Fluoro-edenite Fibers Induce Expression of Hsp70 and Inflammatory Response
Abstract
:Introduction
Materials and Methods
Preparation of Cell Cultures
Preparation and Composition of Asbestos Fibers
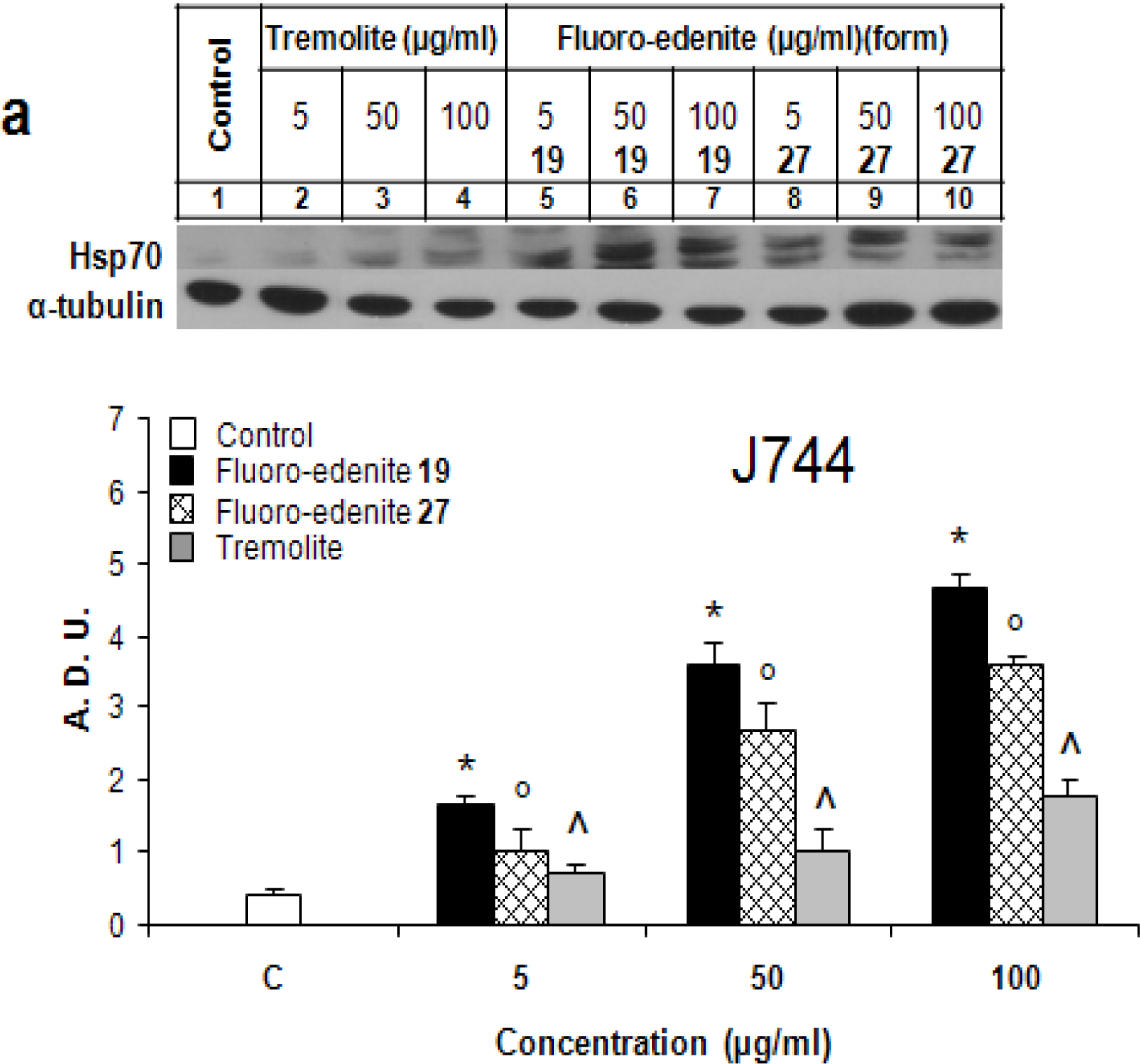
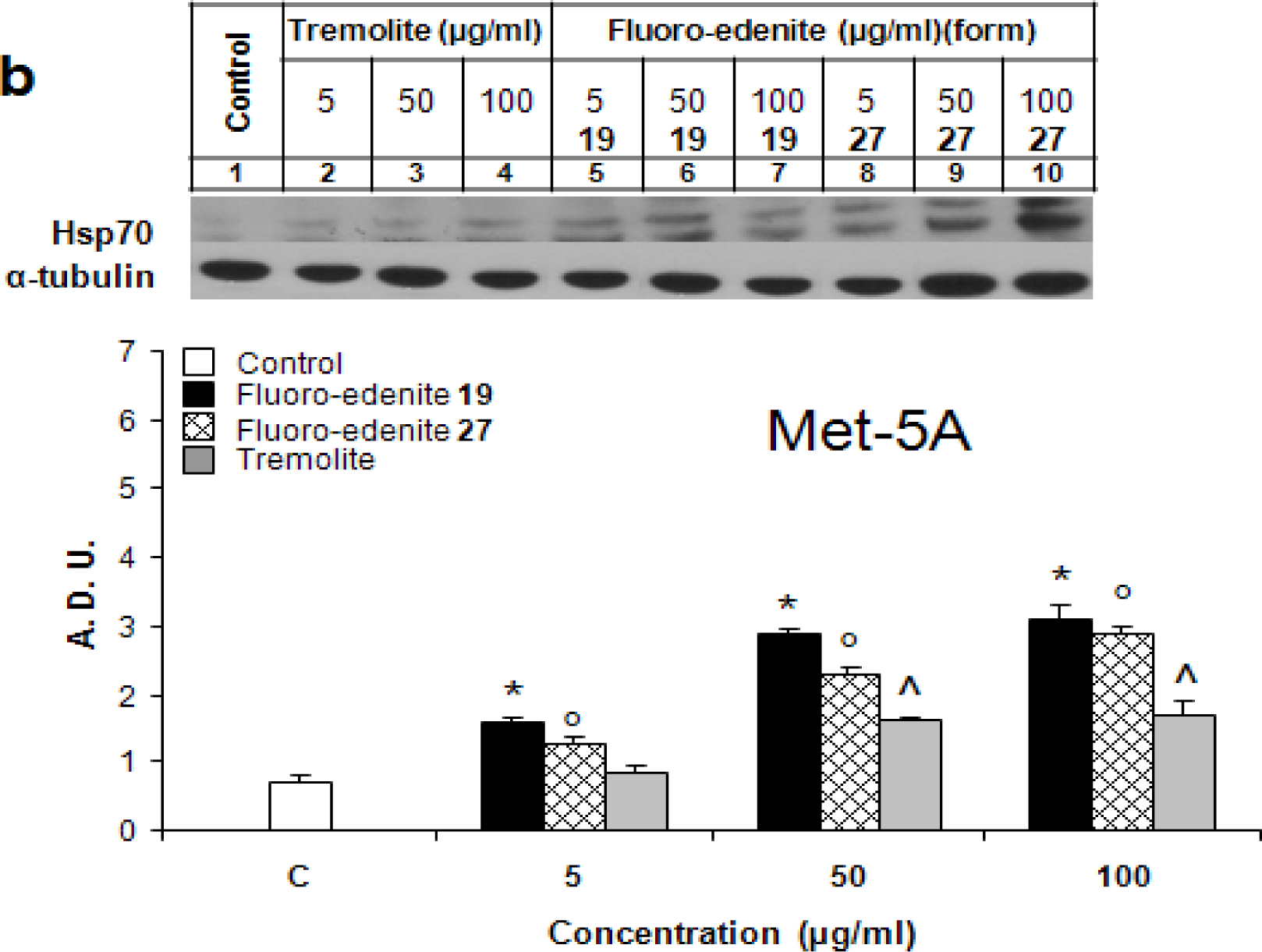
Identification of Hsp70
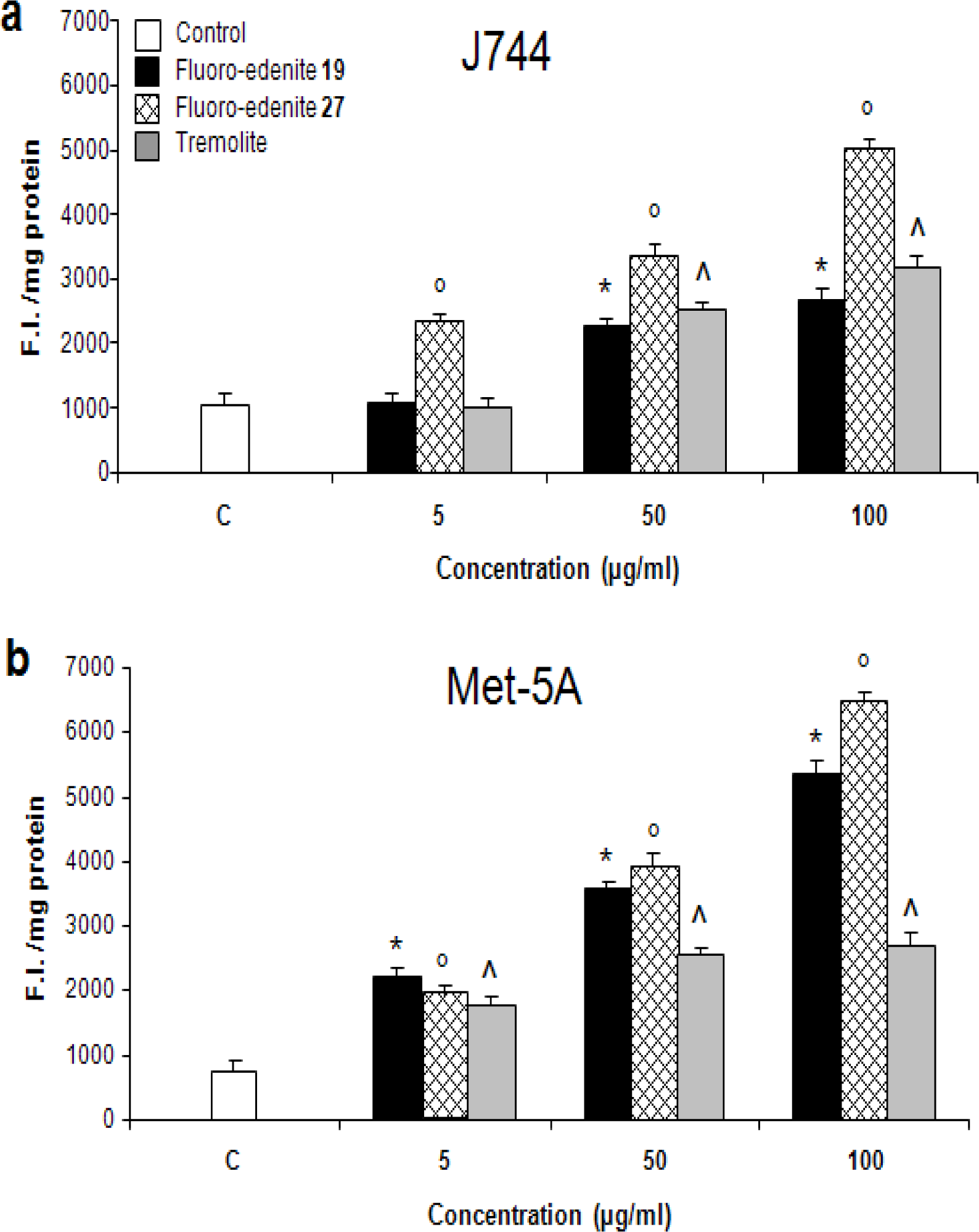
Analysis of Reactive Oxygen Species
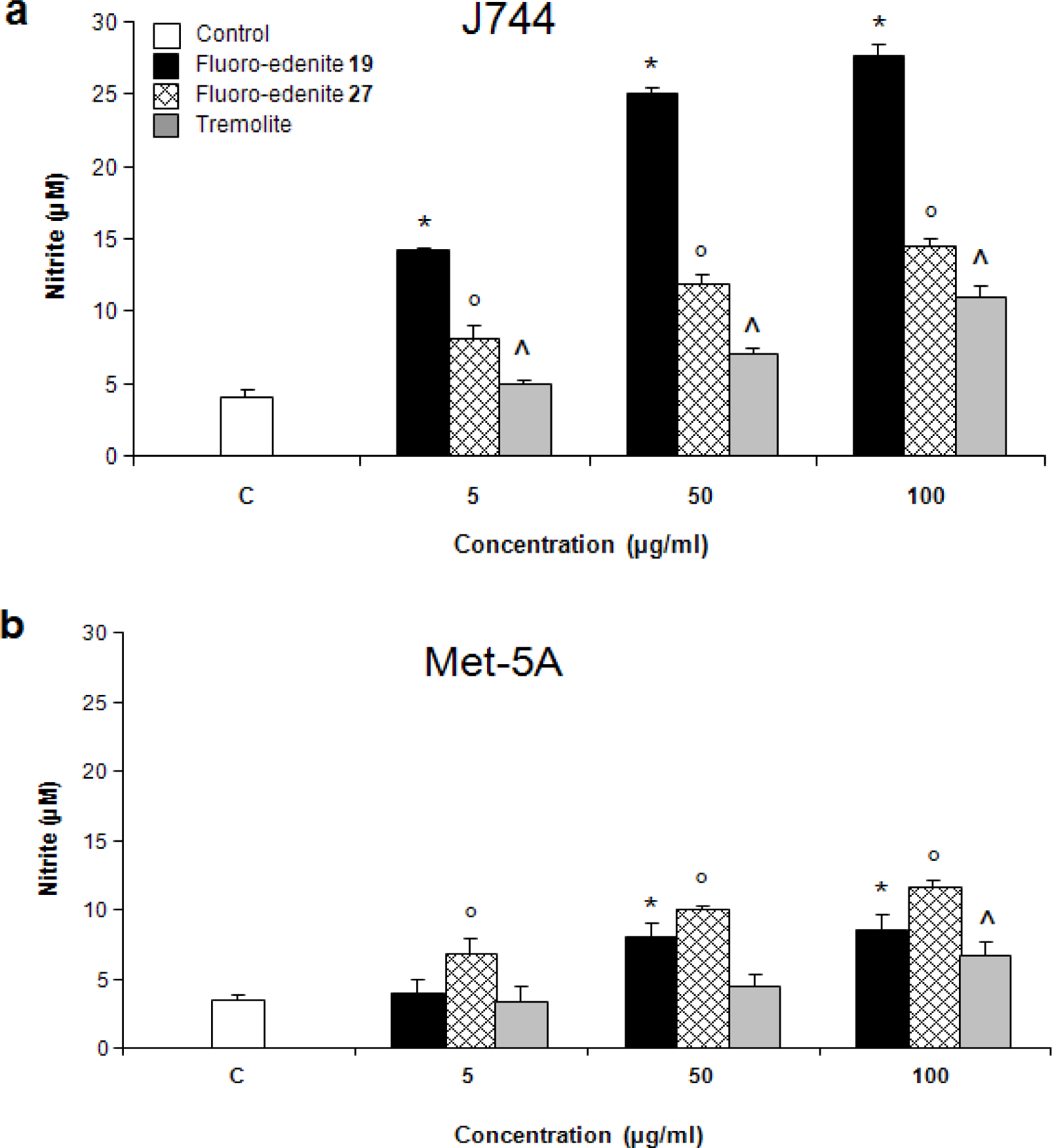
Analysis of Nitrite and LDH
Determination of Cell Viability
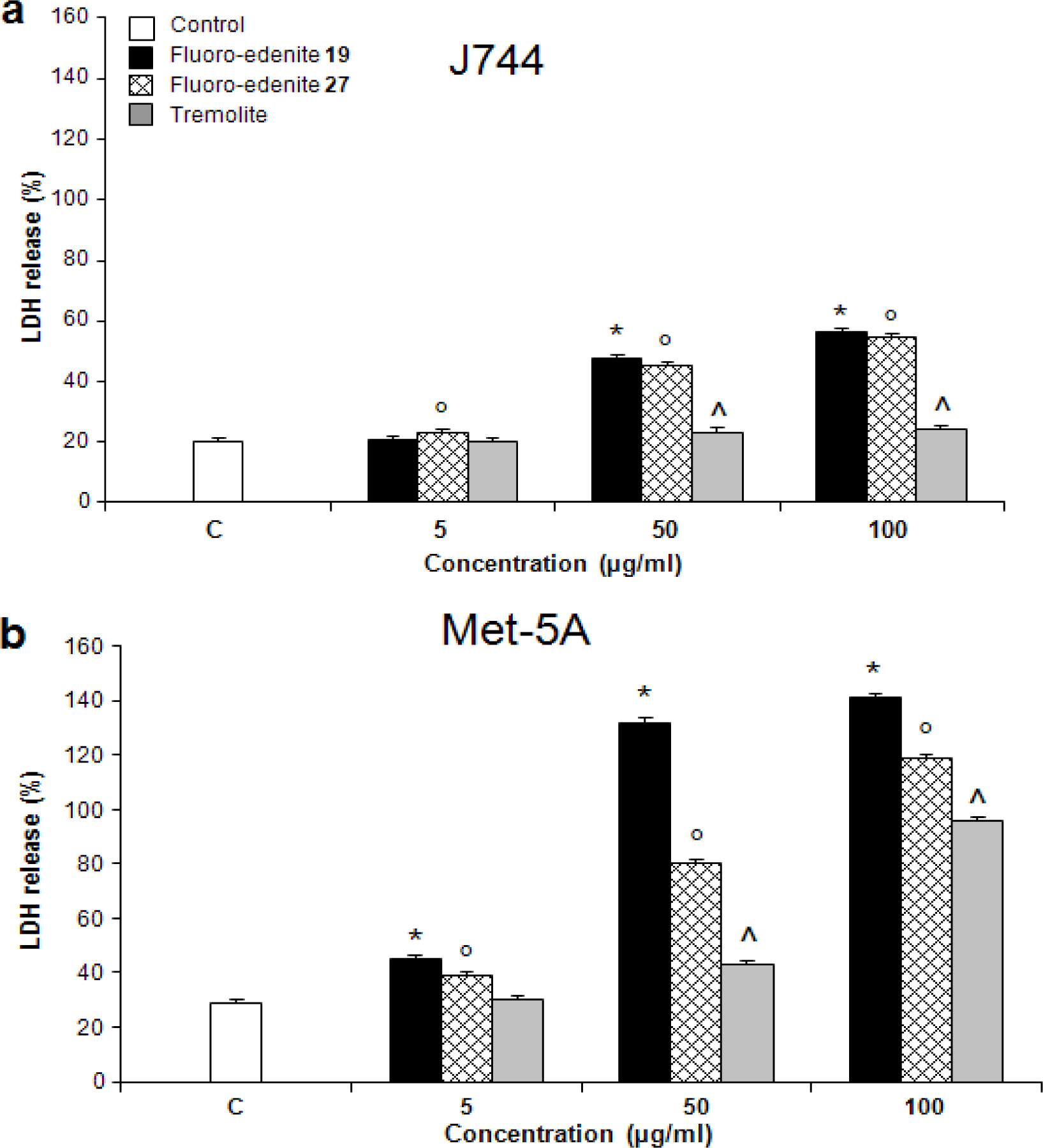
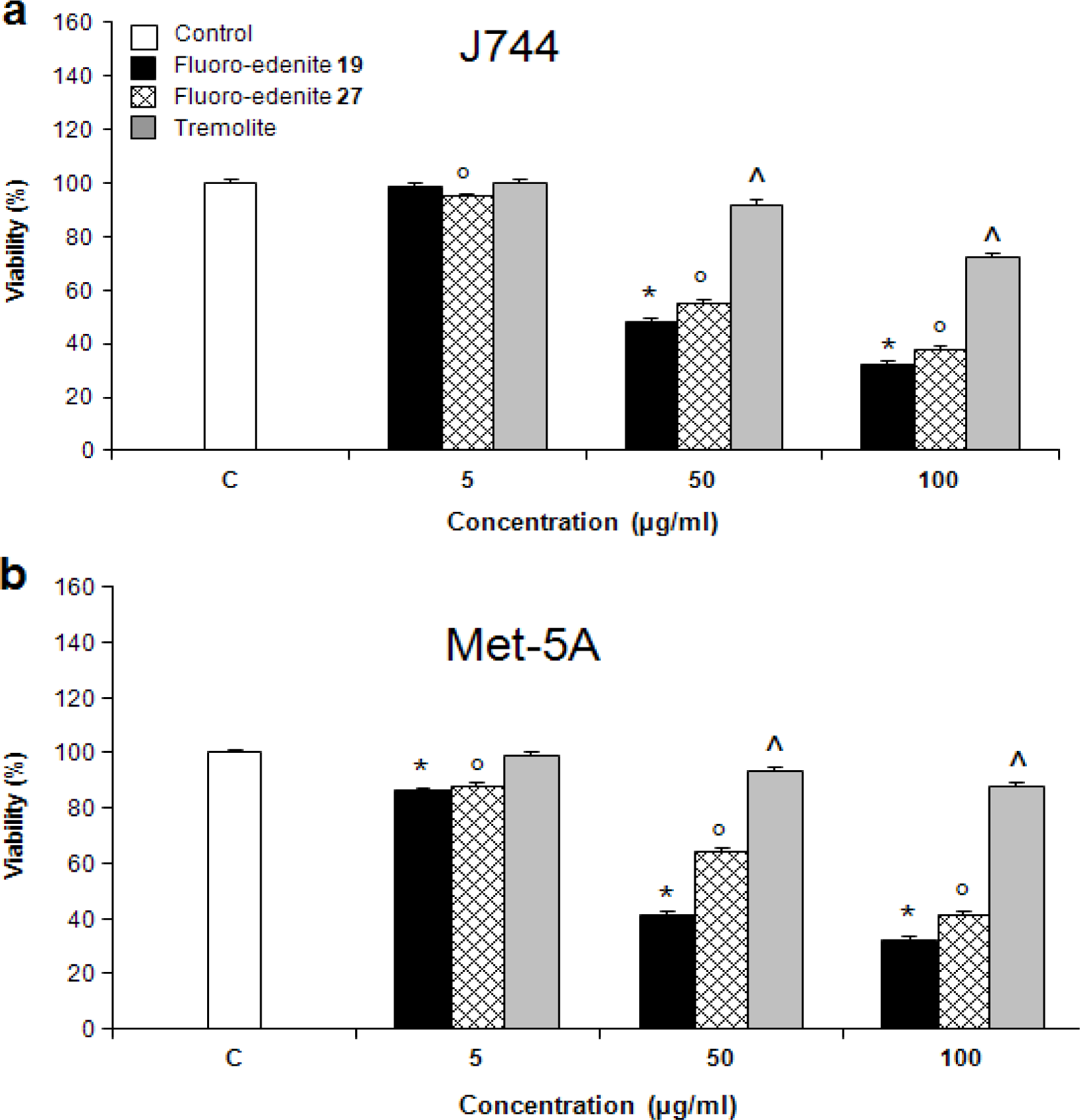
Results
Discussion
List of Abbreviations
| DMEM | Dulbecco’s modified Eagle’s medium |
| FBS | Foetal bovine serum |
| Hsp 70 | Heat shock protein 70 kDalton |
| DCFH-DA | 2’7’-dichlorodihydrofluorescein diacetate |
| DMSO | Dimethylsulphoxide |
| LDH | Lactate dehydrogenase |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide test |
| NF-κB | Nuclear factor kappa B |
| PBS | Phosphate buffered saline |
| ROS, RNS | Reactive oxygen/nitrogen species |
| SEM-EDS | Scanning electron microscopy-energy dispersive spectrometer |
| TEM-EDS | Transmission electronic microscopy-energy dispersive spectrometer |
Acknowledgments
