Risk of Lactic Acidosis in Hospitalized Diabetic Patients Prescribed Biguanides in Japan: A Retrospective Total-Population Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Dataset
2.2. Study Design
2.3. Study Population
2.4. Exposure: Biguanide Prescriptions
2.5. Outcome: LA Incidence in Hospitalized DM Patients Retrieved from the NDB
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Inzucchi, S.E.; Bergenstal, R.M.; Buse, J.B.; Diamant, M.; Ferrannini, E.; Nauck, M.; Peters, A.L.; Tsapas, A.; Wender, R.; Matthews, D.R. Management of hyperglycemia in type 2 diabetes, 2015: A patient-centered approach: Update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2015, 38, 140–149. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. EMPA-REG OUTCOME Investigators. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; Poulter, N.R.; Ravn, L.S.; et al. LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Nesti, L.; Natali, A. Metformin effects on the heart and the cardiovascular system: A review of experimental and clinical data. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 657–669. [Google Scholar] [CrossRef] [PubMed]
- Eibich, P.; Green, A.; Hattersley, A.T.; Jennison, C.; Lonergan, M.; Pearson, E.R.; Gray, A.M. Costs and Treatment Pathways for Type 2 Diabetes in the UK: A Mastermind Cohort Study. Diabetes Ther. 2017, 8, 1031–1045. [Google Scholar] [CrossRef] [PubMed]
- Montvida, O.; Shaw, J.; Atherton, J.J.; Stringer, F.; Paul, S.K. Long-term Trends in Antidiabetes Drug Usage in the U.S.: Real-world Evidence in Patients Newly Diagnosed with Type 2 Diabetes. Diabetes Care 2018, 41, 69–78. [Google Scholar] [CrossRef]
- Chu, W.M.; Ho, H.E.; Huang, K.H.; Tsan, Y.T.; Liou, Y.S.; Wang, Y.H.; Lee, M.C.; Li, Y.C. The prescribing trend of oral antidiabetic agents for type 2 diabetes in Taiwan: An 8-year population-based study. Medicine 2017, 96, e8257. [Google Scholar] [CrossRef]
- Nishioka, Y.; Noda, T.; Kubo, S.; Myojin, T.; Imamura, T. Table 5.5-3. Utilization of the Receipt Information/Specific Medical Examination Information Database (NDB): Overview of Diabetes Prescriptions in Japan. Available online: http://www.nstac.go.jp/services/pdf/171117_5-3.pdf (accessed on 2 May 2021). (In Japanese).
- DeFronzo, R.; Fleming, G.A.; Chen, K.; Bicsak, T.A. Metformin-associated lactic acidosis: Current perspectives on causes and risk. Metabolism 2016, 65, 1432–1433. [Google Scholar] [CrossRef]
