Strategies for Monitoring Microbial Life in Beach Sand for Protection of Public Health
Abstract
1. Introduction
2. Sand Characteristics and Surrounding Environment
2.1. Types of Sand and Artificial Beaches
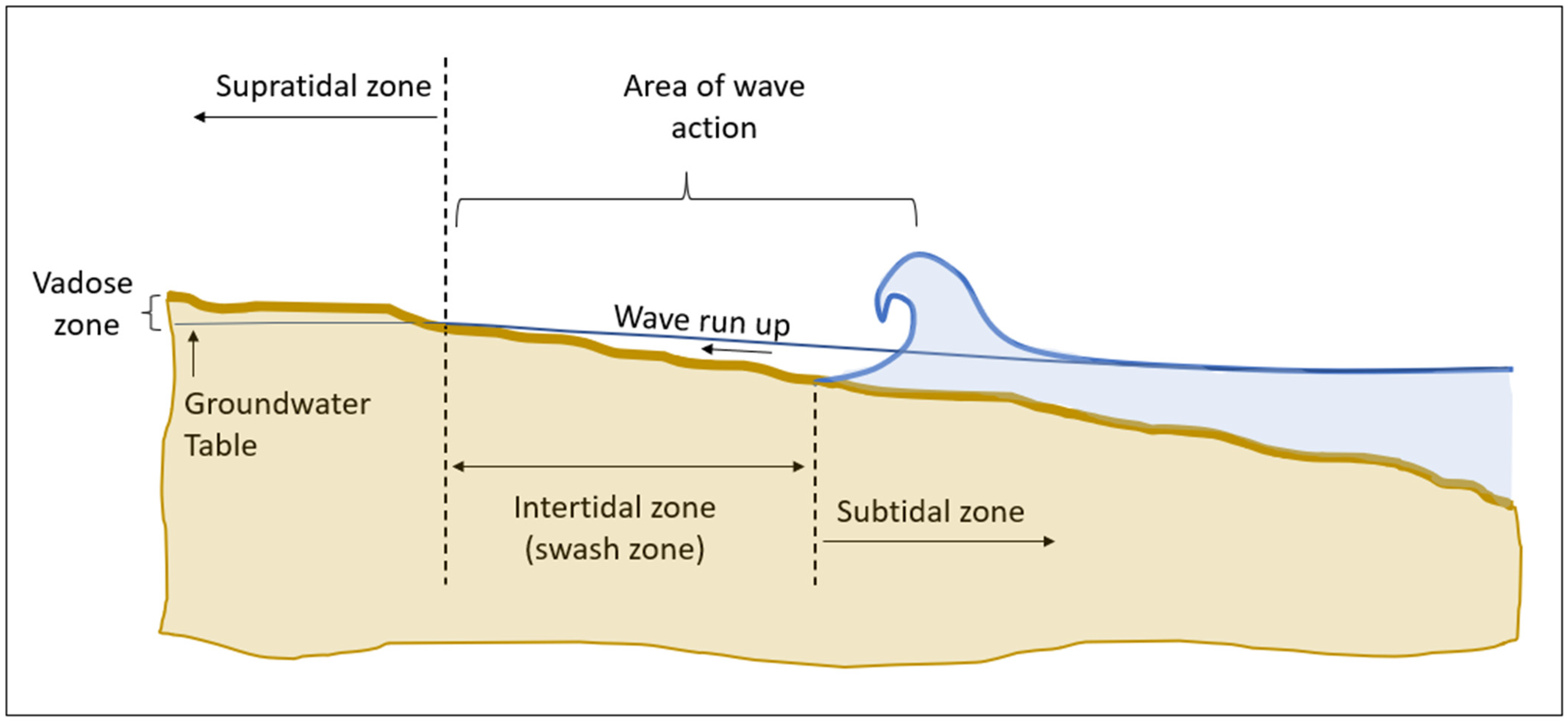
2.2. Swash Zone vs. Supratidal Zone
3. Microbial Parameters
3.1. History of Microbial Tracking in the Sand
3.2. Fecal Indicator Bacteria (FIB)
3.3. Other Bacteria
3.4. Fungi
4. Sampling Time and Sampling Methods
5. Analytical Approaches for Characterizing Microbial Communities of Beach Sand
5.1. Two Tiers Analytical Approach
5.1.1. Analysis during Outbreak Conditions
5.1.2. Quality Assessment Schemes
5.1.3. Outbreak Response
5.2. Microbial Source Tracking (MST)
5.3. Analyzing Microbes in Beach Sand
5.3.1. Fecal Indicator Bacteria (FIB)
5.3.2. Detection of Other Bacteria
5.3.3. Isolation of Fungi
5.3.4. Identification and Taxonomic Classification of Fungi
5.4. Analyzing Insects and Helminths in Beach Sand
6. Beach Sand Quality—Current Situation
7. The Way Forward
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Phillips, M.C.; Solo-Gabriele, H.M.; Piggot, A.M.; Klaus, J.S.; Zhang, Y. Relationships between sand and water quality at recreational beaches. Water Res. 2011, 45, 6763–6769. [Google Scholar] [CrossRef] [PubMed]
- Weiskerger, C.J.; Brandão, J.; Ahmed, W.; Aslan, A.; Avolio, L.; Badgley, B.D.; Boehm, A.B.; Edge, T.A.; Fleisher, J.M.; Heaney, C.D.; et al. Impacts of a changing earth on microbial dynamics and human health risks in the continuum between beach water and sand. Water Res. 2019, 162, 456–470. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines for Safe Recreational Water Environments: Coastal and Fresh Waters; World Health Organization: Geneva, Switzerland, 2003; Volume 1, ISBN 92 4 154580 1. [Google Scholar]
- Heaney, C.D.; Sams, E.; Dufour, A.P.; Brenner, K.P.; Haugland, R.A.; Chern, E.; Wing, S.; Marshall, S.; Love, D.C.; Serre, M.; et al. Fecal Indicators in Sand, Sand Contact, and Risk of Enteric Illness Among Beachgoers. Epidemiology 2012, 23, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, R.; Roll, B.; Byappanahalli, M. Appropriate recreational water quality standards for Hawaii and other tropical regions based on concentrations of Clostridium perfringens. In Proceedings of the Water Environment Federation’s 70th Annual Conference and Exposition, Chicago, IL, USA, 18–22 October 1997; Water Environment Federation: Alexandria, VA, USA, 1997; Volume 4, pp. 405–411. [Google Scholar]
- Fujioka, R.; Shizumura, L.K. Clostridium perfringens, a reliable indicator of stream water quality. J. Water Pollut. Control Fed. 1985, 57, 986–992. [Google Scholar]