References
- Wegele, H; Muller, L; Buchner, J. Hsp70 and Hsp90--a relay team for protein folding. Rev. Physiol Biochem. Pharmacol. 2004, 151, 1–44. [Google Scholar]
- Milani, V; Noessner, E; Ghose, S; Kuppner, M; Ahrens, B; Scharner, A; Gastpar, R; Issels, RD. Heat shock protein 70: role in antigen presentation and immune stimulation. Int. J. Hyperthermia 2002, 18, 563–575. [Google Scholar]
- Multhoff, G. Activation of natural killer cells by heat shock protein 70. Int. J. Hyperthermia 2002, 18, 576–585. [Google Scholar]
- Wirth, D; Christians, E; Munaut, C; Dessy, C; Foidart, JM; Gustin, P. Differential heat shock gene hsp70-1 response to toxicants revealed by in vivo study of lungs in transgenic mice. Cell Stress. Chaperones 2002, 7, 387–395. [Google Scholar]
- Croute, F; Beau, B; Arrabit, C; Gaubin, Y; Delmas, F; Murat, JC; Soleilhavoup, JP. Pattern of stress protein expression in human lung cell-line A549 after short- or long-term exposure to cadmium. Environ. Health Perspect. 2000, 108, 55–60. [Google Scholar]
- Wong, HR; Wispe, JR. The stress response and the lung. Am. J. Physiol 1997, 273, L1–L9. [Google Scholar]
