UVA Photoirradiation of Oxygenated Benz[a]anthracene and 3-Methylcholanthene - Generation of Singlet Oxygen and Induction of Lipid Peroxidation
Abstract
:Introduction
Materials and Methods
Materials
Light Source
Photoirradiation of Oxygenated BA and 3-MC with UVA Light in the Presence of Methyl Linoleate
ESR Spectral Measurements of Singlet Oxygen

Results
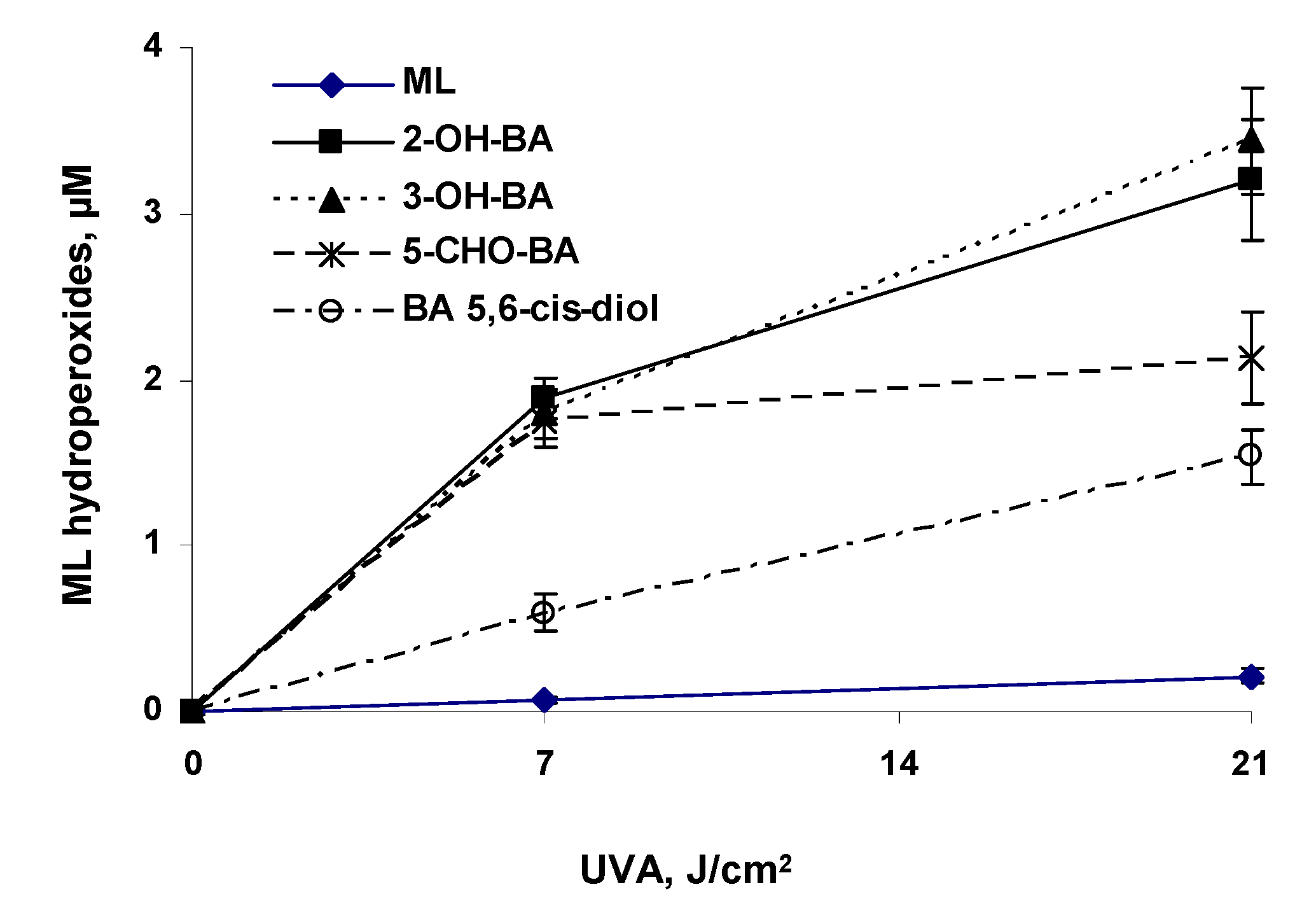
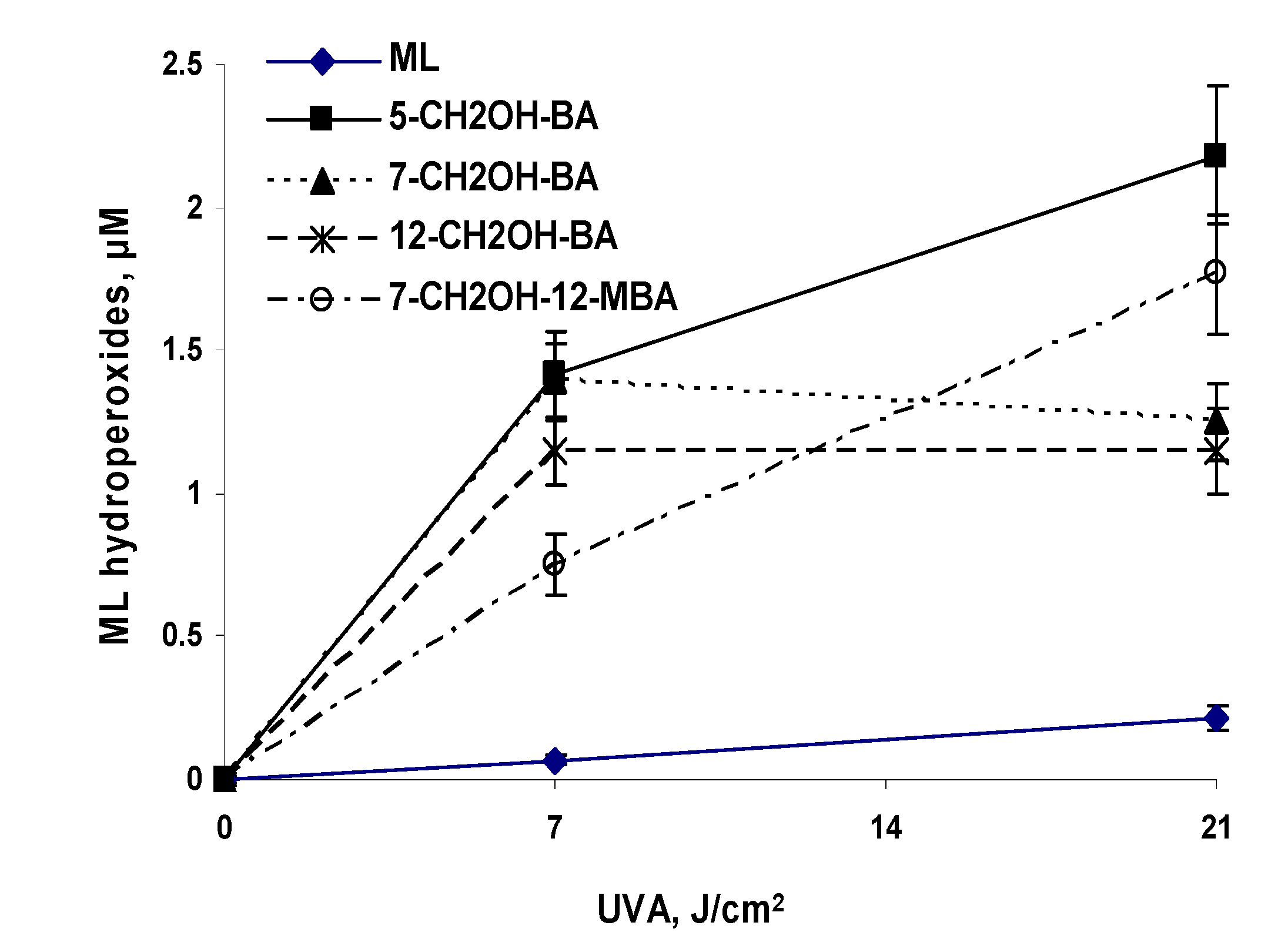
UVA Photoirradiation of Oxygenated BA and 3-MC in the Presence of Methyl Linoleate
| Compound | 0 J/cm2 | 7 J/cm2 | 21 J/cm2 |
| Methyl linoleate (ML) | 178 ± 15 | 267 ± 25 | 452 ± 59 |
| 2-Hydroxy-BA (2-OH-BA) | 187 ± 13 | 2597 ± 153a | 4283 ± 469a |
| 3-Hydroxy-BA (3-OH-BA) | 164 ± 15 | 2458 ± 190 | 4558 ± 412 |
| 5-Formyl-BA (5-CHO-BA) | 191 ± 10 | 2434 ± 206 | 2919 ± 351 |
| 5-Hydroxymethyl-BA (5-CH2OH-BA) | 205 ± 21 | 2012 ± 189 | 2990 ± 311 |
| 7-Hydroxymethyl-BA (7-CH2OH-BA) | 177 ± 19 | 1956 ± 171 | 1777 ± 169 |
| 12-Hydroxymethyl-BA (12-CH2OH-BA) | 185 ± 12 | 1654 ± 150 | 1651 ± 193 |
| 7-Hydroxymethyl-12-methyl-BA (7-CH2OH-12-MBA) | 161 ± 17 | 1121 ± 132 | 2419 ± 270 |
