardiol 2006, 97, 216–222. [Google Scholar]
- Nestel, PJ. Fish oil and cardiovascular disease: lipids and arterial function. Am. J. Clin. Nutr 2000, 71, 228S–231S. [Google Scholar]
- Christensen, JH. n-3 fatty acids and the risk of sudden cardiac death. Emphasis on heart rate variability. Dan. Med. Bull 2003, 50, 347–367. [Google Scholar]
- Mori, TA; Beilin, LJ. Omega-3 fatty acids and inflammation. Curr. Atheroscler. Rep 2004, 6, 461–467. [Google Scholar]
- Kristensen, SD; Iversen, AM; Schmidt, EB. n-3 polyunsaturated fatty acids and coronary thrombosis. Lipids 2001, 36(Suppl), S79–82. [Google Scholar]
- Clandinin, MT; Cheema, S; Field, CJ; Garg, ML; Venkatraman, J; Clandinin, TR. Dietary fat: exogenous determination of membrane structure and cell function. Faseb. J 1991, 5, 2761–2769. [Google Scholar]
- Feller, SE; Gawrisch, K. Properties of docosahexaenoic-acid-containing lipids and their influence on the function of rhodopsin. Curr. Opin. Struct. Biol 2005, 15, 416–422. [Google Scholar]
- Vanden Heuvel, JP. Diet, fatty acids, and regulation of genes important for heart disease. Curr. Atheroscler. Rep 2004, 6, 432–440. [Google Scholar]
- Oomen, CM; Feskens, EJ; Rasanen, L; Fidanza, F; Nissinen, AM; Menotti, A; Kok, FJ; Kromhout, D. Fish consumption and coronary heart disease mortality in Finland, Italy, and The Netherlands. Am. J. Epidemiol 2000, 151, 999–1006. [Google Scholar]
- Mozaffarian, D; Lemaitre, RN; Kuller, LH; Burke, GL; Tracy, RP; Siscovick, DS. Cardiac benefits of fish consumption may depend on the type of fish meal consumed: the Cardiovascular Health Study. Circulation 2003, 107, 1372–1377. [Google Scholar]
- Harris, WS; Mozaffarian, D; Lefevre, M; Toner, CD; Colombo, J; Cunnane, SC; Holden, JM; Klurfeld, DM; Morris, MC; Whelan, J. Towards establishing dietary reference intakes for eicosapentaenoic and docosahexaenoic acids. J. Nutr 2009, 139, 804S–819S. [Google Scholar]
- Sun, Q; Ma, J; Campos, H; Rexrode, KM; Albert, CM; Mozaffarian, D; Hu, FB. Blood concentrations of individual long-chain n-3 fatty acids and risk of nonfatal myocardial infarction. Am. J. Clin. Nutr 2008, 88, 216–223. [Google Scholar]
- He, K; Song, Y; Daviglus, ML; Liu, K; Van Horn, L; Dyer, AR; Goldbourt, U; Greenland, P. Fish consumption and incidence of stroke: a meta-analysis of cohort studies. Stroke 2004, 35, 1538–1542. [Google Scholar]
- Mozaffarian, D; Psaty, BM; Rimm, EB; Lemaitre, RN; Burke, GL; Lyles, MF; Lefkowitz, D; Siscovick, DS. Fish intake and risk of incident atrial fibrillation. Circulation 2004, 110, 368–373. [Google Scholar]
- Morris, MC; Evans, DA; Tangney, CC; Bienias, JL; Wilson, RS. Fish consumption and cognitive decline with age in a large community study. Arch. Neurol 2005, 62, 1849–1853. [Google Scholar]
- Lin, PY; Su, KP. A meta-analytic review of double-blind, placebo-controlled trials of antidepressant efficacy of omega-3 fatty acids. J. Clin. Psychiatry 2007, 68, 1056–1061. [Google Scholar]
- U.S. Food and Drug Administration. Mercury in fish: Cause for concern?
- The Risk Assessment Information System. Toxicity Summary for Mercury.
- U.S. Geological Survey. Mercury in the Environment.
