Pyrrolizidine Alkaloids from Monofloral and Multifloral Italian Honey
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Chemicals and Standards
2.3. Materials
2.4. Working Solutions
2.5. Sample Preparation
2.6. Melissopalynological Analysis
2.7. Instrumentation
2.8. Quantification
2.9. Performance Evaluation
2.10. Statistical Analysis
2.11. Exposure Assessment and Risk Characterization
3. Results and Discussion
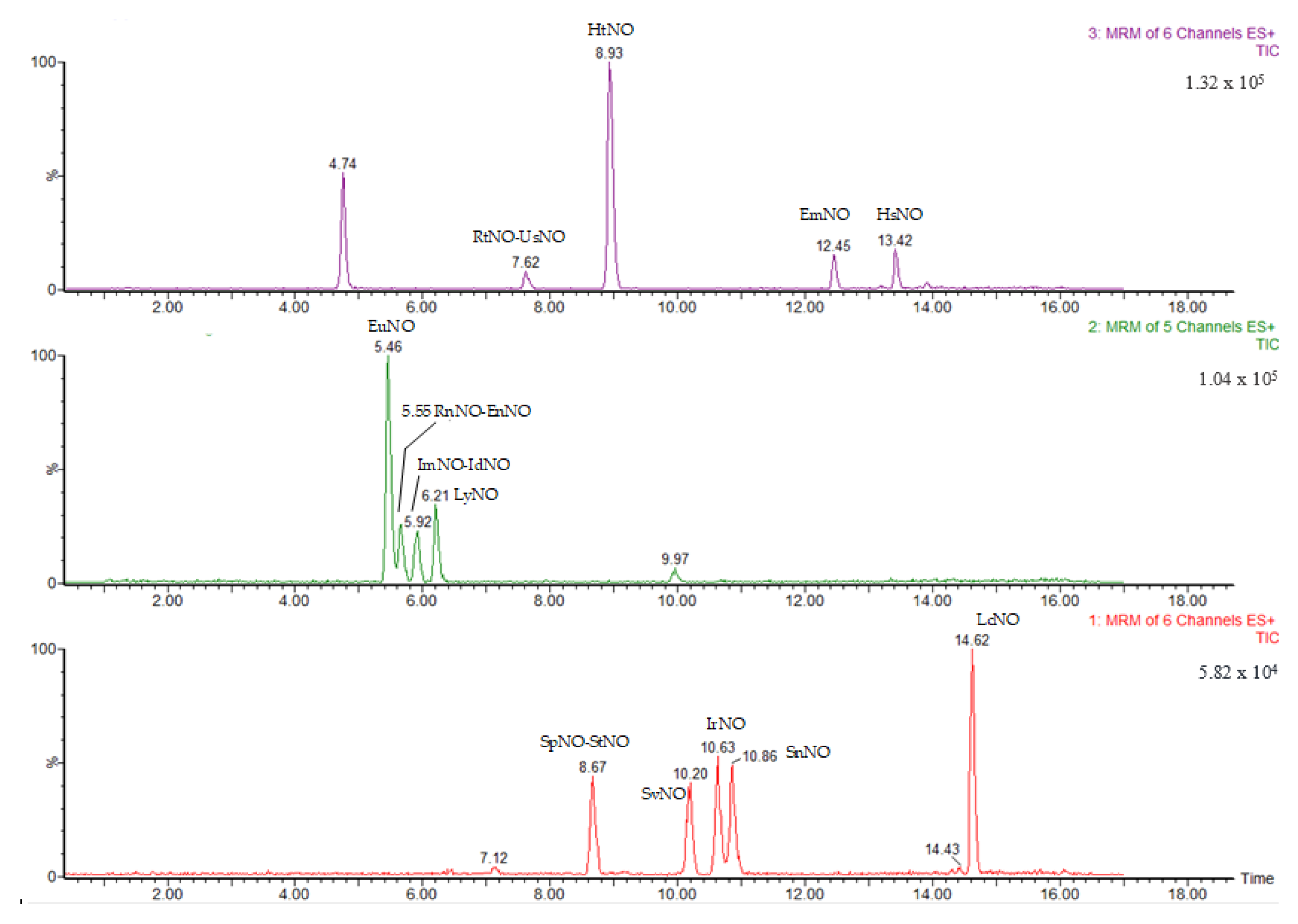
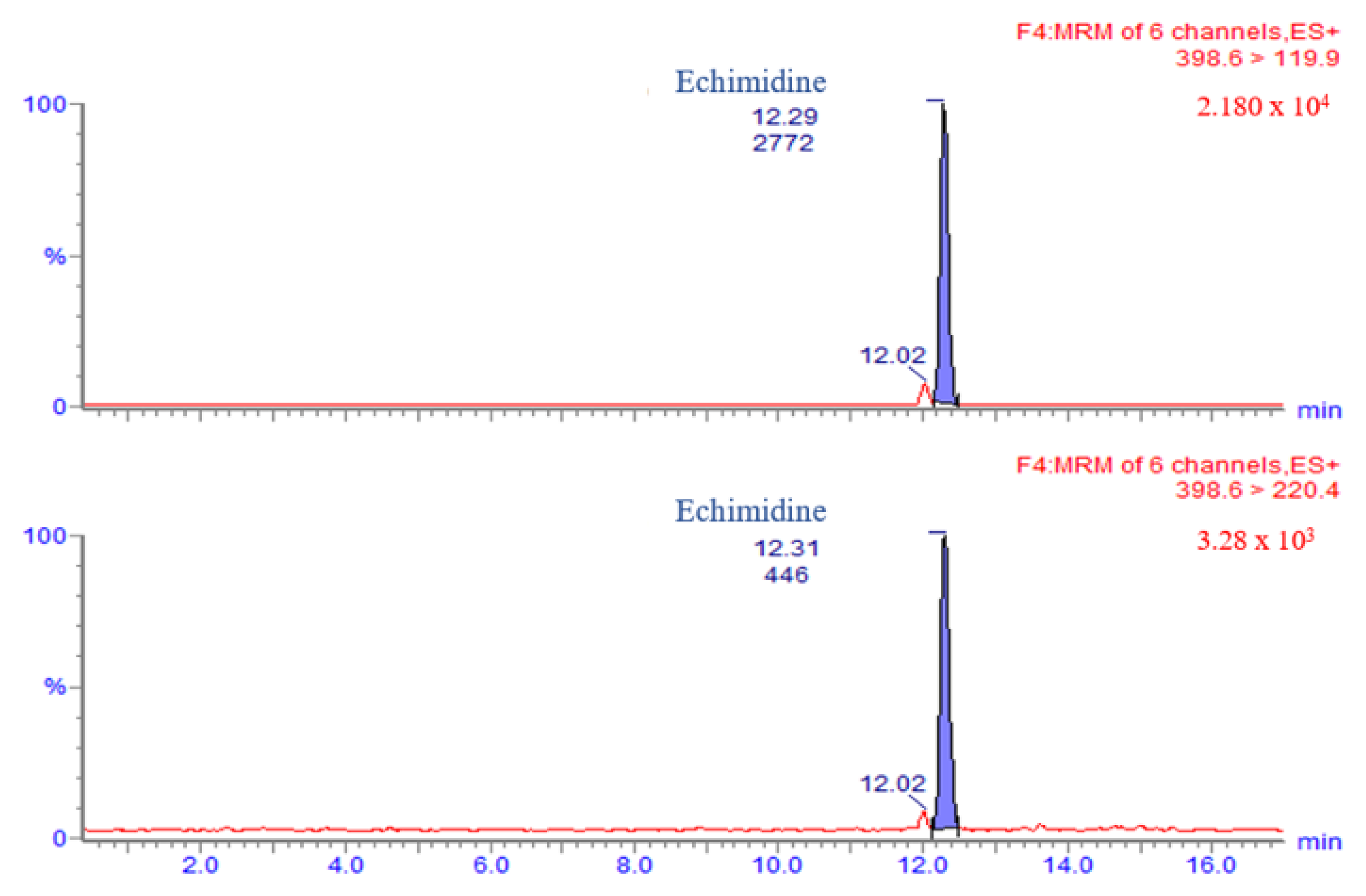
3.1. LC-MS/MS Method Validation
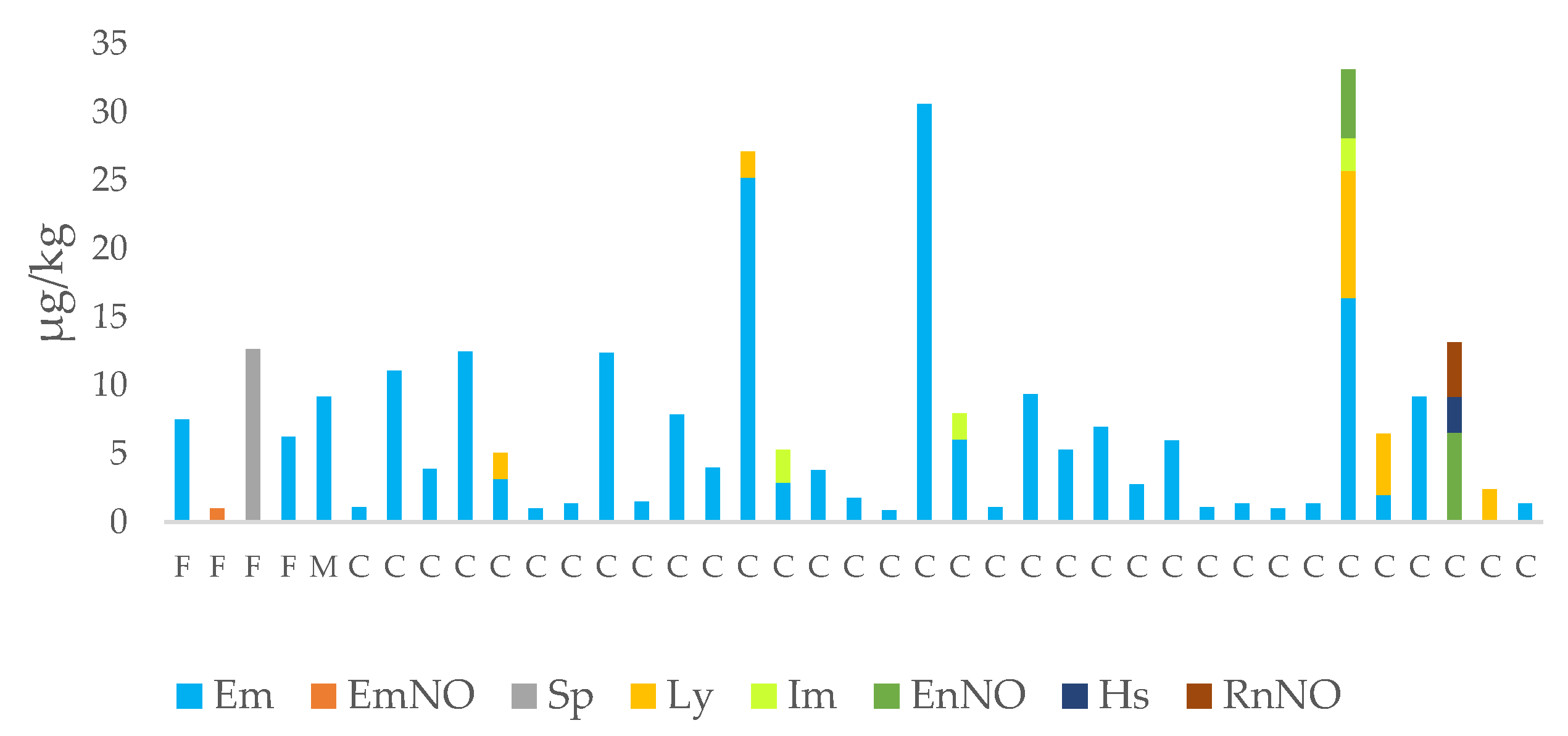
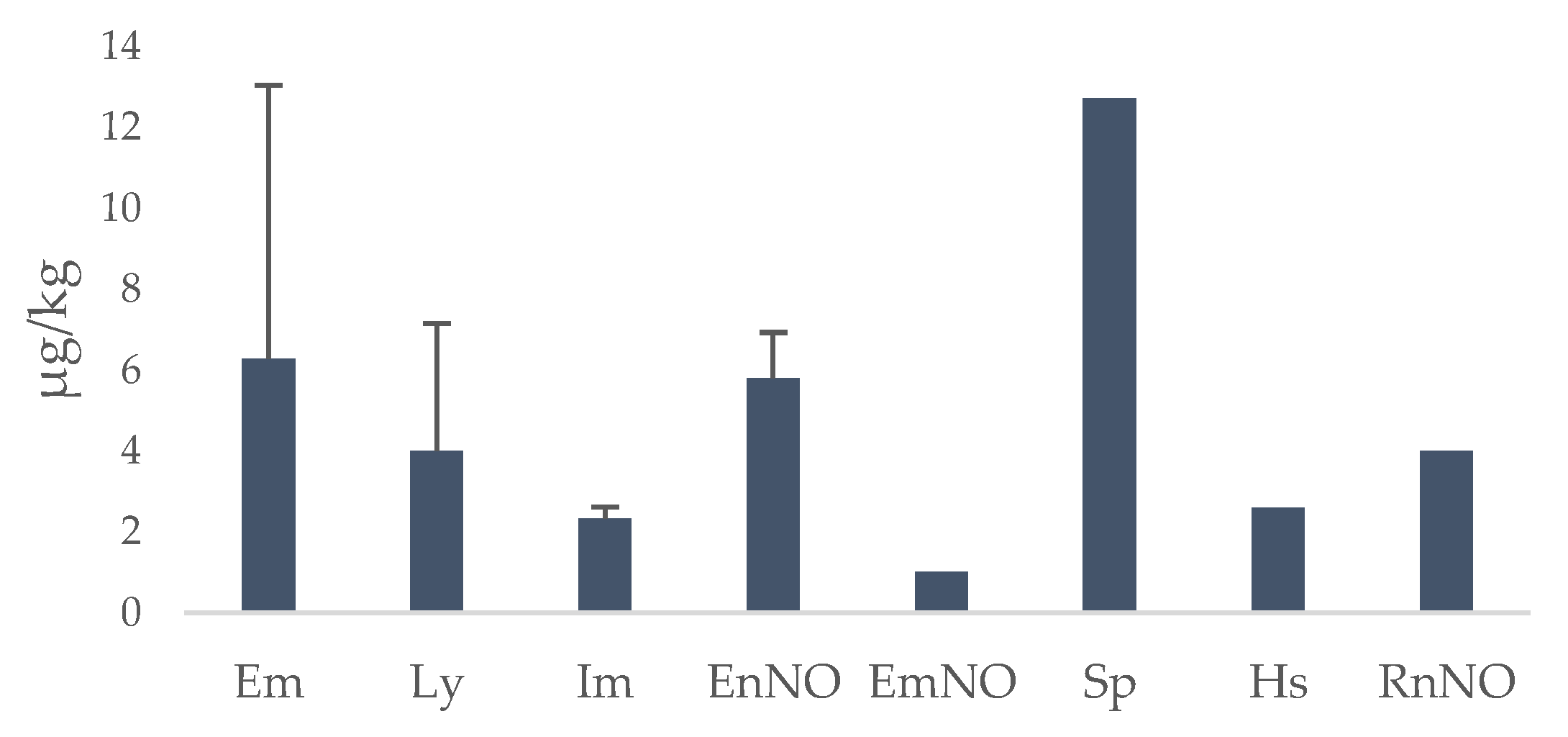
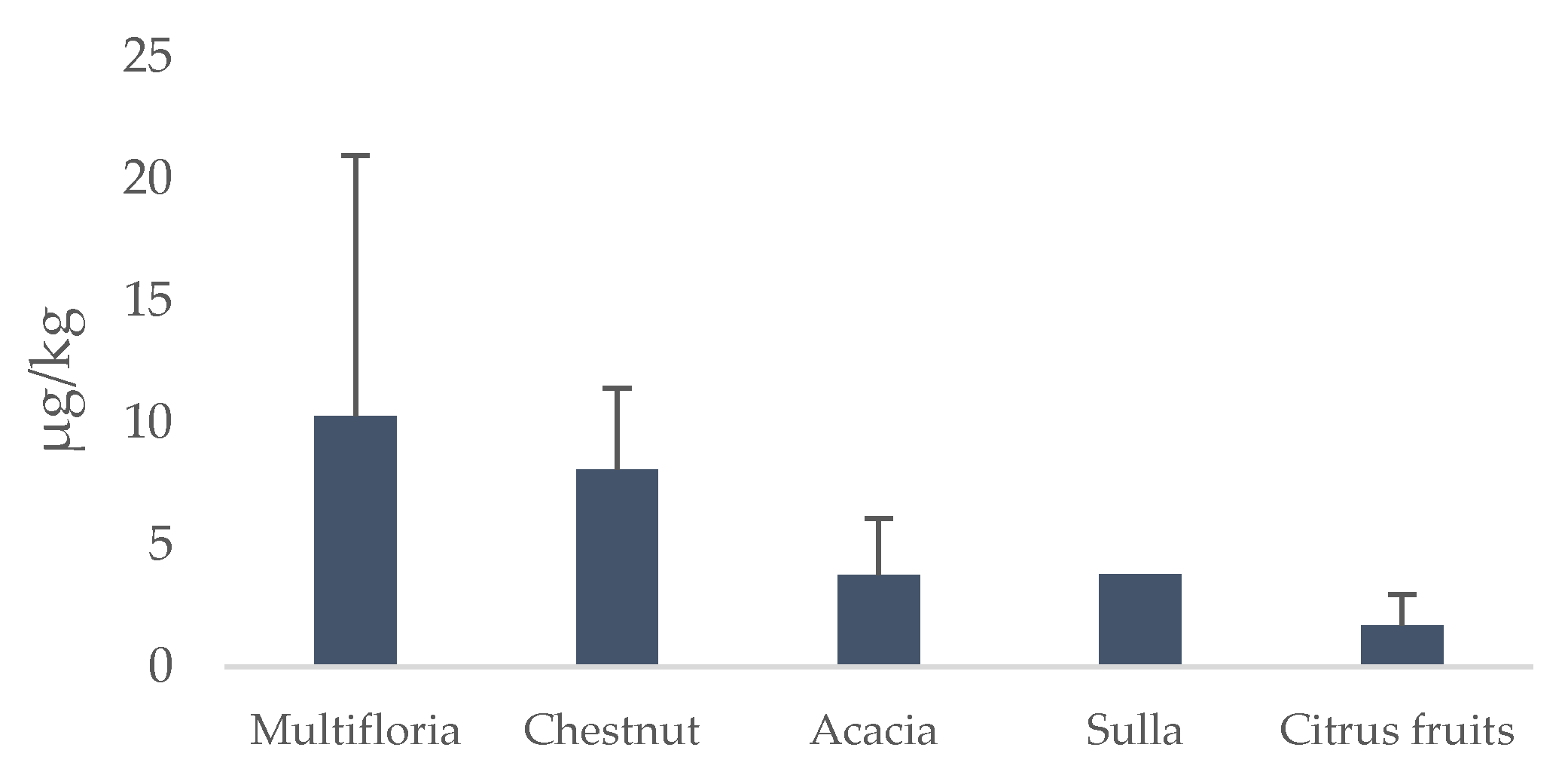
3.2. PA/PANO Content in Honey Samples
3.3. Variables Affecting the Content of PAs/PANOs in Honey Samples
3.4. Exposure Assessment and Health Risk Characterization
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moreira, R.; Pereira, D.; Valentão, P.; Andrade, P. Pyrrolizidine Alkaloids: Chemistry, Pharmacology, Toxicology and Food Safety. Int. J. Mol. Sci. 2018, 19, 1668. [Google Scholar] [CrossRef] [PubMed]
- Ebmeyer, J.; Braeuning, A.; Glatt, H.; These, A.; Hessel-Pras, S.; Lampen, A. Human CYP3A4-Mediated Toxification of the Pyrrolizidine Alkaloid Lasiocarpine. Food Chem. Toxicol. 2019, 130, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Stegemann, T.; Kruse, L.H.; Brütt, M.; Ober, D. Specific Distribution of Pyrrolizidine Alkaloids in Floral Parts of Comfrey (Symphytum officinale) and Its Implications for Flower Ecology. J. Chem. Ecol. 2019, 45, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Brugnerotto, P.; Seraglio, S.K.T.; Schulz, M.; Gonzaga, L.V.; Fett, R.; Costa, A.C.O. Pyrrolizidine Alkaloids and Beehive Products: A Review. Food Chem. 2021, 342, 128384. [Google Scholar] [CrossRef]