| Benz[a]anthracene 5,6-cis-Diol (BA 5,6-cis-Diol) | 159 ± 15 | 920 ± 135 | 2120 ± 208 |
| 1-Hydroxy-3-MC (1-OH-3-MC) | 201 ± 22 | 3370 ± 305 | 5160 ± 397 |
| 1-Keto-3-MC (1-Keto-3-MC) | 210 ± 18 | 3316 ± 321 | 6181 ± 462 |
| 3-MC 1,2-cis-dihydrodiol (3-MC 1,2-cis-diol) | 196 ± 26 | 4543 ± 489 | 10424 ± 580 |

ESR Spectral Measurements of Singlet Oxygen
Discussion
References
- Connell, D. W.; Hawker, D. W.; Warne, M. J.; Vowles, P. P. : Polycyclic aromatic hydrocarbons (PAHs). In Introduction into Environmental Chemistry (McCombs, K., and Starkweather, A. W., eds), CRC Press LLC, Boca Raton, FL., 1997, pp. 205-217.
- Harvey, R. G. : Polycyclic Aromatic Hydrocarbons, Wiley-VCH, New York, 1996.
- National Toxicology Program, P. H. S. , US Department of Health and Human Services, 10th Report on Carcinogens. In, Integrated Laboratory Systems, Inc., Research Triangle Park, NC. 2002, pp. III-201-204,.
- RamaKrishna, N. V. S.; Devanesan, P. D.; Rogan, E. G.; Cavalieri, E. L.; Jeong, H.; Jankowiak, R.; Small, G. J. : Mechanism of Metabolic Activation of the Potent Carcinogen 7,12-Dimethylbenz[a]anthracene. Chemical research in toxicolog, 1992, 5, 220–26. [Google Scholar]