- Hightower, JM; Moore, D. Mercury levels in high-end consumers of fish. Environ. Health Perspect 2003, 111, 604–608. [Google Scholar]
- Joshi, A; Douglass, CW; Kim, HD; Joshipura, KJ; Park, MC; Rimm, EB; Carino, MJ; Garcia, RI; Morris, JS; Willett, WC. The relationship between amalgam restorations and mercury levels in male dentists and nondental health professionals. J. Public Health Dent 2003, 63, 52–60. [Google Scholar]
- MacIntosh, DL; Williams, PL; Hunter, DJ; Sampson, LA; Morris, SC; Willett, WC; Rimm, EB. Evaluation of a food frequency questionnaire-food composition approach for estimating dietary intake of inorganic arsenic and methylmercury. Cancer Epidemiol. Biomarkers Prev 1997, 6, 1043–1050. [Google Scholar]
- Garland, M; Morris, JS; Rosner, BA; Stampfer, MJ; Spate, VL; Baskett, CJ; Willett, WC; Hunter, DJ. Toenail trace element levels as biomarkers: reproducibility over a 6-year period. Cancer Epidemiol. Biomarkers Prev 1993, 2, 493–497. [Google Scholar]
- Rosner, B; Hennekens, CH; Kass, EH; Miall, WE. Age-specific correlation analysis of longitudinal blood pressure data. Am. J. Epidemiol 1977, 106, 306–313. [Google Scholar]
- Berglund, M; Lind, B; Bjornberg, KA; Palm, B; Einarsson, O; Vahter, M. Inter-individual variations of human mercury exposure biomarkers: a cross-sectional assessment. Environ. Health 2005, 4, 20. [Google Scholar]
- Lebel, J; Mergler, D; Branches, F; Lucotte, M; Amorim, M; Larribe, F; Dolbec, J. Neurotoxic effects of low-level methylmercury contamination in the Amazonian Basin. Environ. Res 1998, 79, 20–32. [Google Scholar]
- Yokoo, EM; Valente, JG; Grattan, L; Schmidt, SL; Platt, I; Silbergeld, EK. Low level methylmercury exposure affects neuropsychological function in adults. Environ. Health 2003, 2, 8. [Google Scholar]
- Auger, N; Kofman, O; Kosatsky, T; Armstrong, B. Low-level methylmercury exposure as a risk factor for neurologic abnormalities in adults. Neurotoxicology 2005, 26, 149–157. [Google Scholar]
- Weil, M; Bressler, J; Parsons, P; Bolla, K; Glass, T; Schwartz, B. Blood mercury levels and neurobehavioral function. JAMA 2005, 293, 1875–1882. [Google Scholar]
- Johansson, N; Basun, H; Winblad, B; Nordberg, M. Relationship between mercury concentration in blood, cognitive performance, and blood pressure, in an elderly urban population. Biometals 2002, 15, 189–195. [Google Scholar]
- Peet, M; Stokes, C. Omega-3 fatty acids in the treatment of psychiatric disorders. Drugs 2005, 65, 1051–1059. [Google Scholar]
- Young, G; Conquer, J. Omega-3 fatty acids and neuropsychiatric disorders. Reprod Nutr. Dev 2005, 45, 1–28. [Google Scholar]
- Wakita, Y. Hypertension induced by methyl mercury in rats. Toxicol. Appl. Pharmacol 1987, 89, 144–147. [Google Scholar]
- Kostka, B; Michalska, M; Krajewska, U; Wierzbicki, R. Blood coagulation changes in rats poisoned with methylmercuric chloride (MeHg). Pol. J. Pharmacol. Pharm 1989, 41, 183–189. [Google Scholar]
- Halbach, S. Mercury compounds: lipophilicity and toxic effects on isolated myocardial tissue. Arch. Toxicol 1990, 64, 315–319. [Google Scholar]