- Fujioka, R.S.; Solo-Gabriele, H.M.; Byappanahalli, M.N.; Kirs, M.U.S. Recreational Water Quality Criteria: A Vision for the Future. Int. J. Environ. Res. Public Health 2015, 12, 7752–7776. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, P.; Salvador, D.; Brandão, J.; Ahmed, W.; Sadowsky, M.J.; Valério, E. Environmental and adaptive changes necessitate a paradigm shift for indicators of fecal contamination. Microbiol. Spectr. 2020, 8, 1–20. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2004; ISBN 978-92-4-154995-0. [Google Scholar]
- European Union. Directive 2006/7/EC of the European Parliament and of the Council of 15 February 2006 Concerning the Management of Bathing Water Quality and Repealing Directive 76/160/EEC. OJ L 64, 4.3.2006, 37–51. 2006. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32006L0007 (accessed on 28 April 2023).
- WHO. Guidelines for Recreational Water Quality; World Health Organization: Geneva, Switzerland, 2021; ISBN 978-92-4-003130-2. [Google Scholar]
- Micallef, A.; Williams, A. Beach Management: Principles and Practice; Routledge: Oxfordshire, UK, 2009; pp. 1–445. [Google Scholar] [CrossRef]
- Defeo, O.; McLachlan, A.; Schoeman, D.S.; Schlacher, T.A.; Dugan, J.; Jones, A.; Lastra, M.; Scapini, F. Threats to sandy beach ecosystems: A review. Estuar. Coast. Shelf Sci. 2009, 81, 1–12. [Google Scholar] [CrossRef]
- Piggot, A.M.; Klaus, J.S.; Johnson, S.; Phillips, M.C.; Solo-Gabriele, H.M. Relationship between enterococcal levels and sediment biofilms at recreational beaches in South Florida. Appl. Environ. Microbiol. 2012, 78, 5973–5982. [Google Scholar] [CrossRef]
- Abreu, R.; Figueira, C.; Romão, D.; Brandão, J.; Freitas, M.C.; Andrade, C.; Graça, C.; Ferreira, C.; Campos, A.; Prada, S. Sediment characteristics and microbiological contamination of beach sand—A case-study in the archipelago of Madeira. Sci. Total Environ. 2016, 573, 627–638. [Google Scholar] [CrossRef]
- Valério, E.; Santos, M.L.; Teixeira, P.; Matias, R.; Mendonça, J.; Ahmed, W.; Brandão, J. Microbial Source Tracking as a Method of Determination of Beach Sand Contamination. Int. J. Environ. Res. Public Health 2022, 19, 7934. [Google Scholar] [CrossRef]
- Hernandez, R.J.; Hernandez, Y.; Jimenez, N.H.; Piggot, A.M.; Klaus, J.S.; Feng, Z.; Reniers, A.; Solo-Gabriele, H.M. Effect of full-scale beach renovation on fecal indicator levels in shoreline sand and water. Water Res. 2014, 48, 579–591. [Google Scholar] [CrossRef]
- Bitan, M.; Zviely, D. Sand Beach Nourishment: Experience from the Mediterranean Coast of Israel. J. Mar. Sci. Eng. 2020, 8, 273. [Google Scholar] [CrossRef]
- Solo-Gabriele, H.M.; Harwood, V.J.; Kay, D.; Fujioka, R.S.; Sadowsky, M.J.; Whitman, R.L.; Wither, A.; Caniça, M.; Carvalho da Fonseca, R.; Duarte, A.; et al. Beach sand and the potential for infectious disease transmission: Observations and recommendations. J. Mar. Biol. Assoc. UK 2016, 96, 101–120. [Google Scholar] [CrossRef]
- Outro Planeta. “Areal da Calheta” Last Modified 10 August 2004. 2004. Available online: http://outroplaneta.blogspot.com/2004/08/areal-da-calheta.html (accessed on 28 April 2023).