- Paoletti, L; Batisti, D; Bruno, C; Di Paola, M; Gianfagna, A; Mastrantonio, M; Nesti, M; Comba, P. Unusually high incidence of malignant pleural mesothelioma in a town of eastern Sicily: an epidemiological and environmental study. Arch. Environ. Health 2000, 55, 392–398. [Google Scholar]
- Comba, P; Gianfagna, A; Paoletti, L. Pleural mesothelioma cases in Biancavilla are related to a new fluoro-edenite fibrous amphibole. Arch. Environ. Health 2003, 58, 229–232. [Google Scholar]
- Pelin, K; Hirvonen, A; Taavitsainen, M; Linnainmaa, K. Cytogenetic response to asbestos fibers in cultured human primary mesothelial cells from 10 different donors. Mutat. Res. 1995, 334, 225–233. [Google Scholar]
- Fenoglio, I; Croce, A; Di Renzo, F; Tiozzo, R; Fubini, B. Pure-silica zeolites (Porosils) as model solids for the evaluation of the physicochemical features determining silica toxicity to macrophages. Chem Res. Toxicol. 2000, 13, 489–500. [Google Scholar]
- Rinaudo, C; Gastaldi, D; Belluso, E. Characterization of chrysotile, antigorite and lizardite by FT Raman spectroscopy. Can. Miner. 2003, 41, 883–890. [Google Scholar]
- Laemmli, UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar]