- Rungby, J; Ernst, E. Experimentally induced lipid peroxidation after exposure to chromium, mercury or silver: interactions with carbon tetrachloride. Pharmacol. Toxicol 1992, 70, 205–207. [Google Scholar]
- Jansson, G; Harms-Ringdahl, M. Stimulating effects of mercuric- and silver ions on the superoxide anion production in human polymorphonuclear leukocytes. Free Radic. Res. Commun 1993, 18, 87–98. [Google Scholar]
- Oliveira, EM; Vassallo, DV; Sarkis, JJ; Mill, JG. Mercury effects on the contractile activity of isolated heart muscle. Toxicol. Appl. Pharmacol 1994, 128, 86–91. [Google Scholar]
- InSug, O; Datar, S; Koch, CJ; Shapiro, IM; Shenker, BJ. Mercuric compounds inhibit human monocyte function by inducing apoptosis: evidence for formation of reactive oxygen species, development of mitochondrial membrane permeability transition and loss of reductive reserve. Toxicology 1997, 124, 211–224. [Google Scholar]
- da Cunha, V; Souza, HP; Rossoni, LV; Franca, AS; Vassallo, DV. Effects of mercury on the isolated perfused rat tail vascular bed are endothelium-dependent. Arch. Environ. Contam. Toxicol 2000, 39, 124–130. [Google Scholar]
- de Assis, GP; Silva, CE; Stefanon, I; Vassallo, DV. Effects of small concentrations of mercury on the contractile activity of the rat ventricular myocardium. Comp. Biochem. Physiol. C Toxicol. Pharmacol 2003, 134, 375–383. [Google Scholar]
- Moreira, CM; Oliveira, EM; Bonan, CD; Sarkis, JJ; Vassallo, DV. Effects of mercury on myosin ATPase in the ventricular myocardium of the rat. Comp. Biochem. Physiol. C Toxicol. Pharmacol 2003, 135C, 269–275. [Google Scholar]
- Sorensen, N; Murata, K; Budtz-Jorgensen, E; Weihe, P; Grandjean, P. Prenatal methylmercury exposure as a cardiovascular risk factor at seven years of age. Epidemiology 1999, 10, 370–375. [Google Scholar]
- Grandjean, P; Murata, K; Budtz-Jorgensen, E; Weihe, P. Cardiac autonomic activity in methylmercury neurotoxicity: 14-year follow-up of a Faroese birth cohort. J Pediatr 2004, 144, 169–176. [Google Scholar]
- Thurston, SW; Bovet, P; Myers, GJ; Davidson, PW; Georger, LA; Shamlaye, C; Clarkson, TW. Does prenatal methylmercury exposure from fish consumption affect blood pressure in childhood? Neurotoxicology 2007, 28, 924–930. [Google Scholar]
- Fillion, M; Mergler, D; Sousa Passos, CJ; Larribe, F; Lemire, M; Guimaraes, JR. A preliminary study of mercury exposure and blood pressure in the Brazilian Amazon. Environ. Health 2006, 5, 29. [Google Scholar]
- Valera, B; Dewailly, E; Poirier, P. Cardiac autonomic activity and blood pressure among Nunavik Inuit adults exposed to environmental mercury: a cross-sectional study. Environ. Health 2008, 7, 29. [Google Scholar]
- Choi, AL; Weihe, P; Budtz-Jorgensen, E; Jorgensen, PJ; Salonen, JT; Tuomainen, TP; Murata, K; Nielsen, HP; Petersen, MS; Askham, J; Grandjean, P. Methylmercury exposure and adverse cardiovascular effects in Faroese whaling men. Environ. Health Perspect 2009, 117, 367–372. [Google Scholar]
- Ahlqwist, M; Bengtsson, C; Lapidus, L; Gergdahl, IA; Schutz, A. Serum mercury concentration in relation to survival, symptoms, and diseases: results from the prospective population study of women in Gothenburg, Sweden. Acta Odontol Scand 1999, 57, 168–174. [Google Scholar]