- Romão, D.; Staley, C.; Ferreira, F.; Rodrigues, R.; Sabino, R.; Veríssimo, C.; Wang, P.; Sadowsky, M.; Brandão, J. Next-generation sequencing and culture-based techniques offer complementary insights into fungi and prokaryotes in beach sands. Mar. Pollut. Bull. 2017, 119, 351–358. [Google Scholar] [CrossRef]
- Whitman, R.L.; Harwood, V.J.; Edge, T.A.; Nevers, M.B.; Byappanahalli, M.; Vijayavel, K.; Brandão, J.; Sadowsky, M.J.; Alm, E.W.; Crowe, A.; et al. Microbes in beach sands: Integrating environment, ecology and public health. Rev. Environ. Sci. Biotechnol. 2014, 13, 329–368. [Google Scholar] [CrossRef] [PubMed]
- Sahwell, P.J.; Abdool-Ghany, A.; Klaus, J.; Gaston, C.J.; Solo-Gabriele, H.M. Sampling method comparison of enterococci aerosolization during continuous bubble bursting generation. FEMS Microbiol. Lett. 2021, 368, fnac003. [Google Scholar] [CrossRef] [PubMed]
- Abdool-Ghany, A.A.; Sahwell, P.J.; Klaus, J.; Gidley, M.L.; Sinigalliano, C.D.; Solo-Gabriele, H.M. Fecal indicator bacteria levels at a marine beach before, during, and after the COVID-19 shutdown period and associations with decomposing seaweed and human presence. Sci. Total Environ. 2022, 851 Pt 2, 158349. [Google Scholar] [CrossRef] [PubMed]
- Brandão, J.; Weiskerger, C.; Valério, E.; Pitkänen, T.; Meriläinen, P.; Avolio, L.; Heaney, C.D.; Sadowsky, M.J. Climate Change Impacts on Microbiota in Beach Sand and Water: Looking Ahead. Int. J. Environ. Res. Public Health 2022, 19, 1444. [Google Scholar] [CrossRef]
- Macreadie, I.G.; Johnson, G.; Schlosser, T.; Macreadie, P.I. Growth inhibition of Candida species and Aspergillus fumigatus by statins. FEMS Microbiol. Lett. 2006, 262, 9–13. [Google Scholar] [CrossRef]
- Cornforth, D.M.; Foster, K.R. Antibiotics and the art of bacterial war. Proc. Natl. Acad. Sci. USA 2015, 112, 10827–10828. [Google Scholar] [CrossRef]
- Brandão, J.; Albergaria, I.; Albuquerque, J.; José, S.; Grossinho, J.; Ferreira, F.C.; Raposo, A.; Rodrigues, R.; Silva, C.; Jordao, L.; et al. Untreated sewage contamination of beach sand from a leaking underground sewage system—An episode of skin rash was experienced by 30 people at a beach. Sci. Total Environ. 2020, 740, 140237. [Google Scholar] [CrossRef]
- Brandão, J. Microorganisms in Beach Sand: What Do We Still Not Know? In Encyclopedia of Environmental Health; Nriagu, J., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 4, pp. 390–392. [Google Scholar] [CrossRef]
- Kelly, R. Stormwater Conference. 2018. Available online: https://www.waternz.org.nz/Attachment?Action=Download&Attachment_id=3194 (accessed on 28 April 2023).
- Novak Babič, M.; Zupančič, J.; Gunde-Cimerman, N.; de Hoog, S.; Zalar, P. Ecology of the Human Opportunistic Black Yeast Exophiala dermatitidis Indicates Preference for Human-Made Habitats. Mycopathologia 2018, 183, 201–212. [Google Scholar] [CrossRef]
- Schönfeld, J.; Rieth, H.; Thianprasit, M. Experimenteller Beitrag zur Dermatophytenflora des Ostsee-Badestrandes. Arch. Klin. Exp. Derm. 1960, 212, 78–87. [Google Scholar] [CrossRef]
- Müller, G. Occurrence of dermatophytes in the sands of European beaches. Sci. Total Environ. 1973, 2, 116–118. [Google Scholar] [CrossRef]
- Valiente, M.F.C. Micological Study of the Sand of the Beaches of the City of Alicante. Ph.D. Thesis, University of Alicante, Alicante, Spain, 1990. [Google Scholar]
- Sousa, M.L.R. Micoses. [Fungi.] Lisbon, National Institute of Health (INSA), Centre of Epidemiologic Surveillance of Transmissible Diseases. Epidemiol. Bull. 1990. [Google Scholar]
- Brandão, J.; Veríssimo, C.; Rosado, M.L.; Falcão, M.L.; Giraldes, A.; Rosado, C.; Simões, M.; Noronha, M.G. Qualidade Microbiológica de Areias de Praias Litorais, Relatório Final; Agência Portuguesa do Ambiente, I.P., Ed.; Associação Bandeira Azul da Europa (ABAE): Oeiras, Portugal, 2002. [Google Scholar] [CrossRef]
- Zhan, P.; Liu, W. The changing face of dermatophytic infections worldwide. Mycopathologia 2017, 182, 77–86. [Google Scholar] [CrossRef]
- Oshiro, R.; Fujioka, R. Soil, and Pigeon Droppings: Sources of Indicator Bacteria in the Waters of Hanauma Bay, Oahu, Hawaii. Water Sci. Technol. 1995, 31, 251–254. [Google Scholar] [CrossRef]
- City and County of Honolulu. Hanauma Bay Information. Last Updated: 11 February. 2021. Available online: http://www.honolulu.gov/cms-dpr-menu/site-dpr-sitearticles/1704-hanauma-bay-information.html (accessed on 28 April 2023).
- Ambiental, D.d.S. Directrices Sanitarias para uso Seguro de Aguas Recreativas, Modulo II: Directrices Sanitarias para Enteropatogenos y Microorganismos Oportunistas en Agua Ambiente. Ministerio de Salud, Departamento de Salud Ambiental. 1–66. Argentina. Aprobado por COPERANCAUCH: 2017. Available online: https://www.argentina.gob.ar/sites/default/files/directrices_sanitarias_para_enteropatogenos.pdf (accessed on 28 April 2023).