- Nagai, Y. Beneficial Effects of Biguanides on Cardiovascular Events. Prog. Med. 2021, 41, 795–798. (In Japanese) [Google Scholar]
- Lalau, J.D.; Kajbaf, F.; Protti, A.; Christensen, M.M.; De Broe, M.E.; Wiernsperger, N. Metformin-associated lactic acidosis (MALA): Moving towards a new paradigm. Diabetes Obes. Metab. 2017, 11, 1502–1512. [Google Scholar] [CrossRef]
- Chang, C.H.; Sakaguchi, M.; Dolin, P. Epidemiology of lactic acidosis in type 2 diabetes patients with metformin in Japan. Pharmacoepidemiol. Drug. Saf. 2016, 25, 1196–1203. [Google Scholar] [CrossRef]
- Aharaz, A.; Pottegård, A.; Henriksen, D.P.; Hallas, J.; Beck-Nielsen, H.; Lassen, A.T. Risk of lactic acidosis in type 2 diabetes patients using metformin: A case control study. PLoS ONE 2018, 13, e0196122. [Google Scholar] [CrossRef]
- Salpeter, S.; Greyber, E.; Pasternak, G.; Salpeter, E.E. Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2010, 4, CD002967. [Google Scholar] [CrossRef]
- Bicsak, T.A.; Walsh, B.; Fineman, M. Metformin-associated lactic acidosis: Moving towards a new paradigm? Diabetes Obes. Metab. 2017, 11, 1499–1501. [Google Scholar] [CrossRef]
- Yokoyama, S.; Tsuji, H.; Hiraoka, S.; Nishihara, M. Investigation of Risk Factors Affecting Lactate Levels in Japanese Patients Treated with Metformin. Biol. Pharm. Bull. 2016, 39, 2022–2027. [Google Scholar] [CrossRef]
- Connelly, P.J.; Lonergan, M.; Soto-Pedre, E.; Donnelly, L.; Zhou, K.; Pearson, E.R. Acute kidney injury, plasma lactate concentrations and lactic acidosis in metformin users: AGoDartS study. Diabetes Obes. Metab. 2017, 11, 1579–1586. [Google Scholar] [CrossRef]
- Scale, T.; Harvey, J.N. Diabetes, metformin and lactic acidosis. Clin. Endocrinol. 2011, 74, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Oh, H.J.; Kwon, S.H.; Jeon, J.S.; Noh, H.; Han, D.C.; Kim, H.; Ryu, D.R. Metformin use and cardiovascular outcomes in patients with diabetes and chronic kidney disease: A nationwide cohort study. Kidney Res. Clin. Pract. 2021, 40, 660–672. [Google Scholar] [CrossRef]
- Salvatore, T.; Pafundi, P.C.; Marfella, R.; Sardu, C.; Rinaldi, L.; Monaco, L.; Ricozzi, C.; Imbriani, S.; Nevola, R.; Adinolfi, L.E.; et al. Metformin lactic acidosis: Should we still be afraid? Diabetes Res. Clin. Pract. 2019, 157, 107879. [Google Scholar] [CrossRef]