- Casado, N.; Morante-Zarcero, S.; Sierra, I. The Concerning Food Safety Issue of Pyrrolizidine Alkaloids: An Overview. Trends Food Sci. Technol. 2022, 120, 123–139. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, Q.; Yi, Z.; Chen, Y.; Xiao, W.; Su, D.; Shi, W. Risk Assessment of (Herbal) Teas Containing Pyrrolizidine Alkaloids (PAs) Based on Margin of Exposure Approach and Relative Potency (REP) Factors. Foods 2022, 11, 2946. [Google Scholar] [CrossRef]
- Dusemund, B.; Nowak, N.; Sommerfeld, C.; Lindtner, O.; Schäfer, B.; Lampen, A. Risk Assessment of Pyrrolizidine Alkaloids in Food of Plant and Animal Origin. Food Chem. Toxicol. 2018, 115, 63–72. [Google Scholar] [CrossRef]
- Habs, M.; Binder, K.; Krauss, S.; Müller, K.; Ernst, B.; Valentini, L.; Koller, M. A Balanced Risk–Benefit Analysis to Determine Human Risks Associated with Pyrrolizidine Alkaloids (PA)—The Case of Tea and Herbal Infusions. Nutrients 2017, 9, 717. [Google Scholar] [CrossRef]
- Edgar, J.A.; Colegate, S.M.; Boppré, M.; Molyneux, R.J. Pyrrolizidine Alkaloids in Food: A Spectrum of Potential Health Consequences. Food Addit. Contam. Part A 2011, 28, 308–324. [Google Scholar] [CrossRef]
- Hama, J.R.; Strobel, B.W. Pyrrolizidine Alkaloids Quantified in Soil and Water Using UPLC-MS/MS. RSC Adv. 2019, 9, 30350–30357. [Google Scholar] [CrossRef]
- Hama, J.R.; Strobel, B.W. Occurrence of Pyrrolizidine Alkaloids in Ragwort Plants, Soils and Surface Waters at the Field Scale in Grassland. Sci. Total Environ. 2021, 755, 142822. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific Opinion on Pyrrolizidine Alkaloids in Food and Feed. EFSA J. 2011, 9, 2406. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM); Knutsen, H.K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; et al. Risks for Human Health Related to the Presence of Pyrrolizidine Alkaloids in Honey, Tea, Herbal Infusions and Food Supplements. EFSA J. 2017, 15, e04908. [Google Scholar] [CrossRef]
- Ruan, J.; Yang, M.; Fu, P.; Ye, Y.; Lin, G. Metabolic Activation of Pyrrolizidine Alkaloids: Insights into the Structural and Enzymatic Basis. Chem. Res. Toxicol. 2014, 27, 1030–1039. [Google Scholar] [CrossRef]
- Xia, Q.; He, X.; Shi, Q.; Lin, G.; Fu, P.P. Quantitation of DNA Reactive Pyrrolic Metabolites of Senecionine—A Carcinogenic Pyrrolizidine Alkaloid by LC/MS/MS Analysis. J. Food Drug Anal. 2020, 28, 167–174. [Google Scholar] [CrossRef]
- Xu, J.; Wang, W.; Yang, X.; Xiong, A.; Yang, L.; Wang, Z. Pyrrolizidine Alkaloids: An Update on Their Metabolism and Hepatotoxicity Mechanism. Liver Res. 2019, 3, 176–184. [Google Scholar] [CrossRef]
- Kakar, F.; Akbarian, Z.; Leslie, T.; Mustafa, M.L.; Watson, J.; van Egmond, H.P.; Omar, M.F.; Mofleh, J. An Outbreak of Hepatic Veno-Occlusive Disease in Western Afghanistan Associated with Exposure to Wheat Flour Contaminated with Pyrrolizidine Alkaloids. J. Toxicol. 2010, 2010, 313280. [Google Scholar] [CrossRef]
- IARC (International Agency for Research on Cancer). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, 1st ed.; World Health Organization: Lyon, France, 1987; ISBN 978-92-832-1411-3. [Google Scholar]
- IARC (International Agency for Research on Cancer). Some Traditional Herbal Medicines, Some Mycotoxins, Naphthalene and Styrene. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2002; ISBN 978-92-832-1282-9. [Google Scholar]