- Ministry of Health of the Republic of Lithuania. Lithuanian Hygiene Standard hn 92: 2018 “Beaches and Their Quality of Bathing Water”. Consolidated Version from 01.08.2018 to 25/07/2019. Identification Code 1072250ISAK00V-1055. 2017. Available online: https://e-seimas.lrs.lt/portal/legalAct/lt/TAD/TAIS.311976/wTyZKKekTr (accessed on 28 April 2023).
- Abdelzaher, A.M.; Wright, M.E.; Ortega, C.; Solo-Gabriele, H.M.; Miller, G.; Elmir, S.; Newman, X.; Shih, P.; Bonilla, J.A.; Bonilla, T.D.; et al. Presence of pathogens and indicator microbes at a non-point source subtropical recreational marine beach. Appl. Environ. Microbiol. 2010, 76, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Sabino, R.; Rodrigues, R.; Costa, I.; Carneiro, C.; Cunha, M.; Duarte, A.; Faria, N.; Ferreira, F.C.; Gargaté, M.J.; Júlio, C.; et al. Routine screening of harmful microorganisms in beach sands: Implications to public health. Sci. Total Environ. 2014, 472, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.I.Z.; Di Bari, M.; Lamparelli, C.C.; Truzzi, A.C.; Coelho, M.C.L.S.; Hachich, E.M. Sanitary quality of sands from marine recreational beaches of São Paulo, Brazil. Braz. J. Microbiol. 2005, 36, 321–326. [Google Scholar] [CrossRef]
- Alwakeel, S.S. Molecular identification of fungi isolated from coastal regions of Red Sea, Jeddah, Saudi Arabia. J. Assoc. Arab Univ. Basic Appl. Sci. 2017, 24, 115–119. [Google Scholar] [CrossRef]
- Mohiuddin, M.M.; Salama, Y.; Schellhorn, H.E.; Golding, G.B. Shotgun metagenomic sequencing reveals freshwater beach sands as reservoir of bacterial pathogens. Water Res. 2017, 115, 360–369. [Google Scholar] [CrossRef]
- Taylor, H.B.; Kurtz, H.D., Jr. Microbial community structure shows differing levels of temporal stability in intertidal beach sands of the grand strand region of South Carolina. PLoS ONE 2020, 15, e0229387. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, K.D.; McNay, M.; Cao, Y.P.; Ebentier, D.; Madison, M.; Griffith, J.F. A multi-beach study of Staphylococcus aureus, MRSA, and enterococci in seawater and beach sand. Water Res. 2012, 46, 4195–4207. [Google Scholar] [CrossRef]
- Escandón, P. Novel environmental niches for Candida auris: Isolation from a coastal habitat in Colombia. J. Fungi 2022, 8, 748. [Google Scholar] [CrossRef]
- Sabino, R.; Veríssimo, C.; Cunha, M.A.; Wergikoski, B.; Ferreira, F.C.; Rodrigues, R.; Parada, H.; Falcão, L.; Rosado, L.; Pinheiro, C.; et al. Pathogenic fungi: An unacknowledged risk at coastal resorts? New insights on microbiological sand quality in Portugal. Mar. Pollut. Bull. 2011, 62, 1506–1511. [Google Scholar] [CrossRef]
- Shah, A.H.; Abdelzaher, A.M.; Phillips, M.; Hernandez, R.; Solo-Gabriele, H.M.; Kish, J.; Scorzetti, G.; Fell, J.W.; Diaz, M.R.; Scott, T.M.; et al. Indicator microbes correlate with pathogenic bacteria, yeasts and helminthes in sand at a subtropical recreational beach site. J. Appl. Microbiol. 2011, 110, 1571–1583. [Google Scholar] [CrossRef] [PubMed]
- Vogel, L.J.; O’Carroll, D.M.; Edge, T.A.; Robinson, C.E. Release of Escherichia coli from foreshore sand and pore water during intensified wave conditions at a recreational beach. Environ. Sci. Technol. 2016, 50, 5676–5684. [Google Scholar] [CrossRef]
- Ishii, S.; Hansen, D.L.; Hicks, R.E.; Sadowsky, M.J. Beach sand and sediments are temporal sinks and sources of Escherichia coli in Lake Superior. Environ. Sci. Technol. 2007, 41, 2203–2209. [Google Scholar] [CrossRef]
- Feng, Z.; Reniers, A.; Haus, B.K.; Solo-Gabriele, H.M. Modeling sediment related enterococci loading, transport and inactivation at an embayed, nonpoint source beach. Water Resour. Res. 2013, 49, 693–712. [Google Scholar] [CrossRef]
- Phillips, M.C.; Feng, Z.; Vogel, L.J.; Reniers, A.J.; Haus, B.K.; Enns, A.A.; Zhang, Y.; Hernandez, D.B.; Solo-Gabriele, H.M. Microbial release from seeded beach sediments during wave conditions. Mar. Pollut. Bull. 2014, 79, 114–122. [Google Scholar] [CrossRef]
- Monteiro, S.; Brandão, J.; Santos, R. Viruses in beach sand. In Proceedings of the 5th Food and Environmental Virology Conference, Kusatsu, Japan, 13–16 September 2016. [Google Scholar]
- WHO/UNICEF Joint Monitoring Programme for Water Supply and Sanitation. Progress on Drinking Water and Sanitation: 2012 Update; World Health Organisation: Geneva, Switzerland, 2012; ISBN 978 92 806 4632 0. [Google Scholar]