- Smith, F.C.; Stocker, S.L.; Danta, M.; Carland, J.E.; Kumar, S.S.; Liu, Z.; Greenfield, J.R.; Braithwaite, H.E.; Cheng, T.S.; Graham, G.G.; et al. The safety and pharmacokinetics of metformin in patients with chronic liver disease. Aliment. Pharmacol. Ther. 2020, 51, 565–575. [Google Scholar] [CrossRef]
- Chen, C.C.; Ko, Y.; Chen, C.H.; Hung, Y.J.; Wei, T.E.; Chang, T.H.; Ke, S.S.; Kuo, K.N.; Chen, C. Relationship between metformin use and lactic acidosis in advanced chronic kidney disease: The REMIND-TMU study. Am. J. Med. Sci. 2022, 364, 575–582. [Google Scholar] [CrossRef] [PubMed]
- The Japan Diabetes Society. Committee on the Proper Use of Biguanides: Recommendation on the Proper Use of Biguanide Drugs. 2012. (revised 2020) (In Japanese). Available online: http://www.jds.or.jp/modules/important/index.php?content_id=20 (accessed on 2 May 2021).
- Kadowaki, T. Proper use of biguanide. Diabetes J. 2012, 40, 42–44. (In Japanese) [Google Scholar]
- Gregorio, F.; Ambrosi, F.; Filipponi, P.; Manfrini, S.; Testa, I. Is metformin safe enough for ageing type 2 diabetic patients? Diabetes Metab. 1996, 22, 43–50. [Google Scholar] [PubMed]
- Lin, Y.C.; Lin, L.Y.; Wang, H.F.; Lin, H.D. Fasting plasma lactate concentrations in ambulatory elderly patients with type 2 diabetes receiving metformin therapy: A retrospective cross-sectional study. J. Chin. Med. Assoc. 2010, 73, 617–622. [Google Scholar] [CrossRef]
- Kubo, S.; Noda, T.; Myojin, T.; Nishioka, Y.; Higashino, T.; Matsui, H.; Kato, G.; Imamura, T. National Database of Health Insurance Claims and Specific Health Checkups of Japan (NDB): Outline and Patient-Matching Technique. bioRxiv 2018. [Google Scholar] [CrossRef]
- Noda, T.; Kubo, S.; Myojin, T.; Higashino, T.; Matsui, H.; Kato, G.; Imamura, T. Improvement and verification of the patient matching method in National Database of Health Insurance Claims and Specific Health Checkups of Japan. J. Health Welf. Stat. 2017, 64, 7–13. (In Japanese) [Google Scholar]
- Nishioka, Y.; Okada, S.; Noda, T.; Myojin, T.; Kubo, S.; Ohtera, S.; Kato, G.; Kuroda, T.; Ishii, H.; Imamura, T. Absolute risk of acute coronary syndrome after severe hypoglycemia: A population-based 2-year cohort study using the National Database in Japan. J. Diabetes Investig. 2020, 11, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Myojin, T.; Noda, T.; Kubo, S.; Nishioka, Y.; Higashino, T.; Imamura, T. Development of a New Method to Trace Patient Data Using the National Database in Japan. Adv. Biomed. Eng. 2022, 11, 203–217. [Google Scholar] [CrossRef]