- European Commission. Commission Regulation (EU) 2020/2040 of 11 December 2020 amending Regulation (EC) No 1881/2006 as regards maximum levels of pyrrolizidine alkaloids in certain foodstuffs. Off. J. Eur. Union 2020, 1–5. [Google Scholar]
- Alvarado-Avila, L.Y.; Moguel-Ordóñez, Y.B.; García-Figueroa, C.; Ramírez-Ramírez, F.J.; Arechavaleta-Velasco, M.E. Presencia de Alcaloides Pirrolizidínicos En Miel y Los Efectos de Su Consumo En Humanos y Abejas. Revisión. Rev. Mex. Cienc. Pecu. 2022, 13, 787–802. [Google Scholar] [CrossRef]
- Lucatello, L.; Merlanti, R.; De Jesus Inacio, L.; Bisutti, V.; Montanucci, L.; Capolongo, F. Pyrrolizidine Alkaloid Concentrations in Local Italian and Retail Honeys of Different Origin: A Scenario of Human Exposure. J. Food Compos. Anal. 2021, 104, 104182. [Google Scholar] [CrossRef]
- Griffin, C.T.; Danaher, M.; Elliott, C.T.; Glenn Kennedy, D.; Furey, A. Detection of Pyrrolizidine Alkaloids in Commercial Honey Using Liquid Chromatography–Ion Trap Mass Spectrometry. Food Chem. 2013, 136, 1577–1583. [Google Scholar] [CrossRef] [PubMed]
- Von Der Ohe, W.; Persano Oddo, L.; Piana, M.L.; Morlot, M.; Martin, P. Harmonized Methods of Melissopalynology. Apidologie 2004, 35, S18–S25. [Google Scholar] [CrossRef]
- Bundesinstitut für Risikobewertung. Determination of Pyrrolizidine Alkaloids (PA) in Honey by SPE-LC-MS/MS Method Protocol BfR-PA-Honey-1.0/2013; Bundesinstitut für Risikobewertung: Berlin, Germany, 2013; pp. 1–17. [Google Scholar]
- EURL-MP-Method_002 (Version 2) Determination of Pyrrolizidine Alkaloids in Plant-Based Food and Feed Materials, Including (Herbal) Teas, Herbal Food Supplements, Fodder and Feedstuffs by LC-MS/MS; RIKILT Wageningen University & Research: Wageningen, The Netherlands, 2019; pp. 1–26.