- Hallgren, CG; Hallmans, G; Jansson, JH; Marklund, SL; Huhtasaari, F; Schutz, A; Stromberg, U; Vessby, B; Skerfving, S. Markers of high fish intake are associated with decreased risk of a first myocardial infarction. Br. J. Nutr 2001, 86, 397–404. [Google Scholar]
- Guallar, E; Sanz-Gallardo, MI; van’t Veer, P; Bode, P; Aro, A; Gomez-Aracena, J; Kark, JD; Riemersma, RA; Martin-Moreno, JM; Kok, FJ. Mercury, fish oils, and the risk of myocardial infarction. N. Engl. J. Med 2002, 347, 1747–1754. [Google Scholar]
- Yoshizawa, K; Rimm, EB; Morris, JS; Spate, VL; Hsieh, CC; Spiegelman, D; Stampfer, MJ; Willett, WC. Mercury and the risk of coronary heart disease in men. N. Engl. J. Med 2002, 347, 1755–1760. [Google Scholar]
- Virtanen, JK; Voutilainen, S; Rissanen, TH; Mursu, J; Tuomainen, TP; Korhonen, MJ; Valkonen, VP; Seppanen, K; Laukkanen, JA; Salonen, JT. Mercury, fish oils, and risk of acute coronary events and cardiovascular disease, coronary heart disease, and all-cause mortality in men in eastern Finland. Arterioscler. Thromb. Vasc. Biol 2005, 25, 228–233. [Google Scholar]
- Wennberg, M; Bergdahl, IA; Stegmayr, B; Hallmans, G; Lundh, T; Skerfving, S; Stromberg, U; Vessby, B; Jansson, JH. Fish intake, mercury, long-chain n-3 polyunsaturated fatty acids and risk of stroke in northern Sweden. Br. J. Nutr 2007, 98, 1038–1045. [Google Scholar]
- Thom, T; Haase, N; Rosamond, W; Howard, VJ; Rumsfeld, J; Manolio, T; Zheng, ZJ; Flegal, K; O’Donnell, C; Kittner, S; Lloyd-Jones, D; Goff, DC, Jr; Hong, Y; Adams, R; Friday, G; Furie, K; Gorelick, P; Kissela, B; Marler, J; Meigs, J; Roger, V; Sidney, S; Sorlie, P; Steinberger, J; Wasserthiel-Smoller, S; Wilson, M; Wolf, P. Heart disease and stroke statistics-2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2006, 113, e85–151. [Google Scholar]
- Konig, A; Bouzan, C; Cohen, JT; Connor, WE; Kris-Etherton, PM; Gray, GM; Lawrence, RS; Savitz, DA; Teutsch, SM. A quantitative analysis of fish consumption and coronary heart disease mortality. Am. J. Prev. Med 2005, 29, 335–346. [Google Scholar]
- Mozaffarian, D; Rimm, EB. Methylmercury and risk in adults - A balanced view, and more research, are needed. Environ. Health 2007, 6, 31. [Google Scholar]
- Rissanen, T; Voutilainen, S; Nyyssonen, K; Lakka, TA; Salonen, JT. Fish oil-derived fatty acids, docosahexaenoic acid and docosapentaenoic acid, and the risk of acute coronary events: the Kuopio ischaemic heart disease risk factor study. Circulation 2000, 102, 2677–2679. [Google Scholar]
- Levander, OA. A global view of human selenium nutrition. Annu. Rev. Nutr 1987, 7, 227–250. [Google Scholar]
- Holben, DH; Smith, AM. The diverse role of selenium within selenoproteins: a review. J. Am. Diet. Assoc 1999, 99, 836–843. [Google Scholar]
- Brown, KM; Arthur, JR. Selenium, selenoproteins and human health: a review. Public Health Nutr 2001, 4, 593–599. [Google Scholar]
- National Institutes of Health; Office of Dietary Supplements. Dietary supplement fact sheet: Selenium.