- Gelting, R.; Bliss, K.; Patrick, M.; Lockhart, G.; Handzel, T. Water, sanitation and hygiene in Haiti: Past, present, and future. Am. J. Trop. Med. Hyg. 2013, 89, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Abdool-Ghany, A.A.; Klaus, J.S.; Sosa Villegas, L.E.; D’Alessio, T.; Gidley, M.L.; Sinigalliano, C.D.; Gaston, C.; Solo-Gabriele, H.M. Microbial Communities in the Water Surface Microlayer and Potential Associations with Microbes in Aerosols, Sediment and Bulk Water. FEMS Microbiol. Ecol. 2023, 97, fiad039. [Google Scholar] [CrossRef]
- Goodwin, K.D.; Pobuda, M. Performance of CHROMagar Staph aureus and CHROMagar MRSA for detection of Staphylococcus aureus in seawater and beach sand--comparison of culture, agglutination, and molecular analyses. Water Res. 2009, 43, 4802–4811. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, S.J.; Wylam, M.E. Methicillin-Resistant Staphylococcus aureus Infection and Treatment Options. Methods Mol. Biol. 2020, 2069, 229–251. [Google Scholar]
- Plano, L.R.W.; Shibata, T.; Garza, A.C.; Kish, J.; Fleisher, J.; Sinigalliano, C.D.; Gidley, M.L.; Withum, K.; Elmir, S.M.; Hower, S.; et al. Human-Associated Methicillin-Resistant Staphylococcus aureus from a Subtropical Recreational Marine Beach. Microb. Ecol. 2013, 65, 1039–1051. [Google Scholar] [CrossRef] [PubMed]
- Akanbi, O.E.; Njom, H.A.; Fri, J.; Otigbu, A.C.; Clarke, A.M. Antimicrobial Susceptibility of Staphylococcus aureus Isolated from Recreational Waters and Beach Sand in Eastern Cape Province of South Africa. Int. J. Environ. Res. Public Health 2017, 14, 1001. [Google Scholar] [CrossRef]
- Hainer, B.L. Dermatophyte infections. Am. Fam. Physician 2003, 67, 101–108. [Google Scholar]
- Brandão, J.; Gangneux, J.P.; Arikan-Akdagli, S.; Barac, A.; Bostanaru, A.C.; Brito, S.; Bull, M.; Çerikçioğlu, N.; Chapman, B.; Efstratiou, M.A.; et al. Mycosands: Fungal diversity and abundance in beach sand and recreational waters—Relevance to human health. Sci. Total Environ. 2021, 781, 146598. [Google Scholar] [CrossRef]
- Anderson, J.H. In vitro survival of human pathogenic fungi in Hawaiian beach sand. Sabouraudia 1979, 17, 13–22. [Google Scholar] [CrossRef]
- Salzer, H.; Burchard, G.; Cornely, O.A.; Lange, C.; Rolling, T.; Schmiedel, S.; Libman, M.; Capone, D.; Le, T.; Dalcolmo, M.P.; et al. Diagnosis and Management of Systemic Endemic Mycoses Causing Pulmonary Disease. Respiration 2018, 96, 283–301. [Google Scholar] [CrossRef] [PubMed]
- Applen Clancey, S.; Ciccone, E.J.; Coelho, M.A.; Davis, J.; Ding, L.; Betancourt, R.; Glaubiger, S.; Lee, Y.; Holland, S.M.; Gilligan, P.; et al. Cryptococcus deuterogattii VGIIa Infection Associated with Travel to the Pacific Northwest Outbreak Region in an Anti-Granulocyte-Macrophage Colony-Stimulating Factor Autoantibody-Positive Patient in the United States. MBio 2019, 10, e02733-18. [Google Scholar] [CrossRef] [PubMed]
- Neblett Fanfair, R.; Benedict, K.; Bos, J.; Bennett, S.D.; Lo, Y.C.; Adebanjo, T.; Etienne, K.; Deak, E.; Derado, G.; Shieh, W.J.; et al. Necrotizing cutaneous mucormycosis after a tornado in Joplin, Missouri, in 2011. N. Engl. J. Med. 2012, 367, 2214–2225. [Google Scholar] [CrossRef]
- Sympardi, S.; Drogari-Apiranthitou, M.; Zervakakis, I.; Papakonstantinou, E.; Liapi, G.; Arvaniti, A.; Karaiskou, A.; Giannopoulou, P.; Eleftheria, E. A mucormycosis case during the catastrophic flood in Mandra, Greece, November 2017. In Proceedings of the 9th Trends in Medical Mycology, Nice, France, 11–14 October 2019; Available online: https://www.researchgate.net/publication/338684660_A_mucormycosis_case_during_the_catastrophic_flood_in_Mandra_Attica_Greece_November_2017 (accessed on 28 April 2023).
- Cogliati, M.; Arikan-Akdagli, S.; Barac, A.; Bostanaru, A.C.; Brito, S.; Çerikçioğlu, N.; Efstratiou, M.A.; Ergin, Ç.; Esposto, M.C.; Frenkel, M.; et al. Environmental and bioclimatic factors influencing yeasts and molds distribution along European shores. Sci. Total Environ. 2023, 859 Pt 1, 160132. [Google Scholar] [CrossRef] [PubMed]
- Kellogg, C.A.; Griffin, D.W. African Dust Carries Microbes Across the Ocean: Are They Affecting Human and Ecosystem Health? USGS Open-File Report 03-028. 2003. Available online: https://pubs.usgs.gov/of/2003/0028/report.pdf (accessed on 28 April 2023).