- European Commissions. Commission Regulation (EC) No 401/2006 of 23 February 2006 Laying down the Methods of Sampling and Analysis for the Official Control of the Levels of Mycotoxins in Foodstuffs. Off. J. Eur. Union. 2006, 12–34. [Google Scholar]
- European Commission. Health and Food Safety Directorate-General (DG SANTE). Guidance Document on Identification of Mycotoxin in Food and Feed. SANTE/12089/2016; European Commission DG SANTE: Bruxelles, Belgium, 2016. [Google Scholar]
- Mascha, E.J.; Vetter, T.R. Significance, Errors, Power, and Sample Size: The Blocking and Tackling of Statistics. Anesth. Analg. 2018, 126, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Cramér, H. The Two-Dimensional Case. In Mathematical Methods of Statistics; Princeton University Press: Princeton, NJ, USA, 1946; p. 282. ISBN 0-691-08004-6. [Google Scholar]
- Fürst, P.; Milana, M.R.; Pfaff, K.; Tlustos, C.; Vleminckx, C.; Arcella, D.; Barthélémy, E.; Colombo, P.; Goumperis, T.; Pasinato, L.; et al. Risk Evaluation of Chemical Contaminants in Food in the Context of RASFF Notifications. EFSA Support. Publ. 2019, 16, 1625E. [Google Scholar] [CrossRef]
- Leclercq, C.; Arcella, D.; Piccinelli, R.; Sette, S.; Le Donne, C.; Turrini, A.; INRAN-SCAI 2005–06 Study Group. The Italian National Food Consumption Survey INRAN-SCAI 2005–06: Main Results in Terms of Food Consumption. Public Health Nutr. 2009, 12, 2504–2532. [Google Scholar] [CrossRef]
- FAO-WHO. Pesticide residues in food. In Proceedings of the Joint Meeting of the FAO Panel of Experts on Pesticide Residues in Food and the Environment and the WHO Core Assessment Group on Pesticide Residues, Rome, Italy, 20–29 September 2004, ISSN 0259-2517. [Google Scholar]
- Dakeishi, M.; Murata, K.; Tamura, A.; Iwata, T. Relation Between Benchmark Dose and No-Observed-Adverse-Effect Level in Clinical Research: Effects of Daily Alcohol Intake on Blood Pressure in Japanese Salesmen. Risk Anal. 2006, 26, 115–123. [Google Scholar] [CrossRef]
- European Union Reference Laboratory (EURL). EURLMP-Background doc_003 Guidance Document Performance Criteria v1.1 draft of 17 September 2021. Available online: https://www.wur.nl/en/research-results/research-institutes/food-safety-research/reference-laboratory/european-union-reference-laboratory/eurl-mycotoxins-plant-toxins/library-eurl-mp.htm (accessed on 16 January 2023).
- Cao, X.; Deng, W.; Fu, M.; Zhu, Y.; Liu, H.; Wang, L.; Zeng, J.; Wei, Y.; Xu, X.; Yu, J. Seventy-Two-Hour Release Formulation of the Poorly Soluble Drug Silybin Based on Porous Silica Nanoparticles: In Vitro Release Kinetics and In Vitro/In Vivo Correlations in Beagle Dogs. Eur. J. Pharm. Sci. 2013, 48, 64–71. [Google Scholar] [CrossRef]
- Dübecke, A.; Beckh, G.; Lüllmann, C. Pyrrolizidine Alkaloids in Honey and Bee Pollen. Food Addit. Contam. Part A 2011, 28, 348–358. [Google Scholar] [CrossRef]
- Griffin, C.T.; O’Mahony, J.; Danaher, M.; Furey, A. Liquid Chromatography Tandem Mass Spectrometry Detection of Targeted Pyrrolizidine Alkaloids in Honeys Purchased within Ireland. Food Anal. Methods 2015, 8, 18–31. [Google Scholar] [CrossRef]
- Huybrechts, B.; Callebaut, A. Pyrrolizidine Alkaloids in Food and Feed on the Belgian Market. Food Addit. Contam. Part A 2015, 32, 1939–1951. [Google Scholar] [CrossRef]
- Kempf, M.; Wittig, M.; Schönfeld, K.; Cramer, L.; Schreier, P.; Beuerle, T. Pyrrolizidine Alkaloids in Food: Downstream Contamination in the Food Chain Caused by Honey and Pollen. Food Addit. Contam. Part A 2011, 28, 325–331. [Google Scholar] [CrossRef]
- Martinello, M.; Cristofoli, C.; Gallina, A.; Mutinelli, F. Easy and Rapid Method for the Quantitative Determination of Pyrrolizidine Alkaloids in Honey by Ultra Performance Liquid Chromatography-Mass Spectrometry: An Evaluation in Commercial Honey. Food Control 2014, 37, 146–152. [Google Scholar] [CrossRef]
- Orantes-Bermejo, F.J.; Serra Bonvehí, J.; Gómez-Pajuelo, A.; Megías, M.; Torres, C. Pyrrolizidine Alkaloids: Their Occurrence in Spanish Honey Collected from Purple Viper’s Bugloss (Echium spp.). Food Addit. Contam. Part A 2013, 30, 1799–1806. [Google Scholar] [CrossRef]
- Kowalczyk, E.; Sieradzki, Z.; Kwiatek, K. Determination of Pyrrolizidine Alkaloids in Honey with Sensitive Gas Chromatography-Mass Spectrometry Method. Food Anal. Methods 2018, 11, 1345–1355. [Google Scholar] [CrossRef]
- Kowalczyk, E.; Kwiatek, K. Simultaneous Determination of Pyrrolizidine and Tropane Alkaloids in Honey by Liquid Chromatography–Mass Spectrometry. J. Vet. Res. 2022, 66, 235–243. [Google Scholar] [CrossRef]