- Li, JM; Shah, AM. Endothelial cell superoxide generation: regulation and relevance for cardiovascular pathophysiology. Am. J. Physiol. Regul. Integr. Comp. Physiol 2004, 287, R1014–1030. [Google Scholar]
- Huang, K; Liu, H; Chen, Z; Xu, H. Role of selenium in cytoprotection against cholesterol oxide-induced vascular damage in rats. Atherosclerosis 2002, 162, 137–144. [Google Scholar]
- Traulsen, H; Steinbrenner, H; Buchczyk, DP; Klotz, LO; Sies, H. Selenoprotein P protects low-density lipoprotein against oxidation. Free Radic. Res 2004, 38, 123–128. [Google Scholar]
- Tanguy, S; Boucher, F; Besse, S; Ducros, V; Favier, A; de Leiris, J. Trace elements and cardioprotection: increasing endogenous glutathione peroxidase activity by oral selenium supplementation in rats limits reperfusion-induced arrhythmias. J. Trace Elem. Med. Biol 1998, 12, 28–38. [Google Scholar]
- Tanguy, S; Toufektsian, MC; Besse, S; Ducros, V; De Leiris, J; Boucher, F. Dietary selenium intake affects cardiac susceptibility to ischaemia/reperfusion in male senescent rats. Age Ageing 2003, 32, 273–278. [Google Scholar]
- Tanguy, S; Morel, S; Berthonneche, C; Toufektsian, MC; de Lorgeril, M; Ducros, V; Tosaki, A; de Leiris, J; Boucher, F. Preischemic selenium status as a major determinant of myocardial infarct size in vivo in rats. Antioxid. Redox. Signal 2004, 6, 792–796. [Google Scholar]
- Ayaz, M; Ozdemir, S; Ugur, M; Vassort, G; Turan, B. Effects of selenium on altered mechanical and electrical cardiac activities of diabetic rat. Arch. Biochem. Biophys 2004, 426, 83–90. [Google Scholar]
- Ayaz, M; Can, B; Ozdemir, S; Turan, B. Protective effect of selenium treatment on diabetes-induced myocardial structural alterations. Biol. Trace Elem. Res 2002, 89, 215–226. [Google Scholar]
- Longnecker, MP; Stram, DO; Taylor, PR; Levander, OA; Howe, M; Veillon, C; McAdam, PA; Patterson, KY; Holden, JM; Morris, JS; Swanson, CA; Willett, WC. Use of selenium concentration in whole blood, serum, toenails, or urine as a surrogate measure of selenium intake. Epidemiology 1996, 7, 384–390. [Google Scholar]
- Hunter, DJ; Morris, JS; Chute, CG; Kushner, E; Colditz, GA; Stampfer, MJ; Speizer, FE; Willett, WC. Predictors of selenium concentration in human toenails. Am. J. Epidemiol 1990, 132, 114–122. [Google Scholar]
- Flores-Mateo, G; Navas-Acien, A; Pastor-Barriuso, R; Guallar, E. Selenium and coronary heart disease: a meta-analysis. Am. J. Clin. Nutr 2006, 84, 762–773. [Google Scholar]