- Shen, HM; Shi, CY; Shen, Y; Ong, CN. Detection of elevated reactive oxygen species level in cultured rat hepatocytes treated with aflatoxin B1. Free Radic. Biol. Med. 1996, 21, 139–146. [Google Scholar]
- Green, LC; Wagner, DA; Glogowski, J; Skipper, PL; Wishnok, JS; Tannenbaum, SR. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar]
- Ding, AH; Nathan, CF; Stuehr, DJ. Release of reactive nitrogen intermediates and reactive oxygen intermediates from mouse peritoneal macrophages. Comparison of activating cytokines and evidence for independent production. J. Immunol. 1988, 141, 2407–2412. [Google Scholar]
- Renis, M; Cardile, V; Russo, A; Campisi, A; Collova, F. Glutamine synthetase activity and HSP70 levels in cultured rat astrocytes: effect of 1-octadecyl-2-methyl-rac-glycero-3-phosphocholine. Brain Res. 1998, 783, 143–150. [Google Scholar]
- Bradford, MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar]
- Mossman, BT; Bignon, J; Corn, M; Seaton, A; Gee, JB. Asbestos: scientific developments and implications for public policy. Science 1990, 247, 294–301. [Google Scholar]
- Barrett, JC. Cellular and molecular mechanisms of asbestos carcinogenicity: implications for biopersistence. Environ. Health Perspect. 1994, 102 Suppl 5, 19–23. [Google Scholar]