- Penning, T. M.; Burczynski, M. E.; Hung, C. F.; McCoull, K. D.; Palackal, N. T.; Tsuruda, L. S. : Dihydrodiol Dehydrogenases and Polycyclic Aromatic Hydrocarbon Activation: Generation of Reactive and Redox Active o-Quinones. Chemical research in toxicology, 1999, 12, 1–18. [Google Scholar]
- Yu, H. : Environmental Carcinogenic polycyclic aromatic hydrocarbons: photochemistry and phototoxicity. J. Environ. Sci. Health. Part C, Environ. Carcinog. & Ecotox. Revs. 2002, C20, 149–83. [Google Scholar]
- Wislocki, P. G.; Fiorentini, K. M.; Fu, P. P.; Yang, S. K.; Lu, A. Y. H. :Tumor-initiating ability of the twelve monomethylbenz[a]anthracenes. Carcinogenesis, 1982, 3, 215–17. [Google Scholar]
- Mastrangelo, G.; Fadda, E.; Marzia, V. : Polycyclic aromatic hydrocarbons and cancers in man. Environmental health perspectives, 1996, 104, 1166–70. [Google Scholar] [CrossRef] [PubMed]
- National Toxicology Program Soots, Tars, and Mineral Oils. Eighth Report on Carcinogens, 1998, pp 42-46: 178-181.