- Yoshizawa, K; Ascherio, A; Morris, JS; Stampfer, MJ; Giovannucci, E; Baskett, CK; Willett, WC; Rimm, EB. Prospective study of selenium levels in toenails and risk of coronary heart disease in men. Am. J. Epidemiol 2003, 158, 852–860. [Google Scholar]
- Tribble, DL. AHA Science Advisory. Antioxidant consumption and risk of coronary heart disease: emphasison vitamin C, vitamin E, and beta-carotene: A statement for healthcare professionals from the American Heart Association. Circulation 1999, 99, 591–595. [Google Scholar]
- Burk, RF. Selenium, an antioxidant nutrient. Nutr. Clin. Care 2002, 5, 75–79. [Google Scholar]
- Levander, OA. Selenium requirements as discussed in the 1996 joint FAO/IAEA/WHO expert consultation on trace elements in human nutrition. Biomed. Environ. Sci 1997, 10, 214–219. [Google Scholar]
- Parizek, J; Ostadalova, I. The protective effect of small amounts of selenite in sublimate intoxication. Experientia 1967, 23, 142–143. [Google Scholar]
- Ganther, HE; Goudie, C; Sunde, ML; Kopecky, MJ; Wagner, P. Selenium: relation to decreased toxicity of methylmercury added to diets containing tuna. Science 1972, 175, 1122–1124. [Google Scholar]
- Skerfving, S. Interaction between selenium and methylmercury. Environ. Health Perspect 1978, 25, 57–65. [Google Scholar]
- Ganther, HE. Modification of methylmercury toxicity and metabolism by selenium and vitamin E: possible mechanisms. Environ. Health Perspect 1978, 25, 71–76. [Google Scholar]
- Cuvin-Aralar, ML; Furness, RW. Mercury and selenium interaction: a review. Ecotoxicol. Environ. Saf 1991, 21, 348–364. [Google Scholar]
- Watanabe, C. Modification of mercury toxicity by selenium: practical importance? Tohoku. J. Exp. Med 2002, 196, 71–77. [Google Scholar]
- Stoewsand, GS; Bache, CA; Lisk, DJ. Dietary selenium protection of methylmercury intoxication of Japanese quail. Bull. Environ. Contam. Toxicol 1974, 11, 152–156. [Google Scholar]
- Iwata, H; Masukawa, T; Kito, H; Hayashi, M. Degradation of methylmercury by selenium. Life Sci 1982, 31, 859–866. [Google Scholar]
- Iwata, H; Masukawa, T; Kito, H; Hayashi, M. Degradation of methylmercury by selenium. Life Sci 1982, 31, 859–866. [Google Scholar]
- Goyer, RA. Nutrition and metal toxicity. Am. J. Clin. Nutr 1995, 61, 646S–650S. [Google Scholar]
- El-Demerdash, FM. Effects of selenium and mercury on the enzymatic activities and lipid peroxidation in brain, liver, and blood of rats. J. Environ. Sci. Health B 2001, 36, 489–499. [Google Scholar]
- Frisk, P; Wester, K; Yaqob, A; Lindh, U. Selenium protection against mercury-induced apoptosis and growth inhibition in cultured K-562 cells. Biol. Trace Elem. Res 2003, 92, 105–114. [Google Scholar]