- Kane, AB. Mechanisms of mineral fibre carcinogenesis. IARC Sci. Publ. 1996, 11–34. [Google Scholar]
- Vallyathan, V; Shi, X. The role of oxygen free radicals in occupational and environmental lung diseases. Environ. Health Perspect. 1997, 105 Suppl 1, 165–177. [Google Scholar]
- Kamp, DW; Graceffa, P; Pryor, WA; Weitzman, SA. The role of free radicals in asbestos-induced diseases. Free Radic. Biol. Med. 1992, 12, 293–315. [Google Scholar]
- Hardy, JA; Aust, AE. The effect of iron binding on the ability of crocidolite asbestos to catalyze DNA single-strand breaks. Carcinogenesis 1995, 16, 319–325. [Google Scholar]
- Mehrotra, S; Kakkar, P; Viswanathan, PN. Mitochondrial damage by active oxygen species in vitro. Free Radic. Biol. Med. 1991, 10, 277–285. [Google Scholar]
- Fach, E; Waldman, WJ; Williams, M; Long, J; Meister, RK; Dutta, PK. Analysis of the biological and chemical reactivity of zeolite-based aluminosilicate fibers and particulates. Environ. Health Perspect. 2002, 110, 1087–1096. [Google Scholar]
- Pantopoulos, K; Weiss, G; Hentze, MW. Nitric oxide and the post-transcriptional control of cellular iron traffic. Trends Cell Biol. 1994, 4, 82–86. [Google Scholar]