- Buskirk, A.D.; Green, B.J.; Lemons, A.R.; Nayak, A.P.; Goldsmith, W.T.; Kashon, M.L.; Anderson, S.E.; Hettick, J.M.; Templeton, S.P.; Germolec, D.R.; et al. A murine inhalation model to characterize pulmonary exposure to dry Aspergillus fumigatus conidia. PLoS ONE 2014, 9, e109855. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, R.J.; Boon, N.J.; Vrcelj, K.; Nguyen, A.; Vinci, C.; Armstrong-James, D.; Bignell, E. In silico modeling of spore inhalation reveals fungal persistence following low dose exposure. Sci. Rep. 2015, 5, 13958. [Google Scholar] [CrossRef]
- Cho, S.H.; Seo, S.C.; Schmechel, D.; Grinshpun, S.A.; Reponen, T. Aerodynamic characteristics and respiratory deposition of fungal fragments. Atmos. Environ. 2005, 39, 5454–5465. [Google Scholar] [CrossRef]
- Wålinder, R.; Ernstgård, L.; Johanson, G.; Norbäck, D.; Venge, P.; Wieslander, G. Acute effects of a fungal volatile compound. Environ. Health Perspect. 2005, 113, 1775–1778. [Google Scholar] [CrossRef]
- Aleksic, B.; Draghi, M.; Ritoux, S.; Bailly, S.; Lacroix, M.; Oswald, I.P.; Bailly, J.D.; Robine, E. Aerosolization of Mycotoxins after Growth of Toxinogenic Fungi on Wallpaper. Appl. Environ. Microbiol. 2017, 83, e01001-17. [Google Scholar] [CrossRef]
- de Hoog, G.S.; Guarro, J.; Gené, J.; Ahmed, S.; Al-Hatmi, A.M.S.; Figueras, M.J.; Al-Hatmi, A.M.S.; Figueras, M.J.; Vitale, R.G. Atlas of Clinical Fungi, 3rd ed.; Reus: Utrecht, The Netherlands, 2019; ISBN 9070351439. [Google Scholar]
- Revankar, S.G.; Sutton, D.A. Melanized fungi in human disease. Clin. Microbiol. Rev. 2010, 23, 884–928. [Google Scholar] [CrossRef]
- Irinyi, L.; Roper, M.; Malik, R.; Meyer, W. In Silico Environmental Sampling of Emerging Fungal Pathogens via Big Data Analysis. Fungal Ecol. 2023, 62, 101212. Available online: https://www.sciencedirect.com/science/article/pii/S1754504822000733?via%3Dihub (accessed on 28 April 2023). [CrossRef]
- Steffen, H.C.; Smith, K.; van Deventer, C.; Weiskerger, C.; Bosch, C.; Brandão, J.; Wolfaardt, G.; Botha, A. Health risk posed by direct ingestion of yeasts from polluted river water. Water Res. 2023, 231, 119599. [Google Scholar] [CrossRef] [PubMed]
- Brandão, J.; Harwood, V.J. Chapter 24—Sand and Soil Sampling. In Environmental Mycology in Public Health; Academic Press: Cambridge, MA, USA, 2016; pp. 411–413. [Google Scholar] [CrossRef]
- Vogel, L.J.; Edge, T.A.; O’Carroll, D.M.; Solo-Gabriele, H.M.; Kushnir, C.; Robinson, C.E. Evaluation of methods to sample fecal indicator bacteria in foreshore sand and pore water at freshwater beaches. Water Res. 2017, 121, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Brewer, R.; Peard, J.; Heskett, M. A critical review of discrete soil sample data reliability: Part 1d—Field study results. Soil Sediment Contam. Int. J. 2016, 1, 1–22. [Google Scholar] [CrossRef]
- Hadley, P.W.; Petrisor, I.G. Incremental Sampling: Challenges and Opportunities for Environmental Forensics. Environ. Forens. 2013, 14, 109–120. [Google Scholar] [CrossRef]
- ISO Standard No. 13528; Statistical Methods for Use in Proficiency Testing by Interlaboratory Comparison. International Organization for Standardization: Geneva, Switzerland, 2015. Available online: https://www.iso.org/standard/56125.html (accessed on 28 April 2023).