- Aoki, N. How to calculate and use the person-year method. J. Jpn. Assoc. Cereb. Cardiovasc. Dis. Control. 1991, 26, 64–66. (In Japanese) [Google Scholar]
- Lazarus, B.; Wu, A.; Shin, J.I.; Sang, Y.; Alexander, G.C.; Secora, A.; Inker, L.A.; Coresh, J.; Chang, A.R.; Grams, M.E. Association of Metformin Use with Risk of Lactic Acidosis Across the Range of Kidney Function: A Community-Based Cohort Study. JAMA Intern. Med. 2018, 178, 903–910. [Google Scholar] [CrossRef]
- Haloob, I.; de Zoysa, J.R. Metformin associated lactic acidosis in Auckland City Hospital 2005 to 2009. World J. Nephrol. 2016, 5, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Mariano, F.; Pozzato, M.; Inguaggiato, P.; Guarena, C.; Turello, E.; Manes, M.; David, P.; Berutti, S.; Consiglio, V.; Amore, A.; et al. Metformin-Associated Lactic Acidosis Undergoing Renal Replacement Therapy in Intensive Care Units: A Five-Million Population-Based Study in the North-West of Italy. Blood Purif. 2017, 44, 198–205. [Google Scholar] [CrossRef]
- Chu, P.Y.; Hackstadt, A.J.; Chipman, J.; Griffin, M.R.; Hung, A.M.; Greevy, R.A., Jr.; Grijalva, C.G.; Elasy, T.; Roumie, C.L. Hospitalization for Lactic Acidosis Among Patients with Reduced Kidney Function Treated with Metformin or Sulfonylureas. Diabetes Care 2020, 43, 1462–1470. [Google Scholar] [CrossRef]
- Zhang, X.; Harmsen, W.S.; Mettler, T.A.; Kim, W.R.; Roberts, R.O.; Therneau, T.M.; Roberts, L.R.; Chaiteerakij, R. Continuation of metformin use after a diagnosis of cirrhosis significantly improves survival of patients with diabetes. Hepatology 2014, 60, 2008–2016. [Google Scholar] [CrossRef]
- Cheng, X.; Liu, Y.M.; Li, H.; Zhang, X.; Lei, F.; Qin, J.J.; Chen, Z.; Deng, K.Q.; Lin, L.; Chen, M.M. Metformin Is Associated with Higher Incidence of Acidosis, but Not Mortality, in Individuals with COVID-19 and Pre-existing Type 2 Diabetes. Cell Metab. 2020, 32, 537–547.e3. [Google Scholar] [CrossRef] [PubMed]
- Salber, G.J.; Wang, Y.B.; Lynch, J.T.; Pasquale, K.M.; Rajan, T.V.; Stevens, R.G.; Grady, J.J.; Kenny, A.M. Metformin use in Practice: Compliance with guidelines for patients with diabetes and preserved renal function. Clin. Diabetes 2017, 35, 154–161. [Google Scholar] [CrossRef]