- Weiss, G; Werner-Felmayer, G; Werner, ER; Grunewald, K; Wachter, H; Hentze, MW. Iron regulates nitric oxide synthase activity by controlling nuclear transcription. J. Exp. Med. 1994, 180, 969–976. [Google Scholar]
- Aldieri, E; Ghigo, D; Tomatis, M; Prandi, L; Fenoglio, I; Costamagna, C; Pescarmona, G; Bosia, A; Fubini, B. Iron inhibits the nitric oxide synthesis elicited by asbestos in murine macrophages. Free Radic. Biol. Med. 2001, 31, 412–417. [Google Scholar]
- Balazy, M; Poff, CD. Biological nitration of arachidonic acid. Curr. Vasc. Pharmacol. 2004, 2, 81–93. [Google Scholar]
- Wong, HR; Ryan, M; Menendez, IY; Denenberg, A; Wispe, JR. Heat shock protein induction protects human respiratory epithelium against nitric oxide-mediated cytotoxicity. Shock 1997, 8, 213–218. [Google Scholar]
- Wong, HR; Mannix, RJ; Rusnak, JM; Boota, A; Zar, H; Watkins, SC; Lazo, JS; Pitt, BR. The heat-shock response attenuates lipopolysaccharide-mediated apoptosis in cultured sheep pulmonary artery endothelial cells. Am. J. Respir. Cell Mol. Biol. 1996, 15, 745–751. [Google Scholar]