- Kagan, J.; Tuveson, R. W.; Gong, H. H. : The light- dependent cytotoxicity of benzo[a]pyrene: effect on human erythrocytes, Escherichia coli cells, and Haemophilus influenzae transforming DNA. Mutation research 1989, 216, 231–42. [Google Scholar]
- Fernandez, M.; L’Haridon, J. : Influence of lighting conditions on toxicity and genotoxicity of various PAH in the newt in vivo. Mutation research 1992, 298, 31–41. [Google Scholar]
- Pelletier, M. C.; Burgess, R. M.; Ho, K. T.; Kuhn, A.; McKinney, R. A.; Ryba, S. A. : Phototoxicity of individual polycyclic aromatic hydrocarbons and petroleum to marine invertebrate larvae and juveniles. Environmental toxicology and chemistry, 1997, 16, 2190–99. [Google Scholar]
- Swartz, R. C.; Ferraro, S. P.; Lamberson, J. O.; Cole, F. A.; Ozretich, R. J.; Boese, B. L.; Schults, D. W.; Behrenfeld, M.; Ankley, G.T. : Photoactivation and toxicity of mixtures of polycyclic aromatic hydrocarbon compounds in marine sediment. Environmental toxicology and chemistry, 1997, 16, 2151–57. [Google Scholar]
- Stern, R. S.; Zierler, S.; Parrish, J. A. : Skin carcinoma in patients with psoriasis treated with topical tar and artificial ultraviolet radiation. Lancet 1980, 2, 732–33. [Google Scholar]
- Reed, L. D.; Liss, G. M. : PAH exposure among pitch and asphalt roofing workers, Polynuclear Aromatic Hydrocarbons: Mechanisms, Method, and Metabolism (Cooke and Dennis Ed.), Bartelle Press, Columbus, 1985, pp 1089-95.
- Wang, L; Yan, J.; Cohly, H.; Hwang, H.-M.; Wang, S.; Fu, P. P.; Yu, H.: Photo-acute toxicity and genotoxicity of azulene on human Jurkat T-cells. Mutation Res.: Gen. Toxicl. Environ. Mutag., 2004, 562 (1-2), 143–150.
- Yu, H.; Yan, J.; Jiao, Y.; Fu, P. P. : Photochemical reaction of 7,12-dimethylbenz[a]anthracene (DMBA) and formation of DNA covalent adducts. Int. J. Environ. Res. Public Health, 2005, 2(1), 114–122. [Google Scholar]
- Xia, Q.; Chou, M. W.; Yin, J. J.; Howard, P. C.; Yu, H.; Fu, P. P. : Photoirradiation of representative polycyclic aromatic hydrocarbons and twelve isomeric ethylbenz[a]anthracene with UVA light – formation of lipid peroxidation. Toxicol. Ind. Health, 2006, 22, 147–156. [Google Scholar]
- Yu, H.; Xia, Q.; Yan, J.; Herreno-Saenz, D.; Wu, Y.- S.; Tang, I-W.; Fu, P. P. : Photoirradiation of polycyclic aromatic hydrocarbons with UVA light – a pathway leading to the generation of reactive oxygen species, lipid peroxidation, and DNA damage. Int. J. Environ. Res. Public Health 2006, 3(4), 348–354. [Google Scholar]
- Herreno-Sáenz, D.; Xia, Q.; Chiu, L. T.; Fu, P. P. : UVA photoirradiation of halogenated-polycyclic aromatic hydrocarbons leading to induction of lipid peroxidation. Int. J. Environ. Res. Public Health. 2006, 3(2), 191–195. [Google Scholar] [CrossRef] [PubMed]
- Herreño-Sáenz, D.; Xia, Q.; Fu, P. P. : UVA Photoirradiation of methylated benzo[a]pyrene and benzo[e]pyrene leading to induction of lipid peroxidation. Int. J. Environ. Res. Pub. Health, 2007, 4, 145-152.
- Fu, P. P.; Yang, S. K. : A simple method for the synthesis of specific [2H] and [3H] labelled methyl-hydroxylated derivatives of 7,12-dimethyl- benz[a]anthracene, J. Label. Cpds. Radiopharm., 1979, 16, 819-826.
- Lee, H.; Harvey, R. G. : Synthesis of oxygenated derivatives of 3-methylcholanthrene, Org. Prep. Proc. Int., 1988, 20(1-2), 123-128.
- Lee, H.; Harvey, R. G. : 2,3-Dichloro-5,6-dicyano- 1,4-benzoquinone (DDQ) in aqueous acetic acid, a convenient new reagent for the synthesis of aryl ketones and aldehydes via benzylic oxidation, J. Org. Chem., 1988, 53(19), 4587-4589.
- Tokita, M.; Morita, M. : Identification of new geometric isomers of methyl linoleate hydroperoxide and their chromatographic behavior. Biosci Biotechnol Biochem, 2000, 64(5):1044-6.
- Gibian M., J.; Vandenberg, P. : Product yield in oxygenation of linoleate by soybean lipoxygenase: the value of the molar extinction coefficient in the spectrophotometric assay. Anal Biochem, 1987, 163(2):343-9.