- Krzywanski, J.; Kuchar, E.; Mierzynski, R. Cutaneous larva migrans in a beach volleyball player. IDCases 2021, 24, e01084. [Google Scholar] [CrossRef]
- Weatherhead, J.E.; Hotez, P.J.; Mejia, R. The Global State of Helminth Control and Elimination in Children. Pediatr. Clin. N. Am. 2017, 64, 867–877. [Google Scholar] [CrossRef]
- Boehm, A.B.; Griffith, J.; McGee, C.; Edge, T.A.; Solo-Gabriele, H.M.; Whitman, R.; Cao, Y.; Getrich, M.; Jay, J.A.; Ferguson, D.; et al. Faecal indicator bacteria enumeration in beach sand: A comparison study of extraction methods in medium to coarse sands. Journal of applied microbiology. J. Appl. Microbiol. 2009, 107, 1740–1750. [Google Scholar] [CrossRef]
- Bernhard, A.E.; Field, K.G. Identification of nonpoint sources of fecal pollution in coastal waters by using host-specific 16S ribosomal DNA genetic markers from fecal anaerobes. Appl. Environ. Microbiol. 2000, 66, 1587–1594. [Google Scholar] [CrossRef]
- Teaf, C.M.; Flores, D.; Garber, M.; Harwood, V.J. Toward Forensic Uses of Microbial Source Tracking. Microbiol. Spectr. 2018, 6, 1–14. [Google Scholar] [CrossRef]
- Green, H.C.; Haugland, R.A.; Varma, M.; Millen, H.T.; Borchardt, M.A.; Field, K.G.; Walters, W.A.; Knight, R.; Sivaganesan, M.; Kelty, C.A.; et al. Improved HF183 quantitative real-time PCR assay for characterization of human fecal pollution in ambient surface water samples. Appl. Environ. Microbiol. 2014, 80, 3086–3094. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Domingo, J.W.S.; Lamendella, R.; Edge, T.; Hill, S. Phylogenetic Diversity and Molecular Detection of Bacteria in Gull Feces. Appl. Environ. Microbiol. 2008, 74, 3969–3976. [Google Scholar] [CrossRef]
- Shanks, O.C.; Atikovic, E.; Blackwood, A.D.; Lu, J.; Noble, R.T.; Domingo, J.S.; Seifring, S.; Sivaganesan, M.; Haugland, R.A. Quantitative PCR for detection and enumeration of genetic markers of bovine fecal pollution. Appl. Environ. Microbiol. 2008, 74, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Hussein, K.; Waines, P.; Nisr, R.; Glegg, G.; Bradley, G. Development and use of Bacteroides 16S rRNA polymerase chain reaction assay for source tracking dog faecal pollution in bathing waters. Hydrol. Curr. Res. 2014, 5, 163. [Google Scholar] [CrossRef]
- Nevers, M.B.; Byappanahalli, M.N.; Nakatsu, C.H.; Kinzelman, J.L.; Phanikumar, M.S.; Shively, D.A.; Spoljaric, A. Interaction of bacterial communities and indicators of water quality in shoreline sand, sediment, and water of Lake Michigan. Water Res. 2020, 178, 115671. [Google Scholar] [CrossRef]
- Raza, S.; Kim, J.; Sadowsky, M.J.; Unno, T. Microbial source tracking using metagenomics and other new technologies. J. Microbiol. 2021, 59, 259–269. [Google Scholar] [CrossRef] [PubMed]
- ISO Standard No. 7899-2; Water Quality—Detection and Enumeration of Intestinal Enterococci—Part 2: Membrane Filtration Method. International Organization for Standardization: Geneva, Switzerland, 2000. Available online: https://www.iso.org/standard/14854.html (accessed on 28 April 2023).
- Denissen, J.; Reyneke, B.; Waso-Reyneke, M.; Havenga, B.; Barnard, T.; Khan, S.; Khan, W. Prevalence of ESKAPE pathogens in the environment: Antibiotic resistance status, community-acquired infection and risk to human health. Int. J. Hyg. Environ. Health 2022, 244, 114006. [Google Scholar] [CrossRef] [PubMed]
- Henson, O.E. Dichloran as an inhibitor of mold spreading in fungal plating media: Effects on colony diameter and enumeration. Appl. Environ. Microbiol. 1981, 42, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Shtienberg, D. Rhizopus head rot of confectionery sunflower: Effects on yield quantity and quality and implications for disease management. Phytopathology 1997, 87, 1226–1232. [Google Scholar] [CrossRef]
- Joseph, J. Commentary: Dematiaceous fungal keratitis: Importance of ocular microbiology. Indian J. Ophthalmol. 2019, 67, 1053–1054. [Google Scholar] [CrossRef]
- Harman, G.; Howell, C.; Viterbo, A.; Chat, I.; Lorito, M. Trichoderma species—Opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol. 2004, 2, 43–56. [Google Scholar] [CrossRef]
- Endo, A.; Monacloin, K. A new hypochlesterolemic agent produced by a monascus species. J. Antibiot. 1979, 32, 853–854. [Google Scholar] [CrossRef] [PubMed]
- Franklin, R.B.; Garland, J.L.; Bolster, C.H.; Mills, A.L. Impact of dilution on microbial community structure and functional potential: Comparison of numerical simulations and batch culture experiments. Appl. Environ. Microbiol. 2001, 67, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Money, N.P. Against the naming of fungi. Fungal Biol. 2013, 117, 463–465. [Google Scholar] [CrossRef] [PubMed]
- Novak Babič, M.; Gunde-Cimerman, N.; Breskvar, M.; Džeroski, S.; Brandão, J. Occurrence, Diversity and Anti-Fungal Resistance of Fungi in Sand of an Urban Beach in Slovenia-Environmental Monitoring with Possible Health Risk Implications. J. Fungi 2022, 8, 860. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control; Versteirt, V.; Schaffner, F.; Medlock, J. Guidelines for the Surveillance of Native Mosquitoes in Europe; European Centre for Disease Prevention and Control: Solna, Sweden, 2015. [Google Scholar] [CrossRef]
- Zimmer, J.-Y.; Haubruge, E.; Francis, F. Synthèse bibliographique: L’écologie larvaire des culicoïdes (Diptera: Ceratopogonidae). Biotechnol. Agron. Soc. 2014, 18, 301–312. [Google Scholar]
- EMCA. Guidelines for the Control of Mosquitoes of Public Health Importance in Europe. 2011. Available online: http://www.emca-online.eu/assets/PDFs/EMCA_guidelines_Speyer_2011.pdf (accessed on 28 April 2023).