- Szabo, C; Wong, HR; Salzman, AL. Pre-exposure to heat shock inhibits peroxynitrite-induced activation of poly(ADP) ribosyltransferase and protects against peroxynitrite cytotoxicity in J774 macrophages. Eur. J. Pharmacol. 1996, 315, 221–226. [Google Scholar]
- Wang, YR; Xiao, XZ; Huang, SN; Luo, FJ; You, JL; Luo, H; Luo, ZY. Heat shock pretreatment prevents hydrogen peroxide injury of pulmonary endothelial cells and macrophages in culture. Shock 1996, 6, 134–141. [Google Scholar]
- Kim, HP; Morse, D; Choi, AM. Heat-shock proteins: new keys to the development of cytoprotective therapies. Expert. Opin. Ther. Targets 2006, 10, 759–769. [Google Scholar]
- Kregel, KC. Heat shock proteins: modifying factors in physiological stress responses and acquired thermotolerance. J. Appl. Physiol 2002, 92, 2177–2186. [Google Scholar]
- Wheeler, DS; Wong, HR. Heat shock response and acute lung injury. Free Radic. Biol. Med. 2007, 42, 1–14. [Google Scholar]
- Njemini, R; Lambert, M; Demanet, C; Mets, T. Elevated serum heat-shock protein 70 levels in patients with acute infection: use of an optimized enzyme-linked immunosorbent assay. Scand. J. Immunol. 2003, 58, 664–669. [Google Scholar]
© 2007 MDPI All rights reserved.
Share and Cite