- Di Mauro, S.; Filippello, A.; Scamporrino, A.; Purrello, F.; Piro, S.; Malaguarnera, R. Metformin: When Should We Fear Lactic Acidosis? Int. J. Mol. Sci. 2022, 23, 8320. [Google Scholar] [CrossRef]
- Imai, E. Chronic kidney disease in the elderly. Jpn. J. Geriatr. 2014, 51, 385–400. (In Japanese) [Google Scholar]
- Van Berlo-van de Laar, I.R.F.; Gedik, A.; van’t Riet, E.; de Meijer, A.; Taxis, K.; Jansman, F.G.A. Identifying patients with metformin associated lactic acidosis in the emergency department. Int. J. Clin. Pharm. 2020, 42, 1286–1292. [Google Scholar] [CrossRef]
- Diabetes Prevention Program Research Group, Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care 2012, 35, 731–737. [CrossRef] [PubMed]
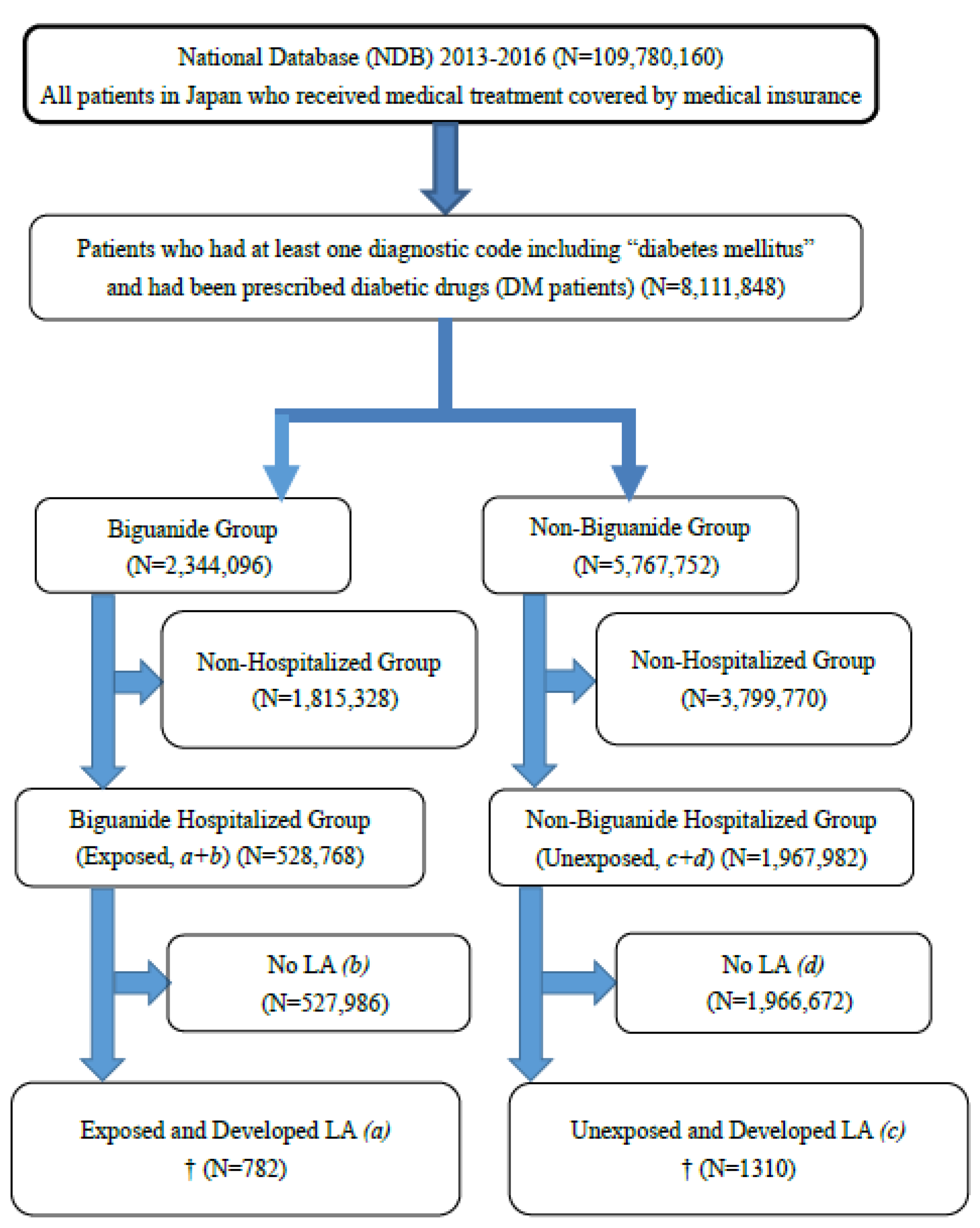
| Hospitalized DM Patients | Lactic Acidosis Diagnosis (Disease) | No Lactic Acidosis Diagnosis (No Disease) | Total |
|---|---|---|---|
| Biguanide prescription (Exposed) | 782 (a) | 527,986 (b) | 528,768 (a + b) |
| No biguanide prescription (Unexposed) | 1310 (c) | 1,966,672 (d) | 1,967,982 (c + d) |
| Total | 2092 (a + c) | 2,494,658 (b + d) | 2,496,750 (a + b + c + d) |
| DM Patients | Number of Person-Days at Risk of LA | Patients | Incidence Rate of LA † | Rate Ratio of Developing LA ‡ (95% CI) |
|---|---|---|---|---|
| Biguanide prescription (Exposed) | 2,128,760,399 person-days | Diagnosed with LA (a) (N = 782) | 13.41 /100,000 person-years | 1.44 (1.32–1.58) |
| Not diagnosed with LA (b) (N = 527,986) | ||||
| No biguanide prescription (Unexposed) | 5,118,605,720 person-days | Diagnosed with LA (c) (N = 1310) | 9.34 /100,000 person-years | |
| Not diagnosed with LA (d) (N = 1,966,672) |
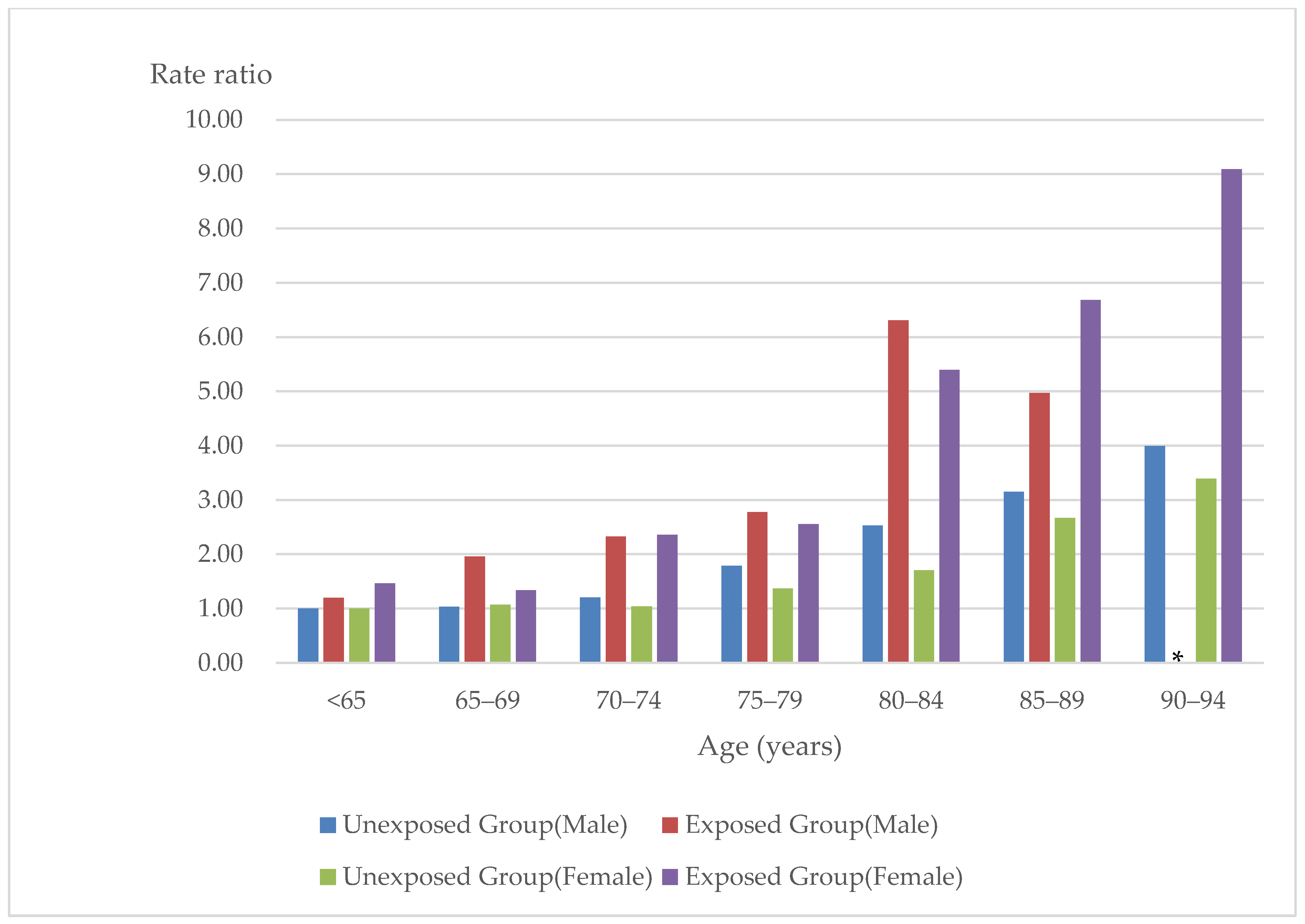
| Biguanide-LA-Hospitalized Group | Non-Biguanide-LA-Hospitalized Group | |||||
|---|---|---|---|---|---|---|
| (Exposed Group) | (Unexposed Group) | |||||
| Age (years) | Incidence Rate /100,000 Person-Years | Rate Ratio (95% CI) | Number | Incidence Rate /100,000 Person-Years | Rate Ratio (95% CI) | Number |
| Males | ||||||
| <65 | 7.61 | 1.20 (0.96–1.50) | 140 | 6.36 | Reference | 182 |
| 65–69 | 12.47 | 1.96 (1.50–2.56) | 78 | 6.55 | 1.03 (0.80–1.32) | 91 |
| 70–74 | 14.79 | 2.32 (1.76–3.06) | 70 | 7.66 | 1.20 (0.94–1.53) | 103 |
| 75–79 | 17.66 | 2.78 (2.06–3.74) | 57 | 11.39 | 1.79 (1.43–2.23) | 137 |
| 80–84 | 40.12 | 6.31 (4.75–8.39) | 64 | 16.08 | 2.53 (2.03–3.16) | 137 |