- Sasakura, C; Suzuki, KT. Biological interaction between transition metals (Ag, Cd and Hg), selenide/sulfide and selenoprotein P. J. Inorg. Biochem 1998, 71, 159–162. [Google Scholar]
- Seppanen, K; Kantola, M; Laatikainen, R; Nyyssonen, K; Valkonen, VP; Kaarlopp, V; Salonen, JT. Effect of supplementation with organic selenium on mercury status as measured by mercury in pubic hair. J. Trace. Elem. Med. Biol 2000, 14, 84–87. [Google Scholar]
- Buettner, C. Mercury and the risk of myocardial infarction. N. Engl. J. Med 2003, 348, 2151–2154. [Google Scholar]
- Aylward, LL; Hays, SM. Temporal trends in human TCDD body burden: decreases over three decades and implications for exposure levels. J. Expo. Anal. Environ. Epidemiol 2002, 12, 319–328. [Google Scholar]
- Hites, RA; Foran, JA; Carpenter, DO; Hamilton, MC; Knuth, BA; Schwager, SJ. Global assessment of organic contaminants in farmed salmon. Science 2004, 303, 226–229. [Google Scholar]
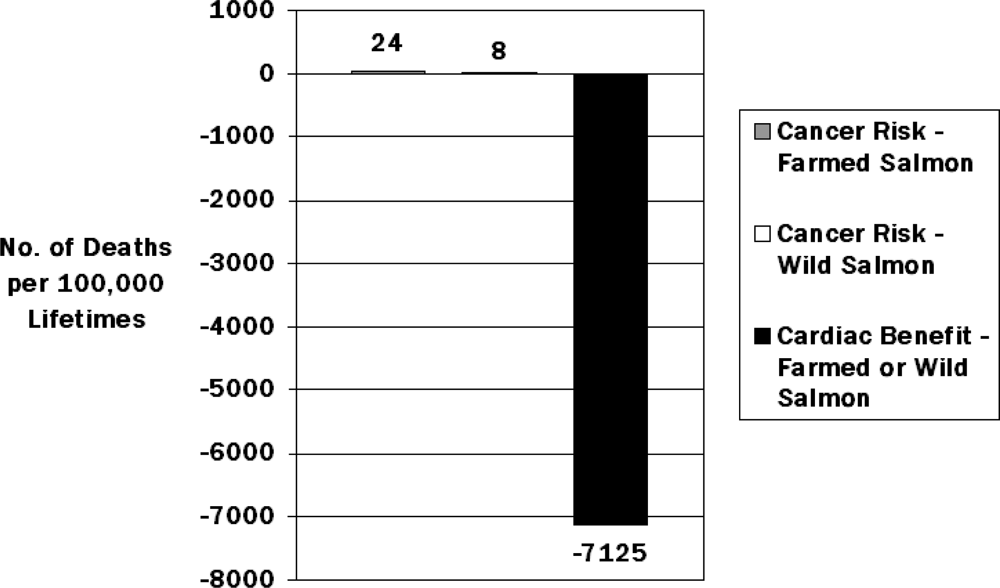
- Foran, JA; Good, DH; Carpenter, DO; Hamilton, MC; Knuth, BA; Schwager, SJ. Quantitative analysis of the benefits and risks of consuming farmed and wild salmon. J. Nutr 2005, 135, 2639–2643. [Google Scholar]
- Schecter, A; Cramer, P; Boggess, K; Stanley, J; Papke, O; Olson, J; Silver, A; Schmitz, M. Intake of dioxins and related compounds from food in the U.S. population. J. Toxicol. Environ. Health A 2001, 63, 1–18. [Google Scholar]
- Judd, N; Griffith, WC; Faustman, EM. Contribution of PCB exposure from fish consumption to total dioxin-like dietary exposure. Regul. Toxicol. Pharmacol 2004, 40, 125–135. [Google Scholar]

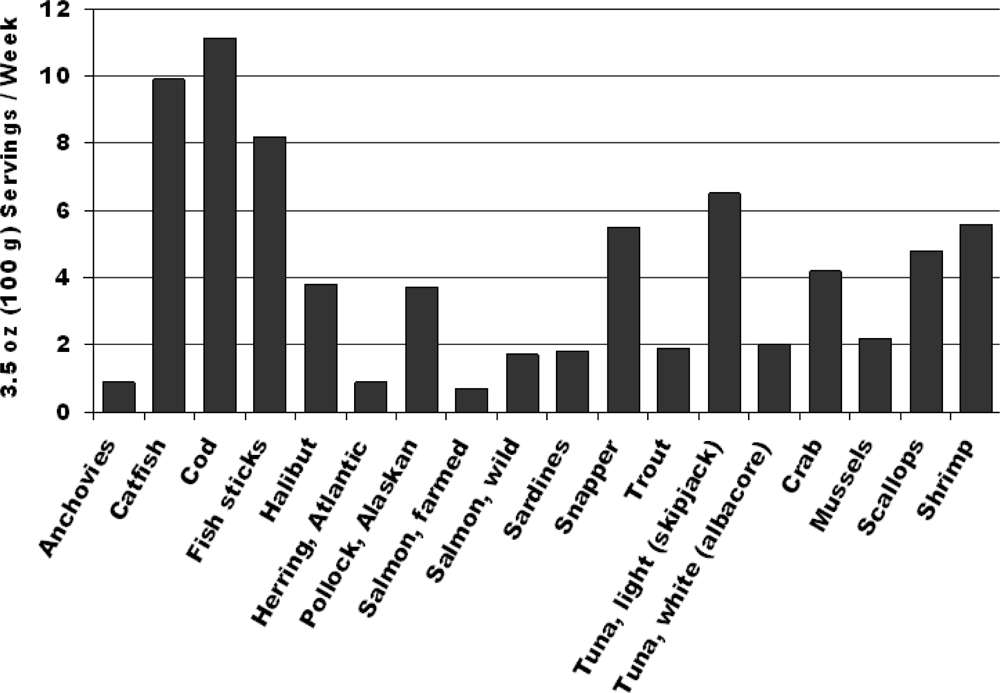
| Systemic Effects |
| Promotion of free radicals and reactive oxygen species |
| Inhibition of antioxidant systems (glutathione peroxidase, catalase, superoxide dismutase) |
| Increased lipid (e.g., LDL cholesterol) peroxidation |
| Promotion of blood coagulation (clotting) |
| Inhibition of endothelial cell migration |
| Direct Cardiovascular Effects |
| Reduction in myocardial contractile force |
| Increased calcium release from myocardial sarcoplasmic reticulum |
| Reduction in left ventricular myosin ATPase activity |
| Decreased heart rate variability and increased blood pressure |
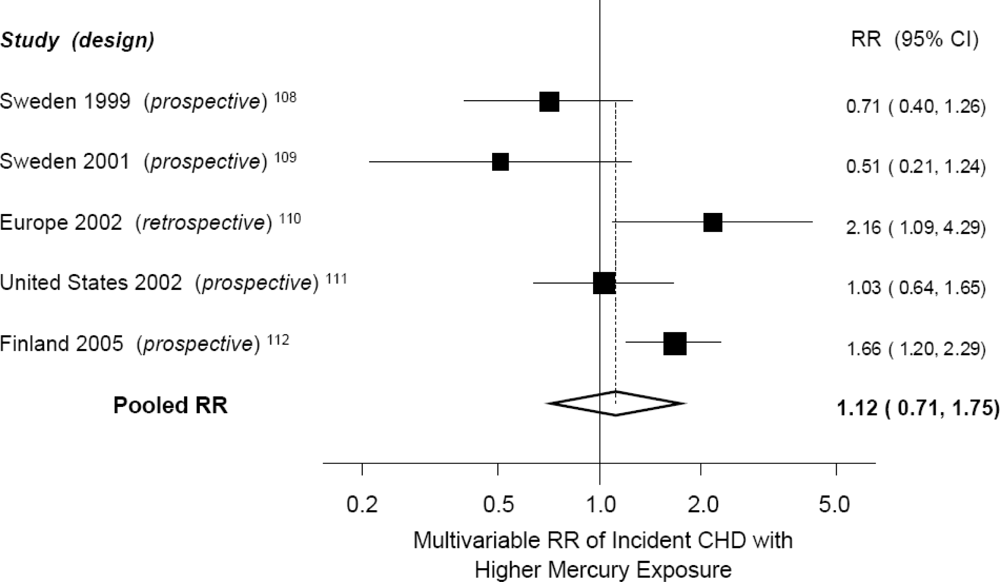
| Author, Year | Design | Country | Exposure | Outcome | No. of Cases | Relative Risk (5% CI) with High Mercury * |
|---|---|---|---|---|---|---|