- Cherng, S.-H.; Xia, Q.; Blankenship, L. R.; Freeman, J. P.; Wamer, W. G.; Howard, P. C.; Fu, P. P.: Photodecomposition of retinyl palmitate in ethanol by UVA light - formation of photodecomposition products, reactive oxygen species, and lipid peroxides. Chem. Res. Toxicol., 2005, 18, 129-138.
- Xia, Q.; Yin, J. J.; Cherng, S.-H.; Wamer, W. G.; Boudreau, M.; Howard, P. C.; Fu, P. P. : UVA Photodecomposition of retinyl palmitate – formation of singlet oxygen and superoxide, and their role in induction of lipid peroxidation. Toxicol. Lett., 2006, 163, 30-43.
- Rinalducci, S.; Pedersen, J. Z.; Zolla, L. : Formation of radical from signet oxygen produced during photoinhibition of isolated light-harvesting protein of photosystem II. Biochim. Biophy. Acta, 2004, 1608, 63-73.
- Lion, Y.; Delmelle, M.; Van De Vorst, A. : New method of detecting singlet oxygen production. Nature, 1976, 263, 442-443.
- Lion, Y.; Grandin, E.; Van De Vorst, A. : On the production of nitroxide radicals by singlet oxygen reaction: an ESR study, Photochem. Photobiol., 1980, 31, 305-309.
- Xia, Q.; Yin, J. J.; Fu, P. P.; Boudreau, M. D. : Photo- irradiation of Aloe vera by UVA - formation of free radicals, singlet oxygen, superoxide, and induction of lipid peroxidation. Toxicol. Lett., 2007, 168, 165-175.
- Fu, P. P.; Xia, Q.; Yin, J. J.; Cherng, S.-H.; Yan, J.; Mei, N.; Chen, T.; Boudreau, M. D.; Howard, P. C. ; Wamer. W. G.: Photodecomposition of vitamin A and photobiological implications for the skin. Photochem. Photobiol., 2007, 83, 409-424.
- Dipple, A.; Moschel, R. C.; Bigger, C. A. H. : Polynuclear aromatic carcinogens. Chemical Carcinogens (C. E. Searle, ed.), American Chemical Society Monograph 182, American Chemical Society, Washington, D.C., 1984, Vol. 1. pp. 41-163.
- Tyrrell, R. M. : Role for singlet oxygen in biological effects of ultraviolet A radiation. Methods in enzymology, 2000, 319, 290–96. [Google Scholar] [PubMed]
- Loft, S.; Poulsen, H. E. : Cancer risk and oxidative DNA damage in man. Journal of molecular medicine 1996, 74(6), 297–312. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2008 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yin, J.-J.; Xia, Q.; Cherng, S.-H.; Tang, I.-W.; Fu, P.P.; Lin, G.; Yu, H.; Sáenz, D.H. UVA Photoirradiation of Oxygenated Benz[a]anthracene and 3-Methylcholanthene - Generation of Singlet Oxygen and Induction of Lipid Peroxidation. Int. J. Environ. Res. Public Health 2008, 5, 26-31. https://doi.org/10.3390/ijerph5010026
Yin J-J, Xia Q, Cherng S-H, Tang I-W, Fu PP, Lin G, Yu H, Sáenz DH. UVA Photoirradiation of Oxygenated Benz[a]anthracene and 3-Methylcholanthene - Generation of Singlet Oxygen and Induction of Lipid Peroxidation. International Journal of Environmental Research and Public Health. 2008; 5(1):26-31. https://doi.org/10.3390/ijerph5010026
Chicago/Turabian StyleYin, Jun-Jie, Qingsu Xia, Shu-Hui Cherng, I-Wah Tang, Peter P. Fu, Ge Lin, Hongtao Yu, and Diógenes Herreño Sáenz. 2008. "UVA Photoirradiation of Oxygenated Benz[a]anthracene and 3-Methylcholanthene - Generation of Singlet Oxygen and Induction of Lipid Peroxidation" International Journal of Environmental Research and Public Health 5, no. 1: 26-31. https://doi.org/10.3390/ijerph5010026
APA StyleYin, J.-J., Xia, Q., Cherng, S.-H., Tang, I.-W., Fu, P. P., Lin, G., Yu, H., & Sáenz, D. H. (2008). UVA Photoirradiation of Oxygenated Benz[a]anthracene and 3-Methylcholanthene - Generation of Singlet Oxygen and Induction of Lipid Peroxidation. International Journal of Environmental Research and Public Health, 5(1), 26-31. https://doi.org/10.3390/ijerph5010026