- Leonard, A.; Zhang, L.; Balfour, A.J.; Garside, R.; Hawkey, P.M.; Murray, A.K.; Ukoumunne, O.C.; Gaze, W.H. Exposure to and colonisation by antibiotic-resistant E. coli in UK coastal water users: Environmental surveillance, exposure assessment, and epidemiological study (Beach Bum Survey). Environ. Int. 2018, 114, 326–333. [Google Scholar] [CrossRef]
| When the sand quality is documented as poor and action needs to be taken, chlorination is the most used approach. Biocides, if needed, do not go beyond the superficial sand layers, unless a dispersing agent is added [22]. If too polluted, sand may be replaced as a remediation approach [29]. |
| Implementation of covered garbage receptacles that are animal-proof and protected from precipitation, in designated areas. |
| Proper solid waste management to prevent non-native bird usage at the beach. |
| Toilet facilities that can minimise contamination impacts from human faecal matter and encourage proper personal hygiene at the beach. |
| Appropriate design and implementation of drainage systems for beach areas so that drainage from parking lots and backshore areas cannot flow into the nearshore beach zone [30]. |
| Re-direction of stormwater drainage from nearby communities away from the beach, combined with limited recreational access to downstream areas if stormwater does reach the beach area, to minimise exposure to beachgoers. |
| Cases Reported | Details |
|---|---|
| Staphylococcus aureus and MRSA Increased persistence in California, USA [48] | Found in 2.7% of the samples |
| Candida spp. including C. auris. increased presence with decreased precipitation (predominantly found in dry sands) in Portugal, Florida, USA and Colombia [49,50,51] | “85 of 495 samples were positive for Candida spp.” [49], “C. auris isolated from water and harsh wetlands” [50]; “Association between indicators and pathogens; C. tropicalis was the most frequent Candida spp. isolated from dry sand with an average count of 34.3 CFU/g of dry sand, followed by C. parapsilosis with 1.6 CFU/g, C. glabrata with 1.2 CFU/g and C. guilliermondii with 0.6 CFU/g” [51]. |
| E. coli elevated numbers in water correlated with increased wave height in Lake Huron, Canada [52] and associated with wider dispersal of microbes and water releases into Lake Superior, USA [53] | “E. coli concentrations in the surface (unsaturated) sand 1 m landward of the initial shoreline (P1) were 1.23 ± 0.99 log CFU/g” [52]; “When E. coli concentrations in sand and sediment samples were converted to CFU per interstitial water, the greatest numbers of E. coli were observed in nearshore and upshore sands, followed by shoreline sands and sediment. These numbers were, on average, 63, 74, 1087, and 4982 times greater in sediment, shoreline, nearshore, and upshore sand samples, respectively, than the concentration of E. coli in lake water expressed as CFU/mL” [53]. |
| Enterococci in mobilized sand caused a spike in water contamination with increasing wave action in Florida, USA (experimental, laboratory conditions) [54,55] | “The time duration for a certain percent die-off is inversely proportional to the solar radiation intensity. It takes 2.2 h to deactivate 90% of the total enterococci at noon time with a near maximum solar insolation of 800 W m−2“ [54]; “For trials with waves, analysis of the top and bottom layers of sediment in the ‘‘final’’ seeded sand revealed that more enterococci were removed from the top layer (78% removal and standard deviation of 17%) as compared to the bottom layer (58% removal and standard deviation of 28%) (p = 0.05, average difference between top and bottom layers = 194 CFU/g dry sand)” [55]. |
| Helminths found in Lithuanian beach sand [41] | “The soil must not be contaminated with helminths, enteroviruses, pathogenic enterobacteria, intestinal rods.” |
| Hepatitis A virus (HAV) and Human Adenovirus (HadV) genetic material found in beach sand with qPCR, although viability of viral particles was not tested [56] | “HAV were detected in 6.25% (3 out of 48) and HadV were detected in 8.33% (4 out of 48) samples using qPCR. These results corroborate the reporting of sand as an independent source of GI illness.” |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brandão, J.; Valério, E.; Weiskerger, C.; Veríssimo, C.; Sarioglou, K.; Novak Babič, M.; Solo-Gabriele, H.M.; Sabino, R.; Rebelo, M.T. Strategies for Monitoring Microbial Life in Beach Sand for Protection of Public Health. Int. J. Environ. Res. Public Health 2023, 20, 5710. https://doi.org/10.3390/ijerph20095710
Brandão J, Valério E, Weiskerger C, Veríssimo C, Sarioglou K, Novak Babič M, Solo-Gabriele HM, Sabino R, Rebelo MT. Strategies for Monitoring Microbial Life in Beach Sand for Protection of Public Health. International Journal of Environmental Research and Public Health. 2023; 20(9):5710. https://doi.org/10.3390/ijerph20095710
Chicago/Turabian StyleBrandão, João, Elisabete Valério, Chelsea Weiskerger, Cristina Veríssimo, Konstantina Sarioglou, Monika Novak Babič, Helena M. Solo-Gabriele, Raquel Sabino, and Maria Teresa Rebelo. 2023. "Strategies for Monitoring Microbial Life in Beach Sand for Protection of Public Health" International Journal of Environmental Research and Public Health 20, no. 9: 5710. https://doi.org/10.3390/ijerph20095710
APA StyleBrandão, J., Valério, E., Weiskerger, C., Veríssimo, C., Sarioglou, K., Novak Babič, M., Solo-Gabriele, H. M., Sabino, R., & Rebelo, M. T. (2023). Strategies for Monitoring Microbial Life in Beach Sand for Protection of Public Health. International Journal of Environmental Research and Public Health, 20(9), 5710. https://doi.org/10.3390/ijerph20095710