| Ahlqwist, 1999 | Prospective cohort | Sweden | Serum mercury | Total myocardial infarction, Stroke | 87, 77 | RR = 0.71 (0.40, 1.26)† for MI; RR = NS (data not reported) for stroke |
| Hallgren, 2001 | Prospective (nested) case-control | Sweden | Erythrocyte mercury | Total myocardial infarction | 78 | RR = 0.51 (0.21, 1.24) |
| Guallar, 2002 | Retrospective case-control | 8 European countries and Israel | Toenail mercury | Nonfatal myocardial infarction | 684 | RR = 2.16 (1.09, 4.29) |
| Yoshizawa, 2002 | Prospective (nested) case-control | USA | Toenail mercury | Total myocardial infarction + coronary revascularization | 470 | RR = 1.03 (0.65, 1.65) |
| Virtanen, 2005 | Prospective cohort | Finland | Hair mercury | Total acute coronary events | 282 | RR = 1.66 (1.20, 2.29) |
| Wennberg, 2007 | Prospective (nested) case-control | Sweden | Erythrocyte mercury | Stroke | 369 | RR = 0.99 (0.93, 1.06) in men; RR = 1.00 (0.94, 1.08) in women |
| Systemic Effects |
| Antioxidant defense against free radicals and reactive oxygen species |
| Decreased lipid peroxidation |
| Protection against vascular damage from oxidized LDL cholesterol particles |
| Antithrombotic effects from decreased plasma thromboxane A2 |
| Direct Cardiovascular Effects |
| Increased myocardial antioxidant glutathione peroxidase activity |
| Improved cardiac recovery from ischemia-reperfusion injury |
| Limitation of ischemia-induced and diabetes-induced ultrastructural damage |
| Reduction in myocardial infarct size |
| Restoration of altered myocyte ion currents |
| Reduced incidence of ischemia-induced ventricular arrhythmias |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mozaffarian, D. Fish, Mercury, Selenium and Cardiovascular Risk: Current Evidence and Unanswered Questions. Int. J. Environ. Res. Public Health 2009, 6, 1894-1916. https://doi.org/10.3390/ijerph6061894
Mozaffarian D. Fish, Mercury, Selenium and Cardiovascular Risk: Current Evidence and Unanswered Questions. International Journal of Environmental Research and Public Health. 2009; 6(6):1894-1916. https://doi.org/10.3390/ijerph6061894
Chicago/Turabian StyleMozaffarian, Dariush. 2009. "Fish, Mercury, Selenium and Cardiovascular Risk: Current Evidence and Unanswered Questions" International Journal of Environmental Research and Public Health 6, no. 6: 1894-1916. https://doi.org/10.3390/ijerph6061894
APA StyleMozaffarian, D. (2009). Fish, Mercury, Selenium and Cardiovascular Risk: Current Evidence and Unanswered Questions. International Journal of Environmental Research and Public Health, 6(6), 1894-1916. https://doi.org/10.3390/ijerph6061894