| 85–89 | 31.62 | 4.97 (2.98–8.29) | 16 | 20.04 | 3.15 (2.41–4.11) | 77 |
| 90–94 | † | † | ‡ | 25.41 | 3.99 (2.59–6.16) | 23 |
| ≥95 | † | † | ‡ | † | † | ‡ |
| Females | ||||||
| <65 | 9.47 | 1.46 (1.09–1.96) | 89 | 6.48 | Reference | 90 |
| 65–69 | 8.64 | 1.33 (0.91–1.94) | 38 | 6.93 | 1.07 (0.77–1.49) | 58 |
| 70–74 | 15.26 | 2.36 (1.70–3.27) | 60 | 6.73 | 1.04 (0.75–1.43) | 64 |
| 75–79 | 16.54 | 2.55 (1.80–3.61) | 49 | 8.87 | 1.37 (1.02–1.84) | 87 |
| 80–84 | 34.96 | 5.40 (3.91–7.46) | 62 | 11.02 | 1.70 (1.28–2.27) | 96 |
| 85–89 | 43.29 | 6.68 (4.50–9.91) | 34 | 17.29 | 2.67 (2.01–3.55) | 98 |
| 90–94 | 58.9 | 9.09 (5.08–16.26) | 13 | 21.99 | 3.39 (2.40–4.78) | 51 |
| ≥95 | † | † | ‡ | † | † | ‡ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohri, T.; Okamoto, S.; Nishioka, Y.; Myojin, T.; Kubo, S.; Higashino, T.; Okada, S.; Akai, Y.; Noda, T.; Ishii, H.; et al. Risk of Lactic Acidosis in Hospitalized Diabetic Patients Prescribed Biguanides in Japan: A Retrospective Total-Population Cohort Study. Int. J. Environ. Res. Public Health 2023, 20, 5300. https://doi.org/10.3390/ijerph20075300
Mohri T, Okamoto S, Nishioka Y, Myojin T, Kubo S, Higashino T, Okada S, Akai Y, Noda T, Ishii H, et al. Risk of Lactic Acidosis in Hospitalized Diabetic Patients Prescribed Biguanides in Japan: A Retrospective Total-Population Cohort Study. International Journal of Environmental Research and Public Health. 2023; 20(7):5300. https://doi.org/10.3390/ijerph20075300
Chicago/Turabian StyleMohri, Takako, Sawako Okamoto, Yuichi Nishioka, Tomoya Myojin, Shinichiro Kubo, Tsuneyuki Higashino, Sadanori Okada, Yasuhiro Akai, Tatsuya Noda, Hitoshi Ishii, and et al. 2023. "Risk of Lactic Acidosis in Hospitalized Diabetic Patients Prescribed Biguanides in Japan: A Retrospective Total-Population Cohort Study" International Journal of Environmental Research and Public Health 20, no. 7: 5300. https://doi.org/10.3390/ijerph20075300
APA StyleMohri, T., Okamoto, S., Nishioka, Y., Myojin, T., Kubo, S., Higashino, T., Okada, S., Akai, Y., Noda, T., Ishii, H., & Imamura, T. (2023). Risk of Lactic Acidosis in Hospitalized Diabetic Patients Prescribed Biguanides in Japan: A Retrospective Total-Population Cohort Study. International Journal of Environmental Research and Public Health, 20(7), 5300. https://doi.org/10.3390/ijerph20075300