- Bodi, D.; Ronczka, S.; Gottschalk, C.; Behr, N.; Skibba, A.; Wagner, M.; Lahrssen-Wiederholt, M.; Preiss-Weigert, A.; These, A. Determination of Pyrrolizidine Alkaloids in Tea, Herbal Drugs and Honey. Food Addit. Contam. Part A 2014, 31, 1886–1895. [Google Scholar] [CrossRef]
- Kast, C.; Dübecke, A.; Kilchenmann, V.; Bieri, K.; Böhlen, M.; Zoller, O.; Beckh, G.; Lüllmann, C. Analysis of Swiss Honeys for Pyrrolizidine Alkaloids. J. Apic. Res. 2014, 53, 75–83. [Google Scholar] [CrossRef]
- Picron, J.-F.; Herman, M.; Van Hoeck, E.; Goscinny, S. Monitoring of Pyrrolizidine Alkaloids in Beehive Products and Derivatives on the Belgian Market. Environ. Sci. Pollut. Res. 2020, 27, 5693–5708. [Google Scholar] [CrossRef]
- Abdulla, C.O.; Ayubi, A.; Zulfiquer, F.; Santhanam, G.; Ahmed, M.A.S.; Deeb, J. Infant Botulism Following Honey Ingestion. Case Rep. 2012, 2012, bcr1120115153. [Google Scholar] [CrossRef]
- Tanzi, M.G.; Gabay, M.P. Association Between Honey Consumption and Infant Botulism. Pharmacotherapy 2002, 22, 1479–1483. [Google Scholar] [CrossRef] [PubMed]
- Vică, M.L.; Glevitzky, M.; Dumitrel, G.-A.; Bostan, R.; Matei, H.V.; Kartalska, Y.; Popa, M. Qualitative Characterization and Antifungal Activity of Romanian Honey and Propolis. Antibiotics 2022, 11, 1552. [Google Scholar] [CrossRef] [PubMed]
- Fratianni, F.; Amato, G.; d’Acierno, A.; Ombra, M.N.; De Feo, V.; Coppola, R.; Nazzaro, F. In Vitro Prospective Healthy and Nutritional Benefits of Different Citrus Monofloral Honeys. Sci. Rep. 2023, 13, 1088. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.L.; Lim, L.Y.; Hammer, K.; Hettiarachchi, D.; Locher, C. A Review of Commonly Used Methodologies for Assessing the Antibacterial Activity of Honey and Honey Products. Antibiotics 2022, 11, 975. [Google Scholar] [CrossRef]
- Zulkifli, N.A.; Hassan, Z.; Mustafa, M.Z.; Azman, W.N.W.; Hadie, S.N.H.; Ghani, N.; Mat Zin, A.A. The Potential Neuroprotective Effects of Stingless Bee Honey. Front. Aging Neurosci. 2023, 14, 1048028. [Google Scholar] [CrossRef]

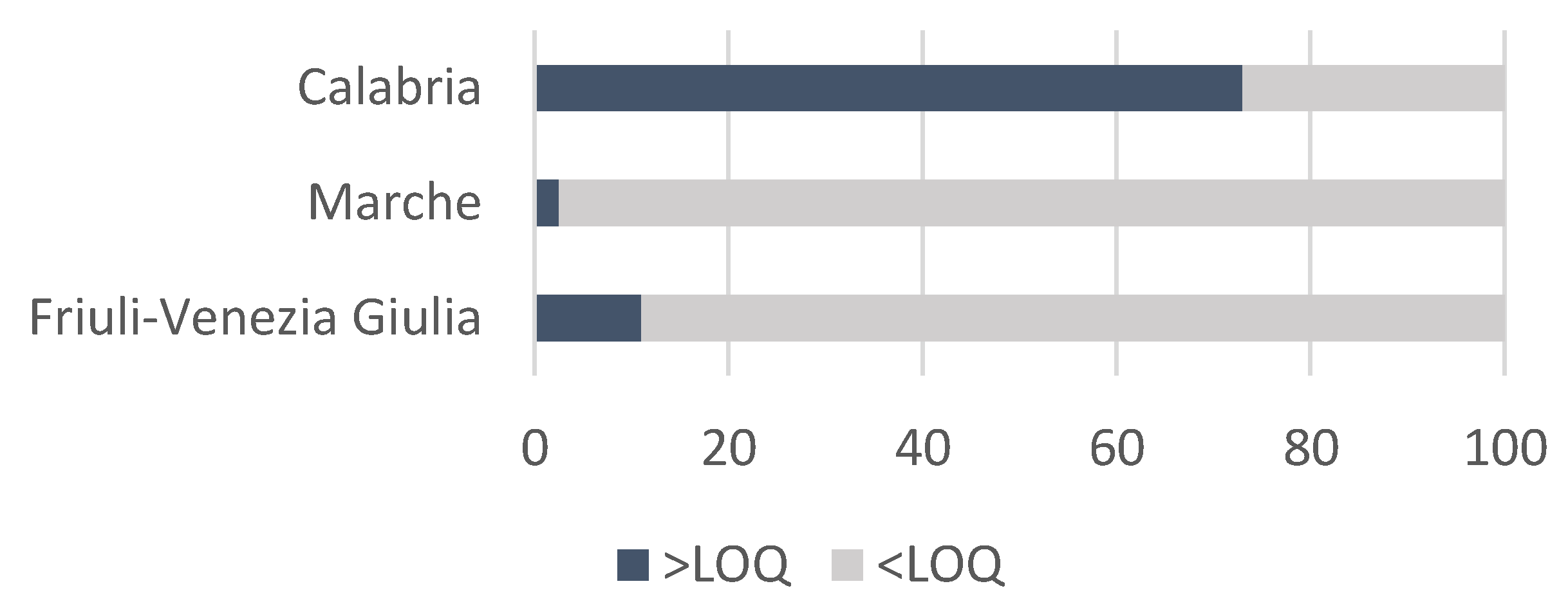
| Honey Types | Origin |
|---|---|
| Multifloral, Winter heath, Common whitebeam, Dandelion, Acacia, Chestnut and Linden | Udine (Friuli-Venezia Giulia) |
| Multifloral, Rapeseed, Linden, Acacia, Bastard Indigobush and Chestnut | Pordenone (Friuli-Venezia Giulia) |
| Multifloral and Mahaleb Cherry | Trieste (Friuli-Venezia Giulia) |
| Multifloral, Sunflower, Rapeseed, Honeydew, Mustard, Clover and Betony | Ancona (Marche) |
| Multifloral, Chestnut and Acacia | Fermo (Marche) |
| Multifloral and Sunflower | Macerata (Marche) |
| Multifloral, Chestnut, Honeydew and Acacia | Ascoli Piceno (Marche) |
| Multifloral, Acacia, Sulla, Citrus Fruits and Chestnut | Vibo Valentia (Calabria) |
| Acacia, Citrus Fruits and Chestnut | Cosenza (Calabria) |
| Multifloral, Acacia, Sulla, Citrus Fruits, Eucalyptus and Chestnut | Catanzaro (Calabria) |
| Chestnut and Winter heath | Crotone (Calabria) |
| Acacia and Citrus Fruits | Reggio Calabria (Calabria) |
| Monofloral Honey Types | Total Samples |
|---|---|
| Acacia or robinia (Robinia pseudoacacia) | 25 |
| Chestnut (Castanea sativa) | 15 |
| Sunflower (Heliantus annuus L.) | 14 |
| Citrus fruits | 7 |
| Linden (Tilia L.) | 6 |
| Rapeseed (Brassica napus) | 4 |
| Honeydew | 3 |
| Winter heath (Erica carnea) | 2 |
| Sulla (Sulla coronaria) | 2 |
| Eucalyptus (E. camaldulensis, E. occidentalis) | 2 |
| Mustard (Sinapis arvensis) | 1 |
| Clover (Trifolium pratense) | 1 |
| Betony (Stachys officinalis) | 1 |
| Common whitebeam (Sorbus aria) | 1 |
| Bastard indigobush (Amorpha fruticosa) | 1 |
| Dandelion (Taraxacum officinale) | 1 |
| Mahaleb cherry (Prunus mahaleb) | 1 |
| Pyrrolizidine Alkaloids | MH+ | CE | m/z | Q, q |
|---|---|---|---|---|
| Sn-group | 336.2 | 25 25 | 120.2 138.0 | Q q |
| Ly-group | 299.7 | 20 25 | 138.0 156.0 | Q q |
| Ht | 314.1 | 20 25 | 138.0 156.0 | Q q |
| Eu | 330 | 20 15 | 138.0 156.0 | Q q |
| Sk | 366.1 | 30 25 | 122.0 167.9 | Q q |
| ErN | 366.1 | 35 25 | 94.1 119.1 | Q q |
| LyN-group | 316.1 | 25 25 | 172.0 138.0 | Q q |
| HtN | 330.2 | 25 25 | 172.0 111.0 | Q q |
| EuN | 346.2 | 25 20 | 172.0 328.1 | Q q |
| Lc | 412.1 | 25 18 | 120.1 220.0 | Q q |
| Sp-group | 334 | 25 25 | 120.1 138.0 | Q q |
| Em-group | 398.6 | 20 15 | 119.9 220.4 | Q q |
| Rt-group | 352.1 | 25 25 | 120.0 138.3 | Q q |
| SnN-group | 352.1 | 25 30 | 94.0 118.0 | Q q |