Cardile, V.; Lombardo, L.; Belluso, E.; Panico, A.; Renis, M.; Gianfagna, A.; Balazy, M. Fluoro-edenite Fibers Induce Expression of Hsp70 and Inflammatory Response. Int. J. Environ. Res. Public Health 2007, 4, 195-202. https://doi.org/10.3390/ijerph2007030001
Cardile V, Lombardo L, Belluso E, Panico A, Renis M, Gianfagna A, Balazy M. Fluoro-edenite Fibers Induce Expression of Hsp70 and Inflammatory Response. International Journal of Environmental Research and Public Health. 2007; 4(3):195-202. https://doi.org/10.3390/ijerph2007030001
Chicago/Turabian StyleCardile, Venera, Laura Lombardo, Elena Belluso, Annamaria Panico, Marcella Renis, Antonio Gianfagna, and Michael Balazy. 2007. "Fluoro-edenite Fibers Induce Expression of Hsp70 and Inflammatory Response" International Journal of Environmental Research and Public Health 4, no. 3: 195-202. https://doi.org/10.3390/ijerph2007030001
APA StyleCardile, V., Lombardo, L., Belluso, E., Panico, A., Renis, M., Gianfagna, A., & Balazy, M. (2007). Fluoro-edenite Fibers Induce Expression of Hsp70 and Inflammatory Response. International Journal of Environmental Research and Public Health, 4(3), 195-202. https://doi.org/10.3390/ijerph2007030001