| LcN | 428.1 | 30 25 | 254.0 94.0 | Q q |
| SpN-group | 350.1 | 30 25 | 94.0 120.0 | Q q |
| EmN-group | 414.2 | 30 25 | 254.0 220.0 | Q q |
| RtN-group | 368.3 | 30 20 | 94.0 120.0 | Q q |
| Analyte | Conc (µg/kg) | Recovery % | RSDr | RSDR |
|---|---|---|---|---|
| Em-Hs | 1 | 94.5 | 13.53 | 14.81 |
| 10 | 85.3 | 13.21 | 23.08 | |
| 25 | 76.9 | 4.52 | 6.28 | |
| EmN | 1 | 82.9 | 10.79 | 11.63 |
| 10 | 75.4 | 10.04 | 10.75 | |
| 25 | 72.9 | 9.41 | 11.21 | |
| Eu | 1 | 77.5 | 14.83 | 17.97 |
| 10 | 60.3 | 4.47 | 18.61 | |
| 25 | 84.8 | 0.96 | 23.87 | |
| EuN | 1 | 83.6 | 13.24 | 15.28 |
| 10 | 79.3 | 4.18 | 16.96 | |
| 25 | 71.9 | 5.33 | 10.58 | |
| Ht | 1 | 90.6 | 11.06 | 12.52 |
| 10 | 75.7 | 8.91 | 14.42 | |
| 25 | 70.4 | 5.91 | 7.13 | |
| HtN | 1 | 91.3 | 11.45 | 12.87 |
| 10 | 80.6 | 4.48 | 5.16 | |
| 25 | 71.8 | 8.19 | 9.81 | |
| Im | 1 | 79.9 | 13.17 | 20.19 |
| 10 | 70.1 | 2.95 | 19.46 | |
| 25 | 62.6 | 5.28 | 7.50 | |
| ImN | 1 | 74.3 | 0.69 | 15.41 |
| 10 | 71.1 | 7.12 | 9.81 | |
| 25 | 67.1 | 17.75 | 9.66 | |
| Lc | 1 | 93.7 | 7.01 | 15.83 |
| 10 | 84.3 | 11.65 | 23.03 | |
| 25 | 76.2 | 4.81 | 7.64 | |
| LcN | 1 | 74.8 | 17.86 | 20.46 |
| 10 | 68.3 | 12.96 | 13.77 | |
| 25 | 67.6 | 4.51 | 9.87 | |
| Ly, Id, En, Rn | 1 | 84.9 | 7.72 | 20.84 |
| 10 | 72.5 | 6.81 | 24.21 | |
| 25 | 65.6 | 3.64 | 13.70 | |
| LyN, IdN | 1 | 74.8 | 9.56 | 9.99 |
| 10 | 68.3 | 5.54 | 8.86 | |
| 25 | 67.6 | 7.52 | 11.24 | |
| Rt, Us | 1 | 75.8 | 11.34 | 16.64 |
| 10 | 71.5 | 7.89 | 19.31 | |
| 25 | 64.7 | 7.28 | 7.63 | |
| RtN, UsN | 1 | 87.6 | 8.92 | 21.43 |
| 10 | 74.7 | 8.36 | 10.56 | |
| 25 | 66.7 | 6.95 | 11.48 | |
| Sp, St | 1 | 80.3 | 15.31 | 19.67 |
| 10 | 87.6 | 7.01 | 23.97 | |
| 25 | 79.6 | 6 | 9.28 | |
| SpN, StN | 1 | 79.7 | 11.9 | 19.64 |
| 10 | 70.2 | 6.51 | 14.11 | |
| 25 | 65.2 | 6.41 | 11.89 | |
| Sn, Ir | 1 | 96.4 | 10.37 | 11.41 |
| 10 | 84.9 | 6.45 | 18.22 | |
| 25 | 81 | 5.18 | 5.87 | |
| SnN | 1 | 85.1 | 11.37 | 13.61 |
| 10 | 78.5 | 5.72 | 12.22 | |
| 25 | 74.2 | 2.88 | 12.05 | |
| Sv | 1 | 88.1 | 12.1 | 22.88 |
| 10 | 71.9 | 8.39 | 20.51 | |
| 25 | 72 | 4.6 | 20.16 | |
| SvN | 1 | 97.9 | 9.38 | 10.21 |
| 10 | 85.8 | 8.14 | 11.68 | |
| 25 | 84.5 | 7.83 | 10.81 | |
| Sk | 1 | 87.4 | 12.85 | 18.87 |
| 10 | 76 | 11.11 | 17.55 | |
| 25 | 77.5 | 2.58 | 16.36 | |
| EnN | 1 | 92.1 | 12.85 | 20.56 |
| 10 | 75.3 | 11.11 | 9.22 | |
| 25 | 61.9 | 2.58 | 10.03 | |
| RnN | 1 | 76.1 | 14.45 | 15.31 |
| 10 | 70.5 | 5.86 | 17.63 | |
| 25 | 68.9 | 8.37 | 13.12 | |
| IrN | 1 | 88.4 | 6.77 | 12.48 |
| 10 | 81.6 | 4.28 | 7.22 | |
| 25 | 77.2 | 10.13 | 13.36 | |
| HsN | 1 | 95.1 | 8.64 | 10.21 |
| 10 | 86.4 | 7.7 | 9.04 | |
| 25 | 87.7 | 3.49 | 11.66 |
| Kowalczyk et al., 2022 | Picron et al., 2020 | Martinello et al., 2017 | Lucatello et al., 2016 | Griffin et al., 2014 | Martinello et al., 2014 | Orantes-Bermejo et al., 2013 | This Research | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PA/PANO (µg/kg) | Polish Honey | Foreign Honey | Belgian Honey | Foreign Honey | Retail Honey | Italian Beekeepers | Retail Honey | Retail Honey | Spanish Honey by Beekeepers | Italian Honey | |
| Echimidine | 7.3 | 120.0 | 5.91 | 8.84 | 0.4–3.3 | 0.3–1.0 | 545.5 | 169 | 36.9 ± 44.36 | 237 | 1.0–30.6 |
| Echinatine | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Europine | n.i. | n.i. | 0.009 | 134.85 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Heliosupine | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | LOQ-2.6 |
| Heliotrine | n.i. | n.i. | <LOQ | 39.44 | <LOQ | <LOQ | <LOQ | <LOQ | n.i. | n.i. | <LOQ |
| Indicine | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Integerrimine | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Intermedine | 9.2 | 23.3 | n.i. | n.i. | <LOQ | <LOQ | n.i. | 31 | n.i. | n.i. | 1.9–2.4 |
| Lasiocarpine | n.i. | n.i. | <LOQ | 5.77 | <LOQ | <LOQ | n.i. | <LOQ | n.i. | n.i. | <LOQ |
| Lycopsamine | 14.1 | 22.5 | n.i. | n.i. | 0.2–74.7 | <LOQ | 392.6 | 42 | 5.7 ± 4.28 | 18 | 1.9–9.3 |
| Retrorsine | 4.3 | 4.3 | 5.82 | 5.32 | <LOQ | 0.9–14.5 | <LOQ | <LOQ | n.i. | n.i. | <LOQ |
| Rinderine | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Senecionine | 2.2 | 2.7 | 9.67 | 1.46 | <LOQ | 0.8–2.1 | 8.4 | <LOQ | n.i. | n.i. | <LOQ |
| Seneciphylline | 4.1 | 4.0 | 7.35 | 4.04 | <LOQ | 0.6–1.1 | 5.7 | <LOQ | 4.1 ± 4.79 | 20 | LOQ-12.71 |
| Senecivernine | <LOQ | 3.0 | 4.15 | 0.41 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Senkirkine | n.i. | n.i. | 42.44 | 1.15 | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ |
| Spartioidine | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Usaramine | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Echimidine-N-oxide | n.i. | n.i. | 8.24 | 0.17 | n.i. | n.i. | n.i. | n.i. | 21.4 ± 23.09 | 70 | LOQ-1.01 |
| Echinatine-N-oxide | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | 5–6.6 |
| Erucifoline-N-oxide | n.i. | n.i. | 0.09 | 0.14 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Europine-N-oxide | n.i. | n.i. | 0.54 | 1.30 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Heliosupine-N-oxide | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Heliotrine-N-oxide | n.i. | n.i. | <LOQ | 0.37 | n.i. | n.i. | n.i. | n.i. | 2.3 ± 0.58 | 4 | <LOQ |
| Indicine-N-oxide | n.i. | n.i. | 0.47 | 0.18 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Intermedine-N-oxide | n.i. | n.i. | 0.26 | 0.21 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Integerrimine-N-oxide | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Lycopsamine-N-oxide | n.i. | n.i. | 0.29 | 0.09 | n.i. | n.i. | n.i. | n.i. | 4.0 ± 2.79 | 8 | <LOQ |
| Retrorsine-N-oxide | n.i. | n.i. | 0.86 | 0.90 | n.i. | n.i. | <LOQ | n.i. | <LOQ | <LOQ | <LOQ |
| Rinderine-N-oxide | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | LOQ-4 |
| Senecionine-N-oxide | n.i. | n.i. | 0.54 | 0.26 | n.i. | n.i. | <LOQ | n.i. | <LOQ | <LOQ | <LOQ |
| Seneciphylline-N-oxide | n.i. | n.i. | 0.16 | 0.28 | n.i. | n.i. | <LOQ | n.i. | <LOQ | <LOQ | <LOQ |
| Senecivernine-N-oxide | n.i. | n.i. | 0.67 | <LOQ | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Spartioidine-N-oxide | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| Usaramine-N-oxide | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | <LOQ |
| RACE Parameters | |
|---|---|
| Contaminant | Pyrrolizidine alkaloids (sum of 1,2-unsaturated) |
| Food description | Honey |
| Analytical result | 33.1 µg/kg |
| Reference value acute (ARfD) | 2 mg/kg bw |
| Reference value chronic (BMDL10) | 237 µg/kg bw |
| Survey country | Italy [32] |
| Output (% ARfD) | |||
|---|---|---|---|
| Acute Consumers Only | Mean | Median | 95th Percentile |
| Toddlers | 0.0022 | 0.0024 | 0.0047 1 |
| Other children | 0.0012 | 0.0008 | 0.0024 1 |
| Adolescents | 0.0007 | 0.0005 | 0.0016 1 |
| Adults | 0.0005 | 0.0003 | 0.0011 |
| Elderly | 0.0004 | 0.0003 | 0.0010 |
| Very elderly | 0.0006 | 0.0005 | 0.0009 1 |
| Acute whole population | Mean | Median | 95th percentile |
| Toddlers | 0.00023 | - 2 | 0.00236 |
| Other children | 0.00008 | - 2 | 0.00072 |
| Adolescents | 0.00001 | - 2 | - |
| Adults | 0.00003 | - 2 | 0.00018 |
| Elderly | 0.00003 | - 2 | 0.00026 |
| Very elderly | 0.00004 | - 2 | 0.00031 |
| Output (MoE) | |||
|---|---|---|---|
| Chronic Consumers Only | Mean | Median | 95th Percentile |
| Toddlers | 7303 | 7876 | 3759 1 |
| Other children | 12,496 | 15,752 | 5783 1 |
| Adolescents | 24,134 | 24,702 | 17,184 1 |
| Adults | 36,740 | 50,476 | 12,411 |
| Elderly | 44,500 | 42,961 | 20,764 1 |
| Very elderly | 27,294 | 31,147 | 7279 1 |
| Chronic whole population | Mean | Median | 95th percentile |
| Toddlers | 52,581 | - 2 | 4654 |
| Other children | 141,868 | - 2 | 16,468 |
| Adolescents | 851,578 | - 2 | - 2 |
| Adults | 456,875 | - 2 | 62,293 |
| Elderly | 391,059 | - 2 | 39,953 |
| Very elderly | 327,532 | - 2 | 37,591 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roncada, P.; Isani, G.; Peloso, M.; Dalmonte, T.; Bonan, S.; Caprai, E. Pyrrolizidine Alkaloids from Monofloral and Multifloral Italian Honey. Int. J. Environ. Res. Public Health 2023, 20, 5410. https://doi.org/10.3390/ijerph20075410
Roncada P, Isani G, Peloso M, Dalmonte T, Bonan S, Caprai E. Pyrrolizidine Alkaloids from Monofloral and Multifloral Italian Honey. International Journal of Environmental Research and Public Health. 2023; 20(7):5410. https://doi.org/10.3390/ijerph20075410
Chicago/Turabian StyleRoncada, Paola, Gloria Isani, Mariantonietta Peloso, Thomas Dalmonte, Stefania Bonan, and Elisabetta Caprai. 2023. "Pyrrolizidine Alkaloids from Monofloral and Multifloral Italian Honey" International Journal of Environmental Research and Public Health 20, no. 7: 5410. https://doi.org/10.3390/ijerph20075410
APA StyleRoncada, P., Isani, G., Peloso, M., Dalmonte, T., Bonan, S., & Caprai, E. (2023). Pyrrolizidine Alkaloids from Monofloral and Multifloral Italian Honey. International Journal of Environmental Research and Public Health, 20(7), 5410. https://doi.org/10.3390/ijerph20075410







